Experimental Study on the Performance of an SI Engine Fueled by Waste Plastic Pyrolysis Oil–Gasoline Blends †
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
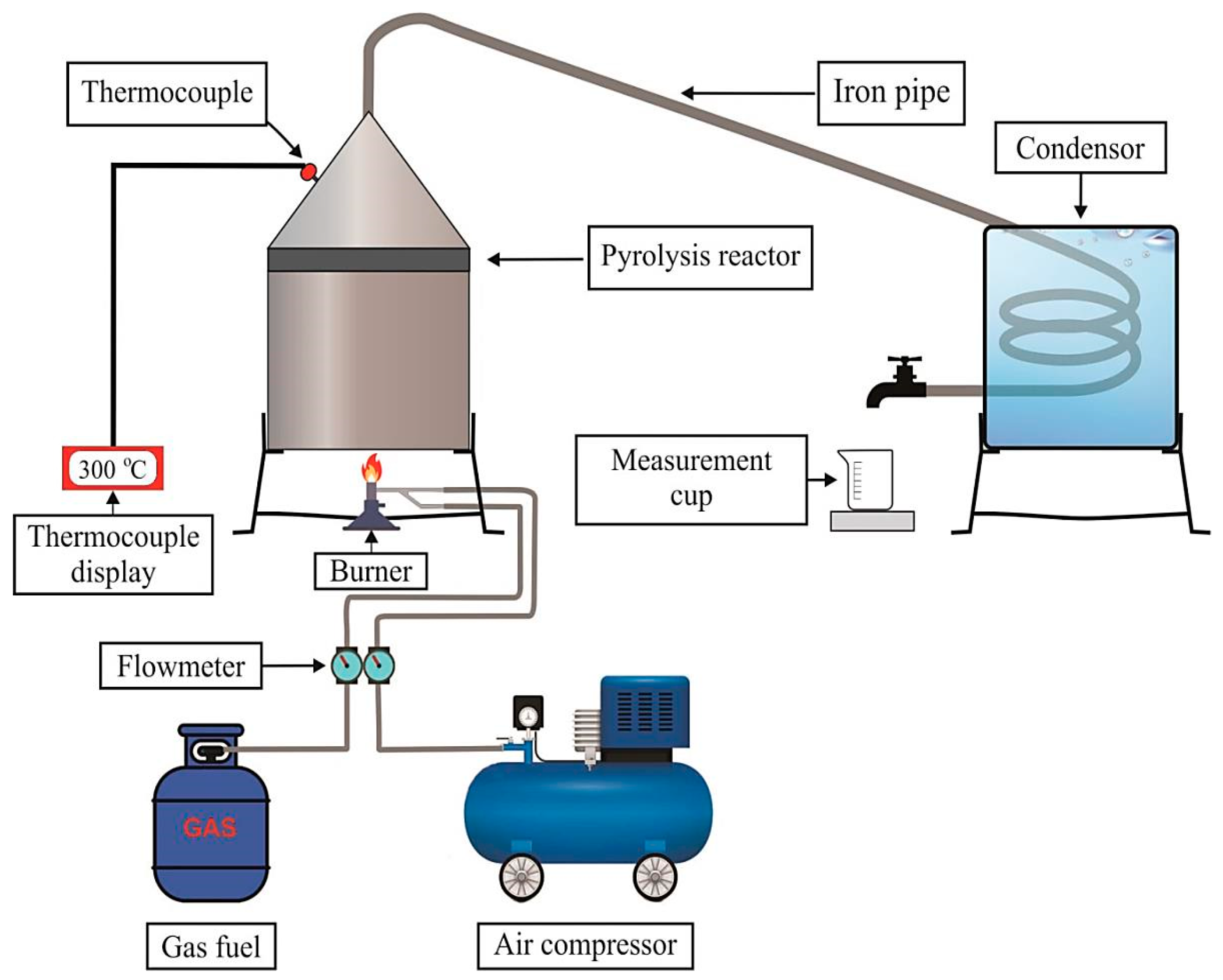
2.2. Production of WPO via Pyrolysis
2.3. Fuel Preparation
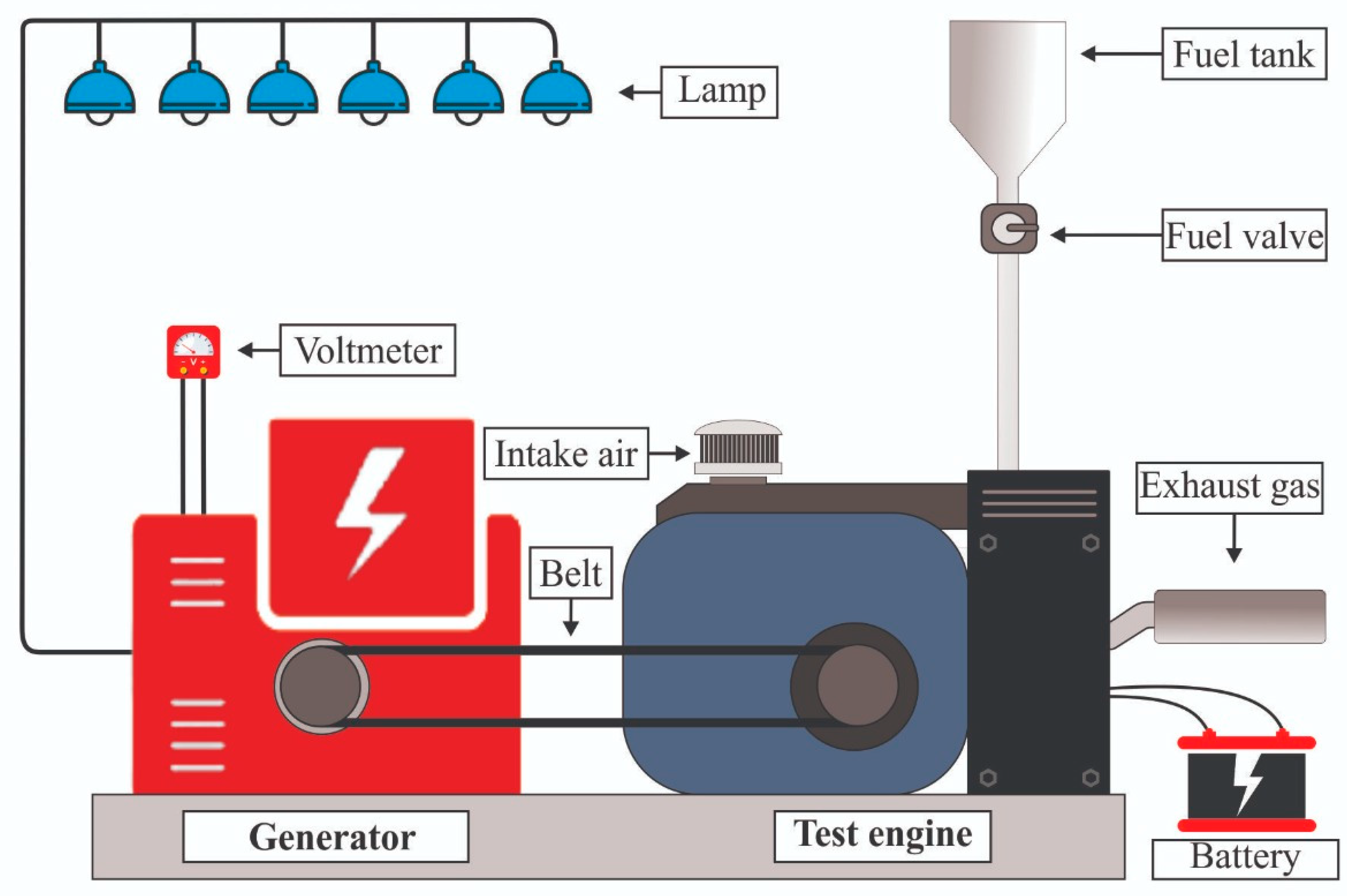
2.4. Engine Performance Test Procedure
