1. Introduction
Ammonium nitrate (AN) is a crystalline, odorless and white, grey or brown solid in the form of prills, pellets or flakes. AN may be applied either in agriculture as a fertilizer (AN-F) or in mining as an oxidizer component in explosives [
1]. The shape of AN-F is similar to granules. AN-F is characterized by a ca. 35% content of nitrogen. Furthermore, it is highly soluble in water, has a high density and low retention. The absorption index is ca. 8% [
2]. In turn, the ammonium nitrate porous prill (AN-PP) has a lower density than AN-F. However, porosity, retention and absorption index are higher by ca. 12–16% in comparison with its AN-F counterpart [
3,
4,
5]. Landucci et al. indicate that ammonium(V) nitrate, which is applied in the mining industry, has a 20% void fraction and is much more energy-efficient [
6]. AN-PP is therefore recommended for blasting companies instead of AN-F.
Under normal pressure, AN may be present in five different phases, which occur depending strictly on the temperature range I (ε-regular), II (δ-trigonal), III (γ-rhombic), IV (β-rhombic) and V (α-tetragonal) [
7]. It was reported that in the production of ANFO from AN, the most crucial transformation is II→III at ca. 32 °C due to a distinct increase of crystal volume by ca. 3.4%, which leads to the prilling process [
3,
4]. Harju explains this is due to vast differences of AN phase III in comparison with other polymorphic forms [
8]. In stage III, the position of the central nitrogen atom is less regular than in different phases [
9,
10]. Harju, Choi and Prask stated that the amplitude of the nitrate ions’ reorientations was negligible in comparison to phase II [
8,
11].
It was reported that some physicochemical properties of ammonium nitrate depend on the provenance of AN [
2,
12,
13,
14,
15,
16]. Lotspeich and Petr, Biessikirski et al. and Biessikirski and Kuterasiński investigated the morphology of AN mini-prills [
2], AN-F, AN-PP in both prill and powder form and non-ideal explosives [
12,
13] using SEM. It was indicated that the number of surface deformations depended on the type of AN grade. Rao et al. made an evaluation of AN morphology, prill caking, rheology, friability and ANFO’s explosive characteristics depending on the AN provenance [
14]. Miyake et al. [
15] reported that the increase of pore diameter resulted in a decrease in detonation velocity (VOD). Viktorov et al. [
16] stated that a change in the microstructure of AN granules led to an increase in retention capacity. Kwok and Jones [
17] indicated that different forms of AN had an impact on octane thermodesorption from the AN surface. According to [
17,
18,
19,
20,
21,
22], the melting and decomposition points of AN are ca. 170 °C and over 210 °C respectively [
22].
The influence of density and confinement on the detonation velocity (VOD) of ANFO was discussed in [
23,
24,
25,
26,
27,
28]. Buczkowski and Zygmunt studied the importance of the physical properties of AN prills on non-ideal explosive blasting parameters, e.g., velocity of detonation (VOD) [
29]. Analysis of the reported data led to the conclusion that the increased density of ANFO caused an increase in VOD.
Despite access to numerous reports concerning AN research, information on the relationship between either morphology or the absorption index and the blasting properties of ANFO was not found. Moreover, some of the blasting representatives state that AN-PP of a higher absorption ratio always allows ANFO of the higher VOD to be obtained, in comparison with the ANFO which is obtained on another assortment of the same AN-PP, whose main difference is only the lower absorption ratio. Hence, this research aims to investigate the dependence of either VOD and the toxic fumes of ANFO-type materials on the two different assortments of AN-PP and their absorption indices (with an assumption of a constant mass of the fuel component).
2. Materials and Methods
AN-PP7 was supplied by Yara’s International A SA and donated to the research by the SSE Polska Sp. z o.o. blasting company. AN-PP contained ca. 35.0% nitrogen. The prill diameter and bulk density were 0.8 mm and 740 kg∙m−3, respectively. The water content did not exceed 0.3%. The absorption index was ca. 14% and was provided by the blasting company according to technical sheets.
AN-PP8 was manufactured by the Yara’s International A SA and donated to the research by the SEE Polska Sp. z o.o. AN-PP contained ca. 35.0% nitrogen. The prill diameter was 1 mm. Bulk density of AN-PP8 was 820 kg∙m−3. The water content did not exceed 0.3%. The absorption index was ca. 12% and was provided by the blasting company according to technical sheets.
