Potential of the Middle Cambrian Aquifer for Carbon Dioxide Storage in the Baltic States
Abstract
:1. Introduction
2. Materials and Methods
2.1. Regulatory Guiding Principles for CO2 Storage in Europe
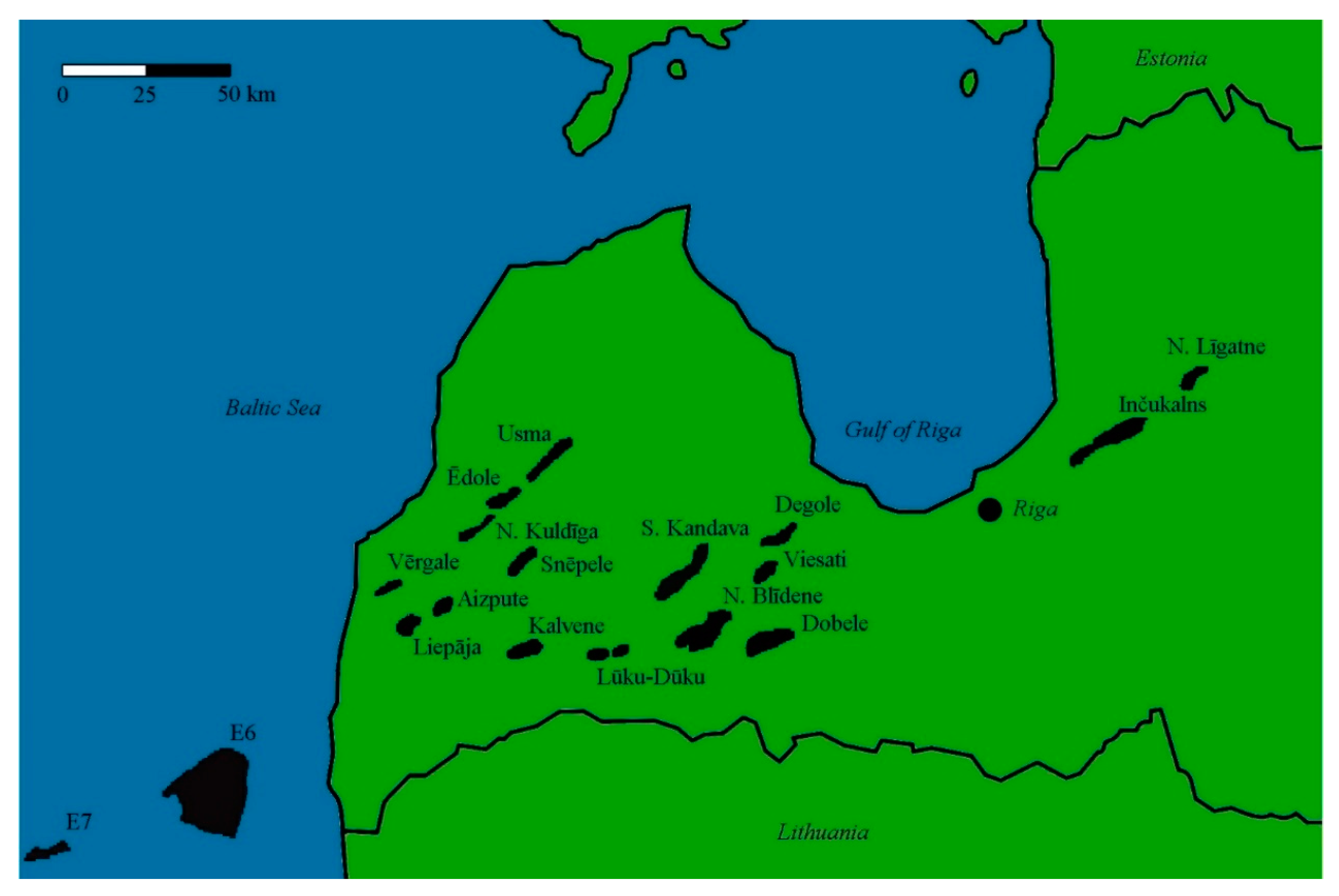
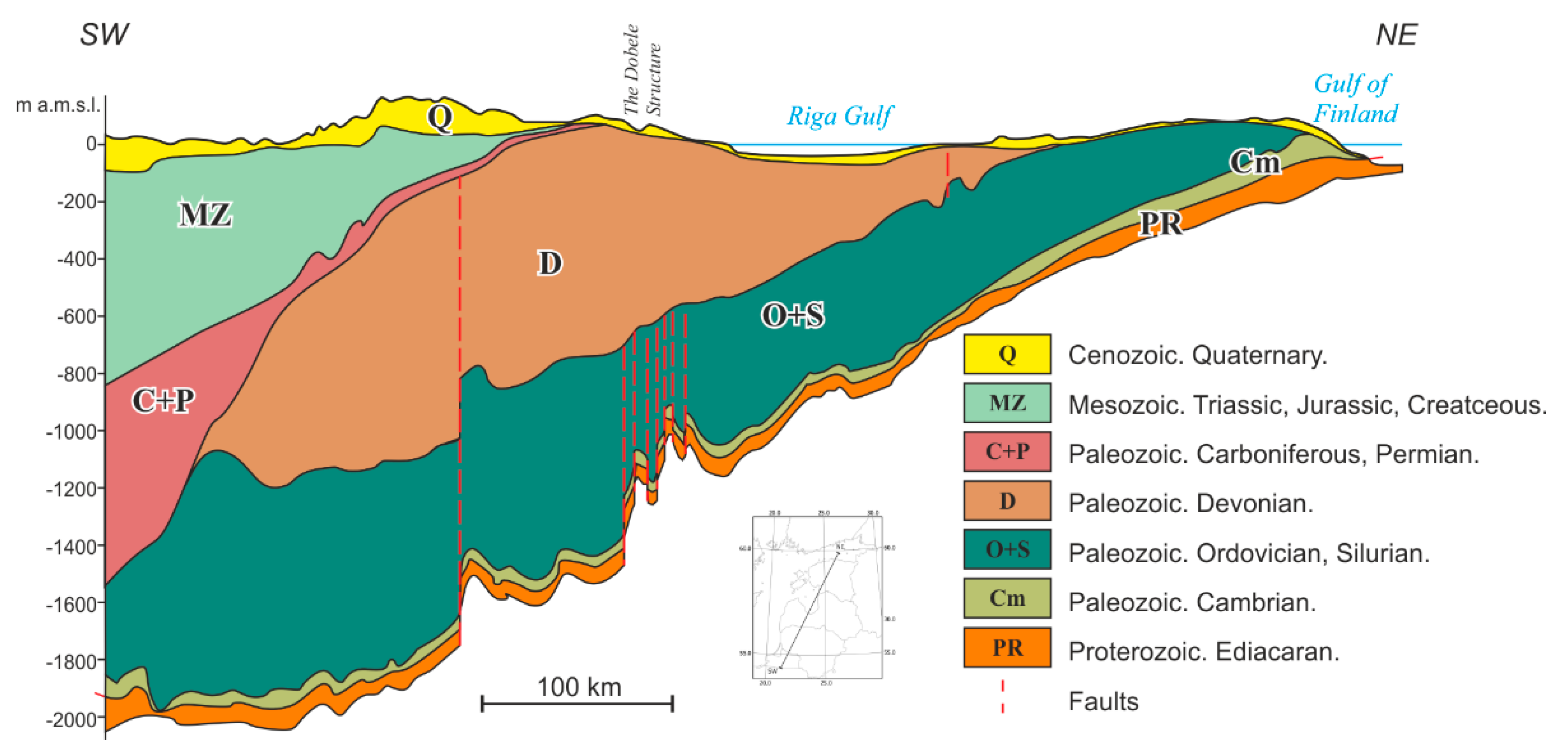
2.2. Location of the Study Subject: The Dobele Structure
2.3. Calculation of the Storage Capacity of the Structure
2.4. Additional Methods for Evaluation of a CO2 Storage Site
2.5. Factors Determining CO2 Storage Efficiency
2.6. Evaluation of the Criteria and Requirements for a Prospective CO2 Trap
3. Results and Discussion
3.1. Fossil CO2 Emission Sources in the Baltic States
3.2. Structures in the Baltic States with the Potential to Store Fossil CO2
3.3. Geological Feasibility of the Dobele Structure for Fossil CO2 Geological Storage
3.4. Justification of Fossil CO2 Geological Storage in Latvia
3.5. Construction of a System of CO2 Delivery to the Geological Storage Sites
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Trends in Atmospheric Carbon Dioxide. Global Monthly Mean CO2. Available online: https://www.esrl.noaa.gov/gmd/ccgg/trends/global.html (accessed on 18 May 2021).
