Effects of Concentration Variations on the Performance and Microbial Community in Microbial Fuel Cell Using Swine Wastewater
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
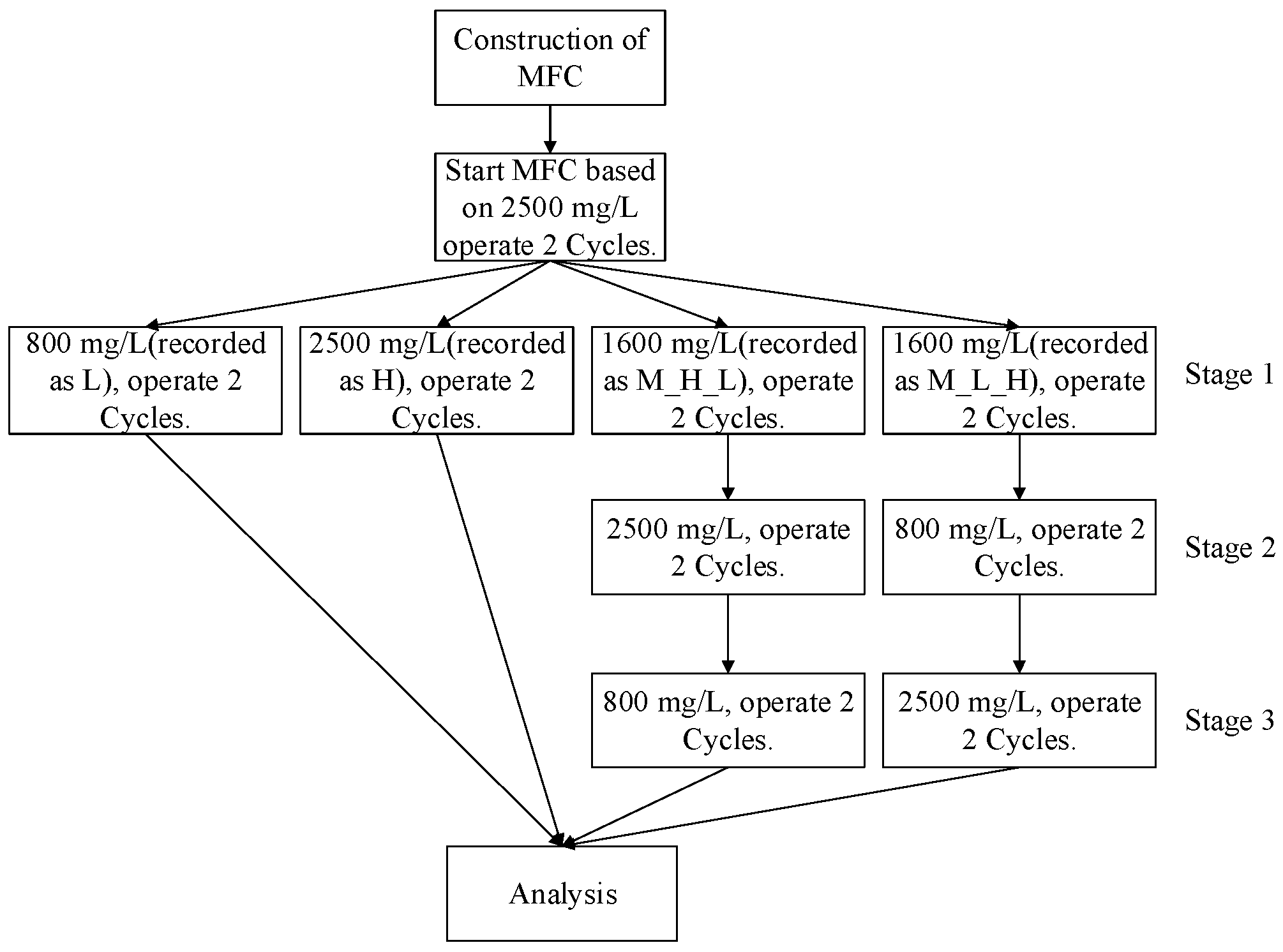
2.2. Construction and Operation
2.3. Statistical Analysis
3. Results
3.1. MFC Performance
3.1.1. Power Generation Performance
3.1.2. Decontamination Performance
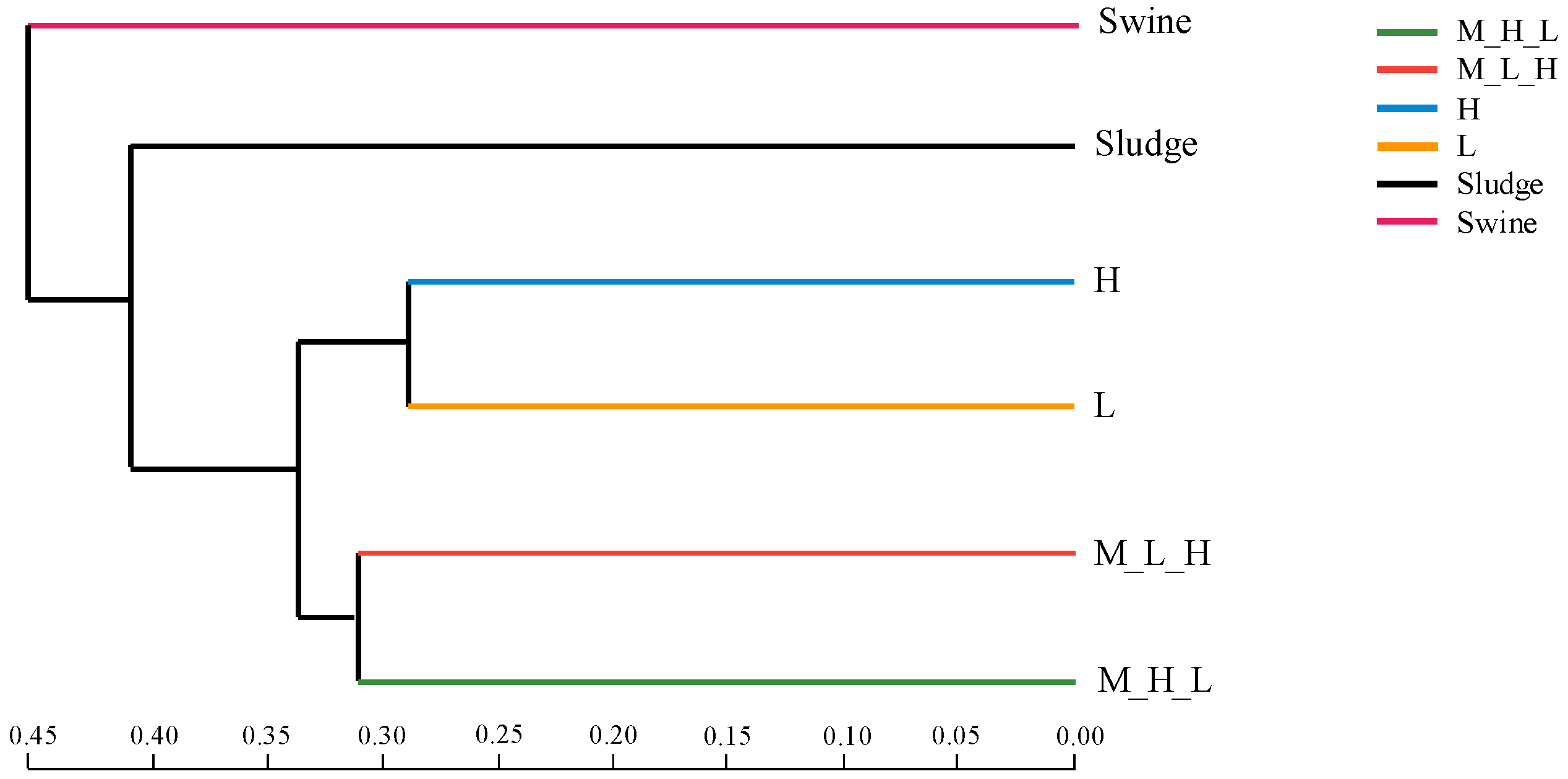
3.2. Microbial Community Diversity
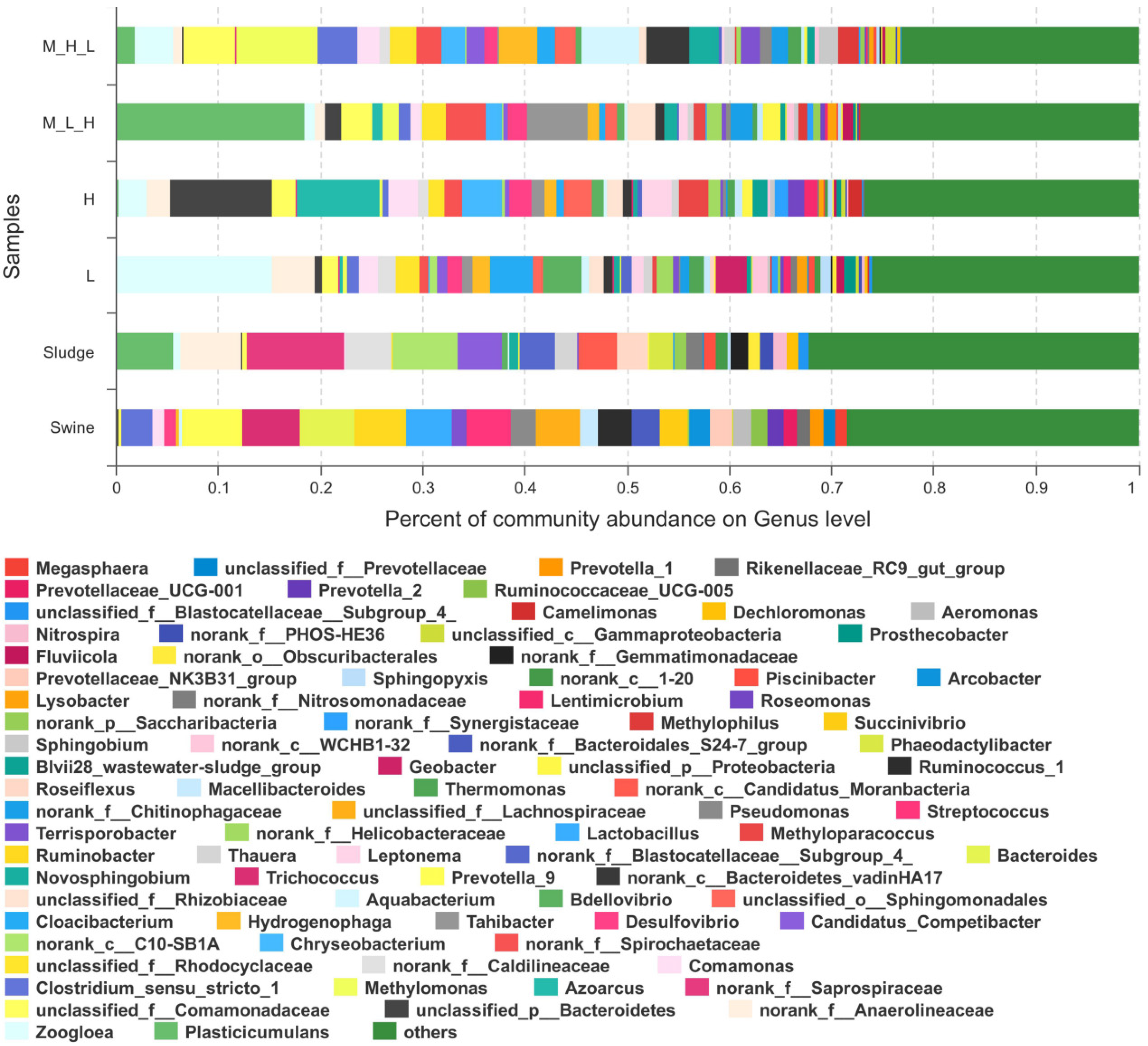
3.3. Microbial Community Structure
4. Conclusions
5. Patents
Author Contributions
Funding
Conflicts of Interest
References
- Zhang, Y.; Zhao, Y.Y.; Zhou, M.H. A photosynthetic algal microbial fuel cell for treating swine wastewater. Environ. Sci. Pollut. Res. 2019, 26, 6182–6190. [Google Scholar] [CrossRef] [PubMed]
- Cheng, D.L.; Ngo, H.H.; Guo, W.S. Problematic effects of antibiotics on anaerobic treatment processes in swine wastewater. Bioresour. Technol. 2018, 263, 642–653. [Google Scholar] [CrossRef] [PubMed]
- Cheng, D.L.; Ngo, H.H.; Guo, W.S. Microalgae biomass from swine wastewater and its conversion to bioenergy. Bioresour. Technol. 2019, 275, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.; Jiang, Z.H.; Lay, C.H. Electricity generation from swine wastewater in microbial fuel cell: Hydraulic reaction time effect. Int. J. Hydrog. Energy 2016, 41, 21820–21826. [Google Scholar] [CrossRef]
- Li, X.H.; Zhu, N.W.; Li, C. Electricity generation and treatment of swine wastewater. Chin. J. Environ. Eng. 2012, 6, 2189–2194. [Google Scholar]
