Recent Advances on Materials for Lithium-Ion Batteries
Abstract
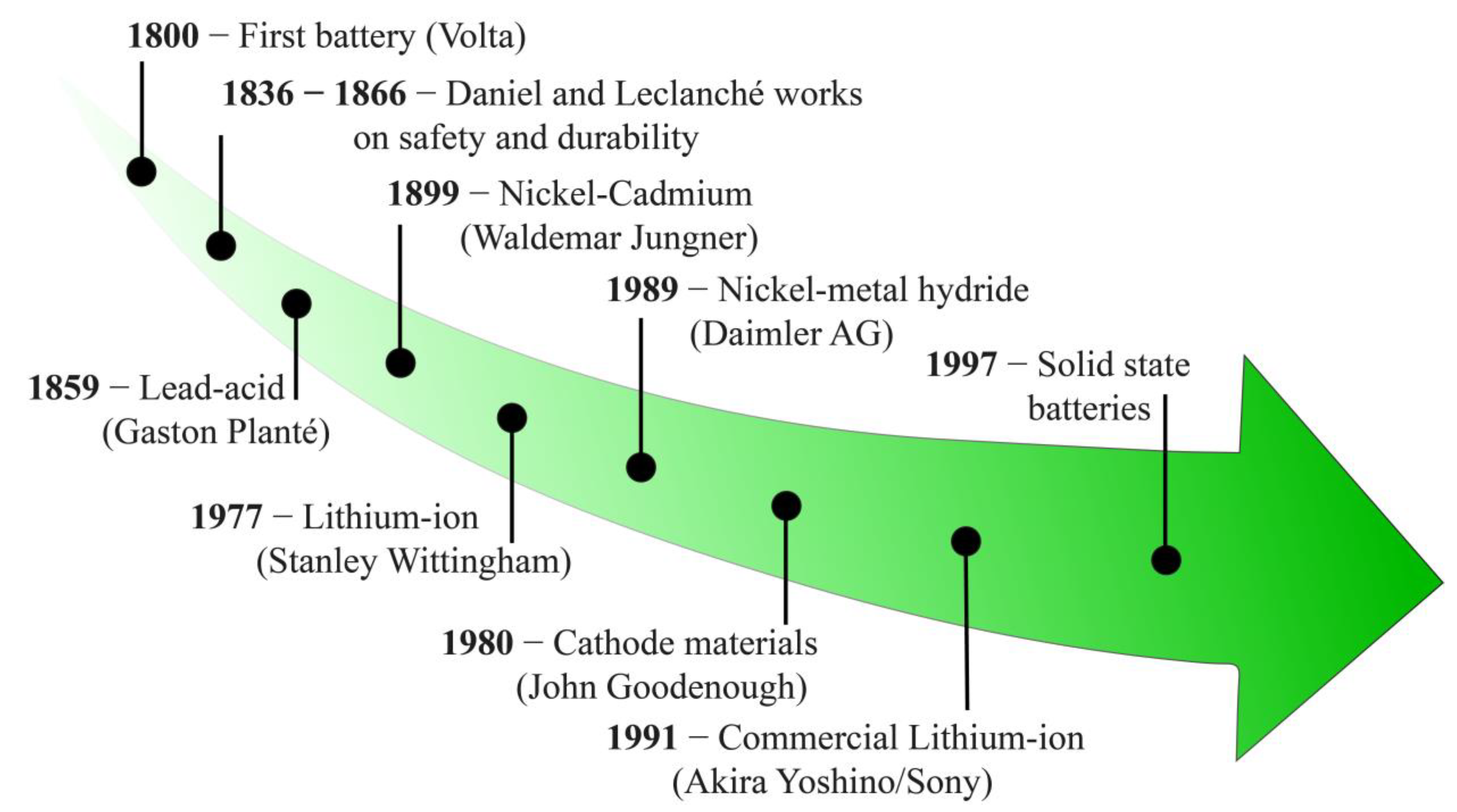
1. Introduction
2. Lithium-Ion Batteries
3. Materials for Electrodes
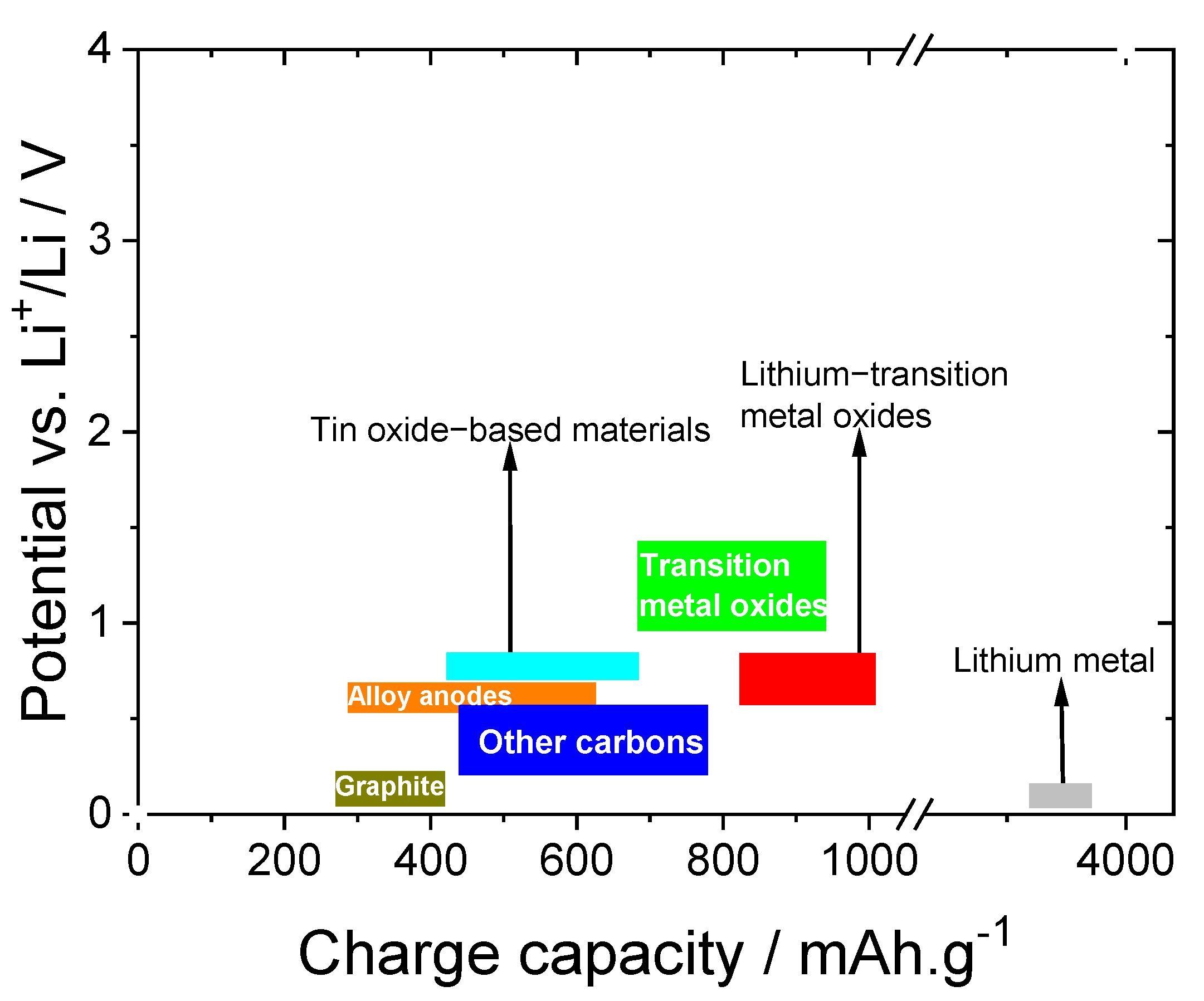
3.1. Active Anode Materials
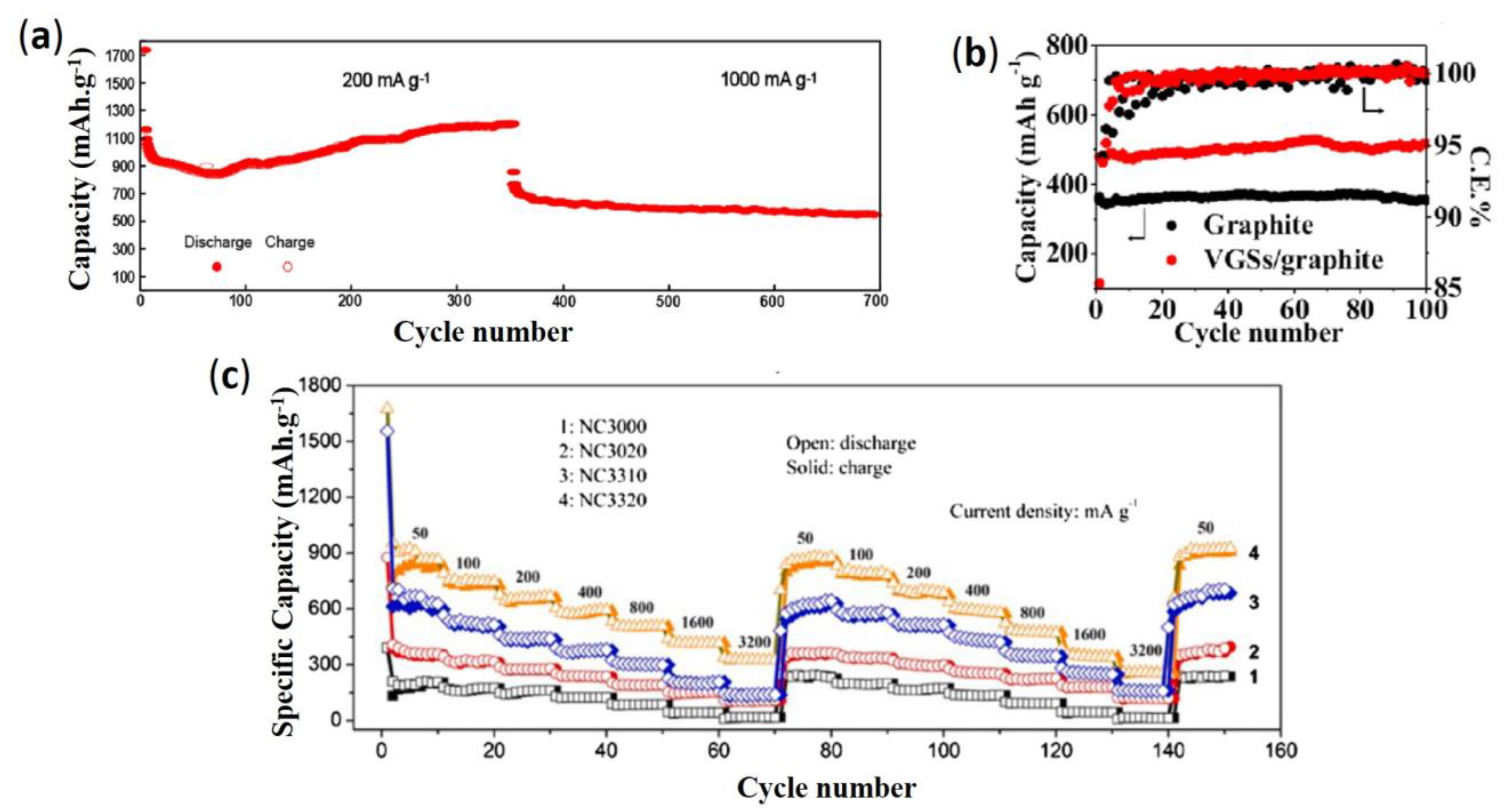
3.1.1. Carbon and Metal Alloys-Based Anode Materials
3.1.2. Silicon-Based Anode Materials
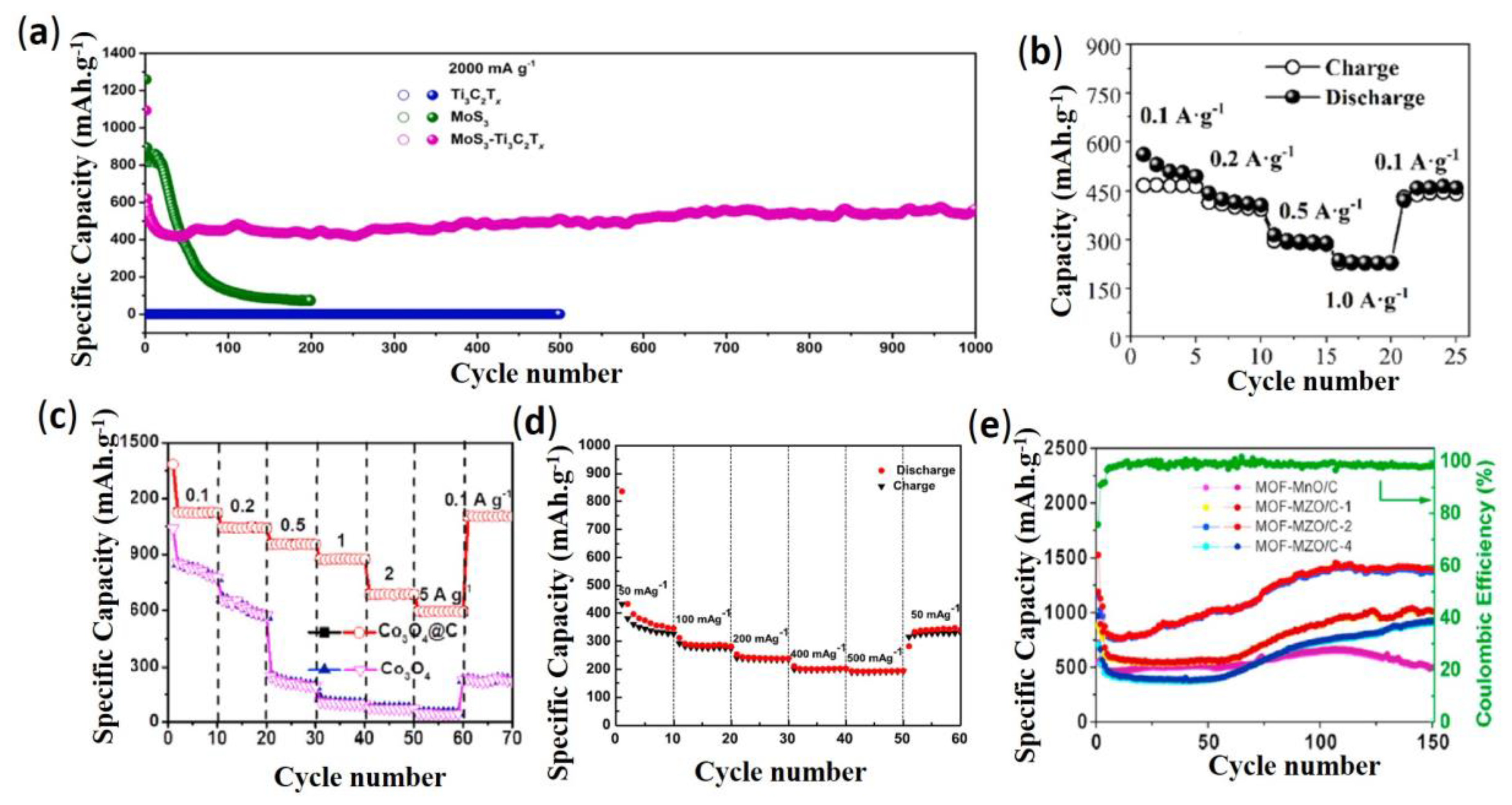
3.1.3. Conversion-Type Transition-Metals and Their Composites-Based Anode Active Materials
3.2. Active Cathode Materials
4. Materials for Separators/Electrolytes
4.1. Separator Membrane
- Low thickness (<25 µm) and good permeability;
- Porosity > 50% and pore size below <1 micron;
- Excellent wettability: absorption and retention of electrolytes;
- Chemical and dimensional stability;
- Good thermal stability and excellent mechanical properties.
