Hydrogen Fuel Cell Road Vehicles and Their Infrastructure: An Option towards an Environmentally Friendly Energy Transition
Abstract
1. Introduction
2. FCV: Maturity Level of Technological Bricks and Associated Trends
2.1. Lifetime
2.2. Cost
2.3. Platinium
2.4. Road FCV Roadmap
3. Development of an Environment Friendly H2 Infrastructure
3.1. H2 Production
3.1.1. Grey Hydrogen
3.1.2. Blue Hydrogen
3.1.3. Green Hydrogen
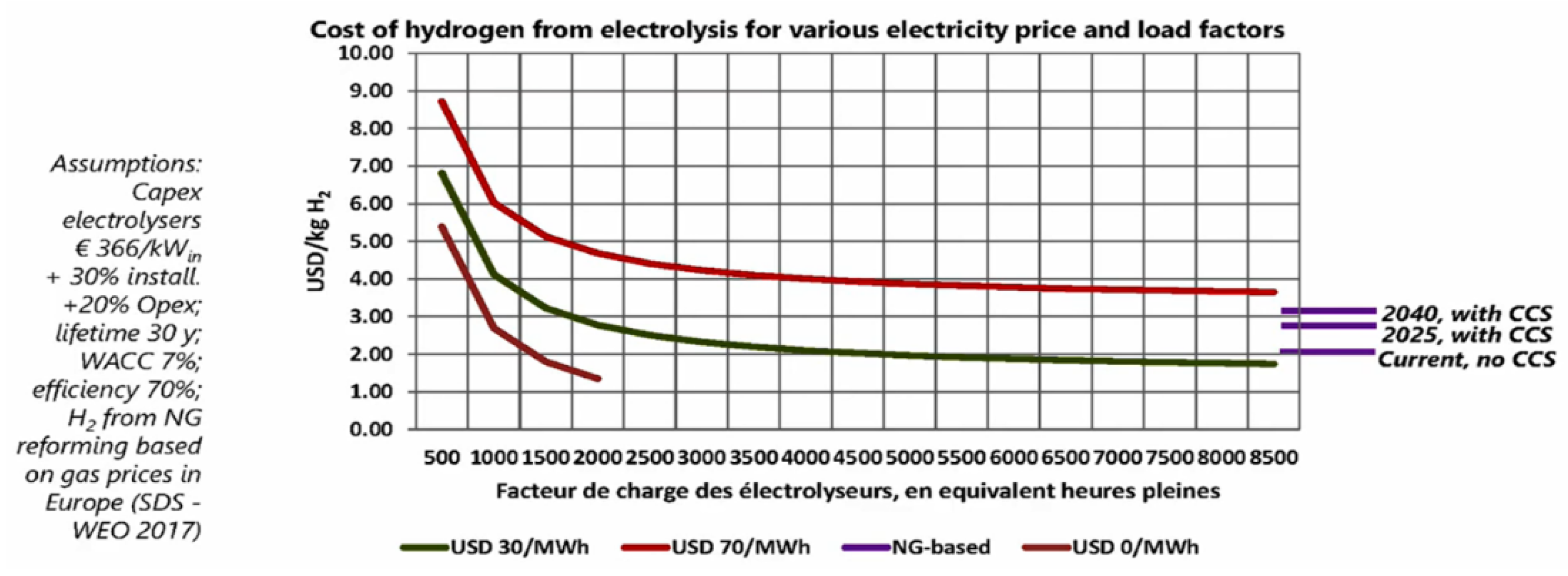
Water Electrolysis Based on Renewable Electricity
Biomass-Based H2 generation
The Thermochemical Biomass Conversion Technology
Biological Hydrogen Production
Transformation of Methanol to Hydrogen
Natural Hydrogen
3.2. H2 Storage
3.2.1. Gaseous Storage
3.2.2. Liquid Storage
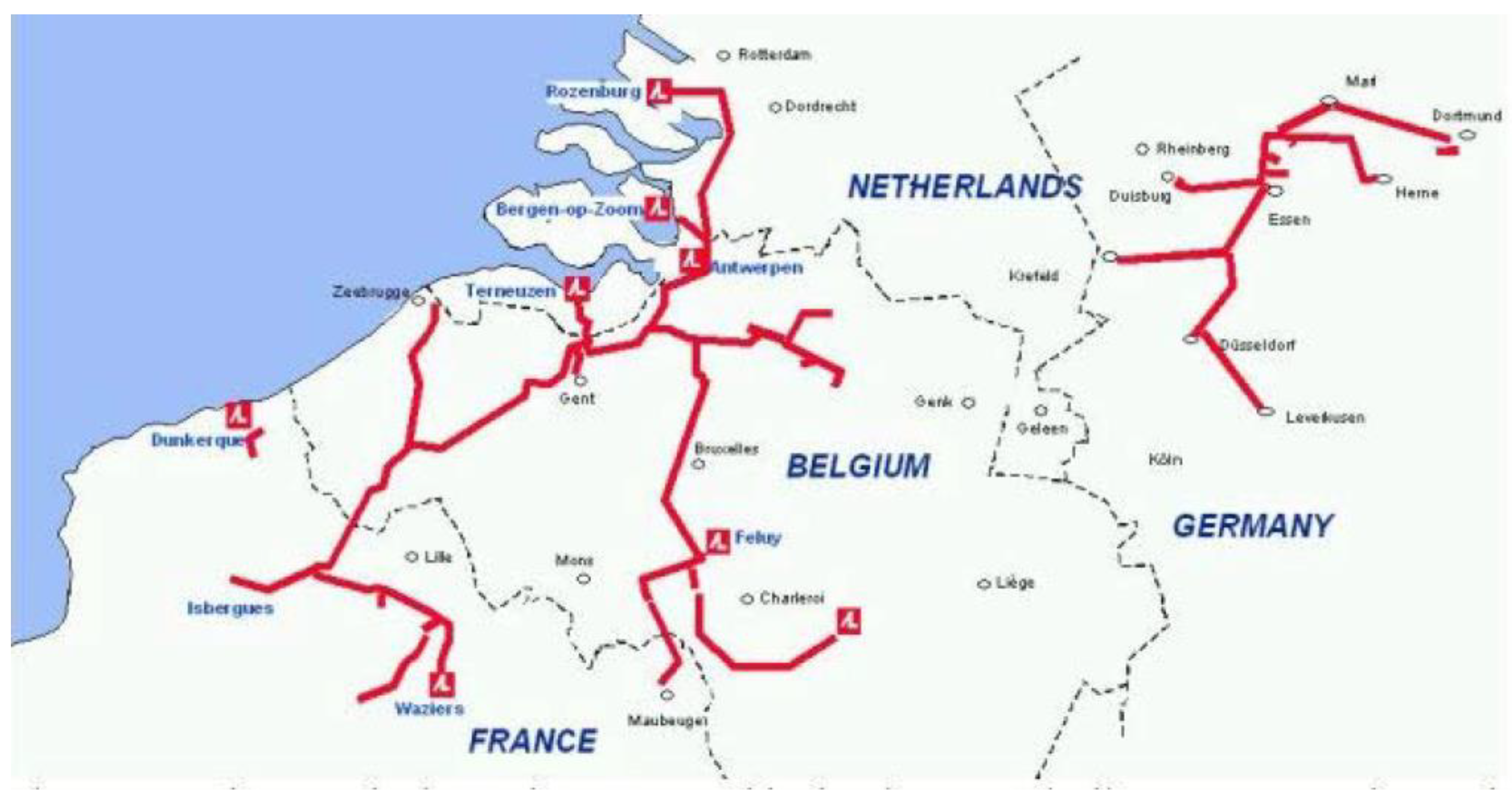
3.3. H2 Transport
3.4. H2 distribution
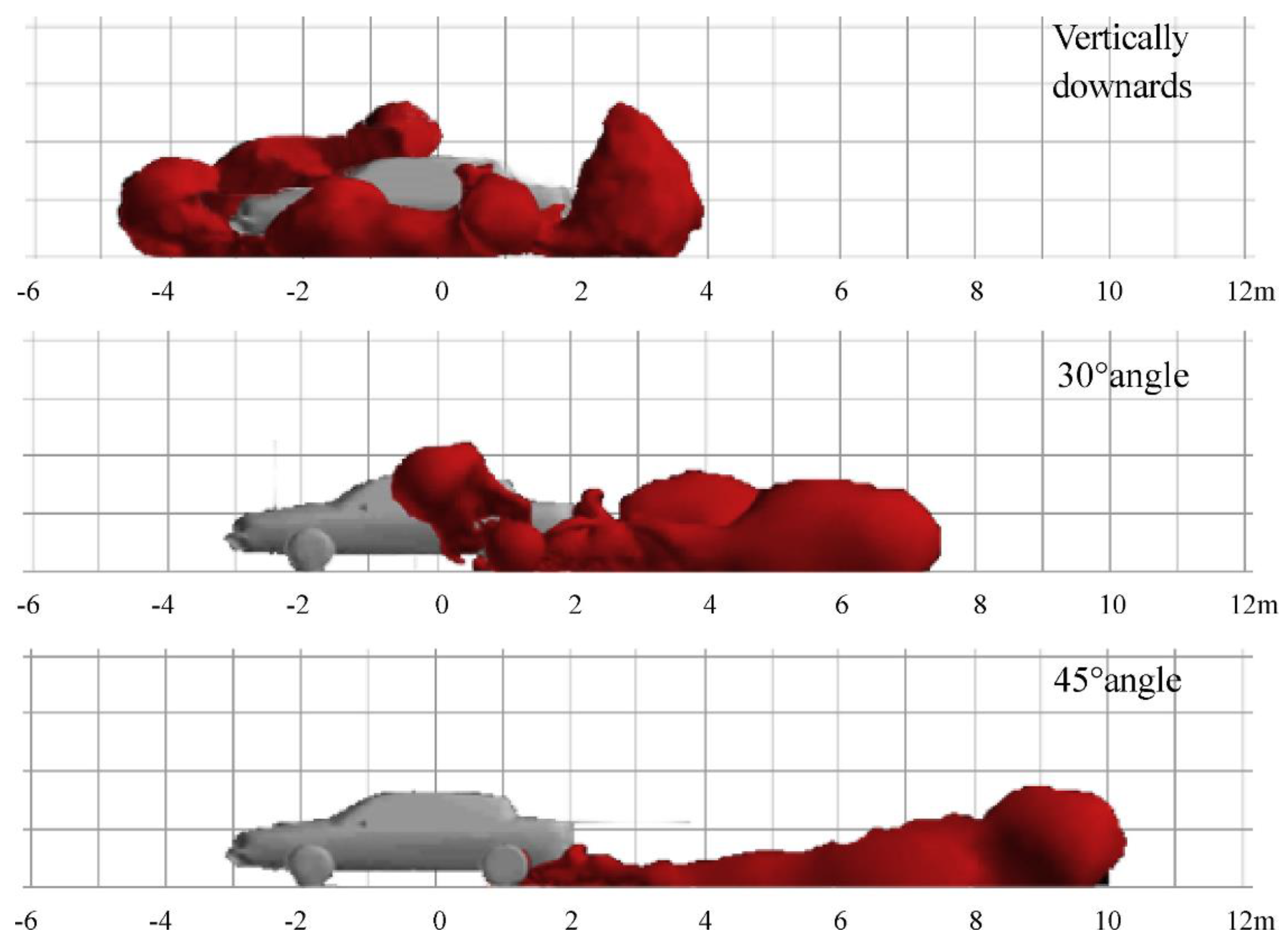
3.5. H2 Safety
4. Conclusions
- First, onboard FCS still requires further enhancements in term of cost, durability and environmental footprint. Mainly, the physical (mass transport) and electrochemical phenomena occurring at nano, micro and milli scales in the active layer of the FC electrode still need better fundamental understanding. Previous continuous progress and current investigation efforts promise many new conceptual advances which would initiate real technological breakthroughs. At the beginning of next decade, it would be reasonable to expect that FCS will address low cost, high durability car/truck specifications. At that point, mature technology could enable to develop environmental-friendly manufacturing and disposal processes. Apart from the FC, the H2 storage also requires both an improvement of its life cycle and new shape to enhance its integration capacity inside the vehicle.
