Decarbonization of the Iron and Steel Industry with Direct Reduction of Iron Ore with Green Hydrogen
Abstract
1. Introduction
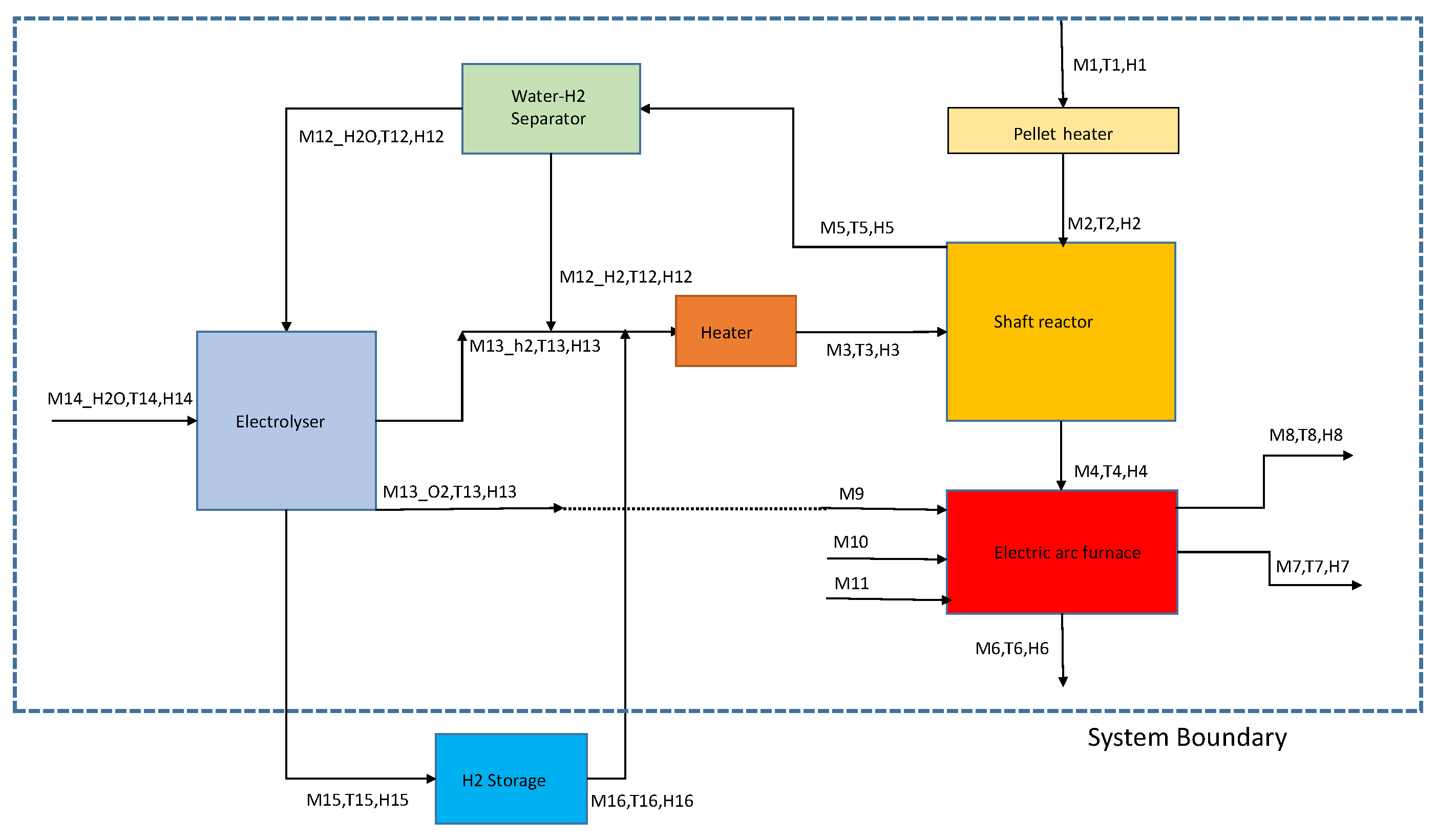
2. Concept
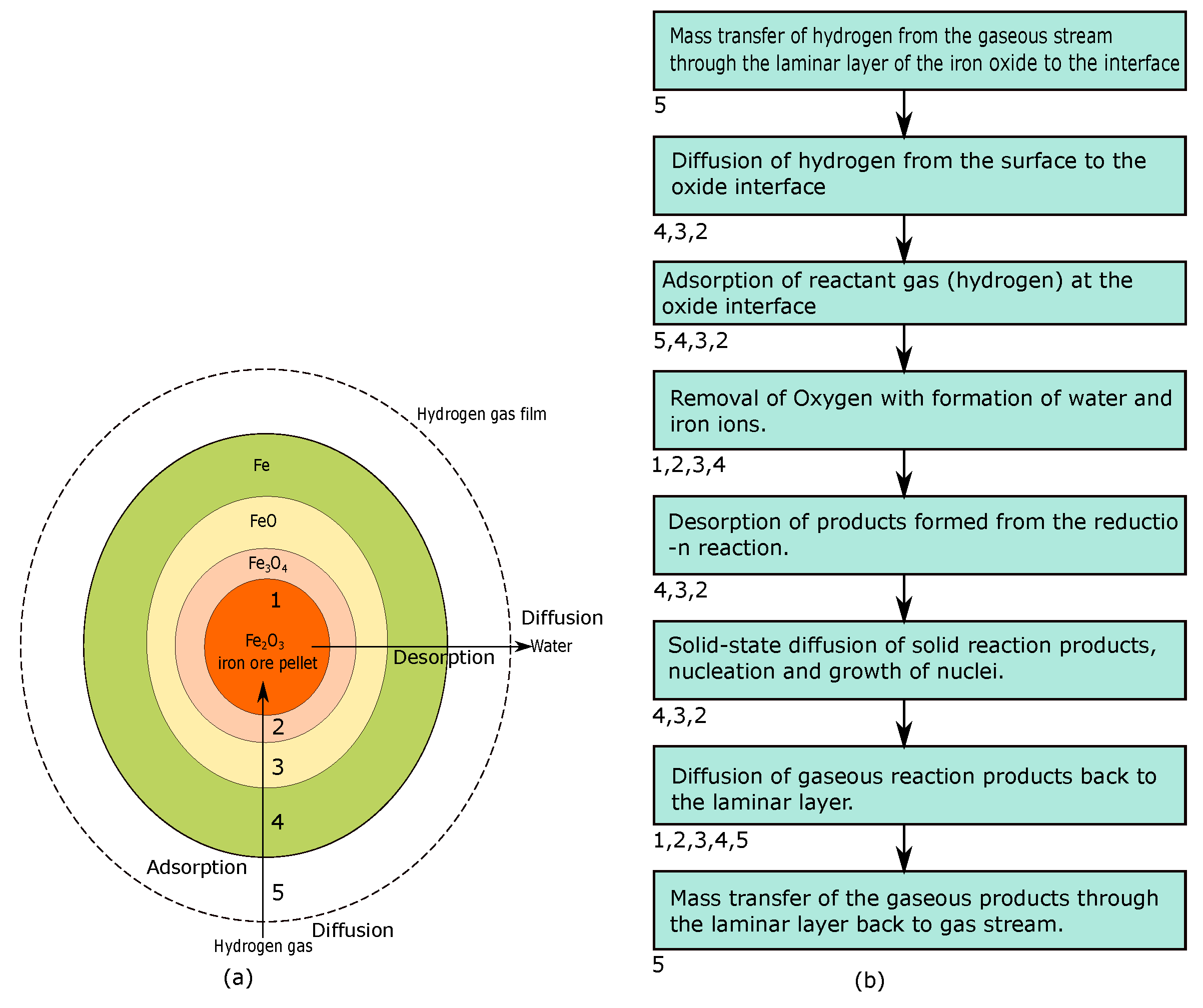
Hydrogen Direct Reduction of Iron Ore
3. Methodology
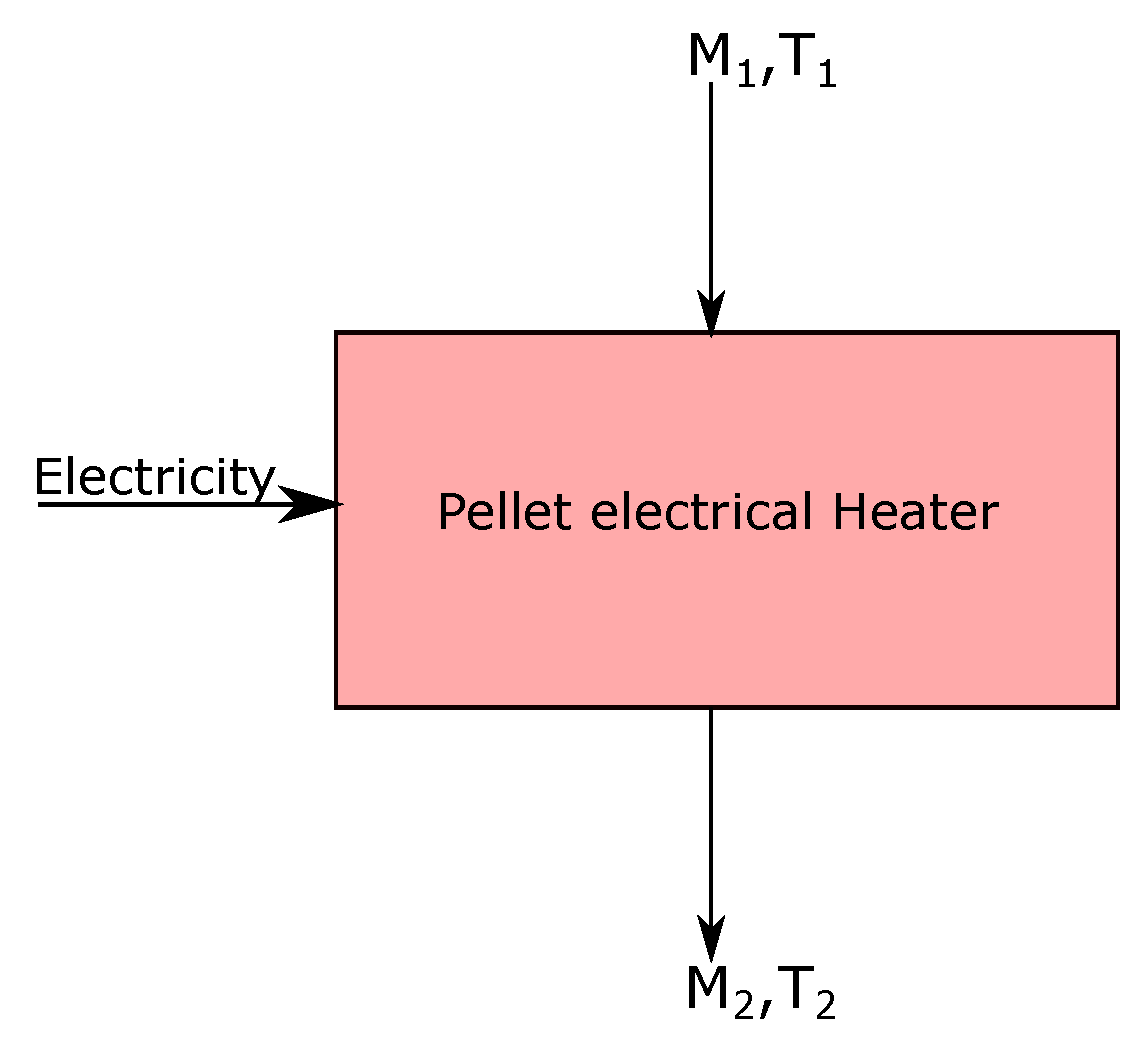
3.1. Pellet Heating
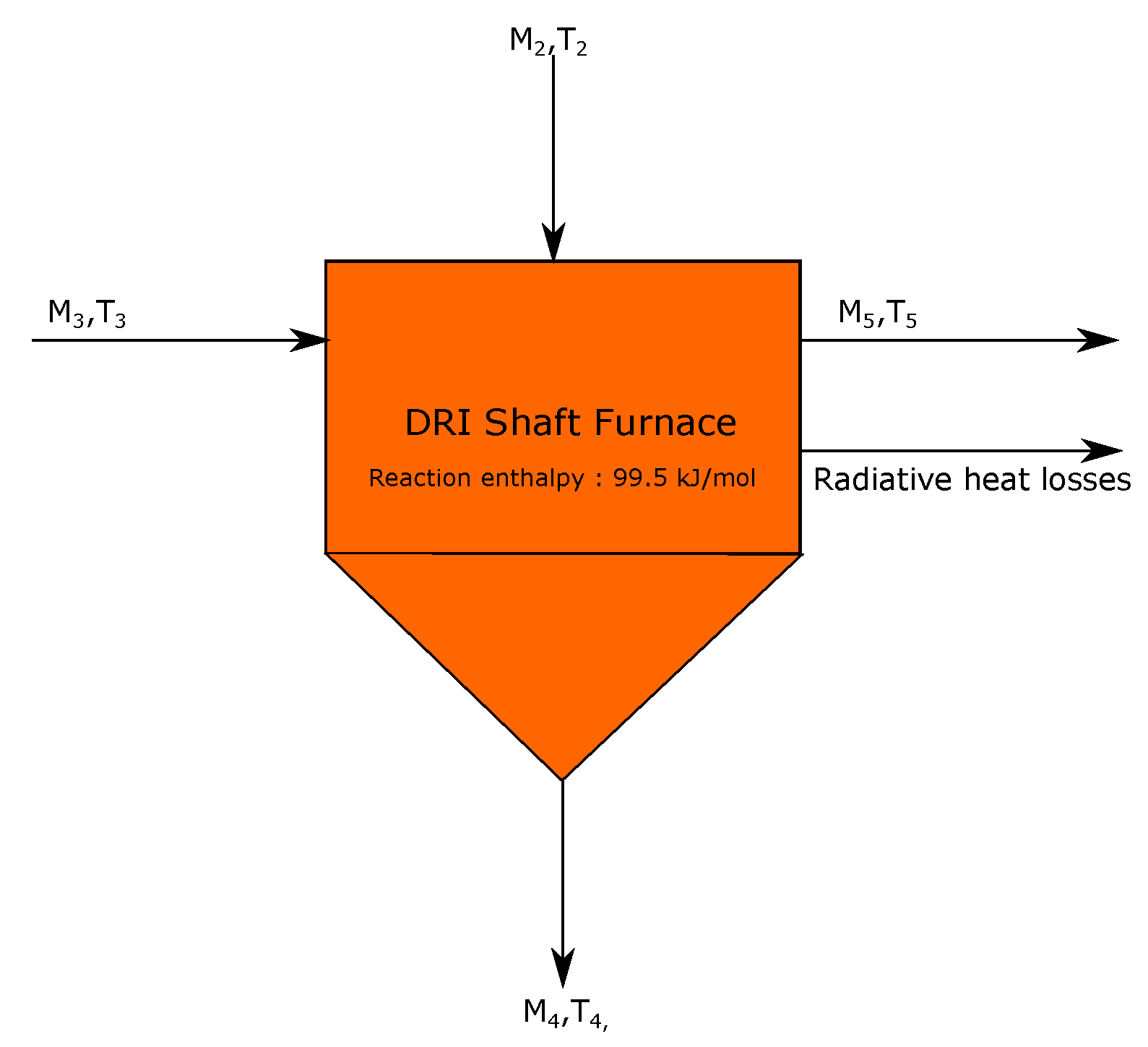
3.2. Direct Reduction Shaft Furnace
3.3. Electric Arc Furnace
3.4. Electrolyzer
3.5. Waste Gas Separation Unit
3.6. Electric Heater for Hydrogen Stream
4. Results and Discussions
4.1. Mass and Energy Flow
4.2. Electricity Consumption
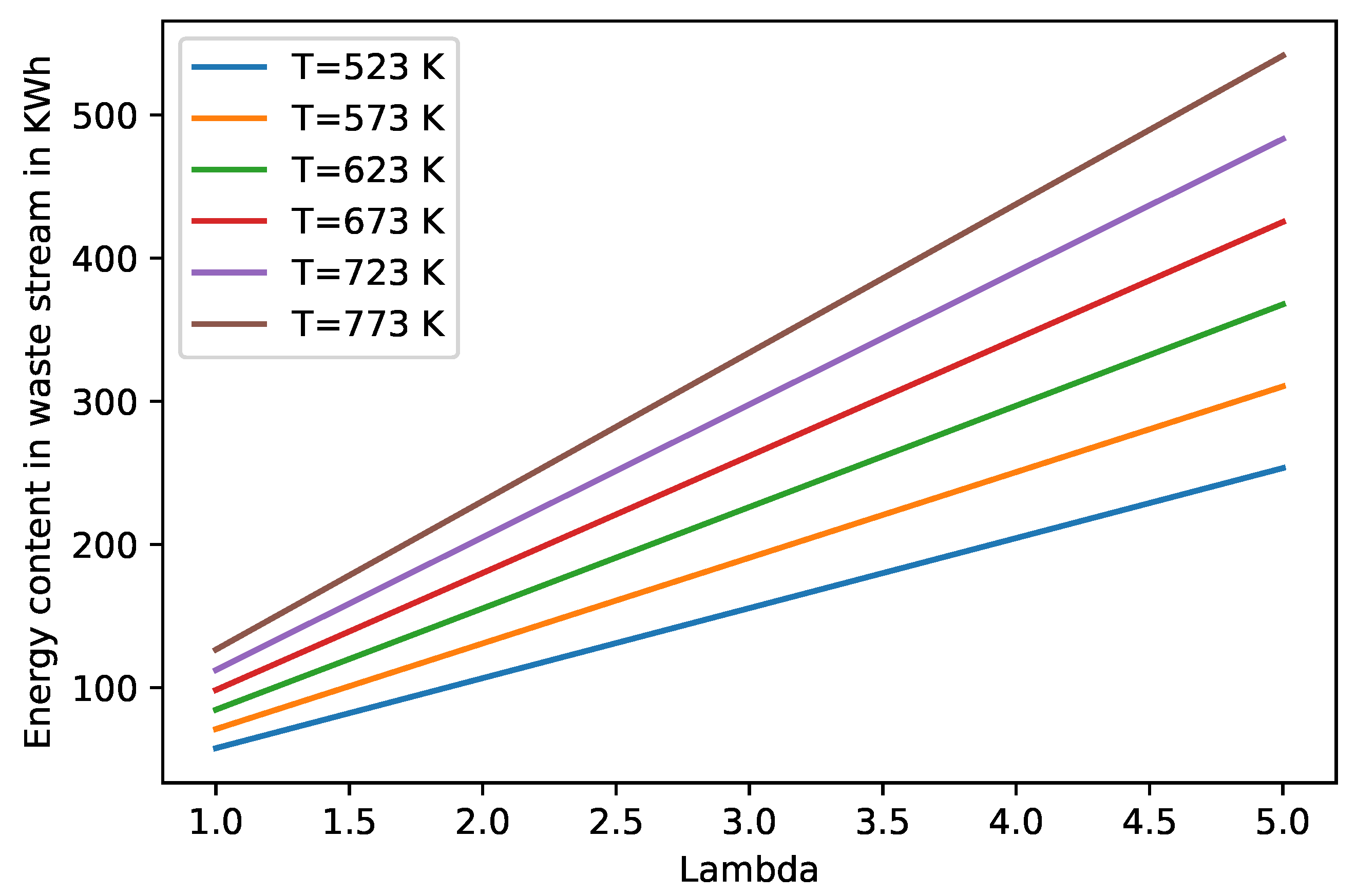
Waste Gas Enthalpy
4.3. Emissions from the HDRI-EAF System
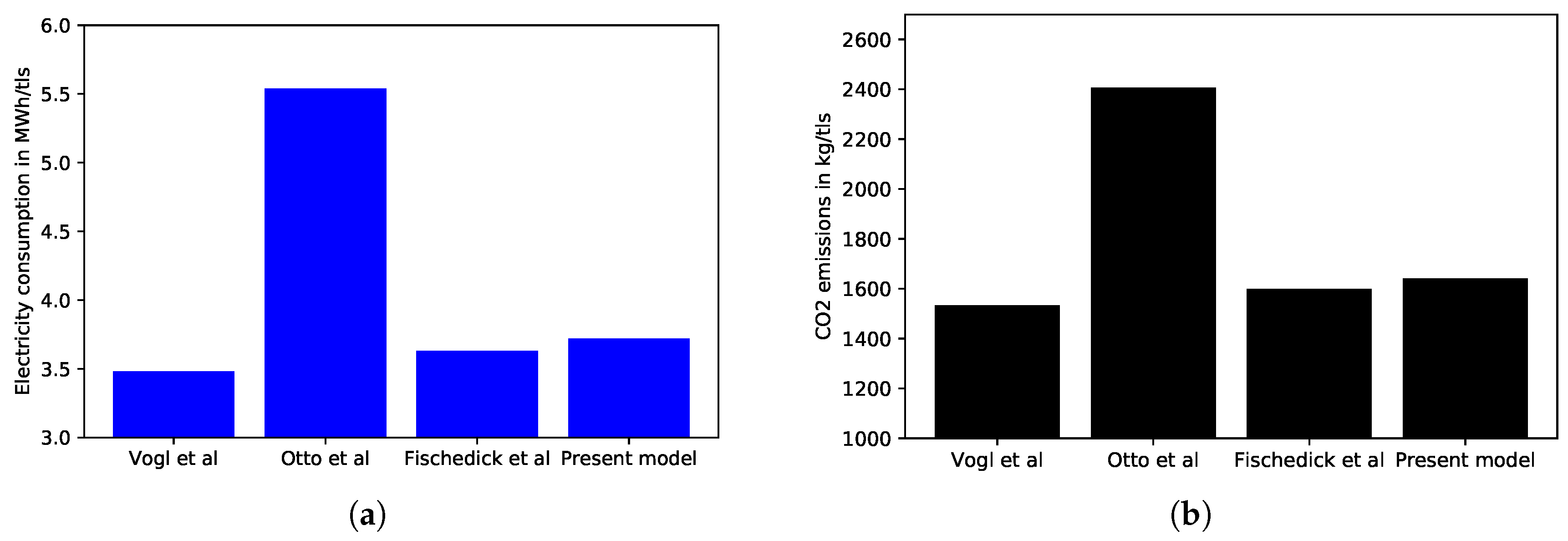
4.4. Comparison with Literature Values
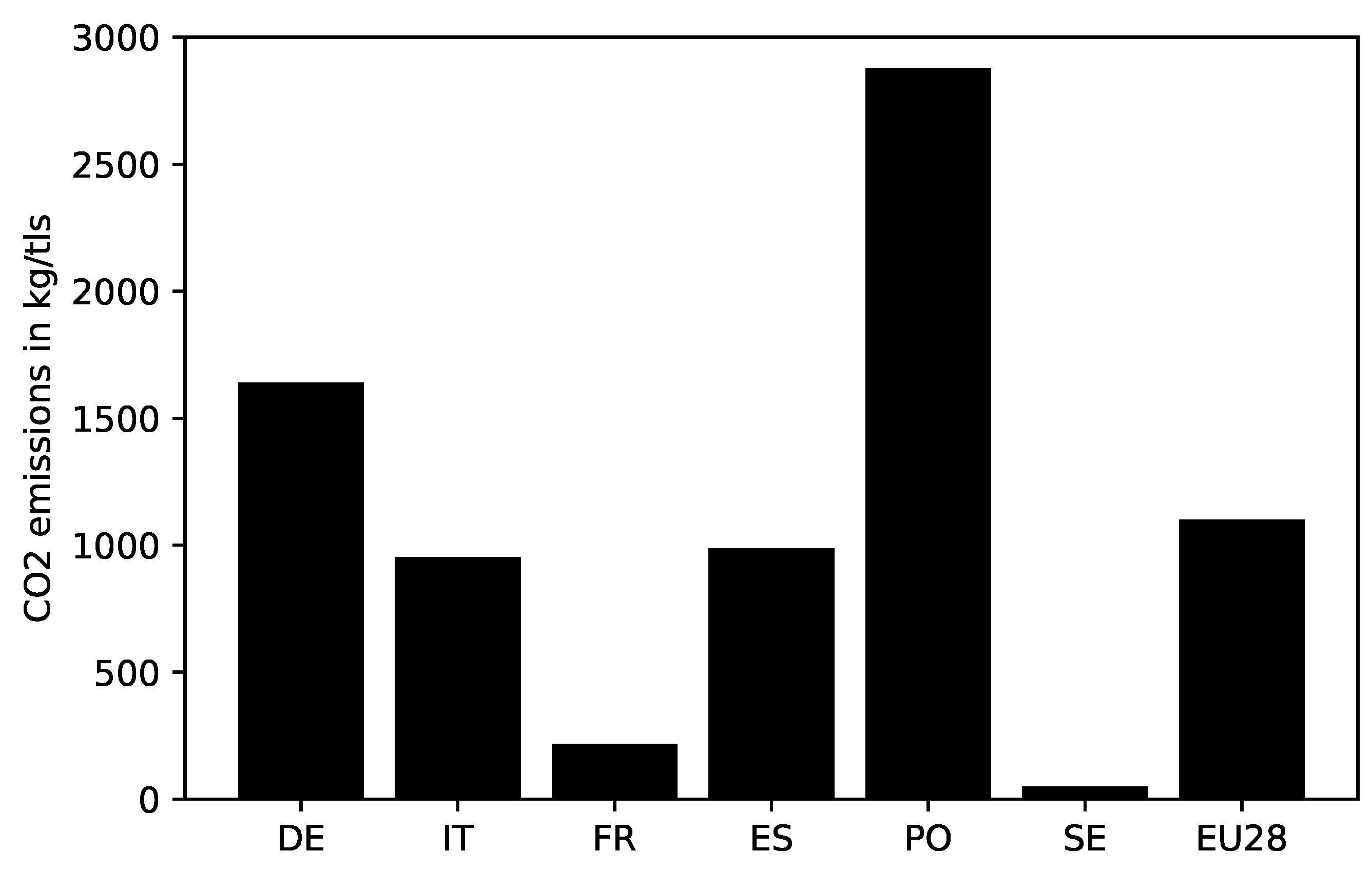
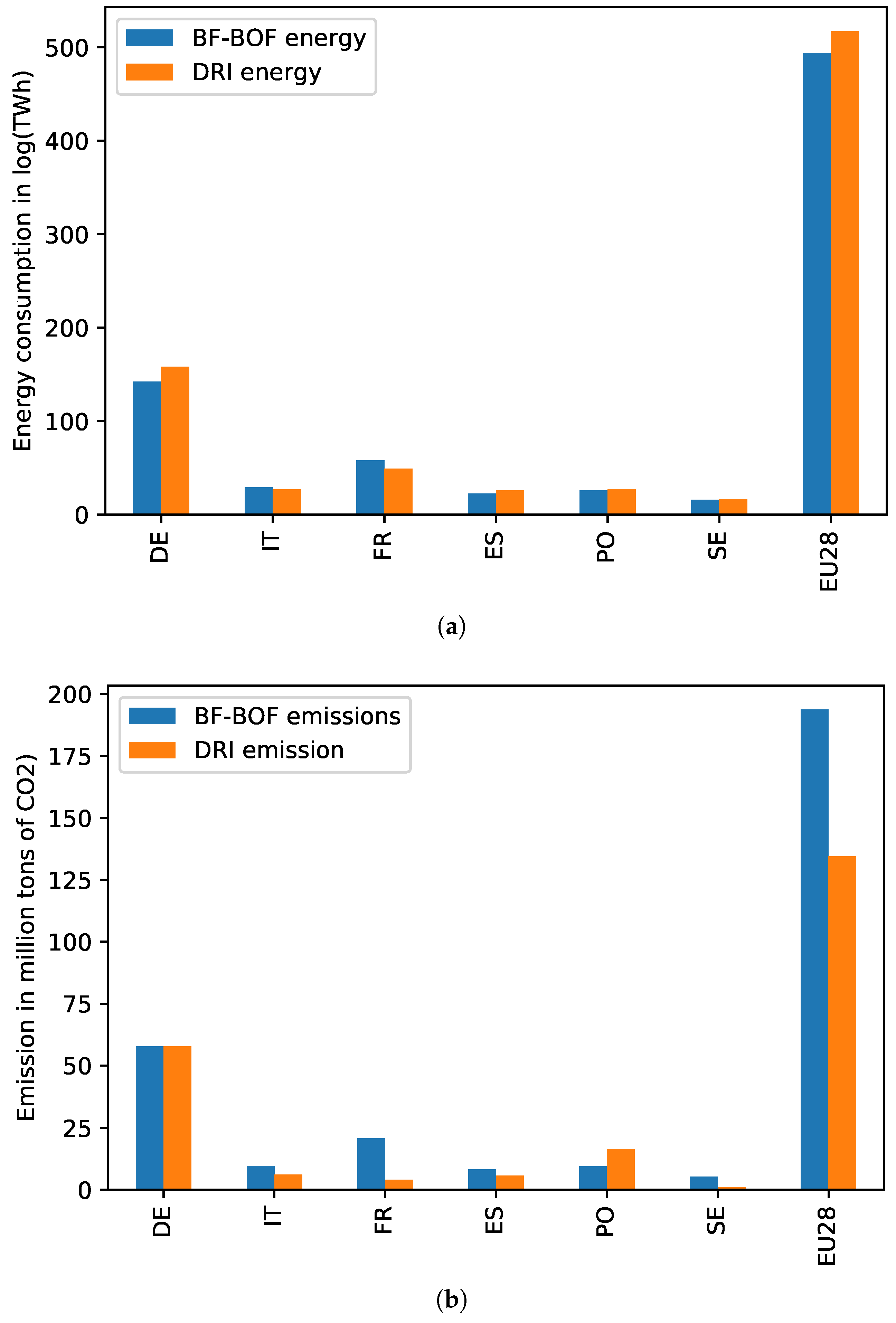
4.5. Energy Consumption and Emissions in EU Countries
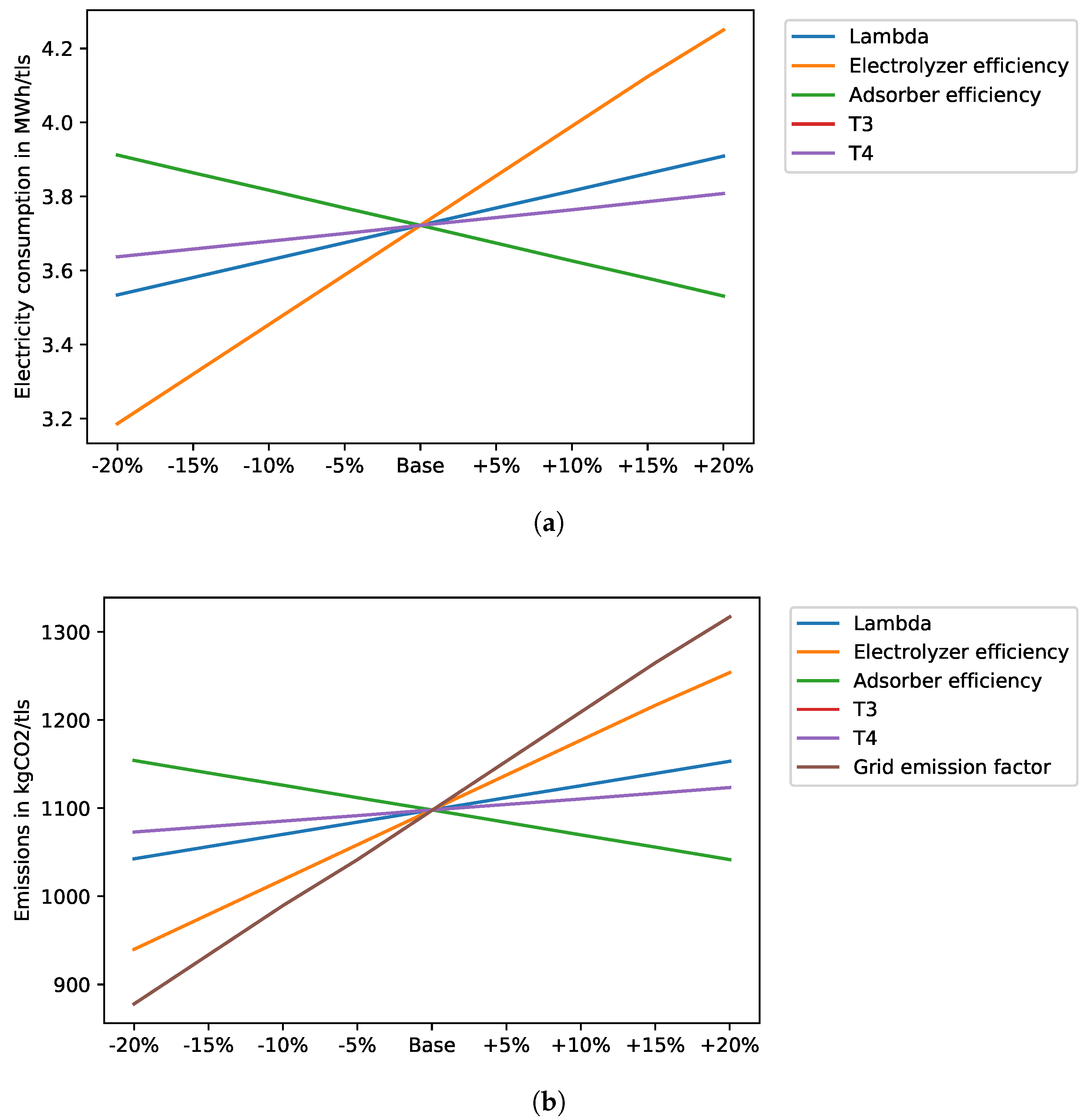
4.6. Sensitivity Analysis
5. Conclusions
6. Future Work
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| Abbreviations | |
| DRI | Direct reduced iron |
| HDRI | Hydrogen direct reduced iron |
| EAF | Electric arc furnace |
| BF-BOF | Blast furnace basic oxygen furnace |
| SMR | Steam methane reforming |
| BASF | Badische Anilin und Soda Fabrik |
| SSAB | Svenskt Stål AB |
| LKAB | Luossavaara-Kiirunavaara Aktiebolag |
| MMBTU | Metric Million British Thermal Unit |
| SEC | Specific energy consumption |
| TLS | Ton of liquid steel |
| kgCO2 | Kilogram of carbon dioxide |
| GtCO2 | Gigaton of carbon dioxide |
| kg | kilogram |
| kJ | KiloJoule |
| KWh | Kilowatthour |
| MWh | Megawatthour |
| $ | US Dollar |
| € | Euro |
| Symbols | |
