Significance of Hydrogen as Economic and Environmentally Friendly Fuel
Abstract
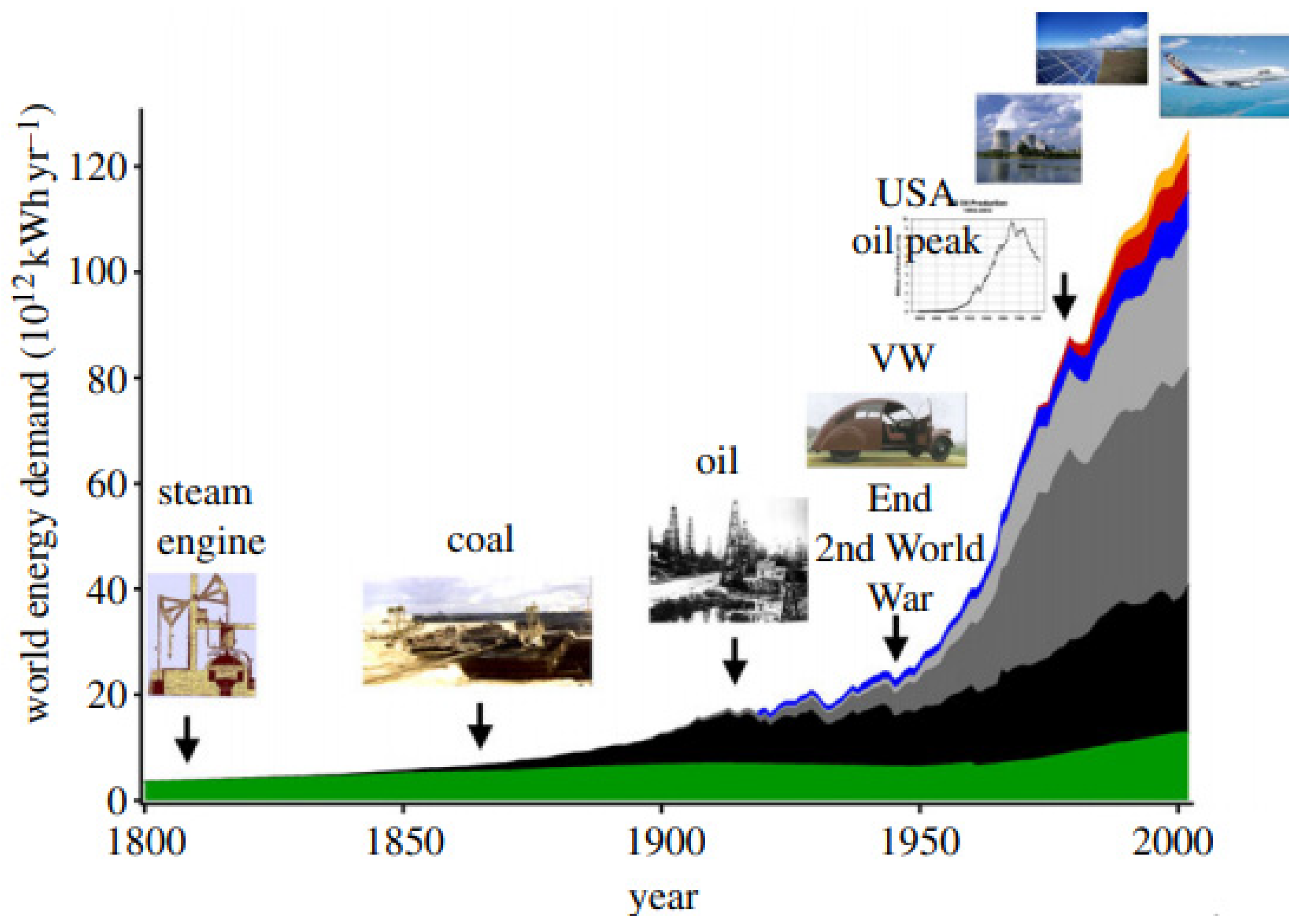
:1. Present Energy Infrastructure and Its Problems: Economic and Environmental Point of View
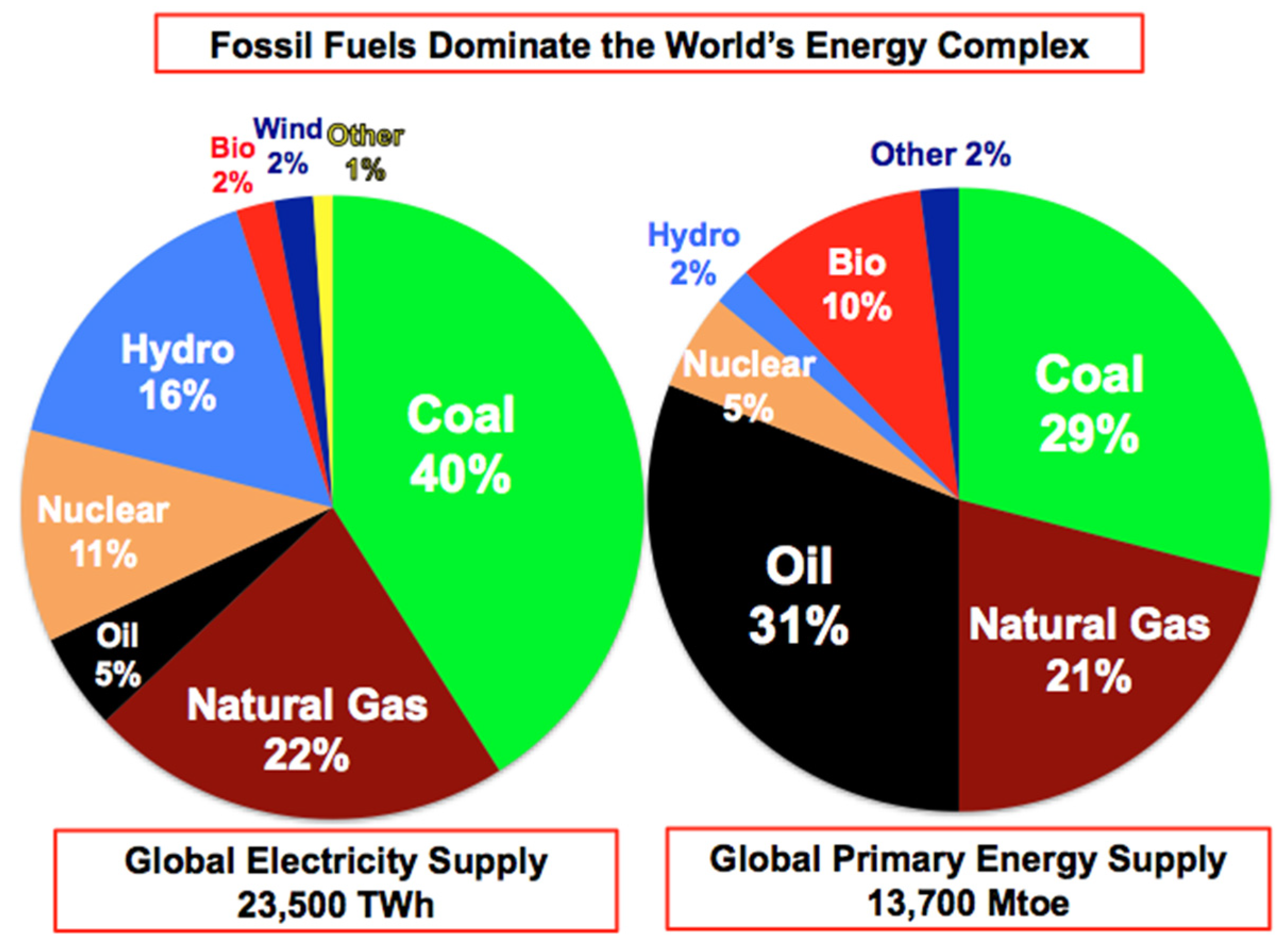
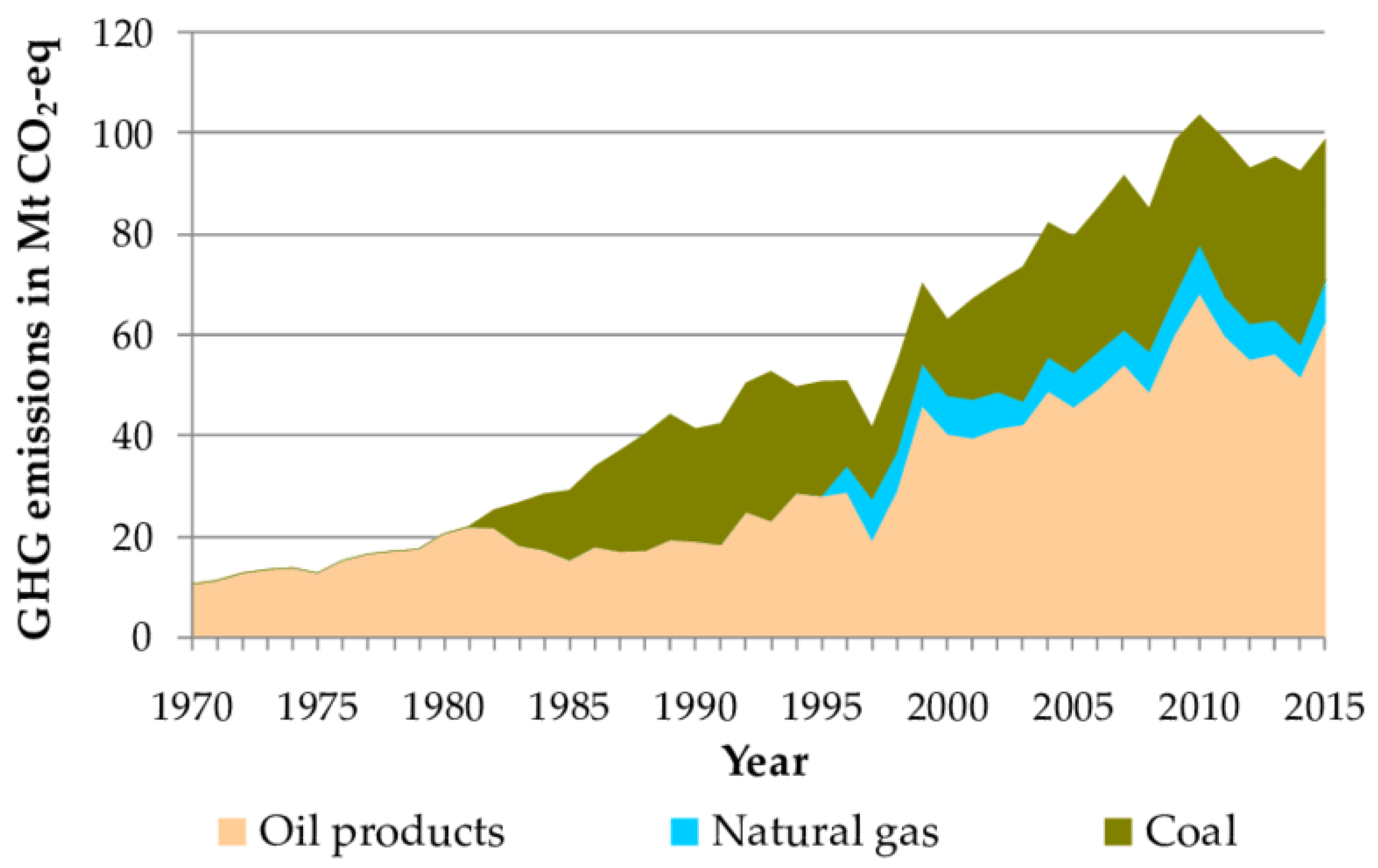
1.1. Environmental Impact
1.2. Economic Feasibility
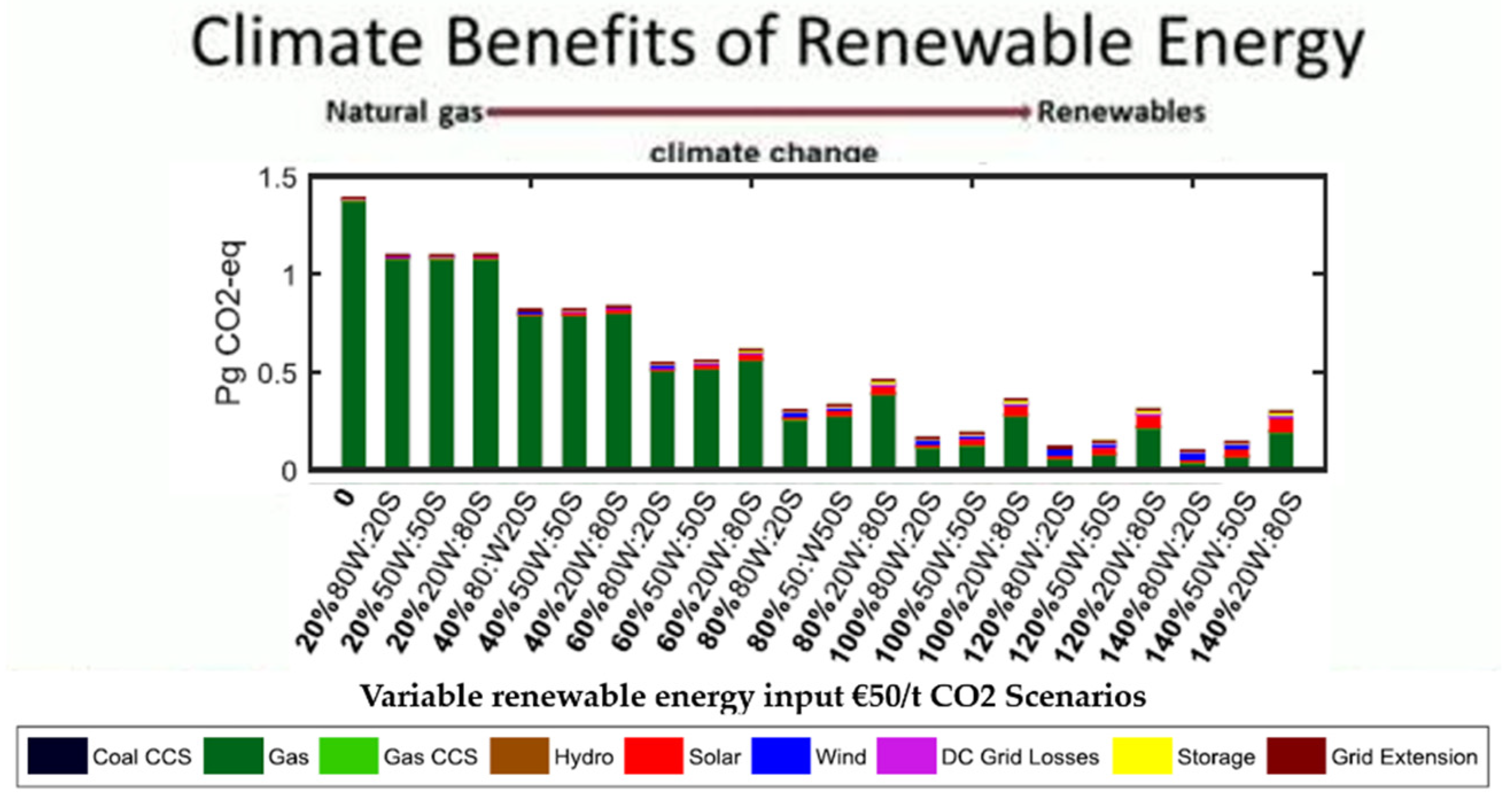
2. Renewable Energy as a Solution of Problems Associated with Conventional Energy Sources
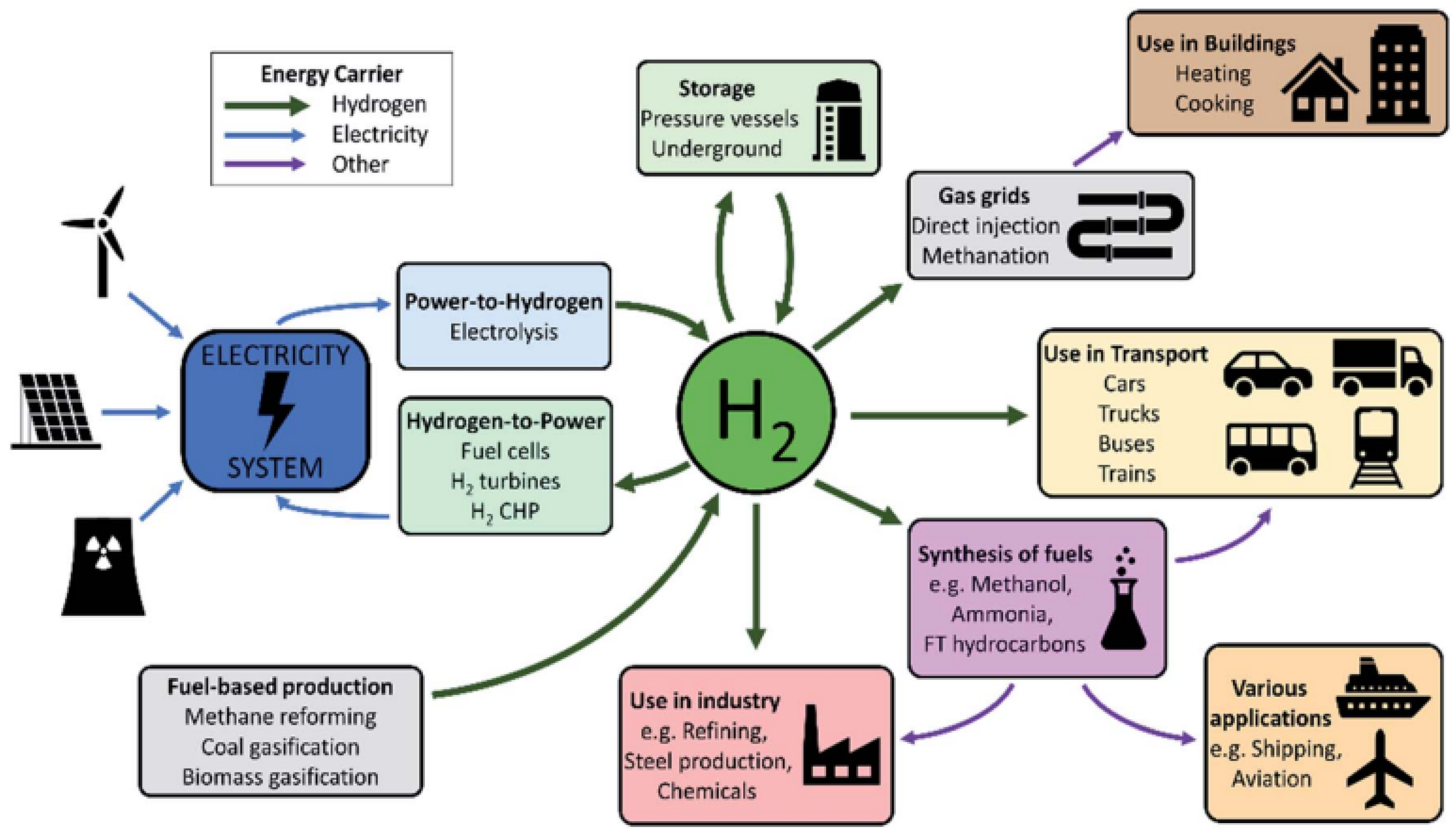
3. Hydrogen as Future Energy Carrier
3.1. Hydrogen Production
3.1.1. Hydrogen Production Methods Based on Hydrocarbons
3.1.2. Hydrogen Production Methods Based on Non-Hydrocarbons
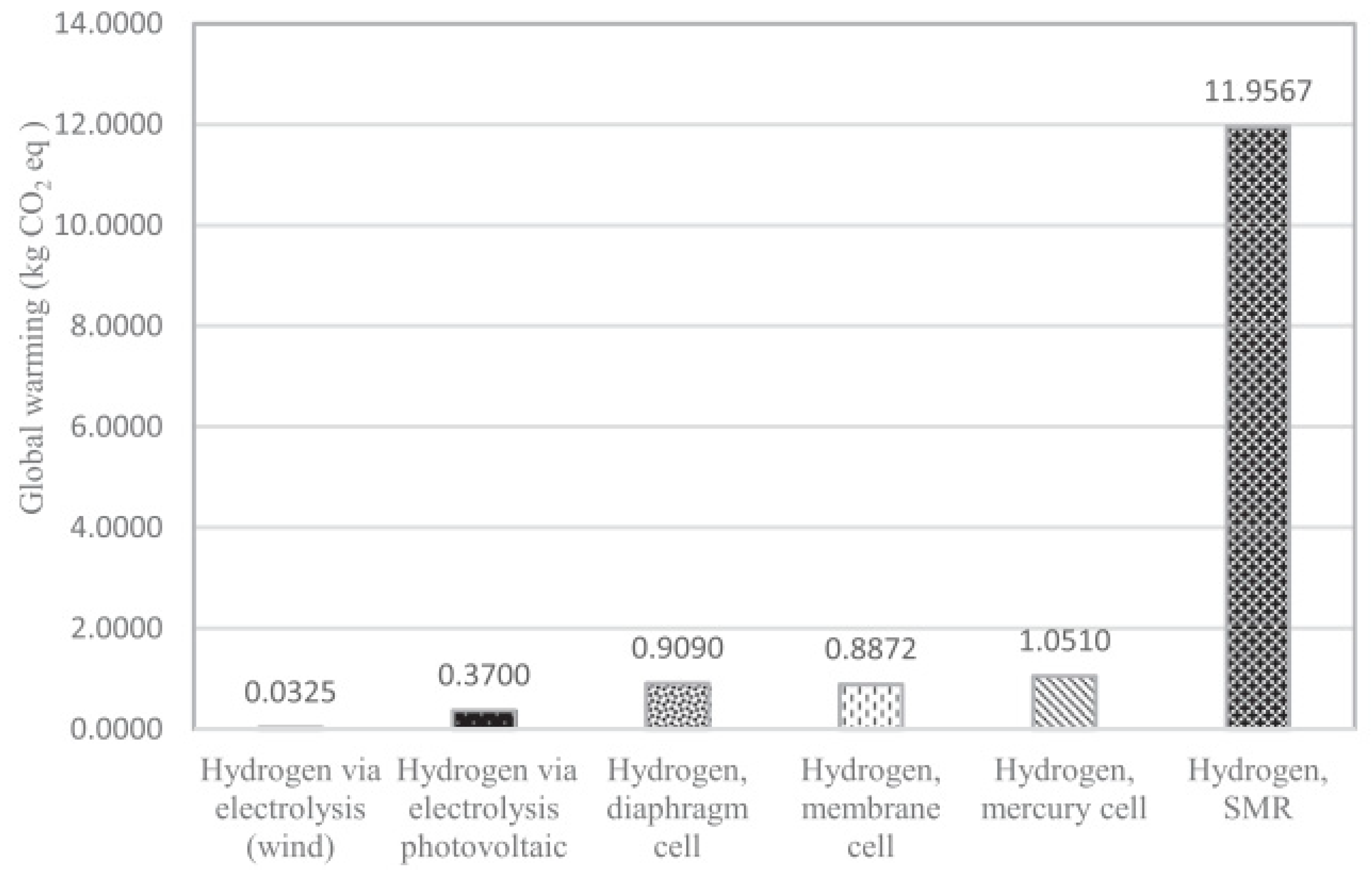
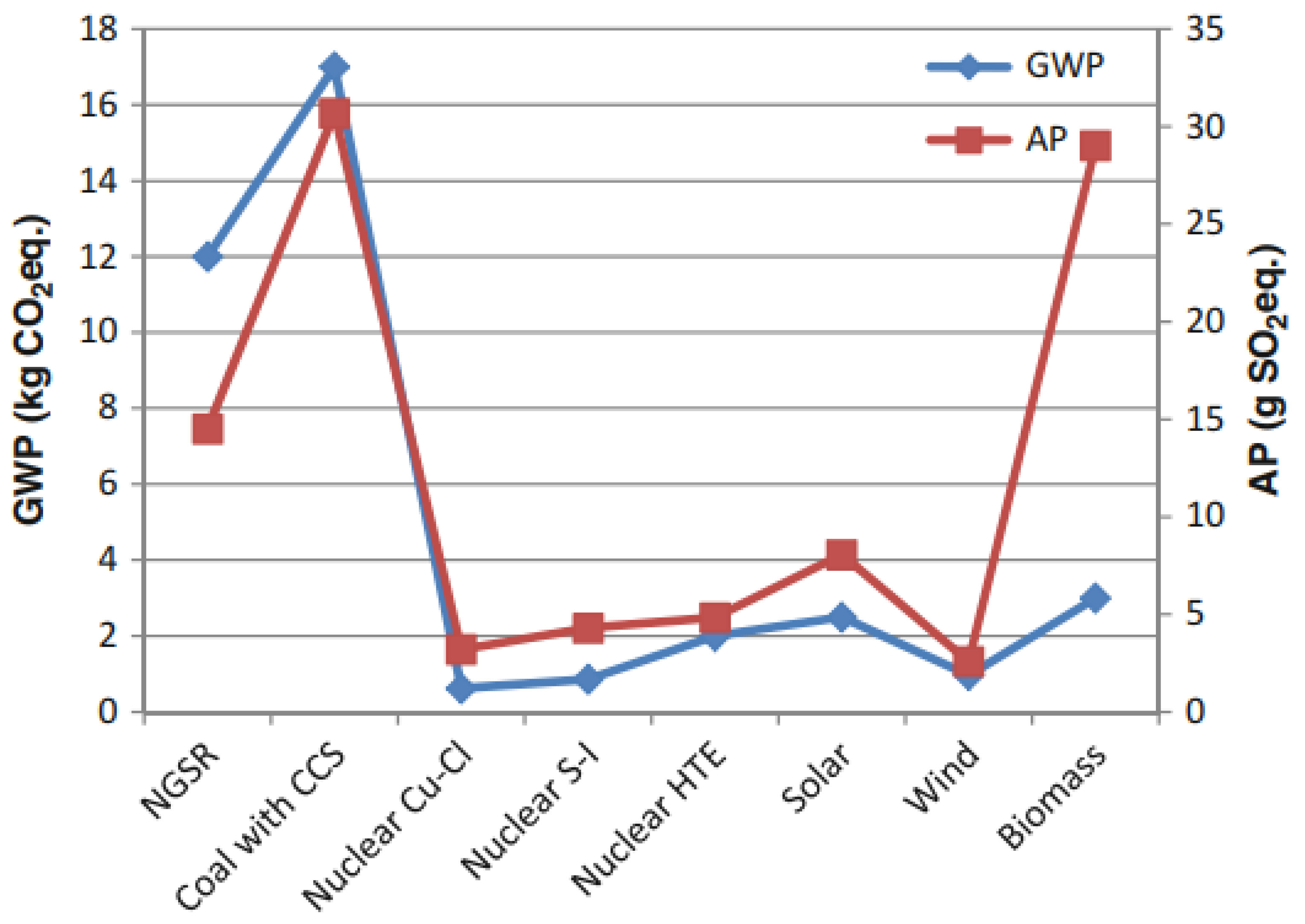
3.1.3. Environmental Aspects of Production Methods
3.1.4. Economic Aspects of Production Methods
3.2. Hydrogen Storage
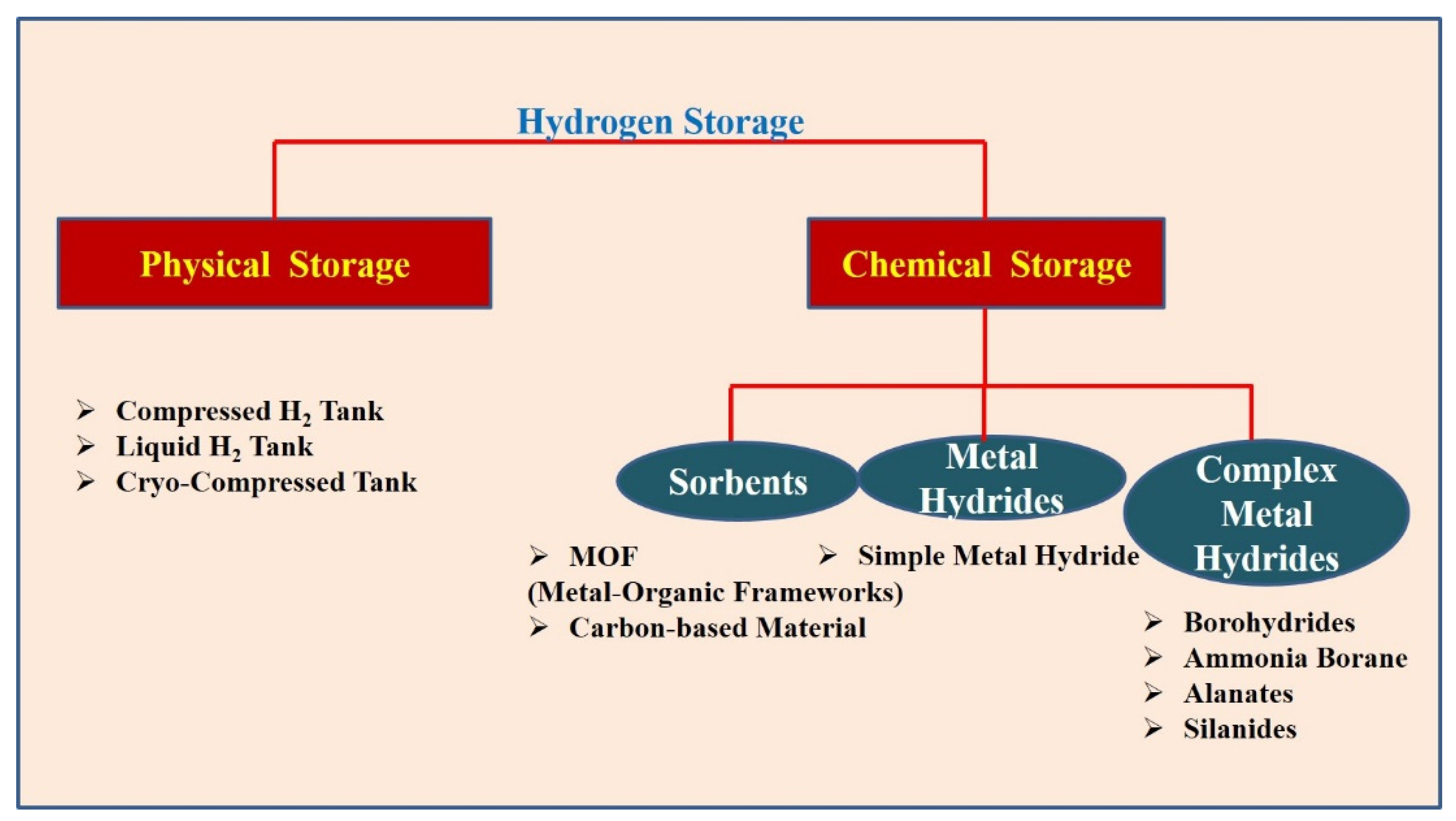
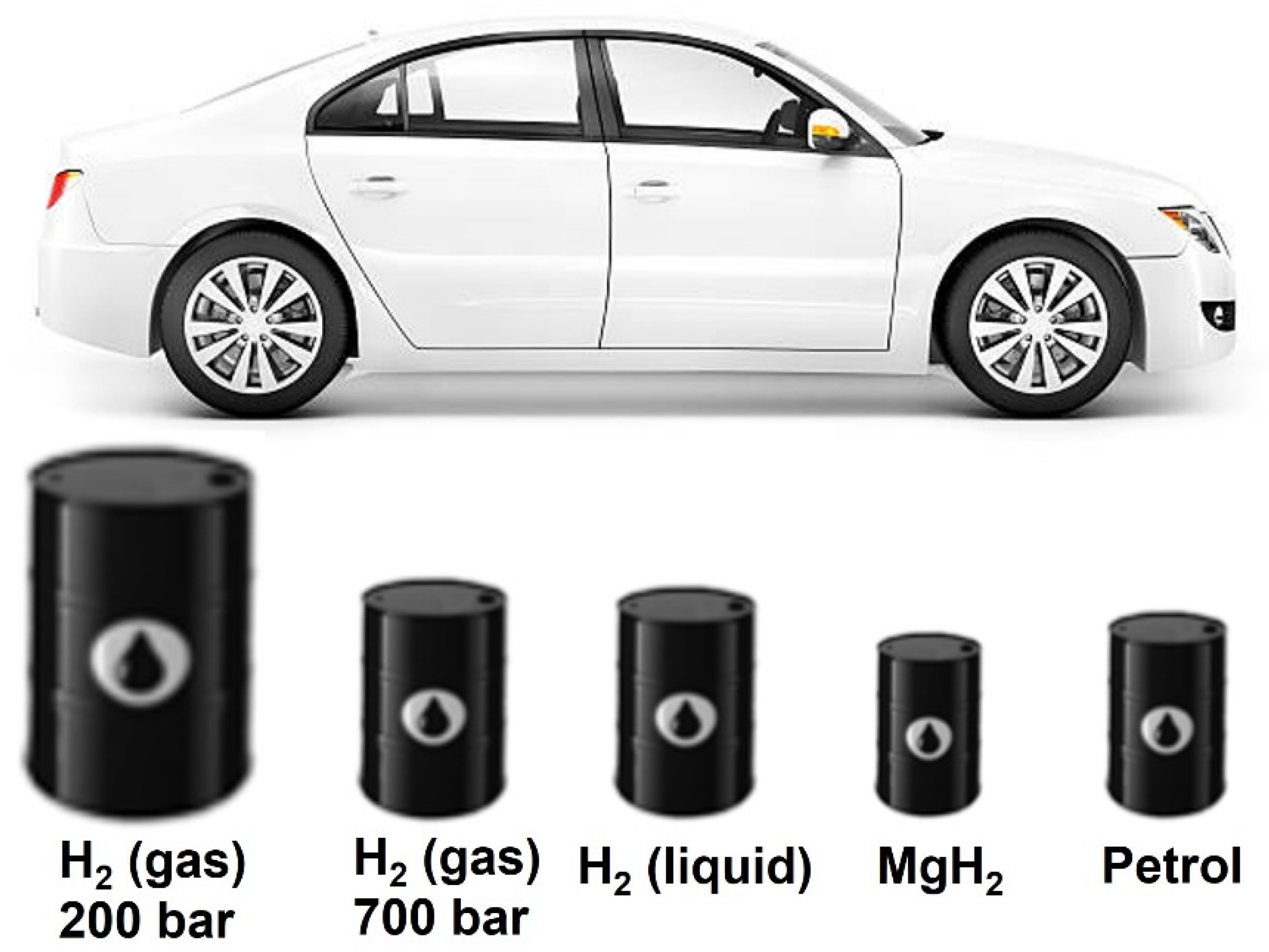
3.2.1. Physical Storage
3.2.2. Chemical Storage
3.2.3. Environmental Aspects
3.2.4. Economic Aspects
3.3. Utilization of Hydrogen
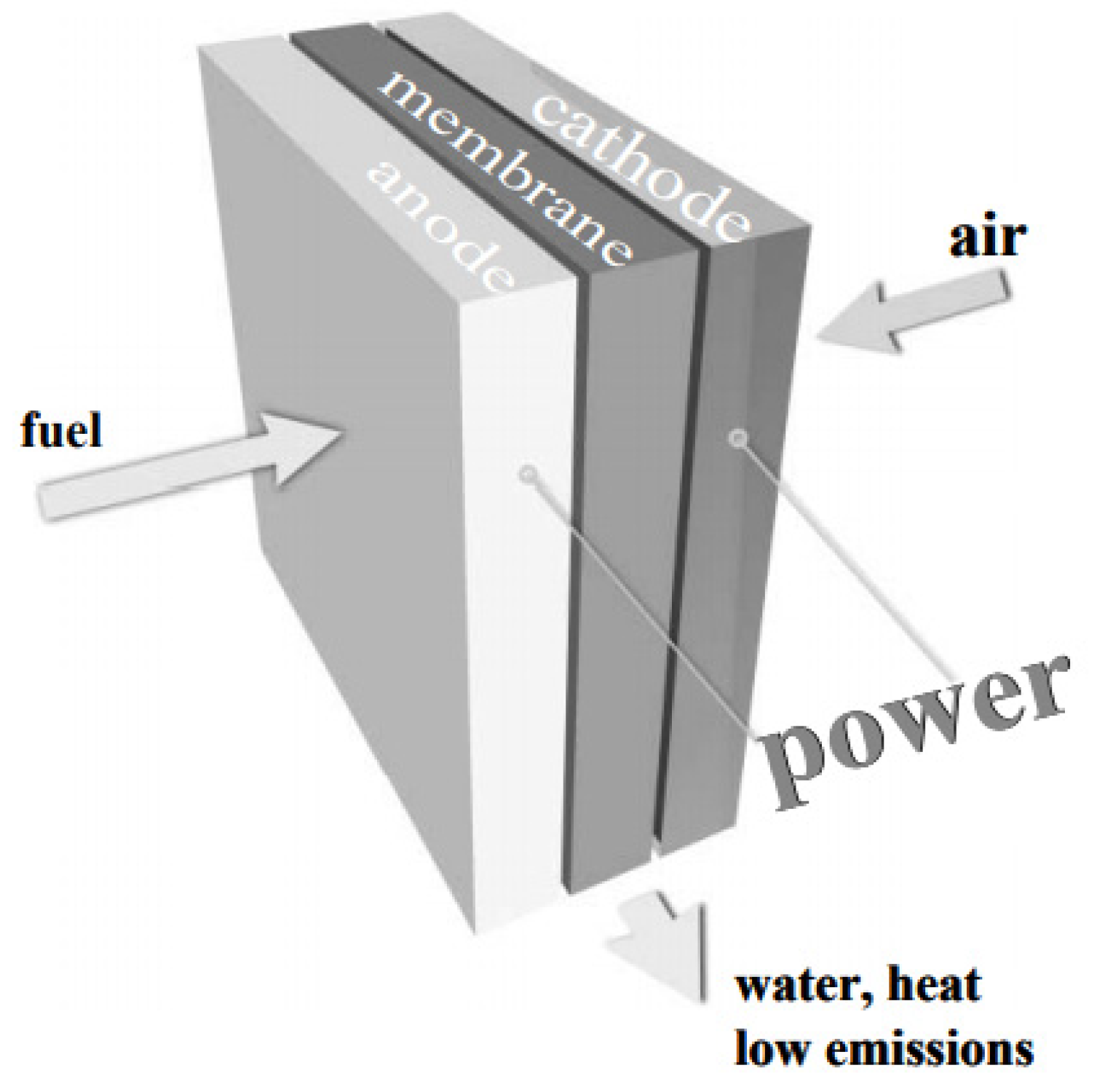
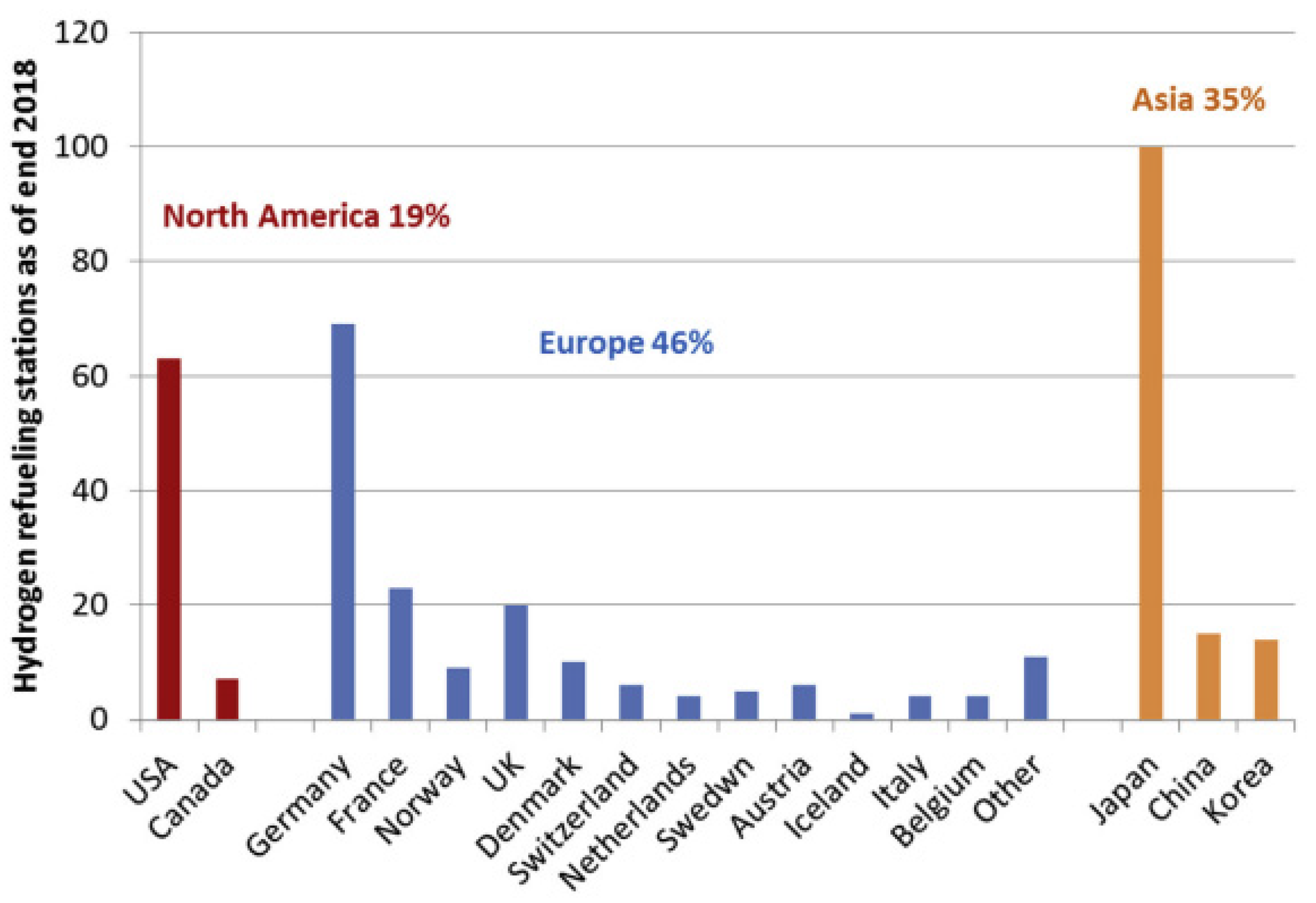
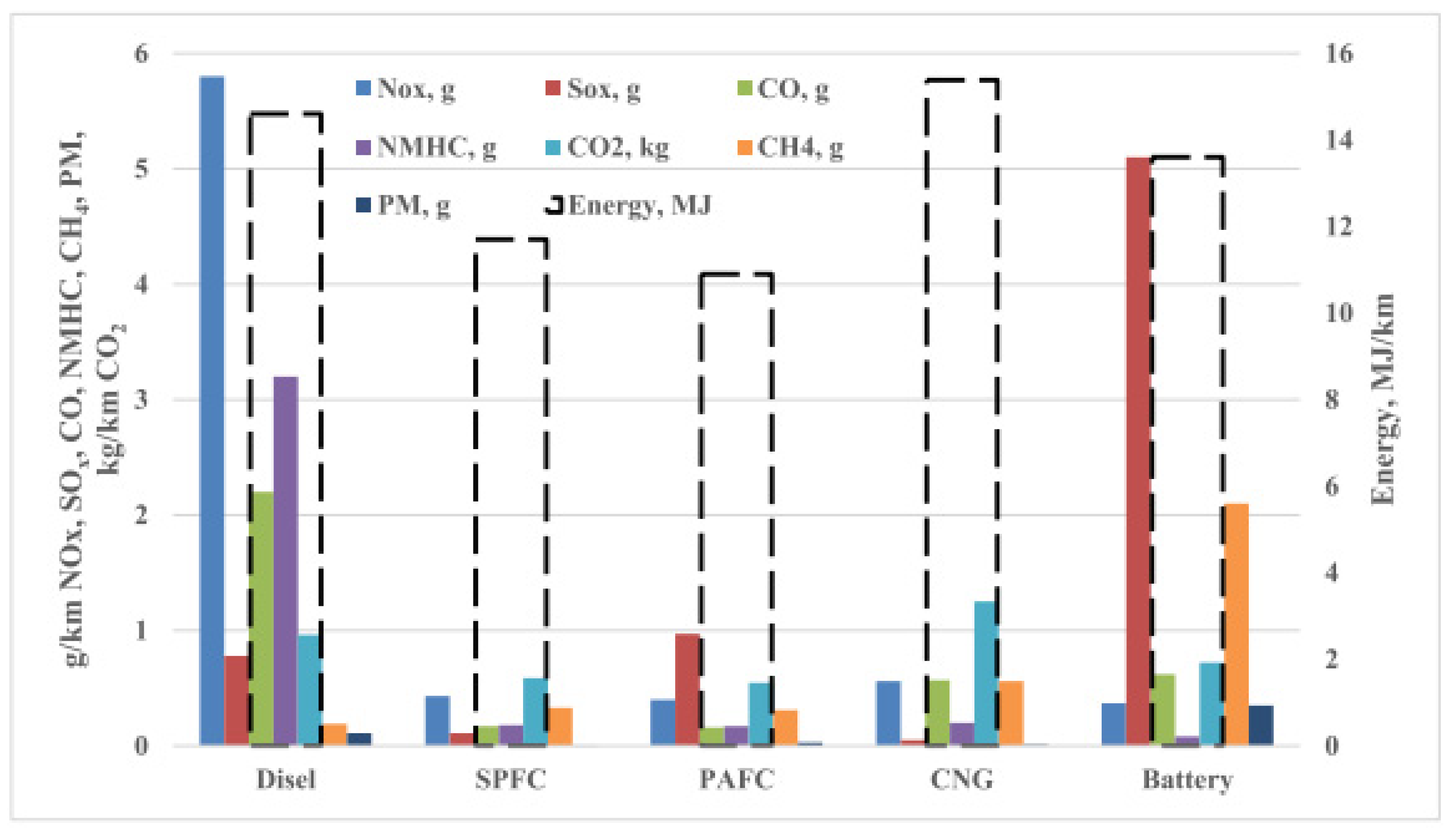
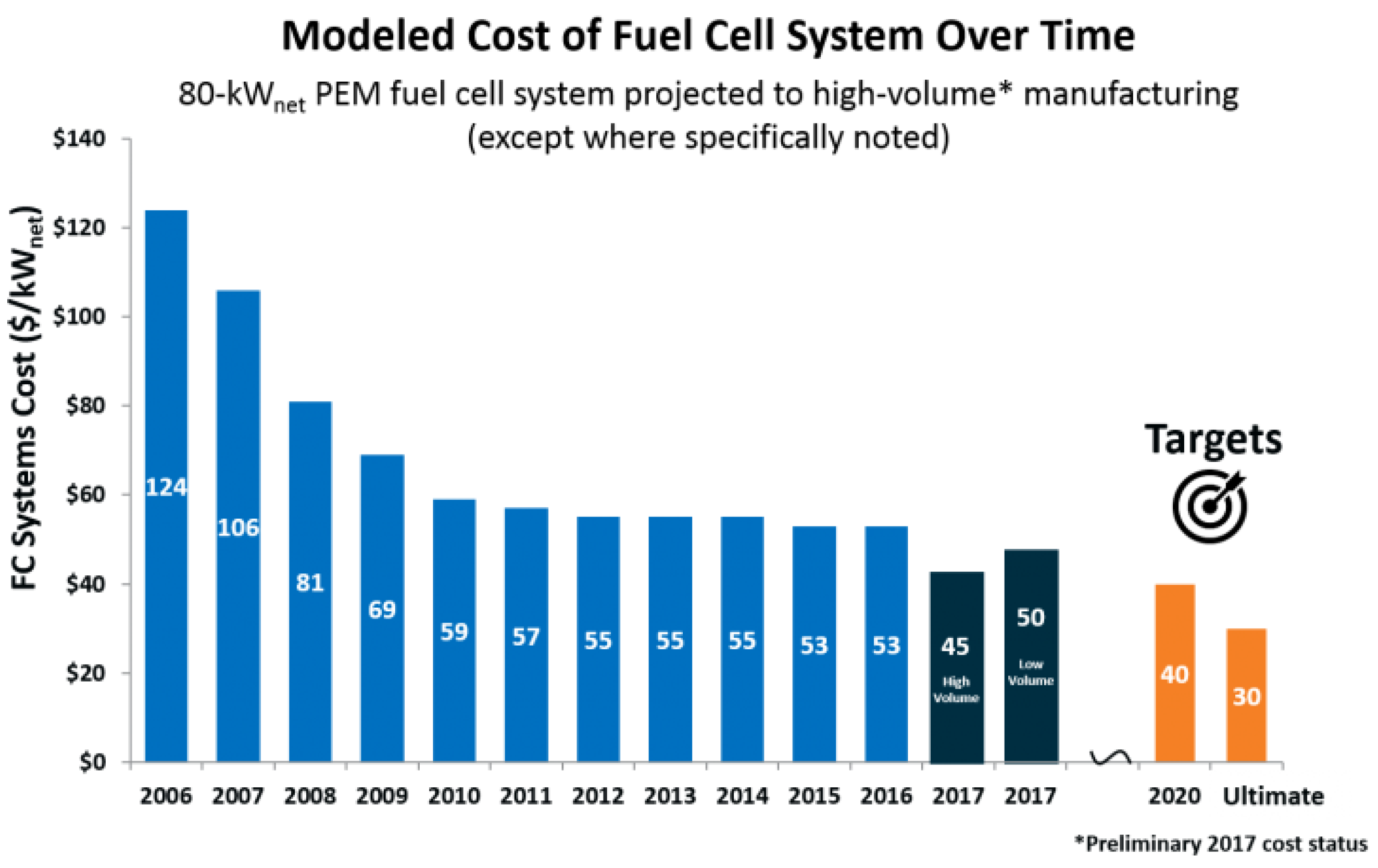