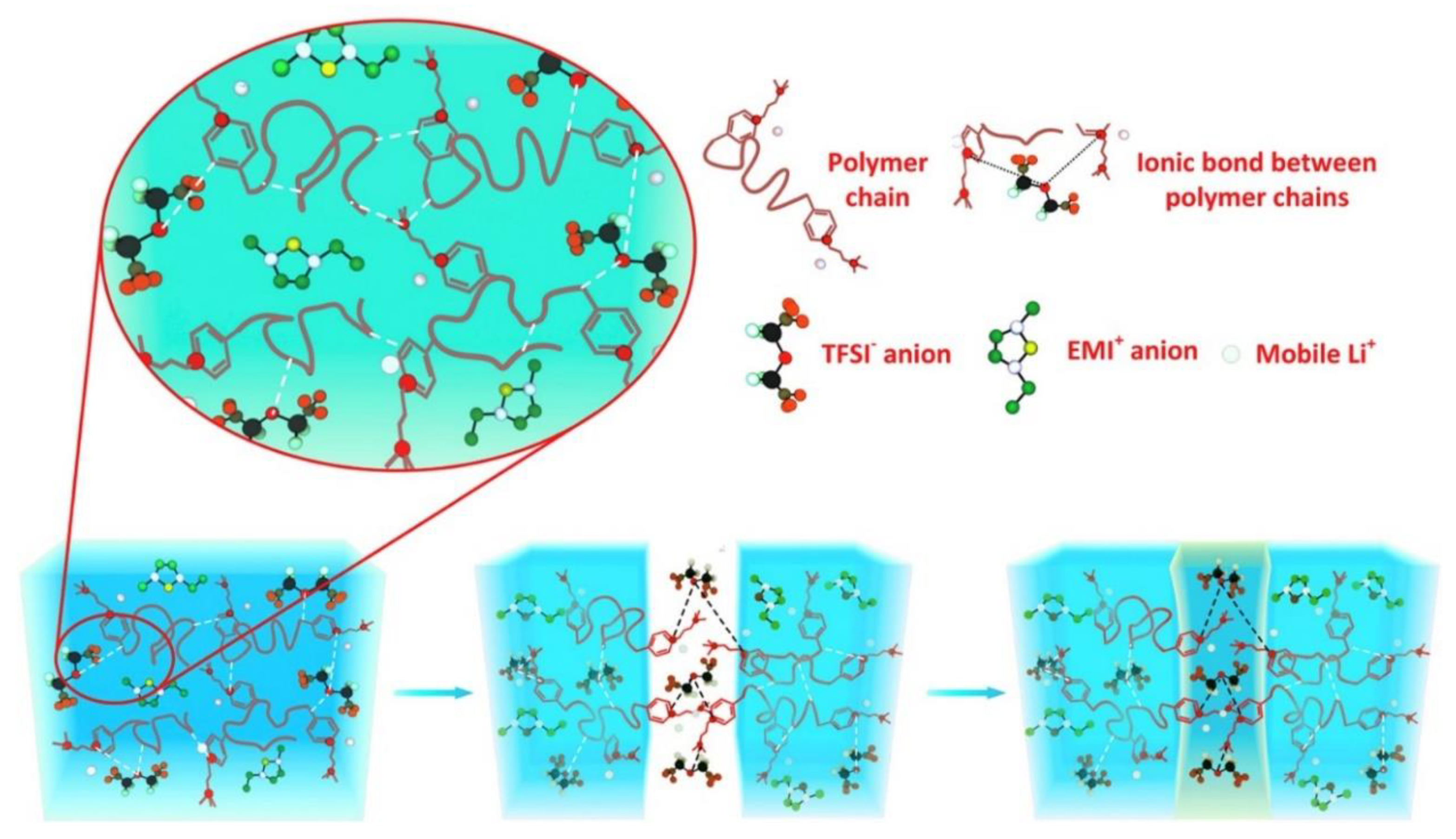
4.2. Solid Polymer Electrolytes
5. Main Conclusions and Future Trends
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Xu, M.; David, J.M.; Kim, S.H. The fourth industrial revolution: Opportunities and challenges. Int. J. Financ. Res. 2018, 9, 90–95. [Google Scholar] [CrossRef]
- Abdelkareem, M.A.; Elsaid, K.; Wilberforce, T.; Kamil, M.; Sayed, E.T.; Olabi, A. Environmental aspects of fuel cells: A review. Sci. Total Environ. 2021, 752, 141803. [Google Scholar] [CrossRef]
- Kaltschmitt, M.; Streicher, W.; Wiese, A. Renewable Energy: Technology, Economics and Environment; Springer Science & Business Media: New York, NY, USA, 2007. [Google Scholar]
- Scrosati, B.; Hassoun, J.; Sun, Y.-K. Lithium-ion batteries. A look into the future. Energy Environ. Sci. 2011, 4, 3287–3295. [Google Scholar] [CrossRef]
- Volta, A. On the electricity excited by the mere contact of conducting substances of different kinds. In a letter from Mr. Alexander Volta, F.R.S. Professor of Natural Philosophy in the University of Pavia, to the Rt. Hon. Sir Joseph Banks, Bart. K.B. P. R. S. Philos. Trans. R. Soc. Lond. 1800, 90, 403–431. [Google Scholar]
- Abraham, K.M. Prospects and Limits of Energy Storage in Batteries. J. Phys. Chem. Lett. 2015, 6, 830–844. [Google Scholar] [CrossRef]
- Broussely, M.; Pistoia, G. Industrial Applications of Batteries: From Cars to Aerospace and Energy Storage; Elsevier: Amsterdam, The Netherlands, 2007. [Google Scholar]
- Diouf, B.; Pode, R. Potential of lithium-ion batteries in renewable energy. Renew. Energy 2015, 76, 375–380. [Google Scholar] [CrossRef]
- Väyrynen, A.; Salminen, J. Lithium ion battery production. J. Chem. Thermodyn. 2012, 46, 80–85. [Google Scholar] [CrossRef]
- Laing, D.; Bahl, C.; Bauer, T.; Fiss, M.; Breidenbach, N.; Hempel, M. High-Temperature Solid-Media Thermal Energy Storage for Solar Thermal Power Plants. Proc. IEEE 2012, 100, 516–524. [Google Scholar] [CrossRef]
- Jiang, F.; Peng, P. Elucidating the Performance Limitations of Lithium-ion Batteries due to Species and Charge Transport through Five Characteristic Parameters. Sci. Rep. 2016, 6, 32639. [Google Scholar] [CrossRef] [PubMed]
- Whittingham, M.S. Electrical Energy Storage and Intercalation Chemistry. Science 1976, 192, 1126. [Google Scholar] [CrossRef] [PubMed]
- Mizushima, K.; Jones, P.C.; Wiseman, P.J.; Goodenough, J.B. LixCoO2 (0 < x < −1): A new cathode material for batteries of high energy density. Mater. Res. Bull. 1980, 15, 783–789. [Google Scholar]
- Thackeray, M.M.; David, W.I.F.; Bruce, P.G.; Goodenough, J.B. Lithium insertion into manganese spinels. Mater. Res. Bull. 1983, 18, 461–472. [Google Scholar] [CrossRef]
- Padhi, A.; Nanjundaswamy, K.S.; Goodenough, J. Phospho-Olivines as Positive-Electrode Materials for Rechargeable Lithium Batteries. J. Electrochem. Soc. 1997, 144, 1188–1194. [Google Scholar] [CrossRef]
- Nishi, Y. The development of lithium ion secondary batteries. Chem. Rec. 2001, 1, 406–413. [Google Scholar] [CrossRef] [PubMed]
- Takada, K. Progress and prospective of solid-state lithium batteries. Acta Mater. 2013, 61, 759–770. [Google Scholar] [CrossRef]
- Liu, C.; Neale, Z.G.; Cao, G. Understanding electrochemical potentials of cathode materials in rechargeable batteries. Mater. Today 2016, 19, 109–123. [Google Scholar] [CrossRef]
- Winter, M.; Besenhard, J.O.; Spahr, M.E.; Novák, P. Insertion Electrode Materials for Rechargeable Lithium Batteries. Adv. Mater. 1998, 10, 725–763. [Google Scholar] [CrossRef]
- Deng, W.; Zhu, W.; Zhou, X.; Liu, Z. Graphene nested porous carbon current collector for lithium metal anode with ultrahigh areal capacity. Energy Storage Mater. 2018, 15, 266–273. [Google Scholar] [CrossRef]
- Miranda, D.; Gören, A.; Costa, C.M.; Silva, M.M.; Almeida, A.M.; Lanceros-Méndez, S. Theoretical simulation of the optimal relation between active material, binder and conductive additive for lithium-ion battery cathodes. Energy 2019, 172, 68–78. [Google Scholar] [CrossRef]
- Lisbona, D.; Snee, T. A review of hazards associated with primary lithium and lithium-ion batteries. Process Saf. Environ. Prot. 2011, 89, 434–442. [Google Scholar] [CrossRef]
- Zhao, Q.; Stalin, S.; Zhao, C.-Z.; Archer, L.A. Designing solid-state electrolytes for safe, energy-dense batteries. Nat. Rev. Mater. 2020, 5, 229–252. [Google Scholar] [CrossRef]
- Zhang, W.-J. Structure and performance of LiFePO4 cathode materials: A review. J. Power Sources 2011, 196, 2962–2970. [Google Scholar] [CrossRef]
- Lyu, Y.; Wu, X.; Wang, K.; Feng, Z.; Cheng, T.; Liu, Y.; Wang, M.; Chen, R.; Xu, L.; Zhou, J. An Overview on the Advances of LiCoO2 Cathodes for Lithium-Ion Batteries. Adv. Energy Mater. 2020, 11, 2000982. [Google Scholar] [CrossRef]
- Tong, W.; Chu, Q.; Meng, Y.; Wang, X.; Bin, Y.; Gao, J.; Zhao, X.; Liu, X. Synthesis of mesoporous orthorhombic LiMnO2 cathode materials via a one-step flux method for high performance lithium-ion batteries. Mater. Res. Express 2018, 5, 065511. [Google Scholar] [CrossRef]
- Chikkannanavar, S.B.; Bernardi, D.M.; Liu, L. A review of blended cathode materials for use in Li-ion batteries. J. Power Sources 2014, 248, 91–100. [Google Scholar] [CrossRef]
- Kamali, A.R.; Fray, D.J. Review on carbon and silicon based materials as anode materials for lithium ion batteries. J. New Mater. Electrochem. Syst 2010, 13, 147–160. [Google Scholar]
- Zhou, D.; Zhang, M.; Sun, F.; Arlt, T.; Frerichs, J.E.; Dong, K.; Wang, J.; Hilger, A.; Wilde, F.; Kolek, M.; et al. Performance and behavior of LLZO-based composite polymer electrolyte for lithium metal electrode with high capacity utilization. Nano Energy 2020, 77, 105196. [Google Scholar] [CrossRef]
- Xie, W.; Wu, L.; Liu, W.; Dang, Y.; Tang, A.; Luo, Y. Modelling electrolyte-immersed tensile property of polypropylene separator for lithium-ion battery. Mech. Mater. 2021, 152, 103667. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhou, M.; Yu, J.; Cai, J.; Yang, Z. Poly (vinylidene fluoride) Modified Commercial Paper as a Separator with Enhanced Thermal Stability and Electrolyte Affinity for Lithium-ion Battery. Energy Environ. Mater. 2020, 1–7. [Google Scholar] [CrossRef]
- Barbosa, J.C.; Dias, J.P.; Lanceros-Mendez, S.; Costa, C.M. Recent Advances in Poly(vinylidene fluoride) and Its Copolymers for Lithium-Ion Battery Separators. Membranes 2018, 8, 45. [Google Scholar] [CrossRef]
- Li, Q.; Chen, J.; Fan, L.; Kong, X.; Lu, Y. Progress in electrolytes for rechargeable Li-based batteries and beyond. Green Energy Environ. 2016, 1, 18–42. [Google Scholar] [CrossRef]
- Okumura, T.; Taminato, S.; Miyazaki, Y.; Kitamura, M.; Saito, T.; Takeuchi, T.; Kobayashi, H. LISICON-Based Amorphous Oxide for Bulk-Type All-Solid-State Lithium-Ion Battery. ACS Appl. Energy Mater. 2020, 3, 3220–3229. [Google Scholar] [CrossRef]
- DeWees, R.; Wang, H. Synthesis and Properties of NaSICON-type LATP and LAGP Solid Electrolytes. ChemSusChem 2019, 12, 3713–3725. [Google Scholar] [CrossRef]
- Li, Y.; Xu, H.; Chien, P.-H.; Wu, N.; Xin, S.; Xue, L.; Park, K.; Hu, Y.-Y.; Goodenough, J.B. A Perovskite Electrolyte That Is Stable in Moist Air for Lithium-Ion Batteries. Angew. Chem. Int. Ed. 2018, 57, 8587–8591. [Google Scholar] [CrossRef]
- Mindemark, J.; Lacey, M.J.; Bowden, T.; Brandell, D. Beyond PEO—Alternative host materials for Li+-conducting solidpolymer electrolytes. Prog. Polym. Sci. 2018, 81, 114–143. [Google Scholar] [CrossRef]
- Wu, F.; Zhang, K.; Liu, Y.; Gao, H.; Bai, Y.; Wang, X.; Wu, C. Polymer electrolytes and interfaces toward solid-state batteries: Recent advances and prospects. Energy Storage Mater. 2020, 33, 26–54. [Google Scholar] [CrossRef]
- Christensen, J.; Albertus, P.; Sanchez-Carrera, R.S.; Lohmann, T.; Kozinsky, B.; Liedtke, R.; Ahmed, J.; Kojic, A. A Critical Review of Li/Air Batteries. J. Electrochem. Soc. 2011, 159, R1–R30. [Google Scholar] [CrossRef]
- Li, T.; Bai, X.; Gulzar, U.; Bai, Y.-J.; Capiglia, C.; Deng, W.; Zhou, X.; Liu, Z.; Feng, Z.; Proietti Zaccaria, R. A Comprehensive Understanding of Lithium–Sulfur Battery Technology. Adv. Funct. Mater. 2019, 29, 1901730. [Google Scholar] [CrossRef]
- Nunes-Pereira, J.; Costa, C.M.; Lanceros-Méndez, S. Polymer composites and blends for battery separators: State of the art, challenges and future trends. J. Power Sources 2015, 281, 378–398. [Google Scholar] [CrossRef]
- Imanishi, N.; Yamamoto, O. Rechargeable lithium–air batteries: Characteristics and prospects. Mater. Today 2014, 17, 24–30. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, Y.; Guo, Z. Approaching high-performance potassium-ion batteries via advanced design strategies and engineering. Sci. Adv. 2019, 5, eaav7412. [Google Scholar] [CrossRef]
- Canepa, P.; Sai Gautam, G.; Hannah, D.C.; Malik, R.; Liu, M.; Gallagher, K.G.; Persson, K.A.; Ceder, G. Odyssey of Multivalent Cathode Materials: Open Questions and Future Challenges. Chem. Rev. 2017, 117, 4287–4341. [Google Scholar] [CrossRef]
- Abraham, K.M. How Comparable Are Sodium-Ion Batteries to Lithium-Ion Counterparts? ACS Energy Lett. 2020, 5, 3544–3547. [Google Scholar] [CrossRef]
- Costa, C.M.; Barbosa, J.C.; Gonçalves, R.; Castro, H.; Campo, F.J.D.; Lanceros-Méndez, S. Recycling and environmental issues of lithium-ion batteries: Advances, challenges and opportunities. Energy Storage Mater. 2021, 37, 433–465. [Google Scholar] [CrossRef]
- Slater, M.D.; Kim, D.; Lee, E.; Johnson, C.S. Sodium-Ion Batteries. Adv. Funct. Mater. 2013, 23, 947–958. [Google Scholar] [CrossRef]
- Hosaka, T.; Kubota, K.; Hameed, A.S.; Komaba, S. Research Development on K-Ion Batteries. Chem. Rev. 2020, 120, 6358–6466. [Google Scholar] [CrossRef] [PubMed]
- Liang, M.; Liu, Y.; Xiao, B.; Yang, S.; Wang, Z.; Han, H. An analytical model for the transverse permeability of gas diffusion layer with electrical double layer effects in proton exchange membrane fuel cells. Int. J. Hydrogen Energy 2018, 43, 17880–17888. [Google Scholar] [CrossRef]
- Joos, J.; Buchele, A.; Schmidt, A.; Weber, A.; Ivers-Tiffée, E. Virtual Electrode Design for Lithium-Ion Battery Cathodes. Energy Technol. 2021, 2000891. [Google Scholar] [CrossRef]
- Gören, A.; Costa, C.M.; Silva, M.M.; Lanceros-Méndez, S. State of the art and open questions on cathode preparation based on carbon coated lithium iron phosphate. Compos. Part B Eng. 2015, 83, 333–345. [Google Scholar] [CrossRef]
- Zhou, W.; Hao, F.; Fang, D. The effects of elastic stiffening on the evolution of the stress field within a spherical electrode particle of lithium-ion batteries. Int. J. Appl. Mech. 2013, 5, 1350040. [Google Scholar] [CrossRef]
- Christensen, J.; Newman, J. A Mathematical Model of Stress Generation and Fracture in Lithium Manganese Oxide. J. Electrochem. Soc. 2006, 153, A1019. [Google Scholar] [CrossRef]
- Zhou, W. Effects of external mechanical loading on stress generation during lithiation in Li-ion battery electrodes. Electrochim. Acta 2015, 185, 28–33. [Google Scholar] [CrossRef]
- Zhu, Z.; He, Y.; Hu, H.; Zhang, F. Evolution of Internal Stress in Heterogeneous Electrode Composite during the Drying Process. Energies 2021, 14, 1683. [Google Scholar] [CrossRef]