- Second, hydrogen production route reveals to be a crucial choice to ensure a positive impact on energy consumption and GHG emissions. Electrolysis is one of the pathway since it is based on excess of renewable electricity. Similarly, biomass-based hydrogen generation is expected to be attractive processes provided that biomass feedstock relies on wastes. In both cases, production on an industrial scale of such green hydrogen will gradually grow up. Obviously, regarding global footprint reduction, the first best use of such green hydrogen is to convert present grey industrial use into green one. Consequently, green hydrogen as an energy carrier for transportation should actually start in the next decade. Basically, this lack of an immediate green hydrogen is a bottleneck to low-carbon FCV development but is fully in line with the current forecast of the FCV commercial maturity.
- Third, hydrogen-powered transportation and especially road vehicles sector has to face safety issue in order to warranty lower probability of hazardous accidents than standard gasoline option. This issue has already been successfully addressed for more than a century by the chemical industry which has develop a reliable and safe infrastructure (production—transport—storage—distribution) for its own usage of hydrogen (as a process agent). However, switching from these highly centralized applications managed by skilled staff to a consumer product used in various operational conditions is a challenge. Research studies show real opportunities to control risk. Taking into account any hazardous circumstances without significantly impacting the costs use and maintenance complexity remains an open question. That is the reason why the first step to really develop hydrogen mobility in intensive applications is to start with captive fleets (transport and distribution truck fleets, bus fleets and company utility vehicle fleets). These experiments, although limited in scope, focus on use cases that can be comprehensively monitored and studied. Their concrete deployment, with its unprecedented articulation of the entire sector involved in this mobility, will allow manufacturers to capitalize on real feedback and thus to learn and build the best trade-offs with a view to generalization. These pre-industrial demonstrators, by improving technological knowledge, working on the question of societal acceptance and revealing the true ways of using every device of the value chain, will allow technological solutions to emerge at limited cost and appropriate regulations to answer the crucial question of security.
- Finally, the key outcome is that the beginning of large-scale commercialization of hydrogen-powered vehicles will possibly begin in the 2030–2035 years when the yet unresolved questions will be fully addressed. In the meantime, it will be crucial to design the whole hydrogen economy to achieve a low environmental footprint. Achieving consistent comparative studies of each transportation option will enable to define efficient deployment strategies. It requires steady data set based on both well-established processes and business models. In this respect, the present pilot projects will also enable to draw initial meaningful conclusions on the benefits and limits of hydrogen-powered transportation sector.
