The Feasibility of Replacing Coal with Biomass in Iron-Ore Pelletizing Plants with Respect to Melt-Induced Slagging
Abstract
1. Introduction
2. Materials and Methods
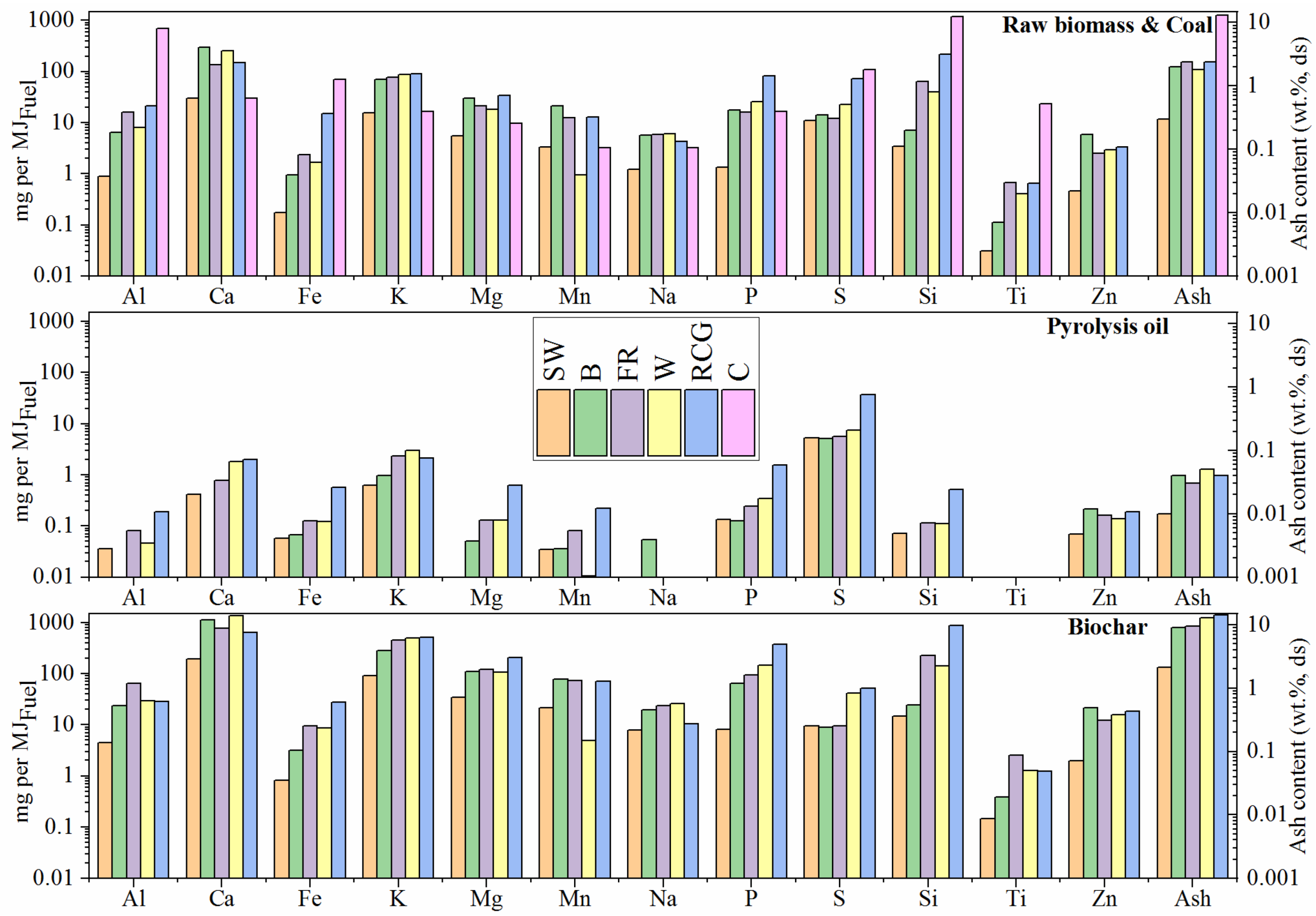
2.1. Proximate, Ultimate, and Calorimetric Analysis of the Investigated Fuels
2.2. Thermochemical Equilibrium Calculations (TECs) and Viscosity Estimations
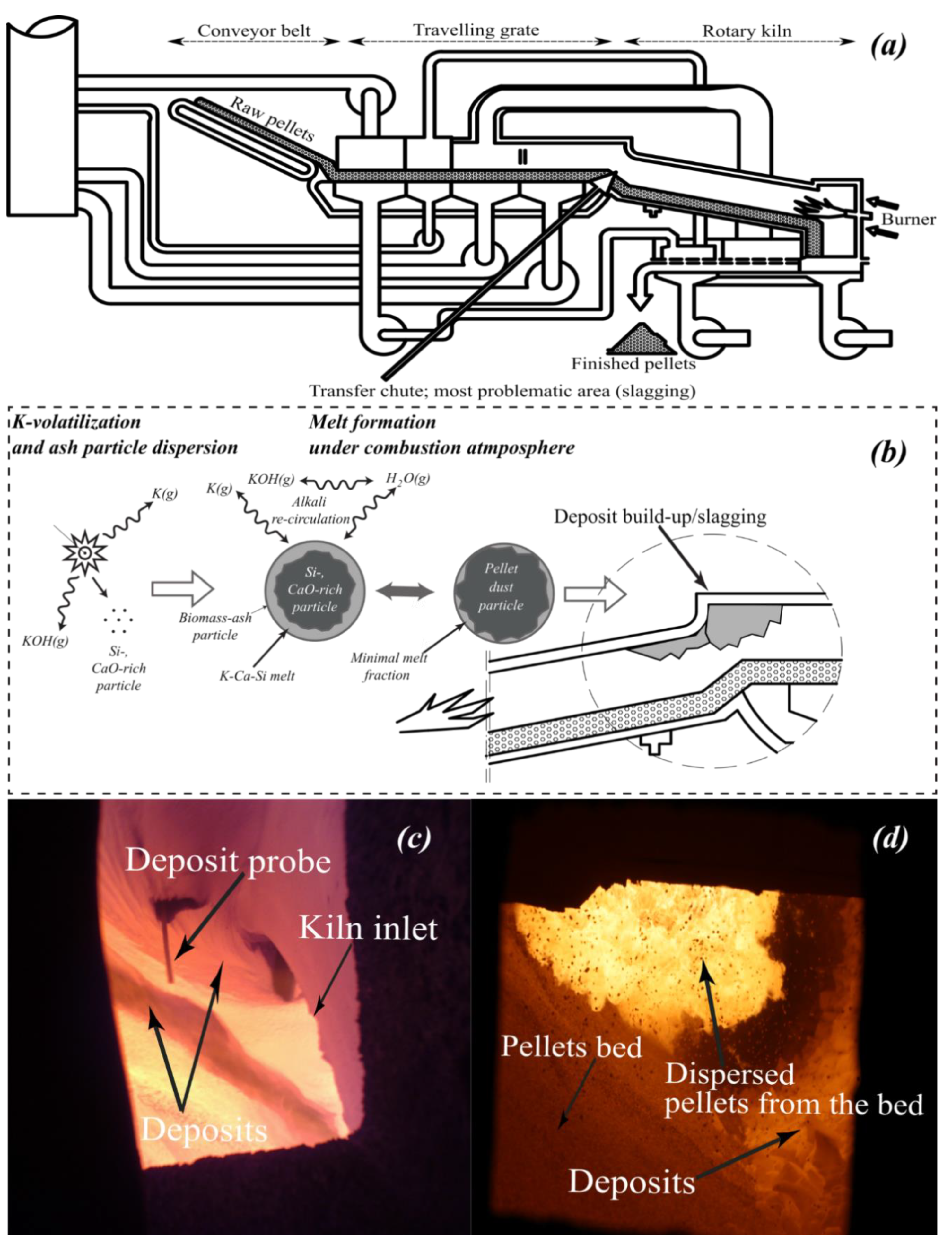
2.3. The Previously Introduced Qualitative Slagging Indicator and the Slagging/Deposition Tendency
2.4. The Absolute Slagging/Deposition Tendency (ADT)
3. Results and Discussion
- Interaction of iron-ore pellet dust and fuel ash: from 0 to 100 wt.% share. Due to the uncertainty regarding the extent of interaction, the amount of ash particles that can potentially interact with the pellet dust can differ from the ratio of their corresponding bulk flows, hence the necessity to study the entire range from 0 to 100 wt.% share of pellet dust (X-axis in the plots).
- Oxidizing gaseous atmosphere at 1 atm, where the partial pressure of oxygen was 16 vol.% (equivalent to an air factor of approximately 4.5), saturated H2O (g) was used, and N2 was used as balance.
