Biocrude Oil Production by Integrating Microalgae Polyculture and Wastewater Treatment: Novel Proposal on the Use of Deep Water-Depth Polyculture of Mixotrophic Microalgae
Abstract
1. Introduction
- stable culture and production because of the mixed component of algae with diverse of tolerance levels and the absence of pond crash;
- the efficient use of resources (nutrients) by niche complementarity;
- cheap and easy operation and maintenance.
2. Wastewater in the World and Japan
3. Algae Biomass Production and Wastewater Treatment
3.1. Mixotrophic Algae
3.2. Polyculture
3.3. Wastewater Treatment by Algae
4. Harvesting and Dewatering
4.1. Sedimentation
- Flocculation by inorganic chemicals
- Flocculation by organic chemicals
- Bio-flocculation
- Auto-flocculation
- Ultrasound and Electro-flocculation/flotation
4.2. Filtration
4.3. Centrifugation
5. Conversion of Algae Biomass into Bio-Crude Oil through HTL
- Robust and reliable: can be applied to wide range of carbon rich feedstocks at similar processing conditions.
- Conceptually simple (feed preparation, pump, heated pipe, separation of biocrude).
- Wet feedstocks with 70–90% moisture such as microalgae biomass, agriculture residues, sludge, manure, and pulp mill residue can be processed.
- The HTL biocrude oil is thermally stable and can be readily upgraded.
- High carbon efficiency of product: greater than 50% carbon conversion from the feedstock to hydrocarbon fuel product.
- Favorably comparable in economics with other biomass conversion technologies such as pyrolysis.
- Biocrude oil―the primary product: it has properties similar to conventional petroleum.
- Solid residues: it is used as a soil amendment for agricultural applications.
- Aqueous phase: it contains essential nutrients including phosphorus that can be used to cultivate algae or other biomass resources. Alternatively, the remaining carbon content in the aqueous phase can produce synthetic natural gas by gasification or methane by anaerobic digestion.
- Gas phase, which contains more than 90% of CO2 and trace amounts of hydrocarbon gases (CH4, C2H4, and C2H6).
6. Life-Cycle Assessment (LCA) of Microalgae-Based Biofuel Production
7. Conclusions
- Mixotrophic algae play a significant role in reducing COD/BOD in wastewater such as heterotrophic microbes.
- Mixotrophic algae have a high potential for solving the bottlenecks of CO2 supply because they can utilize atmospheric CO2 and organic carbon as a carbon source.
- Mixotrophic algae enable deep water culture, which significantly increases the capacities of aerial productivity of biomass/biocrude oil and wastewater treatment and reduces their land footprints.
- Polyculture outperforms the best monoculture by performing multiple functions simultaneously, such as producing resilient communities, enabling efficient nutrient uptake, resisting population crashes and undesirable invasive species, and showing fewer tendencies to trade-off between desirable functions.
- Energy-efficient filtration technologies combined with high-efficiency sedimentation through flocculation will significantly reduce the carbon footprint for algae biofuel production.
- Algal polycultures using sewage have HTL biocrude oil with a yield and quality that match the average monoculture-derived biocrude oil; it is available for existing oil refineries.
- HTL has bioavailability of the recycled nitrogen and phosphorus from the aqueous phase.
- HTL biocrude oil is more sustainable than conventional liquid fuels and other biofuels for combining GHG and EROI.
- Polyculture has the potential to improve the GHG and EROI significantly, compared with the best monoculture.
8. Future Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef] [PubMed]
- Henry, R.J. Evaluation of plant biomass resources available for replacement of fossil oil. Plant Biotechnol. J. 2010, 8, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Lundquist, T.J.; Woertz, I.C.; Quinn, N.W.T.; Benemann, J.R. A Realistic Technology and Engineering Assessment of Algae Biofuel Production; Energy Biosciences Institute, University of California: Berkeley, CA, USA, 2010. [Google Scholar]
- Gu, X.; Martinez-Fernandez, J.S.; Pang, N.; Fu, X.; Chen, S. Recent development of hydrothermal liquefaction for algal biorefinery. Renew. Sustain. Energy Rev. 2020, 121, 109707. [Google Scholar] [CrossRef]
- Wibawa, D.S.; Nasition, M.A.; Noguchi, R.; Ahamed, T.; Demura, M.; Watanabe, M.M. Microalgae oil production: A downstream approach to energy requirements for the Minamisoma pilot plant. Energies 2018, 11, 521. [Google Scholar] [CrossRef]
- Sasongko, N.A.; Noguchi, R.; Ito, J.; Demura, M.; Ichikawa, S.; Nakajima, M.; Watanabe, M.M. Engineering study of a pilot scale process plant for microalgae-oil production utilizing municipal wastewater and flue gases: Fukushima pilot plant. Energies 2018, 11, 1693. [Google Scholar] [CrossRef]
- Ishizaki, R.; Noguchi, R.; Putra, A.S.; Ichikawa, S.; Ahamed, T.; Watanabe, M.M. Reduction in energy requirement and CO2 emission for microalgae oil production using wastewater. Energies 2020, 13, 1641. [Google Scholar] [CrossRef]
- Ishizaki, R.; Putra, A.S.; Ichikawa, S.; Ahamed, T.; Watanabe, M.M.; Noguchi, R. Microalgae oil production using wastewater in Japan-Introducing operational cost function for sustainable management of WWTP. Energies 2020, 13, 5310. [Google Scholar] [CrossRef]
- Cui, Z.; Cheng, F.; Jarvis, J.M.; Brewer, C.E.; Jena, U. Roles of co-solvent in hydrothermal liqauefaction of low-lipid, high protein algae. Bioresour. Technol. 2020, 123454. [Google Scholar] [CrossRef]
- Lu, J.; Zhang, Z.; Zhang, L.; Fan, G.; Wu, Y. Catalytic hydrothermal liquefaction of microalgae over different biochars. Catal. Commun. 2021, 149, 106236. [Google Scholar] [CrossRef]
- Hamilton, C.E.; Rosmeissl, N. Exploring the Utilization of Complex Algal Communities to Address Algal Pond Crash and Increase Annual Biomass Production for Algal Biofuels; U.S. Department of Energy, Energy Efficiency & Renewable Energy: Washington, DC, USA, 2014.
- Lam, M.K.; Lee, K.T. Microalgae biofuels: A critical review of issues, problems and the way forward. Biotechnol. Adv. 2012, 30, 673–690. [Google Scholar] [CrossRef]
- Dalrymple, O.K.; Halfhide, T.; Udom, I.; Gilles, B.; Wolan, J.; Zhang, Q.; Ergas, S. Wastewater use in algae production for generation of renewable resources: A review and preliminary results. Aquat. Biosyst. 2013, 9, 2. [Google Scholar] [CrossRef]
- Demura, M.; Yoshida, M.; Yokoyama, A.; Ito, J.; Kobayashi, H.; Kayano, S.; Tamangawa, T.; Watanobe, M.; Date, N.; Osada, M.; et al. Biomass productivity of native algal communities in Minamisoma city, Fukushima Prefecture, Japan. Algal Res. 2018, 29, 22–35. [Google Scholar] [CrossRef]
- Chen, G.; Zhao, L.; Qi, Y. Enhancing the productivity of microalgae cultivated in wastewater toward biofuel production: A critical review. Appl. Energy 2015, 137, 282–291. [Google Scholar] [CrossRef]
- Craggs, R.; Sutherland, D.; Campbell, H. Hectare-scale demonstration of high rate algal ponds for enhanced wastewater treatment and biofuel production. J. Appl. Phycol. 2012, 24, 329–337. [Google Scholar] [CrossRef]
- Craggs, R.J.; Lundqusit, T.J.; Benemann, J.R. Wastewater treatment and algal biofuel production. In Algae for Biofuels and Energy; Borowitzka, M.A., Moheimanii, N.R., Eds.; Springer: Dordrecht, The Netherlands, 2013; pp. 153–163. [Google Scholar]
- Craggs, R.; Park, J.; Heubeck, S.; Sutherland, D. High rate algal pond systems for low-energy wastewater treatment, nutrient recovery and energy production. N. Z. J. Bot. 2014, 52, 60–73. [Google Scholar] [CrossRef]
- Sutherland, D.L.; Turnbull, M.H.; Craggs, R.J. Increased pond depth improve algal productivity and nutrient removal in wastewater treatment high rate algal ponds. Water Res. 2014, 2014, 271–281. [Google Scholar] [CrossRef]
- Mehrabadi, A.; Craggs, R.; Farid, M.M. Wastewater treatment high rate algal ponds (WWT HRAP) for low-cost biofuel producton. Bioresour. Technol. 2015, 184, 202–214. [Google Scholar] [CrossRef]
- Sheehan, J.; Dunahay, T.; Benemann, J.; Roessler, P. A Look Back at the U.S. Department of Energy’s Aquatic Species Program-Biodiesel from Algae; NERL/TP-580-24190; National Renewable Energy Laboratory: Golden, CO, USA, 1998. [Google Scholar]
- Acién, F.G.; Gómez-Serrano, C.; Morales-Amaral, M.M.; Fernández-Sevilla, J.M.; Mollina-Grima, E. Wastewater treatment using microalgae: How realistic a contribution night it be to significant urban wastewater treatment? Appl. Microbiol. Botechnol. 2016, 100, 9013–9022. [Google Scholar] [CrossRef]
- Acién Fernández, F.G.; Gómez-Serrano, C.; Fernández-Sevilla, J.M. Recovery of nutrients from wastewaters using microalgae. Front. Sustain. Food Syst. 2018, 2, 59. [Google Scholar] [CrossRef]
- Biddy, M.; Davis, R.; Jones, S.; Zhu, Y. Whole Algae Hydrothermal Liquefaction Technology Pathway; Technical Report NREL/TP-5100-58051, PNNL-22314; U.S. Department of Commerce National Technical Information Service: Springfield, VA, USA, 2013.