Fuel oil (FO) was manufactured by Silesia Oil Sp. z o.o. It was a wide-range sample fraction of C10-C20 hydrocarbons. FO was characterized by the bulk density of 800 kg∙m
−3. Kinetic viscosity of the donated sample was 13.6 mm
2·s
−1 at 40 °C. The detailed characteristics and discussion on applied FO low-temperature properties may be found in [
30].
Three types of sample were prepared: single AN, AN blended with FO and AN mixed with an excess of FO.
All AN-based samples wetted with FO were obtained by blending AN-PP7 or AN-PP8 with FO for 20 min at 250 rpm, using a mixer, according to the weight ratio of 94.5:5.5.
All AN-based samples wetted with an excess of FO were obtained by mixing AN-PP7 or AN-PP8 with fuel oil according to the wt ratio of 90.0:10.0.
ANFO based on AN-PP7 and AN-PP8 were designated as samples 1 and 2, respectively.
The SEM micrographs of all tested non-ideal explosives were taken by Nova NanoSEM 200 produced by FEI Europe (5–18 keV). Prior to the SEM measurements, all samples were coated with a carbon layer in a carbon coater. Non-ideal explosives samples were placed in holders using double-sided, conductive, vacuum compatible carbon tape. Images were taken under low vacuum, ca. 60 Pa, and electron beam voltage of 10 kV. The Low Vacuum Detector (LVC) used was operated in secondary electron mode. The FT-IR research was conducted in an iS-10 spectrometer, which was produced by Thermo Scientific. The spectrometer was equipped with both a MCT detector and ATR adapter. IR spectra were recorded in the wavenumber range of 650–4000 cm−1 with 32 scans per spectrum.
X-ray Powder Diffraction (XRD) patterns were obtained using the PANalytical X’Pert PRO MPD. Applied CuKα radiation was at 40kV and 30 mA. XRD scanning was carried out in the 2θ range of 5 ÷ 50°. The applied step was 0.033°. All tested non-ideal explosive samples were in the form of powder and were placed in holders. All XRD measurements were performed at room temperature.
Differential Scanning Calorimetry (DSC) and Thermogravimetry (TG) analyses were conducted using NETZSCH STA 409 PC/PG with a heating rate of 10 °C·min−1.