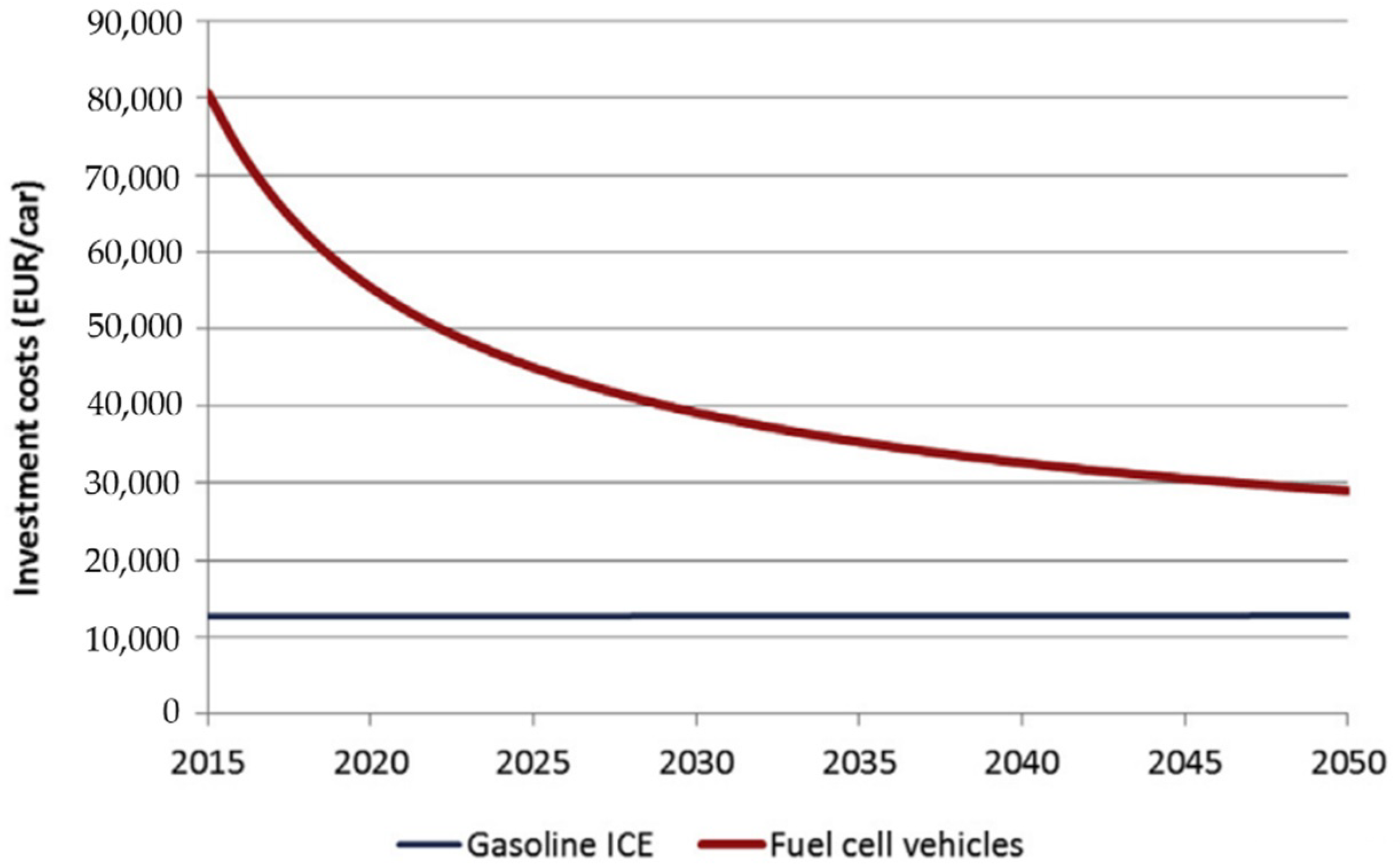
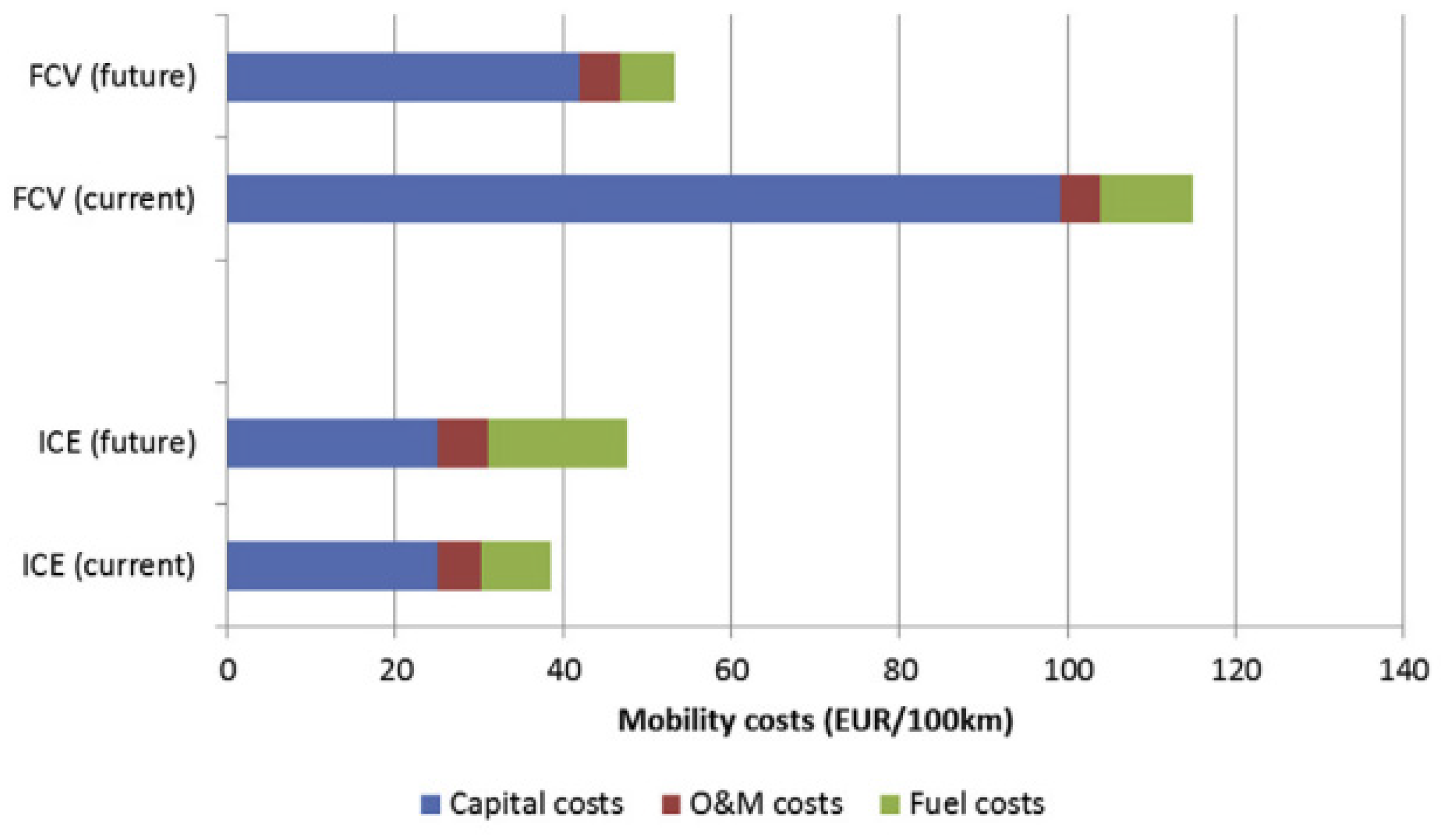
3.3.1. Hydrogen Fuel Cell in Automotive Market, Transport Industries, and Their Infrastructural Issues
3.3.2. Environmental Aspects
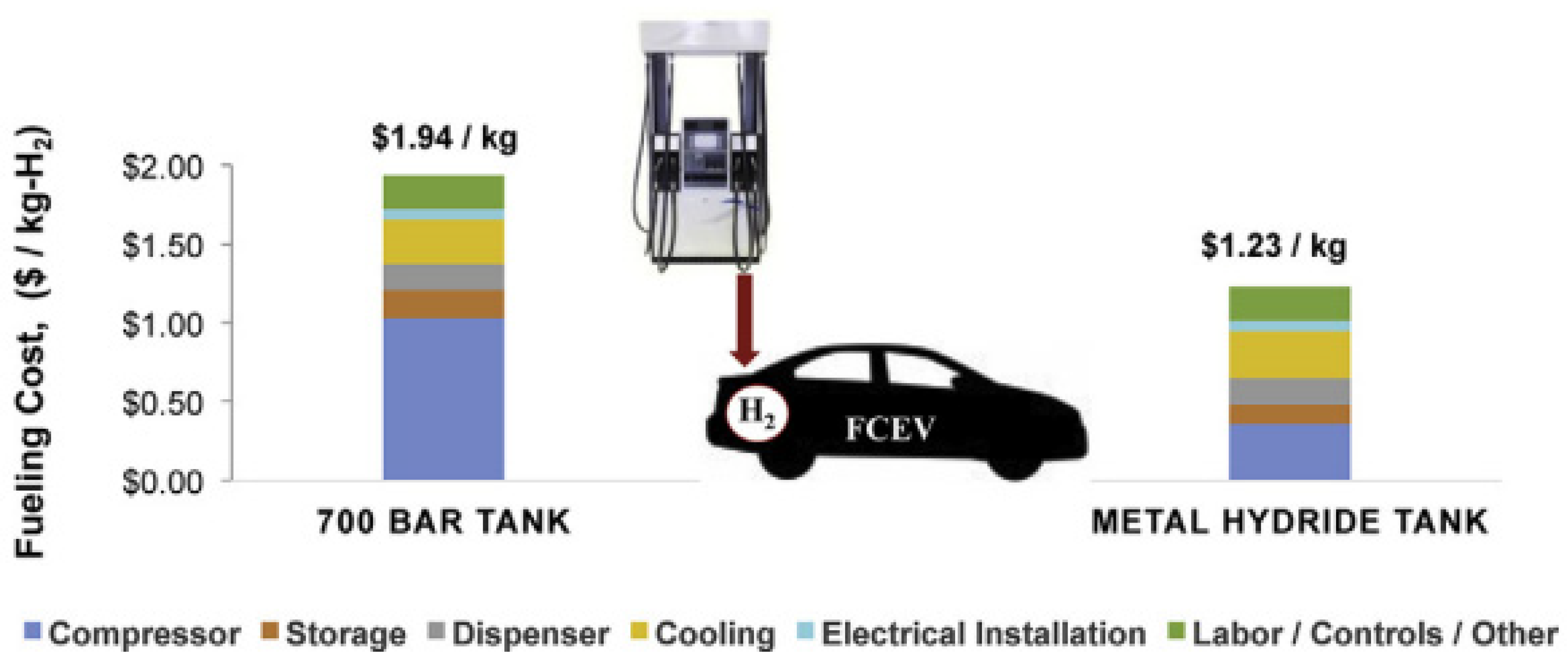
3.3.3. Economic Aspects
3.4. Commercial Applications of Hydrogen
3.5. Hydrogen Safety
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References and Notes
- Apostolou, D.; Xydis, G. A literature review on hydrogen refueling stations and infrastructure, Current status and future prospectus. Renew. Sustain. Energy Rev. 2019, 113, 109292. [Google Scholar] [CrossRef]
- Dincer, I.; Acar, C. Smart energy solutions with hydrogen options. Int. J. Hydrogen Energy 2018, 43, 8579–8599. [Google Scholar] [CrossRef]
- Khan, N.; Kalair, E.; Abas, N.; Kalair, A. Energy transition from molecules to atoms and photons. Eng. Sci. Technol. Int. J. 2019, 22, 185–214. [Google Scholar] [CrossRef]
- To, W.-M.; Lee, P.K.C. Energy Consumption and Economic Development in Hong Kong, China. Energies 2017, 10, 1883. [Google Scholar] [CrossRef] [Green Version]
- Mohsin, M.; Kamran, H.W.; Nawaz, M.A.; Hussain, M.S.; Dahri, A.S. Assessing the impact of transition from non-renewable to renewable energy consumption on economic growth-environmental nexus from developing Asian economies. J. Environ. Manag. 2021, 284, 111999. [Google Scholar] [CrossRef] [PubMed]