Funding
Conflicts of Interest
Abbreviations
| ADEME | French Environment and Energy Management |
| APU | Auxiliary Power Unit |
| AWE | Alkaline Water Electrolyzer |
| BEV | Battery Electric Vehicle |
| BP | Bipolar plate |
| CAPEX | Capital Expenditure |
| CCUS | Carbon Capture, Utilization and Storage |
| D | Diesel |
| DICEV | Diesel Internal Combustion Engine Vehicles |
| DoE | US Department of Energy |
| EC | Energy Consumption |
| EOT | Equivalent Outage Time |
| FCS | Fuel Cell Systems |
| FCH-JU | Fuel Cells and Hydrogen - Joint Undertaking |
| FCV | Fuel cell vehicle |
| G | Gasoline |
| GICEV | Gasoline Internal Combustion Engine Vehicles |
| GHG | Greenhouse Gas |
| H2 | Dihydrogen |
| HCV | Higher Calorific Value (or gross calorific value) |
| HGV | Heavy Good Vehicle |
| ICEV | Internal Combustion Engine Vehicle |
| IEA | International Energy Agency |
| JRC | Joint Research Centre of the European commission |
| LCA | Life Cycle Assessment |
| LCV | Lower Calorific Value (or net calorific value) |
| MEA | Membrane Electrode Assembly |
| OPEX | Operational Expenditure |
| PEME | Proton Exchange Membrane Electrolysers |
| PEMFC | Proton exchange membrane fuel cell |
| PGM | Platinum-Group Metal (Ru, Rh, Pd, Os, Ir and Pt) |
| Pt | Platinum |
| PFA | French automotive industry players association |
| RTE | The electricity transmission system operator of France |
| SOE | Solid Oxide Electrolyzer |
| TPRD | Thermal Pressure Relief Device |
| TRL | Technology Readiness Level |
| V | Vehicle |
| WtW | Well-to-Wheel |
| ZE | Zero-Emission |
| ZEV | Zero-Emission Vehicle |
References
- Thompson, S.T.; James, B.D.; Huya-Kouadio, J.M.; Houchins, C.; DeSantis, D.A.; Ahluwalia, R.; Wilson, A.R.; Kleen, G.; Papageorgopoulos, D. Direct hydrogen fuel cell electric vehicle cost analysis: System and high-volume manufacturing description, validation, and outlook. J. Power Sources 2018, 399, 304–313. [Google Scholar] [CrossRef]
- Béthoux, O. Hydrogen Fuel Cell Road Vehicles: State of the Art and Perspectives. Energies 2020, 13, 5843. [Google Scholar] [CrossRef]
- Marx, N.; Hissel, D.; Gustin, F.; Boulon, L.; Agbossou, K. On Maximizing the Steady-State Efficiency of a Multi-Stack Fuel Cell System. In Proceedings of the 2018 IEEE Vehicle Power and Propulsion Conference (VPPC), Chicago, IL, USA, 27–30 August 2018; pp. 1–6. [Google Scholar] [CrossRef]
- Marx, N.; Hissel, D.; Gustin, F.; Boulon, L.; Agbossou, K. On the sizing and energy management of an hybrid multistack fuel cell—Battery system for automotive applications. Int. J. Hydrog. Energy 2017, 42, 1518–1526. [Google Scholar] [CrossRef]
- Marx, N.; Cárdenas, D.C.T.; Boulon, L.; Gustin, F.; Hissel, D. Degraded mode operation of multi-stack fuel cell systems. IET Electr. Syst. Transp. 2016, 6, 3–11. [Google Scholar] [CrossRef]
- de Bernardinis, A.; Frappé, E.; Béthoux, O.; Marchand, C.; Coquery, G. Multi-port Power Converter for Segmented PEM Fuel Cell in Transport Application. Simulation with fault tolerant strategy. Eur. Phys. J. Appl. Phys. 2012, 58, 1–15. [Google Scholar] [CrossRef]
- Eckert, V.; Revill, J. Powered by Hydrogen, Hyundai’s Trucks Aim to Conquer the Swiss Alps; Reuters Agency: London, UK, 2020. [Google Scholar]
- Yang, Z.; Wang, B.; Jiao, K. Life cycle assessment of fuel cell, electric and internal combustion engine vehicles under different fuel scenarios and driving mileages in China. Energy 2020, 198, 117365. [Google Scholar] [CrossRef]
- Evangelisti, S.; Tagliaferri, C.; Brett, D.J.L.; Lettieri, P. Life cycle assessment of a polymer electrolyte membrane fuel cell system for passenger vehicles. J. Clean. Prod. 2017, 142, 4339–4355. [Google Scholar] [CrossRef]
- Bekel, K.; Pauliuk, S. Prospective cost and environmental impact assessment of battery and fuel cell electric vehicles in Germany. Int. J. Life Cycle Assess. 2020, 25, 831. [Google Scholar] [CrossRef]