The controlled temperature desorption of FO from the surface of the AN prill was carried out at 20–700 °C. All studied samples were kept under an airflow of 30 mL·min−1, both in the furnace and the balance chamber. Air was applied to stimulate detonation conditions. For each experiment, the prepared sample was placed in the DSC aluminum pan using a spatula. Sample weight was 20 mg. The baseline of the TG was confirmed by application of the same heating profile for the empty pan. The TG drift was ca. 5 μg, which represents 0.02 mass%.
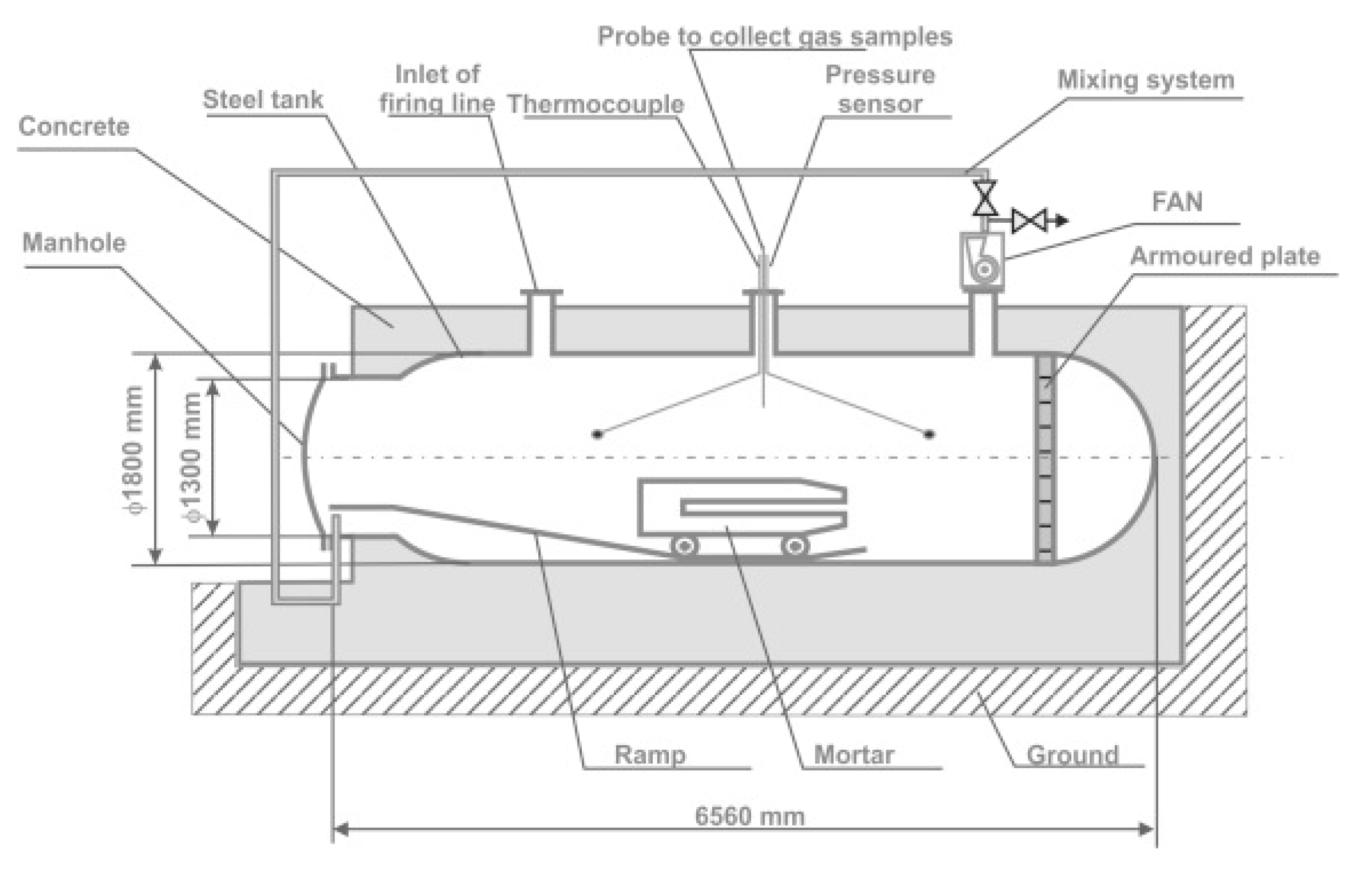
Post-blast oxides were determined according to the standard [
31], which complies with the directive [
32]. Explosive charges of a mass of 600 g were detonated inside the steel mortar in the blasting chamber (
Figure 1).
After detonation, post-blast fumes were homogenized for 3 min, using a mixing procedure. Subsequently, post-blast gases were collected for 20 min in the ventilation system. The amounts of COx and NOx were determined by IR (MIR 25e) and chemiluminescent (TOPAZE 32M) analyzers, respectively.
Additionally, the relative general toxicity (
LCO) was calculated according to Equation (1):
where CO and NO
x are concentrations of carbon monoxide and nitrogen oxides in post-blast gases, obtained from 1 kg of explosive.
VOD was calculated according to [
34]. The ANFO charge (
Figure 2) was introduced into a glass tube with an internal diameter of 46 mm. Two short circuit probes were placed via small holes which were drilled throughout the tube. The distance between the probes was 150 mm. The measurement consisted of the time difference which describes the first recorded signal change on each probe. VOD was calculated according to Equation (2).
where
l is the test base distance between two probes and
t is a time difference.
VOD and post-blast gases were measured simultaneously at the blasting chamber. The Royal Demolition Explosive (RDX)-based booster initiated ANFO with a mass of 14 g.