- BPSTATS. BP Statistical Review of World Energy Statistical Review of World, 68th ed.; Heriot-Watt University: Edinburgh, UK, 2019. [Google Scholar]
- Enerdata. Enerdata: Global Energy Statistical Yearbook 2018. Global Energy Trends. 2018. Available online: https://energydata.info/dataset/key-world-energy-statistics-enerdata/resource/dcda6530-8d2c-436e-9d1d-1e2809ad303e (accessed on 2 November 2021).
- Norton, R. An Overview of a Sustainable City Strategy. Report Prepared for the Global Energy Assessment Planning for Cities and Municipalities. Montreal, QC, Canada, 1991.
- Dincer, I.; Rosen, M.A. A worldwide perspective on energy, environment and sustainable development. Int. J. Energy Res. 1998, 22, 1305–1321. [Google Scholar] [CrossRef]
- Rosen, M. The role of energy efficiency in sustainable development. In Proceedings of the 1995 Interdisciplinary Conference: Knowledge Tools for a Sustainable Civilization, Fourth Canadian Conference on Foundations and Applications of General Science Theory, Toronto, ON, Canada, 8–10 June 1995. [Google Scholar] [CrossRef]
- Solomon, B.D.; Banerjee, A. A global survey of hydrogen energy research, development and policy. Energy Policy 2006, 34, 781–792. [Google Scholar] [CrossRef]