- Lotrič, A.; Sekavčnik, M.; Kuštrin, I.; Mori, M. Life-cycle assessment of hydrogen technologies with the focus on EU critical raw materials and end-of-life strategies. Int. J. Hydrog. Energy 2020. in Press. [Google Scholar] [CrossRef]
- Pollet, B.G.; Kocha, S.S.; Staffell, I. Current status of automotive fuel cells for sustainable transport. Curr. Opin. Electrochem. 2019, 16, 90–95. [Google Scholar] [CrossRef]
- Duclos, L.; Chattot, R.; Dubau, L.; Thivel, P.; Mandil, G.; Laforest, V.; Bolloli, M.; Vincent, R.; Svecova, L. Closing the loop: Life cycle assessment and optimization of a PEMFC platinum-based catalyst recycling process, 2020. Green Chem. 2020, 22, 1919–1933. [Google Scholar] [CrossRef]
- Benitez, A.; Wulf, C.; de Palmenaer, A.; Lengersdorf, M.; Röding, T.; Grube, T.; Robinius, M.; Stolten, D.; Kuckshinrichs, W. Ecological assessment of fuel cell electric vehicles with special focus on type IV carbon fiber hydrogen tank. J. Clean. Prod. 2021, 278, 123277. [Google Scholar] [CrossRef]
- Edwards, R.; Hass, H.; Larivé, J.; Lonza, L.; Maas, H.; Rickeard, D. Well-to-Wheels Analysis of Future Automotive Fuels and Powertrains in the European Context. European Commission Joint Research Center, Scientific and Technical Research Report. Available online: https://core.ac.uk/download/pdf/38627607.pdf (accessed on 22 November 2020).
- U.S. DRIVE. Fuel Cell Technical Team Roadmap. November 2017. Available online: https://www.energy.gov/sites/prod/files/2017/11/f46/FCTT_Roadmap_Nov_2017_FINAL.pdf (accessed on 22 November 2020).
- DOE. Hydrogen and Fuel Cells Program, 2019 Annual Merit Review and Peer Evaluation Report. Available online: https://www.hydrogen.energy.gov/annual_review19_report.html (accessed on 22 November 2020).
- Hydrogen Roadmap Europe, Report; Fuel Cells and Hydrogen—Joint Undertaking (FCH-JU): Saint-Gilles, Belgium, 2019; ISBN 978-92-9246-331-1. EG-06-18-327-EN-N, fch.europa.eu. [CrossRef]
- Hydrogène et Piles à Combustible: Position Technique de Filière. Available online: https://pfa-auto.fr/wp-content/uploads/2020/02/CRA1-PTF-HYDROGENE-ET-PILES-A-COMBUSTIBLE-VF2.pdf (accessed on 22 November 2020).
- Borup, R.L.; Kusoglu, A.; Neyerlin, K.C.; Mukundan, R.; Ahluwalia, R.K.; Cullen, D.A.; More, K.L.; Weber, A.Z.; Myers, D.J. Recent developments in catalyst-related PEM fuel cell durability. Curr. Opin. Electrochem. 2020, 21, 192–200. [Google Scholar] [CrossRef]
- Wang, J.; Wang, H.; Fan, Y. Techno-economic challenges of fuel cell commercialization. Engineering 2018, 4, 352–360. [Google Scholar] [CrossRef]
- Bal, B.; Sahin, I.; Uzun, A.; Canadinc, D. A New Venue toward Predicting the Role of Hydrogen Embrittlement on Metallic Materials. Metall. Mater. Trans. A 2016, 47, 5409–5422. [Google Scholar] [CrossRef]
- Leng, Y.; Ming, P.; Yang, D.; Zhang, C. Stainless steel bipolar plates for proton exchange membrane fuel cells: Materials, flow channel design and forming processes. J. Power Sources 2020, 451, 227783. [Google Scholar] [CrossRef]
- JongHee KI, M.; Jo, K.; Kim, Y.; Lee, J.; Lee, Y.; Kim, J.; Seok, J. Stainless Steel for Polymer Fuel Cell Separator and Method for Preparing Same. U.S. Patent 9,290,845, 22 March 2016. [Google Scholar]
- Ide, S.; Honda, A.; Ishikawa, S.; Ujiro, T. Stainless Steel Having Good Conductivity for Use in Fuel Cell and Method for Producing the Same. U.S. Patent 8,440,029, 14 May 2013. [Google Scholar]
- Fink, C.; Fouquet, N. Three-dimensional simulation of polymer electrolyte membrane fuel cells with experimental validation. Electrochim. Acta 2011, 56, 10820–10831. [Google Scholar] [CrossRef]
- Nonobe, Y. Development of the fuel cell vehicle mirai. IEEJ Trans. Electr. Electron. Eng. 2017, 12, 5–9. [Google Scholar] [CrossRef]
- Sverdrup, H.U.; Ragnarsdottir, K.V. A system dynamics model for platinum group metal supply, market price, depletion of extractable amounts, ore grade, recycling and stocks-in-use. Resour. Conserv. Recycl. 2016, 114, 130–152. [Google Scholar] [CrossRef]