- Doug, A.; Costas, T.; Hamilton, C.Y. Assessment of the Effects of Flow Rate and Ionic Strength on the Performance of an Air-Cathode Microbial Fuel Cell Using Electrochemical Impedance Spectroscopy. Energies 2010, 3, 672–675. [Google Scholar]
- Kim, T.; Yeo, J.; Yang, Y. Boosting voltage without electrochemical degradation using energy-harvesting circuits and power management system-coupled multiple microbial fuel cells. J. Power Sources 2019, 410, 171–178. [Google Scholar] [CrossRef]
- Li, H.; Tian, Y.; Zuo, W. Electricity generation from food wastes and characteristics of organic matters in microbial fuel cell. Bioresour. Technol. 2016, 205, 104–110. [Google Scholar] [CrossRef]
- Escalante-Estrada, V.E.; Garzón-Zúñiga, M.A.; Valle-Cervantes, S. Swine Wastewater Treatment for Small Farms by a New Anaerobic-Aerobic Biofiltration Technology. Water Air Soil Pollut. 2019, 230, 145. [Google Scholar] [CrossRef]
- Goto, Y.; Yoshida, N. Scaling up Microbial Fuel Cells for Treating Swine Wastewater. Water 2019, 11, 1803. [Google Scholar] [CrossRef]
- Gao, C.Y.; Wu, W.M.; Zhao, Y.H. Response of microbial fuel cell anodic microbial communities to substrate switch of lactate-propionate-lactate. Acta Microbiol. Sin. 2015, 55, 1495–1504. [Google Scholar]
- Kondaveeti, S.; Patel, S.K.S.; Pagolu, R. Conversion of simulated biogas to electricity: Sequential operation of methanotrophic reactor effluents in microbial fuel cell. Energy 2019, 189, 116309. [Google Scholar] [CrossRef]
- Ma, J.; Ni, H.J.; Su, D.Y. Bioelectricity generation from pig farm wastewater in microbial fuel cell using carbon brush as electrode. Int. J. Hydrog. Energy 2016, 41, 16191–16195. [Google Scholar] [CrossRef]