- Friedlingstein, P.; O’Sullivan, M.; Jones, M.W.; Andrew, R.M.; Hauck, J.; Olsen, A.; Peters, G.P.; Peters, W.; Pongratz, J.; Sitch, S.; et al. Global Carbon Budget 2020. Earth Syst. Sci. Data 2020, 12, 3269–3340. [Google Scholar] [CrossRef]
- The Paris Agreement. Available online: https://treaties.un.org/doc/Publication/MTDSG/Volume%20II/Chapter%20XXVII/XXVII-7-d.en.pdf (accessed on 18 May 2021).
- Masson-Delmotte, V.; Zhai, P.; Pörtner, H.-O.; Roberts, D.; Skea, J.; Shukla, P.R.; Pirani, A.; Moufouma-Okia, W.; Péan, C.; Pidcock, R.; et al. Global Warming of 1.5°C. An IPCC Special Report on the Impacts of Global Warming of 1.5°C above Pre-Industrial Levels and Related Global Greenhouse Gas Emission Pathways, in the Context of Strengthening the Global Response to the Threat of Climate Change, Sustainable Development, and Efforts to Eradicate Poverty; Intergovernmental Panel on Climate Change: Geneva, Switzerland, 2019; p. 630. [Google Scholar]
- Fawzy, S.; Osman, A.I.; Doran, W.J.; Rooney, D.W. Strategies for mitigation of climate change: A review. Environ. Chem. Lett. 2020, 18, 2069–2094. [Google Scholar] [CrossRef]
- Baines, S.J.; Worden, R.H. Geological storage of carbon dioxide. Geol. Soc. Lond. Spéc. Publ. 2004, 233, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Z.; Huisingh, D. Carbon dioxide storage schemes: Technology, assessment and deployment. J. Clean. Prod. 2017, 142, 1055–1064. [Google Scholar] [CrossRef]
- USA. Department of Energy. Post-Combustion CO2 Capture. Available online: https://www.netl.doe.gov/coal/carbon-capture/post-combustion (accessed on 8 June 2021).
- USA. Department of Energy. Pre-Combustion Carbon Capture Research. Available online: https://www.energy.gov/fe/science-innovation/carbon-capture-and-storage-research/carbon-capture-rd/pre-combustion-carbon (accessed on 8 June 2018).
- Stanger, R.; Wall, T.; Spörl, R.; Paneru, M.; Grathwohl, S.; Weidmann, M.; Scheffknecht, G.; McDonald, D.; Myöhänen, K.; Ritvanen, J.; et al. Oxyfuel combustion for CO2 capture in power plants. Int. J. Greenh. Gas Control. 2015, 40, 55–125. [Google Scholar] [CrossRef]
- Manning, D.A.C.; Renforth, P. Passive sequestration of atmospheric CO2 through coupled plant-mineral reactions in urban soils. Environ. Sci. Technol. 2013, 47, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Jorat, M.E.; Goddard, M.A.; Manning, P.; Lau, H.K.; Ngeow, S.; Sohi, S.P.; Manning, D.A.C. Passive CO2 removal in urban soils: Evidence from brownfield sites. Sci. Total Environ. 2020, 703, 135573. [Google Scholar] [CrossRef]
- Washbourne, C.L.; Lopez-Capel, E.; Renforth, P.; Ascough, P.L.; Manning, D.A.C. Rapid removal of atmospheric CO2 by urban soils. Environ. Sci. Technol. 2015, 49, 5434–5440. [Google Scholar] [CrossRef]
- Jo, H.K.; McPherson, G.E. Carbon storage and flux in urban residential greenspace. J. Environ. Manag. 1995, 45, 109–133. [Google Scholar] [CrossRef] [Green Version]
- Orr, F.M. Storage of carbon dioxide in geological formations. J. Pet. Technol. 2004, 56, 90–97. [Google Scholar] [CrossRef]
- Lashgari, H.R.; Sun, A.; Zhang, T.; Pope, G.A.; Lake, L.W. Evaluation of carbon dioxide storage and miscible gas EOR in shale oil reservoirs. Fuel 2019, 241, 1223–1235. [Google Scholar] [CrossRef]