| Mass of iron ore at heater inlet for production of 1 ton of liquid steel in kg/tls | |
| Mass of iron ore at heater outlet for production of 1 ton of liquid steel in kg/tls | |
| Percentage of pure iron ore in the iron ore stream (assumed to be 0.95) | |
| Ratio of molecular weight of iron contained in iron oxide (0.7) | |
| Electrical energy required for heating the pellets in kJ | |
| Specific enthalpy of iron ore at ambient temperature in kJ/kg | |
| Specific enthalpy of iron ore at reactor temperature in kJ/kg | |
| Efficiency of the electrical heater | |
| Stoichiometric mass flow of hydrogen in kg/tls | |
| 1.5 Moles of hydrogen required for production of one mole of iron | |
| 2.015 g/mol | |
| 55.845 g/mol | |
| Mass of FeO exiting the shaft furnace in kg/tls | |
| Mass of Fe exiting the shaft furnace in kg/tls | |
| Mass of impurities exiting the shaft furnace | |
| Metallization rate | |
| Mass flow of exhaust gases from the shaft furnace in kg/tls | |
| Mass of unused hydrogen as exhaust from the DRI shaft furnace in kg/tls | |
| Mass of water/steam produced as exhaust from the DRI shaft furnace in kg/tls | |
| Specific enthalpy of hydrogen entering the shaft furnace in kJ/kg | |
| Specific enthalpy of metallic stream exiting the shaft furnace in kJ/kg | |
| Specific enthalpy of DRI exhaust gases in kJ/kg | |
| Enthalpy of unreacted hydrogen from the DRI shaft furnace in kJ/kg | |
| Enthalpy of water/steam from the DRI shaft furnace in kJ/kg | |
| Reaction enthalpy of the reduction reaction in kJ/kg | |
| Heat losses | |
| Mass of molten metal from the EAF in Tons | |
| Specific enthalpy of molten metal exiting the EAF in kJ/kg | |
| Mass of scrap from the scrap in kg/tls | |
| Specific enthalpy of scrap exiting the EAF in kJ/kg | |