- Isdepsky, A.; Gotoh, H.A.; Watanabe, S.; Watanabe, M.M. Extraordinarily High Areal Production of Mixotrophic Microalgae in 1.4 m Water-Depth Culture Tank Using Primarily-Treated Wastewater. Partly in: Annual Report of Development of Low Carbon Technologies Based on Collaboration between Efficient Productions of Algae Biomass and Their Conversions to High Performance Plastics, Low Carbon Technology Research, Development and Demonstration Program, Ministry of Environment, Tokyo Japan. 2019. Available online: https://www.env.go.jp/earth/pndanka/cpttv.funds/pdf/db/205.pdf (accessed on 22 October 2021). (In Japanese)
- AIIC (Algae Industry Incubation Consortium Japan). Annual Report on R&D Project for Biofuel Production Using Algae; Agency for Natural Resources and Energy, Ministry of Economy, Trade, and Industry (METI): Tokyo, Japan, 2019; pp. 1–140. (In Japanese)
- AIIC (Algae Industry Incubation Consortium Japan). Annual Report on R&D Project for Biofuel Production Using Indigenous Algae; Next Generation Renewable Energy Technology Development Project: Fukushima, Japan, 2016. (In Japanese) [Google Scholar]
- Ichikawa, S.; Jain, A.; Gohara, T.; Matsumoto, S.; Aramaki, T.; Ito, J.; Noguchi, R.; Watanabe, M.M.; Nakajima, M. Continuous biocrude production from polycultured microalgae biomass using a bench-scale hydrothermal tubular reactor. In Proceedings of the 7th Conference of International Society for Applied Phycology (ISAP2020–2021 Virtual), Virtual, 14 May–13 August 2021. [Google Scholar]
- WWAP (World Water Assessment Program). The United Nations World Water Development Report 2017, Wastewater: The Untapped Resource; UNESCO: Paris, France, 2017. [Google Scholar]
- Sato, T.; Qadir, M.; Yamamoto, S.; Endo, T.; Zahoor, A. Global, reginal, and country level need for data on wastewater generation, treatment, and use. Agric. Water Manag. 2013, 130, 1–13. [Google Scholar] [CrossRef]
- WWAP (World Water Assessment Program). The United Nations World Water Development Report 2012, Managing Water under Uncertainty and Risk; UNESCO: Paris, France, 2012. [Google Scholar]
- Ministry of Land, Infrastructure, Transport and Tourism (MLIT). Survey for Formulating a Comprehensive Sewerage Development Plan. for Each Basin: Guideline and Commentary; MLIT: Tokyo, Japan, 2015. Available online: https://www.mlit.go.jp/common/001065300.pdf (accessed on 22 October 2021). (In Japanese)
- Daverey, A.; Pandey, D.; Verma, P.; Verma, S.; Shah, V.; Dutta, K.; Arunachalam, K. Recent advances in energy efficient biological treatment of municipal wastewater. Bioresour. Technol. Rep. 2019, 7, 100252. [Google Scholar] [CrossRef]
- Magill, B. Sewage Plants Overlooked Source of CO2. Researching and Reporting the Science and Impacts of Climate Change; Climate Central: Princeton, NJ, USA, 2016; Available online: https://www.climatecentral.org/news/sewage-plants-overlooked-co2-source-20840 (accessed on 22 October 2021).
- Sewage and Wastewater Management Department and National Institute for Land and Infrastructure Management, Ministry of Land, Infrastructure (MLIT), Transport and Tourism. Sewage Technology Vision. 2021. Available online: http://www.nilim.go.jp/lab/eag/pdf/01_r0302_ichibukaitei.pdf (accessed on 22 October 2021). (In Japanese)
- Ministry of Land, Infrastructure, Transport and Tourism (MLIT). About Global Warming Countermeasures in the Sewerage Field. 2019. Available online: https://www.mlit.go.jp/singikai/infra/kankyou/6/images/05.pdf (accessed on 22 October 2021). (In Japanese)
- Steen, I. Phosphorus availability in the 21st century: Management of a non-renewable resource. Phosphorus Potassium 1998, 217, 25–31. [Google Scholar]
- Ministry of Land, Infrastructure, Transport and Tourism (MLIT). Guide to the Recycling of Phosphorus in Sewerage. 2010. Available online: https://www.mlit.go.jp/common/000113958.pdf (accessed on 22 October 2021). (In Japanese)
- De-Bashan, L.E.; Bashan, Y. Recent advances in removing phosphorus from wastewater and its future use as fertilizer (1997–2003). Water Res. 2004, 38, 4222–4246. [Google Scholar] [CrossRef] [PubMed]
- Hashizume, M.; Yoshida, M.; Demura, M.; Watanabe, M.M. Culture study on utilization of phosphite by green microalgae. J. Appl. Phycol. 2020, 32, 889–899. [Google Scholar] [CrossRef]
- Liu, Y.-J.; Gu, J.; Liu, Y. Energy self-sufficient biological municipal wastewater reclamation: Present status, challenges and solutions forward. Bioresour. Technol. 2018, 269, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Kong, Z.; Xue, Y.; Wang, T.; Kato, H.; Li, Y.-Y. A comparative long-term operation using up-flow anaerobic sludge blanket (UASB) and anaerobic membrane bioreactor (AnMBR) for the upgrading of anaerobic treatment of N, N-dimethylformamide-containing wastewater. Sci. Total Environ. 2020, 699, 134370. [Google Scholar] [CrossRef] [PubMed]
- Yadav, G.; Shanmugam, S.; Sivaramakrishnan, R.; Kumar, D.; Mathimani, T.; Brindhadevi, K.; Pugazhendhi, A.; Rajendran, K. Mechanism and challenges behind algae as a wastewater treatment choice for bioenergy production and beyond. Fuel 2021, 285, 119093. [Google Scholar] [CrossRef]
- Do, M.H.; Ngo, H.H.; Guo, W.S.; Liu, Y.; Chang, S.W.; Nguyen, D.D.; Nghiem, L.D.; Ni, B.J. Challenges in the application of microbial fuel cells to wastewater treatment and energy production: A mini review. Sci. Total Environ. 2018, 639, 910–920. [Google Scholar] [CrossRef]
- Goto, Y.; Yoshida, N. Scaling up microbial fuel cells for treating swine wastewater. Water 2019, 11, 1803. [Google Scholar] [CrossRef]
- Ma, B.; Xu, X.; Wei, Y.; Ge, C.; Peng, Y. Recent advances in controlling denitritation for achieving denitratation/anamox in mainstream wastewater treatment plants. Bioresour. Technol. 2020, 299, 122697. [Google Scholar] [CrossRef]
- Yoshida, M.; Tanabe, Y.; Yonezawa, N.; Watanabe, M.M. Energy innovation potential of oleaginous microalgae. Bifuels 2012, 3, 761–781. [Google Scholar] [CrossRef]
- Carney, L.T.; Reinsch, S.S.; Lane, P.D.; Solberg, O.D.; Jansen, L.S.; Williams, K.P.; Trent, J.D.; Lane, T.W. Microbiome analysis of a microalgal mass culture growing in municipal wastewater in a prototype OMEGA photobioreactor. Algal Res. 2014, 4, 52–61. [Google Scholar] [CrossRef]
- Plöhn, M.; Spain, O.; Sirin, S.; Silva, M.; Escudero-Oñate, C.; Ferrando-Climent, L.; Allahverdiyeva, Y.; Funk, C. Wastewater treatment by microalgae. Physiol. Plant. 2021, 172, 568–578. [Google Scholar] [CrossRef]
- Bohutskyi, P.; Spiering, R.E.; Phan, D.; Kopachevsky, A.M.; Tang, Y.; Betenbaugh, M.J.; Bouwer, E.J.; Lundquist, T.J. Bioenergy from wastewater resurces: Nutrine removal, productivity and sttleabilitynof indigenous algal-bacteria polyculture, an effect of biomass composition variability on methane production kinetics and anaerobic digestion energy balance. Algal. Res. 2018, 36, 217–228. [Google Scholar] [CrossRef]
- Zhang, T.-U.; Wu, Y.-H.; Zhuang, L.-L.; Wang, X.-X.; Hu, H.-Y. Screening heterotrophic microalgal strains by using the Biolog method for biofuel production from organic wastewater. Algal Res. 2014, 6, 175–179. [Google Scholar]