- Nandy, A.; Sharma, M.; Venkatesan, S. Comparative Evaluation of Coated and Non-Coated Carbon Electrodes in a Microbial Fuel Cell for Treatment of Municipal Sludge. Energies 2019, 12, 1034. [Google Scholar] [CrossRef]
- Lan, L.H.; Li, J.; Feng, Q. Enhanced current production of the anode modified by microalgae derived nitrogen-rich biocarbon for microbial fuel cells. Int. J. Hydrog. Energy 2019, 45, 3833–3839. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhu, Y.Y.; Wang, X.X. Research Progress of Active Components of the Eletrocatalysts for Direct Enthanol Fuel Cell. J. Nantong Univ. 2017, 16, 58–63. [Google Scholar]
- Oberauner, L.; Zachow, C.; Lackner, S. The ignored diversity: Complex bacterial communities in intensive care units revealed by 16S pyrosequencing. Sci. Rep. 2013, 3, 1413. [Google Scholar] [CrossRef]
- Ma, X.; Feng, C.; Zhou, W. Municipal sludge-derived carbon anode with nitrogen- and oxygen-containing functional groups for high-performance microbial fuel cells. J. Power Sources 2016, 307, 105–111. [Google Scholar] [CrossRef]
- Schilirò, T.; Tommasi, T.; Armato, C. The study of electrochemically active planktonic microbes in microbial fuel cells in relation to different carbon-based anode materials. Energy 2016, 106, 277–284. [Google Scholar] [CrossRef]
- Mei, X.; Xing, D.; Yang, Y. Adaptation of microbial community of the anode biofilm in microbial fuel cells to temperature. Bioelectrochemistry 2017, 117, 29–33. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, L.; Hu, Y. Bacterial community shift and incurred performance in response to in situ microbial self-assembly graphene and polarity reversion in microbial fuel cell. Bioresour. Technol. 