- Lu, X.; Li, H.; Chen, N. Analysis of the Properties of Fractional Heat Conduction in Porous Electrodes of Lithium-Ion Batteries. Entropy 2021, 23, 195. [Google Scholar] [CrossRef]
- Nguyen, T.-T.; Demortière, A.; Fleutot, B.; Delobel, B.; Delacourt, C.; Cooper, S.J. The electrode tortuosity factor: Why the conventional tortuosity factor is not well suited for quantifying transport in porous Li-ion battery electrodes and what to use instead. NPJ Comput. Mater. 2020, 6, 123. [Google Scholar] [CrossRef]
- Roy, P.; Srivastava, S.K. Nanostructured anode materials for lithium ion batteries. J. Mater. Chem. A 2015, 3, 2454–2484. [Google Scholar] [CrossRef]
- Liu, N.; Li, W.; Pasta, M.; Cui, Y. Nanomaterials for electrochemical energy storage. Front. Phys. 2014, 9, 323–350. [Google Scholar] [CrossRef]
- Mahmood, N.; Tang, T.; Hou, Y. Nanostructured Anode Materials for Lithium Ion Batteries: Progress, Challenge and Perspective. Adv. Energy Mater. 2016, 6, 1600374. [Google Scholar] [CrossRef]
- Kumar, T.P.; Kumari, T.S.D.; Stephan, M.A. Carbonaceous anode materials for lithium-ion batteries–the road ahead. J. Indian Inst. Sci. 2009, 89, 393–424. [Google Scholar]
- Ni, W.; Shi, L. Review Article: Layer-structured carbonaceous materials for advanced Li-ion and Na-ion batteries: Beyond graphene. J. Vac. Sci. Technol. A 2019, 37, 040803. [Google Scholar] [CrossRef]
- González, M.A.; Marschilok, A.C.; Reichmanis, E. Perspective—Enhancing Active Anode Material Performance for Lithium-Ion Batteries via Manipulation of Interfacial Chemistry. J. Electrochem. Soc. 2020, 167, 050507. [Google Scholar] [CrossRef]
- Lu, Y.; Yu, L.; Lou, X.W. Nanostructured Conversion-type Anode Materials for Advanced Lithium-Ion Batteries. Chem 2018, 4, 972–996. [Google Scholar] [CrossRef]
- Zhong, Y.; Yang, M.; Zhou, X.; Zhou, Z. Structural design for anodes of lithium-ion batteries: Emerging horizons from materials to electrodes. Mater. Horiz. 2015, 2, 553–566. [Google Scholar] [CrossRef]
- Qi, X.; Li, B.-T.; Peng, S.-K.; Wang, N.; Chen, X.; Yan, S.-J. Cobalt chloride-ferric chloride-graphite Bi-Intercalation compounds as anode materials for high-performance lithium-ion batteries. J. Alloys Compd. 2021, 854, 157178. [Google Scholar] [CrossRef]
- Mu, Y.; Han, M.; Li, J.; Liang, J.; Yu, J. Growing vertical graphene sheets on natural graphite for fast charging lithium-ion batteries. Carbon 2021, 173, 477–484. [Google Scholar] [CrossRef]
- Xie, Q.; Qu, S.; Zhang, Y.; Zhao, P. Nitrogen-enriched graphene-like carbon architecture with tunable porosity derived from coffee ground as high performance anodes for lithium ion batteries. Appl. Surf. Sci. 2021, 537, 148092. [Google Scholar] [CrossRef]
- Berger, P.; Flandorfer, H. Sb–Sn alloy anodes for Li-ion batteries: The ternary system Li–Sb–Sn. J. Alloys Compd. 2021, 855, 157381. [Google Scholar] [CrossRef]
- Zhao, X.; Lehto, V.-P. Challenges and prospects of nanosized silicon anodes in lithium-ion batteries. Nanotechnology 2020, 32, 042002. [Google Scholar] [CrossRef]
- Shi, Q.; Zhou, J.; Ullah, S.; Yang, X.; Tokarska, K.; Trzebicka, B.; Ta, H.Q.; Rümmeli, M.H. A review of recent developments in Si/C composite materials for Li-ion batteries. Energy Storage Mater. 2021, 34, 735–754. [Google Scholar] [CrossRef]
- Casimir, A.; Zhang, H.; Ogoke, O.; Amine, J.C.; Lu, J.; Wu, G. Silicon-based anodes for lithium-ion batteries: Effectiveness of materials synthesis and electrode preparation. Nano Energy 2016, 27, 359–376. [Google Scholar] [CrossRef]
- Di, F.; Wang, N.; Li, L.; Geng, X.; Yang, H.; Zhou, W.; Sun, C.; An, B. Coral-like porous composite material of silicon and carbon synthesized by using diatomite as self-template and precursor with a good performance as anode of lithium-ions battery. J. Alloys Compd. 2021, 854, 157253. [Google Scholar] [CrossRef]
- Yang, Y.; Lu, Z.; Xia, J.; Liu, Y.; Wang, K.; Wang, X. Crystalline and amorphous carbon double-modified silicon anode: Towards large-scale production and superior lithium storage performance. Chem. Eng. Sci. 2021, 229, 116054. [Google Scholar] [CrossRef]
- Zeng, Y.; Huang, Y.; Liu, N.; Wang, X.; Zhang, Y.; Guo, Y.; Wu, H.-H.; Chen, H.; Tang, X.; Zhang, Q. N-doped porous carbon nanofibers sheathed pumpkin-like Si/C composites as free-standing anodes for lithium-ion batteries. J. Energy Chem. 2021, 54, 727–735. [Google Scholar] [CrossRef]
- Zhao, H.; Tu, N.; Zhang, W.; Zhang, M.; Wang, J. Novel synthesis of Silicon/Carbon nanotubes microspheres as anode additives through chemical vapor deposition in fluidized bed reactors. Scr. Mater. 2021, 192, 49–54. [Google Scholar] [CrossRef]
- Chung, W.-Y.; Brahma, S.; Hou, S.-C.; Chang, C.-C.; Huang, J.-L. Petroleum waste hydrocarbon resin as a carbon source modified on a Si composite as a superior anode material in lithium ion batteries. Mater. Chem. Phys. 2021, 259, 124011. [Google Scholar] [CrossRef]
- Nazir, A.; Le, H.T.T.; Kasbe, A.; Park, C.-J. Si nanoparticles confined within a conductive 2D porous Cu-based metal–organic framework (Cu3(HITP)2) as potential anodes for high-capacity Li-ion batteries. Chem. Eng. J. 2021, 405, 126963. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhu, C.; Ma, Z. Si@Cu3Si nano-composite prepared by facile method as high-performance anode for lithium-ion batteries. J. Alloys Compd. 2021, 851, 156854. [Google Scholar] [CrossRef]
- Jin, C.; Lai, Q.; Dan, J.; Xu, G.; Yue, Z.; Li, X.; Sun, F.; Huang, H.; Zhou, L.; Wang, L. A novel scalable synthesis of high-rate performance silicon anode materials by liquid-phase coating doping method. Appl. Surf. Sci. 2021, 540, 148326. [Google Scholar] [CrossRef]
- Yu, J.; Wang, K.; Song, W.; Huang, H.; Liang, C.; Xia, Y.; Zhang, J.; Gan, Y.; Wang, F.; Zhang, W. A low temperature MgH2-AlCl3-SiO2 system to synthesize nano-silicon for high-performance Li-ion batteries. Chem. Eng. J. 2021, 406, 126805. [Google Scholar] [CrossRef]
- Lee, S.H.; Park, C.; Do, K.; Ahn, H. Maximizing the utilization of active sites through the formation of native nanovoids of silicon oxycarbide as anode materials in lithium-ion batteries. Energy Storage Mater. 2021, 35, 130–141. [Google Scholar] [CrossRef]
- Lim, H.; Kim, H.; Kim, S.-O.; Kim, K.J.; Choi, W. Novel approach for controlling free-carbon domain in silicone oil-derived silicon oxycarbide (SiOC) as an anode material in secondary batteries. Chem. Eng. J. 2021, 404, 126581. [Google Scholar] [CrossRef]
- Narsimulu, D.; Kakarla, A.K.; Su Yu, J. Cerium vanadate/carbon nanotube hybrid composite nanostructures as a high-performance anode material for lithium-ion batteries. J. Energy Chem. 2021, 58, 25–32. [Google Scholar] [CrossRef]
- Ding, W.; Wu, X.; Li, Y.; Wang, S.; Miao, Z.; Zhou, P.; Zhou, J.; Zhuo, S. Three-dimensional graphene-wrapped Co0.85Se@C as high volumetric capacity anode material for lithium-ion batteries. Appl. Surf. Sci. 2021, 536, 147746. [Google Scholar] [CrossRef]
- Li, W.; Ma, Q.; Shen, P.; Zhou, Y.; Soule, L.; Li, Y.; Wu, Y.; Zhang, H.; Liu, M. Yolk-shell structured CuSi2P3@Graphene nanocomposite anode for long-life and high-rate lithium-ion batteries. Nano Energy 2021, 80, 105506. [Google Scholar] [CrossRef]
- Wu, B.-S.; Wang, P.; Teng, S.-H. Controllable synthesis and coating-thickness-dependent electrochemical properties of mesoporous carbon-coated α-Fe2O3 nanoparticles for lithium-ion batteries. Colloids Surf. A Physicochem. Eng. Asp. 2021, 610, 125907. [Google Scholar] [CrossRef]
- Cao, K.; Jia, Y.; Wang, S.; Huang, K.-J.; Liu, H. Mn3O4 nanoparticles anchored on carbon nanotubes as anode material with enhanced lithium storage. J. Alloys Compd. 2021, 854, 157179. [Google Scholar] [CrossRef]
- Han, J.; Ren, J. Enhanced cycling stability of hierarchical Cu2WS4/C hollow octahedron anode for lithium-ion batteries by tuning its potential window. J. Alloys Compd. 2021, 851, 156730. [Google Scholar] [CrossRef]
- Trukawka, M.; Wenelska, K.; Singer, L.; Klingeler, R.; Chen, X.; Mijowska, E. Hollow carbon spheres loaded with uniform dispersion of copper oxide nanoparticles for anode in lithium- ion batteries. J. Alloys Compd. 2021, 853, 156700. [Google Scholar] [CrossRef]
- Thauer, E.; Zakharova, G.S.; Wegener, S.A.; Zhu, Q.; Klingeler, R. Sol-gel synthesis of Li3VO4/C composites as anode materials for lithium-ion batteries. J. Alloys Compd. 2021, 853, 157364. [Google Scholar] [CrossRef]
- Chu, K.; Li, Z.; Xu, S.; Yao, G.; Xu, Y.; Niu, P.; Zheng, F. NiO nanocrystals encapsulated into a nitrogen-doped porous carbon matrix as highly stable Li-ion battery anodes. J. Alloys Compd. 2021, 854, 157264. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, M.; Yan, X.; Han, Q.; Dong, C.; Sun, X.; You, D.; Jiang, F. Spherical-graphite/nano-Mn2O3 composites as advanced anode materials for lithium half/full batteries. J. Alloys Compd. 2021, 853, 157109. [Google Scholar] [CrossRef]
- Wang, X.; Jia, Z.; Zhang, J.; Ou, X.; Zhang, B.; Feng, J.; Hou, F.; Liang, J. Nanophase MnV2O4 particles as anode materials for lithium-ion batteries. J. Alloys Compd. 2021, 852, 156999. [Google Scholar] [CrossRef]
- Lu, X.; Chen, Y.; Tian, Q.; Zhang, W.; Sui, Z.; Chen, J. Enabling improved cycling stability of hollow SnO2/C composite anode for lithium-ion battery by constructing a built-in porous carbon support. Appl. Surf. Sci. 2021, 537, 148052. [Google Scholar] [CrossRef]
- Ni, X.; Cui, Z.; Jiang, N.; Chen, H.; Wu, Q.; Ju, A.; Zhu, M. Hollow multi-nanochannel carbon nanofiber/MoS2 nanoflower composites as binder-free lithium-ion battery anodes with high capacity and ultralong-cycle life at large current density. J. Mater. Sci. Technol. 2021, 77, 169–177. [Google Scholar] [CrossRef]
- Ni, X.; Cui, Z.; Luo, H.; Chen, H.; Liu, C.; Wu, Q.; Ju, A. Hollow multi-nanochannel carbon nanofibers@MoSe2 nanosheets composite as flexible anodes for high performance lithium-ion batteries. Chem. Eng. J. 2021, 404, 126249. [Google Scholar] [CrossRef]
- Sun, Y.-H.; Huang, M.-X.; Guan, D.-C.; Zhang, G.-L.; Wei, J.-L.; Nan, J.-M.; Yi, F.-Y. Influence of the Sn(Fe)–C bonds content in SnFe2O4@reduced graphene oxide composites on the electrochemical behavior of lithium-ion batteries. J. Alloys Compd. 2021, 854, 157297. [Google Scholar] [CrossRef]
- Wu, Y.-Q.; Zhao, Y.-S.; Meng, W.-J.; Xie, Y.; Zhang, J.; He, C.-J.; Zhao, D.-L. Nanoplates-assembled SnS2 nanoflowers with carbon coating anchored on reduced graphene oxide for high performance Li-ion batteries. Appl. Surf. Sci. 2021, 539, 148283. [Google Scholar] [CrossRef]
- Zhang, Z.; Huang, Q.; Ma, W.; Li, H. Interfacial engineering of polyhedral carbon@hollowed carbon@SiO2 nanobox with tunable structure for enhanced lithium ion battery. Appl. Surf. Sci. 2021, 538, 148039. [Google Scholar] [CrossRef]
- Liu, D.; Wei, Z.; Liu, L.; Pan, H.; Duan, X.; Xia, L.; Zhong, B.; Wang, H.; Jia, D.; Zhou, Y.; et al. Ultrafine SnO2 anchored in ordered mesoporous carbon framework for lithium storage with high capacity and rate capability. Chem. Eng. J. 2021, 406, 126710. [Google Scholar] [CrossRef]