2.5. Engine Performance
2.5.1. Engine Power
2.5.2. Specific Fuel Consumption
2.5.3. Thermal Efficiency
3. Results and Discussion
3.1. The Characteristics of Physicochemical Properties of WPO
3.2. Engine Performance Fueled by Waste Plastic Oil–Gasoline (RON 88) Blends
3.2.1. The Effect of WPO Blends on Brake Power
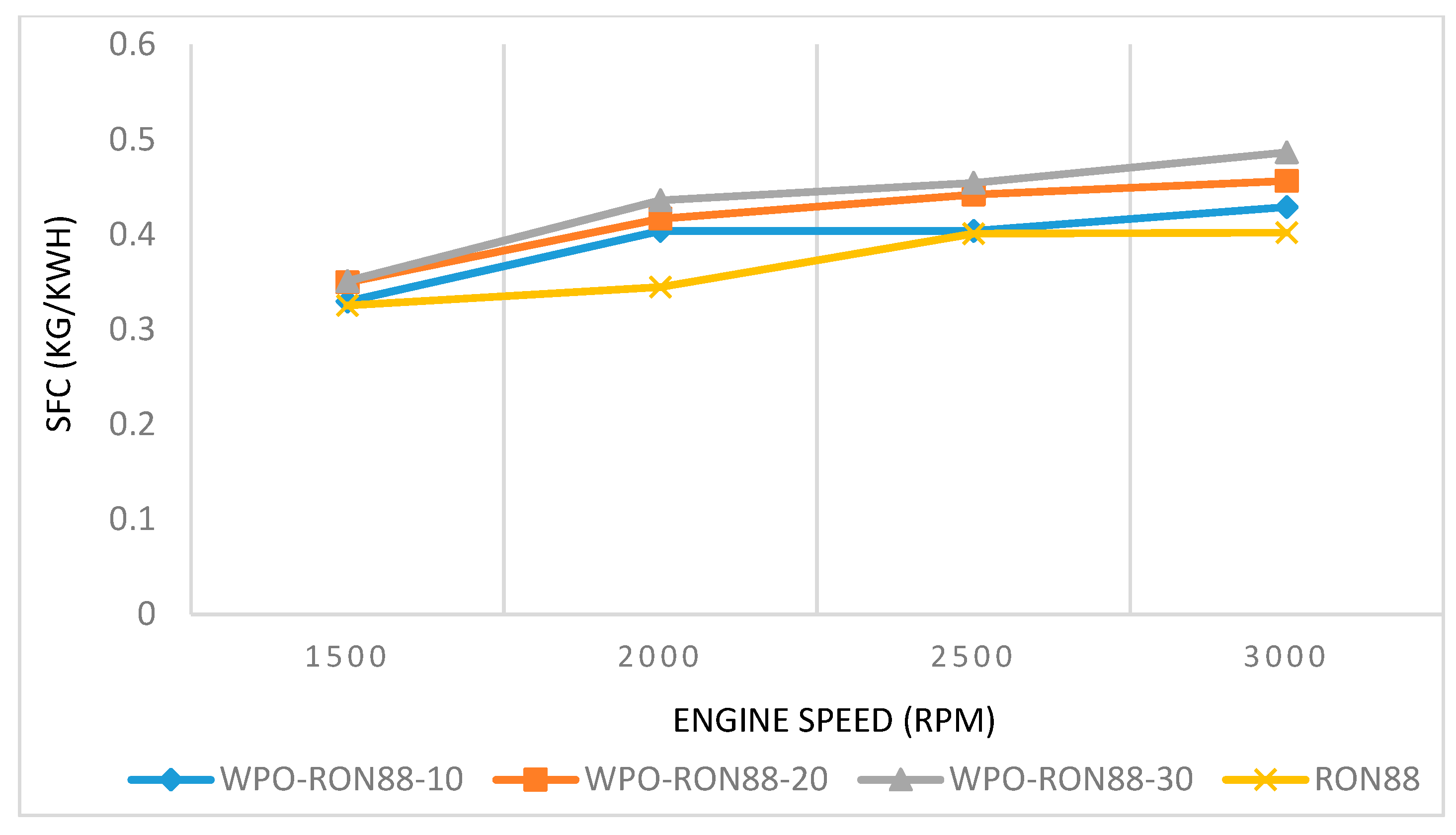