- Godrijan, J.; Drapeau, D.; Balch, W.M. Mixotrophc uptake of organic compounds by coccolithophores. Limnol. Oceanogr. 2020, 65, 1410–1421. [Google Scholar] [CrossRef]
- Girard, J.-M.; Roy, M.-L.; Hafsa, M.B.; Gagnon, J.; Faucheux, N.; Heitz, M.; Tremblay, R.; Deschênes, J.-S. Mixotrophic cultivation of green microalgae Scenedesmus obliquus on cheese whey permeate for biodiesel production. Algal Res. 2014, 5, 241–248. [Google Scholar] [CrossRef]
- Ji, Y.; Hu, W.; Li, X.; Ma, G.; Song, M.; Pei, H. Mixotrophic growth and biochemical analysis of Chlorella vulgaris cultivated with diluted monosodium glutamate wastewater. Bioresour. Technol. 2014, 152, 471–476. [Google Scholar] [CrossRef]
- Park, W.-K.; Moon, M.; Kwak, M.-S.; Jeon, S.; Choi, G.-G.; Yang, J.-W.; Lee, B. Use of orange peel extract for mixotrophic cultivation of Chlorella vulgaris: Increased production of biomass and FAMEs. Bioresour. Technol. 2014, 171, 343–349. [Google Scholar] [CrossRef]
- Moreno-Garcia, L.; Adjallé, K.; Barnabé, S.; Raghavan, G.S.V. Microalgae biomass production for a biorefinery system: Recent advances and the way toward sustainability. Renew. Sustain. Energy Rev. 2017, 76, 493–506. [Google Scholar] [CrossRef]
- Villarejo, A.; Orús, M.I.; Martínez, F. Coordination of photosynthetic and respiratory metabolism in Chlorella vulgaris UAM 101 in the light. Physiol. Plant. 1995, 94, 680–686. [Google Scholar] [CrossRef]
- Liang, Y.; Sarkany, N.; Cui, Y. Biomass and lipid productivities of Chlorella vulgaris under autotrophic, heterotrophic and mixotrophic growth conditions. Biotechnol. Lett. 2009, 31, 1043–1049. [Google Scholar] [CrossRef]
- Perez-Garcia, O.; Escalante, F.M.E.; de-Bashan, L.E.; Bashan, Y. Heterotrophic cultures of microalgae: Metabolism and potential products. Water Res. 2011, 45, 11–36. [Google Scholar] [CrossRef]
- Davis, R.; Markham, J.N.; Kinchin, C.M.; Canter, C.; Han, J.; Li, Q.; Coleman, A.; Wigmosta, M.; Zhu, Y. 2017 Algae Harmonization Study: Evaluating the Potential for Future Algal Biofuel Costs, Sustainability, and Resource Assessment from Harmonized Modeling; Technical Report ANL-18/12; NREL/TP-5100-70715; PNNL-27547; National Renewable Energy Laboratory: Golden, CO, USA, 2018. [Google Scholar]
- Tilman, D. The ecological consequences of changes in biodiversity: A search for general principles. Ecology 1999, 80, 1455–1474. [Google Scholar] [CrossRef]
- Dzialowski, A.R.; Smith, V.H. Nutrient dependent effects of consumer identity and diversity on freshwater ecosystem function. Freshw. Biol. 2008, 53, 148–158. [Google Scholar] [CrossRef]
- Zeller, S.L.; Kalinina, O.; Flynn, D.F.B.; Schmid, B. Mixtures of genetically modified wheat lines outperform monocultures. Ecol. Appl. 2012, 22, 1817–1826. [Google Scholar] [CrossRef]
- Cardinale, B.J.; Gross, K.; Fritschie, K.; Flombaum, P.; Fox, J.W.; Rixen, C.; van Ruijven, J.; Reich, P.B.; Scherer-Lorenzen, M.; Wilsey, B.J. Biodiversity simultaneously enhances the production and stability of community biomass, but the effects are independent. Ecology 2013, 94, 1697–1707. [Google Scholar] [CrossRef]
- Hulot, F.D.; Lacroix, G.; Lescher-Moutoué, F.; Loreau, M. Functional diversity governs ecosystem response to nutrient enrichment. Nature 2000, 405, 340–344. [Google Scholar] [CrossRef]
- Worm, B.; Lotze, H.K.; Hillebrand, H.; Sommer, U. Consumer versus resource control of species diversity and ecosystem functioning, Nature 2002, 417, 848–851. Nature 2002, 417, 848–851. [Google Scholar] [CrossRef]
- Norberg, J. Biodiversity and ecosystem functioning: A complex adaptive systems approach. Limnol. Oceanogr. 2004, 49, 1269–1277. [Google Scholar] [CrossRef]
- Tylianakis, J.M.; Rand, T.A.; Kahmen, A.; Klein, A.-M.; Buchmann, N.; Perner, J.; Tscharntke, T. Resource heterogeneity moderates the biodiversity-function relationship in real world ecosystems. PLoS Biol. 2008, 6, e122. [Google Scholar] [CrossRef]
- Behl, S.; de Schryver, V.; Diehl, S.; Stibor, H. Trophic transfer of biodiversity effects: Functional equivalence of prey diversity and enrichment? Ecol. Evol. 2012, 2, 3110–3122. [Google Scholar] [CrossRef] [PubMed]
- Novoveská, L.; Zaparta, A.K.M.; Zabolotney, J.B.; Atwood, M.C.; Sundstrom, E.R. Optimizing microalgae cultivation and wastewater treatment in large-scale offshore photobioreactors. Algal Res. 2016, 2016, 85–94. [Google Scholar] [CrossRef]
- Narwani, A.; Lashaway, A.R.; Hietala, D.C.; Savage, P.E.; Cardinale, B.J. Power of plankton: Effect of algal biodiversity on biocrude production and stability. Environ. Sci. Technol. 2016, 50, 13142–13150. [Google Scholar] [CrossRef]
- Hietala, D.C.; Koss, C.K.; Narwani, A.; Lashaway, A.R.; Godwin, C.M.; Cardinale, B.J.; Savage, P.E. Influence of biodiversity, biochemical composition, and species identity on the quality of biomass and biocrude oil produced via hydrothermal liquefaction. Algal Res. 2017, 26, 203–217. [Google Scholar] [CrossRef]
- Godwin, C.M.; Hietala, D.C.; Lashaway, A.R.; Narwani, A.; Savage, P.E.; Cardinal, B.J. Ecological stoichiometry meets ecological engineering: Using polycultures to enhance the multifunctionality of algal biocrude system. Environ. Sci. Technol. 2017, 51, 11450–11458. [Google Scholar] [CrossRef]
- Godwin, C.M.; Lashaway, A.R.; Hietala, D.C.; Savage, P.E.; Cardinal, B.J. Biodiversity improves the ecological design of sustainable biofuel systems. GCB Bioenergy 2018, 10, 752–765. [Google Scholar] [CrossRef]
- Carruthers, D.N.; Godwin, C.M.; Hietala, D.C.; Cardinal, B.J.; Lin, X.N.; Savage, P.E. Biodiversity improves life cycle sustainability metrics in algal biofuel production. Environ. Sci. Technol. 2019, 53, 9279–9288. [Google Scholar] [CrossRef]
- Chen, W.-T.; Zhang, Y.; Zhang, J.; Yu, G.; Schdeman, L.C.; Zhang, P.; Minarick, M. Hydrothermal liquefaction of mixed-culture algal biomass from wastewater treatment system into bio-crude oil. Bioresour. Technol. 2014, 152, 130–139. [Google Scholar] [CrossRef]
- Thomas, P.K.; Dunn, G.P.; Coats, E.R.; Newby, D.T.; Feris, K.P. Algal diversity and traits predict biomass yield and grazing resistance in wastewater cultivation. J. Appl. Phycol. 2019, 31, 2323–2334. [Google Scholar] [CrossRef]
- Kim, B.-H.; Choi, J.-E.; Cho, K.; Kang, Z.; Ramanan, R.; Moon, D.-G.; Kim, H.-S. Influence of water depth on microalgal production, biomass harvest, and energy consumption in high rate algal pond using municipal wastewater. J. Microbiol. Biotechnol. 2018, 28, 630–637. [Google Scholar] [CrossRef]
- Audu, M.; Myint, M.T.; Cheng, F.; Mallick, K.; Jena, U.; Nirmalakhandan, N.; Brewer, C.E. Hydrothermal liquefaction of algae grown on brackish dairy wastewater. In Proceedings of the ASABE 2018 Annual International Meeting, Detroit, MI, USA, 29 July–1 August 2018. [Google Scholar] [CrossRef]
- Podkuiko, L.; Kasemets, M.-L.; Kikas, T.; Lips, I. Cultivation of algae polyculture in municipal wastewater with CO2 supply. Environ. Clim. Technol. 2020, 24, 188–200. [Google Scholar] [CrossRef]
- Thomas, P.K.; Dunn, G.P.; Good, A.R.; Callahan, M.P.; Coats, E.R.; Newby, D.T.; Feris, K.P. A natural algal polyculture outperforms an assembled polyculture in wastewater-based open pond biofuel production. Algal Res. 2019, 40, 101488. [Google Scholar] [CrossRef]