- Bachu, S. Sequestration of CO2 in geological media: Criteria and approach for site selection in response to climate change. Energy Convers. Manag. 2000, 41, 953–970. [Google Scholar] [CrossRef]
- Metz, B.; Davidson, O.; de Coninck, H.; Loos, M.; Meyer, L. Carbon Dioxide Capture and Storage; Cambridge University Press: Cambridge, UK, 2005; p. 481. [Google Scholar]
- Urych, T.; Smolinski, A. Numerical modelling of CO2 migration in saline aquifers of selected areas in the upper Silesian coal basin in Poland. Energies 2019, 12, 3093. [Google Scholar] [CrossRef] [Green Version]
- Koteras, A.; Checko, J.; Urych, T.; Madziarczyk, M.; Smolinski, A. An assessment of the formations and structures suitable for safe CO2 geological storage in the upper Silesian coal basin in Poland in the context of the regulation relating to the CSS. Energies 2020, 13, 195. [Google Scholar] [CrossRef] [Green Version]
- Pu, H.; Wang, Y.; Li, Y. How CO2 storage mechanisms are different in organic shale: Characterization and simulation studies. SPE J. 2017, 3, 661–671. [Google Scholar] [CrossRef]
- Goldberg, D.S.; Takahashi, T.; Slagle, A.L. Carbon dioxide sequestration in deep-sea basalt. Proc. Natl. Acad. Sci. USA 2018, 105, 9920–9925. [Google Scholar] [CrossRef] [Green Version]
- Talapatra, A. A study on the carbon dioxide injection into coal seam aiming at enhancing coal bed methane (ECBM) recovery. J. Petrol. Explor. Prod. Technol. 2020, 10, 1965–1981. [Google Scholar] [CrossRef] [Green Version]
- Raza, A.; Gholami, R.; Rezaee, R.; Bing, C.H.; Nagarajan, R.; Hamid, M.A. CO2 storage in depleted gas reservoirs: A study on the effect of residual gas saturation. Petroleum 2018, 4, 95–107. [Google Scholar] [CrossRef]
- Duncan, D.C.; Swanson, V.E. Organic-Rich Shale of the United States and World Land Areas; Geological Survey: Washington, DC, USA, 1965; p. 30. [Google Scholar]
- Tomic, L.; Karovic-Maricic, V.; Danilovic, D.; Crnogorac, M. Criteria for CO2 storage in geological formations. Und. Min. Eng. 2018, 32, 61–67. [Google Scholar] [CrossRef]
- National Academies of Sciences, Engineering, and Medicine. Negative Emissions Technologies and Reliable Sequestrations: A Research Agenda; The National Academies Press: Washington, DC, USA, 2019; p. 510. [Google Scholar] [CrossRef]
- Bickle, M.J. Geological carbon storage. Nat. Geosci. 2009, 2, 815–818. [Google Scholar] [CrossRef]
- Cai, B.; Li, Q.; Liu, G.; Liu, L.; Jin, T.; Shi, H. Environmental concern-based site screening of carbon dioxide geological storage in China. Sci. Rep. 2017, 7, 7598. [Google Scholar] [CrossRef]
- Sliaupa, S.; Shogenova, A.; Shogenov, K.; Sliaupiene, R.; Zabele, A.; Vaher, R. Industrial carbon dioxide emissions and potential geological sinks in the Baltic States. Oil Shale 2008, 25, 465–484. [Google Scholar] [CrossRef] [Green Version]
- O’Brien, P.N.S. Aspects of seismic reflection prospecting for oil and gas. Geophys. J. Int. 1983, 74, 97–127. [Google Scholar] [CrossRef] [Green Version]
- Greenhouse Gas Emissions by Source Sector. Available online: http://appsso.eurostat.ec.europa.eu/nui/show.do?lang=en&dataset=env_air_gge (accessed on 18 May 2021).