- The temperature range studied was from 1000 to 1500 °C. However, according to the previously conducted temperature measurements at the transfer-chute [10,11,12,13], only the results corresponding to the temperatures up to 1350 °C are presented in Section 3.1, Section 3.2 and Section 3.3. However, in Section 3.4 the calculations that are taken into consideration cover the entire studied temperature range (1000–1500 °C).
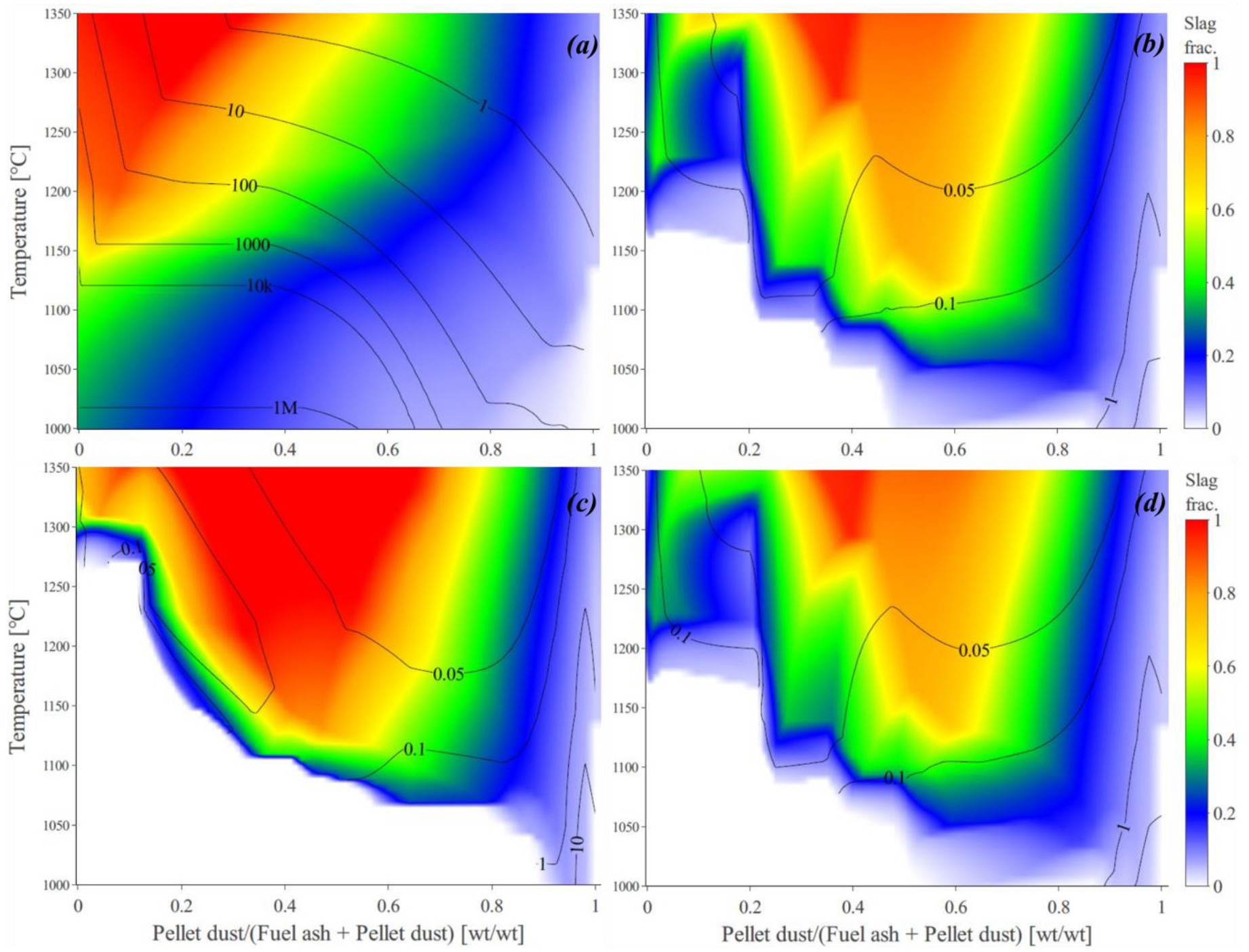
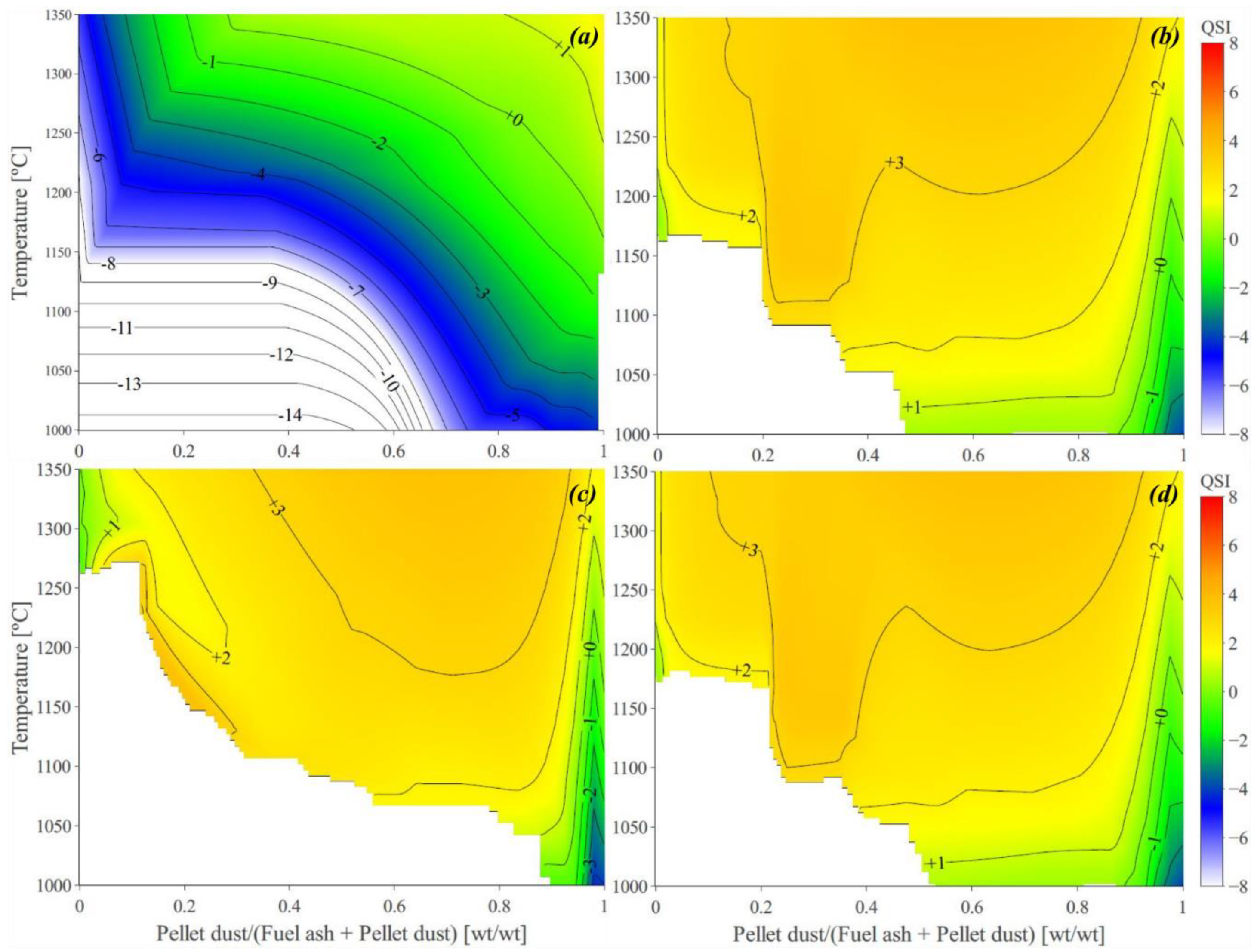
3.1. Predicted Melt Fraction, Viscosity Estimations, and the Calculated QSIs
3.2. The Predicted Absolute Slagging/Deposition Tendency (ADT)
3.3. The Effect of Ash Composition on Slagging/Deposition
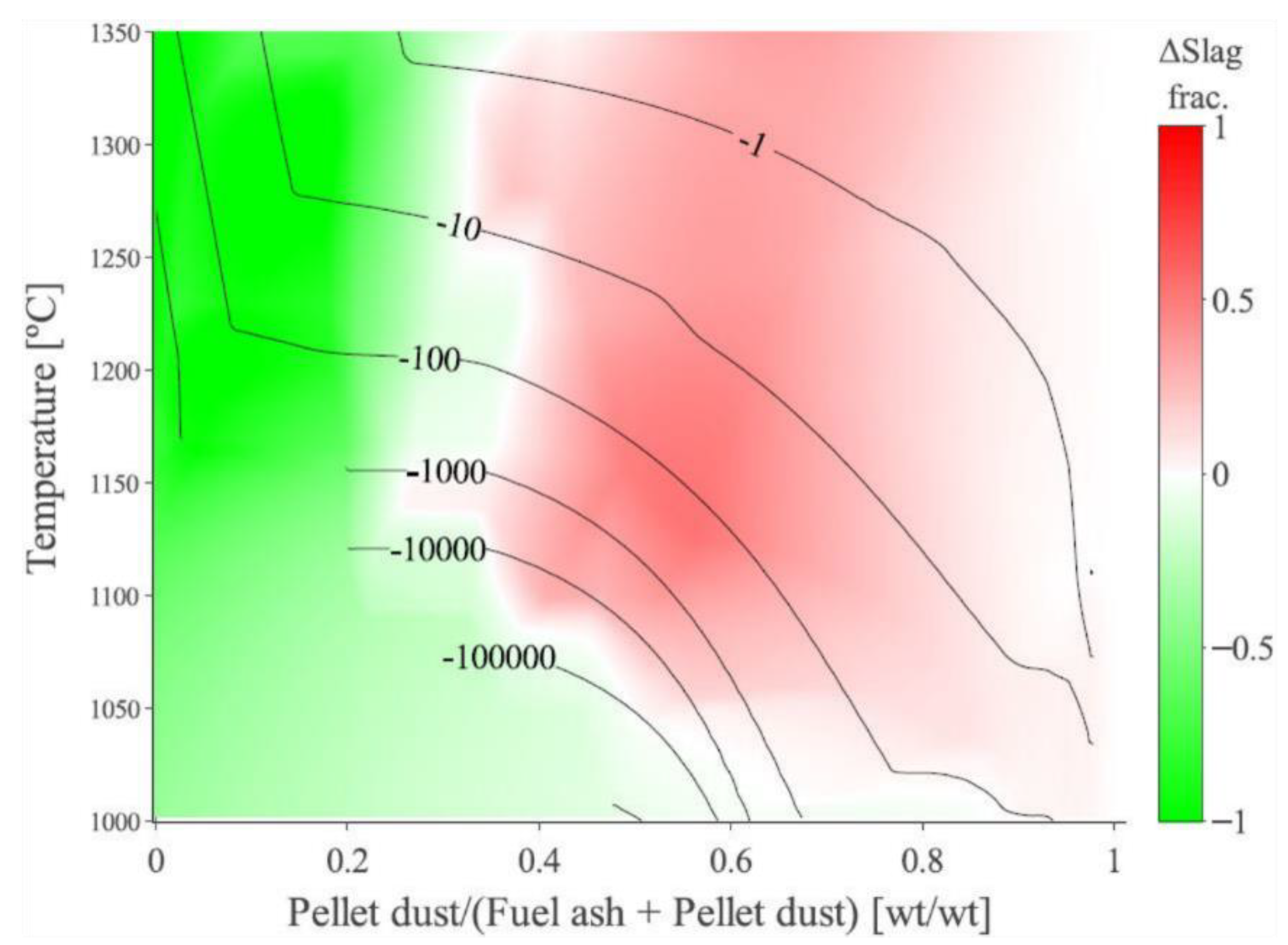
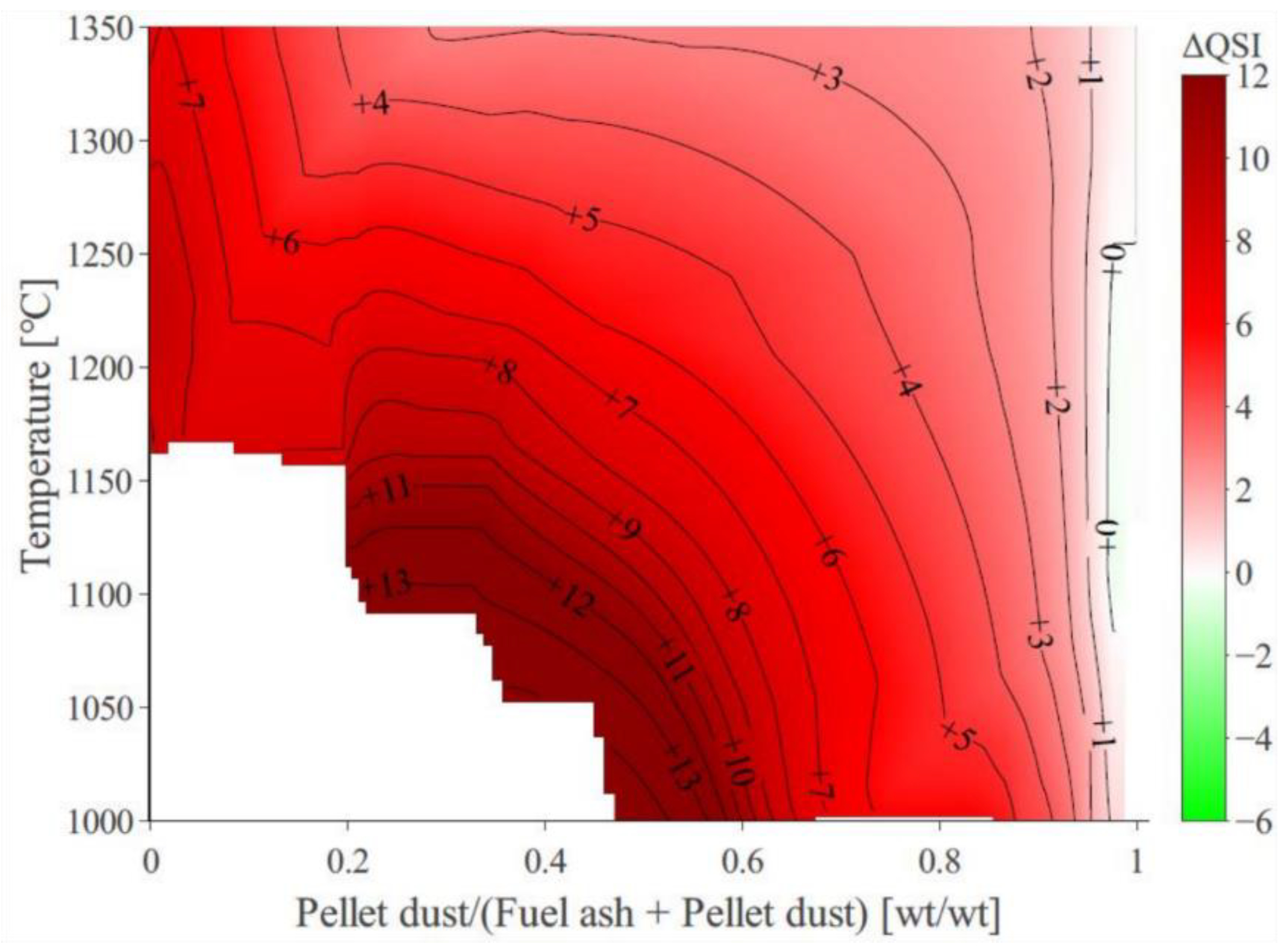
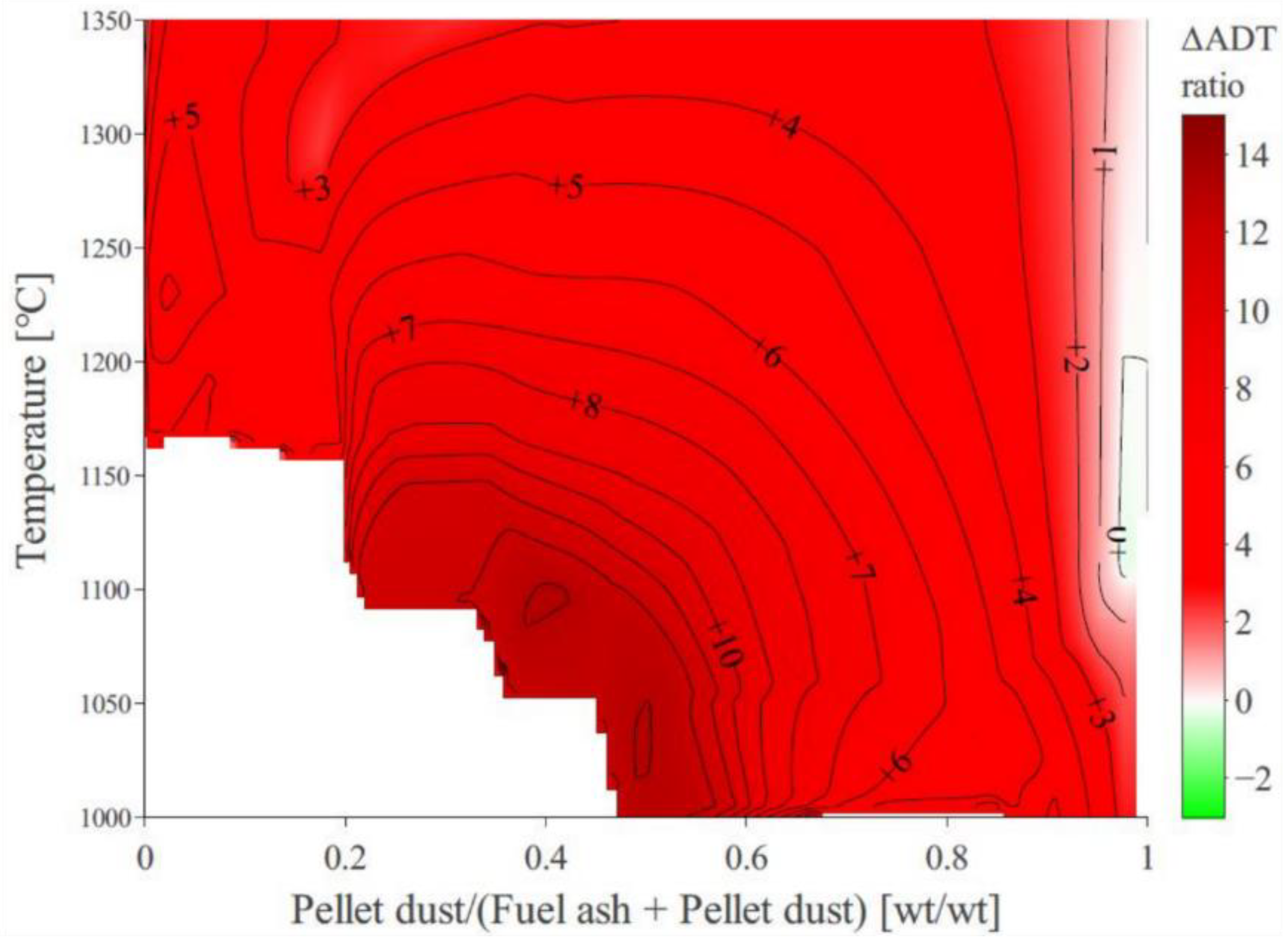
3.4. Quantification of the Absolute Deposition Tendency across the Studied Domain
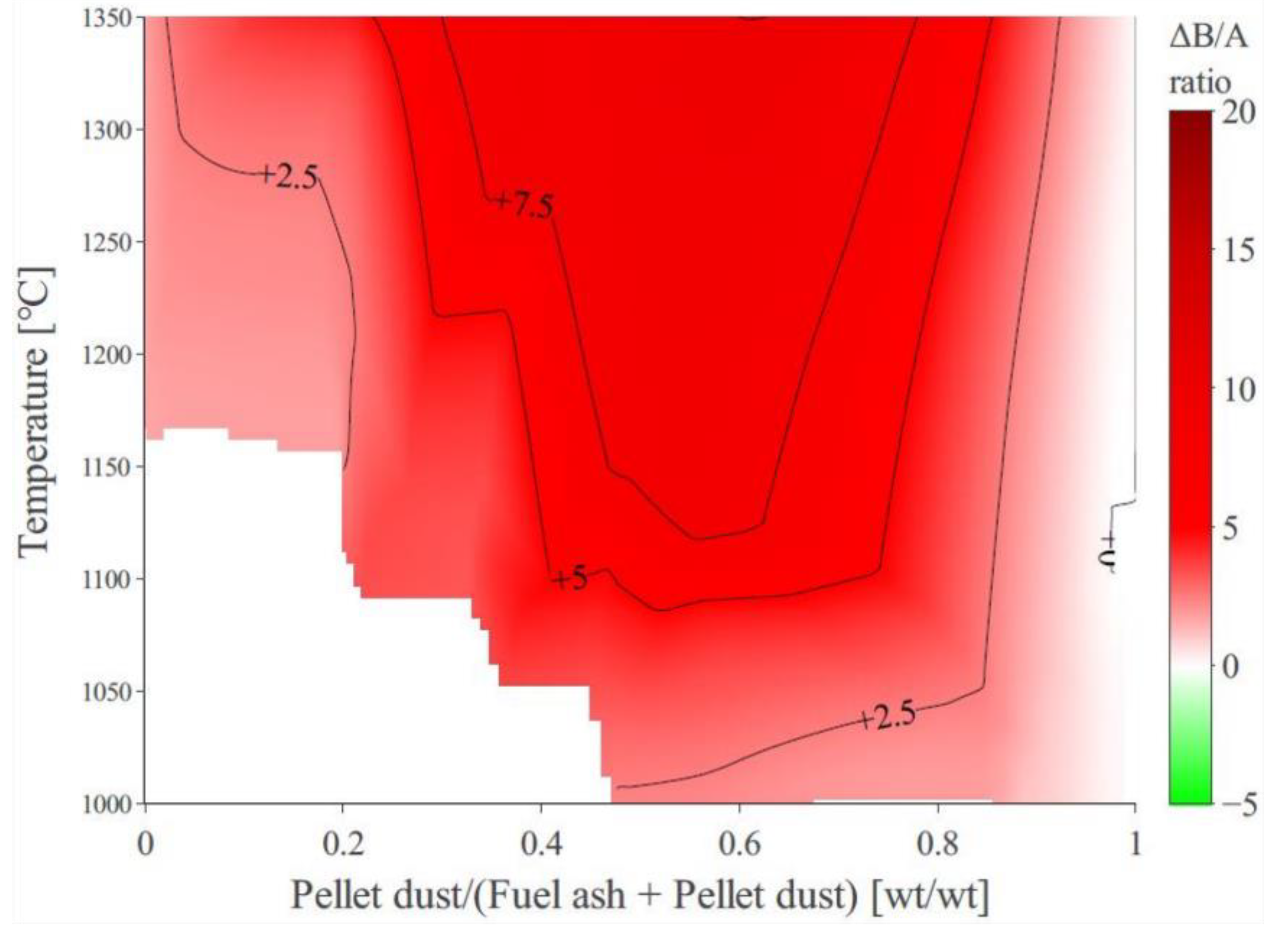
3.5. The Effect of Biomass-Ash Composition upon Slagging When Interacting with Pellet Dust
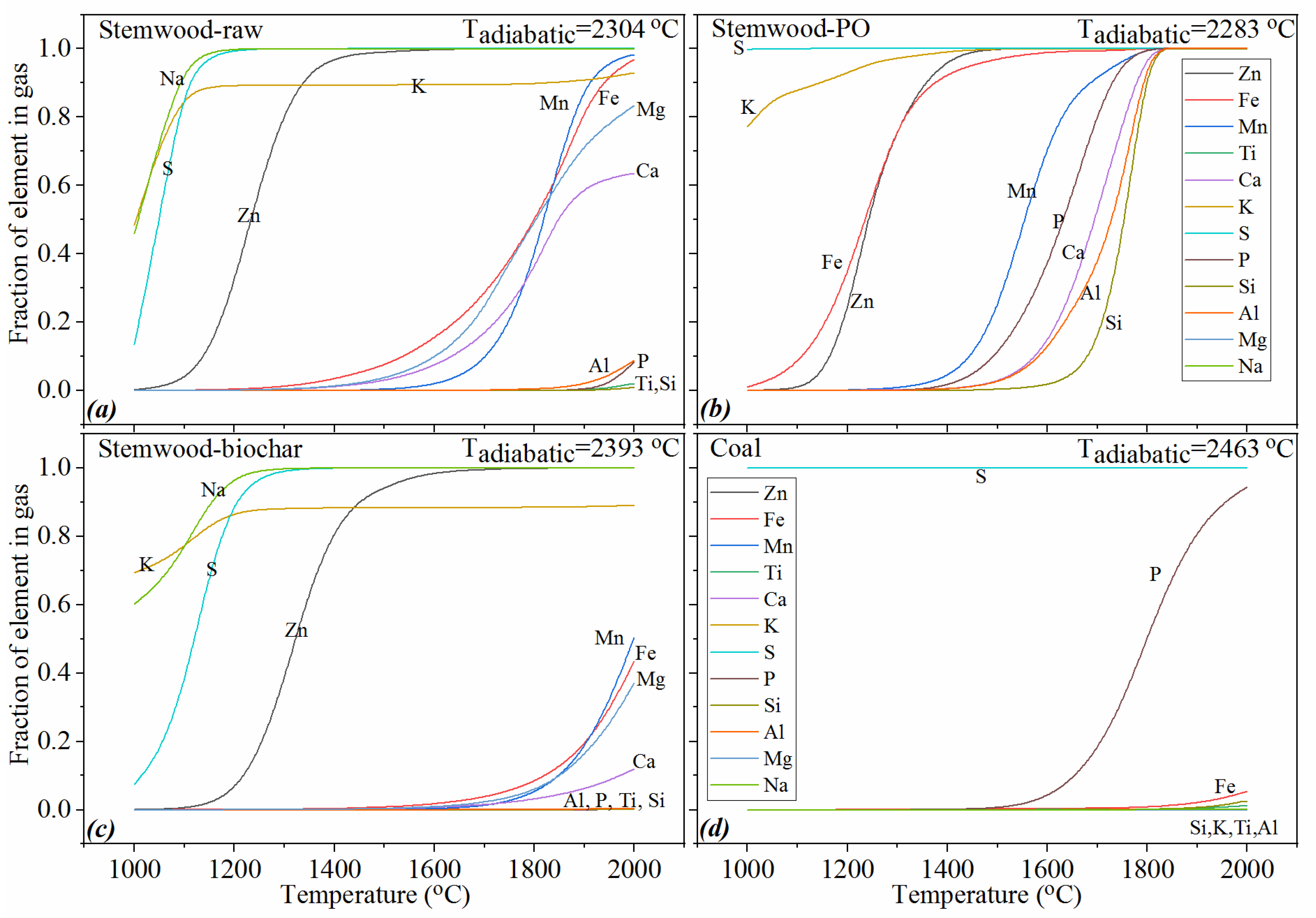
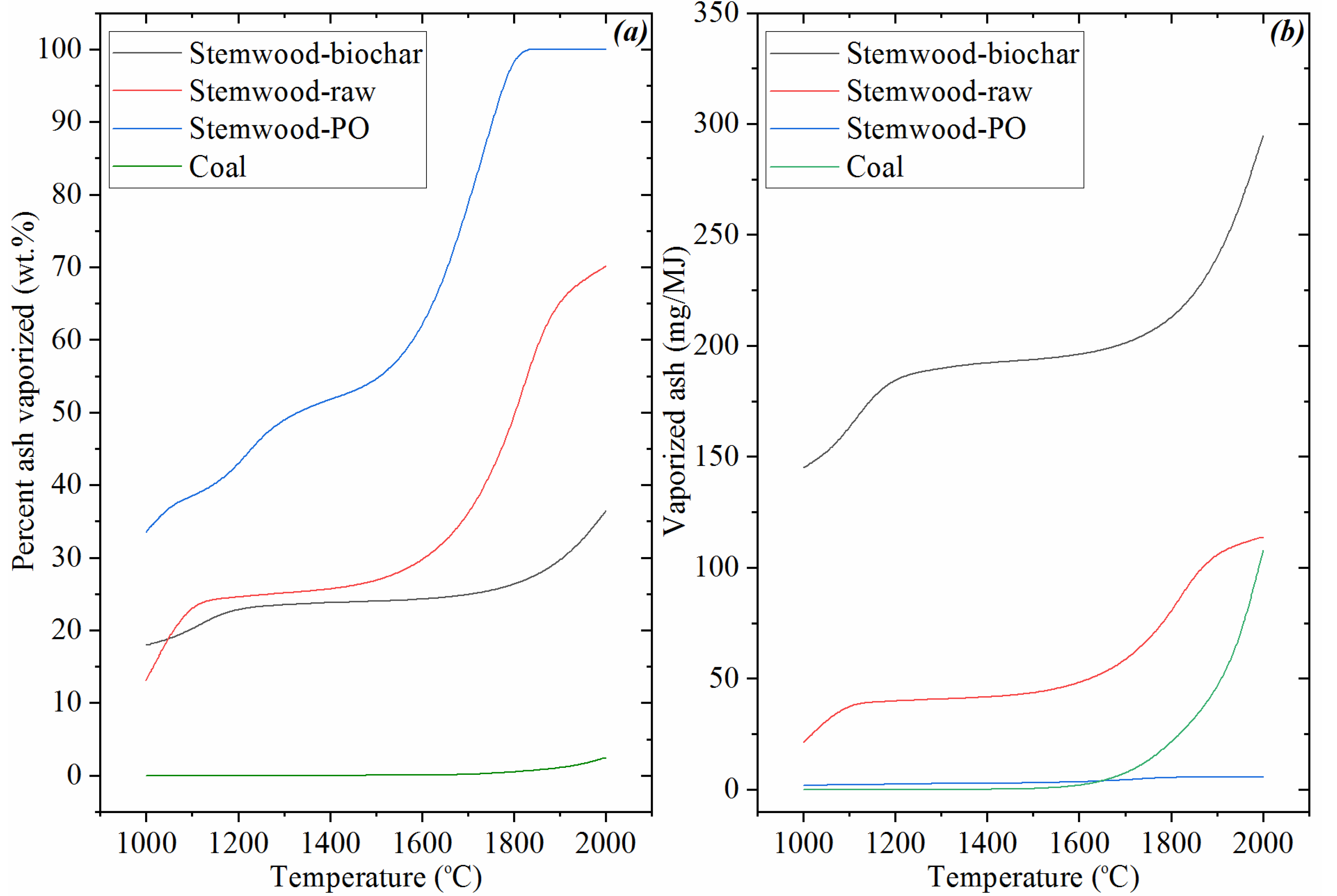
3.6. The Volatile Behavior of Ash in the Investigated Cases
3.7. Practical Implications
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- De Moraes, S.L.; De Lima, J.R.B.; Ribeiro, T.R. Iron Ore Pelletizing Process: An Overview. In Iron Ores and Iron Oxide Materials; IntechOpen: London, UK, 2018. [Google Scholar]
- Forsmo, S.; Vuori, J. The determination of porosity in iron ore green pellets by packing in silica sand. Powder Technol. 2005, 159, 71–77. [Google Scholar] [CrossRef]
- Forsmo, S.; Forsmo, S.-E.; Samskog, P.-O.; Björkman, B. Mechanisms in oxidation and sintering of magnetite iron ore green pellets. Powder Technol. 2008, 183, 247–259. [Google Scholar] [CrossRef]
- Potts, R. Induration of taconite pellets using a grate kiln system. In Annual Meeting of the Minnesota Section, AIME; The Section: Minneapolis, MN, USA, 1991. [Google Scholar]
- Forsmo, S. Influence of Green Pellet Properties on Pelletizing of Magnetite Iron Ore; Luleå Tekniska Universitet: Luleå, Sweden, 2007. [Google Scholar]
- Sefidari, H.; Lindblom, B.; Wiinikka, H.; Nordin, L.-O.; Mouzon, J.; Bhuiyan, I.U.; Öhman, M. The effect of disintegrated iron-ore pellet dust on deposit formation in a pilot-scale pulverized coal combustion furnace. Part I: Characterization of process gas particles and deposits. Fuel Process. Technol. 2018, 177, 283–298. [Google Scholar] [CrossRef]
- Sefidari, H.; Lindblom, B.; Wiinikka, H.; Nordin, L.-O.; Lennartsson, A.; Mouzon, J.; Bhuiyan, I.U.; Öhman, M. The effect of disintegrated iron-ore pellet dust on deposit formation in a pilot-scale pulverized coal combustion furnace. Part II: Thermochemical equilibrium calculations and viscosity estimations. Fuel Process. Technol. 2018, 180, 189–206. [Google Scholar] [CrossRef]
- Bandyopadhyay, R.; Gupta, S.; Lindblom, B.; Jonsson, S. In effect of coal ash characteristics on ash deposition in rotary kilns. In Proceedings of the 4th International Conference on Process Development in Iron and Steelmaking (SCANMET IV), Swerea Mefos, Lulea, Sweden, 10–13 June 2012. [Google Scholar]