3.2.2. The Effect of WPO Blends on SFC
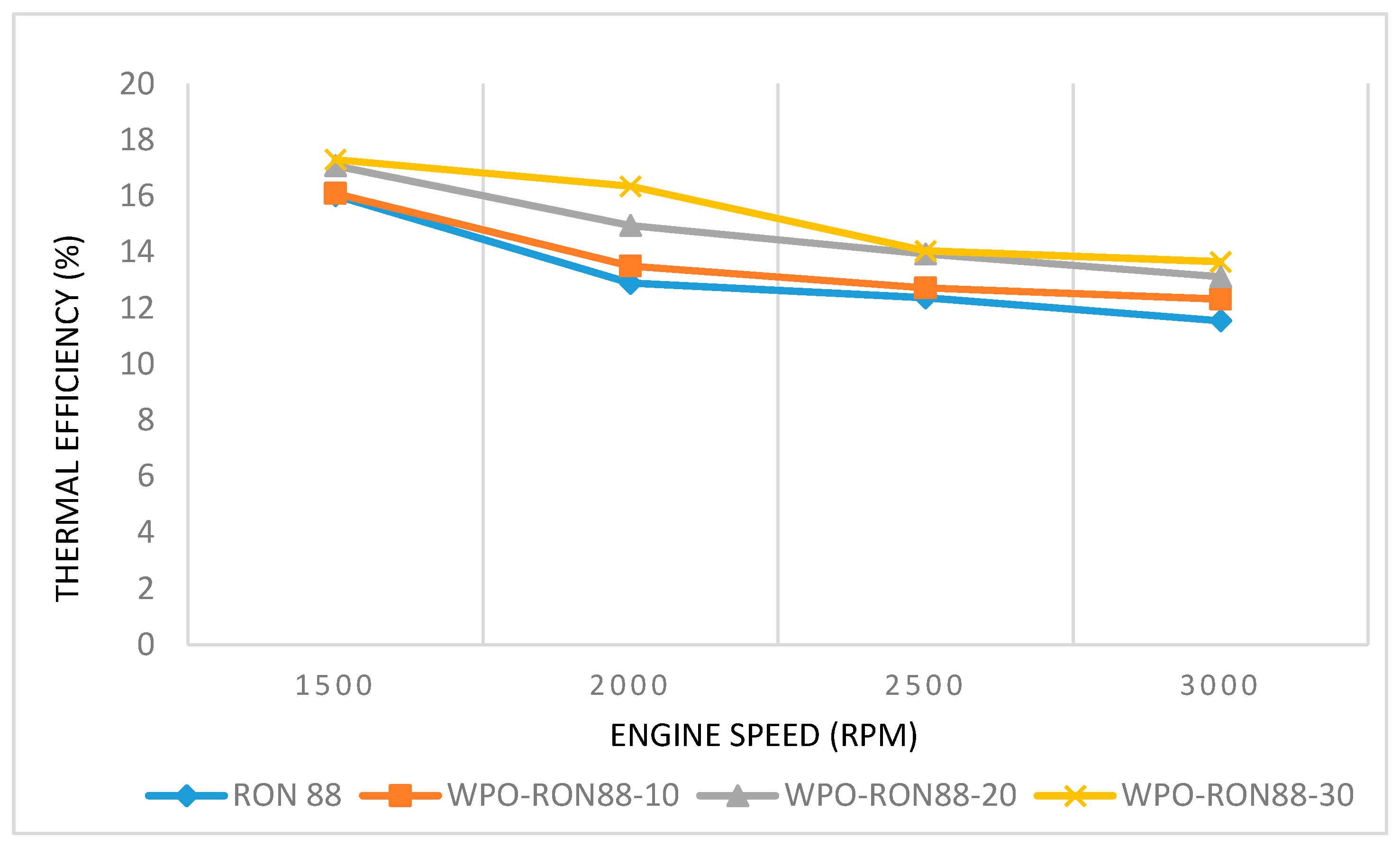
3.2.3. The Effect of WPO Blends on Thermal Efficiency
3.3. Engine Performance Fueled by WPO–Gasoline (RON 90) Blends
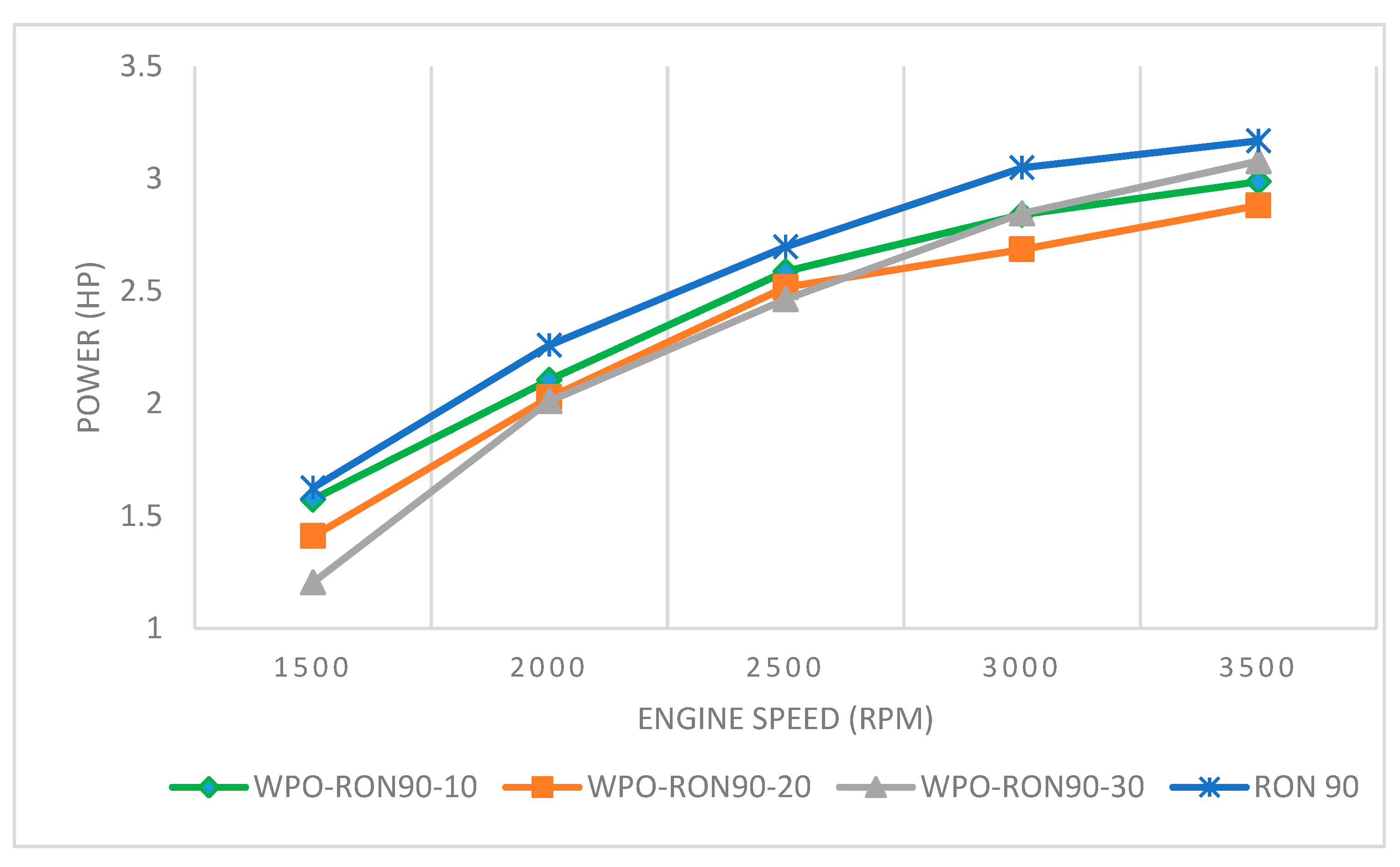
3.3.1. The Effect of WPO–Gasoline RON 90 Blends on Engine Power
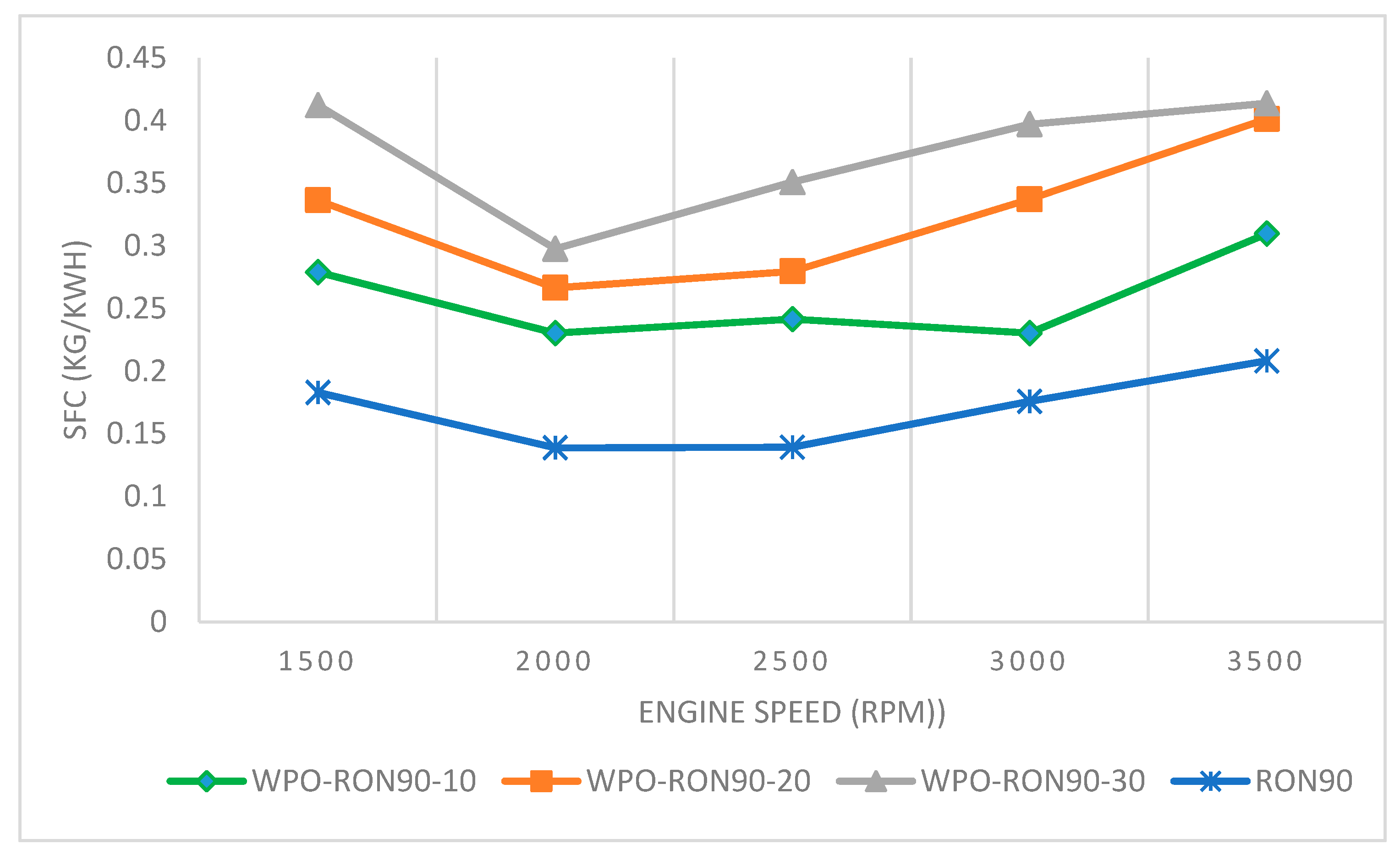
3.3.2. The Effect of WPO–Gasoline RON 90 Blends on SFC
3.3.3. The Effect of WPO–Gasoline RON 90 Blends on Thermal Efficiency
4. Conclusions
- The blend ratio of WPO with gasoline (RON88 and RON 90) was varied from 10% to 30% with gasoline. It was found that the engine power outputs of WPO-RON88 and WPO-RON90 blends were close to that of pure RON88 and RON90 fuels.
- The increasing concentration on the blending of WPO with gasoline (RON88 and RON 90) can reduce engine output power slightly. By contrast, the WPO-RON90-30 obtained satisfactory results at the maximum engine speed of 3500 rpm, which gave results very close to the engine power fueled by pure RON90 fuel.