3. Results and Discussion
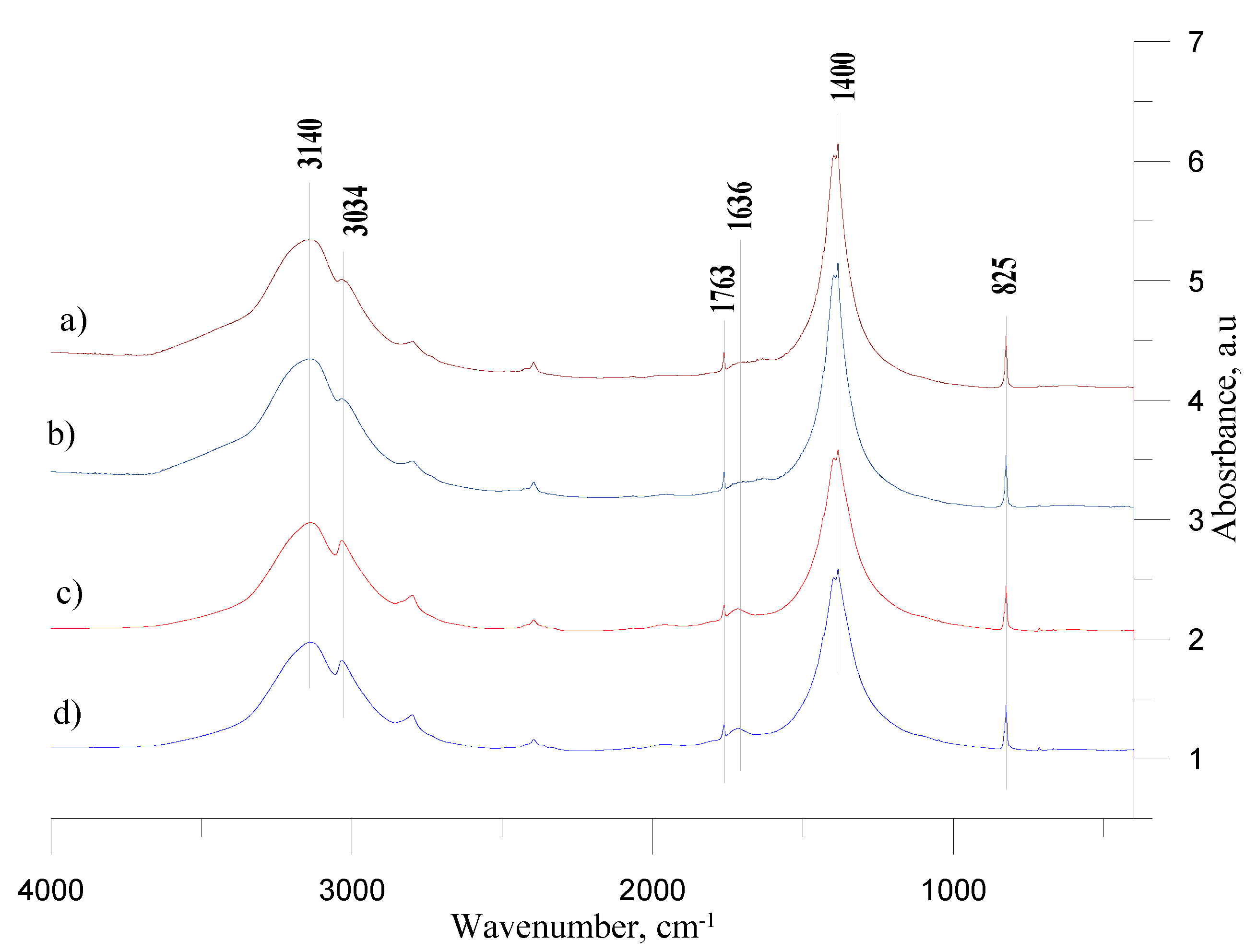
In the case of AN-PP7 and AN-PP8, the peak at 3140 cm
−1 should be explained by the asymmetric NH
4+ stretching mode. The visible shoulder at 3034 cm
−1 corresponds to asymmetric ammonium ion deforming vibrations (
Figure 3, line a and line b). The weak band at 1763 cm
−1 can be explained by the in-plane deformation of the nitrate anion. However, it may be in line with a combination of an asymmetric distortion of NH
4+ with a lattice mode [
9,
35]. The small signal at ca. 16.36 cm
−1 is probably due to H
2O deformation vibration from moisture [
36]. The distinct band at 1400 cm
−1 corresponds to triple degenerated ammonium cation deformation as well as to the double degenerated stretching vibration of nitrate anions. The small signal at 825 cm
−1 corresponds with an out-of-plane deformation of
3− [
7,
37].
The addition of fuel oil to ammonium nitrate caused the appearance of the bands in the range of 2960 ÷ 2854 cm
−1, (line c and line d in
Figure 3), which can be assigned to the symmetric (2854 cm
−1) and asymmetric –CH
3 (2920 cm
−1) stretching and symmetric -CH
2- stretching (2926 cm
−1) vibrations.