2017, 241, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Song, X.; Wang, Y. Bioenergy generation and rhizodegradation as affected by microbial community distribution in a coupled constructed wetland-microbial fuel cell system associated with three macrophytes. Sci. Total Environ. 2017, 607, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Tamis, J.; Luzkov, K.; Jiang, Y. Enrichment of Plasticicumulans acidivorans at pilot-scale for PHA production on industrial wastewater. J. Biotechnol. 2014, 192, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.Y.; Ngo, H.H.; Guo, W.S. Microbial fuel cell for nutrient recovery and electricity generation from municipal wastewater under different ammonium concentrations. Bioresour. Technol. 2019, 292, 121992. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Wei, J.; Peng, L. Microbial community analysis in biocathode microbial fuel cells packed with different materials. AMB Express 2012, 2, 21. [Google Scholar] [CrossRef]
- Zhu, J.P.; Wang, J.; Zhang, T.P. Phylogenetic Diversity of Bacterial and Archaeal Communities in Anode biofilm of Sediment Microbial Fuel Cells. Acta Sci. Circumstantiae 2016, 36, 4017–4024. [Google Scholar]
- Miran, W.; Jang, J.; Nawaz, M. Mixed sulfate-reducing bacteria-enriched microbial fuel cells for the treatment of wastewater containing copper. Chemosphere 2017, 189, 134. [Google Scholar] [CrossRef]
- Pandey, P.; Shinde, V.N.; Deopurkar, R.L. Recent advances in the use of different substrates in microbial fuel cells toward wastewater treatment and simultaneous energy recovery. J. Appl. Energy 2016, 168, 706–723. [Google Scholar] [CrossRef]




| Group | Influent COD (mg/L) | COD Removal Rate in Phase 1 (%) | COD Removal Rate in Phase 2 (%) | COD Removal Rate in Phase 3 (%) | COD Removal Rate of Effluent (%) |