- The engine power, SFC, and thermal efficiency improvement due to WPO blending showed more pronounced effects for WPO-RON90 blends than for WPO-RON88 blends.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Silitonga, A.S.; Masjuki, H.H.; Mahlia, T.M.I.; Ong, H.C.; Chong, W.T.; Boosroh, M.H. Overview properties of biodiesel diesel blends from edible and non-edible feedstock. Renew. Sustain. Energy Rev. 2013, 22, 346–360. [Google Scholar] [CrossRef]
- Shahir, S.A.; Masjuki, H.H.; Kalam, M.A.; Imran, A.; Rizwanul Fattah, I.M.; Sanjid, A. Feasibility of diesel–biodiesel–ethanol/bioethanol blend as existing CI engine fuel: An assessment of properties, material compatibility, safety and combustion. Renew. Sustain. Energy Rev. 2014, 32, 379–395. [Google Scholar] [CrossRef]
- Milano, J.; Ong, H.C.; Masjuki, H.H.; Silitonga, A.S.; Chen, W.-H.; Kusumo, F.; Dharma, S.; Sebayang, A.H. Optimization of biodiesel production by microwave irradiation-assisted transesterification for waste cooking oil-Calophyllum inophyllum oil via response surface methodology. Energy Convers. Manag. 2018, 158, 400–415. [Google Scholar] [CrossRef]
- Mason, I.G.; Page, S.C.; Williamson, A.G. A 100% renewable electricity generation system for New Zealand utilising hydro, wind, geothermal and biomass resources. Energy Policy 2010, 38, 3973–3984. [Google Scholar] [CrossRef]
- Ismail, M.S.; Moghavvemi, M.; Mahlia, T.M.I. Characterization of PV panel and global optimization of its model parameters using genetic algorithm. Energy Convers. Manag. 2013, 73, 10–25. [Google Scholar] [CrossRef]
- Mahlia, T.M.I.; Syaheed, H.; Abas, E.P.; Kusumo, F.; Shamsuddin, A.H.; Ong, H.C.; Bilad, M.R. Organic Rankine Cycle (ORC) System Applications for Solar Energy: Recent Technological Advances. Energies 2019, 12, 2930. [Google Scholar] [CrossRef]
- Mofijur, M.; Mahlia, T.M.I.; Silitonga, A.S.; Ong, H.C.; Silakhori, M.; Hasan, M.H.; Putra, N.; Rahman, S.M.A. Phase Change Materials (PCM) for Solar Energy Usages and Storage: An Overview. Energies 2019, 12, 3167. [Google Scholar] [CrossRef]
- Mahlia, T.M.I.; Syazmi, Z.A.H.S.; Mofijur, M.; Abas, A.E.P.; Bilad, M.R.; Ong, H.C.; Silitonga, A.S. Patent landscape review on biodiesel production: Technology updates. Renew. Sustain. Energy Rev. 2020, 118, 109526. [Google Scholar] [CrossRef]
- Ong, H.C.; Masjuki, H.H.; Mahlia, T.M.I.; Silitonga, A.S.; Chong, W.T.; Yusaf, T. Engine performance and emissions using Jatropha curcas, Ceiba pentandra and Calophyllum inophyllum biodiesel in a CI diesel engine. Energy 2014, 69, 427–445. [Google Scholar] [CrossRef]
- Ahvenainen, R. Modern Plastics Handbook; Woodhead Publishing Limited: Cambridge, UK, 2003. [Google Scholar]
- Jambeck, J.R.; Geyer, R.; Wilcox, C.; Siegler, T.R.; Perryman, M.; Andrady, A.; Narayan, R.; Law, K.L. Plastic waste inputs from land into the ocean. Science 2015, 347, 768–771. [Google Scholar] [CrossRef]
- Finance, M.O. Bumi Dalam Kantong Plastik; Kementerian Keuangan: Jakarta, Indonesia, 2019; Volume XIV, No. 144.
- Lamb, J.B.; Willis, B.L.; Fiorenza, E.A.; Couch, C.S.; Howard, R.; Rader, D.N.; True, J.D.; Kelly, L.A.; Ahmad, A.; Jompa, J.; et al. Plastic waste associated with disease on coral reefs. Science 2018, 359, 460–462. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.L.; Ngadi, N.; Abdullah, T.A.T.; Inuwa, I.M. Current state and future prospects of plastic waste as source of fuel: A review. Renew. Sustain. Energy Rev. 2015, 50, 1167–1180. [Google Scholar] [CrossRef]
- Siddiqui, M.N.; Redhwi, H.H. Pyrolysis of mixed plastics for the recovery of useful products. Fuel Process. Technol. 2009, 90, 545–552. [Google Scholar] [CrossRef]
- Lee, J.; Lee, T.; Tsang, Y.F.; Oh, J.-I.; Kwon, E.E. Enhanced energy recovery from polyethylene terephthalate via pyrolysis in CO2 atmosphere while suppressing acidic chemical species. Energy Convers. Manag. 2017, 148, 456–460. [Google Scholar] [CrossRef]