- European Green Deal. Available online: https://ec.europa.eu/info/strategy/priorities-2019-2024/european-green-deal_en (accessed on 18 May 2021).
- Directive 2009/31/EC. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32009L0031 (accessed on 18 May 2021).
- CO2CRC Annual Report 2019–2020. Available online: https://co2crc.com.au/resources/reports/ (accessed on 18 May 2021).
- Chadwick, A.; Arts, R.; Bernstone, C.; May, F.; Thibeau, S.; Zweigel, P. Best Practice for the Storage of CO2 in Saline Aquifers-Observations and Guidelines from the SACS and CO2STORE Projects; British Geological Survey: Nottingham, UK, 2009; p. 267. [Google Scholar]
- Latvia CO2 Emissions. Available online: https://www.worldometers.info/co2-emissions/latvia-co2-emissions/ (accessed on 18 May 2021).
- Informatīvais Ziņojums “Par Iespējām Sekmēt Zemes Dzīļu Derīgo Īpašību Izmantošanu Valsts Ekonomiskā Potenciāla Paaugstināšanai”. Available online: http://tap.mk.gov.lv/mk/tap/?pid=30233795 (accessed on 18 May 2021).
- Beckholmen, M.; Tiren, S.A. The Geological History of the Baltic Sea: A Review of the Literature and Investigation Tools; Geosigma AB: Uppsala, Sweden, 2008; p. 103. [Google Scholar]
- Geological Settings and Structural Elements of the Baltic States. Available online: https://www4.meteo.lv/vgd_oil_page/geological_setting.htm (accessed on 18 May 2021).
- Shogenova, A.; Sliaupa, S.; Vaher, R.; Shogenov, K.; Pomeranceva, R. The Baltic Basin: Structure, properties of reservoir rocks, and capacity for geological storage of CO2. Est. J. Earth Sci. 2009, 58, 259–267. [Google Scholar] [CrossRef]
- Mikelsons, K.; Davis, A.; Zebergs, V.; Zeltins, N. Latvian Underground Gas Storage Facilities for the Development of a Safe Gas and Power Supply System of North European Gas. In Proceedings of the IAEE International Conference on Energy and Security in the Changing World, Teheran, Iran, 25–27 May 2004. [Google Scholar]
- Potential Sites for CO2 Geological Storage in Latvia. Available online: https://www.meteo.lv/fs/CKFinderJava/userfiles/files/Geologija/Potential%20sites.pdf (accessed on 18 May 2021).
- Shogenov, K.; Shogenova, A.; Vizika-Kavvadias, O. Petrophysical properties and capacity of prospective structures for geological storage of CO2 onshore and offshore Baltic. Energy Proced. 2013, 37, 5036–5046. [Google Scholar] [CrossRef] [Green Version]
- Jansons, L.; Zeltins, N. Technical Studies on Development of the Dobele Underground Natural Gas Storage Facility. In Proceedings of the REHVA Annual Conference on Advanced HVAC and Natural Gas Technologies, Riga, Latvia, 6–9 May 2015. [Google Scholar]
- Tuuling, I. The Leba Ridge-Riga-Pskov fault zone—a major East European Craton interior dislocation zone and its role in the early Paleozoic development of the platform cover. Est. J. Earth Sci. 2019, 68, 161–189. [Google Scholar] [CrossRef]
- Structural Traps in Latvia. Available online: https://www4.meteo.lv/vgd_oil_page/traps1.htm (accessed on 18 May 2021).