- Poirot-Crouvezier, J. Road Electric Transportation—Fuel Cell Vehicles; Techniques de l’ingénieur: Paris, France, 2015. (In French) [Google Scholar]
- Shao, Y.; Dodelet, J.; Wu, G.; Zelenay, P. PGM-free cathode catalysts for PEM fuel cells: A mini-review on stability challenges. Adv. Mater. 2019, 31, 1807615. [Google Scholar] [CrossRef]
- Ramaswamy, N.; Gu, W.; Ziegelbauer, J.M.; Kumaraguru, S. Carbon Support Microstructure Impact on High Current Density Transport Resistances in PEMFC Cathode. J. Electrochem. Soc. 2020, 167, 064515. [Google Scholar] [CrossRef]
- Chen, M.; Zhao, C.; Sun, F.; Fan, J.; Li, H.; Wang, H. Research progress of catalyst layer and interlayer interface structures in membrane electrode assembly (MEA) for proton exchange membrane fuel cell (PEMFC) system. eTransportation 2020, 5, 100075. [Google Scholar] [CrossRef]
- Trencher, G. Strategies to accelerate the production and diffusion of fuel cell electric vehicles: Experiences from California. Energy Rep. 2020, 6, 2503–2519. [Google Scholar] [CrossRef]
- IEA. The Future of Hydrogen—Seizing Today’s Opportunities; Technology Report; IEA: Paris, France, 2019. [Google Scholar]
- Ostadi, M.; Paso, K.G.; Rodriguez-Fabia, S.; Øi, L.E.; Manenti, F.; Hillestad, M. Process Integration of Green Hydrogen: Decarbonization of Chemical Industries. Energies 2020, 13, 4859. [Google Scholar] [CrossRef]
- Abdin, Z.; Zafaranloo, A.; Rafiee, A.; Mérida, W.; Lipiński, W.; Khalilpour, K.R. Hydrogen as an energy vector. Renew. Sustain. Energy Rev. 2020, 120, 109620. [Google Scholar] [CrossRef]
- Dawood, F.; Anda, M.; Shafiullah, G.M. Hydrogen production for energy: An overview. Int. J. Hydrog. Energy 2020, 45, 3847–3869. [Google Scholar] [CrossRef]
- Li, S.; Kang, Q.; Baeyens, J.; Zhang, H.L.; Deng, Y.M. Hydrogen Production: State of Technology. In Proceedings of the 2020 IOP Conference Series: Earth and Environmental Science, Vienna, Austria, 18–21 May 2020; Volume 544, p. 012011. [Google Scholar] [CrossRef]
- Muradov, N. Low to near-zero CO2 production of hydrogen from fossil fuels: Status and perspectives. Int. J. Hydrog. Energy 2017, 42, 14058–14088. [Google Scholar] [CrossRef]
- Surla, K. Hydrogen; J6368; Techniques de l’Ingénieur: Saint-Denis, France, May 2019. (In French) [Google Scholar]
- Andrews, J.W. Hydrogen production and carbon sequestration by steam methane reforming and fracking with carbon dioxide. Int. J. Hydrog. Energy 2020, 45, 9279–9284. [Google Scholar] [CrossRef]
- Peng, X.D. Analysis of the thermal efficiency limit of the steam methane reforming process. Ind. Eng. Chem. Res. 2012, 51, 16385–16392. [Google Scholar] [CrossRef]
- Nuttall, W.J.; Bakenne, A.T. Fossil Fuel Hydrogen; Springer: Berlin/Heidelberg, Germany, 2020; ISBN 978-3-030-30907-7. [Google Scholar] [CrossRef]
- Philibert, C. Electrification and Hydrogen in the Energy Transition. In Proceedings of the Energy Sustainability Conference, ENS Rennes, France, 25 October 2018. (In French). [Google Scholar]
- Baeyens, J.; Zhang, H.; Nie, J.; Appels, L.; Dewil, R.; Ansart, R.; Deng, Y. Reviewing the potential of bio-hydrogen production by fermentation. Renew. Sustain. Energy Rev. 2020, 131, 110023. [Google Scholar] [CrossRef]
- IFRI. Carbon Capture, Storage and utilization to the rescue of Coal?—Global Perspectives and Focus on China and the United States. In Sylvie Cornot-Gandolphe; IFRI: Paris, France, 2019. [Google Scholar]
- IRENA. Hydrogen: A Renewable Energy Perspective; International Renewable Energy Agency: Abu Dhabi, UAE, 2019; ISBN 978-92-9260-151-5. [Google Scholar]
- Grigoriev, S.A.; Fateev, V.N.; Bessarabov, D.G.; Millet, P. Current status, research trends, and challenges in water electrolysis science and technology. Int. J. Hydrog. Energy 2020, 45, 26036–26058. [Google Scholar] [CrossRef]
- Yodwong, B.; Guilbert, D.; Phattanasak, M.; Kaewmanee, W.; Hinaje, M.; Vitale, G. Faraday’s Efficiency Modeling of a Proton Exchange Membrane Electrolyzer Based on Experimental Data. Energies 2020, 13, 4792. [Google Scholar] [CrossRef]