- Bai, C.; Feng, Y.; Ke, J.; Wu, K.; He, M. Superior electrochemical performance of sheet-stacked SnO2@ZrO2/C composite as anode material for lithium-ion batteries. Chem. Phys. Lett. 2021, 763, 138220. [Google Scholar] [CrossRef]
- Zhang, M.; Tang, C.; Cheng, W.; Fu, L. The first-principles study on the performance of the graphene/WS2 heterostructure as an anode material of Li-ion battery. J. Alloys Compd. 2021, 855, 157432. [Google Scholar] [CrossRef]
- Li, M.; Su, A.; Qin, Q.; Qin, Y.; Dou, A.; Zhou, Y.; Su, M.; Liu, Y. High-rate capability of columbite CuNb2O6 anode materials for lithium-ion batteries. Mater. Lett. 2021, 284, 128915. [Google Scholar] [CrossRef]
- Zhao, H.; Ren, Y.; Chen, S.; Xu, J.; Lu, Z. Novel synthesis of sea urchins-like TiNb2O7 with hierarchical structure as anode for lithium-ion batteries. Mater. Lett. 2021, 284, 128950. [Google Scholar] [CrossRef]
- Chen, W.; Fan, M.; Yue, M.; Wang, S.; Liu, J. Rational design of nano-structured ammonium manganese phosphate hydrate as lithium ion battery anode. Mater. Lett. 2021, 285, 129084. [Google Scholar] [CrossRef]
- Wu, W.; Wei, Y.; Chen, H.; Wei, K.; Li, Z.; He, J.; Deng, L.; Yao, L.; Yang, H. In-situ encapsulation of α-Fe2O3 nanoparticles into ZnFe2O4 micro-sized capsules as high-performance lithium-ion battery anodes. J. Mater. Sci. Technol. 2021, 75, 110–117. [Google Scholar] [CrossRef]
- Yao, J.; Yang, Y.; Li, Y.; Jiang, J.; Xiao, S.; Yang, J. Interconnected α-Fe2O3 nanoparticles prepared from leaching liquor of tin ore tailings as anode materials for lithium-ion batteries. J. Alloys Compd. 2021, 855, 157288. [Google Scholar] [CrossRef]
- Sun, M.; Chu, X.; Wang, Z.; Yang, H.; Yang, Z.; Ma, J.; Zhou, B.; Yang, T.; Chen, L. Rosa roxburghii-like hierarchical hollow sandwich-structure C@Fe2O3@C microspheres as second nanomaterialsfor superior lithium storage. J. Alloys Compd. 2021, 855, 157518. [Google Scholar] [CrossRef]
- Zhong, W.; Huang, X.; Lin, Y.; Cao, Y.; Wang, Z. Compact Co3O4/Co in-situ nanocomposites prepared by pulsed laser sintering as anode materials for lithium-ion batteries. J. Energy Chem. 2021, 58, 386–390. [Google Scholar] [CrossRef]
- Zheng, J.; He, C.; Li, X.; Wang, K.; Wang, T.; Zhang, R.; Tang, B.; Rui, Y. CoS2–MnS@Carbon nanoparticles derived from metal–organic framework as a promising anode for lithium-ion batteries. J. Alloys Compd. 2021, 854, 157315. [Google Scholar] [CrossRef]
- Xiao, X.; Zhang, Z.; Yang, K.; Mei, T.; Yan, D.; Wang, X. Design and synthesize hollow spindle Ni-doped Co9S8@ZnS composites and their enhanced cycle performance. J. Alloys Compd. 2021, 853, 157118. [Google Scholar] [CrossRef]
- Gao, J.; Wang, X.; Huang, Y.; Meng, Z.; Sun, Y.; Zhang, Y.; Guo, Y.; Tang, X. Hollow core-shell structured CNT/PAN@Co9S8@C coaxial nanocables as high-performance anode material for lithium ion batteries. J. Alloys Compd. 2021, 853, 157354. [Google Scholar] [CrossRef]
- Xia, D.; Yang, J.; Li, M.; Gao, H.; Gong, F. Hybridized cobalt/manganese vanadates as high-performance anodes in lithium ion battery. Mater. Lett. 2021, 283, 128782. [Google Scholar] [CrossRef]
- Chen, H.; Ke, G.; Wu, X.; Li, W.; Li, Y.; Mi, H.; Sun, L.; Zhang, Q.; He, C.; Ren, X. Amorphous MoS3 decoration on 2D functionalized MXene as a bifunctional electrode for stable and robust lithium storage. Chem. Eng. J. 2021, 406, 126775. [Google Scholar] [CrossRef]
- Chen, Z.; Li, H. A novel phosphatizing strategy to engineering CoO/Co1.94P@carbon polyhedron heterostructures for enhanced lithium-ion battery. J. Mater. Sci. 2021, 56, 3346–3353. [Google Scholar] [CrossRef]
- Tan, Y.; Yang, C.; Qian, W.; Sui, X.; Teng, C.; Li, Q.; Lu, Z. Carbon coated porous Co3O4 polyhedrons as anode materials for highly reversible lithium-ion storage. J. Alloys Compd. 2021, 855, 157387. [Google Scholar] [CrossRef]
- Lopez, J.; Gonzalez, R.; Ayala, J.; Cantu, J.; Castillo, A.; Parsons, J.; Myers, J.; Lodge, T.P.; Alcoutlabi, M. Centrifugally spun TiO2/C composite fibers prepared from TiS2/PAN precursor fibers as binder-free anodes for LIBS. J. Phys. Chem. Solids 2021, 149, 109795. [Google Scholar] [CrossRef]
- Guo, F.; Chen, H.; Chen, Y.; Zhang, W.; Li, Z.; Xu, Y.; Wang, Y.; Zhou, J.; Zhang, H. Dual-metal-organic frameworks derived manganese and zinc oxides nanohybrids as high performance anodes for lithium-ion batteries. J. Alloys Compd. 2021, 852, 156814. [Google Scholar] [CrossRef]
- Sun, H.; Xu, J.; Huang, J.; Li, G.; Luo, J.; Rao, M.; Peng, Z.; Jiang, T. Facile synthesis of hetero-structured few-layer MoS2-coated MoO2 as superior anode materials of lithium ion batteries. J. Alloys Compd. 2021, 851, 156726. [Google Scholar] [CrossRef]
- Liu, J.; Li, G.; Wu, J. Fe2O3–TeO2–MoO3 semiconductor glass-ceramics as anode materials for high specific capacity lithium ion batteries. Mater. Chem. Phys. 2021, 258, 123894. [Google Scholar] [CrossRef]
- Shen, H.; Xia, X.; Yan, S.; Jiao, X.; Sun, D.; Lei, W.; Hao, Q. SnO2/NiFe2O4/graphene nanocomposites as anode materials for lithium ion batteries. J. Alloys Compd. 2021, 853, 157017. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, F.; Liu, K.; Zhu, J.; Chen, T.; Gu, Z.; Yin, S. Cobalt phosphide nanoparticles grown on Ti3C2 nanosheet for enhanced lithium ions storage performances. J. Alloys Compd. 2021, 853, 157136. [Google Scholar] [CrossRef]
- Niu, R.; Han, R.; Wang, Y.; Zhang, L.; Qiao, Q.; Jiang, L.; Sun, Y.; Tang, S.; Zhu, J. MXene-based porous and robust 2D/2D hybrid architectures with dispersed Li3Ti2(PO4)3 as superior anodes for lithium-ion battery. Chem. Eng. J. 2021, 405, 127049. [Google Scholar] [CrossRef]
- Wu, J.; Lin, Y.; Huang, X. Aspergillus oryzae spore carbon/niobium oxide composite material as anode for lithium ion batteries. Mater. Res. Bull. 2021, 134, 111062. [Google Scholar] [CrossRef]
- Li, J.; Han, L.; Zhang, X.; Sun, H.; Liu, X.; Lu, T.; Yao, Y.; Pan, L. Multi-role TiO2 layer coated carbon@few-layered MoS2 nanotubes for durable lithium storage. Chem. Eng. J. 2021, 406, 126873. [Google Scholar] [CrossRef]
- Wei, R.; Dong, Y.; Zhang, Y.; Zhang, R.; Al-Tahan, M.A.; Zhang, J. In-situ self-assembled hollow urchins F-Co-MOF on rGO as advanced anodes for lithium-ion and sodium-ion batteries. J. Colloid Interface Sci. 2021, 582, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Liu, J.; Zhang, Q.; Jin, Y.; Wang, H. Metal organic framework derived Co3O4/Co@N–C composite as high-performance anode material for lithium-ion batteries. J. Alloys Compd. 2021, 855, 157538. [Google Scholar] [CrossRef]
- Wang, Z.; Bi, R.; Liu, J.; Wang, K.; Mao, F.; Wu, H.; Bu, Y.; Song, N. Polyoxometalate-based Cu/Zn-MOFs with diverse stereo dimensions as anode materials in lithium ion batteries. Chem. Eng. J. 2021, 404, 127117. [Google Scholar] [CrossRef]
- Costa, C.M.; Gonçalves, R.; Lanceros-Méndez, S. Advances in Cathode Nanomaterials for Lithium-Ion Batteries. In Nanostructured Materials for Next-Generation Energy Storage and Conversion: Advanced Battery and Supercapacitors; Zhen, Q., Bashir, S., Liu, J.L., Eds.; Springer: Berlin/Heidelberg, Germany, 2019; pp. 105–145. [Google Scholar]
- Nitta, N.; Wu, F.; Lee, J.T.; Yushin, G. Li-ion battery materials: Present and future. Mater. Today 2015, 18, 252–264. [Google Scholar] [CrossRef]
- Lu, G.; Peng, W.; Zhang, Y.; Wang, X.; Shi, X.; Song, D.; Zhang, H.; Zhang, L. Study on the formation, development and coating mechanism of new phases on interface in LiNbO3-coated LiCoO2. Electrochim. Acta 2021, 368, 137639. [Google Scholar] [CrossRef]
- Ding, C.-C.; Jia, L.; Wang, X.; Li, Y.; Wang, Z.-Q.; Wu, L.-N. An investigation of local structures for 3d3, 3d5 and 3d7 ions doing in the layered LiCoO2 cathode materials. J. Phys. Chem. Solids 2021, 150, 109800. [Google Scholar] [CrossRef]
- Li, Y.; Wang, L.; Zhang, K.; Liang, F.; Yuan, M.; Zhang, H.; Yao, Y. An encapsulation of phosphorus doped carbon over LiFePO4 prepared under vacuum condition for lithium-ion batteries. Vacuum 2021, 184, 109935. [Google Scholar] [CrossRef]
- Kanagaraj, A.B.; Chaturvedi, P.; Kim, H.J.; Choi, D.S. Controllable synthesis of LiFePO4 microrods and its superior electrochemical performance. Mater. Lett. 2021, 283, 128737. [Google Scholar] [CrossRef]
- Chen, X.; Li, Y.; Wang, J. Enhanced Electrochemical Performance of LiFePO4 Originating from the Synergistic Effect of ZnO and C Co-Modification. Nanomaterials 2021, 11, 12. [Google Scholar] [CrossRef]
- Shi, M.; Li, R.; Liu, Y. In situ preparation of LiFePO4/C with unique copolymer carbon resource for superior performance lithium-ion batteries. J. Alloys Compd. 2021, 854, 157162. [Google Scholar] [CrossRef]
- Wei, X.; Zhang, M.; Miao, B.; Liu, R. Volumetric capacity enhancement in LiFePO4 cathodes by hot isostatic pressing. Scr. Mater. 2021, 194, 113638. [Google Scholar] [CrossRef]
- Xie, X.; Zhang, B.; Hu, G.; Du, K.; Wu, J.; Wang, Y.; Gan, Z.; Fan, J.; Su, H.; Cao, Y.; et al. A new route for green synthesis of LiFe0.25Mn0.75PO4/C@rGO material for lithium ion batteries. J. Alloys Compd. 2021, 853, 157106. [Google Scholar] [CrossRef]
- Umam, K.; Sin, B.C.; Singh, L.; Moon, C.; Choi, J.; Lee, I.; Lim, J.; Jung, J.; Lah, M.S.; Lee, Y. Phase transition-induced improvement in the capacity of fluorine-substituted LiFeBO3 as a cathode material for lithium ion batteries. Electrochim. Acta 2021, 367, 137364. [Google Scholar] [CrossRef]
- Gonçalves, R.; Sharma, P.; Ram, P.; Ferdov, S.; Silva, M.M.; Costa, C.M.; Singhal, R.; Sharma, R.K.; Lanceros-Méndez, S. Improved electrochemical performance of LiMn1.5M0.5O4 (M=Ni, Co, Cu) based cathodes for lithium-ion batteries. J. Alloys Compd. 2021, 853, 157208. [Google Scholar] [CrossRef]
- Tian, Y.; Qiu, Y.; Liu, Z.; Wei, X.; Cao, H. LiMnO2@rGO nanocomposites for high-performance lithium-ion battery cathodes. Nanotechnology 2020, 32, 015402. [Google Scholar] [CrossRef] [PubMed]
- Abulikemu, A.; Matsunaga, T.; Watanabe, A.; Yamamoto, K.; Uchiyama, T.; Nakanishi, K.; Kawaguchi, S.; Osaka, K.; Uchimoto, Y. Rocksalt type Li2Nb0·15Mn0·85O3 without structure degradation or redox evolution upon cycling. J. Alloys Compd. 2021, 853, 156984. [Google Scholar] [CrossRef]
- Mereacre, V.; Bohn, N.; Müller, M.; Indris, S.; Bergfeldt, T.; Binder, J.R. Improved performance of high-voltage Li-ion batteries using a novel chemically activated coating process. Mater. Res. Bull. 2021, 134, 111095. [Google Scholar] [CrossRef]
- Kocak, T.; Wu, L.; Wang, J.; Savaci, U.; Turan, S.; Zhang, X. The effect of vanadium doping on the cycling performance of LiNi0.5Mn1.5O4 spinel cathode for high voltage lithium-ion batteries. J. Electroanal. Chem. 2021, 881, 114926. [Google Scholar] [CrossRef]
- Yang, Z.; Yu, H.; Hu, Y.; Zhu, H.; Zhu, Y.; Jiang, H.; Li, C. Pomegranate-like Ti-doped LiNi0.4Mn1.6O4 5 V-class cathode with superior high-voltage cycle and rate performance for Li-ion batteries. Chem. Eng. Sci. 2021, 231, 116297. [Google Scholar] [CrossRef]
- Wu, Q.; Mao, S.; Wang, Z.; Tong, Y.; Lu, Y. Improving LiNixCoyMn1−x−yO2 cathode electrolyte interface under high voltage in lithium ion batteries. Nano Sel. 2020, 1, 111–134. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, J. An overview of modification strategies to improve LiNi0·8Co0·1Mn0·1O2 (NCM811) cathode performance for automotive lithium-ion batteries. eTransportation 2021, 7, 100105. [Google Scholar] [CrossRef]