- Geological Survey of Denmark and Greenland. EU GeoCapacity: Assessing European Capacity for Geological Storage of Carbon Dioxide. Final Report; EU GeoCapacity Consortium: Copenhagen, Denmark, 2009; p. 66. [Google Scholar]
- Yang, X.; Buscheck, T.A.; Mansoor, K.; Wang, Z.; Carrol, S.A. Effectiveness of Geophysical Monitoring Methods in Detecting Brine and CO2 Leakage in Underground Sources of Drinking Water; Lawrence Livermore National Laboratory: Livermore, CA, USA, 2018; p. 30. [Google Scholar] [CrossRef]
- Oyedotun, T.D.T. X-ray fluorescence (XRF) in the investigation of the composition of earth materials: A review and an overview. Geol. Ecol. Land. 2018, 2, 148–154. [Google Scholar] [CrossRef]
- Llamas, B.; Arribas, M.; Hernandez, E.; Mazadiego, L.F. Pre-injection phase: Site selection and characterization. In CO2 Sequestration and Valorization; Morgado, C.R.V., Ed.; IntechOpen: Rijeka, Croatia, 2014; pp. 281–303. [Google Scholar]
- Shogenova, A.; Sliaupa, S.; Shogenov, K.; Sliaupiene, R.; Pomeranceva, R.; Vaher, R.; Uibu, M.; Kuusik, R. Possibilities for geological storage and mineral trapping of industrial CO2 emissions in the Baltic region. Energy Proc. 2009, 1, 2753–2760. [Google Scholar] [CrossRef] [Green Version]
- Global Emissions by Economic Sector. Available online: https://www.epa.gov/ghgemissions/global-greenhouse-gas-emissions-data (accessed on 18 May 2021).
- Global CCS Institute. Carbon Dioxide (CO2) Distribution Infrastructure. The Opportunities and Challenges Confronting CO2 Transport for Purposes of Carbon Capture and Storage (CCS) an Observation Paper; Global CCS Institute: Canberra, Australia, 2012; p. 43. [Google Scholar]
- OECD. Strategies to Reduce Greenhouse Gas Emissions from Road Transport: Analytical Methods; OECD Publishing: Paris, France, 2002; p. 67. [Google Scholar]
- Lithuania CO2 Emissions. Available online: https://www.worldometers.info/co2-emissions/lithuania-co2-emissions/ (accessed on 18 May 2019).
- Estonia CO2 Emissions. Available online: https://www.worldometers.info/co2-emissions/estonia-co2-emissions/ (accessed on 18 May 2021).
- EEA Greenhouse Gas-Data Viewer. Available online: https://www.eea.europa.eu/data-and-maps/data/data-viewers/greenhouse-gases-viewer (accessed on 18 May 2021).
- ACEA. CO2-Based Motor Vehicle Taxes in the European Union; ACEA Publishing: Brussels, Belgium, 2020; p. 5. [Google Scholar]
- Shogenova, A.; Shogenov, K.; Vaher, R.; Ivask, J.; Sliaupa, S.; Vangkilde-Pedersen, T.; Uibu, M.; Kuusik, R. CO2 geological storage capacity analysis in Estonia and neighbouring regions. Energy Proc. 2011, 4, 2785–2793. [Google Scholar] [CrossRef] [Green Version]
- Estonian Government. Estonian National Energy and Climate Plan (NECP 2030); Estonian Government: Tallinn, Estonia, 2019; p. 195. [Google Scholar]
- Paškevičius, J. The Geology of the Baltic Republics; University and Geological Survey of Lithuania: Vilnius, Lithuania, 1997; p. 387. [Google Scholar]
- Delina, A.; Kalvans, A.; Saks, T.; Bethers, U.; Vircavs, V. Highlights of Groundwater Research in the Baltic Artesian Basin; University of Latvia: Riga, Latvia, 2012; p. 15. [Google Scholar]
- Miller, R.D.; Saenz, V.; Huggins, R.J. Feasibility of CDP seismic reflection to image structures in a 220-m deep, 2-m thick coal zone near Palau, Coahila, Mexico. Geophysics 1992, 57, 1373–1380. [Google Scholar] [CrossRef]
- Raidla, V.; Kirsimae, K.; Bityukova, L.; Joeleht, A.; Shogenova, A.; Sliaupa, S. Lithology and diagenesis of the poorly consolidated Cambrian siliciclastic sediments in the northern Baltic sedimentary basin. Geol. Q. 2006, 50, 395–406. [Google Scholar]
