Thermal Stability and Performance Testing of Oil-based CuO Nanofluids for Solar Thermal Applications
Abstract
:1. Introduction
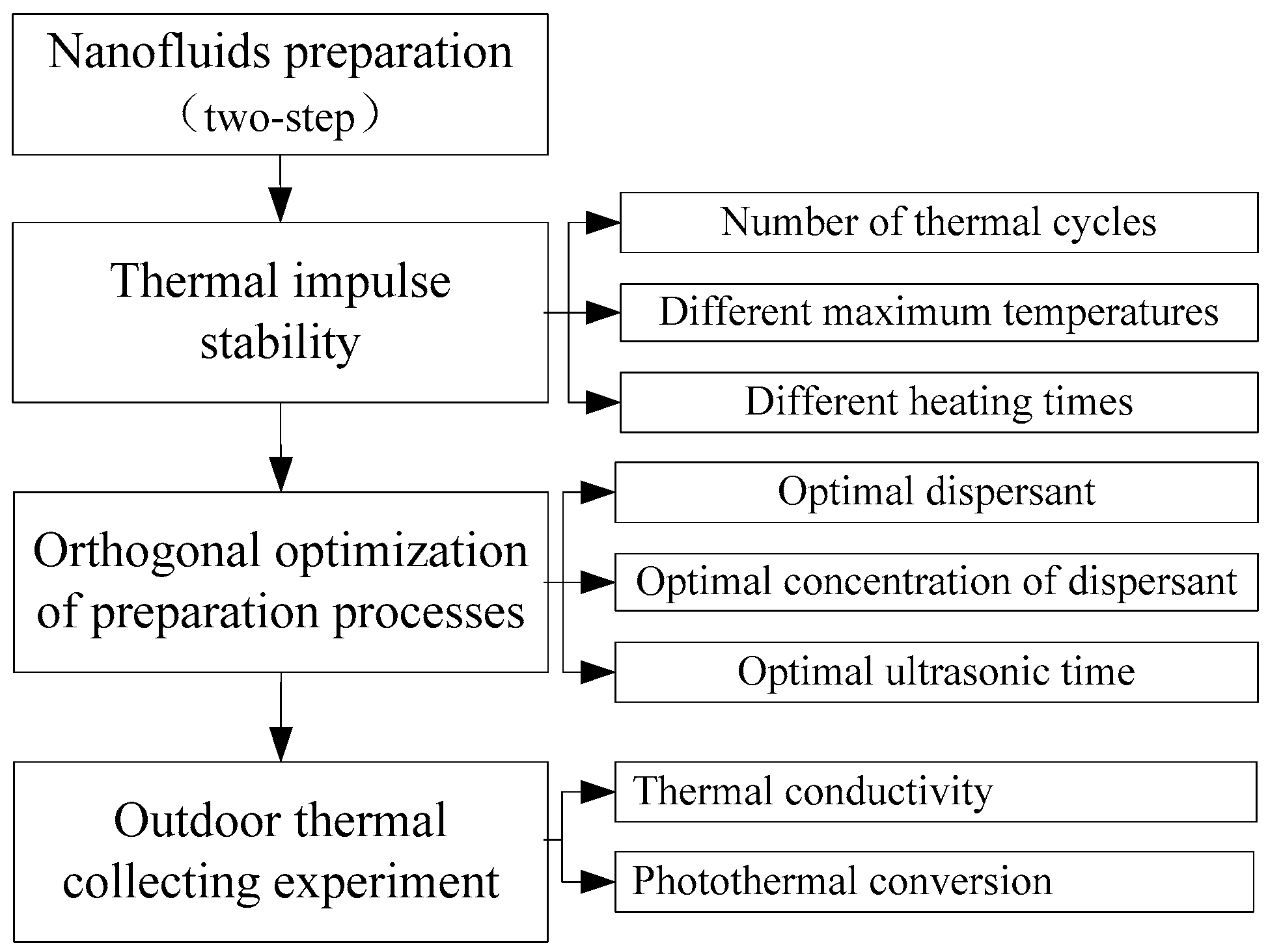
2. Thermal Impulse Stability
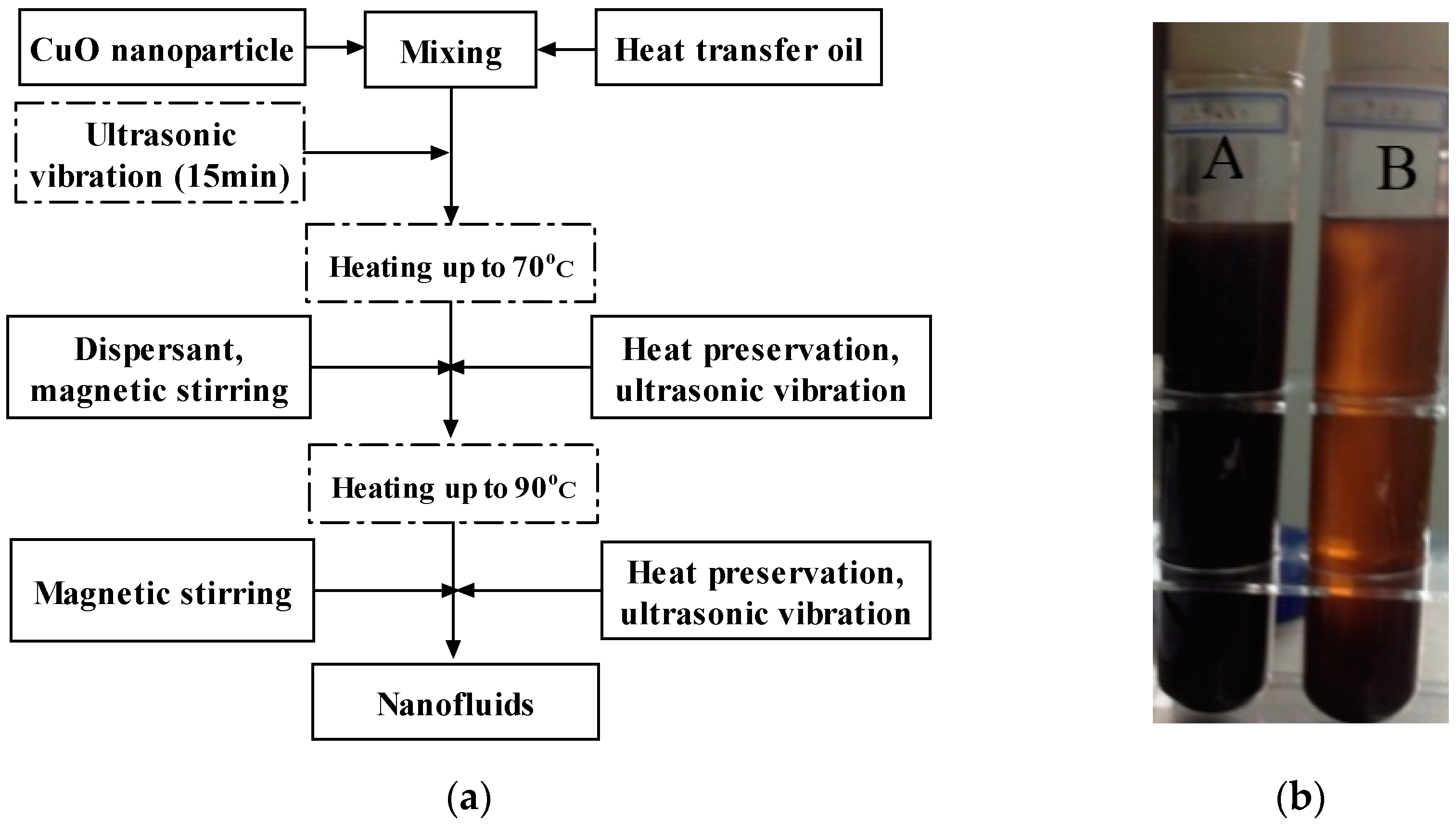
2.1. Nanofluids Preparation
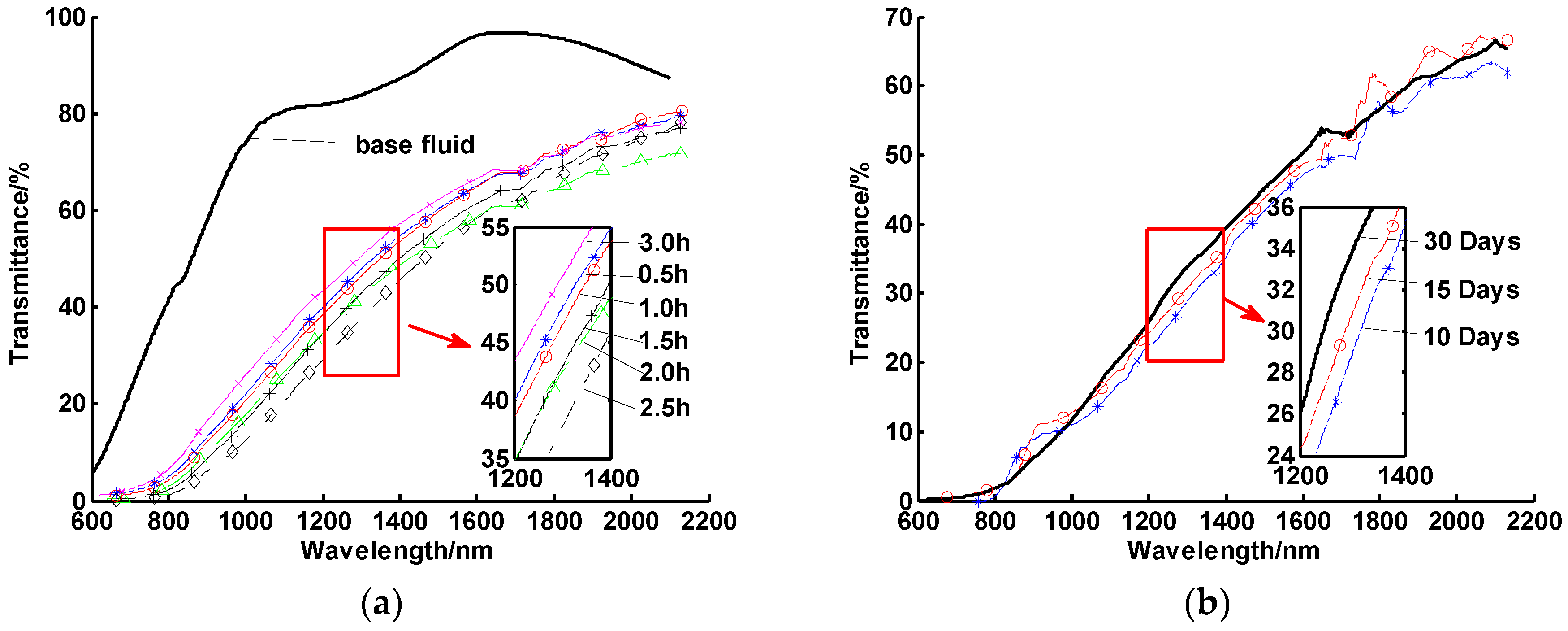
- Soft aggregation of CuO nanoparticles, which occurred during storage/transportation