Characteristic Properties of Alternative Biomass Fuels
Abstract
1. Introduction
- Pollutants from incomplete combustion: CO, CxHy, tar, soot, unburnt hydrocarbon particles, hydrogen, and incompletely oxygenated nitrogen compounds (HCN, NH3, N2O).
- Pollutants from complete combustion: nitrogen oxides (NO, NO2) and CO2.
- Pollutants from trace elements of impurities: incombustible dust particles, sulfur, chlorine compounds, and trace metals (Cu, Pb, Zn, Cd) [5].
2. Materials and Methods
3. Results and Discussion
4. Conclusions
- Some materials examined in this study had not been explored and analyzed yet, where some materials showed great potential for becoming a renewable and sustainable energy source for low-power boilers. A large number of these materials are of waste origin or surpluses from agriculture, and their combustion not only generates energy but also greatly facilitates the solution of disposal or possible waste management problems.
- The moisture content of the analyzed biomass significantly affected the treatment of the material itself and the amount of heat released from a unit amount of the selected material since the lower heating value of the material is reduced by a higher water content of the matrix.
- The use of biomass as fuel also affects the amount of ash formed from combustion. If the material forms a large amount of ash, it is more difficult to remove the ash from the boiler body and to quickly fill the ashpan, which is disadvantageous from a user’s point of view. For small boiler bodies, it is, therefore, preferable to use biomass with low ash and low ballast contents as the energy source. This implies that materials with a high ash content (e.g., waste sludge) should be combined with, for example, a readily available woody mass that forms a minimal amount of ash.
- The volatile content and lower heating value also have a significant effect on fuel quality. These are closely related, as a higher volatile content will increase the fuel higher heating value.
- Fuel of a plant origin shows the influence of its growth location. The composition of the soil in which the plant has grown significantly affects the number of elements and their representation in all its parts. For example, increased nitrogen in plants is caused by the use of fertilizers, which directly affects the increased release of nitrogen oxides in the combustion process.
Author Contributions
Funding
Conflicts of Interest
References
- Vorotinskienė, L.; Paulauskas, R. Parameters influencing wet biofuel drying during combustion in grate furnaces. Fuel 2020, 265, 117013. [Google Scholar] [CrossRef]
- Matúš, M.; Križan, P. The effect of papermaking sludge as an additive to biomass pellets on the final quality of the fuel. Fuel 2018, 219, 196–204. [Google Scholar] [CrossRef]
- Hrdlička, J.; Skopec, P. Emission factors of gaseous pollutants from small scale combustion of biofuels. Fuel 2016, 165, 68–74. [Google Scholar] [CrossRef]
- Greinert, A.; Mrówczyńska, M. The use of plant biomass pellets for energy production by combustion in dedicated furnaces. Energies 2020, 13, 463. [Google Scholar] [CrossRef]
- Ochodek, T.; Koloničný, J.; Janásek, P. Potenciál Biomasy, Druhy, Bilance a Vlastnosti Paliv z Biomasy; Vysoká škola báňská-Technická univerzita Ostrava: Ostrava, Czech Republic, 2006. [Google Scholar]
- Petříková, V.; Punčochář, M. Biomasa—Alternativní Palivo z Hlediska Chemického Složení. Available online: https://biom.cz/cz/odborne-clanky/biomasa-alternativni-palivo-z-hlediska-chemickeho-slozeni (accessed on 19 November 2019).
- Bešenić, T.; Mikulčić, H. Numerical modellign of emissions of nitrogen oxides in solid fuel combustion. J. Environ. Manag. 2018, 215, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Elbl, P.; Baláš, M.; Vavříková, P.; Lisý, M.; Milčák, P. Gaseous Emissions and Solid Particles from the Combustion of Biomass Pellets in 25kW Automatic Boiler. In Proceedings of the 27th European Biomass Conference and Exhibition, Lisbon, Portugal, 27–30 May 2019; pp. 742–748. [Google Scholar]
- Chlor a Anorganické Sloučeniny. Available online: https://www.irz.cz/repository/latky/chlor_a_anorganicke_slouceniny.pdf (accessed on 8 January 2020).