- Gorecki, W.; Lapinskas, P.; Lashkova, L.; Lashkov, E.; Reicher, B.; Sakalauskas, K.; Strzetelski, W. Petroleum perspectives of the Baltic Syneclise. Pol. J. Miner. Resourc. 1992, 1, 65–88. [Google Scholar]
- Brangulis, A.; Kaņevs, S. Latvijas Tektonika; Rīgas Valsts Ģeoloģijas Dienests: Riga, Latvia, 2002; p. 710. [Google Scholar]
- Sliaupa, S.; Hoth, P.; Shogenova, A.; Huenges, E.; Rasteniene, V.; Freimanis, A.; Bityukova, L.; Joeleht, A.; Kirsimae, K.; Laskova, L.; et al. Characterization of Cambrian reservoir rocks and their fluids in the Baltic States (CAMBALTICA). In Cleaner Energy Systems through Utilization of Renewable Geothermal Energy Resources; Bujakowski, W., Ed.; Kajc: Krakow, Poland, 2003; pp. 61–73. [Google Scholar]
- Bergman, B.; Juhojuntti, N.G. Carbon Storage in Swedish Bedrock—Current Status Regarding Potential Storage Areas and Geophysical Information. In Proceedings of the Fall Meeting of the American Geophysical Union, San Francisco, CA, USA, 15 September 2010. [Google Scholar]
- Zemite, L.; Kutjuns, A.; Bode, I.; Kunickis, M.; Zeltins, N. Risk treatment and system recovery analysis of gas system of gas and electricity network of Latvia. Lat. J. Phys. Tech. Sci. 2018, 55, 3–15. [Google Scholar] [CrossRef] [Green Version]
- Jorgensen, P.; Ovlisen, F.R.; Catalini, E.; Petersen, N.; Muro-sune, N.; Littmann, W.; Hehmann, U. Inčukalns Underground Gas Storage—Study of Increased Flexibility and Use as Strategic Gas Storage. Summary; Ramboll: Copenhagen, Denmark, 2017; p. 6. [Google Scholar]
- The Outlook of Latvian Potential Underground Gas Storages and Prospects of Utilization of this Potential for Securing a Reliable Gas Supply to Europe. Available online: https://unece.org/fileadmin/DAM/ie/se/pdfs/wpgas/session/17_countr/latvia.pdf (accessed on 18 May 2021).
- Davis, A.; Jesinska, A.; Kreslins, A.; Zebergs, V.; Zeltins, N. Increasing Role of Underground Gas Storages for Reliable Supply of Gas to Latvia, Lithuania, Estonia, Finland and NW Russia and Prospects of Development of Inčukalns Underground Gas Storage. In Proceedings of the 23rd World Gas Conference, Amsterdam, The Netherlands, 5–9 June 2006. [Google Scholar]
- Regulations for Use of the Section of Subsoil of National Significance the “Dobele Structure”. Cabinet Regulation No. 524. Available online: http://extwprlegs1.fao.org/docs/pdf/lat186898ENG.pdf (accessed on 18 May 2021).
- Levins, I. Hidrodinamiskā Izpēte Valsts Nozīmes Zemes Nogabalā “Dobeles Struktūra”; PS ‘Dobeles Struktūra’: Riga, Latvia, 2010. [Google Scholar]
- Raidla, V.; Kirsimae, K.; Vaikmae, R.; Joeleht, A.; Karro, E.; Marandi, A.; Savitskaja, L. Geochemical evolution of groundwater in the Cambrian-Vendian aquifer system of the Baltic basin. Chem. Geol. 2009, 258, 219–231. [Google Scholar] [CrossRef]
- Shogenov, K.; Shogenova, A.; Vizika-Kavvadias, O.; Nauroy, J.F. Experimental modelling of CO2-fluid-rock interaction: The evolution of the composition and properties of host rocks in the Baltic region. Earth Space Sci. 2015, 2, 262–284. [Google Scholar] [CrossRef]
- Gunter, W.D.; Perkins, E.H.; Hutcheon, I. Aquifer disposal of acid gases: Modelling of water-rock reactions for trapping of acid wastes. Appl. Geochem. 2000, 15, 1086–1095. [Google Scholar] [CrossRef]
- Aszuba, J.P.; Janecky, D.R. Geochemical impacts of sequestering carbon dioxide in brine formations. In Carbon Sequestration and Its Role in the Global Carbon Cycle; McPherson, B.J., Sundquist, E.T., Eds.; AGU: Washington, DC, USA, 2009; Volume 183, pp. 239–247. [Google Scholar]
- OECD. Effective Carbon Rates on Energy; OECD Publishing: Paris, France, 2016; p. 16. [Google Scholar]
- Cost and Economic Potential of the CCS System. Available online: https://www.ipcc.ch/site/assets/uploads/2018/03/srccs_chapter8-1.pdf (accessed on 18 May 2021).