over time at room temperature, can be broken up with about 15 min ultrasonic vibration.
- Oleic acid (the dispersant) was much less viscous and soluble with the heat transfer oil, when the suspension was heated up to 70 °C. Thus, in this step, the dispersant was added and kept warm for one hour to ensure CuO nanoparticles to fully adsorb oleic acid.
- The final heating step was also maintained at 90 °C for one hour to ensure the low viscosity of the Diphyl DT during the final stirring/sonication step.
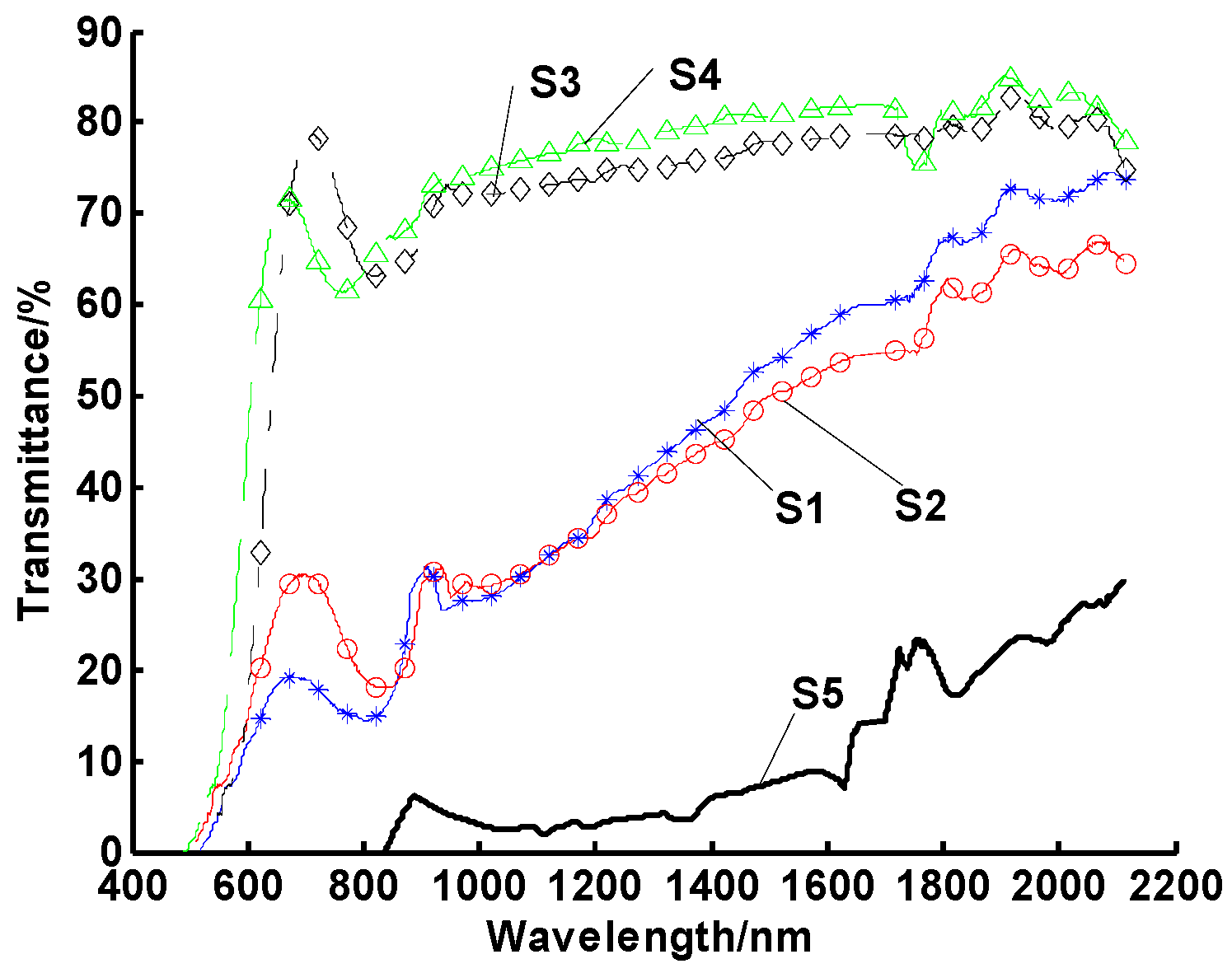
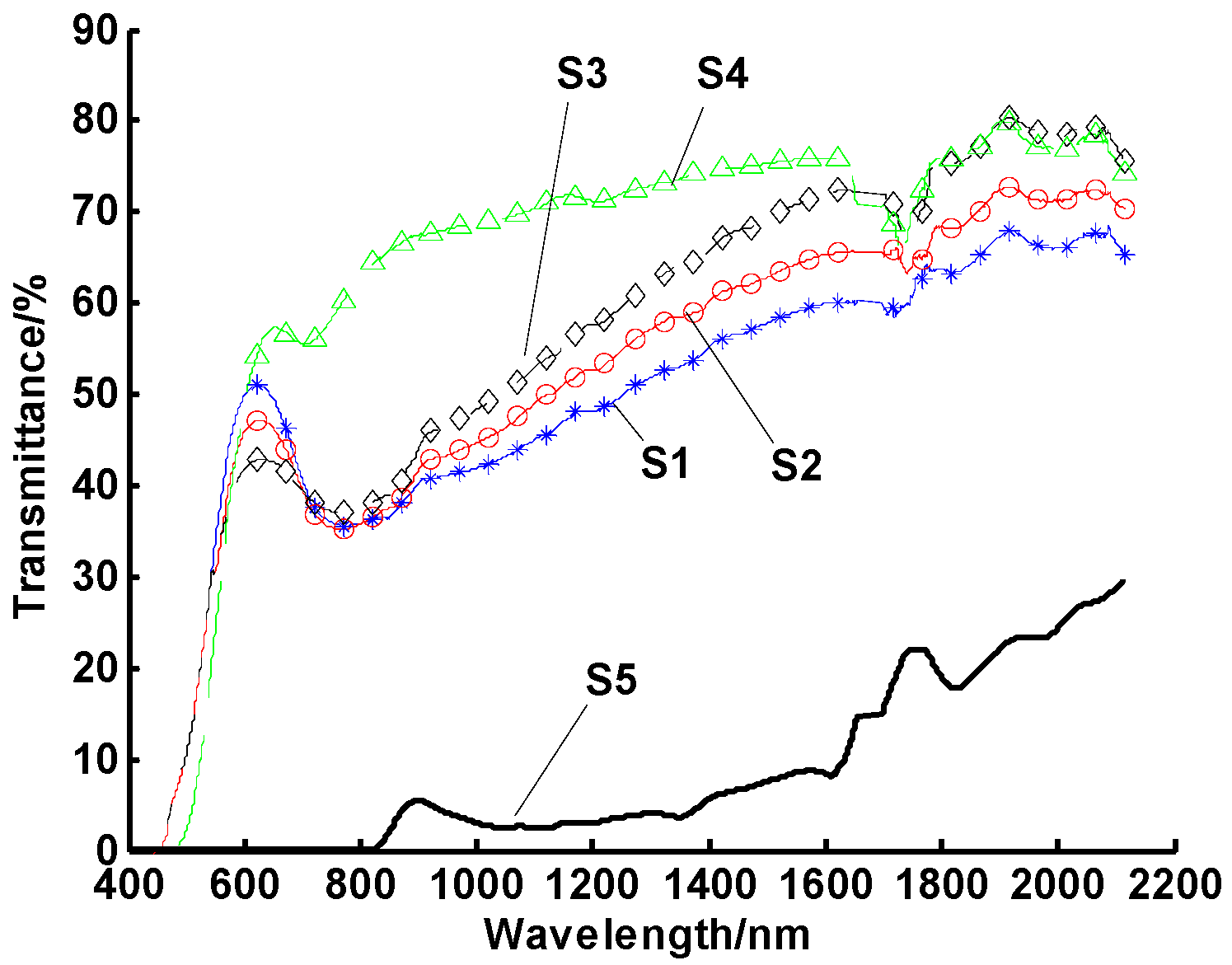
2.2. Thermal Impulse Testing
3. Optimization of Preparation Processes
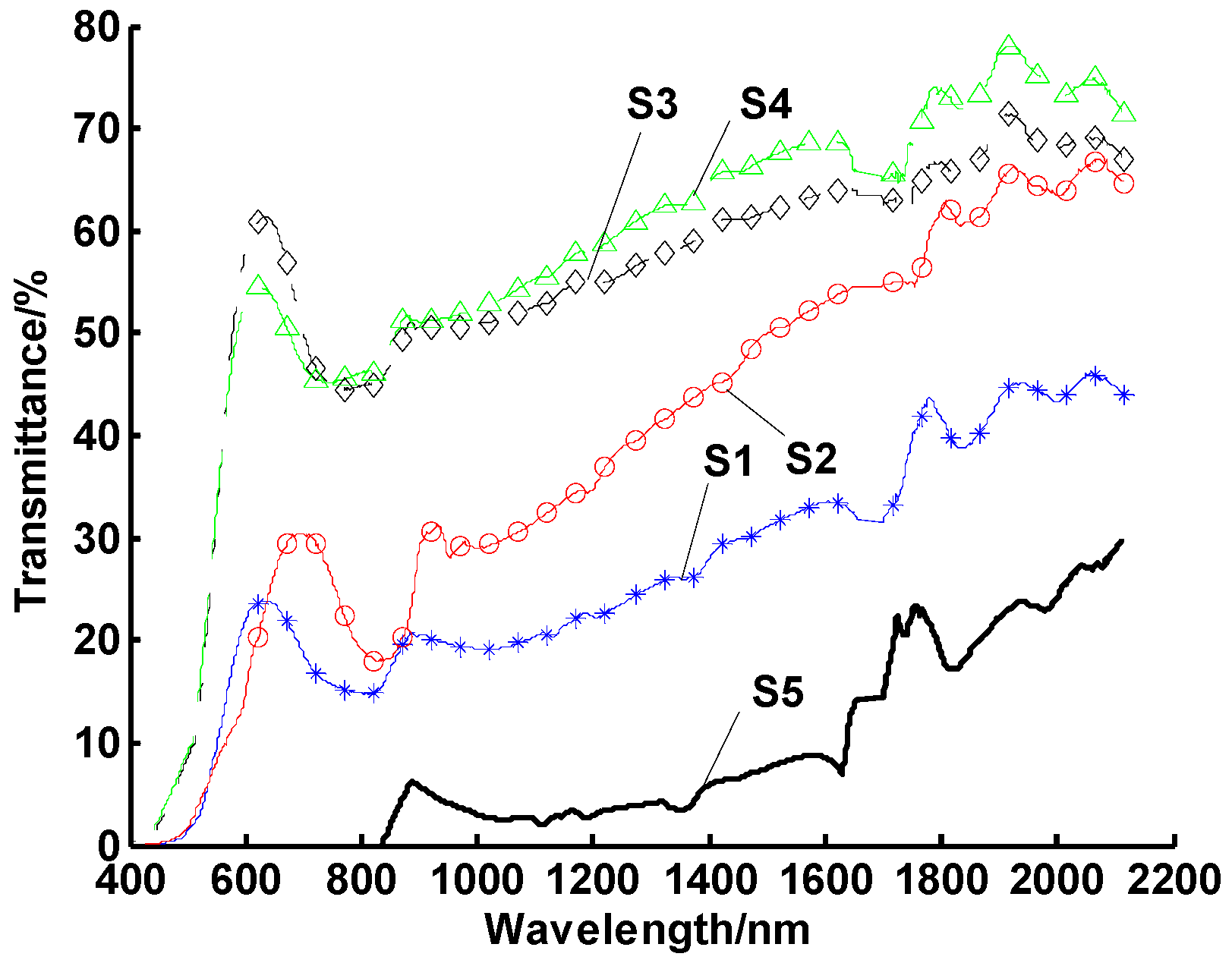
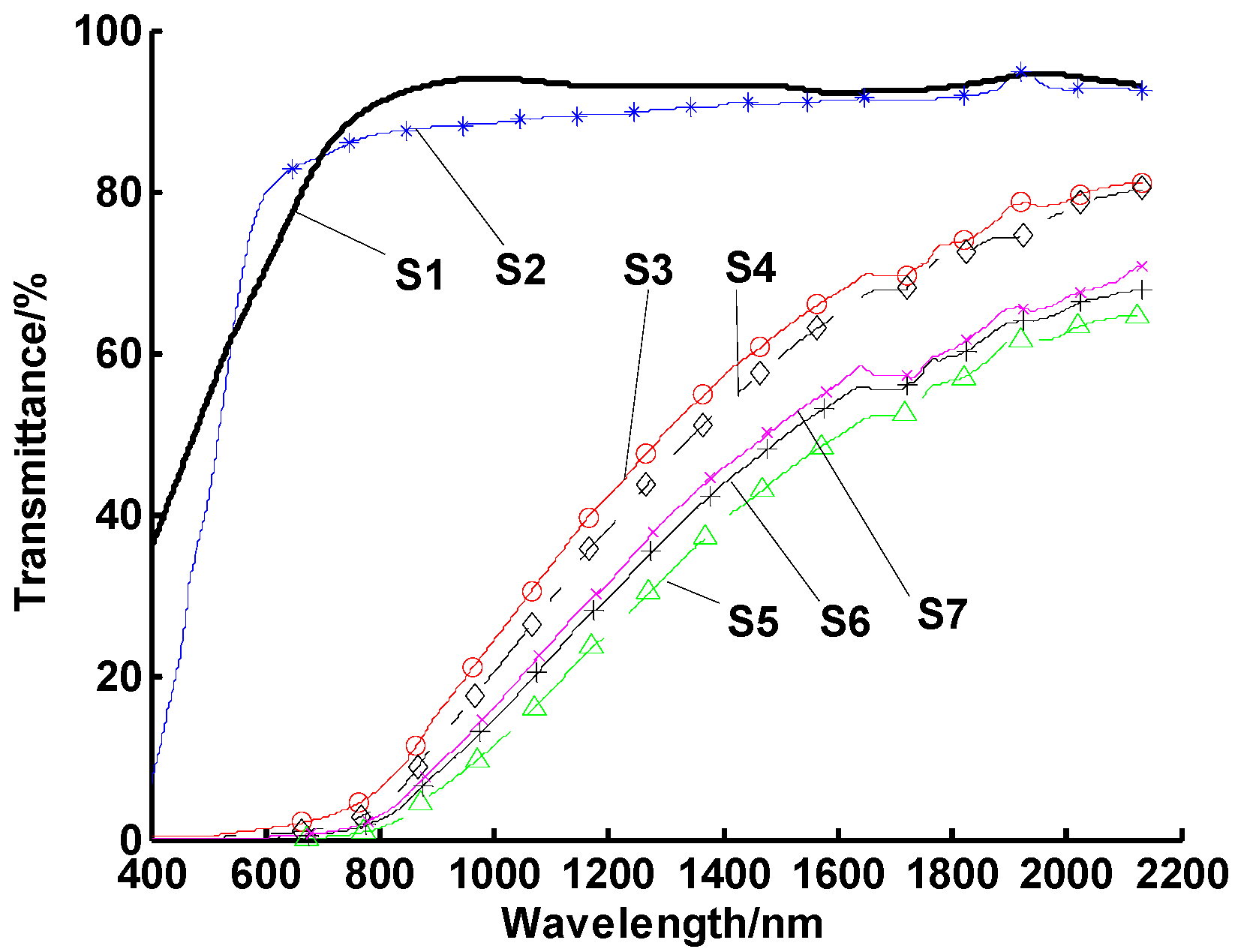