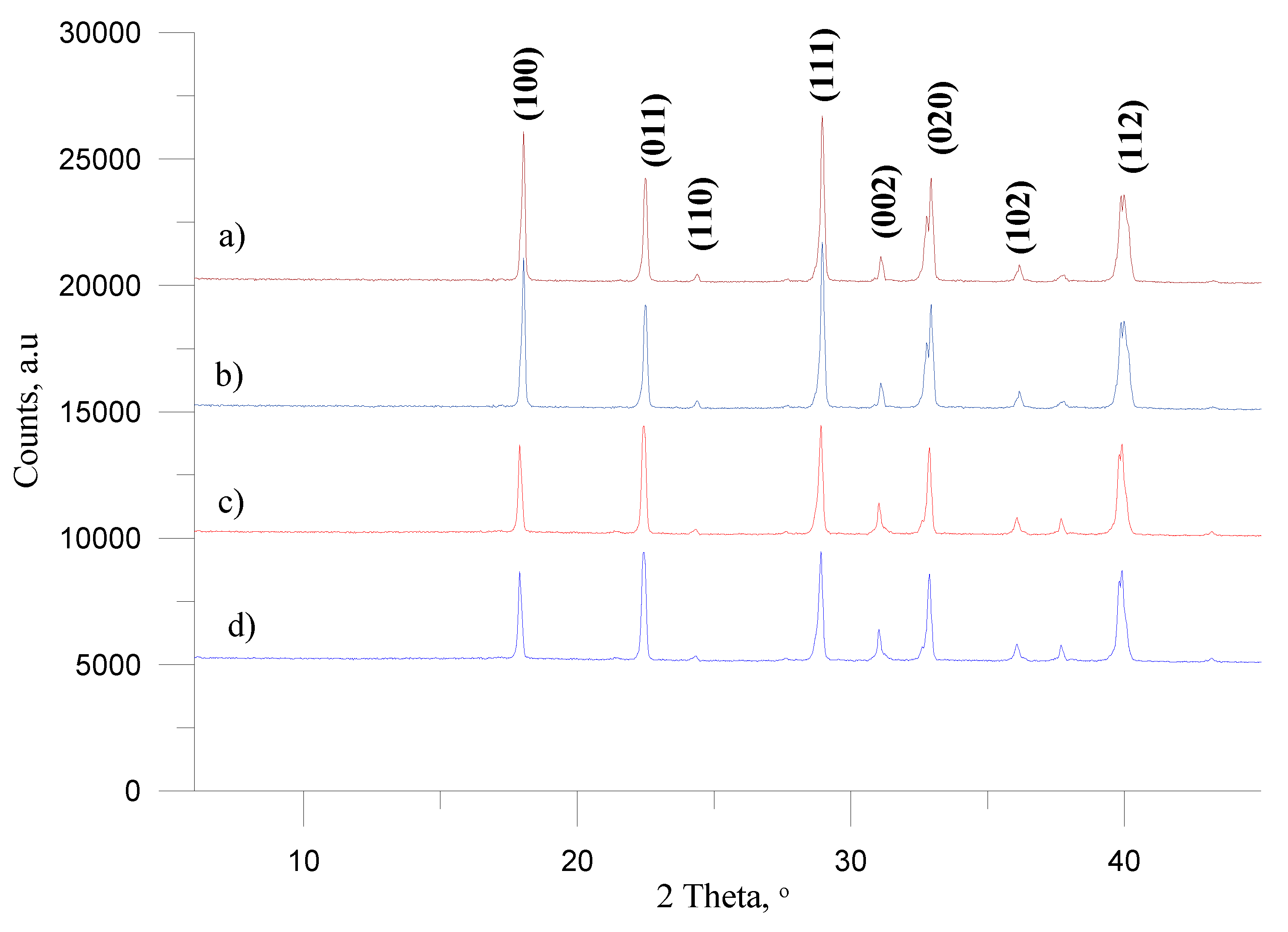
The analysis of XRD images showed that the dominant influence of the ammonium(V) nitrate belonged to the XRD patterns of pure AN-PP7, (
Figure 4, line a) and AN-PP8 (
Figure 4, line b) showed the diffraction peaks at 2θ = 18˚, 22˚, 24˚, 29˚, 31˚, 33˚, 36˚ and 40˚ corresponding to (100), (011), (110), (111), (002), (020), (102) and (112) respectively [
9,
13,
36].