- Khairil; Jihad, M.; Indra Riayatsyah, T.M.; Bahri, S.; Sofyan, S.E.; Jalaluddin. The effect of gasoline-waste plastics oil blends on SI engine performance at high-speed rotation. IOP. Conf. Ser. Earth Environ. Sci. 2020, 463, 012002. [Google Scholar] [CrossRef]
- Surono, U.B. Berbagai metode konversi sampah plastik menjadi bahan bakar minyak. J. Tek. 2013, 3, 32–40. [Google Scholar]
- Bezergianni, S.; Dimitriadis, A.; Faussone, G.-C.; Karonis, D. Alternative Diesel from Waste Plastics. Energies 2017, 10, 1750. [Google Scholar] [CrossRef]
- Chong, C.T.; Mong, G.R.; Ng, J.-H.; Chong, W.W.F.; Ani, F.N.; Lam, S.S.; Ong, H.C. Pyrolysis characteristics and kinetic studies of horse manure using thermogravimetric analysis. Energy Convers. Manag. 2019, 180, 1260–1267. [Google Scholar] [CrossRef]
- Ong, H.C.; Chen, W.-H.; Farooq, A.; Gan, Y.Y.; Lee, K.T.; Ashokkumar, V. Catalytic thermochemical conversion of biomass for biofuel production: A comprehensive review. Renew. Sustain. Energy Rev. 2019, 113, 109266. [Google Scholar] [CrossRef]
- Cleetus, C.; Thomas, S.; Varghese, S. Synthesis of Petroleum-Based Fuel from Waste Plastics and Performance Analysis in a CI Engine. J. Energy 2013, 2013, 608797. [Google Scholar] [CrossRef]
- Khan, M.Z.H.; Sultana, M.; Al-Mamun, M.R.; Hasan, M.R. Pyrolytic waste plastic oil and its diesel blend: Fuel characterization. J. Environ. Public Health 2016, 2016, 7869080. [Google Scholar] [CrossRef] [PubMed]
- Soltes, E.J.; Elder, T.J. Pyrolysis. In Organic Chemicals from Biomass; CRC Press: Boca Raton, FL, USA, 2018; pp. 63–99. [Google Scholar]
- Sultan, M.A.-S.; Yang, Y.; Wang, J.; Gary, A.L. Pyro-Oil and Wax Recovery from Reclaimed Plastic Waste in a Continuous Auger Pyrolysis Reactor. Energies 2020, 13, 2040. [Google Scholar]
- Lee, X.J.; Ong, H.C.; Gan, Y.Y.; Chen, W.-H.; Mahlia, T.M.I. State of art review on conventional and advanced pyrolysis of macroalgae and microalgae for biochar, bio-oil and bio-syngas production. Energy Convers. Manag. 2020, 210, 112707. [Google Scholar] [CrossRef]
- Ramadhan, A.; Ali, M. Pengolahan sampah plastik menjadi minyak menggunakan proses pirolisis. J. Ilm. Tek. Lingkung. 2012, 4, 44–53. [Google Scholar]
- Sarker, M.; Rashid, M.M.; Rahman, M.S.; Molla, M. Environmentally harmful low density waste plastic conversion into kerosene grade fuel. J. Environ. Prot. 2012, 3, 700. [Google Scholar] [CrossRef]
- Indonesia: Fuels: Diesel and Gasoline. Available online: https://www.transportpolicy.net/standard/indonesia-fuels-diesel-and-gasoline/ (accessed on 8 November 2019).
- Silitonga, A.S.; Atabani, A.E.; Mahlia, T.M.I.; Masjuki, H.H.; Badruddin, I.A.; Mekhilef, S. A review on prospect of Jatropha curcas for biodiesel in Indonesia. Renew. Sustain. Energy Rev. 2011, 15, 3733–3756. [Google Scholar] [CrossRef]
- Mohanraj, C.; Senthilkumar, T.; Chandrasekar, M. A review on conversion techniques of liquid fuel from waste plastic materials. Int. J. Energy Res. 2017, 41, 1534–1552. [Google Scholar]
- Thahir, R.; Altway, A.; Juliastuti, S.R.; Susianto. Production of liquid fuel from plastic waste using integrated pyrolysis method with refinery distillation bubble cap plate column. Energy Rep. 2019, 5, 70–77. [Google Scholar] [CrossRef]
- Priambodo, B.; Maleev, V. Operasi dan Pemeliharaan Mesin Diesel; Penerbit Erlangga: Jakarta, Indonesia, 1991. [Google Scholar]
- Marthur, S.; Sharma, R. A Course in Internal Combustion Engine; JC Kapur: For Dhanpat Rai & Son: Delhi/Julundur, India, 1980. [Google Scholar]
- Ananthakumar, S.; Jayabal, S.; Thirumal, P. Investigation on performance, emission and combustion characteristics of variable compression engine fuelled with diesel, waste plastics oil blends. J. Braz. Soc. Mech. Sci. Eng. 2017, 39, 19–28. [Google Scholar] [CrossRef]
- Vijaya Kumar, K.; Puli, R.K. Effects of Waste Plastic Oil Blends on a Multi Cylinder Spark Ignition Engine. MATEC Web Conf. 2017, 108, 08005. [Google Scholar] [CrossRef]