- Kumar, S.S.; Himabindu, V. Hydrogen production by PEM water electrolysis—A review. Mater. Sci. Energy Technol. 2019, 2, 442–454. [Google Scholar] [CrossRef]
- David, M.; Ocampo-Martínez, C.; Sánchez-Peña, R. Advances in alkaline water electrolyzers: A review. J. Energy Storage 2019, 23, 392–403. [Google Scholar] [CrossRef]
- Philibert, C. Renewable Energy for Industry; IEA: Paris, France; OECD: Paris, France, 2017. [Google Scholar]
- Ceran, B. Multi-Criteria Comparative Analysis of Clean Hydrogen Production Scenarios. Energies 2020, 13, 4180. [Google Scholar] [CrossRef]
- Park, S.; Nam, S.; Oh, M.; Choi, I.-J.; Shin, J. Preference Structure on the Design of Hydrogen Refueling Stations to Activate Energy Transition. Energies 2020, 13, 3959. [Google Scholar] [CrossRef]
- Nicita, A.; Maggio, G.; Andaloro, A.P.F.; Squadrito, G. Green hydrogen as feedstock: Financial analysis of a photovoltaic-powered electrolysis plant. Int. J. Hydrog. Energy 2020, 45, 11395–11408. [Google Scholar] [CrossRef]
- Lee, D.; Elgowainy, A.; Dai, Q. Life cycle greenhouse gas emissions of hydrogen fuel production from chlor-alkali processes in the United States. Appl. Energy 2018, 217, 467–479. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, T.; Bao, M.; Su, H.; Xu, P. Microbial Production of Hydrogen by Mixed Culture Technologies: A Review. Biotechnol. J. 2020, 15, 1900297. [Google Scholar] [CrossRef]
- Cao, L.; Yu, I.K.M.; Xiong, X.; Tsang, D.C.W.; Zhang, S.; Clark, J.H.; Hu, C.; Ng, Y.H.; Shang, J.; Ok, Y.S. Biorenewable hydrogen production through biomass gasification: A review and future prospects. Environ. Res. 2020, 186, 109547. [Google Scholar] [CrossRef]
- Dou, B.; Zhang, H.; Song, Y.; Zhao, L.; Jiang, B.; He, M.; Ruan, C.; Chen, H.; Xu, Y. Hydrogen production from the thermochemical conversion of biomass: Issues and challenges. Sustain. Energy Fuels 2019, 3, 314–342. [Google Scholar] [CrossRef]
- El-Emam, R.S.; Özcan, H. Comprehensive review on the techno-economics of sustainable large-scale clean hydrogen production. J. Clean. Prod. 2019, 220, 593–609. [Google Scholar] [CrossRef]
- van de Velden, M.; Baeyens, J.; Brems, A.; Janssens, B.; Dewil, R. Fundamentals, kinetics and endothermicity of the biomass pyrolysis reaction. Renew. Energy 2010, 35, 232–242. [Google Scholar] [CrossRef]
- Niezgoda, A.; Deng, Y.; Sabatier, F.; Ansart, R. From end-of-life tires to storable energy carriers. J. Environ. Manag. 2020, 276, 111318. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Ma, Z.; Su, H. Two-step continuous hydrogen production by immobilized mixed culture on corn stalk. Renew. Energy 2018, 121, 230–235. [Google Scholar] [CrossRef]
- Herdem, M.S.; Sinaki, M.Y.; Farhad, S.; Hamdullahpur, F. An overview of the methanol reforming process: Comparison of fuels, catalysts, reformers, and systems. Int. J. Energy Res. 2019, 43, 5076–5105. [Google Scholar] [CrossRef]
- Prinzhofer, A.; Cissé, C.S.T.; Diallo, A.B. Discovery of a large accumulation of natural ydrogen in Bourakebougou (Mali). Int. J. Hydrog. Energy 2018, 43, 19315–19326. [Google Scholar] [CrossRef]
- Prinzhofer, A.; Moretti, I.; Françolin, J.; Pacheco, C.; D’Agostino, A.; Werly, J.; Rupin, F. Natural hydrogen continuous emission from sedimentary basins: The example of a Brazilian H2-emitting structure. Int. J. Hydrog. Energy 2019, 44, 5676–5685. [Google Scholar] [CrossRef]
- Andersson, J.; Grönkvist, S. Large-scale storage of hydrogen. Int. J. Hydrog. Energy 2019, 44, 11901–11919. [Google Scholar] [CrossRef]
- Kurtz, J.; Sprik, S.; Bradley, T.H. Review of transportation hydrogen infrastructure performance and reliability. Int. J. Hydrog. Energy 2019, 44, 12010–12023. [Google Scholar] [CrossRef]
- RTE. Electricity Report 2018. Available online: https://bilan-electrique-2018.rte-france.com/wp-content/uploads/2019/02/BE-PDF-2018-1.pdf (accessed on 22 November 2020).