- Arashiro, L.T.; Ferrer, I.; Rousseau, D.P.L.; van Hulle, S.W.H.; Garfí, M. The effect of primary treatment of wastewater in high rate algal pond systems: Biomass and bioenergy recovery. Bioresour. Technol. 2019, 280, 27–36. [Google Scholar] [CrossRef]
- Goswami, G.; Makut, B.B.; Das, D. Sustainable production of bio-crude oil via hydrothermal liquefaction of symbiotically grown biomass of microalgae-bacteria coupled with effective wastewater treatment. Sci. Rep. 2019, 9, 15016. [Google Scholar] [CrossRef]
- Liu, J.; Danneels, B.; Vanormelingen, P.; Vyverman, W. Nutrient removal form horticultural wastewater by benthic filamentous algae Klebsormidium sp., Stigeoclonium spp. and their communities: From laboratory flask to outdoor Algal Turf Scrubber (ATS). Water Res. 2016, 92, 61–68. [Google Scholar] [CrossRef]
- Ryšánek, D.; Elster, J.; Kováčik, L.; Škaloud, P. Diversity and dispersal capacities of a terrestrial algal genus Klebsormidium (Streptophyta) in polar regions. FEMS Micobiol. Ecol. 2016, 92, fiw039. [Google Scholar] [CrossRef]
- Molina Grima, E.; Femández Sevilla, J.M.; Sánchez Pérez, J.A.; García Camacho, F. A study on simultaneous photolimitation and photoinhibition in dense microalgal cultures taking into account incident and averaged irradiances. J. Biotechnol. 1996, 45, 59–69. [Google Scholar] [CrossRef]
- Benemann, J. Wastewater treatment with microalgae ponds. In Proceedings of the 7th Conference of International Society for Applied Phycology (ISAP2020–2021 Virtual), Virtual, 14 May–13 August 2021. [Google Scholar]
- Lundquist, T.J.; Blackwell, P.E.S.; Diego, E.; Coyne, J.; Benemann, J. Microalgae for water and nutrient recycling. In Proceedings of the 7th Conference of International Society for Applied Phycology (ISAP2020–2021 Virtual), Virtual, 14 May–13 August 2021. [Google Scholar]
- Craggs, R.J.; Park, J.B.K.; Weaver, L. Removal of faecal indicators and human viral pathogens in hectare-scale wastewater treatment high rate algal pond system. In Proceedings of the 7th Conference of International Society for Applied Phycology (ISAP2020–2021 Virtual), Virtual, 14 May–13 August 2021. [Google Scholar]
- Acién, F.G. Microalgae production coupled to wastewater treatment. In Proceedings of the 7th Conference of International Society for Applied Phycology (ISAP2020–2021 Virtual), Virtual, 14 May–13 August 2021. [Google Scholar]
- Sun, L.; Tian, Y.; Zhang, J.; Cui, H.; Zuo, W.; Li, J. A novel symbiotic system combining algae and sludge membrane bioreactor technology for wastewater treatment and membrane fouling mitigation: Performance and mechanism. Chem. Eng. J. 2018, 344, 246–253. [Google Scholar] [CrossRef]
- Ye, J.; Liang, J.; Wang, L.; Markou, G.; Jia, Q. Operation optimization of a photo-sequencing batch reactor for wastewater treatment: Study on influencing factors and impact on symbiotic microbial ecology. Bioresour.Technol. 2018, 252, 7–13. [Google Scholar] [CrossRef]
- Sun, L.; Tian, Y.; Zhang, J.; Li, H.; Tang, C.; Li, J. Wastewater treatment and membrane fouling with algae-activated sludge culture in a novel membrane bioreactor: Influence of inoculation ratios. Chem. Eng. J. 2018, 343, 455–459. [Google Scholar] [CrossRef]
- Tang, C.-C.; Tian, Y.; He, Z.-W.; Zuo, W.; Zhang, J. Performance and mechanism of a novel algal-bacterial symbiosis system based on sequencing catch suspended biofilm reactor treating domestic wastewater. Bioresour. Technol. 2018, 265, 422–431. [Google Scholar] [CrossRef]
- Mujtaba, G.; Rizwan, M.; Kim, G.; Lee, K. Removal of nutrients and COD through co-culturing activated sludge and immobilized Chlorella vulgaris. Chem. Eng. J. 2018, 343, 155–162. [Google Scholar] [CrossRef]
- Zhu, S.; Qin, L.; Feng, P.; Shang, C.; Wang, Z.; Yuan, Z. Treatment of low C/N ratio wastewater and biomass production using co-culture of Chlorella vulgaris and activated sludge in a batch photobioreactor. Bioresour. Technol. 2019, 274, 313–320. [Google Scholar] [CrossRef]
- Sepehri, A.; Sarrafzadeh, M.-H.; Avateffazeli, M. Interaction between Chlorella vulgaris and nitrifying-enriched activated sludge in the treatment of wastewater with low C/N ratio. J. Clean. Prod. 2020, 247, 119164. [Google Scholar] [CrossRef]
- Fan, J.; Chen, Y.; Zhang, T.C.; Ji, B.; Gao, L. Performance of Chlorella sorokiniana-activated sludge consortium treating wastewater under light-limited heterotrophic condition. Chem. Eng. J. 2020, 382, 122799. [Google Scholar] [CrossRef]
- Nugyen, T.-T.-D.; Niguyen, T.-T.; Binh, Q.A.; Bui, X.-T.; Ngo, H.H.; Vo, H.N.P.; Lin, K.-Y.A.; Vo, T.-D.-H.; Guo, W.; Lin, C.; et al. Co-culture of microalgae-activated sludge for wastewater treatment and biomass production: Exploring their role under different inoculation ratios. Bioresour. Technol. 2020, 314, 123754. [Google Scholar] [CrossRef]
- Randall, H. NMSU Environmental Engineers Examine New Method for Wasterwater Treatment, Reuse. 2019. Available online: https://ce.nmsu.edu/nmsu-environmental-engineers-examine-new-method-for-wastewater-treatment-reuse/ (accessed on 21 October 2021).
- Brennan, L.; Owende, P. Biofuels from microalgae-a review of technologies for production, processing, and extractions of biofuels and co-products. Renew. Sustain. Energy Rev. 2010, 14, 557–577. [Google Scholar] [CrossRef]
- Mohn, F.H. Experiences and strategies in the recovery of biomass from mass cultures of microalgae. In Algae Biomass, Production and Use; Shelef, G., Soeder, C.J., Eds.; Elsevier/North-Holland Biomedical Press: Amsterdam, The Netherlands, 1980; pp. 547–571. [Google Scholar]
- Uduman, N.; Qi, Y.; Danquah, M.K.; Forde, G.M.; Hoadley, A. Dewatering of microalgal cultures: A major bottleneck to algae-based fuels. J. Renew. Sustain. Energy 2010, 2, 012701. [Google Scholar] [CrossRef]
- Fasaei, F.; Bitter, J.H.; Slegers, P.M.; van Boxtel, A.J.B. Techno-economic evaluation of microalgae harvesting and dewatering systems. Algal Res. 2018, 31, 347–362. [Google Scholar] [CrossRef]
- Pahl, S.L.; Lee, A.K.; Kalaitzidis, T.; Ashman, P.J.; Sathe, S.; Lewis, D.M. Harvesting, Thickening and Dewatering Microalgae Biomass. In Algae for Biofuels and Energy; Borowitzka, M.A., Moheimani, N.R., Eds.; Springer: Dordrecht, The Netherlands, 2013; pp. 165–185. [Google Scholar]
- Grima, E.M.; Belarbi, E.H.; Fernandez, F.G.A.; Medina, A.R.; Chisti, Y. Recovery of microalgal biomass and metabolites: Process options and economics. Biotechnol. Adv. 2003, 20, 491–515. [Google Scholar] [CrossRef]
- Danquah, M.K.; Ang, L.; Uduman, N.; Moheimani, N.; Fordea, G.M. Dewatering of microalgal culture for biodiesel production: Exploring polymer flocculation and tangential flow filtration. J. Chem. Technol. Biotechnol. 2009, 84, 1078–1083. [Google Scholar] [CrossRef]
- Shelef, G.; Sukenik, A.; Green, M. Microalgae Harvesting and Processing: A Literature Review; Report SERI/STR-231-2396; Technion Research and Development Foundation Ltd.: Haifa, Israel, 1984. Available online: https://www.nrel.gov/docs/legosti/old/2396.pdf (accessed on 22 October 2021).