- Niu, S.; Xu, J.; Wu, K.; Liang, C.; Zhu, G.; Qu, Q.; Zheng, H. Different mechanical and electrochemical behavior between the two major Ni-rich cathode materials in Li-Ion batteries. Mater. Chem. Phys. 2021, 260, 124046. [Google Scholar] [CrossRef]
- Bai, X.; He, R.; Wei, A.; Li, X.; Zhang, L.; Liu, Z. A Co-Modified strategy for enhanced structural stability and long cycling life of Ni-Rich LiNi0.8Co0.1Mn0.1O2 cathode material. J. Alloys Compd. 2021, 857, 157877. [Google Scholar] [CrossRef]
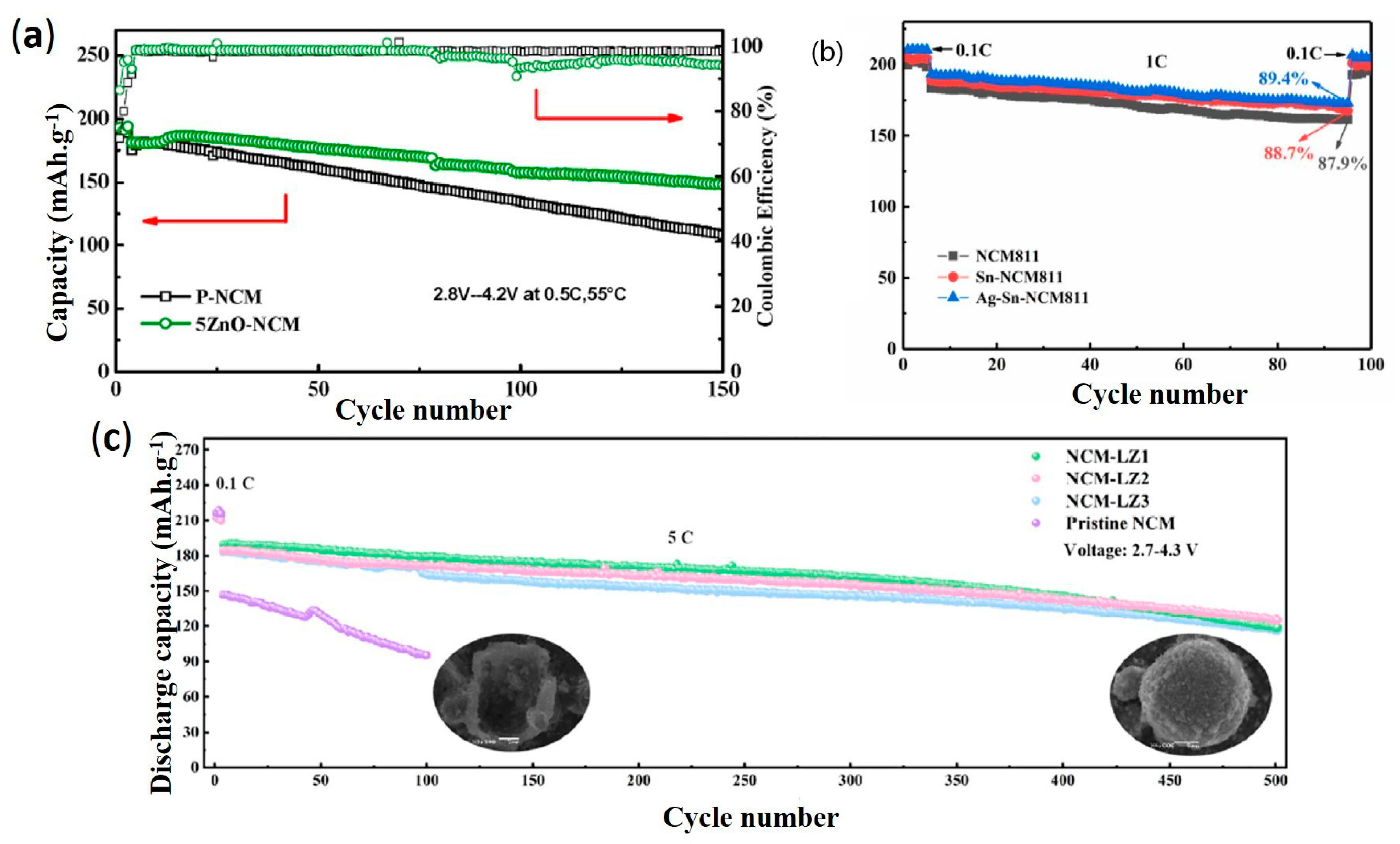
- Sun, H.; Wang, J.; Liu, Q.; Zhang, Y.; Zhang, D.; Wang, Q.; Li, Z.; Li, W.; Wang, B. Ag–Sn dual-modified LiNi0.8Co0.1Mn0.1O2 as cathode for lithium storage. J. Alloys Compd. 2021, 850, 156763. [Google Scholar] [CrossRef]
- Ning, R.; Yuan, K.; Zhang, K.; Shen, C.; Xie, K. A scalable snowballing strategy to construct uniform rGO-wrapped LiNi0.8Co0.1Mn0.1O2 with enhanced processability and electrochemical performance. Appl. Surf. Sci. 2021, 542, 148663. [Google Scholar] [CrossRef]
- Tang, L.-B.; Liu, Y.; Wei, H.-X.; Yan, C.; He, Z.-J.; Li, Y.-J.; Zheng, J.-C. Boosting cell performance of LiNi0.8Co0.1Mn0.1O2 cathode material via structure design. J. Energy Chem. 2021, 55, 114–123. [Google Scholar] [CrossRef]
- You, L.; Chu, B.; Li, G.; Huang, T.; Yu, A. H3BO3 washed LiNi0.8Co0.1Mn0.1O2 with enhanced electrochemical performance and storage characteristics. J. Power Sources 2021, 482, 228940. [Google Scholar] [CrossRef]
- Hwang, J.; Do, K.; Ahn, H. Highly conductive 3D structural carbon network-encapsulated Ni-rich LiNi0.8Co0.1Mn0.1O2 as depolarized and passivated cathode for lithium-ion batteries. Chem. Eng. J. 2021, 406, 126813. [Google Scholar] [CrossRef]
- Zha, G.; Hu, W.; Agarwal, S.; Ouyang, C.; Hu, N.; Hou, H. High performance layered LiNi0.8Co0.07Fe0.03Mn0.1O2 cathode materials for Li-ion battery. Chem. Eng. J. 2021, 409, 128343. [Google Scholar] [CrossRef]
- Gu, H.; Wang, J.; Wang, Z.; Tong, J.; Qi, N.; Han, G.; Zhang, M. Self-assembled porous LiNi0.8Co0.1Mn0.1O2 cathode materials with micro/nano-layered hollow morphologies for high-power lithium-ion batteries. Appl. Surf. Sci. 2021, 539, 148034. [Google Scholar] [CrossRef]
- Wu, Y.-S.; Pham, Q.-T.; Yang, C.-C.; Chern, C.-S.; Musuvadhi Babulal, L.; Seenivasan, M.; Brunklaus, G.; Placke, T.; Hwang, B.-J.; Winter, M. Study of electrochemical performance and thermal property of LiNi0.5Co0.2Mn0.3O2 cathode materials coated with a novel oligomer additive for high-safety lithium-ion batteries. Chem. Eng. J. 2021, 405, 126727. [Google Scholar] [CrossRef]
- Zhang, Y.; Cui, C.; He, Y.; Liu, J.; Song, Y.; Song, Z.; Xu, H.; Huang, S.; Bei, Y. The effect of drying methods on the structure and performance of LiNi0.5Co0.2Mn0.3O2 cathode material for lithium-ion batteries. Mater. Chem. Phys. 2021, 262, 124269. [Google Scholar] [CrossRef]
- Yao, L.; Li, Y.; Gao, X.; Cai, M.; Jin, J.; Yang, J.; Xiu, T.; Song, Z.; Badding, M.E.; Wen, Z. Microstructure boosting the cycling stability of LiNi0.6Co0.2Mn0.2O2 cathode through Zr-based dual modification. Energy Storage Mater. 2021, 36, 179–185. [Google Scholar] [CrossRef]
- Meng, F.; Hu, R.; Chen, Z.; Tan, L.; Lan, X.; Yuan, B. Plasma assisted synthesis of LiNi0.6Co0.2Mn0.2O2 cathode materials with good cyclic stability at subzero temperatures. J. Energy Chem. 2021, 56, 46–55. [Google Scholar] [CrossRef]
- Li, L.; Liu, Y.; Yu, H.; Sun, R.; Ding, Z.; Li, K.; Yuan, Q.; Wu, J.; Liu, X. Hollow spherical 0.5Li2MnO3·0.5LiMn1/3Ni1/3Co1/3O2 prepared by facile molten salt method for enhanced long-cycle and rate capability of lithium-ion batteries. J. Alloys Compd. 2021, 855, 157376. [Google Scholar] [CrossRef]
- Pang, P.; Tan, X.; Wang, Z.; Cai, Z.; Nan, J.; Xing, Z.; Li, H. Crack-free single-crystal LiNi0.83Co0.10Mn0.07O2 as cycling/thermal stable cathode materials for high-voltage lithium-ion batteries. Electrochim. Acta 2021, 365, 137380. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, B.; Hu, Y.; Li, X.; Zhao, T. Failure analysis of LiNi0·83Co0·12Mn0·05O2/graphite–SiOx pouch batteries cycled at high temperature. J. Power Sources 2021, 482, 228978. [Google Scholar] [CrossRef]
- Jamil, S.; Ran, Q.; Yang, L.; Huang, Y.; Cao, S.; Yang, X.; Wang, X. Improved high-voltage performance of LiNi0.87Co0.1Al0.03O2 by Li+-conductor coating. Chem. Eng. J. 2021, 407, 126442. [Google Scholar] [CrossRef]
- Song, X.; Liu, G.; Yue, H.; Luo, L.; Yang, S.; Huang, Y.; Wang, C. A novel low-cobalt long-life LiNi0.88Co0.06Mn0.03Al0.03O2 cathode material for lithium ion batteries. Chem. Eng. J. 2021, 407, 126301. [Google Scholar] [CrossRef]
- Shan, W.; Zhang, H.; Hu, C.; Zhou, Y.; Lam, K.-h.; Wang, S.; Hou, X. The cycle performance of high nickel cathode materials significantly enhanced by the LiAlO2@Al2O3 dual-modified coating. Electrochim. Acta 2021, 367, 137216. [Google Scholar] [CrossRef]
- Sim, S.-J.; Lee, S.-H.; Jin, B.-S.; Kim, H.-S. Effects of lithium tungsten oxide coating on LiNi0.90Co0.05Mn0.05O2 cathode material for lithium-ion batteries. J. Power Sources 2021, 481, 229037. [Google Scholar] [CrossRef]
- Thien Nguyen, T.; Kim, U.-H.; Yoon, C.S.; Sun, Y.-K. Enhanced cycling stability of Sn-doped Li[Ni0.90Co0.05Mn0.05]O2 via optimization of particle shape and orientation. Chem. Eng. J. 2021, 405, 126887. [Google Scholar] [CrossRef]
- Liu, H.; Li, M.; Xiang, M.; Guo, J.; Bai, H.; Bai, W.; Liu, X. Effects of crystal structure and plane orientation on lithium and nickel co-doped spinel lithium manganese oxide for long cycle life lithium-ion batteries. J. Colloid Interface Sci. 2021, 585, 729–739. [Google Scholar] [CrossRef]
- Liu, Q.; Liang, Q.; Guo, J.; Xiang, M.; Bai, W.; Bai, H.; Liu, X. High rate performance and kinetic investigation of polyhedral Li1·05Mn1.95-xNixO4 cathode material. Ceram. Int. 2021, 47, 2441–2449. [Google Scholar] [CrossRef]
- Yu, M.; Li, J.; Ning, X. Improving electrochemical performance of LiMn0.5Fe0.5PO4 cathode by hybrid coating of Li3VO4 and carbon. Electrochim. Acta 2021, 368, 137597. [Google Scholar] [CrossRef]
- Zhu, L.; Mo, L.; Xie, L.; Cao, X. Synthesis and electrochemical Li-storage performance of Li2ZrO3-Li3V2(PO4)3/C composites. Electrochem. Commun. 2021, 122, 106908. [Google Scholar] [CrossRef]
- Amou, M.; Larhrib, B.; Saadoune, I. Controlled synthesis of Li1.17Ni0.21Mn0.54Co0.08O2 as a cathode material for Li ion batteries. J. Electroanal. Chem. 2021, 881, 114957. [Google Scholar] [CrossRef]
- Tang, W.; Chen, Z.; Huang, H.; Irfan, M.; Huang, C.; Yang, Z.; Zhang, W. PVP-bridged γ-LiAlO2 nanolayer on Li1.2Ni0.182Co0.08Mn0.538O2 cathode materials for improving the rate capability and cycling stability. Chem. Eng. Sci. 2021, 229, 116126. [Google Scholar] [CrossRef]
- Amzil, S.; Zhang, Y.; Qiu, T.; Jiang, F.; Rao, H.; Fang, Z.; Huang, J. {010} oriented Li1.2Mn0.54Co0.13Ni0.13O2@C nanoplate with high initial columbic efficiency. J. Power Sources 2021, 486, 229372. [Google Scholar] [CrossRef]
- Luo, Z.; Zhou, Z.; He, Z.; Sun, Z.; Zheng, J.; Li, Y. Enhanced electrochemical performance of Li1.2Mn0.54Ni0.13Co0.13O2 cathode by surface modification using La–Co–O compound. Ceram. Int. 2021, 47, 2656–2664. [Google Scholar] [CrossRef]
- Yin, C.; Wan, L.; Qiu, B.; Wang, F.; Jiang, W.; Cui, H.; Bai, J.; Ehrlich, S.; Wei, Z.; Liu, Z. Boosting energy efficiency of Li-rich layered oxide cathodes by tuning oxygen redox kinetics and reversibility. Energy Storage Mater. 2021, 35, 388–399. [Google Scholar] [CrossRef]
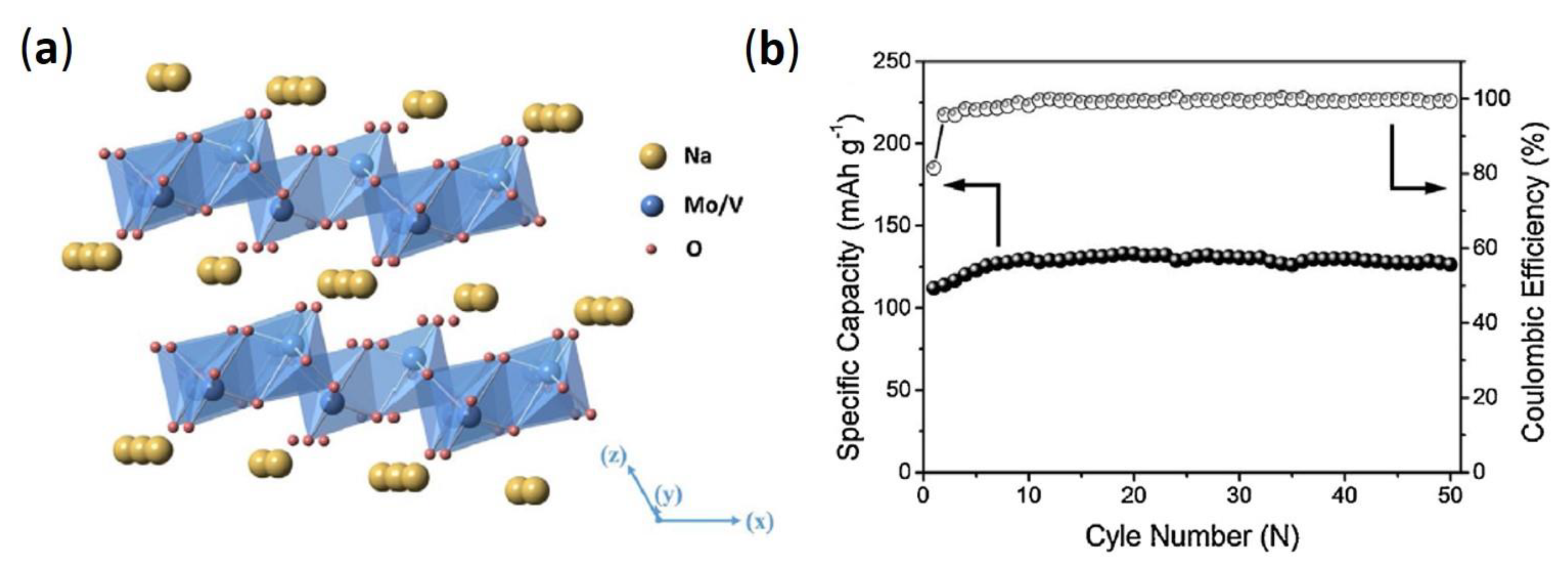
- Huang, C.; Liu, S.; Wang, Y.; Feng, J.; Zhao, Y. A new active NaVMoO6 cathode material for rechargeable Li ion batteries. J. Mater. Sci. Technol. 2021, 66, 97–102. [Google Scholar] [CrossRef]
- Li, C.; Zhang, C.; Xie, J.; Wang, K.; Li, J.; Zhang, Q. Ferrocene-based metal-organic framework as a promising cathode in lithium-ion battery. Chem. Eng. J. 2021, 404, 126463. [Google Scholar] [CrossRef]
- Costa, C.M.; Silva, M.M.; Lanceros-Méndez, S. Battery separators based on vinylidene fluoride (VDF) polymers and copolymers for lithium ion battery applications. RSC Adv. 2013, 3, 11404–11417. [Google Scholar] [CrossRef]
- Costa, C.M.; Lee, Y.-H.; Kim, J.-H.; Lee, S.-Y.; Lanceros-Méndez, S. Recent advances on separator membranes for lithium-ion battery applications: From porous membranes to solid electrolytes. Energy Storage Mater. 2019, 22, 346–375. [Google Scholar] [CrossRef]
- Ren, W.; Zheng, Y.; Cui, Z.; Tao, Y.; Li, B.; Wang, W. Recent progress of functional separators in dendrite inhibition for lithium metal batteries. Energy Storage Mater. 2021, 35, 157–168. [Google Scholar] [CrossRef]
- Li, M.; Sheng, L.; Zhang, H.; Yang, Y.; Xu, R.; Bai, Y.; Song, S.; Liu, G.; Wang, T.; Huang, X.; et al. Effect of the heat treatment temperature on mechanical and electrochemical properties of polyimide separator for lithium ion batteries. J. Mater. Sci. 2020, 55, 16158–16170. [Google Scholar] [CrossRef]
- Jaritphun, S.; Park, J.S.; Chung, O.H.; Nguyen, T.T.T. Sandwiched polyimide-composite separator for lithium-ion batteries via electrospinning and electrospraying. Polym. Compos. 2020, 41, 4478–4488. [Google Scholar] [CrossRef]
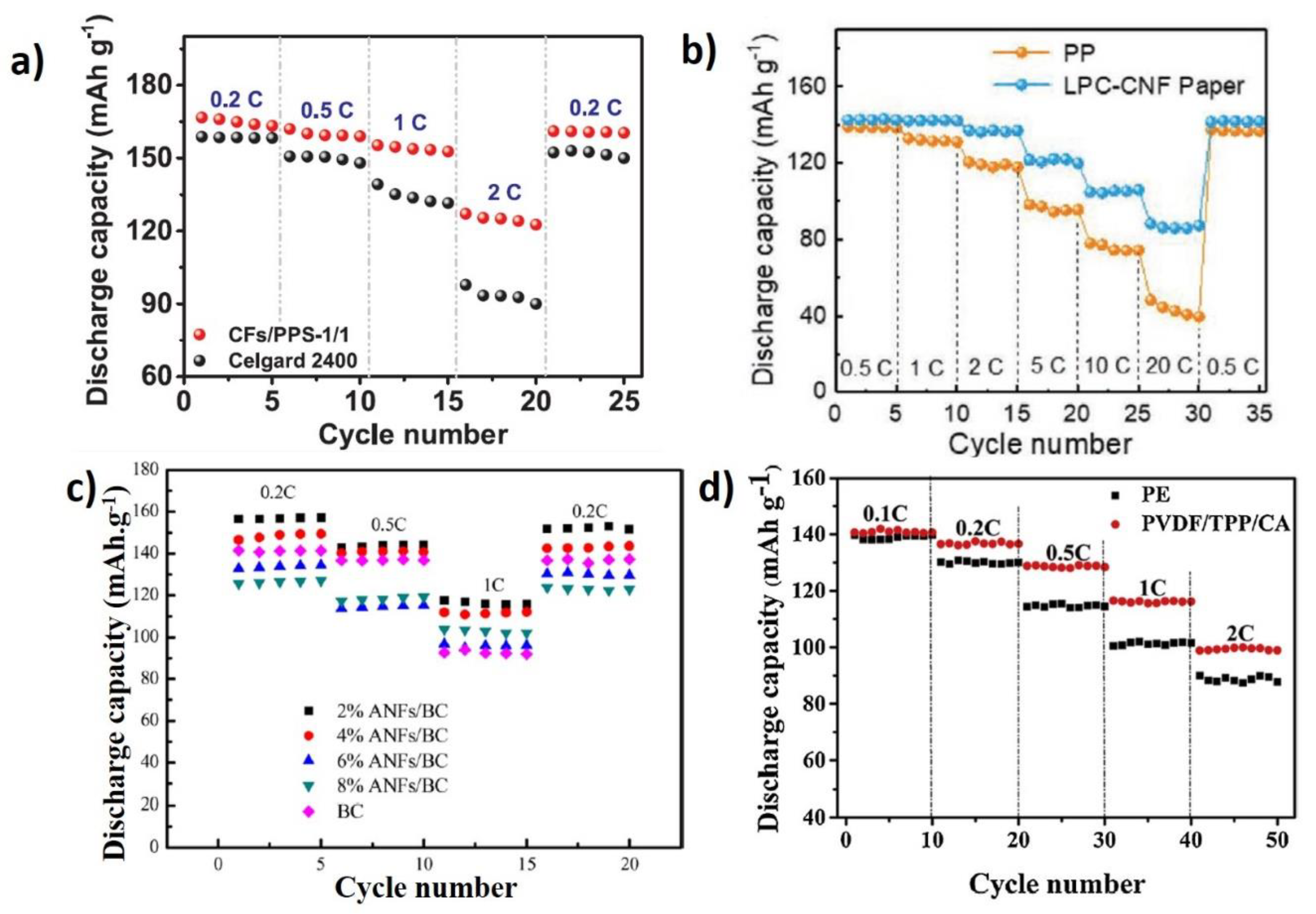
- Gu, Q.-Q.; Xue, H.-J.; Li, Z.-W.; Song, J.-C.; Sun, Z.-Y. High-performance polyethylene separators for lithium-ion batteries modified by phenolic resin. J. Power Sources 2021, 483, 229155. [Google Scholar] [CrossRef]
- Zhang, H.; Sheng, L.; Bai, Y.; Song, S.; Liu, G.; Xue, H.; Wang, T.; Huang, X.; He, J. Amino-Functionalized Al2O3 Particles Coating Separator with Excellent Lithium-Ion Transport Properties for High-Power Density Lithium-Ion Batteries. Adv. Eng. Mater. 2020, 22, 1901545. [Google Scholar] [CrossRef]
- Li, J.; Bi, S.; Li, M.; Xian, Y.; Shui, Y.; Yao, Y.; Wu, M. Rapid homogenization preparation of the mussel-inspired hydrophilic separator for high power lithium-ion batteries. J. Appl. Polym. Sci. 2020, 137, 49052. [Google Scholar] [CrossRef]
- Khassi, K.; Youssefi, M.; Semnani, D. PVDF/TiO2/graphene oxide composite nanofiber membranes serving as separators in lithium-ion batteries. J. Appl. Polym. Sci. 2020, 137, 48775. [Google Scholar] [CrossRef]
- Li, Y.; Wang, X.; Liang, J.; Wu, K.; Xu, L.; Wang, J. Design of A High Performance Zeolite/Polyimide Composite Separator for Lithium-Ion Batteries. Polymers 2020, 12, 764. [Google Scholar] [CrossRef]
- Cui, W.; Shi, L.; Song, W.; Wang, X.; Lin, Z.; Deng, W.; Ma, Y. A heatproof electrospun PES/PVDF composite membrane as an advanced separator for lithium-ion batteries. J. Appl. Polym. Sci. 2020, 137, 49328. [Google Scholar] [CrossRef]
- Zhao, H.; Deng, N.; Kang, W.; Li, Z.; Wang, G.; Cheng, B. Highly multiscale structural Poly(vinylidene fluoridehexafluoropropylene)/poly-m-phenyleneisophthalamide separator with enhanced interface compatibility and uniform lithium-ion flux distribution for dendrite-proof lithium-metal batteries. Energy Storage Mater. 2020, 26, 334–348. [Google Scholar] [CrossRef]