Furthermore, from the XRD data, it may be concluded that different assortments of ammonium nitrate (AN-PP7 vs. AN-PP8) had no influence on the appearance of XRD patterns. The data also indicated that the addition of FO to AN-PP7 (
Figure 4, line c) and AN-PP8 (
Figure 4, line d), did not cause significant changes in the appearance of the XRD pictures of such prepared ANFO materials.
Crystal surface of AN-PP7 and AN-PP8 was presented in
Figure 5.
Based on SEM micrographs, surface deformations on the crystal surfaces of both AN-PP7 and AN-PP8 prill were found. Furthermore, for both samples the appearance of a central cavity was observed. This may be explained by the occurrence of stresses during the solidification of AN [
16,
38]. The surface around the central cavity was similar to the crystal surface of the prill [
16]. The differences observed between AN-PP7 and AN-PP8 corresponded to the prill sizes and the multiplicity of the surface deformations. In the case of AN-PP7, the prill size was smaller, and the number of structural distortions was higher in comparison to AN-PP8.
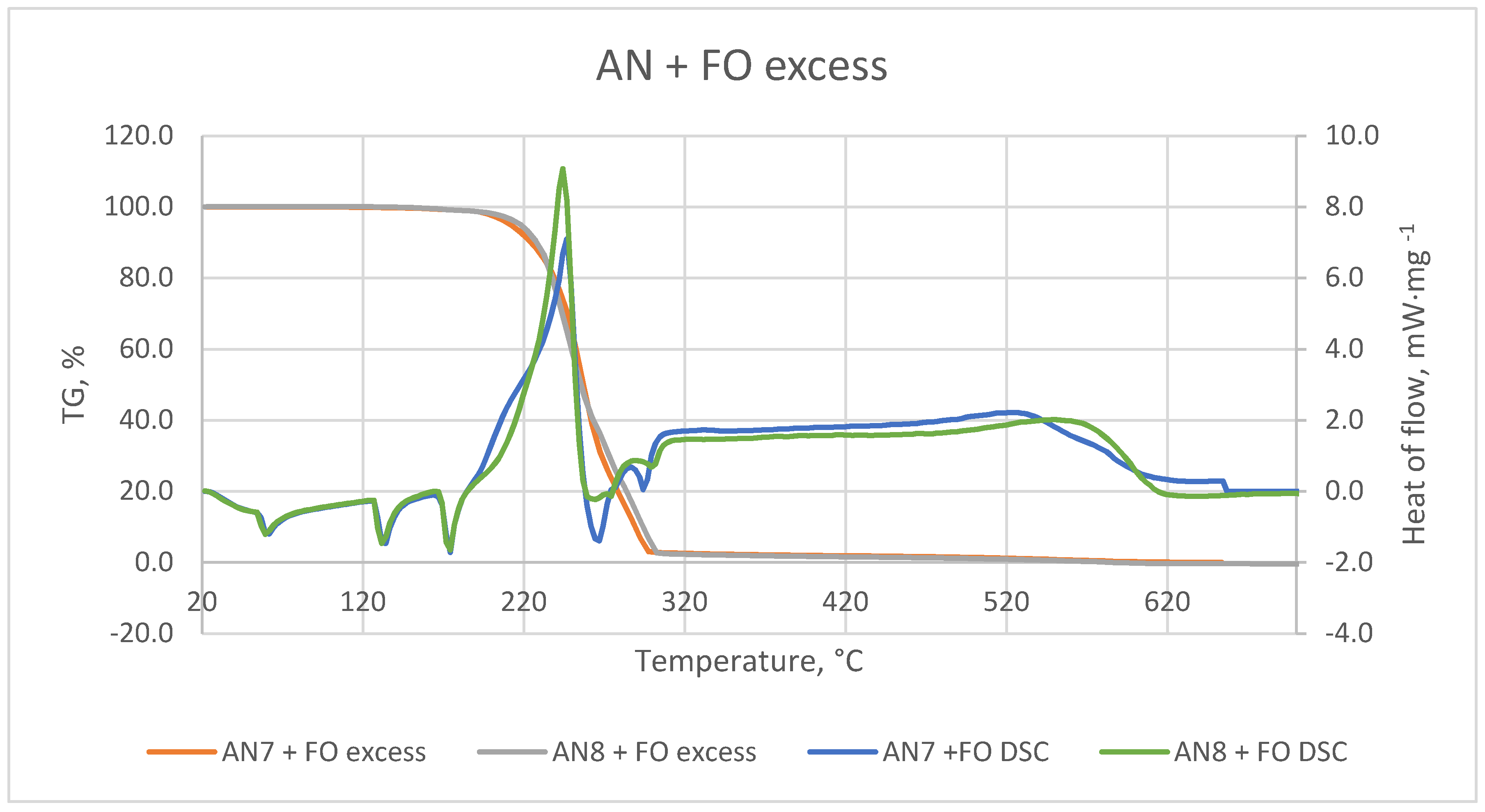
Figure 6 illustrates TG/DSC curves of the samples obtained from the mixing of AN with an excess of fuel oil at 20–700 °C. The DSC profile indicates four endothermic peaks at 60, 134, 176 and 266 °C. Two signals at ca. 60 and ca. 134 °C can be assigned to the crystallographic transformation of AN IV→II and AN II→I, respectively [
37]. A third endothermic peak at ca. 176 °C may be assigned to the melting point of the AN. Furthermore, an increase in temperature caused the thermal decomposition of AN, for which the fourth DSC peak has a maximum temperature of ca. 266 °C.