- Lindblom, B.; Fredriksson, C.; Jonsson, S.; Nordin, L.-O.; Wiinikka, H. Pilot scale tests of different coals for the usage in the grate-kiln process at LKAB. In Proceedings of the International Conference on Applied Energy ICAE, Suzhou, China, 5–8 July 2012. [Google Scholar]
- Stjernberg, J.; Ion, J.; Antti, M.-L.; Nordin, L.-O.; Lindblom, B.; Odén, M. Extended studies of degradation mechanisms in the refractory lining of a rotary kiln for iron ore pellet production. J. Eur. Ceram. Soc. 2012, 32, 1519–1528. [Google Scholar] [CrossRef]
- Jonsson, C.Y.; Stjernberg, J.; Wiinikka, H.; Lindblom, B.; Boström, D.; Öhman, M. Deposit Formation in a Grate-Kiln Plant for Iron-Ore Pellet Production. Part 1: Characterization of Process Gas Particles. Energy Fuels 2013, 27, 6159–6170. [Google Scholar] [CrossRef]
- Stjernberg, J.; Jonsson, C.Y.; Wiinikka, H.; Lindblom, B.; Boström, D.; Öhman, M. Deposit Formation in a Grate–Kiln Plant for Iron-Ore Pellet Production. Part 2: Characterization of Deposits. Energy Fuels 2013, 27, 6171–6184. [Google Scholar] [CrossRef]
- Jonsson, C.; Wiinikka, H.; Lindblom, B.; Stjernberg, J.; Öhman, M. Comparison of particle and deposit formation between a full-scale grate-kiln plant (40 MW) and a pilot-scale pulverised coal-fired furnace (400 kW). In Proceedings of the International Conference on Applied Energy: Energy Solution for a Sustainable World, Pretoria, South Africa, 1–5 July 2013. [Google Scholar]
- Stjernberg, J.; Isaksson, O.; Ion, J. The grate-kiln induration machine—History, advantages, and drawbacks, and outline for the future. J. South. Afr. Inst. Min. Met. 2015, 115, 137–144. [Google Scholar] [CrossRef]
- Bryers, R.W. Fireside slagging, fouling, and high-temperature corrosion of heat-transfer surface due to impurities in steam-raising fuels. Prog. Energy Combust. Sci. 1996, 22, 29–120. [Google Scholar] [CrossRef]
- Benson, S.A.; Hurley, J.P.; Zygarlicke, C.J.; Steadman, E.N.; Erickson, T.A. Predicting ash behavior in utility boilers. Energy Fuels 1993, 7, 746–754. [Google Scholar] [CrossRef]
- Benson, S.A.; Erickson, T.A.; Jensen, R.R.; Laumb, J.D. Transformations model for predicting size and composition of ash during coal combustion. Fuel Chem. Div. Prepr. 2002, 47, 796. [Google Scholar]
- Srinivasachar, S.; Senior, C.L.; Helble, J.; Moore, J. A fundamental approach to the prediction of coal ash deposit formation in combustion systems. Symp Combust. 1992, 24, 1179–1187. [Google Scholar] [CrossRef]
- Raask, E. Mineral Impurities in Coal Combustion: Behavior, Problems, and Remedial Measures; Taylor & Francis: Leatherhead, UK, 1985. [Google Scholar]
- Sefidari, H.; Wiinikka, H.; Lindblom, B.; Nordin, L.; Wu, G.; Yazhenskikh, E.; Müller, M.; Ma, C.; Öhman, M. Comparison of high-rank coals with respect to slagging/deposition tendency at the transfer-chute of iron-ore pelletizing grate-kiln plants: A pilot-scale experimental study accompanied by thermochemical equilibrium modeling and viscosity estimations. Fuel Process. Technol. 2019, 193, 244–262. [Google Scholar] [CrossRef]