- An, M.-Y.; Kim, H.-T.; Chang, D.-R. Multilayered separator based on porous polyethylene layer, Al2O3 layer, and electro-spun PVdF nanofiber layer for lithium batteries. J. Solid State Electrochem. 2014, 18, 1807–1814. [Google Scholar] [CrossRef]
- Ahn, Y.-K.; Kwon, Y.K.; Kim, K.J. Surface-modified polyethylene separator with hydrophilic property for enhancing the electrochemical performance of lithium-ion battery. Int. J. Energy Res. 2020, 44, 6651–6659. [Google Scholar] [CrossRef]
- Badini Pourazar, M.; Mohammadi, T.; Jafari Nasr, M.R.; Bakhtiari, O.; Javanbakht, M. Preparation and characterization of poly(vinylidene fluoride)-13X zeolite mixed matrix membranes for lithium ion batteries’ separator with enhanced performance. J. Appl. Polym. Sci. 2020, 137, 49367. [Google Scholar] [CrossRef]
- Liu, Q.; Jiang, W.; Lu, W.; Mei, Y.; He, F.; Zhang, M.; Liu, Y.; Chen, Y.; Peng, J.; Ding, Y. Anisotropic semi-aligned PAN@PVdF-HFP separator for Li-ion batteries. Nanotechnology 2020, 31, 435701. [Google Scholar] [CrossRef]
- Wang, Z.; Chen, J.; Ye, B.; Pang, P.; Ma, Z.; Chen, H.; Nan, J. A pore-controllable polyamine (PAI) layer-coated polyolefin (PE) separator for pouch lithium-ion batteries with enhanced safety. J. Solid State Electrochem. 2020, 24, 843–853. [Google Scholar] [CrossRef]
- Jia, S.; Long, J.; Li, J.; Yang, S.; Huang, K.; Yang, N.; Liang, Y.; Xiao, J. Biomineralized zircon-coated PVDF nanofiber separator for enhancing thermo- and electro-chemical properties of lithium ion batteries. J. Mater. Sci. 2020, 55, 14907–14921. [Google Scholar] [CrossRef]
- Zhang, C.; Li, H.; Wang, S.; Cao, Y.; Yang, H.; Ai, X.; Zhong, F. A polyethylene microsphere-coated separator with rapid thermal shutdown function for lithium-ion batteries. J. Energy Chem. 2020, 44, 33–40. [Google Scholar] [CrossRef]
- Yang, Y.; Huang, C.; Gao, G.; Hu, C.; Luo, L.; Xu, J. Aramid nanofiber/bacterial cellulose composite separators for lithium-ion batteries. Carbohydr. Polym. 2020, 247, 116702. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, J.C.; Reizabal, A.; Correia, D.M.; Fidalgo-Marijuan, A.; Gonçalves, R.; Silva, M.M.; Lanceros-Mendez, S.; Costa, C.M. Lithium-ion battery separator membranes based on poly(L-lactic acid) biopolymer. Mater. Today Energy 2020, 18, 100494. [Google Scholar] [CrossRef]
- Asghar, M.R.; Anwar, M.T.; Xia, G.; Zhang, J. Cellulose/Poly(vinylidene fluoride hexafluoropropylene) composite membrane with titania nanoparticles for lithium-ion batteries. Mater. Chem. Phys. 2020, 252, 123122. [Google Scholar] [CrossRef]
- Reizabal, A.; Gonçalves, R.; Fidalgo-Marijuan, A.; Costa, C.M.; Pérez, L.; Vilas, J.-L.; Lanceros-Mendez, S. Tailoring silk fibroin separator membranes pore size for improving performance of lithium ion batteries. J. Membr. Sci. 2020, 598, 117678. [Google Scholar] [CrossRef]
- Wang, D.; Yu, J.; Duan, G.; Liu, K.; Hou, H. Electrospun polyimide nonwovens with enhanced mechanical and thermal properties by addition of trace plasticizer. J. Mater. Sci. 2020, 55, 5667–5679. [Google Scholar] [CrossRef]
- Li, J.; Yu, J.; Wang, Y.; Zhu, J.; Hu, Z. Intercalated Montmorillonite Reinforced Polyimide Separator Prepared by Solution Blow Spinning for Lithium-Ion Batteries. Ind. Eng. Chem. Res. 2020, 59, 12879–12888. [Google Scholar] [CrossRef]
- Kim, D.; Munakata, H.; Park, J.; Roh, Y.; Jin, D.; Ryou, M.-H.; Kanamura, K.; Lee, Y.M. Hybrid Effect of Micropatterned Lithium Metal and Three Dimensionally Ordered Macroporous Polyimide Separator on the Cycle Performance of Lithium Metal Batteries. ACS Appl. Energy Mater. 2020, 3, 3721–3727. [Google Scholar] [CrossRef]
- Liao, C.; Wang, W.; Wang, J.; Han, L.; Qiu, S.; Song, L.; Gui, Z.; Kan, Y.; Hu, Y. Magnetron sputtering deposition of silicon nitride on polyimide separator for high-temperature lithium-ion batteries. J. Energy Chem. 2021, 56, 1–10. [Google Scholar] [CrossRef]
- Cai, M.; Yuan, D.; Zhang, X.; Pu, Y.; Liu, X.; He, H.; Zhang, L.; Ning, X. Lithium ion battery separator with improved performance via side-by-side bicomponent electrospinning of PVDF-HFP/PI followed by 3D thermal crosslinking. J. Power Sources 2020, 461, 228123. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, B.; Wu, Y.; Chen, J.; Fang, M.; Wang, L.; Wang, L. The effects of polybenzimidazole nanofiber separator on the safety and performance of lithium-ion batteries: Characterization and analysis from the perspective of mechanism. J. Power Sources 2020, 475, 228624. [Google Scholar] [CrossRef]
- Pan, F.; Qiao, L.; Yuan, B.; Duan, C.; Wang, J.; Wu, W.; Lin, Q.; Shi, Y.; Chen, Z.; Jiang, Z. Polydopamine coated poly(m-phenylene isophthalamid) membrane as heat-tolerant separator for lithium-ion batteries. Ionics 2020, 26, 5471–5480. [Google Scholar] [CrossRef]
- Yu, Y.; Xiong, S.; Huang, H.; Zhao, L.; Nie, K.; Chen, S.; Xu, J.; Yin, X.; Wang, H.; Wang, L. Fabrication and application of poly (phenylene sulfide) ultrafine fiber. React. Funct. Polym. 2020, 150, 104539. [Google Scholar] [CrossRef]
- Zhu, C.; Zhang, J.; Xu, J.; Yin, X.; Wu, J.; Chen, S.; Zhu, Z.; Wang, L.; Wang, H. Facile fabrication of cellulose/polyphenylene sulfide composite separator for lithium-ion batteries. Carbohydr. Polym. 2020, 248, 116753. [Google Scholar] [CrossRef]
- Mu, X.; Zhou, X.; Wang, W.; Xiao, Y.; Liao, C.; Longfei, H.; Kan, Y.; Song, L. Design of compressible flame retardant grafted porous organic polymer based separator with high fire safety and good electrochemical properties. Chem. Eng. J. 2021, 405, 126946. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, W.; Zhang, J. A waterborne superLEphilic and thermostable separator based on natural clay nanorods for high-voltage lithium-ion batteries. Mater. Today Energy 2020, 16, 100420. [Google Scholar] [CrossRef]
- Chao, C.-H.; Hsieh, C.-T.; Ke, W.-J.; Lee, L.-W.; Lin, Y.-F.; Liu, H.-W.; Gu, S.; Fu, C.-C.; Juang, R.-S.; Mallick, B.C.; et al. Roll-to-roll atomic layer deposition of titania coating on polymeric separators for lithium ion batteries. J. Power Sources 2021, 482, 228896. [Google Scholar] [CrossRef]
- Qiu, Z.; Yuan, S.; Wang, Z.; Shi, L.; Jo, J.H.; Myung, S.-T.; Zhu, J. Construction of silica-oxygen-borate hybrid networks on Al2O3-coated polyethylene separators realizing multifunction for high-performance lithium ion batteries. J. Power Sources 2020, 472, 228445. [Google Scholar] [CrossRef]
- Sheng, L.; Song, L.; Gong, H.; Pan, J.; Bai, Y.; Song, S.; Liu, G.; Wang, T.; Huang, X.; He, J. Polyethylene separator grafting with polar monomer for enhancing the lithium-ion transport property. J. Power Sources 2020, 479, 228812. [Google Scholar] [CrossRef]
- Pai, J.-Y.; Hsieh, C.-T.; Lee, C.-H.; Wang, J.-A.; Ku, H.-Y.; Huang, C.-L.; Hardwick, L.J.; Hu, C.-C. Engineering of electrospun polyimide separators for electrical double-layer capacitors and lithium-ion cells. J. Power Sources 2021, 482, 229054. [Google Scholar] [CrossRef]
- Li, X.; Chen, S.; Xia, Z.; Li, L.; Yuan, W. High performance of boehmite/polyacrylonitrile composite nanofiber membrane for polymer lithium-ion battery. RSC Adv. 2020, 10, 27492–27501. [Google Scholar] [CrossRef]
- Yusuf, A.; Avvaru, V.S.; Dirican, M.; Changchun, S.; Wang, D.-Y. Low heat yielding electrospun phosphenanthrene oxide loaded polyacrylonitrile composite separators for safer high energy density lithium-ion batteries. Appl. Mater. Today 2020, 20, 100675. [Google Scholar] [CrossRef]
- Al Rai, A.; Stojanovska, E.; Akgul, Y.; Khan, M.M.; Kilic, A.; Yilmaz, S. Fabrication of co-PVDF/modacrylic/SiO2 nanofibrous membrane: Composite separator for safe and high performance lithium-ion batteries. J. Appl. Polym. Sci. 2021, 138, 49835. [Google Scholar] [CrossRef]
- Fu, Q.; Zhang, W.; Muhammad, I.P.; Chen, X.; Zeng, Y.; Wang, B.; Zhang, S. Coaxially electrospun PAN/HCNFs@PVDF/UiO-66 composite separator with high strength and thermal stability for lithium-ion battery. Microporous Mesoporous Mater. 2021, 311, 110724. [Google Scholar] [CrossRef]
- Dwyer, D.B.; Mera, E.; Gudavalli, G.S.; Bernier, W.E.; Dhakal, T.P.; Jones, W.E. Homogeneous titanium hydroxide yields hydroxyl decorated nanofiber separator with improved electrolyte uptake and device capacitance. Mater. Lett. 2021, 284, 129023. [Google Scholar] [CrossRef]
- Liu, L.; Wang, Z.; Xie, Y.; Peng, Y.; Dong, J.; Chow, Y.T.; Zhang, G.; Zhu, L.; Wang, X.; Xu, D. Zirconia/polyethylene terephthalate ceramic fiber paper separator for high-safety lithium-ion battery. Ionics 2020, 26, 6057–6067. [Google Scholar] [CrossRef]
- Zhang, K.; Xiao, W.; Li, X.; Liu, J.; Yan, C. Highly thermostable expanded polytetrafluoroethylene separator with mussel-inspired silica coating for advanced Li-ion batteries. J. Power Sources 2020, 468, 228403. [Google Scholar] [CrossRef]
- Xiao, W.; Song, J.; Huang, L.; Yang, Z.; Qiao, Q. PVA-ZrO2 multilayer composite separator with enhanced electrolyte property and mechanical strength for lithium-ion batteries. Ceram. Int. 2020, 46, 29212–29221. [Google Scholar] [CrossRef]
- Xie, Y.; Pan, Y.; Cai, P. Novel PVA-Based Porous Separators Prepared via Freeze-Drying for Enhancing Performance of Lithium-Ion Batteries. Ind. Eng. Chem. Res. 2020, 59, 15242–15254. [Google Scholar] [CrossRef]
- Karuppiah, C.; Hsieh, Y.-C.; Beshahwured, S.L.; Wu, X.-W.; Wu, S.-H.; Jose, R.; Lue, S.J.; Yang, C.-C. Poly(vinyl alcohol)/Melamine Composite Containing LATP Nanocrystals as a High-Performing Nanofibrous Membrane Separator for High-Power, High-Voltage Lithium-Ion Batteries. ACS Appl. Energy Mater. 2020, 3, 8487–8499. [Google Scholar] [CrossRef]
- Wang, J.; Yuan, B.; Pan, F.; Qiao, L.; Guo, J.; Duan, C.; Wu, W.; Chen, Z.; Su, Y. Nano-silica-decorated Poly(m-Phenylene Isophthalamide) Separator with Enhanced Mechanical and Electrolyte Wetting Properties for Lithium-Ion Batteries. Trans. Tianjin Univ. 2020, 26, 256–264. [Google Scholar] [CrossRef]
- Kim, S.; Kwon, M.-S.; Han, J.H.; Yuk, J.; Lee, J.Y.; Lee, K.T.; Kim, T.-H. Poly(ethylene-co-vinyl acetate)/polyimide/poly(ethylene-co-vinyl acetate) tri-layer porous separator with high conductivity and tailored thermal shutdown function for application in sodium-ion batteries. J. Power Sources 2021, 482, 228907. [Google Scholar] [CrossRef]
- Zhang, F.; Lan, X.; Peng, H.; Hu, X.; Zhao, Q. A “Trojan Horse” Camouflage Strategy for High-Performance Cellulose Paper and Separators. Adv. Funct. Mater. 2020, 30, 2002169. [Google Scholar] [CrossRef]
- Ajkidkarn, P.; Manuspiya, H. Novel bacterial cellulose nanocrystals/polyether block amide microporous membranes as separators for lithium-ion batteries. Int. J. Biol. Macromol. 2020, 164, 3580–3588. [Google Scholar] [CrossRef]
- Sun, X.; Xu, W.; Zhang, X.; Lei, T.; Lee, S.-Y.; Wu, Q. ZIF-67@Cellulose nanofiber hybrid membrane with controlled porosity for use as Li-ion battery separator. J. Energy Chem. 2021, 52, 170–180. [Google Scholar] [CrossRef]
- Li, L.; Li, H.; Wang, Y.; Zheng, S.; Zou, Y.; Ma, Z. Poly(vinylidenefluoride-hexafluoropropylene)/cellulose/carboxylic TiO2 composite separator with high temperature resistance for lithium-ion batteries. Ionics 2020, 26, 4489–4497. [Google Scholar] [CrossRef]