- Pospíšil, J.; Lisý, M.; Špiláček, M. Optimalization of Afterburner Channel in Biomass Boiler Using CFD Analysis. Acta Polytech. 2016, 56, 379–387. [Google Scholar] [CrossRef]
- Lisý, M.; Pospíšil, J.; Štelcl, O.; Špiláček, M. Optimization of Secondary Air Distribution in Biomass Boiler by CFD Analysis. Appl. Mechan. Mater. 2016, 832, 231–237. [Google Scholar] [CrossRef]
- Carroll, J.; Finnan, J. Emissions and efficiencies from the combustion of agricultural feedstock pellets using a small scale tilting grate boiler. Biosyst. Eng. 2013, 115, 50–55. [Google Scholar] [CrossRef]
- Lamberg, H.; Tissari, J. Fione particle and gaseous emissions from a small-scale boiler fueled by pellets of various raw materials. Energy Fuels 2013, 27, 7044–7053. [Google Scholar] [CrossRef]
- Tissari, J.; Sippula, O.; Kouki, J.; Vuorio, K.; Jokiniemi, J. Fine particle and gas emissions from the combustion oa agricultural fuels fired in a 20 kW burner. 2008, 22, 2033–2042. Energy Fuels 2008, 22, 2033–2042. [Google Scholar] [CrossRef]
- Díaz-Ramírez, M.; Boman, C.; Sebastián, F.; Royo, J.; Xiong, S.; Bostrom, D. Ash characterisation and transformation behavior of the fixed-bed combustion of novel crops: Poplar, brassica, and cassava fuels. Energy Fuels 2012, 26, 3218–3229. [Google Scholar] [CrossRef]
- ČSN EN ISO 18 134-1. Tuhá Biopaliva—Stanovení Obsahu Vody—Metoda Sušení v Sušárně—Část 1: Celková Aoda—Referenční Metoda; UNMZ: Praha, Czech Republic, 2016. [Google Scholar]
- ČSN EN ISO 18 134-2. Tuhá Biopaliva—Stanovení Obsahu Vody—Metoda Sušení v Sušárně—Část 2: Celková Voda—Zjednodušená Metoda; UNMZ: Praha, Czech Republic, 2017. [Google Scholar]
- ČSN EN ISO 18 134-3. Tuhá Biopaliva—Stanovení Obsahu Vody—Metoda Sušení v Sušárně—Část 3: Obsah Vody v Analytickém Vzorku Pro Obecný Rozbor; UNMZ: Praha, Czech Republic, 2016. [Google Scholar]
- ČSN EN ISO 18 122. Tuhá Biopaliva—Stanovení Obsahu Aopela; UNMZ: Praha, Czech Republic, 2016. [Google Scholar]
- ČSN EN ISO 18 123. Tuhá Biopaliva—Stanovení Obsahu Prchavé Hořlaviny; UNMZ: Praha, Czech Republic, 2016. [Google Scholar]
- Elementar Analysensysteme GmbH. [manuál] Návod k obsluze Elementární Analyzátor Vario MACRO Cube; Operating Instructions; Elementar Analysensysteme GmbH: Langenselbold, Germany, 2009. [Google Scholar]
- Rédr, M.; Příhoda, M. Základy Tepelné Techniky; SNTL: Praha, Czech Republic, 1991; Volume 677. [Google Scholar]
- ČSN EN ISO 18 125. Tuhá Biopaliva—Stanovení Spalného Tepla a Aýhřevnosti; UNMZ: Praha, Czech Republic, 2019. [Google Scholar]
- Pastorek, Z.; Kára, J.; Jevič, P. Biomasa: Obnovitelný Adroj Energie; FCC Public: Praha, Czech Republic, 2004. [Google Scholar]
- Effect of Moisture Content, Forest Research. Available online: https://www.forestresearch.gov.uk/tools-and-resources/biomass-energyresources/fuel/woodfuel-production-and-supply/woodfuel-processing/dryingbiomass/effect-of-moisture-content/ (accessed on 25 June 2019).
- Baláš, M. Kotle a Aýměníky Tepla; Akademické Nakladatelství CERM: Brno, Czech Republic, 2013. [Google Scholar]
- Barbanera, M.; Cotana, F. Co-combustion performance and kinetic study of solid digestate with gasification biochar. Renew. Energy 2018, 121, 597–605. [Google Scholar] [CrossRef]
- Şensöz, S.; Angin, D. Pyrrolysis of safflower (Charthamus tinctorius L) seed press cake in a fixed-beed reactor: Part 2, structural characterisation of pyrolysis bio-oils. Bioresour. Technol. 2008, 99, 5498–5504. [Google Scholar] [CrossRef]
- Arromdeee, P.; Kuprianov, V. A comparative study on combustion of sunflower shells in bubbling and swirling fluidised-bed combustors with a cone-shaped bed. Chem. Eng. Proc. 2012, 62, 26–38. [Google Scholar] [CrossRef]
- Wang, C.H.; Wang, X. the thermal behavior and kinetics of co-combustion between sewage sludge and wheat straw. Fuel Proc. Tech. 2019, 189, 1–14. [Google Scholar] [CrossRef]
- Juszczak, M.; Lossy, K. Pollutant emission from a heat station supplied with agriculture biomass and wood pellet mixture. Chem. Process Eng. 2012, 33, 233–234. [Google Scholar] [CrossRef]
- Harvex, R. Potential Use of Combinable Crop Biomass As Fuel for Small Heating Boilers. 2007. Available online: https://www.researchgate.net/publication/237659583_Potential_use_of_combinable_crop_biomass_as_fuel_for_small_heating_boilers (accessed on 4 May 2019).