- Zuttel, A.; Remhof, A.; Borgschulte, A. Hydrogen: The future energy carrie. Philos. Trans. R. Soc. A 2010, 368, 3329–3342. [Google Scholar] [CrossRef] [PubMed]
- Rosen, M.A.; Dincer, I.; Kanoglu, M. Role of exergy in increasing efficiency and sustainability and reducing environmental impact. Energy Policy 2008, 36, 128–137. [Google Scholar] [CrossRef]
- IEA. From Poverty to Prosperity. In World Energy Outlook Special Report; OECD Publishing: Paris, France, 2017; Available online: https://www.oecd.org/publications/energy-access-outlook-2017-9789264285569-en.htm (accessed on 19 February 2020).
- Berrill, P.; Arvesen, A.; Scholz, Y.; Gils, H.C.; Hertwich, E. Environmental impacts of high penetration renewable energy scenarios for Europe. Environ. Res. Lett. 2016, 11, 014012. [Google Scholar] [CrossRef]
- Dunn, S. Hydrogen futures: Toward a sustainable energy system. Int. J. Hydrogen Energy 2002, 27, 235–264. [Google Scholar] [CrossRef]
- Dincer, I.; Rosen, M.A. Sustainability aspects of hydrogen and fuel cell systems. Energy Sustain. Dev. 2011, 15, 137–146. [Google Scholar] [CrossRef]
- Boyle, G. Renewable energy: Power for a sustainable future. Renew. Energy 2004, 19, 456. [Google Scholar]