- Lv, D.; Chai, J.; Wang, P.; Zhu, L.; Liu, C.; Nie, S.; Li, B.; Cui, G. Pure cellulose lithium-ion battery separator with tunable pore size and improved working stability by cellulose nanofibrils. Carbohydr. Polym. 2021, 251, 116975. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Luo, J.; Chen, L.; Long, J.; Hu, J.; Meng, L. Effect of fibrillated fiber morphology on properties of paper-based separators for lithium-ion battery applications. J. Power Sources 2021, 482, 228899. [Google Scholar] [CrossRef]
- Chen, Y.; Qiu, L.; Ma, X.; Dong, L.; Jin, Z.; Xia, G.; Du, P.; Xiong, J. Electrospun cellulose polymer nanofiber membrane with flame resistance properties for lithium-ion batteries. Carbohydr. Polym. 2020, 234, 115907. [Google Scholar] [CrossRef] [PubMed]
- Liang, M.; Fu, C.; Xiao, B.; Luo, L.; Wang, Z. A fractal study for the effective electrolyte diffusion through charged porous media. Int. J. Heat Mass Transf. 2019, 137, 365–371. [Google Scholar] [CrossRef]
- Song, J.Y.; Wang, Y.; Wan, C. Review of gel-type polymer electrolytes for lithium-ion batteries. J. Power Sources 1999, 77, 183–197. [Google Scholar] [CrossRef]
- Koerver, R.; Zhang, W.; de Biasi, L.; Schweidler, S.; Kondrakov, A.O.; Kolling, S.; Brezesinski, T.; Hartmann, P.; Zeier, W.G.; Janek, J. Chemo-mechanical expansion of lithium electrode materials—On the route to mechanically optimized all-solid-state batteries. Energy Environ. Sci. 2018, 11, 2142–2158. [Google Scholar] [CrossRef]
- Li, B.; Su, Q.; Yu, L.; Dong, S.; Zhang, M.; Ding, S.; Du, G.; Xu, B. Ultrathin, flexible, and sandwiched structure composite polymer electrolyte membrane for solid-state lithium batteries. J. Membr. Sci. 2021, 618, 118734. [Google Scholar] [CrossRef]
- Lin, Z.; Guo, X.; Yang, Y.; Tang, M.; Wei, Q.; Yu, H. Block copolymer electrolyte with adjustable functional units for solid polymer lithium metal battery. J. Energy Chem. 2021, 52, 67–74. [Google Scholar] [CrossRef]
- Hu, Z.; Zhang, X.; Chen, S. A graphene oxide and ionic liquid assisted anion-immobilized polymer electrolyte with high ionic conductivity for dendrite-free lithium metal batteries. J. Power Sources 2020, 477, 228754. [Google Scholar] [CrossRef]
- Cui, J.; Zhou, Z.; Jia, M.; Chen, X.; Shi, C.; Zhao, N.; Guo, X. Solid Polymer Electrolytes with Flexible Framework of SiO2 Nanofibers for Highly Safe Solid Lithium Batteries. Polymers 2020, 12, 1324. [Google Scholar] [CrossRef]
- Yang, G.; Lehmann, M.L.; Zhao, S.; Li, B.; Ge, S.; Cao, P.-F.; Delnick, F.M.; Sokolov, A.P.; Saito, T.; Nanda, J. Anomalously high elastic modulus of a poly(ethylene oxide)-based composite electrolyte. Energy Storage Mater. 2021, 35, 431–442. [Google Scholar] [CrossRef]
- Gao, L.; Li, J.; Ju, J.; Wang, L.; Yan, J.; Cheng, B.; Kang, W.; Deng, N.; Li, Y. Designing of root-soil-like polyethylene oxide-based composite electrolyte for dendrite-free and long-cycling all-solid-state lithium metal batteries. Chem. Eng. J. 2020, 389, 124478. [Google Scholar] [CrossRef]
- Zhang, B.; Liu, Y.; Pan, X.; Liu, J.; Doyle-Davis, K.; Sun, L.; Liu, J.; Jiao, X.; Jie, J.; Xie, H.; et al. Dendrite-free lithium metal solid battery with a novel polyester based triblock copolymer solid-state electrolyte. Nano Energy 2020, 72, 104690. [Google Scholar] [CrossRef]
- Wang, S.; Li, Q.; Bai, M.; He, J.; Liu, C.; Li, Z.; Liu, X.; Lai, W.-Y.; Zhang, L. A dendrite-suppressed flexible polymer-in-ceramic electrolyte membrane for advanced lithium batteries. Electrochim. Acta 2020, 353, 136604. [Google Scholar] [CrossRef]
- Yang, Y.; Wu, Q.; Wang, D.; Ma, C.; Chen, Z.; Su, Q.; Zhu, C.; Li, C. Ionic liquid enhanced composite solid electrolyte for high-temperature/long-life/dendrite-free lithium metal batteries. J. Membr. Sci. 2020, 612, 118424. [Google Scholar] [CrossRef]
- Li, L.; Shan, Y.; Yang, X. New insights for constructing solid polymer electrolytes with ideal lithium-ion transfer channels by using inorganic filler. Mater. Today Commun. 2021, 26, 101910. [Google Scholar] [CrossRef]
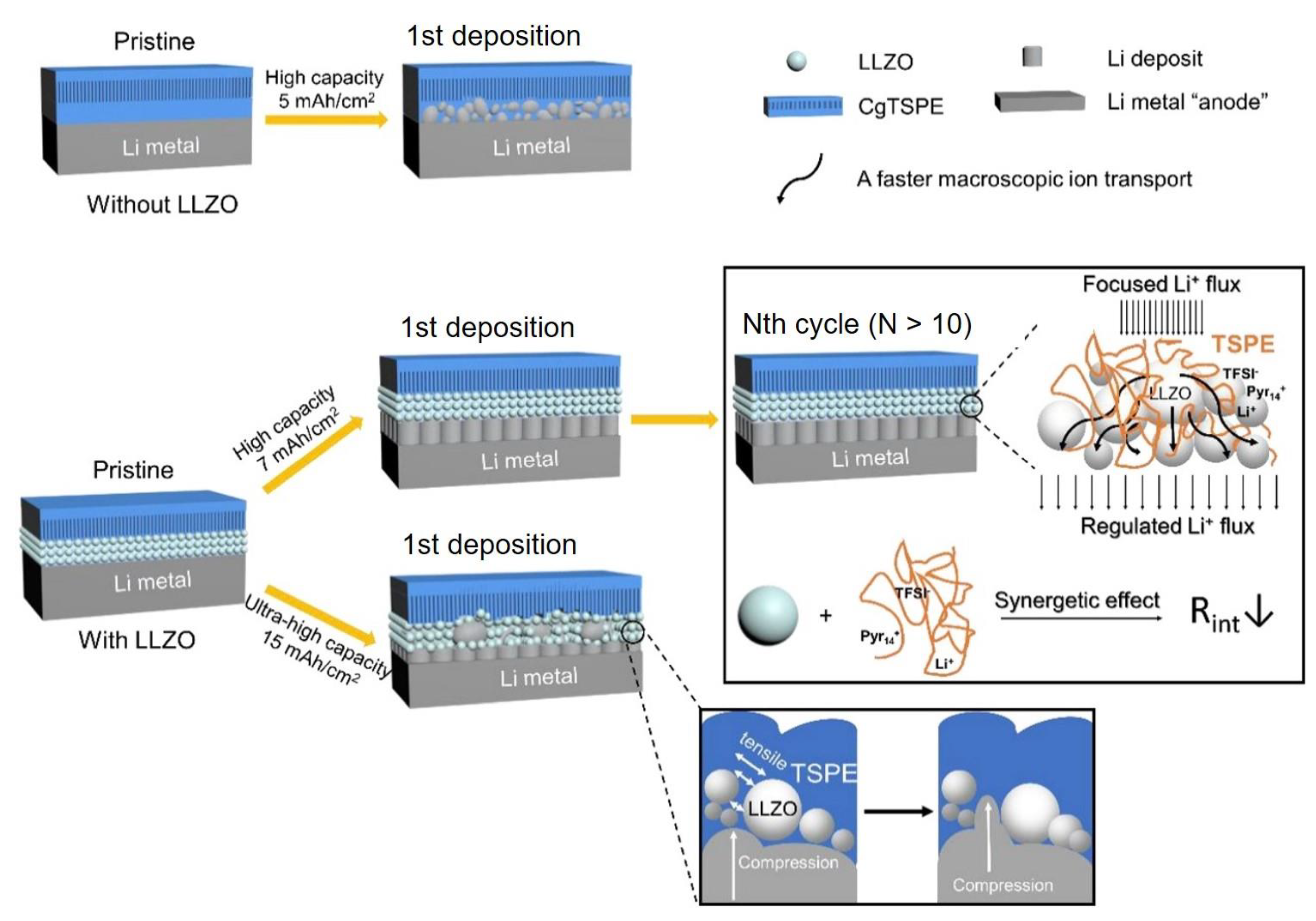
- Zhang, W.; Wang, X.; Zhang, Q.; Wang, L.; Xu, Z.; Li, Y.; Huang, S. Li7La3Zr2O12 Ceramic Nanofiber-Incorporated Solid Polymer Electrolytes for Flexible Lithium Batteries. Acs Appl. Energy Mater. 2020, 3, 5238–5246. [Google Scholar] [CrossRef]
- Angulakshmi, N.; Zhou, Y.; Suriyakumar, S.; Dhanalakshmi, R.B.; Satishrajan, M.; Alwarappan, S.; Alkordi, M.H.; Stephan, A.M. Microporous Metal–Organic Framework (MOF)-Based Composite Polymer Electrolyte (CPE) Mitigating Lithium Dendrite Formation in All-Solid-State-Lithium Batteries. ACS Omega 2020, 5, 7885–7894. [Google Scholar] [CrossRef] [PubMed]
- Fu, F.; Lu, W.; Zheng, Y.; Chen, K.; Sun, C.; Cong, L.; Liu, Y.; Xie, H.; Sun, L. Regulating lithium deposition via bifunctional regular-random cross-linking network solid polymer electrolyte for Li metal batteries. J. Power Sources 2021, 484, 229186. [Google Scholar] [CrossRef]
- Gao, L.; Li, J.; Sarmad, B.; Cheng, B.; Kang, W.; Deng, N. A 3D polyacrylonitrile nanofiber and flexible polydimethylsiloxane macromolecule combined all-solid-state composite electrolyte for efficient lithium metal batteries. Nanoscale 2020, 12, 14279–14289. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Liu, G.; Jiang, Z.; Peng, L.; Xie, J. Zinc bis(2–ethylhexanoate), a homogeneous and bifunctional additive, to improve conductivity and lithium deposition for poly (ethylene oxide) based all-solid-state lithium metal battery. J. Power Sources 2020, 451, 227730. [Google Scholar] [CrossRef]
- Cong, L.; Li, Y.; Lu, W.; Jie, J.; Liu, Y.; Sun, L.; Xie, H. Unlocking the Poly(vinylidene fluoride-co-hexafluoropropylene)/Li10GeP2S12 composite solid-state Electrolytes for Dendrite-Free Li metal batteries assisting with perfluoropolyethers as bifunctional adjuvant. J. Power Sources 2020, 446, 227365. [Google Scholar] [CrossRef]
- Martinez-Ibañez, M.; Sanchez-Diez, E.; Qiao, L.; Zhang, Y.; Judez, X.; Santiago, A.; Aldalur, I.; Carrasco, J.; Zhu, H.; Forsyth, M.; et al. Unprecedented Improvement of Single Li-Ion Conductive Solid Polymer Electrolyte Through Salt Additive. Adv. Funct. Mater. 2020, 30, 2000455. [Google Scholar] [CrossRef]
- Yuan, H.; Luan, J.; Yang, Z.; Zhang, J.; Wu, Y.; Lu, Z.; Liu, H. Single Lithium-Ion Conducting Solid Polymer Electrolyte with Superior Electrochemical Stability and Interfacial Compatibility for Solid-State Lithium Metal Batteries. ACS Appl. Mater. Interfaces 2020, 12, 7249–7256. [Google Scholar] [CrossRef]
- Tang, S.; Zhang, X.; Li, Y.; Tian, J.; Zhao, Y.; Mai, L.; Wang, L.; Cao, Y.-C.; Zhang, W. A fast ionic conductor and stretchable solid electrolyte artificial interphase layer for Li metal protection in lithium batteries. J. Alloys Compd. 2020, 843, 155839. [Google Scholar] [CrossRef]
- Zhou, B.; Yang, M.; Zuo, C.; Chen, G.; He, D.; Zhou, X.; Liu, C.; Xie, X.; Xue, Z. Flexible, Self-Healing, and Fire-Resistant Polymer Electrolytes Fabricated via Photopolymerization for All-Solid-State Lithium Metal Batteries. ACS Macro Lett. 2020, 9, 525–532. [Google Scholar] [CrossRef]
- Tian, X.; Yang, P.; Yi, Y.; Liu, P.; Wang, T.; Shu, C.; Qu, L.; Tang, W.; Zhang, Y.; Li, M.; et al. Self-healing and high stretchable polymer electrolytes based on ionic bonds with high conductivity for lithium batteries. J. Power Sources 2020, 450, 227629. [Google Scholar] [CrossRef]
- Jo, Y.H.; Li, S.; Zuo, C.; Zhang, Y.; Gan, H.; Li, S.; Yu, L.; He, D.; Xie, X.; Xue, Z. Self-Healing Solid Polymer Electrolyte Facilitated by a Dynamic Cross-Linked Polymer Matrix for Lithium-Ion Batteries. Macromolecules 2020, 53, 1024–1032. [Google Scholar] [CrossRef]
- Zeng, L.; Jia, L.; Liu, X.; Zhang, C. A Novel Silicon/Phosphorus Co-Flame Retardant Polymer Electrolyte for High-Safety All-Solid-State Lithium Ion Batteries. Polymers 2020, 12, 2937. [Google Scholar] [CrossRef]
- Tian, Z.; Kim, D. Solid electrolyte membranes prepared from poly(arylene ether sulfone)-g-poly(ethylene glycol) with various functional end groups for lithium-ion battery. J. Membr. Sci. 2021, 621, 119023. [Google Scholar] [CrossRef]
- Lei, Z.; Shen, J.; Zhang, W.; Wang, Q.; Wang, J.; Deng, Y.; Wang, C. Exploring porous zeolitic imidazolate frame work-8 (ZIF-8) as an efficient filler for high-performance poly(ethyleneoxide)-based solid polymer electrolytes. Nano Res. 2020, 13, 2259–2267. [Google Scholar] [CrossRef]
- Kim, J.I.; Choi, Y.G.; Ahn, Y.; Kim, D.; Park, J.H. Optimized ion-conductive pathway in UV-cured solid polymer electrolytes for all-solid lithium/sodium ion batteries. J. Membr. Sci. 2021, 619, 118771. [Google Scholar] [CrossRef]
- Yang, J.; Shao, L.; Wang, X.; Yang, Y.; Tian, Z.; Chen, W.; Zhang, G.; Shen, C. Effect of intermolecular interactions on the performance of UiO-66-laden solid composite polymer electrolytes. J. Alloys Compd. 2020, 845, 155179. [Google Scholar] [CrossRef]
- Tan, J.; Ao, X.; Dai, A.; Yuan, Y.; Zhuo, H.; Lu, H.; Zhuang, L.; Ke, Y.; Su, C.; Peng, X.; et al. Polycation ionic liquid tailored PEO-based solid polymer electrolytes for high temperature lithium metal batteries. Energy Storage Mater. 2020, 33, 173–180. [Google Scholar] [CrossRef]
- Shen, X.; Li, R.; Ma, H.; Peng, L.; Huang, B.; Zhang, P.; Zhao, J. Enhancing Li+ transport kinetics of PEO-based polymer electrolyte with mesoporous silica-derived fillers for lithium-ion batteries. Solid State Ion. 2020, 354, 115412. [Google Scholar] [CrossRef]
- Sha, Y.; Dong, T.; Zhao, Q.; Zheng, H.; Wen, X.; Chen, S.; Zhang, S. A new strategy for enhancing the room temperature conductivity of solid-state electrolyte by using a polymeric ionic liquid. Ionics 2020, 26, 4803–4812. [Google Scholar] [CrossRef]
- Sathyanathan, T.; Pugazhendhi Sugumaran, C. Enhancement of Ionic Conductivity and Dielectric Permittivity for PMMA-Based Polymer Electrolyte with BaTiO3 NanoFiller. Int. J. Nanosci. 2020, 19, 1950035. [Google Scholar] [CrossRef]
- Serra, J.P.; Pinto, R.S.; Barbosa, J.C.; Correia, D.M.; Gonçalves, R.; Silva, M.M.; Lanceros-Mendez, S.; Costa, C.M. Ionic liquid based Fluoropolymer solid electrolytes for Lithium-ion batteries. Sustain. Mater. Technol. 2020, 25, e00176. [Google Scholar] [CrossRef]
- Wang, H.; Cui, X.; Zhang, C.; Gao, H.; Du, W.; Chen, Y. Promotion of Ionic Conductivity of PEO-Based Solid Electrolyte Using Ultrasonic Vibration. Polymers 2020, 12, 1889. [Google Scholar] [CrossRef] [PubMed]
- Cai, D.; Wang, D.; Chen, Y.; Zhang, S.; Wang, X.; Xia, X.; Tu, J. A highly ion-conductive three-dimensional LLZAO-PEO/LiTFSI solid electrolyte for high-performance solid-state batteries. Chem. Eng. J. 2020, 394, 124993. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, K.; Liu, K.; Zhou, L.; Ma, C.; Du, Y. Imidazole containing solid polymer electrolyte for lithium ion conduction and the effects of two lithium salts. Electrochim. Acta 2020, 351, 136342. [Google Scholar] [CrossRef]
- Leo Edward, M.; Dharanibalaji, K.C.; Kumar, K.T.; Chandrabose, A.R.S.; Shanmugharaj, A.M.; Jaisankar, V. Preparation and characterisation of chitosan extracted from shrimp shell (Penaeus monodon) and chitosan-based blended solid polymer electrolyte for lithium-ion batteries. Polym. Bull. 2021. [Google Scholar] [CrossRef]