- Sefidari, H.; Ma, C.; Fredriksson, C.; Lindblom, B.; Wiinikka, H.; Nordin, L.; Wu, G.; Yazhenskikh, E.; Müller, M.; Öhman, M. The effect of co-firing coal and woody biomass upon the slagging/deposition tendency in iron-ore pelletizing grate-kiln plants. Fuel Process. Technol. 2020, 199, 106254. [Google Scholar] [CrossRef]
- Wiinikka, H.; Sepman, A.; Ögren, Y.; Lindblom, B.; Nordin, L.O. Combustion Evaluation of Renewable Fuels for Iron-Ore Pellet Induration. Energy Fuels 2019, 33, 7819–7829. [Google Scholar] [CrossRef]
- Boström, D.; Skoglund, N.; Grimm, A.; Boman, C.; Öhman, M.; Broström, M.; Backman, R. Ash Transformation Chemistry during Combustion of Biomass. Energy Fuels 2011, 26, 85–93. [Google Scholar] [CrossRef]
- Kleinhans, U.; Wieland, C.; Frandsen, F.J.; Spliethoff, H. Ash formation and deposition in coal and biomass fired combustion systems: Progress and challenges in the field of ash particle sticking and rebound behavior. Prog. Energy Combust. Sci. 2018, 68, 65–168. [Google Scholar] [CrossRef]
- Zhou, H.; Ma, W.; Zhang, J.; Xu, Y.; Zhao, M. Ash deposition behavior under coal and wood co-firing conditions in a 300 kW downfired furnace. J. Energy Inst. 2018, 91, 743–755. [Google Scholar] [CrossRef]
- Wiinikka, H.; Carlsson, P.; Johansson, A.-C.; Gullberg, M.; Ylipää, C.; Lundgren, M.; Sandström, L. Fast Pyrolysis of Stem Wood in a Pilot-Scale Cyclone Reactor. Energy Fuels 2015, 29, 3158–3167. [Google Scholar] [CrossRef]
- Johansson, A.-C.; Wiinikka, H.; Sandström, L.; Marklund, M.; Öhrman, O.; Narvesjö, J. Characterization of pyrolysis products produced from different Nordic biomass types in a cyclone pilot plant. Fuel Process. Technol. 2016, 146, 9–19. [Google Scholar] [CrossRef]
- Wiinikka, H.; Johansson, A.-C.; Sandström, L.; Öhrman, O. Fate of inorganic elements during fast pyrolysis of biomass in a cyclone reactor. Fuel 2017, 203, 537–547. [Google Scholar] [CrossRef]
- Sefidari, H. Mechanisms of Deposit Formation in the Grate-kiln Process; Luleå Tekniska Universitet: Luleå, Sweden, 2018. [Google Scholar]
- Carlsson, P.; Ma, C.; Molinder, R.; Weiland, F.; Wiinikka, H.; Öhman, M.; Öhrman, O. Slag Formation during Oxygen-Blown Entrained-Flow Gasification of Stem Wood. Energy Fuels 2014, 28, 6941–6952. [Google Scholar] [CrossRef]
- Ma, C.; Backman, R.; Öhman, M. Thermochemical Equilibrium Study of Slag Formation during Pressurized Entrained-Flow Gasification of Woody Biomass. Energy Fuels 2015, 29, 4399–4406. [Google Scholar] [CrossRef]
- Carlborg, M.; Weiland, F.; Ma, C.; Backman, R.; Landälv, I.; Wiinikka, H. Exposure of refractory materials during high-temperature gasification of a woody biomass and peat mixture. J. Eur. Ceram. Soc. 2018, 38, 777–787. [Google Scholar] [CrossRef]
- Ma, C.; Carlborg, M.; Hedman, H.; Wennebro, J.; Weiland, F.; Wiinikka, H.; Backman, R.; Öhman, M. Ash Formation in Pilot-Scale Pressurized Entrained-Flow Gasification of Bark and a Bark/Peat Mixture. Energy Fuels 2016, 30, 10543–10554. [Google Scholar] [CrossRef]