3.1. Experimental Study on the Optimal Dispersant
3.2. Experimental Study on the Optimal Concentration of the Dispersant
3.3. Experimental Study on the Optimal Ultrasonic Time
3.4. Orthogonal Tests
4. Experiment on Thermal Properties of CuO/Oil Nanofluids
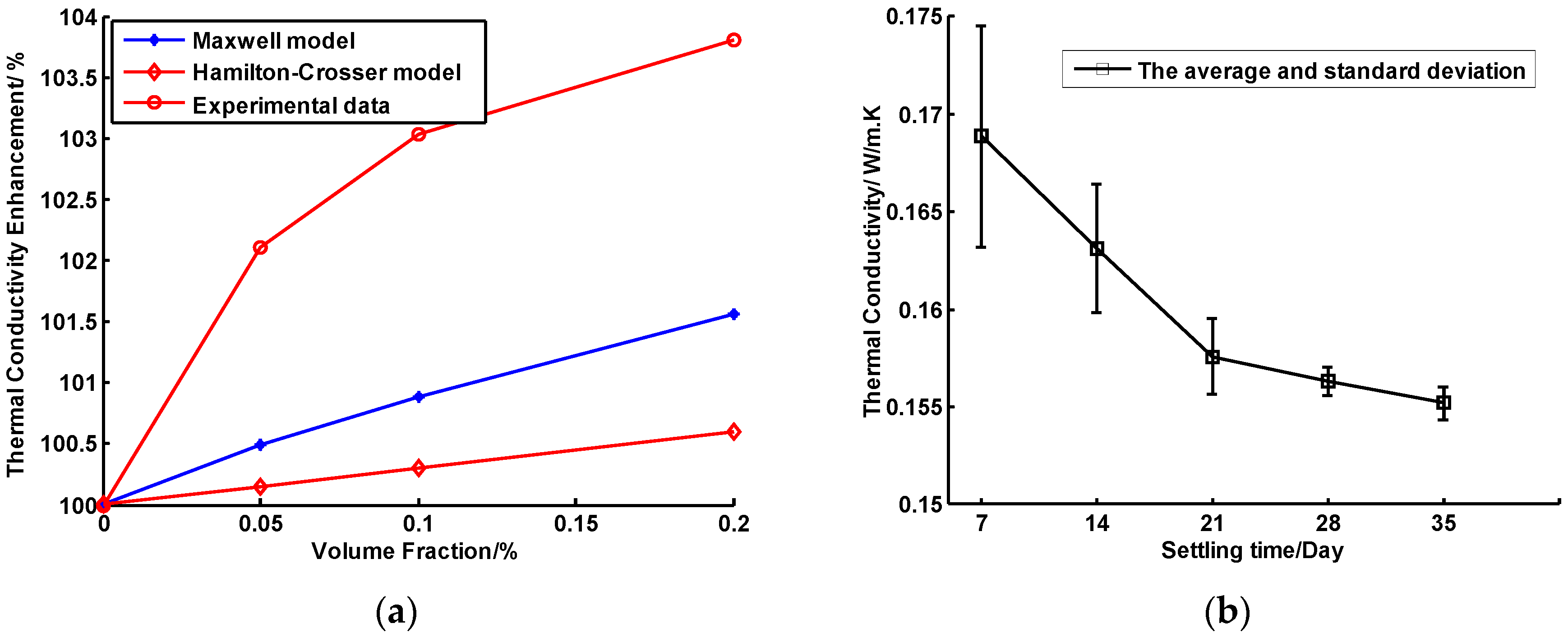
4.1. Experiment on the Thermal Conductivity of CuO/Oil Nanofluids
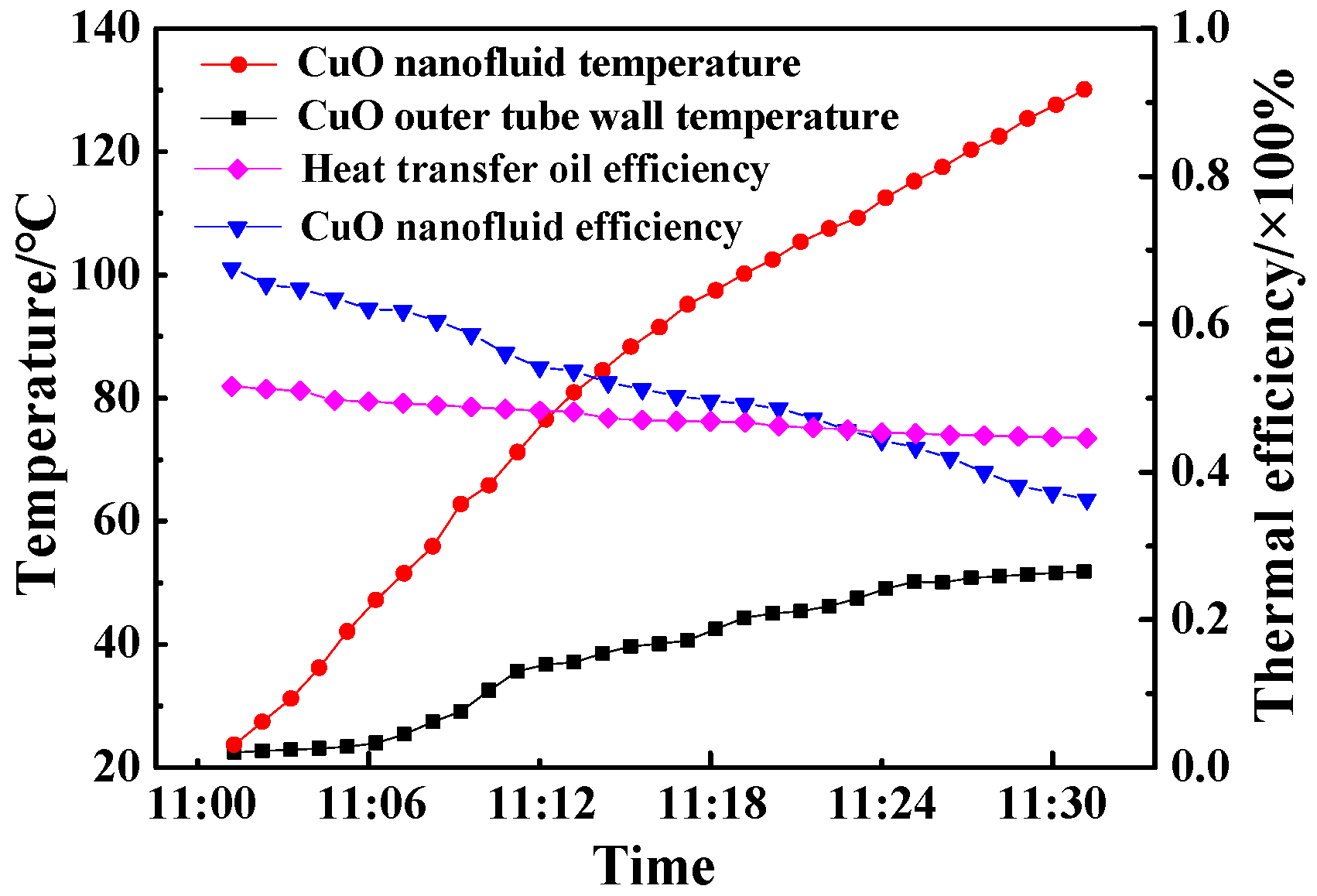
4.2. Photothermal Conversion of CuO/oil Nanofluids
5. Conclusions
- Heating temperature, heating cycles, and heating time were all found to significantly influence the stability of nanofluids;
- Nanofluids tended to aggregate quickly at medium temperatures or after several thermal cycles, even if they had high stability at room temperature (e.g., more than 30 days without settling).