- Becker, E.W.; Venkataraman, L.V. Biotechnology and Exploitation of Algae: The Indian Approach: A Comprehensive Report on the Cultivation and Utilization of Microalgae Performed at the Central Food Technological Research Institute, Mysore, India; German Agency for Technical Cooperation: Eschborn, Germany, 1982. [Google Scholar]
- O’Connell, D.; Savelski, M.; Slater, C.S. Life cycle assessment of dewatering routes for algae derived biodiesel processes. Clean Technol. Environ. Policy 2013, 15, 567–577. [Google Scholar] [CrossRef]
- Lardon, L.; Hélias, A.; Sialve, B.; Steyer, J.-P.; Bernard, O. Life-cycle assessment of biodiesel production from microalgae. Environ. Sci. Technol. 2009, 43, 6475–6481. [Google Scholar] [CrossRef]
- Shelef, G.; Soeder, C.J. (Eds.) Algae Biomass, Production and Use; Elsevier/North Holland Biomedical Press: Amsterdam, The Netherlands, 1980. [Google Scholar]
- Burlew, J.S. (Ed.) Current status of the large-scale culture of Algae 1. In Algal Culture from Laboratory to Pilot Plant; Carnegie Institution of Washington Publication: Washington, DC, USA, 1953; pp. 3–23. [Google Scholar]
- Muylaert, K.; Bastiaens, L.; Vandamme, D.; Gouveia, L. Harvesting of microalgae: Overview of process options and their strengths and drawbacks. In Microalgae-based Biofuels and Bioproducts; Gonzalez-Fernandez, C., Muñoz, R., Eds.; Woodhead Publishing: Oxford, UK, 2017; pp. 113–132. [Google Scholar]
- Singh, G.; Patidar, S.K. Microalgae Harvesting Techniques: A review. J. Environ. Manage. 2018, 217, 499–508. [Google Scholar] [CrossRef]
- Richmond, A. (Ed.) Biological principles of mass cultivation. In Handbook of Microalgal Culture: Biotechnology and Applied Phycology; Blackwell Publishing: Oxford, UK, 2003; pp. 125–177. [Google Scholar]
- Moraine, R.; Shelef, G.; Sandbank, E.; Bahr-Moshe, Z.; Shvartzburd, L. Recovery of sewage-borne algae: Flocculation, flotation, and centrifugation techniques. In Algae Biomass, Production and Use; Shelef, G., Soeder, C.J., Eds.; Elsevier/North-Holland Biomedical Press: Amsterdam, The Netherlands, 1980; pp. 531–545. [Google Scholar]
- Tenney, M.W.; Echelberger, W.F.; Schuessler, R.G.; Pavoni, J.L. Algal flocculation with synthetic organic polyelectrolytes. Appl. Microbiol. 1969, 18, 965–971. [Google Scholar] [CrossRef]
- Tilton, R.C.; Murphy, L.; Dixon, J.K. Flocculation of algae with synthetic polymeric flocculants. Water Res. 1972, 6, 155–164. [Google Scholar] [CrossRef]
- Bilanovic, D.; Shelef, G.; Sukenik, A. Flocculation of microalgae with cationic polymers—Effects of medium salinity. Biomass 1988, 17, 65–76. [Google Scholar] [CrossRef]
- Udom, I.; Zaribaf, B.H.; Halfhide, T.; Gillie, B.; Dalrymple, O.; Zhang, Q.; Ergas, S.J. Harvesting microalgae grown on wastewater. Bioresour. Technol. 2013, 139, 101–106. [Google Scholar] [CrossRef]
- Molina Grima, E.; Acien Fernandez, F.G.; Robles Medina, A. Downstream processing of cell-mass and products. In Handbook of Microalgal Culture: Biotechnology and Applied Phycology; Richmond, A., Ed.; Blackwell Publishing: Oxford, UK, 2003; pp. 215–251. [Google Scholar]
- Mohn, F.H.; Soeder, C.J. Improved technologies for the harvesting and processing of microalgae and their impact on production costs. Arch. Hydrobiol. Bech. Ergebn. Limnol. 1978, 11, 228–253. [Google Scholar]
- Biller, P.; Ross, A. Potential yields and properties of oil from the hydrothermal liquefaction of microalgae with different biochemical content. Bioresour. Technol. 2011, 102, 215–225. [Google Scholar] [CrossRef]
- Jazrawi, C.; Biller, P.; Ross, A.B.; Montoya, A.; Maschmeyer, T.; Haynes, B.S. Pilot Plant Testing of Continuous Hydrothermal Liquefaction of Microalgae. Algal Res. 2013, 2, 268–277. [Google Scholar] [CrossRef]
- Wang, B.; Huang, Y.; Zhang, J. Hydrothermal liquefaction of lignite, wheat straw and plastic waste in sub-critical water for oil: Product distribution. J. Anal. Appl. Pyrolysis 2014, 110, 382–389. [Google Scholar] [CrossRef]
- Gollakota, A.R.K.; Kishore, N.; Gu, S. A review on hydrothermal liquefaction of biomass. Renew. Sustain. Energy Rev. 2018, 81, 1378–1392. [Google Scholar] [CrossRef]
- Kumar, M.; Oyedun, A.O.; Kumar, A. A review on the current status of various hydrothermal technologies on biomass feedstock. Renew. Sustain. Energy Rev. 2018, 81, 1742–1770. [Google Scholar] [CrossRef]
- Chen, P.H.; Quinn, J.C. Microalgae to biofuels through hydrothermal liquefaction: Open-source techno-economic analysis and life cycle assessment. Appl. Energy 2021, 289, 116613. [Google Scholar] [CrossRef]
- Elliott, D.C. Historical developments in hydroprocessing bio-oils. Energy Fuels 2007, 21, 1792–1815. [Google Scholar] [CrossRef]