According to the TG curves, it can be concluded that mass loss was only caused by the evaporation of fuel oil from AN. The evaporation of FO from the crystal surfaces of AN-PP7 and AN-PP8 took place first from the surface pores and then from the FO monolayer. Finally, the evaporation continued from further FO layers [
18].
The DSC curve confirmed that AN-PP7 had a more significant absorption index than the AN-PP8.
Blasting tests (
Table 1) revealed no absorption index influence on the detonation velocity. This can be explained by the assumption of zero oxygen balance, which is used for explosives which are applied in the mining industry. The zero oxygen balance allows the maximum value of energy with the lowest concentration of the post-blast fumes to be obtained. However, this determines a fixed mass of AN-PP and FO in ANFO composition, which meets the condition of 94.5:5.5 wt ratio. An increase in the absorption ratio makes it possible to add more FO to the non-ideal explosive without any negative effects like dribbling FO from ANFO; however, additional amounts of FO will shift the oxygen balance towards negative values. This means that in the case of AN-PP7, additional FO would slightly increase non-ideal explosive density, which only in that case would influence the VOD, but on the other hand would result in the increase of carbon oxide content. From the gathered data it can be observed that the highest VOD (ca. 3140 m·s
−1) was registered for the AN-PP-8-based sample. Even though ANFO based on the AN-PP7 had a higher number of surface deformations, its VOD value was lower (ca. 1700 m·s
−1). On the other hand, some weak relationships between the density of the ANFO samples and their VOD could be observed. Increase in the density of the ANFO samples leads to increased VOD values [
23,
24,
25,
26,
27,
28].
According to
Table 2, none of the investigated explosives exceeded the maximum permissible amounts of NOx and CO, which, according to the Polish standard, are 16 and 27 dm
3·kg
−1, respectively. However, both ANFO explosives exceeded the maximum level of the relative general toxicity (
LCO), which equals 50 dm
3·kg
−1 [
33]. AN-PP-7-based ANFO was characterized by a lower content of toxic fumes (
LCO of ca. 81.5 dm
3·kg
−1, CO of ca. 15.0 dm
3·kg
−1, NOx of ca. 10.2 dm
3·kg
−1) in comparison to ANFO based on AN-PP8. This may be associated with a higher absorption index of AN-PP7 (14%) to AN-PP8 (12%). Taking into account the total volume of post-blast fumes of CO
2, CO and NO
x (AN-PP7 ca. of 150.2 dm
3·kg
−1 and AN-PP8 ca. of 174.0 dm
3·kg
−3), it may be concluded that with increasing volume of post-blast fumes the VOD increases. This could be explained by the influence of temperature and post-blast volume on the Chapman–Jouguet plain pressure, which in turn determines the VOD. As was mentioned before, a higher absorption index allows applying more FO to the non-ideal explosive without any adverse morphological effect (dribbling FO from the prill surface). However, in this case the oxide deficiency in the chemical structure of ANFO would result in an increased volume of carbon monoxide. Moreover, according to the Kistiakowski–Wilson and Springall–Roberts rule [
39], the volume of carbon and nitrogen dioxide should decrease, and a low volume of unreacted carbon particles could be present in post-blast fumes in soot form.