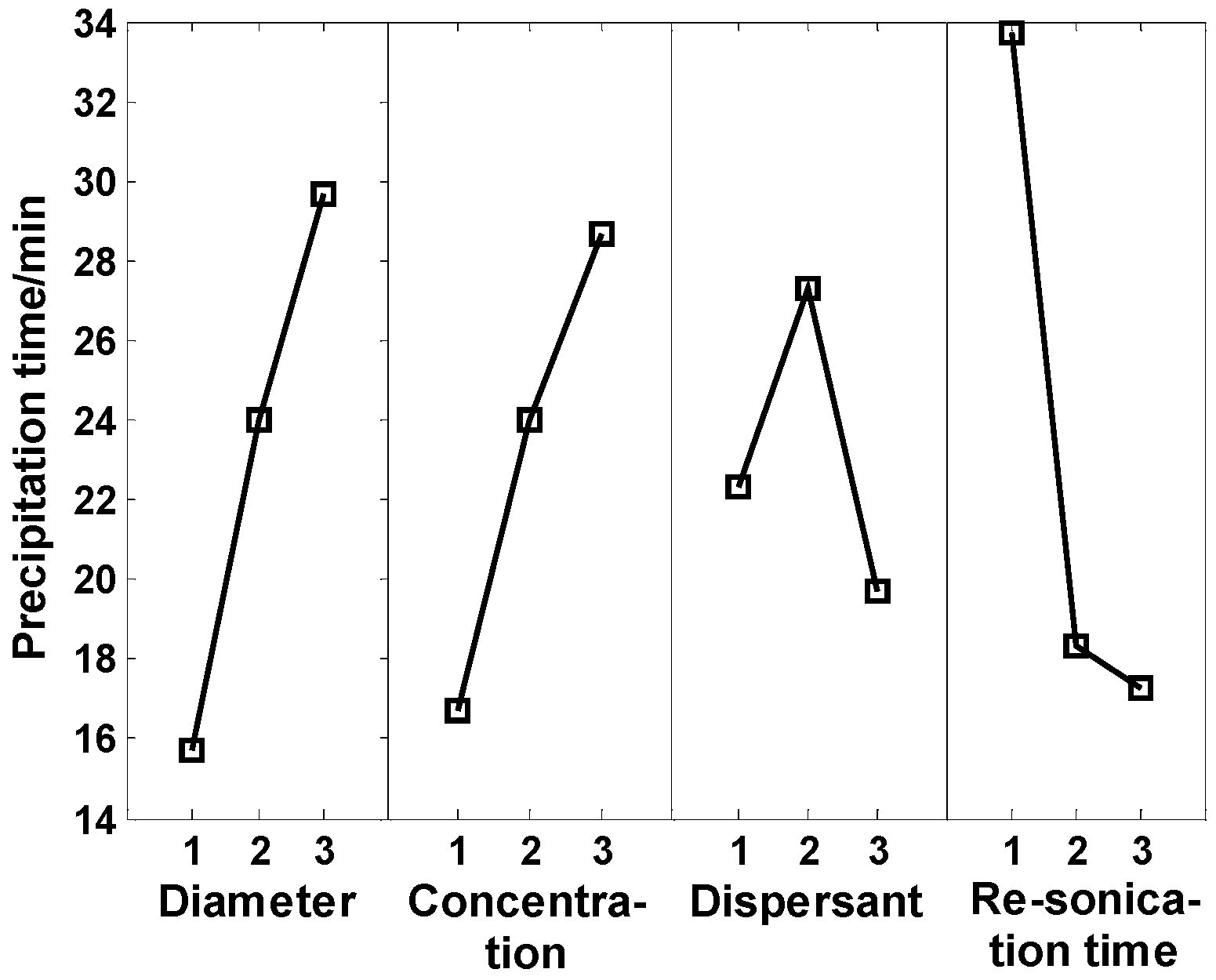
- According to our orthogonal test, the main factors affecting thermal impulse stability were the periodic resonication time, particle size, particle concentration, and the type of dispersant;
- The thermal conductivity of CuO/oil nanofluids with a 0.2% volum fraction was 3.8% higher than that of the pure oil base fluid, enabling its thermal efficiency in the solar collector to achieve 67.6%.
Author Contributions
Funding
Conflicts of Interest
References
- Tehrani, S.S.M.; Taylor, R.A. Off-Design Simulation and Performance of Molten Salt Cavity Receivers in Solar Tower Plants under Realistic Operational Modes and Control Strategies. Appl. Energy 2016, 179, 698–715. [Google Scholar] [CrossRef]
- Otanicar, T.P.; Theisen, S.; Norman, T.; Tyagi, H.; Taylor, R.A. Envisioning Advanced Solar Electricity Generation: Parametric Studies of CPV/T Systems with Spectral Filtering and High Temperature PV. Appl. Energy 2015, 140, 224–233. [Google Scholar] [CrossRef]
- Menbari, A.; Alemrajabi, A.A.; Ghayeb, Y. Investigation on the Stability, Viscosity and Extinction Coefficient of Cuo–Al2O3/Water Binary Mixture Nanofluid. Exp. Therm. Fluid Sci. 2016, 74, 122–129. [Google Scholar] [CrossRef]
- Aguilar, T.; Navas, J.; Sánchez-Coronilla, A.; Martín, E.I.; Gallardo, J.J.; Martínez-Merino, P.; Gómez-Villarejo, R.; Piñero, J.C.; Alcántara, R.; Fernández-Lorenzo, C. Investigation of Enhanced Thermal Properties in NiO-Based Nanofluids for Concentrating Solar Power Applications: A Molecular Dynamics and Experimental Analysis. Appl. Energy 2018, 211, 677–688. [Google Scholar] [CrossRef]
- Sharafeldin, M.A.; Gróf, G. Efficiency of Evacuated Tube Solar Collector Using WO3/water Nanofluid. Renew. Energy 2019, 134, 453–460. [Google Scholar] [CrossRef]
- Du, M.; Tang, G.H. Optical Property of Nanofluids with Particle Agglomeration. Sol. Energy 2015, 122, 864–872. [Google Scholar] [CrossRef]
- Yousefi, T.; Veysi, F.; Shojaeizadeh, E.; Zinadini, S. An Experimental Investigation on the Effect of Al2O3–H2O Nanofluid on the Efficiency of Flat-Plate Solar Collectors. Renew. Energy 2012, 39, 293–298. [Google Scholar] [CrossRef]
- Mohammad, S.; Mohammad, P.F.; Saeed, Z.H. Experimental Investigation of the Effects of Silica/water Nanofluid on PV/T (photovoltaic thermal units). Energy 2014, 66, 264–272. [Google Scholar] [CrossRef]
- Taylor, R.A.; Phelan, P.E.; Otanicar, T.P.; Walker, C.A.; Nguyen, M.; Trimble, S.; Prasher, R. Applicability of Nanofluids in High Flux Solar Collectors. J. Renew. Sustain. Energy 2011, 3. [Google Scholar] [CrossRef] [Green Version]
- Amiri, A.; Øye, G.; Sjöblom, J. Temperature and Pressure Effects on Stability and Gelation Properties of Silica Suspensions. Colloid Surf. A-Physicochem. Eng. Asp. 2011, 378, 14–21. [Google Scholar] [CrossRef]
- Mahbubul, I.M.; Saidur, R.; Amalina, M.A.; Niza, M.E. Influence of Ultrasonication Duration on Rheological Properties of Nanofluid: An Experimental Study with Alumina–water Nanofluid. Int. Commun. Heat Mass Transf. 2016, 76, 33–40. [Google Scholar] [CrossRef]
- Li, F.S.; Li, L.; Zhong, G.J.; Zhai, Y.L.; Li, Z.H. Effects of Ultrasonic Time, Size of Aggregates and Temperature on the Stability and Viscosity of Cu-Ethylene Glycol (EG) Nanofluids. Int. J. Heat Mass Transf. 2019, 129, 278–286. [Google Scholar] [CrossRef]
- Khairul, M.A.; Shah, K.; Doroodchi, E.; Azizian, R.; Moghtaderi, B. Effects of Surfactant on Stability and Thermo-Physical Properties of Metal Oxide Nanofluids. Int. J. Heat Mass Transf. 2016, 98, 778–787. [Google Scholar] [CrossRef]
- Sezer, N.; Koç, M. Stabilization of the Aqueous Dispersion of Carbon Nanotubes Using Different Approaches. Therm. Sci. Eng. Prog. 2018, 8, 411–417. [Google Scholar] [CrossRef]
- Nagarajan, P.K. Influence of Stability and Particle Shape Effects for an Entropy Generation Based Optimized Selection of Magnesia Nanofluid for Convective Heat Flow Applications. Appl. Surf. Sci. 2019, 489, 560–575. [Google Scholar] [CrossRef]