- Chitra, R.; Sathya, P.; Selvasekarapandian, S.; Monisha, S.; Moniha, V.; Meyvel, S. Synthesis and characterization of iota-carrageenan solid biopolymer electrolytes for electrochemical applications. Ionics 2019, 25, 2147–2157. [Google Scholar] [CrossRef]
- Shilpa, R.; Saratha, R. Biodegradable pectin- guar gum blend electrolyte for solid state lithium ion batteries. AIP Conf. Proc. 2020, 2270, 100006. [Google Scholar]
- Park, J.H.; Jung, S.; Song, Y.H.; Aluru, N.R.; Kim, T.; Lee, S.B.; Choi, U.H.; Lee, J. Water-Assisted Increase of Ionic Conductivity of Lithium Poly(acrylic acid)-Based Aqueous Polymer Electrolyte. ACS Appl. Energy Mater. 2020, 3, 10119–10130. [Google Scholar] [CrossRef]
- Chen, H.; Liu, J.; Zhou, X.; Ji, H.; Liu, S.; Wang, M.; Qian, T.; Yan, C. Rapid leakage responsive and self-healing Li-metal batteries. Chem. Eng. J. 2021, 404, 126470. [Google Scholar] [CrossRef]
- Lizundia, E.; Kundu, D. Advances in Natural Biopolymer-Based Electrolytes and Separators for Battery Applications. Adv. Funct. Mater. 2021, 31, 2005646. [Google Scholar] [CrossRef]

| Technology | Specific Energy (Wh/kg) | Number of Cycles | Efficiency (%) | Voltage (V) | Ref. |
|---|---|---|---|---|---|
| Lithium-sulfur | 500 | ~500 | 85 | 3 | [40] |
| Lithium-ion | 100–265 | 1000–2000 | 99.9 | 3.6 | [41] |
| Lithium-air | 3860 | 700 | 65 | 2.91 | [42] |
| Potassium-ion | 120–170 | ~4000 | >90 | 2.0–4.3 | [43] |
| Magnesium-ion | 77 | ~2000 | ~95 | 1.1 | [44] |
| Sodium-ion | 85–125 | ~500 | >90 | 2.7–3.2 | [45] |
| Cathode Active Material | Crystal System/Space Group [Point Group] | Specific Capacity/mAh·g−1 | Typical Voltage Range/V |
|---|---|---|---|
| LiCoO2 | Orthorhombic/R3m [C3V] | 274 | 2.5–4.45 |
| LiFePO4 | Orthorhombic/Pnma [D2h] | 170 | 2.5–4.2 |
| LiMn2O4 | Cubic/Fdm [Oh] | 148 | 3.0–4.3 |
| LiNiO2 | Trigonal/R3m [C3V] | 275 | 3.0–4.3 |
| LiNi1-xCoxO2 (0.2 ≤ x ≤ 0.5) | Rhombohedral/R3m [C3V] | ~275 | 3.5–4.3 |
| LiNi1/3Mn1/3Co1/3O2 | Rhombohedral/R3m [C3V] | 278 | 2.3–4.3 |
| LiNi0.5Mn1.5O2 | Trigonal/R3m [C3V] | 147 | 3.5–4.9 |
| Materials | Electrolyte Solution | Porosity and Uptake (%) | Conductivity (mS·cm−1) and Capacity (mAh·g−1) | Main Goal/Achievement | Ref. |
|---|---|---|---|---|---|
| Polyimide (PI) with poly(amic acid) (PAA) | 1.0 M LiPF6 (ethylene carbonate (EC)/ diethyl carbonate (DEC) (1:1 by weight)) with 5% of fluoroethylene carbonate (FEC) | 89.1/- | 1.79/1.930 mAh | Improved mechanical strength | [184] |
| Polyimide (PI) | 1.0 M LiPF6 (EC/dimethyl carbonate (DMC) (1:1 in vol.)) | -/- | -/- | Improved thermal stability | [185] |
| PE with phenolic resin (AF) | 1mol L−1 LiPF6 (DMC/EC (1:1 in vol.)) | 57/228 | 0.6/119 | Improved thermal stability and electrochemical properties | [186] |
| Untreated Al2O3/PE | 1.0 M LiPF6 (EC/DEC (1:1 by weight)) with 5% of Fluoroethylene carbonate (FEC) | -/- | 0.39/140@0.2C | Good wettability, high thermal stability, and good electrochemical performance | [187] |
| Polyethyleneimine (PEI)/dopamine coating layer in PP separator. | 1.0 M LiPF6 (EC/DMC (1:1 in vol.)) | -/144 | 0.58/128 | High electrolyte uptake | [188] |
| PVDF containing titanium dioxide (TiO2) and graphene oxide (GO) | Commercial electrolyte based LiPF6 | 86.50/494 | 4.87/- | High electrolyte uptake | [189] |
| Polyimide (PI) with ZSM-5 zeolite filler | 1.0 M LiPF6 (EC/DMC (1:1 in vol.)) | 61/260 | 1.04/133@2C | Enhanced wettability and electrolyte uptake | [190] |
| Poly(aryl ether sulfone) (PES) and poly(vinylidene fluoride) (PVDF) | 1.0 M LiPF6 (EC/ethylmethyl carbonate (EMC)/DMC (1:1:1 in vol.)) | -/595 | 1.69/162.8 | Enhanced wettability and high ionic conductivity | [191] |
| Poly(vinylidene fluoridehExafluoropropyl-ene) (PVDF-HFP)/poly-mphenyleneisophthalamide (PMIA) | 1.0 M LiPF6 (EC/DEC/EMC (1:1:1 in vol.)) | 94.28/~900 | -/- | Good electrolyte affinity and enhanced interfacial compatibility | [192] |
| PE with ammonium persulfate (APS) coating | 1.0 M LiPF6 (EC/EMC (3:7 in vol.)) with 2 wt % vinylene carbonate as an additive | -/- | 0.36/~170 | High lithium-ion migration and ionic conductivity | [193,194] |
| PVDF/13X zeolite particles | 1.0 M LiPF6 (EC/DEC/DMC (1:1:1 in vol.)) | 76/475 | ~1/144.14 | Excellent ionic conductivity | [195] |
| PAN@PVdF-HFP | - | -/- | 1.2/170@0.1C | Excellent cycling performance, good rate capability | [196] |
| PE with controllable polyamine (PAI) layer | 1.0 M LiPF6 carbonate solution | 60/- | -/- | Enhanced safety | [197] |
| PVDF coated with ZnO | - | 85.1/352 | 2.3/148@1C | High safety in high temperature. | [198] |
| Thin layer of low-density polyethylene microspheres onto a commercial porous PP | 1.0 M LiPF6 (EC/DMC/EMC (1:1:1 in vol.)) | -/- | 0.30/158 | Rapid thermal shutdown at elevated temperature (≈110 °C) | [199] |
| Aramid nanofiber/bacterial cellulose | 1.0 M LiPF6 (EC/DMC/DEC (1:1:1 in vol.)) | 83.9/- | 12.54/157 | Excellent tensile strength and ionic conductivity. | [200] |
| Poly (L-lactic acid) (PLLA) | 1.0 M LiPF6 (EC/DMC (1:1In vol.)) | ≈72/350 | 1.6/93@1C | Environmentally friendly separator | [201] |
| Cellulose/PVDF-HFP with TiO2 | 1.0 M LiPF6 (EC/DMC (1:1 in vol.)) | 86/403 | 1.68/103.8@8C | Excellent thermal stability and high ion conductivity | [202] |
| Silk fibroin | 1.0 M LiPF6 (EC/DMC (1:1 in vol.)) | 86/350 | 2.2/131.3 @8C | Environmentally friendly separator | [203] |
| Polymer Matrix | Fillers | Method | Ionic Conductivity (S·cm−1) | Discharge Capacity (mAh·g−1) | Main Features | Ref. |
|---|---|---|---|---|---|---|
| PEO | Carbon quantum dots, LiTFSI | Doctor blade | 2.2 × 10−4 (25 °C) | 160.4 (C/10) | Lithium dendrite suppression | [30] |
| PEO | GO, [Bmim][DCA] | Solvent casting | 1 × 10−4 (25 °C) | 156.2 (C/10) | Lithium dendrite suppression | [243] |
| PEO | LiTFSI SiO2 | Solvent casting | 9.32 × 10−5 (30 °C) | 166.9 (C/10) | Lithium dendrite suppression | [244] |
| PEO | Nonwoven glass fiber, LiTFSI | Solvent casting | 1.2 × 10−4 (20 °C) | 128 (C/10) | Lithium dendrite suppression | [245] |
| PEO, PVDF | LiTFSI | Solvent casting | 2.46 × 10−9 (30 °C) | 157.5 (C/5) | Lithium dendrite suppression | [246] |
| PCL-PPC-PCL | LiTFSI | Solvent casting | 3 × 10−5 (30 °C) | 142 (C/20) | Lithium dendrite suppression | [247] |
| PVDF | LiTFSI, LLZTO | Solvent casting | 1.16 × 10−3 (80 °C) | 151 (C/5) | Lithium dendrite suppression | [248] |
| PVDF | LiTFSI, Pyr13TFSI | Solvent casting | 1.23 × 10−3 (25 °C) | 158.2 (1C) | Lithium dendrite suppression | [249] |
| PVDF-CA | Montmorillonite, LiTFSI | Solvent casting | 3.40 × 10−4 (25 °C) | 112 (C/2) | Lithium dendrite suppression | [250] |
| PVDF-HFP | LLZO, IL | Solvent casting | 6.3 × 10−3 (20 °C) | 164 (C/2) | Lithium dendrite suppression | [251] |
| PEO | LiTFSI, UiO-66-NH2@SiO2 | Hot pressing | 8.1 × 10−6 (60 °C) | 151 (C/10) | Lithium dendrite suppression | [252] |
| TMPTA-TEGDME-PEO | LiTFSI | UV curing | 4.36 × 10−4 (30 °C) | 157.8 (C/10) | Lithium dendrite suppression | [253] |
| PEO | LiTFSI, Pyr14TFSI, LLZO | UV curing | 5.0 × 10−4 (60 °C) | - | Lithium dendrite suppression | [29] |
| PVDF | LiClO4, LLTO | Tape casting | 4.7 × 10−4 (25 °C) | 139 (C/5) | Lithium dendrite suppression, interfacial contact | [241] |
| PAN nanofibers, PDMS, PEO | LiTFSI | Solvent casting | 1.2 × 10−3 (60 °C) | 151.7 (C/5) | Lithium dendrite suppression, interfacial contact | [254] |
| PEO | LiTFSI Zn(BEH2) | Solvent casting | 1.1 × 10−5 (30 °C) | 145 (C/10) | Lithium dendrite suppression, interfacial contact | [255] |
| PVDF-HFP | LiTFSI, LGPS | Solvent casting | 1.8 × 10−4 (30 °C) | 158 (C/20) | Lithium dendrite suppression, interfacial contact | [256] |
| PEG | LiTFSI, HDIt | Cross-linked copolymerization | 6.51 × 10−5 (25 °C) | 162 (C/10) | Interfacial contact | [242] |
| PEO | LiFSI, LiPSTFSI | Hot pressing | 3.7 × 10−5 (70 °C) | 150 (C/20) | Interfacial contact | [257] |
| PEO | LiPCSI | Solvent casting | 7.33 × 10−5 (60 °C) | 141 (C/10) | Interfacial contact | [258] |
| PEO | Li3N | Doctor blade | - | 160 (C/10) | Self-healing | [259] |
| HCP-UPyMA, PEGMA | LiTFSI | UV copolymerization | 8.95 × 10−5 (30 °C) | - | Self-healing | [260] |
| PVT | [EMIM][TFSI], LiTFSI | Solvent casting | 1.26 × 10−4 (25 °C) | 145 | Self-healing | [261] |
| PEGA | LiTFSI, Bis(2-methacryloyloxyethyl) Disulfide, 1,2-Bis (ureidoethylenemethacrylate) Hexamethylene | RAFT polymerization | 7.28 × 10−6 (30 °C) | 140.5 (C/10) | Self-healing | [262] |
| PEG600 | Phosphorous and silicon-containing monomers, LiTFSI | Solvent casting | 2.98 × 10−5 (25 °C) | 142.0 (C/10) | Flame retardancy | [263] |
| PAES-g-PEG | PYR14-TFSI, LiTFSI | Solvent casting | 8.9 × 10−4 (40 °C) | 138 (C/10) | Battery stability | [264] |
| PEO | ZIF-8 | Solvent casting | 2.2 × 10−5 (30 °C) | 111 (C/2) | Battery stability | [265] |
| ETPTA-PVDF-HFP | - | UV curing | 9 × 10−4 (25 °C) | 150 (C/5) | Ionic conduction | [266] |
| PEO | UiO-66, LiClO4 | Solvent casting | 4.8 × 10−5 (25 °C) | 148 (C/10) | Ionic conduction | [267] |
| PEO | HACC-TFSI, LiTFSI | Solvent casting | 1.77 × 10−5 (30 °C) | 161.3 (C/5) | Ionic conduction | [268] |
| PEO | LiTFSO, Mesoporous silica | Solvent casting | 4.3 × 10−4 (60 °C) | 150.3 (C/10) | Ionic conduction | [269] |
| PVDF-HFP | LiTFSI, Pyr13TFSI, P(MMA-co-VIm(1O2)) (TFSI) | Solvent casting | 5.1 × 10−4 (25 °C) | 102 (C/10) | Ionic conduction | [270] |
| PMMA | BaTiO3, LiPF6 | Solvent casting | 3.9 × 10−4 (70 °C) | - | Ionic conduction | [271] |
| PVDH-HFP | [Bmim][SCN] | Doctor blade | 1.5 × 10−4 (25 °C) | 148 (C/8) | Ionic conduction | [272] |
| PEO | LiTFSI | Ultrasonic treatment, solvent casting | 3.2 × 10−4 (25 °C) | - | Ionic conduction | [273] |
| PEO | LiTFSI, LLZAO | Solvent casting | 2.51 × 10−4 (25 °C) | 165.9 (C/5) | Ionic conduction | [274] |
| PVO | LiTFSI | Solvent casting | 1.36 × 10−6 (25 °C) | - | Ionic conduction | [275] |
| Chitosan, PEG | LiClO4 | Solvent casting | 4.56 × 10−4 (25 °C) | - | Environmentally friendly | [276] |
| I-Carrageenan | LiCl | Solvent casting | 5.33 × 10−3 (25 °C) | - | Environmentally friendly | [277] |
| Pectin, Guar gum | LiTFSI | Solvent casting | 1.59 × 10−4 (25 °C) | - | Environmentally friendly | [278] |
| PAA | Silica nanoparticles | Free radical polymerization | 1.29 × 10−2 (25 °C) | - | Environmentally friendly | [279] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barbosa, J.C.; Gonçalves, R.; Costa, C.M.; Lanceros-Mendez, S. Recent Advances on Materials for Lithium-Ion Batteries. Energies 2021, 14, 3145. https://doi.org/10.3390/en14113145
Barbosa JC, Gonçalves R, Costa CM, Lanceros-Mendez S. Recent Advances on Materials for Lithium-Ion Batteries. Energies. 2021; 14(11):3145. https://doi.org/10.3390/en14113145
Chicago/Turabian StyleBarbosa, João C., Renato Gonçalves, Carlos M. Costa, and Senentxu Lanceros-Mendez. 2021. "Recent Advances on Materials for Lithium-Ion Batteries" Energies 14, no. 11: 3145. https://doi.org/10.3390/en14113145
APA StyleBarbosa, J. C., Gonçalves, R., Costa, C. M., & Lanceros-Mendez, S. (2021). Recent Advances on Materials for Lithium-Ion Batteries. Energies, 14(11), 3145. https://doi.org/10.3390/en14113145