- Jin, B.; Duan, P.; Xu, Y.; Wang, F.; Fan, Y. Co-liquefaction of micro-and macroalgae in subcritical water. Bioresour. Technol. 2013, 149, 103–110. [Google Scholar] [CrossRef]
- Elliott, D.C.; Biller, P.; Ross, A.B.; Schmidt, A.J.; Jones, S.B. Hydrothermal liquefaction of biomass: Developments from batch to continuous process. Bioresour. Technol. 2015, 178, 147–156. [Google Scholar] [CrossRef]
- Toor, S.S.; Rosendahl, L.; Rudolf, A. Hydrothermal liquefaction of biomass: A review of subcritical water technologies. Energy 2011, 36, 2328–2342. [Google Scholar] [CrossRef]
- Palomino, A.; Godoy-Silva, R.D.; Raikova, S.; Chuck, C.J. The storage stability of biocrude obtained by the hydrothermal liquefaction of microalgae. Renew. Energy 2020, 145, 1720–1729. [Google Scholar] [CrossRef]
- Zhang, L.; Li, C.J.; Zhou, D.; Zhang, S.C.; Chen, J.M. Hydrothermal liquefaction of water hyacinth: Product distribution and identification. Energy Sources A: Recovery Util. Environ. Eff. 2013, 35, 1349–1357. [Google Scholar]
- Nallasivam, J.; Eboibi, B.E.; Isdepsky, A.; Lavanya, M.; Bhaskar, S.; Chinnasamy, S. Hydrothermal liquefaction of water hyacinth (Eichhorniacrassipes): Influence of reaction temperature on product yield, carbon and energy recovery, and hydrocarbon species distribution in biocrude. Biomass Convers. Biorefin. 2020. [Google Scholar] [CrossRef]
- Chen, X.; Peng, X.; Ma, X.; Wang, J. Investigation of Mannich reaction during co-liquefaction of microalgae and sweet potato waste. Bioresour. Technol. 2019, 284, 286–292. [Google Scholar] [CrossRef]
- Hietala, D.C.; Godwin, C.M.; Cardinale, B.J.; Savage, P.E. The independent and coupled effects of feedstock characteristics and reaction conditions on biocrude production by hydrothermal liquefaction. Appl. Energy 2019, 235, 714–728. [Google Scholar] [CrossRef]
- Eboibi, B.E. Impact of time on yield and properties of biocrude during downstream processing of product mixture derived from hydrothermal liquefaction of microalga. Biomass Convers. Biorefin. 2019, 9, 379–387. [Google Scholar] [CrossRef]
- Fortier, M.-O.P.; Roberts, G.W.; Stagg-Williams, S.M.; Sturm, B.S.M. Life cycle assessment of bio-jet fuel from hydrothermal liquefaction of microalgae. Appl. Energy 2014, 122, 73–82. [Google Scholar] [CrossRef]
- Carneiro, M.L.N.M.; Pradelle, F.; Braga, S.L.; Gomes, M.S.P.; Martins, A.R.F.A.; Turkovics, F.; Pradelle, R.N.C. Potential of biofuels from algae: Comparison with fossil fuels, ethanol and biodiesel in Europe and Brazil through life cycle assessment (LCA). Renew. Sustain. Energy Rev. 2017, 73, 632–653. [Google Scholar] [CrossRef]
- Frank, E.; Wang, M.; Han, J.; Elgowainy, A.; Palou-Rivera, I. Life-Cycle Analysis of Algal-Based Fuels with the GREET Model; Energy Systems Division, Argonne Laboratory: San Francisco, CA, USA, 2011. [Google Scholar]
- Frank, E.D.; Elgowainy, A.; Han, J.; Wang, Z. Life cycle comparison of hydrothermal liquefaction and lipid extraction pathways to renewable diesel from algae. Mitig. Adapt. Strateg. Glob. Chang. 2013, 18, 137–158. [Google Scholar] [CrossRef]
- de Boer, K.; Moheimani, N.R.; Borowitzka, M.A.; Bahri, P.A. Extraction and conversion pathways for microalgae to biodiesel: A review focused on energy consumption. J. Appl. Phycol. 2012, 24, 1681–1698. [Google Scholar] [CrossRef]
- Flesch, A.; Beer, T.; Campbell, P.K.; Batten, D.; Grant, T. Greenhouse gas balance and algae-based biodiesel. In Algae for Biofuels and Energy; Springer: Berlin/Heidelberg, Germany, 2013; pp. 233–254. [Google Scholar]
- Woertz, I.C.; Benemann, J.R.; Du, N.; Unnasch, S.; Mendola, D.; Mitchell, B.G.; Lundquist, T.J. Life cycle GHG emissions from microalgal biodiesel- A CA-GREET model. Environ. Sci. Technol. 2014, 48, 6060–6068. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Environment. Total Greenhouse Gas Emissions-Guidelines for Calculation Method 2017. Available online: https://www.env.go.jp/policy/local_keikaku/data/guideline.pdf (accessed on 22 October 2021). (In Japanese)
- Vasudevan, V.; Stratton, R.W.; Pearlson, M.N.; Jersey, G.R.; Beyene, A.G.; Weissman, J.C.; Rubino, M.; Hileman, J.I. Environmental performance of algal biofuel technology options. Environ. Sci. Technol. 2012, 46, 2451–2459. [Google Scholar] [CrossRef] [PubMed]
- Manganaro, J.L.; Lawal, A. CO2 life-cycle assessment of the production of algae-based liquid fuel compared to crude oil to diesel. Energy Fuels. 2016, 30, 3130–3139. [Google Scholar] [CrossRef]
- Bošnjakovíc, M.; Sinaga, N. The perspective of large-scale production of algae biodiesel. Appl. Sci. 2020, 8181. [Google Scholar] [CrossRef]