| Mass of exhaust gases exiting the EAF in kg/tls | |
| Specific enthalpy of exhaust gases exiting the EAF in kJ/kg | |
| Mass of oxygen entering the EAF in kg/tls | |
| Mass of carbon added in the EAF in kg/tls | |
| Mass of lime and dolomite added in the EAF in kg/tls | |
| Efficiency of the EAF for conversion from electricity to heat | |
| Electricity supplied to the EAF in KWh/tls | |
| Mass of water entering the electrolyzer from the waste gas separation unit in kg | |
| Mass of hydrogen entering the electric heater in kg | |
| Enthalpy of hydrogen entering the electrolyzer from the waste gas separation unit in kg | |
| Mass of water supplied to electrolyzer externally in kg | |
| Mass of hydrogen produced in the electrolyzer and supplied to the DRI shaft furnace in kg | |
| Enthalpy of hydrogen produced in the electrolyzer and supplied to the DRI shaft furnace in kg | |
| Mass of hydrogen produced in the electrolyzer and supplied to the hydrogen storage in kg | |
| Mass of oxygen produced in the electrolyzer in kg | |
| Specific energy consumption of the electrolyzer in KWh kg | |
| Electricity consumption in the electrolyzer in KWh | |
| Uncaptured hydrogen exiting the pressure swing adsorber | |
| Electricity consumed for heating the hydrogen stream in KWh/tls |
Appendix A
- All calculations are done for the production of 1 ton of liquid steel from the system.
- Energy consumption and emissions related to iron ore mining, pellet making, and downstream steel finishing steps were not considered in this analysis.
- 5% impurities are present in the raw materials. The assumption is consistent with the plant data available in the literature. The primary components of the impurities are silica and alumina.