- Vassilev, S.V.; Baxter, D.; Vassileva, C.G. An Overview of the Behaviour of Biomass during Combustion: Part II, Ash Fusion and Ash Formation Mechanisms of Biomass Types. 2019. Available online: https://www.sciencedirect.com/science/article/pii/S0016236113008533 (accessed on 2 July 2019).
| Sample | Water Content (wt%) | Dry Matter Content (wt%) | Standard Deviation | Confidence Interval |
|---|---|---|---|---|
| Digestate | 8.43 | 91.57 | 0.07 | 0.08 |
| Softwood pellets (spruce) | 7.67 | 92.33 | 0.04 | 0.04 |
| Hardwood pellets (beech) | 7.84 | 92.16 | 0.07 | 0.08 |
| Composite wood | 9.49 | 90.51 | 0.05 | 0.06 |
| Energo compost | 9.35 | 90.65 | 0.24 | 0.27 |
| Rape straw | 10.62 | 89.38 | 0.08 | 0.09 |
| Wheat straw pellet | 7.16 | 92.84 | 0.02 | 0.02 |
| Hay | 7.96 | 92.04 | 0.08 | 0.09 |
| Straw 60% + sludge 40% | 3.86 | 96.11 | 0.03 | 0.03 |
| Straw 70% + sludge 30% | 3.82 | 96.18 | 0.02 | 0.02 |
| Straw 80% + sludge 20% | 4.39 | 95.61 | 0.04 | 0.04 |
| Straw 90% + sludge 10% | 4.79 | 95.21 | 0.09 | 0.1 |
| Sunflower—peel | 7.71 | 92.29 | 0.05 | 0.06 |
| Sunflower—after the oil press | 6.09 | 93.91 | 0.06 | 0.07 |
| Sunflower—whole plant | 10.58 | 89.42 | 0.32 | 0.36 |
| Mix—seeds rape, sunflower, mustard, husks | 11.92 | 88.08 | 0.05 | 0.06 |
| Mustard—seed | 5.88 | 94.12 | 0.17 | 0.19 |
| Spruce sawdust + digestate | 6.44 | 93.56 | 0.09 | 0.10 |
| Safflower—seed | 5.41 | 94.59 | 0.09 | 0.10 |
| Safflower—peel | 5.49 | 94.51 | 0.09 | 0.10 |
| Safflower—after the oil press | 7.08 | 92.92 | 0.12 | 0.13 |
| Amaranth | 6.69 | 93.31 | 0.02 | 0.02 |
| Flax—waste | 5.42 | 94.58 | 0.08 | 0.09 |
| Crambe abyssinica | 5.49 | 94.51 | 0.04 | 0.04 |
| Camelina—seed | 5.95 | 94.05 | 0.01 | 0.01 |
| Camelina—after the oil press | 7.10 | 92.90 | 0.08 | 0.09 |
| Spelt—waste | 8.06 | 91.94 | 0.04 | 0.04 |
| Cocoa—peel | 7.34 | 92.66 | 0.03 | 0.03 |
| Sorrel pellet (whole plant) | 8.64 | 91.36 | 0.05 | 0.05 |
| Rye straw | 7.69 | 92.31 | 0.12 | 0.13 |
| Quinoa—waste | 8.43 | 91.57 | 0.03 | 0.03 |
| Sample | Ash Content in the Raw Sample (wt%) | Ash Content in the Anhydrous Sample (wt%) | Loss on Annealing the Raw Sample (wt%) | Loss on Annealing the Anhydrous Sample (wt%) | Ballast Ratio in the Raw Sample (wt%) |
|---|---|---|---|---|---|
| Digestate | 10.36 | 11.31 | 89.64 | 87.65 | 18.79 |
| Softwood pellets (spruce) | 2.77 | 3.01 | 97.23 | 96.99 | 10.44 |
| Hardwood pellets (beech) | 2.25 | 2.44 | 97.75 | 97.56 | 10.09 |
| Composite wood | 0.30 | 0.33 | 99.70 | 99.67 | 9.79 |
| Energo compost | 19.92 | 21.97 | 80.08 | 75.76 | 29.27 |