- Jonsson, C. Deposit Formation in the Grate-Kiln Process; Luleå Tekniska Universitet: Luleå, Sweden, 2013. [Google Scholar]
- Tao, J.; Guo-qiang, H.; Min, G.; Guang-hui, L.; Xiao-hui, F.; Li-shun, Y. Forming mechanism of rings in rotary-kiln for oxidized pellet. J. Iron Steel Res. Int. 2009, 16, 292–297. [Google Scholar]
- Allendorf, M.D.; Spear, K.E. Thermodynamic Analysis of Silica Refractory Corrosion in Glass-Melting Furnaces. J. Electrochem. Soc. 2001, 148, B59–B67. [Google Scholar] [CrossRef]
- Hack, K.; Jantzen, T.; Müller, M.; Yazhenskikh, E.; Wu, G. A novel thermodynamic database for slag systems and refractory materials. In Proceedings of the 5th International Congress on the Science and Technology of Steelmaking, Dresden, Germany, 20 October 2012. [Google Scholar]
- Bale, C.; Bélisle, E.; Chartrand, P.; Decterov, S.; Eriksson, G.; Hack, K.; Jung, I.-H.; Kang, Y.-B.; Melançon, J.; Pelton, A.; et al. FactSage thermochemical software and databases—Recent developments. Calphad 2009, 33, 295–311. [Google Scholar] [CrossRef]
- Bale, C.; Chartrand, P.; Degterov, S.; Eriksson, G.; Hack, K.; Ben Mahfoud, R.; Melançon, J.; Pelton, A.; Petersen, S. FactSage thermochemical software and databases. Calphad 2002, 26, 189–228. [Google Scholar] [CrossRef]
- Koukkari, P.; Penttilä, K.; Hack, K.; Petersen, S. CHEMSHEET—An Efficient Worksheet Tool for Thermodynamic Process Simulation. In Microstructures, Mechanical Properties and Processes—Computer Simulation and Modelling; Wiley-VCH: Weinheim, Germany, 2005; Volume 3, pp. 323–330. [Google Scholar]
- Nentwig, T.; Kondratiev, A.; Yazhenskikh, E.; Hack, K.; Müller, M. Viscosity Model for Oxide Melts Relevant to Coal Ash Slags Based on the Associate Species Thermodynamic Model. Energy Fuels 2013, 27, 6469–6476. [Google Scholar] [CrossRef]
- Wu, G.; Yazhenskikh, E.; Hack, K.; Wosch, E.; Müller, M. Viscosity model for oxide melts relevant to fuel slags. Part 1: Pure oxides and binary systems in the system SiO2–Al2O3–CaO–MgO–Na2O–K2O. Fuel Process. Technol. 2015, 137, 93–103. [Google Scholar] [CrossRef]
- Wu, G.; Yazhenskikh, E.; Hack, K.; Müller, M. Viscosity model for oxide melts relevant to fuel slags. Part 2: The system SiO2–Al2O3–CaO–MgO–Na2O–K2O. Fuel Process. Technol. 2015, 138, 520–533. [Google Scholar] [CrossRef]
- Wu, G.; Seebold, S.; Yazhenskikh, E.; Hack, K.; Müller, M. Viscosity model for oxide melts relevant to fuel slags. Part 3: The iron oxide containing low order systems in the system SiO2–Al2O3–CaO–MgO–Na2O–K2O–FeO–Fe2O3. Fuel Process. Technol. 2018, 171, 339–349. [Google Scholar] [CrossRef]
- Hack, K.; Wu, G.; Yazhenskikh, E.; Jantzen, T.; Müller, M. A CALPHAD approach to modelling of slag viscosities. Calphad 2019, 65, 101–110. [Google Scholar] [CrossRef]
- Wu, G.; Seebold, S.; Yazhenskikh, E.; Tanner, J.; Hack, K.; Müller, M. Slag mobility in entrained flow gasifiers optimized using a new reliable viscosity model of iron oxide-containing multicomponent melts. Appl. Energy 2019, 236, 837–849. [Google Scholar] [CrossRef]