- Zainuri, F. Pirolisis Sampah Plastik Hingga Suhu 900 °C Sebagai Upaya Menghasilkan Bahan Bakar Ramah Lingkungan; Universitas Muhammadiyah Surakarta: Surakarta, Indonesia, 2014. [Google Scholar]
- Pratama, A.W.; Budi, A. Design of Polypropylene Distillation Fuel Generator (PDFG) as an effort to create alternative energy for standar engine. In Proceedings of the International Conference on Food and Agriculture, Bali, Indonesia, 20–21 October 2018. [Google Scholar]
- Rao, R.N.; Silitonga, A.S.; Shamsuddin, A.H.; Milano, J.; Riayatsyah, T.M.I.; Sebayang, A.H.; Nur, T.B.; Sabri, M.; Yulita, M.R.; Sembiring, R.W. Effect of Ethanol and Gasoline Blending on the Performance of a Stationary Small Single Cylinder Engine. Arab. J. Sci. Eng. 2020. [Google Scholar] [CrossRef]
- Fattah, I.M.R.; Masjuki, H.H.; Kalam, M.A.; Mofijur, M.; Abedin, M.J. Effect of antioxidant on the performance and emission characteristics of a diesel engine fueled with palm biodiesel blends. Energy Convers. Manag. 2014, 79, 265–272. [Google Scholar] [CrossRef]
- Murni, M. Kaji Eksperimental Pengaruh Temperatur Biodiesel Minyak Sawit Terhadap Performansi Mesin Diesel Direct Injection Putaran Konstan. Master’s Thesis, Diponegoro University, Semarang, Indonesia, 2010. [Google Scholar]



| Fuel Type | WPO (%) | Gasoline Type | Volume | |
|---|---|---|---|---|
| RON88 (%) | RON90 (%) | |||
| WPO-RON88-10 | 10 | 90 | - | 1 L |
| WPO-RON88-20 | 20 | 80 | - | 1 L |
| WPO-RON88-30 | 30 | 70 | - | 1 L |
| RON88-100 | 0 | 100 | 1 L | |
| WPO-RON90-10 | 10 | - | 90 | 1 L |
| WPO-RON90-20 | 20 | - | 80 | 1 L |
| WPO-RON90-30 | 30 | - | 70 | 1 L |
| RON90-100 | 0 | 100 | 1 L | |
| Brand | Specification (Honda) |
|---|---|
| Model | GP160H-SD1 |
| Power | 5.5 HP |
| Engine type | Air-Cooled 4 Stroke OHV Single Cylinder, Horizontal Shaft |
| Cylinder Volume | 163 cc |
| Bore × Stroke | 68 × 45 mm |
| Compression Ratio | 8.5:1 |
| Maximum Torque | 10.3 Nm at 2500 rpm |
| Maximum Output | 5.5 HP at 3600 rpm |
| Output Net | 3.6 HP at 3600 rpm |
| Properties | Unit | ASTM D6751 Standard | RON 88 [11] | RON 90 [11,38] | WPO a | WPO b [36] | Method |
|---|---|---|---|---|---|---|---|
| Kinematic Viscosity | (mm2/s) | 1.9–6.0 | 2.0–5.0 | 2.0–5.0 | 3.5 | 2.64 | ASTM D4052 |
| Density at 15 °C | (Kg/m3) | 880 max | 715–770 | 715–770 | 750 | 830 | ASTMD4052 or D1298 |
| Sulfur Content | % mm | Max. 0.05 | 0.05 | 0.05 | 0.68 | - | ASTM D664 |
| Calorific Value | MJ/kg | - | 47.049 | 48.20 | 45.0196 | 42.8075 | ASTM D240 |
| RON | RON (min) | - | 88 | 90 | 78.8 | - | ASTMD2699 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khairil; Riayatsyah, T.M.I.; Bahri, S.; Sofyan, S.E.; Jalaluddin, J.; Kusumo, F.; Silitonga, A.S.; Padli, Y.; Jihad, M.; Shamsuddin, A.H. Experimental Study on the Performance of an SI Engine Fueled by Waste Plastic Pyrolysis Oil–Gasoline Blends. Energies 2020, 13, 4196. https://doi.org/10.3390/en13164196
Khairil, Riayatsyah TMI, Bahri S, Sofyan SE, Jalaluddin J, Kusumo F, Silitonga AS, Padli Y, Jihad M, Shamsuddin AH. Experimental Study on the Performance of an SI Engine Fueled by Waste Plastic Pyrolysis Oil–Gasoline Blends. Energies. 2020; 13(16):4196. https://doi.org/10.3390/en13164196
Chicago/Turabian StyleKhairil, Teuku Meurah Indra Riayatsyah, Samsul Bahri, Sarwo Edhy Sofyan, Jalaluddin Jalaluddin, Fitranto Kusumo, Arridina Susan Silitonga, Yanti Padli, Muhammad Jihad, and Abd Halim Shamsuddin. 2020. "Experimental Study on the Performance of an SI Engine Fueled by Waste Plastic Pyrolysis Oil–Gasoline Blends" Energies 13, no. 16: 4196. https://doi.org/10.3390/en13164196
APA StyleKhairil, Riayatsyah, T. M. I., Bahri, S., Sofyan, S. E., Jalaluddin, J., Kusumo, F., Silitonga, A. S., Padli, Y., Jihad, M., & Shamsuddin, A. H. (2020). Experimental Study on the Performance of an SI Engine Fueled by Waste Plastic Pyrolysis Oil–Gasoline Blends. Energies, 13(16), 4196. https://doi.org/10.3390/en13164196