- Arulprakasajothi, M.; Elangovan, K.; HemaChandraReddy, K.; Suresh, S. Heat Transfer Study of Water-based Nanofluids Containing Titanium Oxide Nanoparticles. Mater. Today: Proc. 2015, 2, 3648–3655. [Google Scholar] [CrossRef]
- Bashirnezhad, K.; Bazri, S.; Safaei, M.R.; Goodarzi, M.; Dahari, M.; Mahian, O.; Dalkılıçad, A.S.; Wongwisese, S. Viscosity of Nanofluids: A Review of Recent Experimental Studies. Int. Commun. Heat Mass Transf. 2016, 73, 114–123. [Google Scholar] [CrossRef]
- Li, Y.J.; Eric, S.; Simon, T.; Xi, S.Q.; Zhou, J.E. A Review on Development of Nanofluid Preparation and Characterization. Powder Technol. 2009, 196, 89–101. [Google Scholar] [CrossRef]
- Al-Gebory, L.; Mengüç, M.P. The Effect of PH on Particle Agglomeration and Optical Properties of Nanoparticle Suspensions. J. Quant. Spectrosc. Radiat. Transf. 2018, 219, 46–60. [Google Scholar] [CrossRef]
- Sharma, S.K.; Gupta, S.M. Preparation and Evaluation of Stable Nanofluids for Heat Transfer Application: A Review. Exp. Therm. Fluid Sci. 2016, 79, 202–212. [Google Scholar] [CrossRef]
- Taylor, R.A.; Hjerrild, N.; Duhaini, N.; Pickford, M.; Mesgari, S. Stability Testing of Silver Nanodisc Suspensions for Solar Applications. Appl. Surf. Sci. 2018, 455, 465–475. [Google Scholar] [CrossRef]
- Hordy, N.; Rabilloud, D.; Meunier, J.L.; Coulombe, S. High Temperature and Long-Term Stability of Carbon Nanotube Nanofluids for Direct Absorption Solar Thermal Collectors. Sol. Energy 2014, 105, 82–90. [Google Scholar] [CrossRef]
- Bellos, E.; Tzivanidis, C. A Review of Concentrating Solar Thermal Collectors with and without Nanofluids. J. Therm. Anal. Calorim. 2019, 135, 763–786. [Google Scholar] [CrossRef]
- Bellos, E.; Tzivanidis, C. Multi-Criteria Evaluation of a Nanofluid-Based Linear Fresnel Solar Collector. Sol. Energy 2018, 163, 200–214. [Google Scholar] [CrossRef]
- Sani, E.; Barison, S.; Pagura, C.; Mercatelli, L.; Sansoni, P.; Fontani, D.; Jafrancesco, D.; Francini, F. Carbon Nanohorns-Based Nanofluids as Direct Sunlight Absorbers. Opt. Express 2010, 18, 5179–5187. [Google Scholar] [CrossRef]
- Mesgari, S.; Coulombe, S.; Hordy, N.; Taylor, R.A. Thermal Stability of Carbon Nanotube-Based Nanofluids for Solar Thermal Collectors. Mater. Res. Innov. 2015, 19. [Google Scholar] [CrossRef]
- Jiang, H.F.; Li, H.; Zan, C.; Wang, F.Q.; Yang, Q.P.; Shi, L. Temperature Dependence of the Stability and Thermal Conductivity of an Oil-Based Nanofluid. Thermochim. Acta 2014, 579, 27–30. [Google Scholar] [CrossRef]
- Otanicar, T.; Hoyt, J.; Fahar, M.; Jiang, X.; Taylor, R.A. Experimental and Numerical Study on the Optical Properties and Agglomeration of Nanoparticle Suspensions. J. Nanopart. Res. 2013, 15, 2039. [Google Scholar] [CrossRef]
- Mesgari, S.; Taylor, R.A.; Crisostomo, F.; Li, Q.; Hjerrild, N.E.; Scott, J. An Investigation of Thermal Stability of Carbon Nanofluids for Solar Thermal Applications. Sol. Energy Mater. Sol. Cells 2016, 157, 652–659. [Google Scholar] [CrossRef]
- Khanafer, K.; Vafai, K. A Review on the Applications of Nanofluids in Solar Energy Field. Renew. Energy 2018, 123, 398–406. [Google Scholar] [CrossRef]
- Sezer, N.; Atieh, M.A.; Koç, M. A Comprehensive Review on Synthesis, Stability, Thermophysical Properties, and Characterization of Nanofluids. Powder Technol. 2019, 344, 404–431. [Google Scholar] [CrossRef]
- Xu, G.Y.; Chen, W.; Deng, S.M.; Zhang, X.S.; Zhao, S.N. Performance Evaluation of a Nanofluid-Based Direct Absorption Solar Collector with Parabolic Trough Concentrator. Nanomaterials 2015, 5, 2131–2147. [Google Scholar] [CrossRef] [Green Version]
- Mwesigye, A.; Huan, Z.J.; Meyer, J.P. Thermodynamic Optimisation of the Performance of a Parabolic Trough Receiver Using Synthetic Oil-Al2O3 Nanofluid. Appl. Energy 2015, 156, 398–412. [Google Scholar] [CrossRef] [Green Version]