- Vargas, S.; Frandsen, F.J.; Dam-Johansen, K. Rheological properties of high-temperature melts of coal ashes and other silicates. Prog. Energy Combust. Sci. 2001, 27, 237–429. [Google Scholar] [CrossRef]
- Browning, G.J.; Bryant, G.W.; Hurst, H.J.; Lucas, J.; Wall, T.F. An Empirical Method for the Prediction of Coal Ash Slag Viscosity. Energy Fuels 2003, 17, 731–737. [Google Scholar] [CrossRef]
- Duchesne, M.A.; Bronsch, A.M.; Hughes, R.W.; Masset, P.J. Slag viscosity modeling toolbox. Fuel 2013, 114, 38–43. [Google Scholar] [CrossRef]
- Yazhenskikh, E.; Hack, K.; Müller, M. Critical thermodynamic evaluation of oxide systems relevant to fuel ashes and slags. Part 1: Alkali oxide–silica systems. Calphad 2006, 30, 270–276. [Google Scholar] [CrossRef]
- Yazhenskikh, E.; Hack, K.; Müller, M. Critical thermodynamic evaluation of oxide systems relevant to fuel ashes and slags Part 2: Alkali oxide–alumina systems. Calphad 2006, 30, 397–404. [Google Scholar] [CrossRef]
- Yazhenskikh, E.; Hack, K.; Müller, M. Critical thermodynamic evaluation of oxide systems relevant to fuel ashes and slags. Part 3: Silica–alumina system. Calphad 2008, 32, 195–205. [Google Scholar] [CrossRef]
- Yazhenskikh, E.; Hack, K.; Müller, M. Critical thermodynamic evaluation of oxide systems relevant to fuel ashes and slags, Part 4: Sodium oxide–potassium oxide–silica. Calphad 2008, 32, 506–513. [Google Scholar] [CrossRef]
- Yazhenskikh, E.; Hack, K.; Müller, M. Critical thermodynamic evaluation of oxide systems relevant to fuel ashes and slags, Part 5: Potassium oxide–alumina–silica. Calphad 2011, 35, 6–19. [Google Scholar] [CrossRef]
- Yazhenskikh, E.; Jantzen, T.; Hack, K.; Müller, M. Critical thermodynamic evaluation of oxide systems relevant to fuel ashes and slags: Potassium oxide–magnesium oxide–silica. Calphad 2014, 47, 35–49. [Google Scholar] [CrossRef]
- Brown, G.J. X-ray scattering and X-ray spectroscopy studies of silicate melts. Struct. Dyn. Prop. Silic. Melts 1995, 32, 317–410. [Google Scholar]
- Urbain, G. Viscosity estimation of slags. Steel Res. 1987, 58, 111–116. [Google Scholar] [CrossRef]
- Kondratiev, A.; Hayes, P.C.; Jak, E. Development of a Quasi-chemical Viscosity Model for Fully Liquid Slags in the Al2O3–CaO–‘FeO’–MgO–SiO2 System. Part 1. Description of the Model and Its Application to the MgO, MgO–SiO2, Al2O3–MgO and CaO–MgO Sub-systems. ISIJ Int. 2006, 46, 359–367. [Google Scholar] [CrossRef]
- Senior, C.L.; Srinivasachar, S. Viscosity of Ash Particles in Combustion Systems for Prediction of Particle Sticking. Energy Fuels 1995, 9, 277–283. [Google Scholar] [CrossRef]
- Watt, J. The Physical and Chemical Behaviour of the Mineral Matter in the Coal under the Conditions Met in Combustion Plant: A Literature Survey; Bcura Industrial Laboratories: Leatherhead, UK, 1968. [Google Scholar]
- Watt, J.; Fereday, F. The flow properties of slag formed from the ashes of British coals. J. Ind. Fuel 1969, 42, 89. [Google Scholar]
- MacKenzie, J.K.; Shuttleworth, R. A Phenomenological Theory of Sintering. Proc. Phys. Soc. Sect. B 1949, 62, 833–852. [Google Scholar] [CrossRef]