| Rape straw | 3.82 | 4.27 | 96.18 | 95.23 | 14.44 |
| Wheat straw pellet | 6.16 | 6.64 | 93.84 | 93.36 | 13.32 |
| Hay | 4.83 | 5.25 | 95.17 | 94.30 | 12.79 |
| Straw 60% + sludge 40% | 27.93 | 29.05 | 72.07 | 69.77 | 31.79 |
| Straw 70% + sludge 30% | 25.07 | 26.07 | 74.93 | 72.90 | 28.89 |
| Straw 80% + sludge 20% | 17.24 | 18.03 | 82.76 | 81.14 | 21.63 |
| Straw 90% + sludge 10% | 10.24 | 10.75 | 89.76 | 88.71 | 15.03 |
| Sunflower — peel | 3.92 | 4.20 | 96.08 | 95.50 | 10.60 |
| Sunflower— fter the oil press | 5.67 | 6.04 | 94.33 | 93.96 | 11.76 |
| Sunflower—whole plant | 3.93 | 4.39 | 96.07 | 95.10 | 14.51 |
| Mix—seeds rape, sunflower, mustard, husks | 6.98 | 7.93 | 93.02 | 91.00 | 18.90 |
| Mustard—seed | 14.68 | 15.58 | 85.32 | 84.42 | 20.56 |
| Spruce sawdust + digestate | 2.53 | 2.70 | 97.47 | 97.11 | 8.97 |
| Safflower—seed | 6.60 | 7.00 | 93.40 | 93.00 | 12.01 |
| Safflower—peel | 2.69 | 2.97 | 97.31 | 97.03 | 8.18 |
| Safflower–after the oil press | 3.68 | 3.96 | 96.32 | 96.04 | 10.76 |
| Amaranth—whole plant | 7.13 | 7.70 | 92.87 | 92.30 | 13.82 |
| Flax—waste | 17.05 | 17.90 | 82.95 | 82.10 | 22.47 |
| Crambe abyssinica | 5.82 | 6.16 | 94.18 | 93.84 | 11.31 |
| Camelina—seed | 12.57 | 13.36 | 87.43 | 86.64 | 18.52 |
| Camelina—after the oil press | 10.09 | 10.87 | 89.92 | 89.14 | 17.18 |
| Spelt—waste | 4.43 | 4.82 | 95.57 | 95.18 | 12.49 |
| Cocoa—peel | 5.92 | 6.39 | 94.08 | 93.10 | 13.26 |
| Sorrel pellet (whole plant) | 4.73 | 5.18 | 95.27 | 94.33 | 13.37 |
| Rye straw | 12.51 | 13.67 | 87.49 | 86.33 | 20.19 |
| Quinoa—waste | 4.76 | 5.20 | 95.24 | 94.80 | 13.19 |
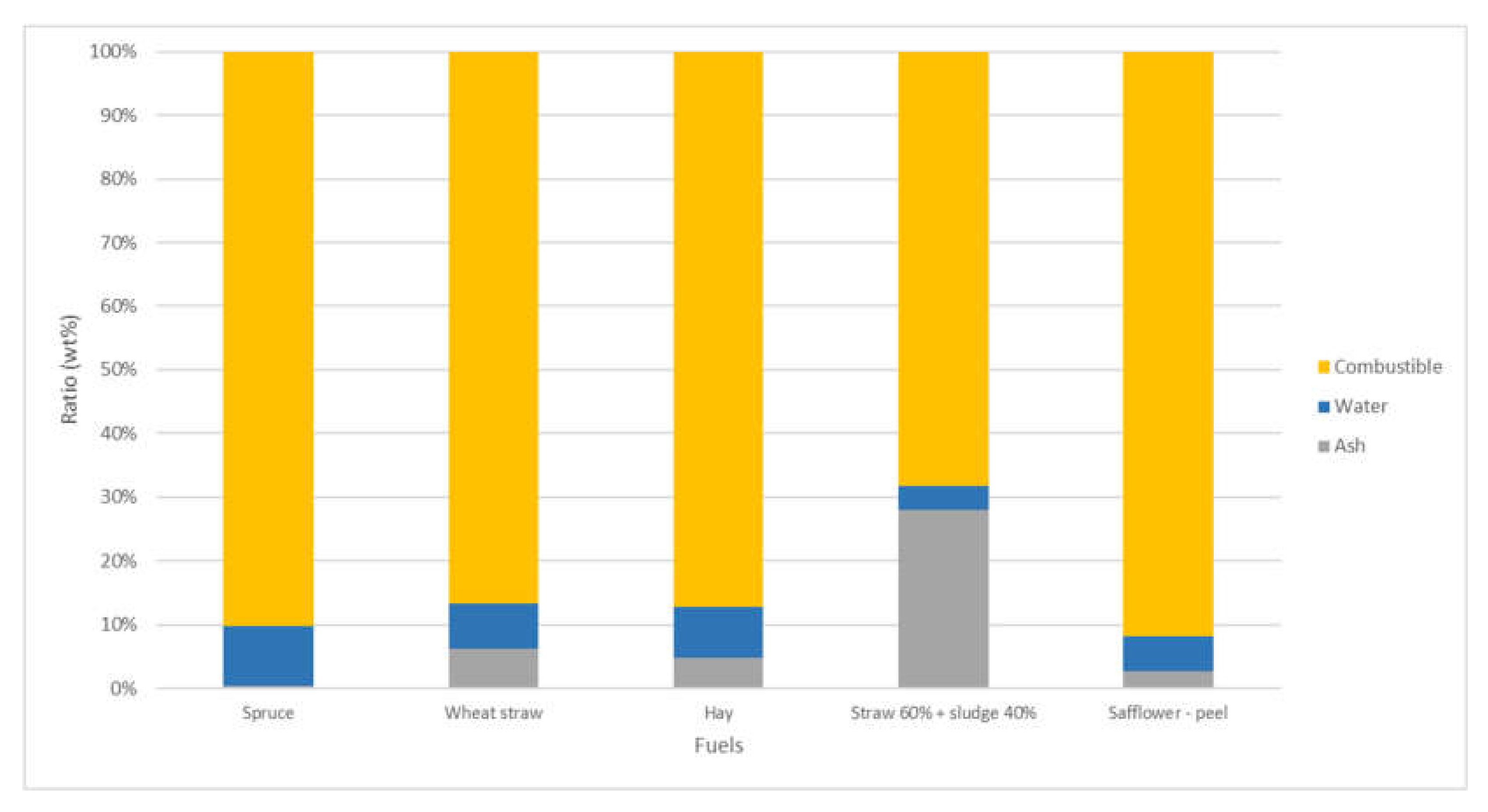
| Proximate Analysis | Spruce | Wheat Straw Pellets | Hay | Straw:Sludge 60:40 | Safflower—Peel | |
|---|---|---|---|---|---|---|
| Combustible | (wt%) | 90.21 | 86.68 | 87.21 | 68.21 | 91.82 |
| Water | (wt%) | 9.49 | 7.16 | 7.96 | 3.86 | 5.49 |
| Ash | (wt%) | 0.3 | 6.16 | 4.83 | 27.93 | 2.69 |
| Ratio of Combustible | Wood | Straw Pellets | Safflower—Peel |
|---|---|---|---|
| Combustible (wt%) | 90.21 | 86.68 | 91.82 |
| Volatile content (wt%) | 75.45 | 71.37 | 79.15 |
| Solids (wt%) | 14.76 | 15.31 | 12.67 |
| Sample | Volatile Content in Raw Sample (wt%) | Volatile Content in Anhydrous Sample (wt%) |
|---|---|---|
| Softwood pellets (spruce) | 76.44 | 82.79 |
| Hardwood pellets (beech) | 76.34 | 82.84 |
| Composite wood | 75.45 | 83.36 |
| Wheat straw pellet | 71.37 | 76.88 |
| Hay | 78.39 | 89.99 |
| Sunflower—peel | 47.49 | 51.45 |
| Sunflower—after the oil press | 75.73 | 80.64 |