- ADEME. Hydrogen in the Energy Transition. 2018. Available online: https://www.ademe.fr/sites/default/files/assets/documents/fiche-technique_hydrogene_dans_la_te_avril2018_2.pdf (accessed on 22 November 2020). (In French).
- van de Kaa, G.; Scholten, D.; Rezaei, J.; Milchram, C. The Battle between Battery and Fuel Cell Powered Electric Vehicles: A BWM Approach. Energies 2017, 10, 1707. [Google Scholar] [CrossRef]
- Bansal, S.; Zong, Y.; You, S.; Mihet-Popa, L.; Xiao, J. Technical and Economic Analysis of One-Stop Charging Stations for Battery and Fuel Cell EV with Renewable Energy Sources. Energies 2020, 13, 2855. [Google Scholar] [CrossRef]
- Briottet, L.; Moro, I.; Lemoine, P. Quantifying the hydrogen embrittlement of pipeline steels for safety considerations. Int. J. Hydrog. Energy 2012, 37, 17616–17623. [Google Scholar] [CrossRef]
- Darkrim-Lamari, F.; Malbrunot, P. Hydrogen Fuel—Use; Techniques de l’Ingénieur: Paris, France, 2013. [Google Scholar]
- Dryer, F.L.; Chaos, M.; Zhao, Z.; Stein, J.N.; Alpert, J.Y.; Homer, C.J. Spontaneous ignition of pressurized releases of hydrogen and natural gas into air. Combust. Sci. Technol. 2007, 179, 663–694. [Google Scholar] [CrossRef]
- Tamura, Y.; Takabayashi, M.; Takeuchi, M. The spread of fire from adjoining vehicles to a hydrogen fuel cell vehicle. Int. J. Hydrog. Energy 2014, 39, 6169–6175. [Google Scholar] [CrossRef]
- Hussein, H.; Brennan, S.; Molkov, V. Dispersion of hydrogen release in a naturally ventilated covered car park. Int. J. Hydrog. Energy 2020, 45, 23882–23897. [Google Scholar] [CrossRef]
- Dadashzadeh, M.; Kashkarov, S.; Makarov, D.; Molkov, V. Risk assessment methodology for onboard hydrogen storage. Int. J. Hydrog. Energy 2018, 43, 6462–6475. [Google Scholar] [CrossRef]
- Li, Z.; Sun, K. Mitigation measures for intended hydrogen release from thermally activated pressure relief device of onboard storage. Int. J. Hydrog. Energy 2020, 5, 9260–9267. [Google Scholar] [CrossRef]
- Huang, E. A hydrogen Fueling Station Fire in Norway Has Left Fuel-Cell Cars Nowhere to Charge; Reuters Agency: London, UK, 2019. [Google Scholar]
- Jin, H.; Chung, J. Hydrogen Hurdles: A Deadly Blast Hampers South Korea’s Big Fuel Cell Car Bet; Reuters Agency: London, UK, 2019. [Google Scholar]




| Part of the FCS | Cost Part |
|---|---|
| Stack | 50% |
| Air management | 17% |
| Fuel management | 8% |
| Thermal management | 8% |
| Water management | 3% |
| System balancing | 7% |
| System assembly | 7% |
| Part of the FC | Cost Part |
|---|---|
| Active Layer (AL) | 51% |
| Membrane (M) | 4% |
| Gas Diffusion Layer (GDL) | 5% |
| Bipolar Plate (BP) | 24% |
| Seal | 7% |
| Stack balancing | 2% |
| Assembly | 5% |
| Conditioning | 2% |
| Year | 1995 | 2000 | 2005 | 2010 | 2015 | 2020 |
| Pt load (W/mgPt) | 0.54 | 0.93 | 1.595 | 2.71 | 5.00 | 8.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bethoux, O. Hydrogen Fuel Cell Road Vehicles and Their Infrastructure: An Option towards an Environmentally Friendly Energy Transition. Energies 2020, 13, 6132. https://doi.org/10.3390/en13226132
Bethoux O. Hydrogen Fuel Cell Road Vehicles and Their Infrastructure: An Option towards an Environmentally Friendly Energy Transition. Energies. 2020; 13(22):6132. https://doi.org/10.3390/en13226132
Chicago/Turabian StyleBethoux, Olivier. 2020. "Hydrogen Fuel Cell Road Vehicles and Their Infrastructure: An Option towards an Environmentally Friendly Energy Transition" Energies 13, no. 22: 6132. https://doi.org/10.3390/en13226132
APA StyleBethoux, O. (2020). Hydrogen Fuel Cell Road Vehicles and Their Infrastructure: An Option towards an Environmentally Friendly Energy Transition. Energies, 13(22), 6132. https://doi.org/10.3390/en13226132