- The iron ore pellets are heated from ambient temperature to 800 °C, through an electrical heater of efficiency, .
- Output from the shaft furnace would be metallic Fe and FeO. The remaining FeO will be reduced to pure iron in the electric arc furnace. Although, in practice, some amount of FeO does not get reduced and becomes a part of the EAF slag.
- The flow rate of hydrogen is considered to be higher than the stoichiometric requirements.
- Apparent activation energy of 35 kJ/mole has been considered in this model.
- Hydrogen produced from electrolyzers is heated in an electrical heater with an efficiency of .
- DRI stream exiting the shaft furnace is considered to be at a temperature of 800 °C.
- The exhaust gas stream is assumed to be composed of hydrogen and water. The waste stream enthalpy varies with exhaust gas temperature and .
- Energy required to separate hydrogen and water from the waste stream is not considered in the present calculations.
- 100% DRI is fed into the furnace without any scrap. The quality of scrap has a significant effect on energy consumption in a DRI.
- Hot DRI is fed into the DRI at 700 °C as it saves a considerable amount of electrical energy in the EAF.
- Natural gas is not used for heating the material as its the general practice to use natural gas with scrap for initial heating.
- As DRI is reduced only with Hydrogen, it is assumed that it does not contain any ferric carbide. Carbon required for reduction of remaining FeO in the EAF is supplied externally as coal or coke.
- Temperature of the DRI being fed into the EAF is not taken into account into empirical energy models [74]. Thermodynamic modeling of the EAF has been done to get the specific energy consumption of the EAF with 100% DRI.