- Schlapbach, L.; Züttel, A. Hydrogen-storage materials for mobile applications. Nat. Cell Biol. 2001, 414, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Carbon-Based Nanoporous Materials for Hydrogen Storage. Available online: https://pure.unileoben.ac.at/portal/files/2214367/AC14528127n01-2017 (accessed on 30 November 2017).
- Nowotny, J.; Veziroglu, T.N. Impact of hydrogen on the environment. Int. J. Hydrogen Energy 2011, 36, 13218–13224. [Google Scholar] [CrossRef]
- Hydrogen Council. Path to Hydrogen Competitiveness a Cost Perspective; Hydrogen Council: Brussels, Belgium, 2020; pp. 1–88. [Google Scholar]
- Hydrogen Council. Hydrogen Scaling up, a Sustainable Pathway for the Global Energy Transition; Hydrogen Council: Brussels, Belgium, 2017; pp. 1–80. [Google Scholar]
- Rosen, M.A.; Koohi-Fayegh, S. The prospects for hydrogen as an energy carrier: An overview of hydrogen energy and hydrogen energy systems. Energy Ecol. Environ. 2016, 1, 10–29. [Google Scholar] [CrossRef] [Green Version]
- Atilhan, S.; Park, S.; El-Halwagi, M.M.; Atilhan, M.; Moore, M.; Nielsen, R.B. Green hydrogen as an alternative fuel for the shipping industry. Curr. Opin. Chem. Eng. 2021, 31, 100668. [Google Scholar] [CrossRef]
- Global hydrogen market insights, 2020–2024 by production process, end-user, generation system and region. Focus Catal. 2020, 5, 2.