| Mustard—seed | 78.16 | 83.05 |
| Safflower—seed | 73.38 | 71.86 |
| Safflower—peel | 79.15 | 78.85 |
| Safflower—after the oil press | 74.34 | 80.00 |
| Amaranth—whole plant | 73.58 | 78.85 |
| Flax—waste | 72.41 | 76.56 |
| Crambe abyssinica | 78.82 | 83.40 |
| Camelina—seed | 80.36 | 85.45 |
| Camelina—after the oil press | 74.15 | 79.82 |
| Spelt—waste | 81.30 | 88.42 |
| Rye straw | 77.02 | 83.43 |
| Quinoa—waste | 74.77 | 81.65 |
| Sample | Raw (Original) Sample—Elements (wt%) | ||||
|---|---|---|---|---|---|
| Digestate | 1.91 | 41.68 | 5.45 | 0.04 | 39.60 |
| Softwood pellets (spruce) | 0.43 | 46.65 | 6.37 | 0.00 | 43.79 |
| Hardwood pellets (beech) | 0.60 | 46.33 | 6.17 | 0.00 | 44.65 |
| Composite wood | 0.80 | 48.30 | 6.08 | 0.00 | 44.50 |
| Energo compost | 2.13 | 38.22 | 4.79 | 0.33 | 32.56 |
| Rape straw | 0.69 | 41.72 | 6.10 | 0.02 | 47.02 |
| Wheat straw pellet | 0.76 | 43.81 | 6.09 | 0.11 | 43.07 |
| Hay | 0.68 | 42.20 | 5.98 | 0.05 | 45.84 |
| Straw 60% + sludge 40% | 0.40 | 36.04 | 4.23 | 0.01 | 30.27 |
| Straw 70% + sludge 30% | 0.44 | 37.19 | 4.36 | 0.00 | 31.93 |
| Straw 80% + sludge 20% | 0.67 | 39.10 | 4.83 | 0.00 | 37.37 |
| Straw 90% + sludge 10% | 0.39 | 41.88 | 5.52 | 0.00 | 41.46 |
| Sunflower—peel | 0.80 | 46.48 | 5.96 | 0.04 | 42.51 |
| Sunflower—after the oil press | 3.67 | 50.85 | 7.97 | 0.20 | 31.63 |
| Sunflower—whole plant | 0.90 | 45.24 | 5.87 | 0.03 | 43.57 |
| Mix—seeds rape, sunflower, mustard, husks | 2.12 | 40.32 | 5.86 | 0.16 | 43.58 |
| Mustard—seed | 5.02 | 52.37 | 8.04 | 1.08 | 18.80 |
| Spruce sawdust + digestate | 0.61 | 46.23 | 6.12 | 0.00 | 43.34 |
| Safflower—seed | 2.29 | 52.55 | 7.52 | 0.01 | 31.02 |
| Safflower—peel | 1.67 | 50.82 | 7.19 | 0.01 | 37.61 |
| Safflower—after the oil press | 2.70 | 48.14 | 6.87 | 0.04 | 38.57 |
| Amaranth—whole plant | 0.89 | 40.16 | 5.71 | 0.09 | 46.03 |
| Flax—waste | 3.41 | 46.78 | 6.75 | 0.17 | 25.83 |
| Crambe abyssinica | 3.13 | 54.48 | 8.28 | 0.67 | 27.61 |
| Camelina—seed | 4.66 | 54.81 | 8.45 | 0.66 | 18.06 |
| Camelina—after the oil press | 6.17 | 47.21 | 7.41 | 0.90 | 28.23 |
| Spelt—waste | 0.68 | 44.98 | 6.05 | 0.19 | 43.67 |
| Cocoa—peel | 2.39 | 45.49 | 6.19 | 0.11 | 39.43 |
| Sorrel pellet (whole plant) | 1.31 | 43.28 | 5.86 | 0.09 | 44.29 |
| Rye straw | 2.81 | 41.74 | 6.79 | 0.05 | 36.10 |
| Quinoa—waste | 2.87 | 42.99 | 6.84 | 0.18 | 42.34 |
| Sample | Combustible—Elements (wt%) | ||||
|---|---|---|---|---|---|
| Digestate | 2.38 | 51.94 | 5.62 | 0.05 | 40.02 |
| Softwood pellets (spruce) | 0.48 | 52.08 | 6.15 | 0.00 | 41.28 |