- Iron ore pellets generally contain elements such as silicon, manganese, chromium, aluminium, sulphur, phosphorus, molybdenum etc. They get oxidized inside the electric arc furnace, releasing heat and assist in the melting of the iron ore. As iron ore pellets containing only alumina and silica have been considered in this model, additional energy supplied from the oxidation of these elements has not been considered, but a provision for their inclusion in future work has been made in the code.
- Carbon is added into the EAF to reduce the remaining FeO in the mix and also to generate CO for froth formation, which is essential for the operation of the EAF and to extend the life of the graphite electrodes and the refractory.
- CaO and MgO are added in the EAF as slag formers to maintain the basicity of the EAF. The weights of CaO and MgO used are according to data published in the literature [51].
- Efficiency parameters used in the EAF model for electrical and chemical energy are according to the reference [51].
References
- Arens, M.; Worrell, E.; Eichhammer, W.; Hasanbeigi, A.; Zhang, Q. Pathways to a low-carbon iron and steel industry in the medium-term—The case of Germany. J. Clean. Prod. 2017, 163, 84–98. [Google Scholar] [CrossRef]
- UNFCCC’s Technology Executive Commitee; UNFCCC. Industrial Energy and Material Efficiency in Emission-Intensive Sectors; Technical report, United nations framework convention for climate change; UNFCCC’s Technology Executive Commitee: Copenhagen, Denmark, 2017; Available online: https://bit.ly/332v3jl (accessed on 5 January 2020).
- World Steel Association Statistics. 2017. Available online: https://www.worldsteel.org/internet-2017/steel-by-topic/statistics/steel-data-viewer/P1{_}crude{_}steel{_}total/CHN/IND/WORLD{_}ALL/JPN/DEU (accessed on 13 November 2019).
- Arcelor Mittal: Climate Action Report. Available online: https://corporate.arcelormittal.com/sustainability/arcelormittal-climate-action-report?frommobile=true (accessed on 8 September 2019).
- Bataille, C.; Åhman, M.; Neuhoff, K.; Nilsson, L.J.; Fischedick, M.; Lechtenböhmer, S.; Solano-Rodriquez, B.; Denis-Ryan, A.; Stiebert, S.; Waisman, H.; et al. A review of technology and policy deep decarbonization pathway options for making energy-intensive industry production consistent with the Paris Agreement. J. Clean. Prod. 2018, 187, 960–973. [Google Scholar] [CrossRef]
- Åhman, M.; Nilsson, L.J.; Johansson, B. Global climate policy and deep decarbonization of energy-intensive industries. Clim. Policy 2017, 17. [Google Scholar] [CrossRef]
- Fischedick, M.; Marzinkowski, J.; Winzer, P.; Weigel, M. Techno-economic evaluation of innovative steel production technologies. J. Clean. Prod. 2014, 84, 563–580. [Google Scholar] [CrossRef]
- Sarkar, S.; Bhattacharya, R.; Roy, G.G.; Sen, P.K. Modeling MIDREX Based Process Configurations for Energy and Emission Analysis. Steel Res. Int. 2018, 89, 1700248. [Google Scholar] [CrossRef]
- Pardo, N.; Moya, J.A.; Vatopoulos, K. Prospective Scenarios on Energy Efficiency and CO2 Emissions in the EU Iron & Steel Industry; Technical Report LD-NA-25543-EN-C; Joint Research Council, European Union: Luxemburg City, Luxemburg, 2012. [Google Scholar] [CrossRef]
- Wiencke, J.; Lavelaine, H.; Panteix, P.J.; Petitjean, C.; Rapin, C. Electrolysis of iron in a molten oxide electrolyte. J. Appl. Electrochem. 2018, 48, 115–126. [Google Scholar] [CrossRef]
- Stel, J.V.D.; Hattink, M.; Sert, D.; Zagaria, M.; Eklund, N.; Pettersson, M.; Sundqvist, L.; Mefos, B.S.; Feilmayr, C.; Kinnunen, K.; et al. Developments of the ULCOS Low CO2 Blast Furnace Process at the LKAB Experimental BF in Luleå Abstract Key Words History of Recycling of Blast Furnace Top Gas. In Proceedings of the 1st International Conference on Energy Efficiency and CO2 Reduction in the Steel Industry, Dusseldorf, Germany, 1 July 2011; pp. 1–8. [Google Scholar]
- IEA. Technology Roadmap Carbon Capture and Storage in Industrial Applications; IEA: Paris, France, 2011; p. 43. [Google Scholar]
- Hydrogen Future Fuel Cell and Hydrogen Joint Undertaking. Technical Report June. 2019. Available online: https://www.fch.europa.eu/sites/default/files/Hydrogen%20Roadmap%20Europe_Report.pdf (accessed on 5 January 2020).
- The Future of Hydrogen: Seizing Today’s Opportunities. Technical Report June. 2019. Available online: https://webstore.iea.org/the-future-of-hydrogen (accessed on 5 January 2020).
- Hydrogen for Australia’s Future. Technical Report August, Austrlian Government Chief Scientist. 2018. Available online: https://bit.ly/2O0ASJA (accessed on 5 January 2020).
- Philibert, C. Renewable Energy for Industry From Green Energy to Green Materials and Fuels; Technical report; International Energy Agency: Paris, France, 2017; Available online: https://webstore.iea.org/search?q=renewable+energy+for (accessed on 5 January 2020).
- Nuber, D.; Eichberger, H.; Rollinger, B. Circored fine ore direct reduction. Millenium Steel 2006, 2006, 37–40. [Google Scholar]
- Elmquist, S.A.; Weber, P.; Eichberger, H. Operational results of the Circored fine ore direct reduction plant in Trinidad. STAHL UND EISEN 2002, 59–64. Available online: https://www.researchgate.net/publication/288144463_Operational_results_of_the_Circored_fine_ore_direct_reduction_plant_in_Trinidad (accessed on 5 January 2020).
- Duarte, P. Hydrogen-Based Steelmaking; Technical report; TenovaHYL: San Nicolás de los Garza, Mexico, 2015. [Google Scholar]
- Armijo, J.; Philibert, C. Flexible production of green hydrogen and ammonia from variable solar and wind energy. Case study of Chile and Argentina. ResearchGate 2019. [Google Scholar] [CrossRef]
- Glenk, G.; Reichelstein, S. Economics of converting renewable power to hydrogen. Nat. Energy 2019, 4, 216–222. [Google Scholar] [CrossRef]
- The Future of Hydrogen; Number June. OECD: Paris, France, 2019; p. 203. Available online: https://www.oecd-ilibrary.org/energy/the-future-of-hydrogen{_}1e0514c4-en (accessed on 5 February 2020).
- Hybrit Project—Pilot Projects. Available online: http://www.hybritdevelopment.com/articles/three-hybrit-pilot-projects (accessed on 5 January 2020).
- H2future. h2future. Available online: https://www.h2future-project.eu/technology (accessed on 8 September 2019).
- Salcos Salzgitter. Available online: https://salcos.salzgitter-ag.com/ (accessed on 8 September 2019).
- Green Indsustrial Hydrogen: GrInHy2.0. Available online: https://www.green-industrial-hydrogen.com/ (accessed on 8 September 2019).
- Windh2. Available online: https://www.windh2.de/ (accessed on 8 September 2019).
- Brook, D. Tata Steel’s European Operations Take Major Step Towards Becoming Carbon Neutral. 2018. Available online: https://www.tatasteeleurope.com/en/news/news/tata-steel-european-operations-take-major-step-towards-becoming-carbon-neutral (accessed on 8 September 2019).
- ArcelorMittal. World First for Steel: ArcelorMittal Investigates the Industrial Use of Pure Hydrogen—ArcelorMittal. 2019. Available online: https://corporate.arcelormittal.com/news-and-media/news/2019/mar/28-03-2019 (accessed on 5 January 2020).
- Langner, A.; Lorraine, L. ArcelorMittal Commissions Midrex to Design Demonstration Plant for Hydrogen Steel Production in Hamburg. 2019. Available online: https://corporate.arcelormittal.com/news-and-media/news/2019/sep/16-09-2019 (accessed on 18 November 2019).
- Thyssenkrupp. World First in Duisburg as NRW Economics Minister Pinkwart Launches Tests at Thyssenkrupp Into Blast Furnace Use of Hydrogen. 2019. Available online: https://www.thyssenkrupp-steel.com/en/newsroom/press-releases/world-first-in-duisburg.html (accessed on 13 November 2019).
- Otto, A.; Robinius, M.; Grube, T.; Schiebahn, S.; Praktiknjo, A.; Stolten, D. Power-to-steel: Reducing CO2 through the integration of renewable energy and hydrogen into the German steel industry. Energies 2017, 10, 451. [Google Scholar] [CrossRef]
- Cavaliere, P.; Cavaliere, P. Direct Reduced Iron: Most Efficient Technologies for Greenhouse Emissions Abatement; Springer: New York, NY, USA, 2019; pp. 419–484. [Google Scholar] [CrossRef]
- Vogl, V.; Åhman, M.; Nilsson, L.J. Assessment of hydrogen direct reduction for fossil-free steelmaking. J. Clean. Prod. 2018, 203, 736–745. [Google Scholar] [CrossRef]
- Wagner, D.; Devisme, O.; Patisson, F.; Ablitzer, D. A laboratory study of the reduction of iron oxides by hydrogen. 2006 TMS Fall Extr. Process. Divis. Sohn Int. Symp. 2006, 2, 111–120. [Google Scholar]
- Abhinav, B. Hydrogen Direct Reduction of Iron Ore Using Green Hydrogen. Available online: https://zenodo.org/record/3562399#.XjpcSvkzaUk (accessed on 5 January 2020).
