Abstract
Steam explosion is a well-known process to pretreat lignocellulosic biomass in order to enhance sugar yields in enzymatic hydrolysis, but pretreatment conditions have to be optimized individually for each material. In this study, we investigated how the results of a pretreatment optimization procedure are influenced by the chosen reaction conditions in the enzymatic hydrolysis. Beechwood was pretreated by steam explosion and the resulting biomass was subjected to enzymatic hydrolysis at glucan loadings of 1% and 5% employing either washed solids or the whole pretreatment slurry. For enzymatic hydrolysis in both reaction modes at a glucan loading of 1%, the glucose yields markedly increased with increasing severity and with increasing pretreatment temperature at identical severities and maximal values were reached at a pretreatment temperature of 230 °C. However, the optimal severity was 5.0 for washed solids enzymatic hydrolysis, but only 4.75 for whole slurry enzymatic hydrolysis. When the glucan loading was increased to 5%, glucose yields hardly increased for pretreatment temperatures between 210 and 230 °C at a given severity, and a pretreatment temperature of 220 °C was sufficient under these conditions. Consequently, it is important to precisely choose the desired conditions of the enzymatic hydrolysis reaction, when aiming to optimize the pretreatment conditions for a certain biomass.
1. Introduction
Lignocellulosic biomass (e.g., wood, agricultural residues or wastepaper products) plays a major role as a sustainable feedstock for biofuel and biochemical production due to its great abundance and relatively low cost [1,2,3]. Especially in central and western Europe, beechwood (Fagus sylvatica) is a promising but underutilized feedstock which is reflected by an ongoing inventory increase in the past decades e.g., by 18.4% since 1983 in Switzerland [4].
Generally, cellulose, hemicelluloses and lignin are the main components of structural plant material, whereas the latter provides natural protection towards degradation of (hemi-)celluloses and thereby contributes to the recalcitrance towards biochemical conversion [5,6]. To overcome this recalcitrance and to enhance sugar release by enzymatic hydrolysis, a range of physicochemical pretreatment processes can be applied [7,8,9]. The pretreatment improves enzyme access to the cellulose by several mechanisms, including an increase of the surface area, solubilization of lignin and/or hemicelluloses and a decrease of the cellulose crystallinity and degree of polymerization [10,11]. In this work, a steam explosion process was used to pretreat beechwood. Here, the biomass is heated to the target temperature by injection of saturated steam, followed by an explosive pressure release after the desired incubation time. This leads to a disruption of the matrix of the plant material and a reduction of the particle size in an energy efficient way [12,13]. As this method does not require any supplementary chemicals, operating costs and corrosive effects on the equipment are minimized. Steam explosion pretreatment has been shown to yield highly reactive substrates across various feedstocks such as hardwoods [14,15,16,17] or herbaceous biomass [18,19,20] and belongs to the most economical methods [21,22,23]. However, there are only few reports on its application to beechwood [24,25]. Generally, the applied pretreatment conditions (temperature, time and particle size) have to be optimized individually for each feedstock in order to maximize the total sugar yields after pretreatment and enzymatic hydrolysis [26,27,28]. To facilitate the comparison of different pretreatment conditions, the pretreatment severity log R0 can be calculated as a function of the variables temperature T [°C] and time t [min], as introduced by Overend et al. [29]:
The influence of the size of the particles entering the pretreatment process is not reflected in Equation (1), but it has also been shown to influence the effectiveness of the pretreatment [13]. However, no clear general trends could be observed, and the influence of the particle size depends on the type of biomass and the pretreatment method. For steam exploded poplar (a hardwood), no difference in enzymatic digestibility was found for two different particle size fractions (2–5 and 12–15 mm) [30], while, for steam exploded pine (a softwood) and corn stover, higher enzymatic glucose yields from the solid fraction were found for larger particles [31,32]. A typical pretreatment optimization methodology is described in the publications of the Biomass Refining Consortium for Applied Fundamentals and Innovation (CAFI), which compared different pretreatment strategies for corn stover [33], poplar [34] and switchgrass [9,35]. Enzymatic hydrolysis reactions were performed with washed pretreated biomass solids at a low glucan loading of 1% and a medium cellulase dosage of 15 filter paper units (FPU) per gram of glucan. Although those conditions are appropriate to determine the reactivity of the pretreated solids, they hardly match the conditions on industrial biorefinery scale. For instance, a minimal titer of 4 wt% ethanol is required for economic product recovery by distillation [36], which translates to a biomass concentration of approximately 12–15% in the enzymatic hydrolysis step. Furthermore, it is desirable to process the whole pretreated biomass slurry containing different inhibitors without any washing and detoxification steps to reduce the number of necessary unit operations [37,38].