|---|---|---|---|---|---|
| M_H_L | 1600 | 60.56 | 45.2 | 14 | 42.38 |
| M_L_H | 1600 | 66.56 | 27.25 | 63.08 | 72.4 |
| H | 2500 | - | - | - | 64.36 |
| L | 800 | - | - | - | 13.75 |
| Sample | Seqs | OTU | Chao | Heip | Shannon | Coverage |
|---|---|---|---|---|---|---|
| M_H_L | 35,458 | 594 | 695 (658,754) | 0.2124 | 4.84 (4.82,4.86) | 0.9942 |
| M_L_H | 33,177 | 569 | 627 (604,667) | 0.1915 | 4.69 (4.66,4.71) | 0.9952 |
| H | 35,632 | 664 | 783 (739,853) | 0.1920 | 4.85 (4.83,4.87) | 0.9955 |
| L | 35,340 | 692 | 798 (758,862) | 0.2118 | 4.99 (4.97,5.02) | 0.9951 |
| Sludge | 42,403 | 599 | 674 (641,732) | 0.2331 | 4.94 (4.92,4.96) | 0.9971 |
| Swine | 44,636 | 464 | 488 (475,514) | 0.2829 | 4.87 (4.85,4.88) | 0.9981 |
| Phylum | Abundance (Above 0.1%) | SEM | p-Value | |||||
|---|---|---|---|---|---|---|---|---|
| Sludge | Swine | M_H_L | M_L_H | H | L | |||
| Acidobacteria | 6.69% | 0.02% | 0.74% | 0.40% | 1.02% | 2.78% | 1.03% | 0.07 |
| Actinobacteria | 2.32% | 0.83% | 1.92% | 2.31% | 2.17% | 2.23% | 0.23% | 0.02 |
| Bacteroidetes | 17.35% | 32.25% | 15.96% | 15.89% | 25.63% | 15.59% | 2.83% | 0.17 |
| Chloroflexi | 23.62% | 0.00% | 2.43% | 1.50% | 4.54% | 9.65% | 3.60% | 0.24 |
| Cyanobacteria | 1.31% | 0.01% | 0.17% | 0.92% | 0.23% | 0.67% | 0.21% | 0.01 |
| Firmicutes | 0.97% | 44.56% | 7.81% | 4.44% | 3.43% | 3.83% | 6.80% | 0.01 |
| Gemmatimonadetes | 1.81% | 0.00% | 0.11% | 0.01% | 0.07% | 0.13% | 0.29% | 0.02 |
| Ignavibacteriae | 1.43% | 0.00% | 0.13% | 0.03% | 0.15% | 0.33% | 0.22% | 0.01 |
| Nitrospirae | 1.26% | 0.00% | 0.02% | 0.01% | 0.16% | 0.31% | 0.19% | 0.01 |