Another important factor which should be considered is the afterburning effect. This phenomenon is associated with unreacted FO or partially oxidized post-blast fumes. The secondary reactions are obtained from the products of primary reactions which subsequently mix with the surrounding air. In addition to the air volume, afterburning is also influenced by the temperature. During the post-blast analysis, the ANFO charge was placed in the mortar, which resulted in inhibition of the energy dissipation. Because ANFO is a low-sensitivity material with a high critical diameter, it was not possible to obtain reliable results in unconfined conditions due to possible interruption of the detonation. In order to verify the influence of confined (inside mortar) and unconfined (outside mortar) conditions, the reference RDX charge of a mass of 600 g was detonated in both conditions. Results of the post-blast analysis are presented in
Table 3.
Based on
Table 3, it can be concluded that the RDX explosive which was blasted in unconfined conditions was characterized by a lower total volume of post-blast fumes in comparison with confined RDX. This was probably caused by the fact that the outer walls of the unconfined RDX charge were not fully detonated. In addition, a significant increase in the CO-to-CO
2 ratio was reported. In the case of confined RDX, the CO-to-CO
2 ratio was ca. 0.018, and in the case of unconfined RDX it was 0.070. Such a significant difference could be explained by the appearance of secondary reactions. In the case of the non-ideal explosive based on AN-PP7 and AN-PP8, the concentration ratios equaled 0.120 and 0.113 respectively. This indicates that afterburning effects for both ANFO samples were similar. This could be explained by the same zero oxygen balance. A higher ratio of CO to CO
2 in the case of ANFO based on the AN-PP8 may be caused by a higher concentration of explosion energy in comparison with ANFO based on the AN-PP7. Moreover, research [
40] for both TNT and black powder has indicated that afterburning is influenced by the air-to-fuel-mass ratio. A significant and sudden decrease in pressure was found in the ratio range 1 ÷ 10. Above factor 10, carbon monoxide would be completely oxidized to carbon dioxide, and the pressure curve could decrease to volumetric effects. Furthermore, according to [
40], afterburning increases with a negative oxygen balance (oxygen deficiency). In the case of ANFO based on the AN-PP7, the additional FO content could lead to a driving detonation wave away from the charge. In that case, holding off the detonation wave would require a much longer time in comparison with an ANFO of a higher oxygen balance.
4. Conclusions
In this paper, the influence of the physicochemical properties of ammonium nitrate (AN) on the velocity of detonation (VOD) of ammonium nitrate fuel oil samples was presented.
XRD and IR analyses indicate that both assortments of AN (AN-PP7 and AN-PP8) revealed similar properties which resulted in the appearance of the same XRD patterns and IR spectra.
From the SEM micrograph, it can be concluded that both samples were characterized by the presence of numerous surface deformations, which were more prevalent in the case of AN-PP7, and these influenced the absorption index. A higher number of surface deformations increased the active surface contact between AN and fuel oil (FO).
Based on the DSC curves, it was proven that the presence of FO in AN did not affect the crystallographic transformation temperatures. Moreover, TG/DSC curves confirmed that AN-PP7 had a higher absorption ratio in comparison with AN-PP8.
From the analysis of TG curves, it was concluded that both types of AN decomposed at similar temperatures (ca. 164 °C). Furthermore, based on the TG curve, it was shown that mass loss in ANFO samples was mainly related to AN decomposition.
A higher absorption index allows the FO content in ANFO composition to be increased without any adverse effects like dribbling FO from the surface on the prill. This can improve the density of ANFO as well, as it would shift the oxygen balance towards negative values. Shifting the oxygen balance would result in an increase in carbon monoxide concentration, the possible presence of soot in post-blast fumes, as well as the increase of afterburning pressure effects.
Based on blasting tests, it was also demonstrated that VOD was independent of the absorption index due to the assumption of the zero oxygen balance, which imposes a rigid mass of ammonium(V) nitrate and FO according to the wt ratio 94.5:5.5. The absorption index influenced the content of toxic fumes. In the case of the AN-PP7-based ANFO sample, the amount of toxic fumes was lower in relation to the AN-PP8-based sample. Moreover, it was observed that with an elevated total volume of post-blast fumes, the VOD increased. A distinct impact of density on VOD was also found. VOD values increased with density. From the preliminary studies, it may be concluded that the size of grains might also influence the VOD. However, further investigation on the relationship between prill grain size and VOD is required.
Low-velocity detonation has a major significance in the case of ANFO application. Despite the volume of the post-blast gases, low-velocity explosives will not be able to quarry hard resistance rock mass. This observation can be explained by the insufficient amount of energy derived from the detonation process.