| Hardwood pellets (beech) | 0.66 | 51.53 | 5.88 | 0.00 | 41.92 |
| Composite wood | 0.85 | 51.17 | 5.82 | 0.00 | 42.16 |
| Energo compost | 3.10 | 55.65 | 5.45 | 0.48 | 35.32 |
| Rape straw | 0.81 | 49.12 | 5.78 | 0.02 | 44.26 |
| Wheat straw pellet | 0.88 | 50.54 | 6.10 | 0.12 | 42.35 |
| Hay | 0.78 | 48.62 | 5.86 | 0.06 | 44.67 |
| Straw 60% + sludge 40% | 0.60 | 53.72 | 5.66 | 0.01 | 40.01 |
| Straw 70% + sludge 30% | 0.63 | 53.05 | 5.61 | 0.00 | 40.71 |
| Straw 80% + sludge 20% | 0.86 | 50.40 | 5.59 | 0.00 | 43.14 |
| Straw 90% + sludge 10% | 0.46 | 49.59 | 5.90 | 0.00 | 44.05 |
| Sunflower—peel | 0.90 | 52.16 | 5.85 | 0.04 | 41.05 |
| Sunflower—after the oil press | 4.16 | 57.63 | 8.26 | 0.23 | 29.72 |
| Sunflower—whole plant | 1.06 | 53.20 | 5.51 | 0.04 | 40.19 |
| Mix—seeds rape, sunflower, mustard, husks | 2.65 | 50.32 | 5.65 | 0.20 | 41.18 |
| Mustard—seed | 6.32 | 65.93 | 9.30 | 1.36 | 17.09 |
| Spruce sawdust + digestate | 0.68 | 51.45 | 6.01 | 0.00 | 41.87 |
| Safflower—seed | 2.60 | 59.73 | 7.86 | 0.02 | 29.79 |
| Safflower—peel | 1.82 | 55.35 | 7.16 | 0.01 | 35.65 |
| Safflower—after the oil press | 3.03 | 53.94 | 6.81 | 0.04 | 36.17 |
| Amaranth—whole plant | 1.03 | 46.60 | 5.76 | 0.10 | 46.52 |
| Flax—waste | 4.40 | 60.34 | 7.92 | 0.22 | 27.11 |
| Crambe abyssinica | 3.53 | 61.43 | 8.64 | 0.76 | 25.64 |
| Camelina—seed | 5.78 | 67.94 | 9.64 | 0.82 | 15.83 |
| Camelina – after the oil press | 7.45 | 57.00 | 7.98 | 1.08 | 26.49 |
| Spelt—waste | 0.78 | 51.40 | 5.89 | 0.21 | 41.72 |
| Cocoa—peel | 2.77 | 52.73 | 6.22 | 0.13 | 38.15 |
| Sorrel pellet (whole plant) | 1.52 | 50.21 | 5.68 | 0.10 | 42.48 |
| Rye straw | 3.52 | 52.30 | 7.43 | 0.07 | 36.68 |
| Quinoa—waste | 3.30 | 49.47 | 6.80 | 0.21 | 40.22 |
| Sample | Dry Matter—Elements (wt%) | ||||
|---|---|---|---|---|---|
| Digestate | 2.27 | 46.06 | 4.98 | 0.04 | 35.49 |
| Softwood pellets (spruce) | 0.47 | 50.64 | 5.98 | 0.00 | 40.14 |
| Hardwood pellets (beech) | 0.65 | 50.37 | 5.75 | 0.00 | 40.98 |
| Composite wood | 0.85 | 51.00 | 5.80 | 0.00 | 42.02 |
| Energo compost | 2.42 | 43.42 | 4.25 | 0.37 | 27.56 |
| Rape straw | 0.78 | 47.03 | 5.54 | 0.02 | 42.37 |
| Wheat straw pellet | 0.83 | 47.42 | 5.73 | 0.12 | 39.75 |
| Hay | 0.74 | 46.07 | 5.56 | 0.05 | 42.33 |
| Straw 60% + sludge 40% | 0.42 | 38.11 | 4.02 | 0.01 | 28.39 |
| Straw 70% + sludge 30% | 0.46 | 39.22 | 4.15 | 0.00 | 30.10 |
| Straw 80% + sludge 20% | 0.71 | 41.31 | 4.58 | 0.00 | 35.37 |
| Straw 90% + sludge 10% | 0.41 | 44.26 | 5.27 | 0.00 | 39.32 |
| Sunflower—peel | 0.86 | 49.97 | 5.60 | 0.04 | 39.32 |