- Boow, J. Sodium/ash reactions in the formation of fireside deposits in pulverized-fuel-fired boilers. Fuel 1972, 51, 170–173. [Google Scholar] [CrossRef]
- Garba, M.; Ingham, D.; Ma, L.; Degereji, M.; Pourkashanian, M.; Williams, A. Modelling of deposit formation and sintering for the co-combustion of coal with biomass. Fuel 2013, 113, 863–872. [Google Scholar] [CrossRef]
- Balakrishnan, S.; Nagarajan, R.; Karthick, K. Mechanistic modeling, numerical simulation and validation of slag-layer growth in a coal-fired boiler. Energy 2015, 81, 462–470. [Google Scholar] [CrossRef]
- Degereji, M.; Ingham, D.; Ma, L.; Pourkashanian, M.; Williams, A. Numerical assessment of coals/blends slagging potential in pulverized coal boilers. Fuel 2012, 102, 345–353. [Google Scholar] [CrossRef]
- Degereji, M.; Ingham, D.B.; Ma, L.; Pourkashanian, M.; Williams, A. Prediction of ash slagging propensity in a pulverized coal combustion furnace. Fuel 2012, 101, 171–178. [Google Scholar] [CrossRef]
- Zhong, Q.; Yang, Y.; Jiang, T.; Li, Q.; Xu, B. Effect of coal ash on ring behavior of iron-ore pellet powder in kiln. Powder Technol. 2018, 323, 195–202. [Google Scholar] [CrossRef]
- Niininiskorpi, V. Development of Phases and Structures during Pelletizing of Kiruna Magnetite Ore; Åbo Akademi Finland: Turku, Finland, 2004. [Google Scholar]
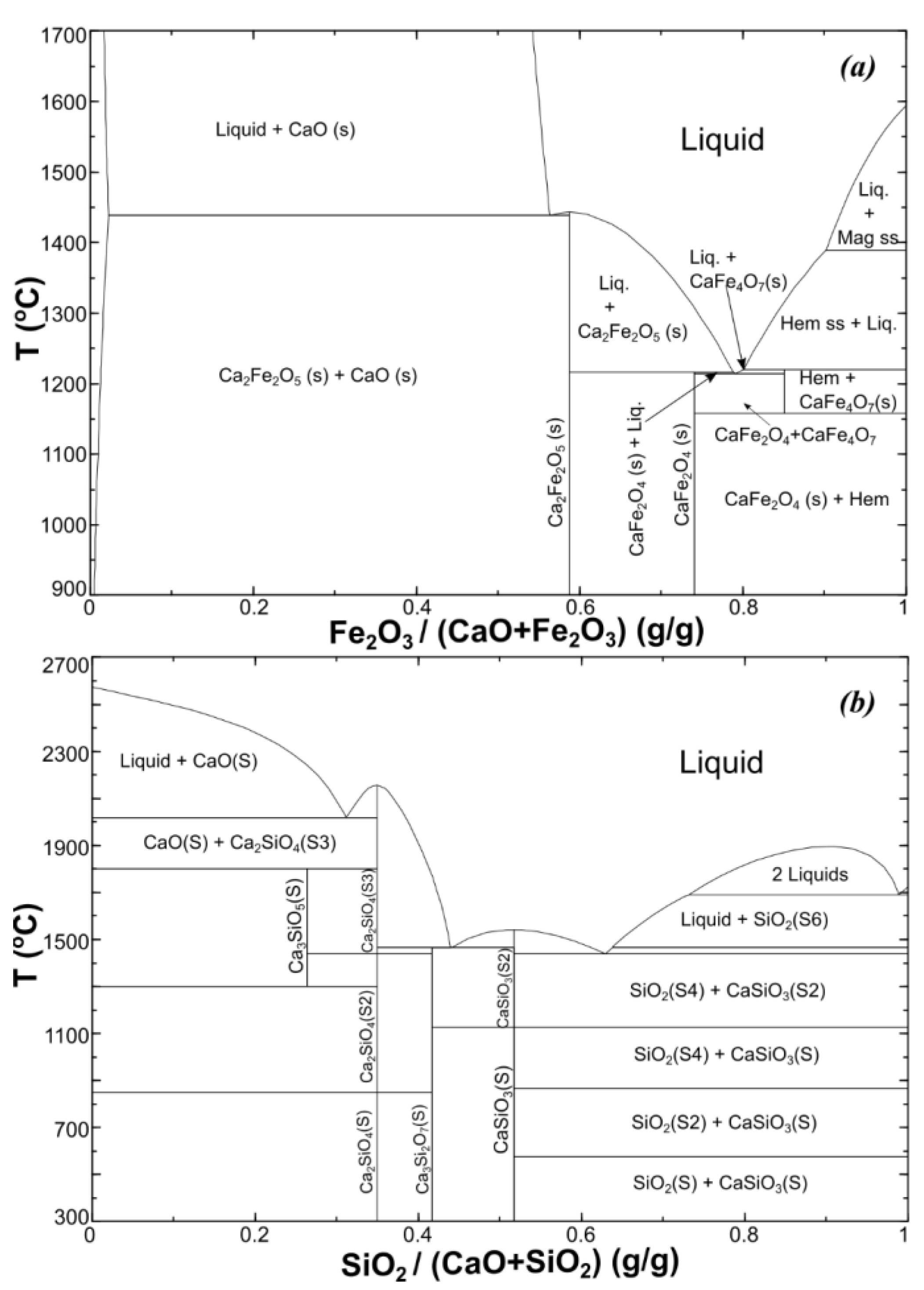
- Phillips, B.; Muan, A. Phase Equilibria in the system CaO-Iron oxide in air and at 1 atm. O2 Pressure. J. Am. Ceram. Soc. 1958, 41, 445–454. [Google Scholar] [CrossRef]
- Phillips, B.; Muan, A. Phase Equilibria in the system CaO-Iron oxide-SiO2, in air. J. Am. Ceram. Soc. 1959, 42, 413–423. [Google Scholar] [CrossRef]
- Nilsson, E.A.A. Degradation Mechanisms of Heat Resistant Steel at Elevated Temperatures: In an Iron Ore Pelletizing Industry. Ph.D. Thesis, Luleå University of Technology, Luleå, Sweden, 1 January 1997. [Google Scholar]
- Flagan, R.; Friedlander, S. Particle formation in pulverized coal combustion—A review. Recent Dev. Aerosol Sci. 1978, 2, 25–59. [Google Scholar]
- Flagan, R.C. Submicron particles from coal combustion. Symp. Combust. 1979, 17, 97–104. [Google Scholar] [CrossRef]
- Neville, M.; Quann, R.; Haynes, B.S.; Sarofim, A. Vaporization and condensation of mineral matter during pulverized coal combustion. Symp. Combust. 1981, 18, 1267–1274. [Google Scholar] [CrossRef]
- Neville, M.; Sarofim, A. The stratified composition of inorganic submicron particles produced during coal combustion. Symp. Combust. 1982, 19, 1441–1449. [Google Scholar] [CrossRef]
- Quann, R.; Sarofim, A. Vaporization of refractory oxides during pulverized coal combustion. Symp. Combust. 1982, 19, 1429–1440. [Google Scholar] [CrossRef]
- Helble, J.; Neville, M.; Sarofim, A. Aggregate formation from vaporized ash during pulverized coal combustion. Symp. Combust. 1988, 21, 411–417. [Google Scholar] [CrossRef]


| A | B | C |  | Increasing slagging potential | |||
| Coal | 26 | Stem wood-PO | 6 | Stem wood-PO | 10 | ||
| Bark-PO | 6446 | Coal | 11 | Forest residues-PO | 30 | ||
| Reed canary grass | 8030 | Bark-PO | 14 | Bark-PO | 37 | ||