- Osman, A.I.; Mehta, N.; Elgarahy, A.M.; Hefny, M.; Al-Hinai, A.; Al-Muhtaseb, A.H.; Rooney, D.W. Hydrogen production, storage, utilisation and environmental impacts: A review. Environ. Chem. Lett. 2021, 1–36. [Google Scholar] [CrossRef]
- Nicoletti, G.; Arcuri, N.; Nicoletti, G.; Bruno, R. A technical and environmental comparison between hydrogen and some fossil fuels. Energy Convers. Manag. 2015, 89, 205–213. [Google Scholar] [CrossRef]
- Dincer, I.; Zamfirescu, C. Sustainable hydrogen production options and the role of IAHE. Int. J. Hydrogen Energy 2012, 37, 16266–16286. [Google Scholar] [CrossRef]
- Chaubey, R.; Sahu, S.; James, O.O.; Maity, S. A review on development of industrial processes and emerging techniques for production of hydrogen from renewable and sustainable sources. Renew. Sustain. Energy Rev. 2013, 23, 443–462. [Google Scholar] [CrossRef]
- Acar, C.; Dincer, I. Comparative assessment of hydrogen production methods from renewable and non-renewable sources. Int. J. Hydrogen Energy 2014, 39, 1–12. [Google Scholar] [CrossRef]
- Dincer, I.; Acar, C. Review and evaluation of hydrogen production methods for better sustainability. Int. J. Hydrogen Energy 2015, 40, 11094–11111. [Google Scholar] [CrossRef]
- Enger, B.C.; Lødeng, R.; Holmen, A. A review of catalytic partial oxidation of methane to synthesis gas with emphasis on reaction mechanisms over transition metal catalysts. Appl. Catal. A Gen. 2008, 346, 1–27. [Google Scholar] [CrossRef]
- Agrafiotis, C.; von Storch, H.; Roeb, M.; Sattler, C. Solar thermal reforming of methane feedstocks for hydrogen and syngas production—A review. Renew. Sustain. Energy Rev. 2014, 29, 656–682. [Google Scholar] [CrossRef]
- Barelli, L.; Bidini, G.; Gallorini, F.; Servili, S. Hydrogen production through sorption-enhanced steam methane reforming and membrane technology: A review. Energy 2008, 33, 554–570. [Google Scholar] [CrossRef]
- Amin, A.; Croiset, E.; Epling, W. Review of methane catalytic cracking for hydrogen production. Int. J. Hydrogen Energy 2011, 36, 2904–2935. [Google Scholar] [CrossRef]
- Abbas, H.F.; Daud, W.W. Hydrogen production by methane decomposition: A review. Int. J. Hydrogen Energy 2010, 35, 1160–1190. [Google Scholar] [CrossRef]
- Demirci, U.B. The hydrogen cycle with the hydrolysis of sodium borohydride: A statistical approach for highlighting the scientific/technical issues to prioritize in the field. Int. J. Hydrogen Energy 2015, 40, 2673–2691. [Google Scholar] [CrossRef]
- Patel, N.; Miotello, A. Progress in Co–B related catalyst for hydrogen production by hydrolysis of boron-hydrides: A review and the perspectives to substitute noble metals. Int. J. Hydrogen Energy 2015, 40, 1429–1464. [Google Scholar] [CrossRef]
- Liu, B.H.; Li, Z.P. A review: Hydrogen generation from borohydride hydrolysis reaction. J. Power Sources 2009, 187, 527–534. [Google Scholar] [CrossRef]
- Moussa, G.; Moury, R.; Demirci, U.B.; Sener, T.; Miele, P. Boron-based hydrides for chemical hydrogen storage. Int. J. Energy Res. 2013, 37, 825–842. [Google Scholar] [CrossRef]
- Rand, D.A.J.; Dell, R.M. Fuels-hydrogen production: Coal gasification. EncyclElecrochem Power Sources 2009, 276–292. [Google Scholar]
- Hallenbeck, P.C.; Abo-Hashesh, M.; Ghosh, D. Strategies for improving biological hydrogen production. Bioresour. Technol. 2012, 110, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Eroglu, E.; Melis, A. Photo biological hydrogen production: Recent advances and state of the art. Bioresour. Technol. 2011, 102, 8403–8413. [Google Scholar] [CrossRef]
- Lee, H.-S.; Vermaas, W.F.; Rittmann, B.E. Biological hydrogen production: Prospects and challenges. Trends Biotechnol. 2010, 28, 262–271. [Google Scholar] [CrossRef]
- Singh, L.; Wahid, Z.A. Methods for enhancing bio-hydrogen production from biological process: A review. J. Ind. Eng. Chem. 2015, 21, 70–80. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, Y.; Zhang, Z.; Lee, D.-J.; Zhou, X.; Jing, Y.; Ge, X.; Jiang, D.; Hu, J.; He, C. Photo-fermentative hydrogen production from crop residue: A mini review. Bioresour. Technol. 2017, 229, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Elsharnouby, O.; Hafez, H.; Nakhla, G.; El Naggar, M.H. A critical literature review on biohydrogen production by pure cultures. Int. J. Hydrogen Energy 2013, 38, 4945–4966. [Google Scholar] [CrossRef]
- Sivagurunathan, P.; Kumar, G.; Bakonyi, P.; Kim, S.-H.; Kobayashi, T.; Xu, K.; Lakner, G.; Tóth, G.; Nemestothy, N.; Bélafi-Bakó, K. A critical review on issues and overcoming strategies for the enhancement of dark fermentative hydrogen production in continuous systems. Int. J. Hydrogen Energy 2016, 41, 3820–3836. [Google Scholar] [CrossRef]
- Edwards, P.P.; Kuznetsov, V.L.; David, W.I.F. Hydrogen energy. Philos. Trans. R. Soc. A 2007, 365, 1043–1056. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, M. Modern and prospective technologies for hydrogen production from fossil fuels. Int. J. Hydrogen Energy 1989, 14, 797–820. [Google Scholar] [CrossRef]
- Bartels, J.R.; Pate, M.B.; Olson, N.K. An economic survey of hydrogen production from conventional and alternative energy sources. Int. J. Hydrogen Energy 2010, 35, 8371–8384. [Google Scholar] [CrossRef]
- Yan, W.; Hoekman, K. Production of CO2-free hydrogen from methane dissociation: A review. Environ. Prog. Sustain. Energy 2014, 33, 213–219. [Google Scholar] [CrossRef]
- Gaudernack, B.; Lynum, S. Hydrogen from natural gas without release of CO2 to the atmosphere. Int. J. Hydrogen Energy 1998, 23, 1087–1093. [Google Scholar] [CrossRef]
- Ashik, U.P.M.; Daud, W.M.A.W.; Abbas, H.F. Production of greenhouse gas free hydrogen by thermo catalytic decomposition of methane a review. Renew. Sustain. Energy Rev. 2015, 44, 221–256. [Google Scholar] [CrossRef] [Green Version]
- Damen, K.; van Troost, M.; Faaij, A.; Turkenburg, W. A comparison of electricity and hydrogen production systems with CO2 capture and storage. Part A: Review and selection of promising conversion and capture technologies. Prog. Energy Combust. Sci. 2006, 32, 215–246. [Google Scholar] [CrossRef] [Green Version]
- Nikolaidis, P.; Poullikkas, A. A comparative overview of hydrogen production processes. Renew. Sustain. Energy Rev. 2017, 67, 597–611. [Google Scholar] [CrossRef]
- Bonner, M.; Botts, T.; McBreen, J.; Mezzina, A.; Salzano, F.; Yang, C. Status of advanced electrolytic hydrogen production in the United States and abroad. Int. J. Hydrogen Energy 1984, 9, 269–275. [Google Scholar] [CrossRef]
- Carpetis, C. An assessment of electrolytic hydrogen production by mans of photovoltaic energy conversion. Int. J. Hydrogen Energy 1984, 9, 969–992. [Google Scholar] [CrossRef]
- Johnson, A.C.; Barnstaple, A.G.; Bates, J.G.; Boardman, B.R.; Dewees, D.N.; Fleck, F.C.; Taylor, J.B.; Schofield, L.J.; Soots, V.; Wank, H. Hydrogen, a Challenging Opportunity. Report of the Ontario Hydrogen Energy Task Force; Ontario Government Publications Services: Toronto, ON, Canada, 1981. [Google Scholar]
- Baykara, S.Z.; Bilgen, E. An overall assessment of hydrogen production by solar water thermolysis. Int. J. Hydrogen Energy 1989, 14, 881–891. [Google Scholar] [CrossRef]
- Baykara, S. Hydrogen production by direct solar thermal decomposition of water, possibilities for improvement of process efficiency. Int. J. Hydrogen Energy 2004, 29, 1451–1458. [Google Scholar] [CrossRef]