- Spreitzer, D.; Schenk, J. Reduction of Iron Oxides with Hydrogen—A Review. Steel Res. Int. 2019, 90, 1900108. [Google Scholar] [CrossRef]
- Hasanbeigi, A.; Price, L.; Chunxia, Z.; Aden, N.; Xiuping, L.; Fangqin, S. Comparison of iron and steel production energy use and energy intensity in China and the U.S. J. Clean. Prod. 2014, 65, 108–119. [Google Scholar] [CrossRef]
- Small, M. Direct Reduction of Iron Ore. J. Metals 1981, 33, 67–75. [Google Scholar] [CrossRef]
- Battle, T.; Srivastava, U.; Kopfle, J.; Hunter, R.; McClelland, J. The Direct Reduction of Iron. Treatise Process Metall. 2014, 3, 89–176. [Google Scholar] [CrossRef]
- Anameric, B.; Kawatra, S.K. Properties and features of direct reduced iron. Mineral Process. Extr. Metall. Rev. 2007, 28, 59–116. [Google Scholar] [CrossRef]
- Béchara, R.; Hamadeh, H.; Mirgaux, O.; Patisson, F. Optimization of the iron ore direct reduction process through multiscale process modeling. Materials 2018, 11, 1094. [Google Scholar] [CrossRef]
- MIDREX. World Direct Reduction Statistics; Midrex Technologies, Inc.: Charlotte, NC, USA, 2013; p. 14. [Google Scholar]
- Cheap Gas to Increase US Direct Reduced Iron Self Sufficiency. 2019. Available online: https://www.reuters.com/article/iron-gas-usa/cheap-gas-to-increase-us-direct-reduced-iron-self-sufficiency-idUSL5N0AYFSC20130129 (accessed on 25 October 2019).
- Carlos, P.E.; Martinis, A.D.; Jorge, B.; Lizcano, C. Energiron direct reduction ironmaking—Economical, flexible, environmentally friendly. Steel Times Int. 2010, 34, 25–30. [Google Scholar]
- Chen, Z.; Dang, J.; Hu, X.; Yan, H. Reduction kinetics of hematite powder in hydrogen atmosphere at moderate temperatures. Metals 2018, 8, 751. [Google Scholar] [CrossRef]
- Van Der Walt, S.; Colbert, S.C.; Varoquaux, G. The NumPy array: A structure for efficient numerical computation. Comput. Sci. Eng. 2011, 13, 22–30. [Google Scholar] [CrossRef]
- Alhumaizi, K.; Ajbar, A.; Soliman, M. Modelling the complex interactions between reformer and reduction furnace in a midrex-based iron plant. Canad. J. Chem. Eng. 2012, 90, 1120–1141. [Google Scholar] [CrossRef]
- Cárdenas, J.G.G.; Conejo, A.N.; Gnechi, G.G. Optimization of Energy Consumption in Electric Arc Furnaces Operated with 100% Dri. Metal 2007, 2007, 1–7. [Google Scholar]
- Rojas-Cardenas, J.C.; Hasanbeigi, A.; Sheinbaum-Pardo, C.; Price, L. Energy efficiency in the Mexican iron and steel industry from an international perspective. J. Clean. Prod. 2017, 158, 335–348. [Google Scholar] [CrossRef]
- Dİlmaç, N.; Yörük, S.; Gülaboğlu, Ů.M. Investigation of Direct Reduction Mechanism of Attepe Iron Ore by Hydrogen in a Fluidized Bed. Metall. Mater. Trans. B Process Metall. Mater. Process. Sci. 2015, 46, 2278–2287. [Google Scholar] [CrossRef]
- Pineau, A.; Kanari, N.; Gaballah, I. Kinetics of reduction of iron oxides by H2. Part II. Low temperature reduction of magnetite. Thermochim. Acta 2007, 456, 75–88. [Google Scholar] [CrossRef]
- Teplov, O.A. Kinetics of the low-temperature hydrogen reduction of magnetite concentrates. Russian Metall. (Metally) 2012, 2012, 8–21. [Google Scholar] [CrossRef]
- Fruehan, R.J.; Li, Y.; Brabie, L.; Kim, E.J. Final stage of reduction of iron ores by hydrogen. Scand. J. Metall. 2005, 34, 205–212. [Google Scholar] [CrossRef]