- Tyner, W.E.; Taheripour, F.; Zhuang, Q.; Birur, D.; Baldos, U.L. Land Use Changes and Consequent CO2 Emissions due to US Corn ethanol Production: A Comprehensive Analysis; Final Report; Department of Agricultural Economics at Purdue University: Lafayette, IN, USA, 2010. [Google Scholar]
- Liu, X.; Saydah, B.; Eranki, P.; Colosi, L.M.; Mitchell, B.G.; Rhodes, J.; Clarens, A.F. Pilot-scale data provide enhanced estimates of the life cycle energy and emissions profile of algae biofuels produced via hydrothermal liquefaction. Bioresour. Technol. 2013, 148, 163–171. [Google Scholar] [CrossRef]
- Yoo, G.; Park, M.S.; Yang, J.-W.; Choi, M. Lipid content in microalgae determines the quality of biocrude and Energy Return on Investment of hydrothermal liquefaction. Appl. Energy 2015, 156, 354–361. [Google Scholar] [CrossRef]
- Mehrabadi, A.; Craggs, R.; Farid, M.M. Wastewater treatment high rate algal pond biomass for bio-crude oil production. Bioresour. Technol. 2017, 224, 255–264. [Google Scholar] [CrossRef]
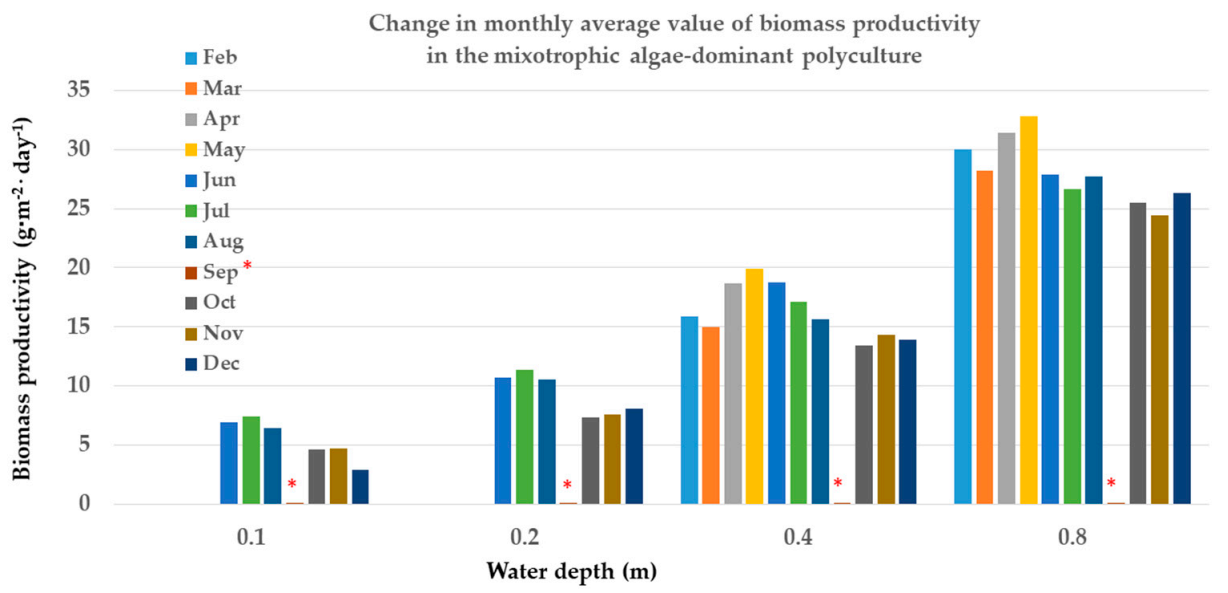
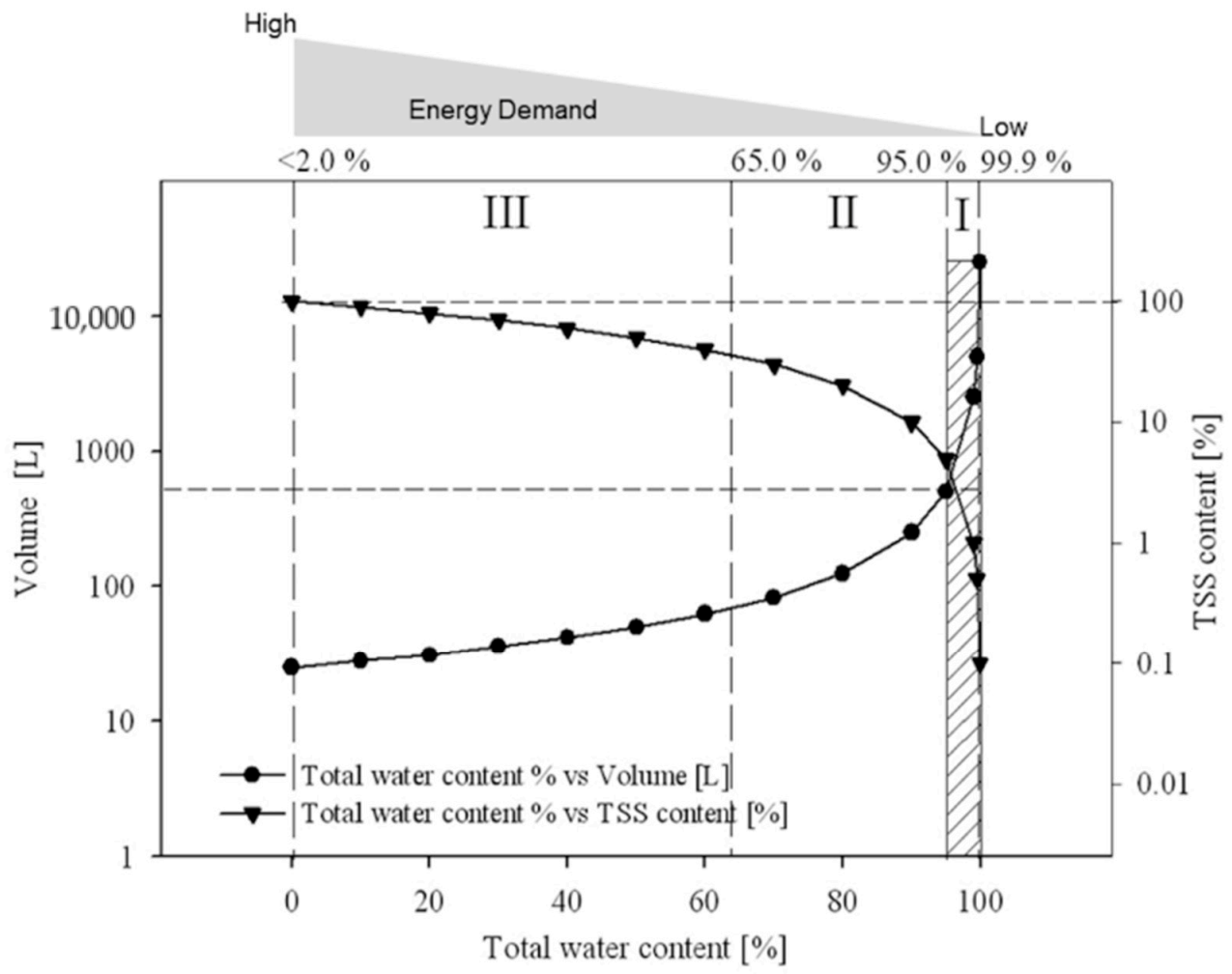
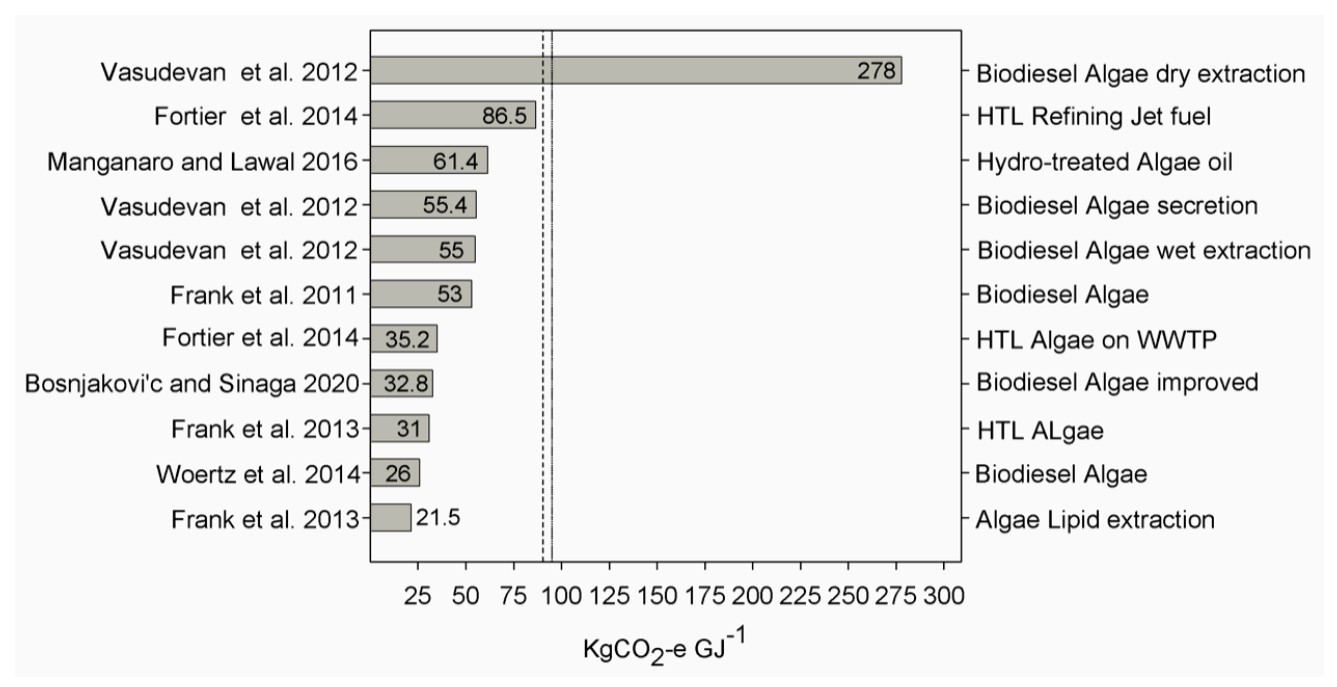
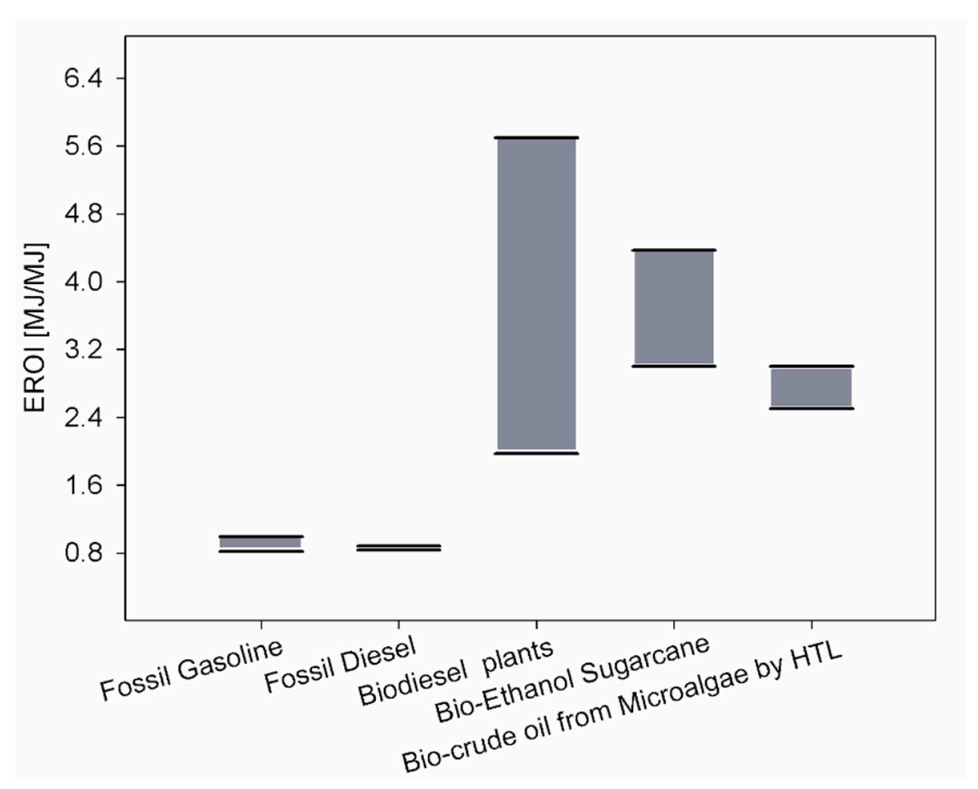
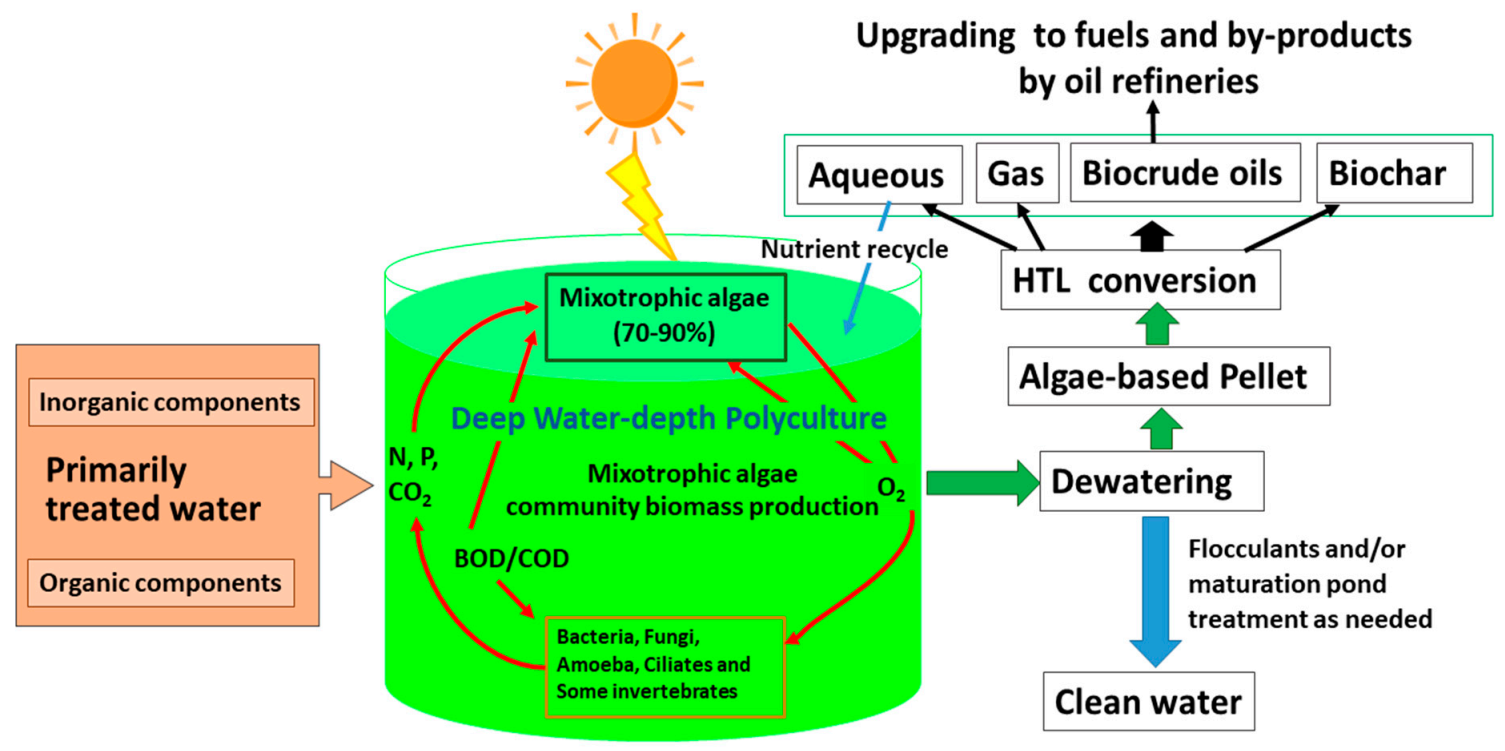
|
| Daily Maximum Inflow of Sewage | 10,000 m3·day−1 | 50,000 m3·day−1 | 100,000 m3·day−1 |
|---|---|---|---|
| Conventional activated sludge process | |||
| Construction fee (million JPY) | 6374 | 16,212 | 24,235 |
| Unit construction fee (million JPY·m−3) | 0.64 | 0.32 | 0.24 |
| Operation/maintenance fee (million JPY·year−1) | 100 | 302 | 488 |
| Unit operation/maintenance fee (JPY·m−3·day−1) | 27 | 17 | 13 |
| Energy consumption (MJ·day−1) | 30,178 | 106,960 | 175,472 |
| Unit energy consumption (MJ·m−3) | 3.02 | 2.14 | 1.76 |
| Anaerobic–anoxic–oxic (A2O) process | |||
| Construction fee (million JPY) | 7329 | 19,642 | 29,979 |
| Unit construction fee (million JPY·m−3) | 0.73 | 0.39 | 0.30 |
| Operation/maintenance fee (million JPY·year−1) | 116 | 371 | 611 |
| Unit operation/maintenance fee (JPY·m−3·day−1) | 32 | 20 | 17 |
| Energy consumption (MJ·day−1) | 38,582 | 116,510 | 187,180 |
| Unit energy consumption (MJ·m−3) | 3.86 | 2.33 | 1.87 |
| Culture | Conditions | Results Highlighted | Ref. |
|---|---|---|---|
| Natural algal polyculture | 5 ha demonstration HRAP system treating primary settled wastewater. 15 month operation. |
| [16] |
| Natural algal polyculture | 5 ha demonstration HRAP system treating primary settled wastewater. | See Table 4 in the present paper | [18] |
| Natural algal polyculture | Three HRAP pond depths (20, 30 and 40 cm) treating primary settled wastewater. |
| [19] |
| Natural algal polyculture | Offshore photobioreactor. Liquid (wastewater) depth ranged from 5 to 25 cm. |
| [70] |
| Monoculture: six microalgae species Polyculture: two-species, four-species, and six-species | 10 L tank. Temperture: constant (22 °C) and variable (17 °C to 27 °C). |
| [71,72] |
| Monoculture: five microalgae species Polyculture: two-species, four-species, and six-species | 9.5 L chemostat with 30% refresh of medium per week. |
| [73] |
| Monoculture: five microalgae species Polyculture: two-species and four species | 1100 L outdoor pond. |
| [74] |
| Monoculture: five microalgae species Polyculture: two-species and four-species. | 1100 L outdoor pond. |
| [75] |
| Mixed-culture microalgae | Directly harvested from wastewater treatment sysytem | Maximum biocrude-oil yield: 49% of volatile matter (=25.7% of dry weight). Elemental composition of C, H, N, and O: 65.7, 8.76, 3.93, and 21.7. | [76] |
| Monoculture: Five microalgae species Polyculture: three-species, six-species, and a natural polyculture | Batch culture using flasks with anaerobic digester effluent. |
| [77] |
| Natural algal polyculture | HRAP with different depths of 20, 30, and 40 cm. |
| [78] |
| Polyculture of microalgae isolated | Photobioreactor with 37 L brackish dairy wastewater. |
| [79] |
| Monoculture: three microalgae species Polyculture: three-species | 300 mL conical flasks with primarily treated municipal wastewater. |
| [80] |
| Natural algal polyculture An assembled polyculture of three-species | Open raceway pond with anaerobic digester effluent. |
| [81] |
| Natural algal polyculture | HRAPs with and without primarily treated water. |
| [82] |
| Consortium of two algae and two bacteria species | 7.5 L phtobioreactor wth wastewater from the paper indusry. |
| [83] |
| Natural algal polyculture | Outdoor vessel-type reactor with water depth 0.8 m. |
| [14,26,27,28] |
| Water Quality Variables (ppm) | Conventional WTP System | HRAP System | HRAP System with CO2 Addition | HRAP System with P Flocculation | ||||