- Baykara, S.Z. Experimental solar water thermolysis. Int. J. Hydrogen Energy 2004, 29, 1459–1469. [Google Scholar] [CrossRef]
- Bhandari, R.; Trudewind, C.A.; Zapp, P. Life cycle assessment of hydrogen production via Electrolysis—A review. J. Clean. Prod. 2014, 85, 151–163. [Google Scholar] [CrossRef]
- Muradov, N.Z.; Veziroglu, T.N. “Green” path from fossil-based to hydrogen economy: An overview of carbon-neutral technologies. Int. J. Hydrogen Energy 2008, 33, 6804–6839. [Google Scholar] [CrossRef]
- Turner, J.; Sverdrup, G.; Mann, M.K.; Maness, P.C.; Kroposki, B.; Ghirardi, M.; Evans, R.J.; Blake, D. Renewable hydrogen production. Int. J. Energy Res. 2008, 32, 379–407. [Google Scholar] [CrossRef] [Green Version]
- Yildiz, B.; Kazimi, M.S. Efficiency of hydrogen production systems using alternative nuclear energy technologies. Int. J. Hydrogen Energy 2006, 31, 77–92. [Google Scholar] [CrossRef]
- Dieguez, P.; Ursúa, A.; Sanchis, P.; Sopena, C.; Guelbenzu, E.; Gandia, L. Thermal performance of a commercial alkaline water electrolyzer: Experimental study and mathematical modeling. Int. J. Hydrogen Energy 2008, 33, 7338–7354. [Google Scholar] [CrossRef]
- Funk, J.E. Thermochemical hydrogen production: Past and present. Int. J. Hydrogen Energy 2001, 26, 185–190. [Google Scholar] [CrossRef]
- Lewis, M.A.; Masin, J.G. The evaluation of alternative thermochemical cycles—Part II: The down-selection process. Int. J. Hydrogen Energy 2009, 34, 4125–4135. [Google Scholar] [CrossRef]
- Andress, R.J.; Huang, X.; Bequette, B.W.; Martin, L.L. A systematic methodology for the evaluation of alternative thermochemical cycles for hydrogen production. Int. J. Hydrogen Energy 2009, 34, 4146–4154. [Google Scholar] [CrossRef]
- Forsberg, C.W.; Peterson, P.F.; Pickard, P.S. Molten-Salt-Cooled Advanced High-Temperature Reactor for Production of Hydrogen and Electricity. Nucl. Technol. 2003, 144, 289–302. [Google Scholar] [CrossRef]
- Forsberg, C.W. Future hydrogen markets for large-scale hydrogen production systems. Int. J. Hydrogen Energy 2007, 32, 431–439. [Google Scholar] [CrossRef]
- Acar, C.; Dincer, I. Comparative Environmental Impact Evaluation of Hydrogen Production Methods from Renewable and Nonrenewable Sources. In Causes, Impacts and Solutions to Global Warming; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2013; pp. 493–514. [Google Scholar]
- Schultz, K.; Herring, S.; Lewis, M.; Summers, W. The hydrogen reaction. Nucl. Eng. Int. 2005, 50, 10–19. [Google Scholar]
- Orhan, M.; Dincer, I.; Naterer, G. Cost analysis of a thermochemical Cu–Cl pilot plant for nuclear-based hydrogen production. Int. J. Hydrogen Energy 2008, 33, 6006–6020. [Google Scholar] [CrossRef]
- Ursúa, A.; Gandia, L.M.; Sanchis, P. Hydrogen Production from Water Electrolysis: Current Status and Future Trends; IEEE: Piscataway, NJ, USA, 2011; Volume 100, pp. 410–426. [Google Scholar]
- Srinivasan, N.; Shiga, Y.; Atarashi, D.; Sakai, E.; Miyauchi, M. A PEDOT-coated quantum dot as efficient visible light harvester for photocatalytic hydrogen production. Appl. Catal. B Environ. 2015, 179, 113–121. [Google Scholar] [CrossRef] [Green Version]
- Engels, H.; Funk, J.E.; Hesselmann, K.; Knoche, K.F. Thermochemical hydrogen production. Int. J. Hydrogen Energy 1987, 12, 291–295. [Google Scholar] [CrossRef]
- Bargigli, S.; Raugei, M.; Ulgiati, S. Comparison of thermodynamic and environmental indexes of natural gas, syngas and hydrogen production processes. Energy 2004, 29, 2145–2159. [Google Scholar] [CrossRef]
- Chiesa, P.; Consonni, S.; Kreutz, T. Co-production of hydrogen, electricity and CO from coal with commercially ready technology. Part A: Performance and emissions. Int. J. Hydrogen Energy 2005, 30, 747–767. [Google Scholar] [CrossRef]
- Cohce, M.; Dincer, I.; Rosen, M. Thermodynamic analysis of hydrogen production from biomass gasification. Int. J. Hydrogen Energy 2010, 35, 4970–4980. [Google Scholar] [CrossRef]
- Sazali, N. Emerging technologies by hydrogen: A review. Int. J. Hydrogen Energy 2020, 45, 18753–18771. [Google Scholar] [CrossRef]
- Suleman, F.; Dincer, I.; Agelin-Chaab, M. Environmental impact assessment and comparison of some hydrogen production options. Int. J. Hydrogen Energy 2015, 40, 6976–6987. [Google Scholar] [CrossRef]
- Baykara, S.Z. Hydrogen: A brief overview on its sources, production and environmental impact. Int. J. Hydrogen Energy 2018, 43, 10605–10614. [Google Scholar] [CrossRef]
- Baykara, S.Z. Hydrogen as fuel: A critical technology? Int. J. Hydrogen Energy 2005, 30, 545–553. [Google Scholar] [CrossRef]
- Kothari, R.; Buddhiand, D.; Sawhney, R.L. Comparison of environmental and economic aspects of various hydrogen production methods. Renew. Sustain. Energy Rev. 2008, 12, 553–563. [Google Scholar] [CrossRef]
- Ajanovic, A. On the economics of hydrogen from renewable energy sources as an alternative fuel in transport sector in Austria. Int. J. Hydrogen Energy 2008, 33, 4223–4234. [Google Scholar] [CrossRef]
- Sara, H.R.; Enrico, B.; Mauro, V.; Andrea, D.C.; Vincenzo, N. Techno-economic Analysis of Hydrogen Production Using Biomass Gasification—A Small Scale Power Plant Study. Energy Procedia 2016, 101, 806–813. [Google Scholar] [CrossRef]
- Sharma, M.; Kaushik, A. Biohydrogen Economy: Challenges and Prospects for Commercialization. In Biohydrogen Production: Sustainability of Current Technology and Future Perspective; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2017; pp. 253–267. [Google Scholar]
- Hay, J.X.W.; Wu, T.Y.; Juan, J.C.; Jahim, J.M. Biohydrogen production through photo fermentation or dark fermentation using waste as a substrate: Overview, economics, and future prospects of hydrogen usage. Biofuels Bioprod. Biorefin. 2013, 7, 334–352. [Google Scholar] [CrossRef]
- Kalamaras, C.; Efstathiou, A.M. Hydrogen Production Technologies: Current State and Future Developments. Conf. Pap. Energy 2013, 2013, 690627. [Google Scholar] [CrossRef] [Green Version]
- Zuttel, A.; Borgschulte, A.; Schlapbach, L. Hydrogen as a future energy Carrier. J. Appl. Electrochem. 2008, 38, 1483. [Google Scholar]
- Sakintuna, B.; Lamari-Darkrim, F.; Hirscher, M. Metal hydride materials for solid hydrogen storage: A review. Int. J. Hydrogen Energy 2007, 32, 1121–1140. [Google Scholar] [CrossRef]
- Sreedhar, I.; Kamani, K.M.; Kamani, B.M.; Reddy, B.M.; Venugopal, A. A Bird’s Eye view on process and engineering aspects of hydrogen storage. Renew. Sustain. Energy Rev. 2018, 91, 838–860. [Google Scholar] [CrossRef]
- Suh, M.P.; Park, H.J.; Prasad, T.K.; Lim, D.W. Hydrogen storage in metal–organic frameworks. Chem. Rev. 2012, 112, 782–835. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.; Agarwal, S.; Ichikawa, T. Catalytic Tuning of Sorption Kinetics of Lightweight Hydrides: A Review of the Materials and Mechanism. Catalysts 2018, 8, 651. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.; Sudik, A.; Wolverton, C.; Siegel, D.J. High capacity hydrogen storage materials: Attributes for automotive applications and techniques for materials discovery. Chem. Soc. Rev. 2010, 39, 656–675. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jain, A.; Miyaoka, H.; Ichikawa, T. Destabilization of lithium hydride by the substitution of group 14 elements: A review. Int. J. Hydrogen Energy 2016, 41, 5969–5978. [Google Scholar] [CrossRef]
- Jain, I.; Lal, C.; Jain, A. Hydrogen storage in Mg: A most promising material. Int. J. Hydrogen Energy 2010, 35, 5133–5144. [Google Scholar] [CrossRef]