- Guo, D.; Hu, M.; Pu, C.; Xiao, B.; Hu, Z.; Liu, S.; Wang, X.; Zhu, X. Kinetics and mechanisms of direct reduction of iron ore-biomass composite pellets with hydrogen gas. Int. J. Hydr. Energy 2015, 40, 4733–4740. [Google Scholar] [CrossRef]
- Ranzani da Costa, A.; Wagner, D.; Patisson, F. Modelling a new, low CO2 emissions, hydrogen steelmaking process. J. Clean. Prod. 2013, 46, 27–35. [Google Scholar] [CrossRef]
- Zuo, H.-B.; Wang, C.; Dong, J.-J.; Jiao, K.-X.; Xu, R.-S. Reduction kinetics of iron oxide pellets with H2 and CO mixtures. Int. J. Miner. Metall. Mater. 2015, 22, 688–696. [Google Scholar] [CrossRef]
- Pérez, F.; Granger, B.E. IPython: A system for interactive scientific computing. Comput. Sci. Eng. 2007, 9, 21–29. [Google Scholar] [CrossRef]
- McKinney, W. Data Structures for Statistical Computing in Python. In Proceedings of the 9th Python in Science Conference, Austin, TX, USA, 28 June–3 July 2010; pp. 51–56. [Google Scholar]
- Hunter, J.D. Matplotlib: A 2D graphics environment. Comput. Sci. Eng. 2007, 9, 99–104. [Google Scholar] [CrossRef]
- Anaconda software distribution, Computer software. 2016. Available online: https://www.anaconda.com/ (accessed on 5 January 2020).
- Schmidt, O.; Gambhir, A.; Staffell, I.; Hawkes, A.; Nelson, J.; Few, S. Future cost and performance of water electrolysis: An expert elicitation study. Int. J. Hydr. Energy 2017, 42, 30470–30492. [Google Scholar] [CrossRef]
- Hydrogen, N. Nel ASA: Receives 4.5 MW Electrolyzer Purchase Order for Fossil Free Steel Production. 2019. Available online: https://nelhydrogen.com/press-release/nel-asa-receives-4-5-mw-electrolyzer-purchase-order-for-fossil-free-steel-production/ (accessed on 12 November 2019).
- Thyssenkrupp. Hydrogen From Large-Scale Electrolysis—Efficienct Solutions For Sustainable Chemicals and Energy Storage. Available online: https://d13qmi8c46i38w.cloudfront.net/media/UCPthyssenkruppBAISUhdeChlorineEngineers/assets.files/products/water{_}electrolysis/thyssenkrupp{_}electrolytic{_}hydrogen{_}brochure.pdf (accessed on 5 January 2020).
- Santos Diogo, M.F.; Sequeira Cesar, A.C.; Figueiredo, J.L. Hydrogen production by alkaline water electrolysis. Quim. Nova Rev. 2013, 36, 1176–1193. [Google Scholar] [CrossRef]
- Rashid, M.M.; Mesfer, M.K.A.; Naseem, H.; Danish, M. Hydrogen Production by Water Electrolysis: A Review of Alkaline Water Electrolysis, PEM Water Electrolysis and High Temperature Water Electrolysis. Int. J. Eng. Adv. Technol. 2015, 4, 2249–8958. [Google Scholar]
- Zeng, K.; Zhang, D. Recent progress in alkaline water electrolysis for hydrogen production and applications. Progress Energy Combust. Sci. 2010, 36, 307–326. [Google Scholar] [CrossRef]
- Zare, A.; Khanipour, M.; Sarverstani, H.K.; Kakavandi, I.A.; Shokroo, E.J.; Farniaei, M.; Baghbani, M. Hydrogen and carbon dioxide recovery from the petrochemical flare gas to methanol production using adsorption and absorption combined high-efficient method. Appl. Petrochem. Res. 2019, 9, 127–145. [Google Scholar] [CrossRef]
- Grande, C.A. Advances in Pressure Swing Adsorption for Gas Separation. ISRN Chem. Eng. 2012, 2012, 1–13. [Google Scholar] [CrossRef]