| Reed canary grass-BC | 8765 | Forest residues-PO | 17 | Reed canary grass-PO | 38 | ||
| Willow-PO | 9686 | Reed canary grass-PO | 26 | Willow-PO | 53 | ||
| Forest residues-PO | 9877 | Willow-PO | 29 | Stem wood | 260 | ||
| Bark-BC | 10,720 | Stem wood | 197 | Stem wood-BC | 1280 | ||
| Bark | 10,779 | Stem wood-BC | 949 | Bark | 1552 | ||
| Stem wood-PO | 10,895 | Reed canary grass | 1075 | Reed canary grass | 1635 | ||
| Reed canary grass-PO | 11,471 | Bark | 1114 | Willow | 1663 | ||
| Stem wood-BC | 11,756 | Willow | 1315 | Forest residues | 1952 | ||
| Stem wood | 12,114 | Forest residues | 1601 | Coal | 4138 | ||
| Willow-BC | 12,346 | Bark-BC | 3696 | Bark-BC | 5218 | ||
| Forest residues | 12,607 | Forest residues-BC | 4662 | Forest residues-BC | 5718 | ||
| Willow | 12,650 | Reed canary grass-BC | 4959 | Reed canary grass-BC | 7127 | ||
| Forest residues-BC | 13,004 | Willow-BC | 6271 | Willow-BC | 8193 | ||
| Key Value_1 | Key Value_2 | Key Value_3 | Key Value_4 |  | Undesirable |
| Stem wood-PO | Stem wood-PO | Coal | Stem wood-PO | ||
| Coal | Forest residues-PO | Bark-PO | Forest residues-PO | ||
| Bark-PO | Bark-PO | Reed canary grass | Reed canary grass-PO | ||
| Forest residues-PO | Reed canary grass-PO | Reed canary grass-BC | Bark-PO | ||
| Reed canary grass-PO | Willow-PO | Willow-PO | Willow-PO | ||
| Willow-PO | Stem wood | Forest residues-PO | Coal | ||
| Stem wood | Stem wood-BC | Bark-BC | Stem wood | ||
| Stem wood-BC | Bark | Bark | Stem wood-BC | ||
| Reed canary grass | Reed canary grass | Stem wood-PO | Bark | ||
| Bark | Willow | Reed canary grass-PO | Reed canary grass | ||
| Willow | Forest residues | Stem wood-BC | Bark-BC | ||
| Forest residues | Coal | Stem wood | Willow | ||
| Bark-BC | Bark-BC | Willow-BC | Forest residues | ||
| Forest residues-BC | Forest residues-BC | Forest residues | Reed canary grass-BC | ||
| Reed canary grass-BC | Reed canary grass-BC | Willow | Forest residues-BC | ||
| Willow-BC | Willow-BC | Forest residues-BC | Willow-BC |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sefidari, H.; Lindblom, B.; Nordin, L.-O.; Wiinikka, H. The Feasibility of Replacing Coal with Biomass in Iron-Ore Pelletizing Plants with Respect to Melt-Induced Slagging. Energies 2020, 13, 5386. https://doi.org/10.3390/en13205386
Sefidari H, Lindblom B, Nordin L-O, Wiinikka H. The Feasibility of Replacing Coal with Biomass in Iron-Ore Pelletizing Plants with Respect to Melt-Induced Slagging. Energies. 2020; 13(20):5386. https://doi.org/10.3390/en13205386
Chicago/Turabian StyleSefidari, Hamid, Bo Lindblom, Lars-Olof Nordin, and Henrik Wiinikka. 2020. "The Feasibility of Replacing Coal with Biomass in Iron-Ore Pelletizing Plants with Respect to Melt-Induced Slagging" Energies 13, no. 20: 5386. https://doi.org/10.3390/en13205386
APA StyleSefidari, H., Lindblom, B., Nordin, L.-O., & Wiinikka, H. (2020). The Feasibility of Replacing Coal with Biomass in Iron-Ore Pelletizing Plants with Respect to Melt-Induced Slagging. Energies, 13(20), 5386. https://doi.org/10.3390/en13205386