- Daily EU ETS Carbon Market Price (Euros). Available online: https://ember-climate.org/data/carbon-price-viewer/ (accessed on 18 May 2021).
- Schmelz, W.J.; Hochman, G.; Miller, K.G. Total cost of carbon capture and storage implemented at a regional scale: North-eastern and midwestern United States. Interface Focus 2020, 10, 10. [Google Scholar] [CrossRef] [PubMed]
- The International Energy Agency Greenhouse Gas R&D Programme. The Costs of CO2 Storage. Post-Demonstration CCS in the EU; European Technology Platform for Zero Emission Fossil Fuel Power Plants: Brussels, Belgium; p. 42. Available online: https://www.globalccsinstitute.com/archive/hub/publications/119816/costs-co2-storage-post-demonstration-ccs-eu.pdf (accessed on 18 May 2019).
- Gusca, J.; Blumberga, D. Simplified dynamic life cycle assessment model of CO2 compression, transportation and injection phase within carbon capture and storage. Energy Proc. 2011, 4, 2526–2532. [Google Scholar] [CrossRef] [Green Version]
- The International Energy Agency Greenhouse Gas R&D Programme. The Costs of CO2 Transport. Post-Demonstration CCS in the EU; European Technology Platform for Zero Emission fossil Fuel Power Plants: Brussels, Belgium; p. 53. Available online: https://www.globalccsinstitute.com/archive/hub/publications/119811/costs-co2-transport-post-demonstration-ccs-eu.pdf (accessed on 18 May 2021).
- Gusca, J. Research on Development of Latvian Energy Sector. Impact Assessment of Carbon Dioxide Capture and Storage Processes. Ph.D. Thesis, University of Latvia, Riga, Latvia, 2011. [Google Scholar]

| Source Sector | Estonia | Latvia | Lithuania | Total |
|---|---|---|---|---|
| Public electricity and heat generation | 12.17 | 1.84 | 1.02 | 15.03 |
| Transport | 2.38 | 3.30 | 6.00 | 11.68 |
| Chemical industry | - | - | 1.83 | 1.83 |
| Petroleum refining | - | - | 1.31 | 1.31 |
| Manufacture of solid fuels | 1.56 | 0.05 | 0.05 | 1.66 |
| Other fuel combustion sectors | 0.46 | 1.28 | 1.34 | 3.08 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krūmiņš, J.; Kļaviņš, M.; Dēliņa, A.; Damkevics, R.; Segliņš, V. Potential of the Middle Cambrian Aquifer for Carbon Dioxide Storage in the Baltic States. Energies 2021, 14, 3681. https://doi.org/10.3390/en14123681
Krūmiņš J, Kļaviņš M, Dēliņa A, Damkevics R, Segliņš V. Potential of the Middle Cambrian Aquifer for Carbon Dioxide Storage in the Baltic States. Energies. 2021; 14(12):3681. https://doi.org/10.3390/en14123681
Chicago/Turabian StyleKrūmiņš, Jānis, Māris Kļaviņš, Aija Dēliņa, Raivo Damkevics, and Valdis Segliņš. 2021. "Potential of the Middle Cambrian Aquifer for Carbon Dioxide Storage in the Baltic States" Energies 14, no. 12: 3681. https://doi.org/10.3390/en14123681
APA StyleKrūmiņš, J., Kļaviņš, M., Dēliņa, A., Damkevics, R., & Segliņš, V. (2021). Potential of the Middle Cambrian Aquifer for Carbon Dioxide Storage in the Baltic States. Energies, 14(12), 3681. https://doi.org/10.3390/en14123681