- Song, C.; Liu, Q.; Ji, N.; Kansha, Y.; Tsutsumi, A. Optimization of steam methane reforming coupled with pressure swing adsorption hydrogen production process by heat integration. Appl. Energy 2015, 154, 392–401. [Google Scholar] [CrossRef]
- Mondal, M.; Datta, A. Energy transfer in hydrogen separation from syngas using pressure swing adsorption (PSA) process: A thermodynamic model. Int. J. Energy Res. 2017, 41, 448–458. [Google Scholar] [CrossRef]
- Mantzos, L.; Wiesenthal, T.; Matei, N.A.; Tchung-Ming, S.; Rozsai, M.; Russ, H.P.; Soria Ramirez, A. JRC-IDEES: Integrated Database of the European Energy Sector: Methodological Note; Technical report; Joint Research Council, European Union: Luxemburg, 2017. [Google Scholar] [CrossRef]
- Hamby, D.M. A review of techniques for parameter sensitivity analysis of environmental models. Environ. Monit. Assess. 1994, 32, 135–154. [Google Scholar] [CrossRef] [PubMed]
- Pfeifer, H.; Kirschen, M. Thermodynamic analysis of EAF energy efficiency and comparison with a statical model of electric energy model of demand. Engineering 2003, 2003, 1–16. [Google Scholar]
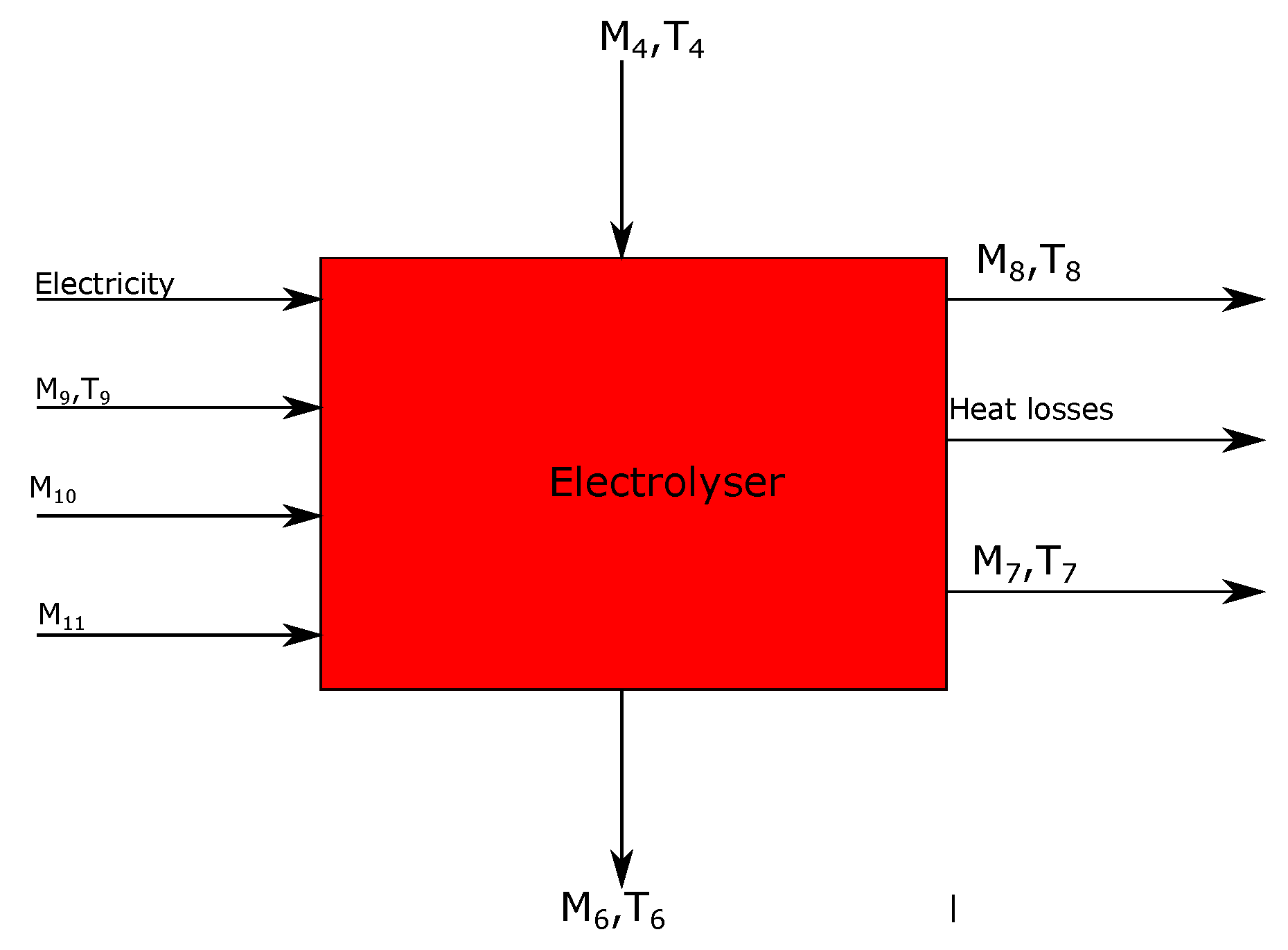
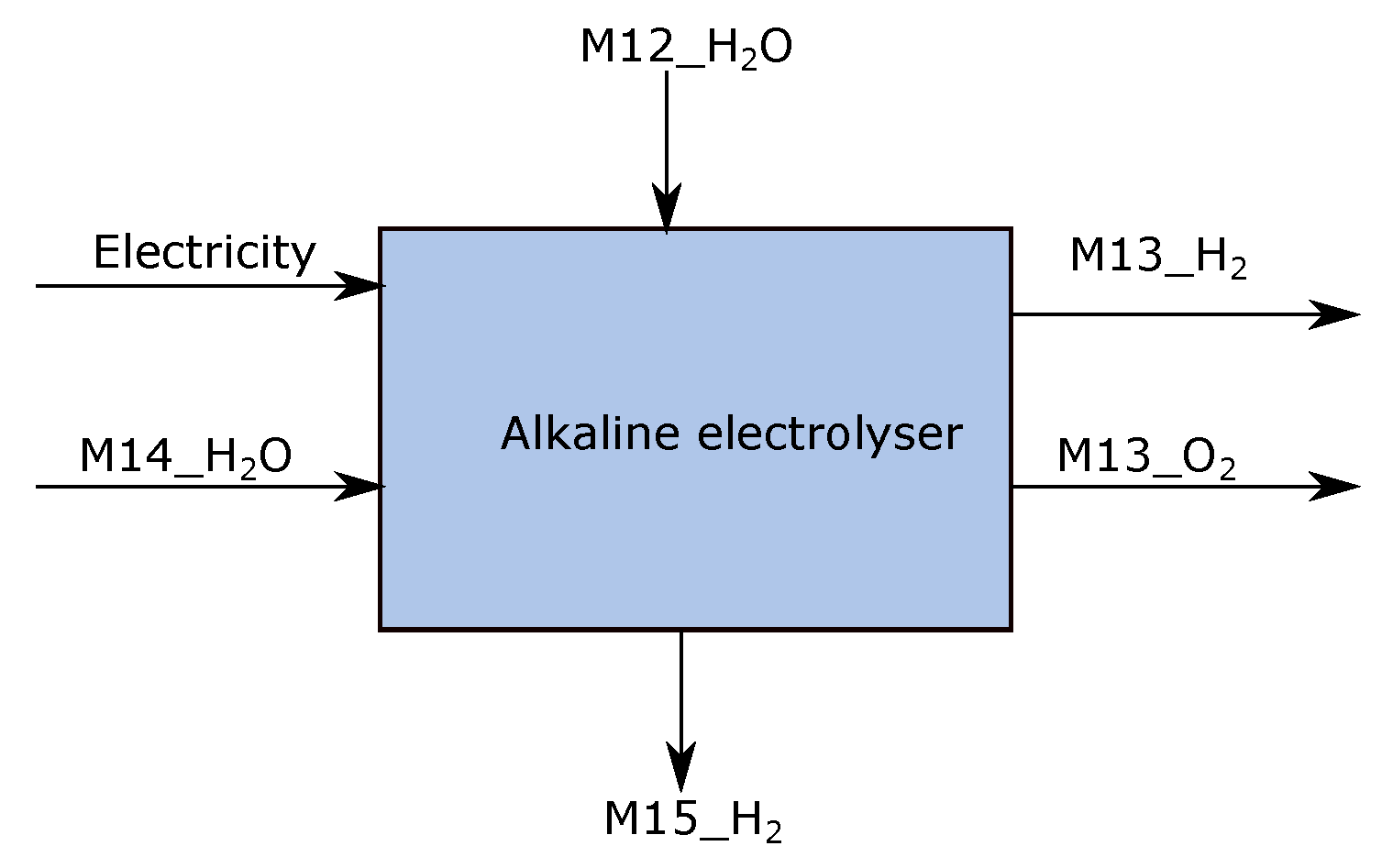
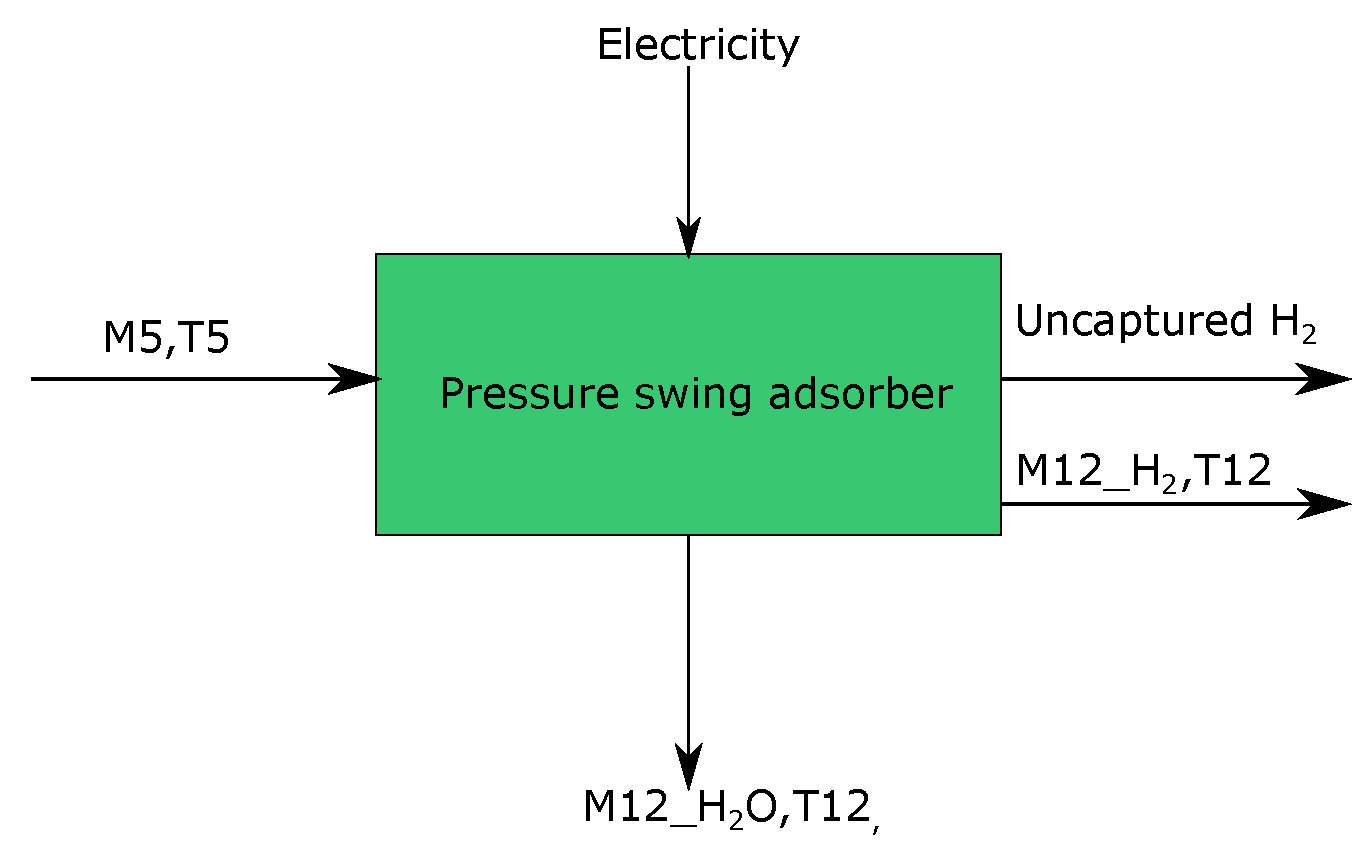
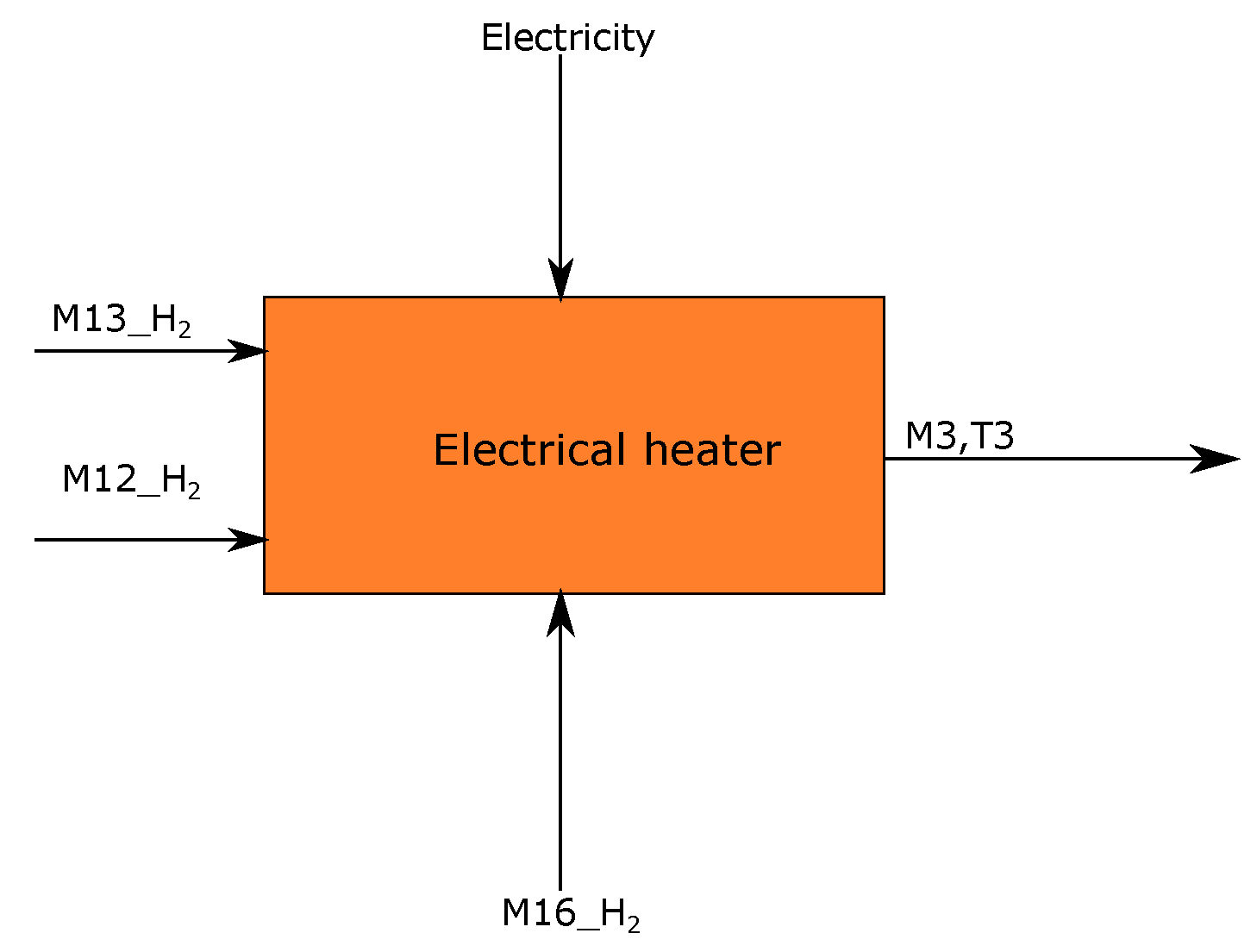
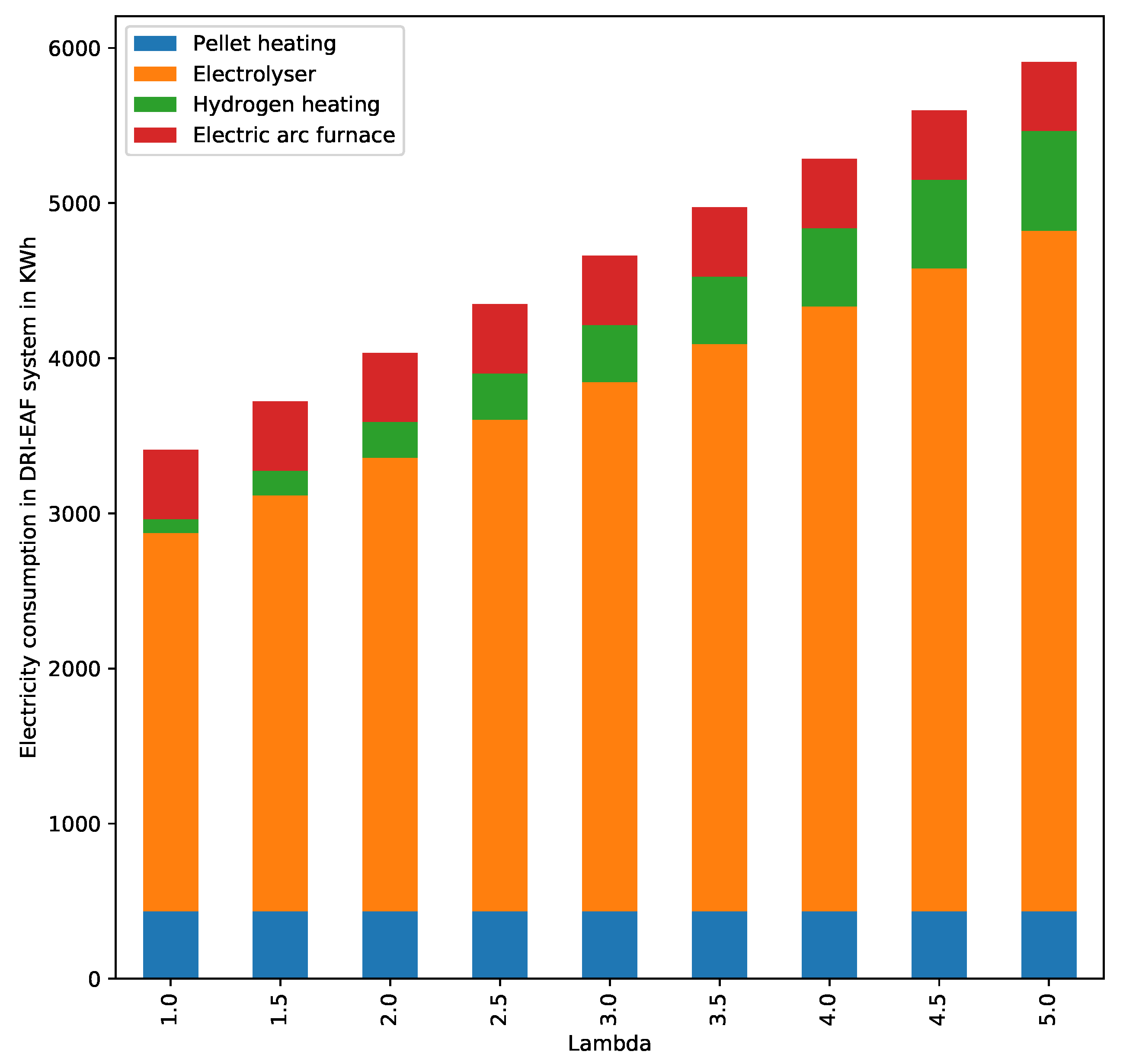
| Stream | Mass Flow (ton/tls) | Temperature (°C) | Energy (KWh) | Short Description | Process Step |
|---|---|---|---|---|---|
| 1.599 | 25 | N.A | Iron ore pellets | Pellet heater | |
| 1.599 | 800 | 370.78 | Heated iron ore pellets | Pellet heater | |
| 0.0812 | 500 | 155.59 | H entering the shaft furnace | Shaft furnace | |
| 1.063 | 700 | 107.498 | Metallic stream exiting the shaft furnace | Shaft furnace | |
| 0.027 | 250 | 24.45 | H from waste stream | Shaft furnace | |
| 0.483 | 250 | 82.18 | HO from Waste stream | Shaft furnace | |
| 1 | 1650 | 239.15 | Molten steel exiting the EAF | EAF | |
| 0.149 | 1650 | 54.25 | Slag exiting the EAF | EAF | |
| 0.021 | 250 | 5.613 | H exiting the adsorber | Adsorber | |
| 0.483 | 90 | N.A | HO exiting the adsorber | Adsorber | |
| 0.059 | 90 | 53.80 | H from electrolyzer | Electrolyzer | |
| 0.171 | 25 | N.A | HO entering the electrolyzer | Electrolyzer |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhaskar, A.; Assadi, M.; Nikpey Somehsaraei, H. Decarbonization of the Iron and Steel Industry with Direct Reduction of Iron Ore with Green Hydrogen. Energies 2020, 13, 758. https://doi.org/10.3390/en13030758
Bhaskar A, Assadi M, Nikpey Somehsaraei H. Decarbonization of the Iron and Steel Industry with Direct Reduction of Iron Ore with Green Hydrogen. Energies. 2020; 13(3):758. https://doi.org/10.3390/en13030758
Chicago/Turabian StyleBhaskar, Abhinav, Mohsen Assadi, and Homam Nikpey Somehsaraei. 2020. "Decarbonization of the Iron and Steel Industry with Direct Reduction of Iron Ore with Green Hydrogen" Energies 13, no. 3: 758. https://doi.org/10.3390/en13030758
APA StyleBhaskar, A., Assadi, M., & Nikpey Somehsaraei, H. (2020). Decarbonization of the Iron and Steel Industry with Direct Reduction of Iron Ore with Green Hydrogen. Energies, 13(3), 758. https://doi.org/10.3390/en13030758