- Okonkwo, E.C.; Essien, E.A.; Akhayere, E.; Abid, M.; Kavaz, D.; Ratlamwala, T.A.H. Thermal Performance Analysis of a Parabolic Trough Collector Using Water-Based Green-Synthesized Nanofluids. Sol. Energy 2019, 170, 658–670. [Google Scholar] [CrossRef]
- Dardan, E.; Afrand, M.; Isfahani, A.H.M. Effect of Suspending Hybrid Nano-Ddditives on Rheological Behavior of Engine Oil and Pumping Power. Appl. Therm. Eng. 2016, 109, 524–534. [Google Scholar] [CrossRef]
- Yang, L.; Hu, Y. Toward TiO2 Nanofluids-Part 1: Preparation and Properties. Nanoscale Res. Lett. 2017, 12, 417. [Google Scholar] [CrossRef] [Green Version]
- Priya, K.R.; Suganthi, K.S.; Rajan, K.S. Transport Properties of Ultra-Low Concentration CuO–Water Nanofluids Containing Non-Spherical Nanoparticles. Int. J. Heat Mass Transf. 2012, 55, 4734–4743. [Google Scholar] [CrossRef]
- Kim, H.J.; Lee, S.H.; Lee, J.H.; Jang, S.P. Effect of Particle Shape on Suspension Stability and Thermal Conductivities of Water-Based Bohemite Alumina Nanofluids. Energy 2015, 90, 1290–1297. [Google Scholar] [CrossRef]
- Yang, L.; Xu, J.; Zhang, X. Recent Developments on Viscosity and Thermal Conductivity of Nanofluids. Powder Technol. 2017, 317, 348–369. [Google Scholar] [CrossRef]



| Materials | Mean Diameter, (nm) | Specific Surface, (m2/g) | Density, (g/cm3) | Thermal Conductivity, (W/(m × K)) |
|---|---|---|---|---|
| Diphyl DT | - | - | 1.035 | 0.1554 |
| CuO | 60 | 60 | 6.4 | 76.5 |
| Dispersants | Oleic Acid | Glyceryl Monostearate | Span 80 | Tween 40 |
|---|---|---|---|---|
| Chemical formula | C18H34O2 | C21H42O4 | C24H44O6 | C22H42O6 |
| Density (20 °C) | 0.891 g/mL | 0.985 g/cm3 | 0.994 g/mL | 1.10 g/mL |
| Boiling point | 350–360 °C | 476.9 °C | 579.3 °C | — |
| Melting point | 13.4 °C | 80 °C | 11 °C | 0.1 °C |
| Hydrophile lipophile balance (HLB) | 1.0 | 3.8 | 4.3 | 5.6 |
| Purity | AR | CP | CP | CP |
| Level | Diameter (A) | Concentration (B) | Dispersant (C) | Periodic Resonication Time (D) |
|---|---|---|---|---|
| 1 2 3 | 100 nm 60 nm 30 nm | 3 vol% 2 vol% 1 vol% | Oleic acid | 2 h 0.5 h 0 h |
| Oleic acid + Span 80 | ||||
| Span 80 |
| Items | A | B | C | D | Results |
|---|---|---|---|---|---|
| 1 | 1 | 1 | 1 | 1 | 19 |
| 2 | 1 | 2 | 2 | 2 | 16 |
| 3 | 1 | 3 | 3 | 3 | 12 |
| 4 | 2 | 1 | 2 | 3 | 16 |
| 5 | 2 | 2 | 3 | 1 | 32 |
| 6 | 2 | 3 | 1 | 2 | 24 |
| 7 | 3 | 1 | 3 | 2 | 15 |
| 8 | 3 | 2 | 1 | 3 | 24 |
| 9 | 3 | 3 | 2 | 1 | 50 |
| k1 | 47 | 50 | 67 | 101 | |
| k2 | 72 | 72 | 82 | 55 | |
| k3 | 89 | 86 | 59 | 52 | |
| 15.7 | 16.7 | 22.3 | 33.7 | ||
| 24 | 24 | 27.3 | 18.3 | ||
| 29.7 | 28.7 | 19.7 | 17.3 | ||
| Range | 14 | 12 | 7.6 | 16.4 |
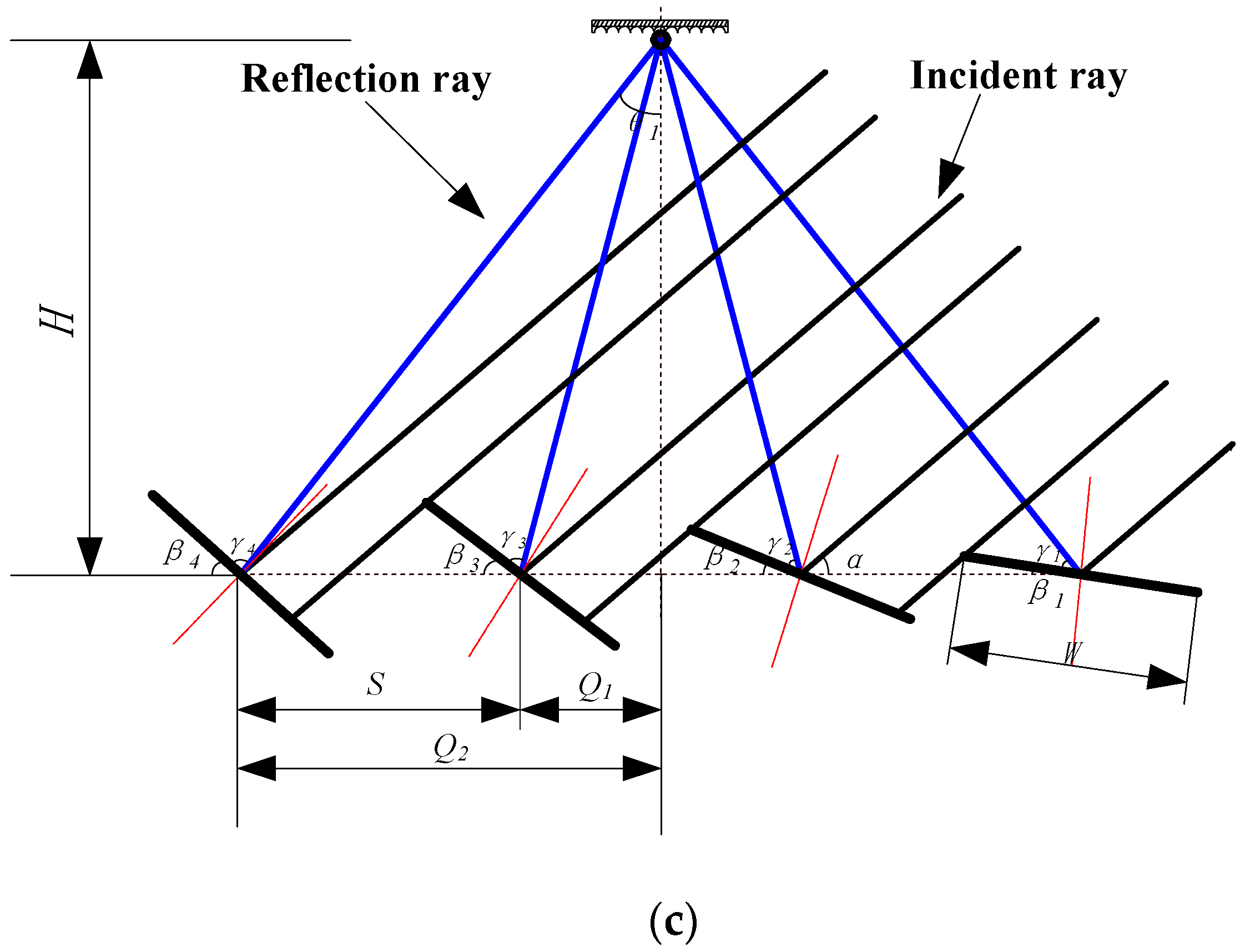
| Parameter | Value |
|---|---|
| Mirror width W (mm) | 800 |
| Mirror length L (mm) | 2200 |
| Q1 (mm) | 475 |
| Center distance S (mm) | 950 |
| Collector height H (mm) | 1800 |
| Tilt angle (°) | 32 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, M.; Wang, S.; Zhu, Y.; Taylor, R.A.; Moghimi, M.A.; Wang, Y. Thermal Stability and Performance Testing of Oil-based CuO Nanofluids for Solar Thermal Applications. Energies 2020, 13, 876. https://doi.org/10.3390/en13040876
Yang M, Wang S, Zhu Y, Taylor RA, Moghimi MA, Wang Y. Thermal Stability and Performance Testing of Oil-based CuO Nanofluids for Solar Thermal Applications. Energies. 2020; 13(4):876. https://doi.org/10.3390/en13040876
Chicago/Turabian StyleYang, Moucun, Sa Wang, Yuezhao Zhu, Robert A. Taylor, M.A. Moghimi, and Yinfeng Wang. 2020. "Thermal Stability and Performance Testing of Oil-based CuO Nanofluids for Solar Thermal Applications" Energies 13, no. 4: 876. https://doi.org/10.3390/en13040876
APA StyleYang, M., Wang, S., Zhu, Y., Taylor, R. A., Moghimi, M. A., & Wang, Y. (2020). Thermal Stability and Performance Testing of Oil-based CuO Nanofluids for Solar Thermal Applications. Energies, 13(4), 876. https://doi.org/10.3390/en13040876