| Sunflower—after the oil press | 2.72 | 37.68 | 5.40 | 0.15 | 19.43 |
| Sunflower—whole plant | 1.01 | 50.87 | 5.27 | 0.03 | 38.43 |
| Mix—seeds rape, sunflower, mustard, husks | 2.44 | 46.33 | 5.20 | 0.18 | 37.91 |
| Mustard—seed | 5.39 | 56.25 | 7.93 | 1.16 | 14.58 |
| Spruce sawdust + digestate | 0.66 | 50.06 | 5.85 | 0.00 | 40.74 |
| Safflower—seed | 2.43 | 55.79 | 7.34 | 0.01 | 27.83 |
| Safflower—peel | 1.77 | 53.86 | 6.97 | 0.01 | 34.69 |
| Safflower—after the oil press | 2.92 | 51.96 | 6.56 | 0.04 | 34.84 |
| Amaranth—whole plant | 0.96 | 43.27 | 5.35 | 0.09 | 43.20 |
| Flax—waste | 3.65 | 50.06 | 6.57 | 0.19 | 22.49 |
| Crambe abyssinica | 3.32 | 57.64 | 8.11 | 0.71 | 24.06 |
| Camelina—seed | 5.00 | 58.86 | 8.35 | 0.71 | 13.71 |
| Camelina—after the oil press | 6.40 | 49.00 | 6.86 | 0.93 | 22.77 |
| Spelt—waste | 0.75 | 49.12 | 5.62 | 0.20 | 39.88 |
| Cocoa—peel | 2.59 | 49.36 | 5.83 | 0.12 | 35.71 |
| Sorrel pellet (whole plant) | 1.44 | 47.61 | 5.38 | 0.10 | 40.28 |
| Rye straw | 3.08 | 45.76 | 6.50 | 0.06 | 32.09 |
| Quinoa—waste | 3.14 | 47.11 | 6.48 | 0.20 | 38.29 |
| Sample | Higher Heating Value (MJ·kg−1) | ||
|---|---|---|---|
| Digestate | 15.769 | 17.222 | 19.649 |
| Softwood pellets (spruce) | 18.207 | 19.718 | 20.330 |
| Hardwood pellets (beech) | 18.044 | 19.579 | 20.069 |
| Composite wood | 18.235 | 20.148 | 20.215 |
| Energo compost | 14.684 | 16.199 | 21.382 |
| Rape straw | 15.572 | 17.422 | 18.292 |
| Wheat straw pellet | 17.238 | 18.567 | 19.888 |
| Hay | 15.790 | 17.155 | 18.192 |
| Straw 60% + sludge 40% | 13.559 | 14.150 | 20.280 |
| Straw 70% + sludge 30% | 13.947 | 14.500 | 19.891 |
| Straw 80% + sludge 20% | 14.467 | 15.131 | 18.648 |
| Straw 90% + sludge 10% | 15.744 | 16.535 | 18.640 |
| Sunflower—peel | 17.699 | 18.966 | 19.861 |
| Sunflower—after the oil press | 22.319 | 23.766 | 25.294 |
| Sunflower—whole plant | 16.925 | 18.928 | 19.904 |
| Mix—seeds rape, sunflower, mustard, husks | 15.246 | 17.308 | 19.021 |
| Mustard—seed | 24.131 | 25.639 | 30.369 |
| Spruce sawdust + digestate | 17.565 | 18.773 | 19.331 |
| Safflower—seed | 23.221 | 24.550 | 26.397 |
| Safflower—peel | 22.677 | 23.994 | 24.728 |
| Safflower—after the oil press | 20.125 | 21.658 | 22.551 |
| Amaranth—whole plant | 16.309 | 17.478 | 18.937 |
| Flax—waste | 20.318 | 21.483 | 26.167 |
| Crambe abyssinica | 25.351 | 26.824 | 28.583 |
| Camelina—seed | 25.154 | 26.746 | 30.871 |
| Camelina—after the oil press | 20.942 | 22.542 | 25.289 |
| Spelt—waste | 16.947 | 18.432 | 19.365 |
| Cocoa—peel | 18.078 | 19.504 | 20.948 |