| Parcubacteria | 4.82% | 0.02% | 0.00% | 0.01% | 0.00% | 0.00% | 0.80% | 0.05 |
| Proteobacteria | 33.17% | 19.91% | 66.07% | 66.55% | 53.14% | 58.81% | 7.76% | 0.33 |
| Saccharibacteria | 1.36% | 0.04% | 0.34% | 0.66% | 0.15% | 0.34% | 0.19% | 0.01 |
| Spirochaetae | 0.33% | 0.73% | 3.54% | 5.86% | 6.11% | 2.53% | 1.04% | <0.01 |
| Synergistetes | 0.05% | 0.06% | 0.29% | 0.64% | 1.35% | 0.55% | 0.20% | <0.01 |
| Verrucomicrobia | 0.05% | 0.14% | 0.03% | 0.42% | 0.51% | 1.31% | 0.20% | <0.01 |
| others | 3.46% | 1.43% | 0.44% | 0.35% | 1.34% | 0.94% | 0.46% | 0.03 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ni, H.; Wang, K.; Lv, S.; Wang, X.; Zhuo, L.; Zhang, J. Effects of Concentration Variations on the Performance and Microbial Community in Microbial Fuel Cell Using Swine Wastewater. Energies 2020, 13, 2231. https://doi.org/10.3390/en13092231
Ni H, Wang K, Lv S, Wang X, Zhuo L, Zhang J. Effects of Concentration Variations on the Performance and Microbial Community in Microbial Fuel Cell Using Swine Wastewater. Energies. 2020; 13(9):2231. https://doi.org/10.3390/en13092231
Chicago/Turabian StyleNi, Hongjun, Kaixuan Wang, Shuaishuai Lv, Xingxing Wang, Lu Zhuo, and Jiaqiao Zhang. 2020. "Effects of Concentration Variations on the Performance and Microbial Community in Microbial Fuel Cell Using Swine Wastewater" Energies 13, no. 9: 2231. https://doi.org/10.3390/en13092231
APA StyleNi, H., Wang, K., Lv, S., Wang, X., Zhuo, L., & Zhang, J. (2020). Effects of Concentration Variations on the Performance and Microbial Community in Microbial Fuel Cell Using Swine Wastewater. Energies, 13(9), 2231. https://doi.org/10.3390/en13092231