|---|---|---|---|---|---|---|---|---|
| Median | 95 PCTL | Median | 95 PCTL | Median | 95 PCTL | Median | 95 PCTL | |
| BOD | <40 | <110 | <15 | <50 | <15 | <40 | <10 | <30 |
| TSS | <80 | <150 | <15 | <60 | <15 | <40 | <10 | <30 |
| NH4-N | <10 | <30 | <5 | <20 | <2 | <10 | <4 | <15 |
| TN | <10 | <50 | <10 | <40 | <5 | <15 | <5 | <15 |
| DRP | <10 | <15 | <5 | <10 | <2 | <5 | <1 | <5 |
| TP | <10 | <15 | <7 | <15 | <7 | <10 | <2 | <10 |
| E. coli (MPN/100 mL) | <40,000 | <400,000 | <100 | <1000 | <100 | <1000 | <100 | <1000 |
| Water Quality Variables | Quality Standard of Discharge Water Based on the Sewage Act in Japan | Influent | Effluent | Effluent after Flocculant Treatment |
|---|---|---|---|---|
| BOD (ppm) | <15 | 252.6 | 43.4 | 7.9 |
| TSS (ppm) | <40 | 158.4 | 81.6 | 2.7 |
| TN (ppm) | <20 | 38.8 | 13.9 | 7.3 |
| TP (ppm) | <3 | 5.6 | 3.8 | 0.2 |
| Dewatering Technology | Biomass Concentration (% TSS) | Energy Consumption (KWh/m3) | Ref. | |
|---|---|---|---|---|
| Input | Output | |||
| Micro-strainer | <0.1 | 1.5 | 0.20 | [102] |
| Gravity sedimentation | 1.5 | 0.10 | [103] | |
| Flocculation | 2–8 | 0.15–2.4 | [104,105] | |
| Filter basket | 5.0 | 0.20 | [106] | |
| Dissolved air flotation | 6.0 | 20.00 | [107] | |
| Vibrating screen | 0.1–0.5 | 6.0 | 0.40 | [102] |
| Filter thickener | 7.0 | 1.60 | [106] | |
| Cylindrical sieve | 7.5 | 0.30 | [102] | |
| Suction filter | 8.0 | 0.10 | [106] | |
| Vacuum drum | 8.0 | 5.90 | [108] | |
| Belt filter | 4.0 | 9.5 | 0.45 | [106] |
| Belt filter press | 0.5 | 18.0 | 0.50 | [102] |
| Centrifugation | 0.1 | 22.0 | 8.00 | [103] |
| Chamber filter | 5.0 | 27.0 | 0.88 | [106] |
| (KWh/Kg BMDW) | ||||
| Drum Dryer | 20 | >90.0 | 8.7 | [109] |
| 96.0 | 3.5 | [110] | ||
| Spray dryer | 2.0 | 95.0 | 35.0 | [108] |
| Biofuel Feedstock | Unit MJ·kg−1 BM | Production Process | Energy Input MJ·kg−1 BM | Accumulated Energy Input MJ·kg−1 BM | Accumulated CO2 Emission * kg-CO2·kg−1 BM |
|---|---|---|---|---|---|
| Dry Algae Biomass (BM) | 20 (polyculture using wastewater) 24 (monoculture of high lipid BM) | 1. Cultivation (pumping, mixing) | 2.50 | 2.50 | 0.04875 |
| 2. Cultivation (Fertilizer use) | 5.43 | 7.93 | 0.15464 | ||
| 3. Harvesting/Dewatering (~80 wt% moisture | 1.99 | 9.92 | 0.19344 | ||
| 4. Drying (from ~80 wt% moisture to <10% moisture) | 9.00 | 18.92 | 0.36894 | ||
| Net Energy in polyculture for HTL (MJ·kg−1 BM): process 1, 3) | 15.51 | ||||
| Net energy in monoculture of high lipid BM (MJ·kg−1 dry BM): process 1–4 | 5.08 | ||||
| Biodiesel (30 wt% of dry BM) | 12 | BM at <10 wt% moisture | 18.92 | 0.36894 | |
| Dry BM extraction (40 wt% lipids) | 0.93 | 19.85 | 0.38708 | ||
| Glycerol (96 g·kg−1 BM) | 1.65 | Transesterification (Methanolysis) | 0.87 | 20.72 | 0.40404 |
| Remaining BM (60 wt%) | 8.15 | ||||
| Total output at 1 kg BM | 21.8 | ||||
| Net Energy (MJ·kg−1 BM) | 1.08 | ||||
| Biocrude oil (HTL) (50 wt% conversion rate) | 17.5 | BM at ~80 wt% moisture | 4.49 | 0.08756 | |
| HTL conversion (MJ·kg−1 biocrude oil) | 3,25 | 7.74 | 0.15093 | ||
| Solid/liquid separation (Tricanter) | 0.15 | 7.89 | 0.15386 | ||
| Dewatering by sedimentation | 0.7 | 8.59 | 0.16751 | ||
| Thermal water removal at ~50 wt% moisture | 1.85 | 10.44 | 0.203580 | ||
| Remaining solids (~35 wt%) for biogas | 10.6 | ||||
| Gas phase (10–15 wt%) | 0.11–0.165 | ||||
| Total HTL Output at 1 kg Biomass | 28.21 | ||||
| Net Energy (MJ·kg−1 BM) | 17.77 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Watanabe, M.M.; Isdepsky, A. Biocrude Oil Production by Integrating Microalgae Polyculture and Wastewater Treatment: Novel Proposal on the Use of Deep Water-Depth Polyculture of Mixotrophic Microalgae. Energies 2021, 14, 6992. https://doi.org/10.3390/en14216992
Watanabe MM, Isdepsky A. Biocrude Oil Production by Integrating Microalgae Polyculture and Wastewater Treatment: Novel Proposal on the Use of Deep Water-Depth Polyculture of Mixotrophic Microalgae. Energies. 2021; 14(21):6992. https://doi.org/10.3390/en14216992
Chicago/Turabian StyleWatanabe, Makoto M., and Andreas Isdepsky. 2021. "Biocrude Oil Production by Integrating Microalgae Polyculture and Wastewater Treatment: Novel Proposal on the Use of Deep Water-Depth Polyculture of Mixotrophic Microalgae" Energies 14, no. 21: 6992. https://doi.org/10.3390/en14216992
APA StyleWatanabe, M. M., & Isdepsky, A. (2021). Biocrude Oil Production by Integrating Microalgae Polyculture and Wastewater Treatment: Novel Proposal on the Use of Deep Water-Depth Polyculture of Mixotrophic Microalgae. Energies, 14(21), 6992. https://doi.org/10.3390/en14216992