| Sorrel pellet (whole plant) | 16.211 | 17.744 | 18.809 |
| Rye straw | 16.835 | 18.237 | 21.126 |
| Quinoa—waste | 17.700 | 19.329 | 20.389 |
| Sample | Lower Heating Value (MJ·kg−1) | ||
|---|---|---|---|
| Digestate | 14.559 | 16.129 | 19.860 |
| Softwood pellets (spruce) | 16.807 | 18.406 | 20.551 |
| Hardwood pellets (beech) | 16.688 | 18.318 | 20.374 |
| Composite wood | 16.848 | 18.876 | 20.925 |
| Energo compost | 13.607 | 15.266 | 21.582 |
| Rape straw | 14.223 | 16.207 | 18.942 |
| Wheat straw pellet | 15.899 | 17.311 | 19.972 |
| Hay | 14.470 | 15.936 | 18.274 |
| Straw 60% + sludge 40% | 12.615 | 13.269 | 19.453 |
| Straw 70% + sludge 30% | 12.976 | 13.590 | 19.113 |
| Straw 80% + sludge 20% | 13.396 | 14.125 | 18.023 |
| Straw 90% + sludge 10% | 14.524 | 15.380 | 18.099 |
| Sunflower—peel | 16.385 | 17.737 | 19.839 |
| Sunflower—after the oil press | 20.566 | 22.581 | 25.591 |
| Sunflower—whole plant | 15.629 | 17.772 | 20.787 |
| Mix—seeds rape, sunflower, mustard, husks | 13.946 | 16.167 | 19.936 |
| Mustard—seed | 22.363 | 23.898 | 30.083 |
| Spruce sawdust + digestate | 16.204 | 17.490 | 19.213 |
| Safflower—seed | 21.567 | 22.939 | 26.071 |
| Safflower—peel | 21.096 | 22.465 | 24.465 |
| Safflower—after the oil press | 18.614 | 20.219 | 22.656 |
| Amaranth—whole plant | 15.053 | 16.305 | 18.920 |
| Flax—waste | 18.835 | 20.042 | 25.852 |
| Crambe abyssinica | 23.530 | 25.044 | 28.237 |
| Camelina—seed | 23.280 | 24.913 | 30.578 |
| Camelina—after the oil press | 19.314 | 21.036 | 25.400 |
| Spelt—waste | 15.617 | 17.198 | 19.652 |
| Cocoa—peel | 16.711 | 18.226 | 21.012 |
| Sorrel pellet (whole plant) | 14.918 | 16.563 | 19.120 |
| Rye straw | 15.342 | 16.810 | 21.063 |
| Quinoa—waste | 16.195 | 17.908 | 20.628 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lisý, M.; Lisá, H.; Jecha, D.; Baláš, M.; Križan, P. Characteristic Properties of Alternative Biomass Fuels. Energies 2020, 13, 1448. https://doi.org/10.3390/en13061448
Lisý M, Lisá H, Jecha D, Baláš M, Križan P. Characteristic Properties of Alternative Biomass Fuels. Energies. 2020; 13(6):1448. https://doi.org/10.3390/en13061448
Chicago/Turabian StyleLisý, Martin, Hana Lisá, David Jecha, Marek Baláš, and Peter Križan. 2020. "Characteristic Properties of Alternative Biomass Fuels" Energies 13, no. 6: 1448. https://doi.org/10.3390/en13061448
APA StyleLisý, M., Lisá, H., Jecha, D., Baláš, M., & Križan, P. (2020). Characteristic Properties of Alternative Biomass Fuels. Energies, 13(6), 1448. https://doi.org/10.3390/en13061448