- Wang, H.; Lin, H.; Cai, W.; Ouyang, L.; Zhu, M. Tuning kinetics and thermodynamics of hydrogen storage in light metal element based systems—A review of recent progress. J. Alloy. Compd. 2016, 658, 280–300. [Google Scholar] [CrossRef]
- Hu, M.; Geanangel, R.; Wendlandt, W. The thermal decomposition of ammonia borane. Thermochim. Acta 1978, 23, 249–255. [Google Scholar] [CrossRef]
- Klerke, A.; Christensen, C.H.; Nørskov, J.K.; Vegge, T. Ammonia for hydrogen storage: Challenges and opportunities. J. Mater. Chem. 2008, 18, 2304–2310. [Google Scholar] [CrossRef]
- Jain, I.P.; Jain, A. Novel hydrogen storage materials: A review of light weight complex hydrides. J. Alloys Compd. 2010, 503, 303–339. [Google Scholar] [CrossRef]
- Frank, E.D.; Elgowainy, A.; Khalid, Y.S.; Peng, J.-K.; Reddi, K. Refueling-station costs for metal hydride storage tanks on board hydrogen fuel cell vehicles. Int. J. Hydrogen Energy 2019, 44, 29849–29861. [Google Scholar] [CrossRef]
- Reuß, M.; Grube, T.; Robinius, M.; Preuster, P.; Wasserscheid, P.; Stolten, D. Seasonal storage and alternative carriers: A flexible hydrogen supply chain model. Appl. Energy 2017, 200, 290–302. [Google Scholar] [CrossRef]
- Raab, M.; Maier, S.; Dietrich, R.-U. Comparative techno-economic assessment of a large-scale hydrogen transport via liquid transport media. Int. J. Hydrogen Energy 2021, 46, 11956–11968. [Google Scholar] [CrossRef]
- Cardella, U.; Decker, L.; Sundberg, J.; Klein, H. Process optimization for large-scale hydrogen liquefaction. Int. J. Hydrogen Energy 2017, 42, 12339–12354. [Google Scholar] [CrossRef]
- Quarton, C.J.; Tlili, O.; Welder, L.; Mansilla, C.; Blanco, H.; Heinrichs, H.; Leaver, J.; Samsatli, N.J.; Lucchese, P.; Robinius, M.; et al. The curious case of the conflicting roles of hydrogen in global energy scenarios. Sustain. Energy Fuels 2020, 4, 80–95. [Google Scholar] [CrossRef] [Green Version]
- Ajanovic, A.; Haas, R. Prospects and impediments for hydrogen and fuel cell vehicles in the transport sector. Int. J. Hydrogen Energy 2021, 46, 10049–10058. [Google Scholar] [CrossRef]
- Dutton, A.G. Hydrogen Energy Technology, Tyndall Working Paper TWP 17; Tyndall Centre for Climate Change: Norwich, UK, 2002; Volume 30. [Google Scholar]
- Available online: https://www.alliedmarketresearch.com/hydrogen-fuel-cell-vehicle-market-2021 (accessed on 2 November 2021).
- Winter, M.; Brodd, R.J. What are batteries, fuel cells, and super capacitors? Chem. Rev. 2004, 104, 4245–4269. [Google Scholar] [CrossRef] [Green Version]
- Hua, T.; Ahluwalia, R.; Eudy, L.; Singer, G.; Jermer, B.; Asselin-Miller, N.; Wessel, S.; Patterson, T.; Marcinkoski, J. Status of hydrogen fuel cell electric buses worldwide. J. Power Sources 2014, 269, 975–993. [Google Scholar] [CrossRef] [Green Version]
- Abdelkareem, M.A.; Elsaid, K.; Wilberforce, T.; Kamil, M.; Sayed, E.T.; Olabi, A. Environmental aspects of fuel cells: A review. Sci. Total Environ. 2021, 752, 141803. [Google Scholar] [CrossRef] [PubMed]
- Fact of the Month April 2018: Fuel Cell Cost Decreased by 60% Since 2006. Available online: https://www.energy.gov/eere/fuelcells/fact-month-april-2018-fuel-cell-cost-decreased-60-2006 (accessed on 2 November 2021).
- Lattin, W.; Utgikar, V. Transition to hydrogen economy in the United States: A 2006 status report. Int. J. Hydrogen Energy 2007, 32, 3230–3237. [Google Scholar] [CrossRef]
- Ball, M.; Weeda, M. The hydrogen economy: Vision or reality? Int. J. Hydrogen Energy 2015, 40, 7903–7919. [Google Scholar] [CrossRef]
- Granovskii, M.; Dincer, I.; Rosen, M.A. Life cycle assessment of hydrogen fuel cell and gasoline vehicles. Int. J. Hydrogen Energy 2006, 31, 337–352. [Google Scholar] [CrossRef]
- Thomas, C. Fuel cell and battery electric vehicles compared. Int. J. Hydrogen Energy 2009, 34, 6005–6020. [Google Scholar] [CrossRef] [Green Version]
- Sharma, S.; Ghoshal, S.K. Hydrogen the future transportation fuel: From production to applications. Renew. Sustain. Energy Rev. 2015, 43, 1151–1158. [Google Scholar] [CrossRef]
- Cox, K.E.; Williamson, K.D. Hydrogen: Its Technology and Implications; CRC Press: Boca Raton, FL, USA, 1979. [Google Scholar]
- Schulte, I.; Hart, D.; van der Vorst, R. Issues affecting the acceptance of hydrogen fuel. Int. J. Hydrogen Energy 2004, 29, 677–685. [Google Scholar] [CrossRef]
- Acar, C.; Dincer, I. Review and evaluation of hydrogen production options for better environment. J. Clean. Prod. 2019, 218, 835–849. [Google Scholar] [CrossRef]
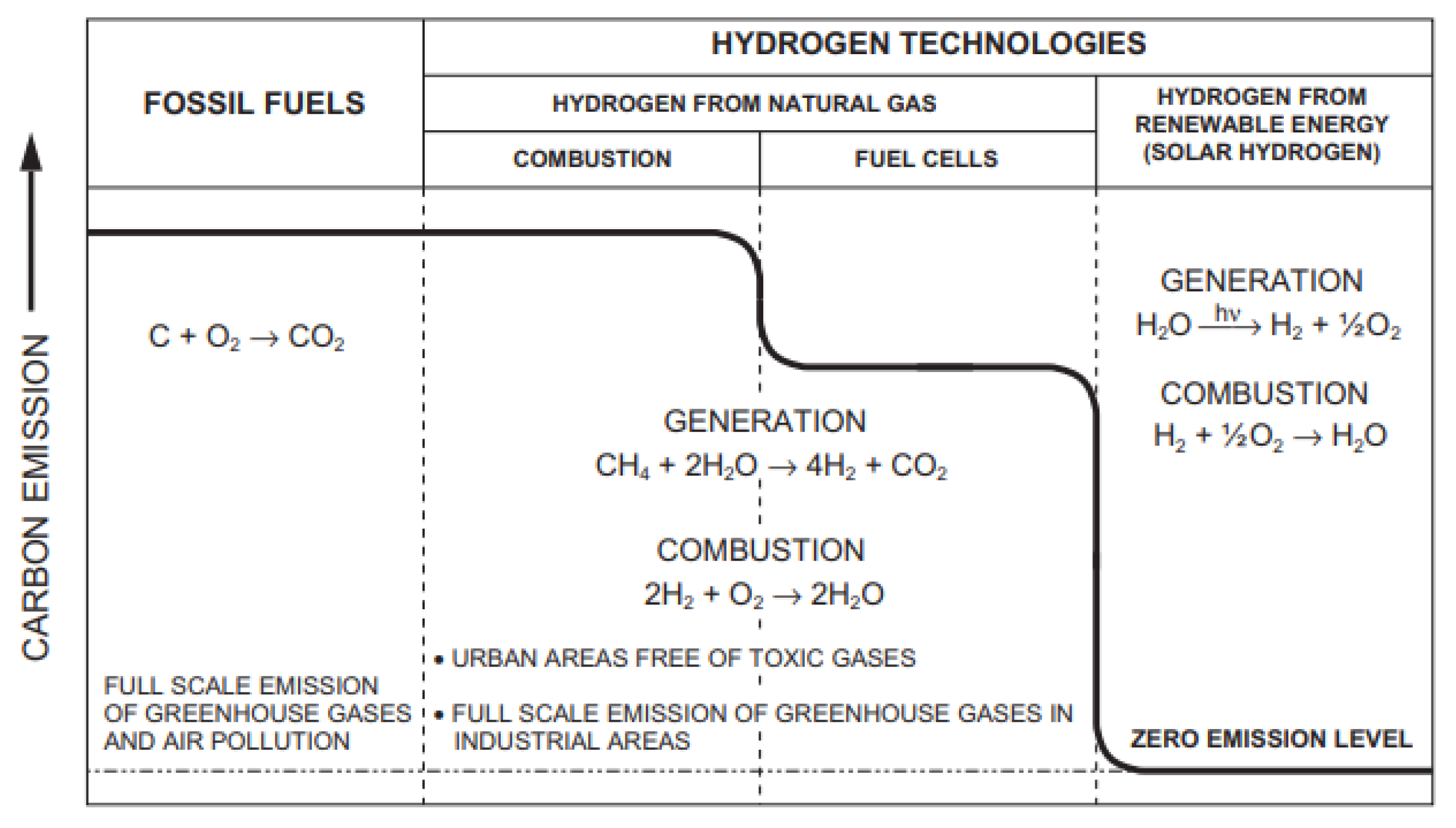
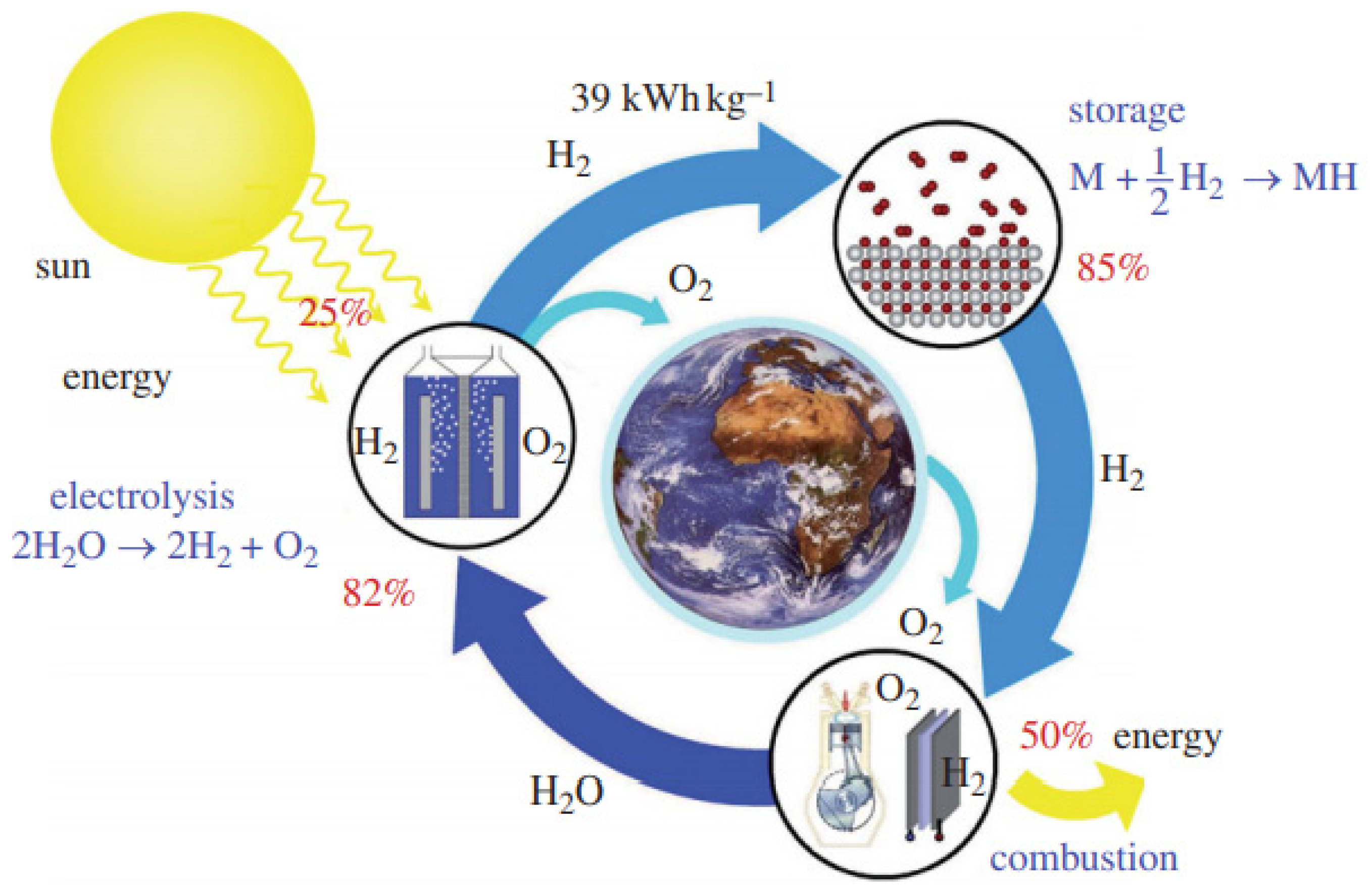
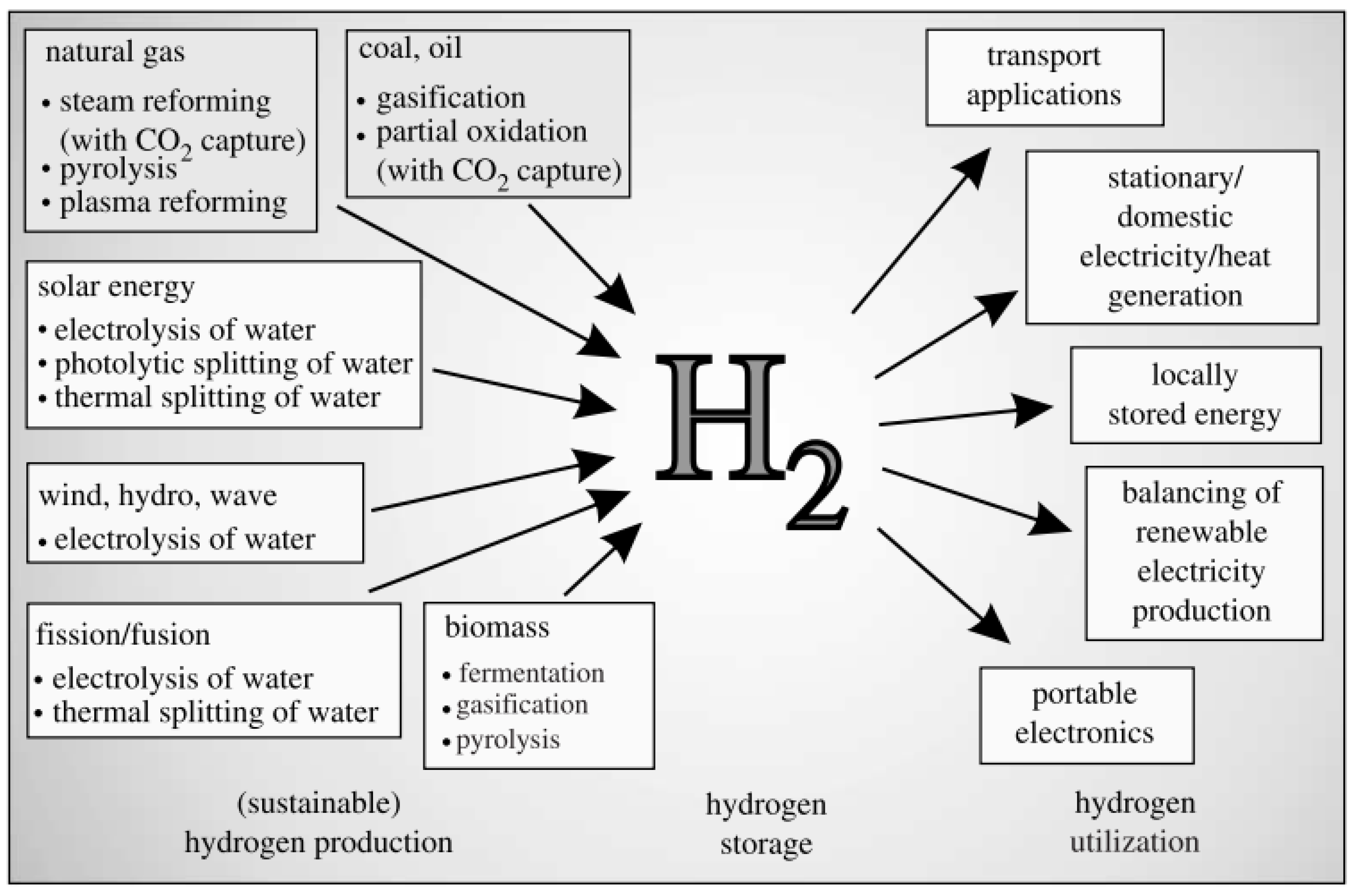
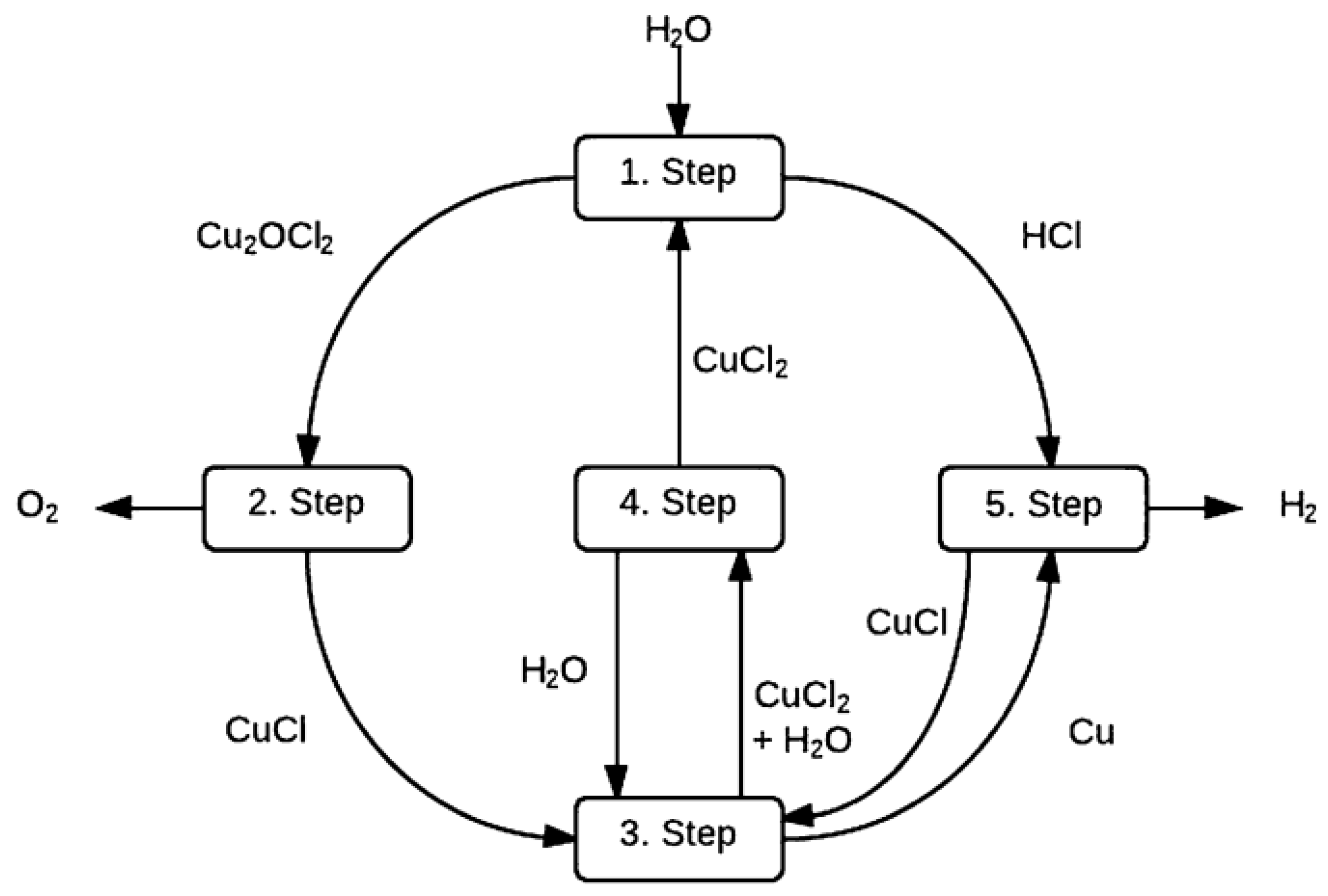

| Atomic number | 1 |
| Atomic mass | 1.00784 amu |
| Molecular mass | 2.01568 amu |
| Energy density | 10.05 MJ/m3 |
| Volumetric density | 0.08376 kg/m3 |
| Boiling point | −252.87 °C |
| Melting point | −259 °C |
| Critical point | −240.2 °C at 12.7 atm |
| Triple point | −259.3 °C at 0.07 atm |
| Higher heating value | 141.86 MJ/kg |
| Lower heating value | 119.93 MJ/kg |
| Energy System | Pollutants (kg/GJ) | Vapour Generation | Environmental Damage Ratio c | Environmental Compatibility Factor | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| CO2 | CO | SO2 | NOx | HC | PM a | Energy System | Global Warming | (%) b | |||
| Fossil fuel | 72.40 | 0.80 | 0.38 | 0.34 | 0.20 | 0.09 | 8.9 | 3900 | 0.782 | 18.24 | 0.055 |
| Coal/Synthetic fossil fuel | 100 | 0.65 | 0.50 | 0.32 | 0.12 | 0.14 | 9.3 | 3900 | 0.782 | 22.62 | 0.044 |
| Solar-hydrogen Energy | 0.00 | 0.00 | 0.00 | 0.00 | 0.10 | 0.00 | 6.0 | 0.0 | 0.001 | 1.0 | 1.00 |
| Source | Technology | Production Cost ($/kg) |
|---|---|---|
| Methane | Steam reforming | 0.75 |
| Coal | Gasification without CO2 sequestration | 0.92 |
| Nuclear | Electrolysis | 2.6–3.0 |
| Water splitting | 1.4–2.3 | |
| Biomass | Centralized biomass gasification | 1.2–2.4 |
| Solar | Photo catalytic water splitting | 5.0 |
| Hydrogen Storage Materials | Hydrogen Storage Capacity | Operating Temperature |
|---|---|---|
| Sorbent Systems Ex.—C-based materials, MOFs | 2–7 wt% | ~77 K |
| Conventional metal hydrides Interstitial Hydrides Ex.—LaNi5, FeTi etc. Ionic/covalent hydride Ex.—LiH, MgH2 etc. | 1~4 wt% >7 wt% | RT >600 K |
| Chemical Hydrides Ex.—NH3, NH3BH3 etc. | 17.8~20 wt% | 373~>773 K |
| Complex Hydrides Alanates Ex.—LiAlH4, NaAlH4, Mg(AlH4)2 etc. Borohydrides Ex.—LiBH4, NaBH4, Mg(BH4)2 etc. Amides Ex.—LiNH2, NaNH2, Mg(NH2)2 etc. Silanides Ex.—KSiH3, RbSiH3, CsSiH3 | 5.8~10.5 wt% 10~18.5 wt% 5~10 wt% 2~4.5 wt% | ≥400 K ≥400 K ≥400 K RT~500 K |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sharma, S.; Agarwal, S.; Jain, A. Significance of Hydrogen as Economic and Environmentally Friendly Fuel. Energies 2021, 14, 7389. https://doi.org/10.3390/en14217389
Sharma S, Agarwal S, Jain A. Significance of Hydrogen as Economic and Environmentally Friendly Fuel. Energies. 2021; 14(21):7389. https://doi.org/10.3390/en14217389
Chicago/Turabian StyleSharma, Shashi, Shivani Agarwal, and Ankur Jain. 2021. "Significance of Hydrogen as Economic and Environmentally Friendly Fuel" Energies 14, no. 21: 7389. https://doi.org/10.3390/en14217389
APA StyleSharma, S., Agarwal, S., & Jain, A. (2021). Significance of Hydrogen as Economic and Environmentally Friendly Fuel. Energies, 14(21), 7389. https://doi.org/10.3390/en14217389