To the best of our knowledge, it has not been investigated how the results of a pretreatment optimization aiming to find the conditions that allow for maximal glucose and xylose release are altered, if enzymatic hydrolysis is performed at a higher solid loading or if whole slurry biomass is used instead of washed biomass solids. For that reason, this work examined steam explosion pretreatment of beechwood applying a broad range of pretreatment severities at temperatures from 160–230 °C. Besides determining the composition of the resulting solid and liquid process streams to obtain a mass balance for xylan, we subjected the pretreated biomass to enzymatic hydrolysis at a glucan loading of 1% employing either washed solids or the whole pretreatment slurry and measured the resulting xylose, glucose and total sugar yields. Finally, enzymatic hydrolysis was performed also at a glucan loading of 5% with beechwood pretreated at 210–230 °C.
2. Materials and Methods
2.1. Biomass Feedstock
In this study, beechwood (Fagus sylvatica) harvested in winter 2015/2016 from a forest in Messen, canton Solothurn, Switzerland, was used as feedstock. It was chopped to the particle size of G30, air-dried to a final dry matter of 94% and milled (Retsch, SM 100, Haan, Germany) through a screen to a particle size of <1.5 mm. The untreated beechwood contained 40.8 ± 1.2% glucan, 19.1 ± 0.6% xylan, 25.3 ± 0.9% acid insoluble lignin, 7.3 ± 0.2% acetyl, 0.5% ash and 0.9% extractives. Here, the difference to full mass closure can be explained by not accounting for acid soluble lignin and for other carbohydrates present in minor amounts.
2.2. Steam Explosion Pretreatment
All steam explosion pretreatment experiments were performed with a custom-built steam gun system (Industrieanlagen Planungsgesellschaft m.b.H., Graz, Austria), as previously described [39]. For steam pretreatment optimization, each run was performed with 250 g of air-dried beechwood. After a defined incubation time (count-down started at 99% of the target temperature typically reached in less than 1 min), the biomass was explosively discharged into a stainless-steel receiving vessel equipped with a drum liner (55 gallon polyethylene drum liner, McMaster-Carr, Elmhurst, IL, USA) to collect the pretreated biomass slurry. After weighing the drum liner with the pretreated biomass slurry to determine the total weight of the pretreated beechwood, the solids and the liquid prehydrolyzate were separated by vacuum filtration through Whatman No. 1 filter paper. The solid filter cake was washed thoroughly with room temperature water until the filtrate was colorless, and the mass and the dry matter content of the washed solids were measured. The mass of the prehydrolyzate was calculated by subtracting the dry mass of the washed solids from the total weight of the pretreated biomass slurry.
The applied pretreatment conditions are summarized in Table 1.

Table 1.
Summary of the tested pretreatment conditions. Shown are the pretreatment durations in [min] to reach the targeted severities log R0 (see Equation (1)) at a given temperature.
Based on literature results for other hardwoods, a pretreatment temperature range of 160–230 °C was selected [40,41,42,43]. Furthermore, we chose the tested severities for each temperature as such that the pretreatment duration is between 2 and 180 min. Preliminary experiments comparing the enzymatic glucose yields after pretreatment at two conditions (log R0 = 5.0, T = 210 and 230 °C) of milled (<1.5 mm) and chopped (G30) beechwood did not show any statistically relevant differences (data not shown), thus particle size was not included as an optimization parameter in this study.
Pretreatments were performed as single experiments, but one condition (log R0 = 5.0, 230 °C) was carried out in triplicates in order to check the reproducibility of the results. For this condition, the mean values together with the standard deviation of the mean are reported.
2.3. Xylan Mass Balance
For the xylan mass balance, the xylose concentration in the prehydrolyzate was analyzed after posthydrolysis and the xylan content in the washed pretreated solids was determined (see Section 2.5). With the measured volume of the prehydrolyzate and the mass of the washed solids after pretreatment, absolute values were calculated to determine the recovery of xylan after the pretreatment.
2.4. Enzymatic Hydrolysis
2.4.1. Enzymatic Hydrolysis of Washed Solids
Washed solids obtained after steam explosion pretreatment were enzymatically hydrolyzed in 50 mL screw-top flasks at 50 °C in an incubation shaker (Infors HT, MultitronPro, Bottmingen, Switzerland) at 150 rpm. Commercial Accellerase 1500 (DuPont, St Joseph, MO, USA) with an activity of 58.7 FPU/mL (measured according to Ghose [44]) was used with a cellulase loading of 15 FPU per g of glucan in the raw biomass. A 50 mmol/L citric acid buffer maintained a pH of 5.0 and 0.01 g/L sodium azide prevented microbial growth in the hydrolysis mixtures. The glucan loading was set to 1% or 5%, respectively, based on the composition of the raw beechwood to consider any carbohydrate loss that might take place during pretreatment. To determine the necessary amount of washed pretreated beechwood for enzymatic hydrolysis, a conversion factor relating the mass of the raw beechwood to the mass of washed pretreated solids was utilized. Samples were taken after 7 days, centrifuged and the supernatant was stored at −20 °C until analysis for glucose, xylose and cellobiose.
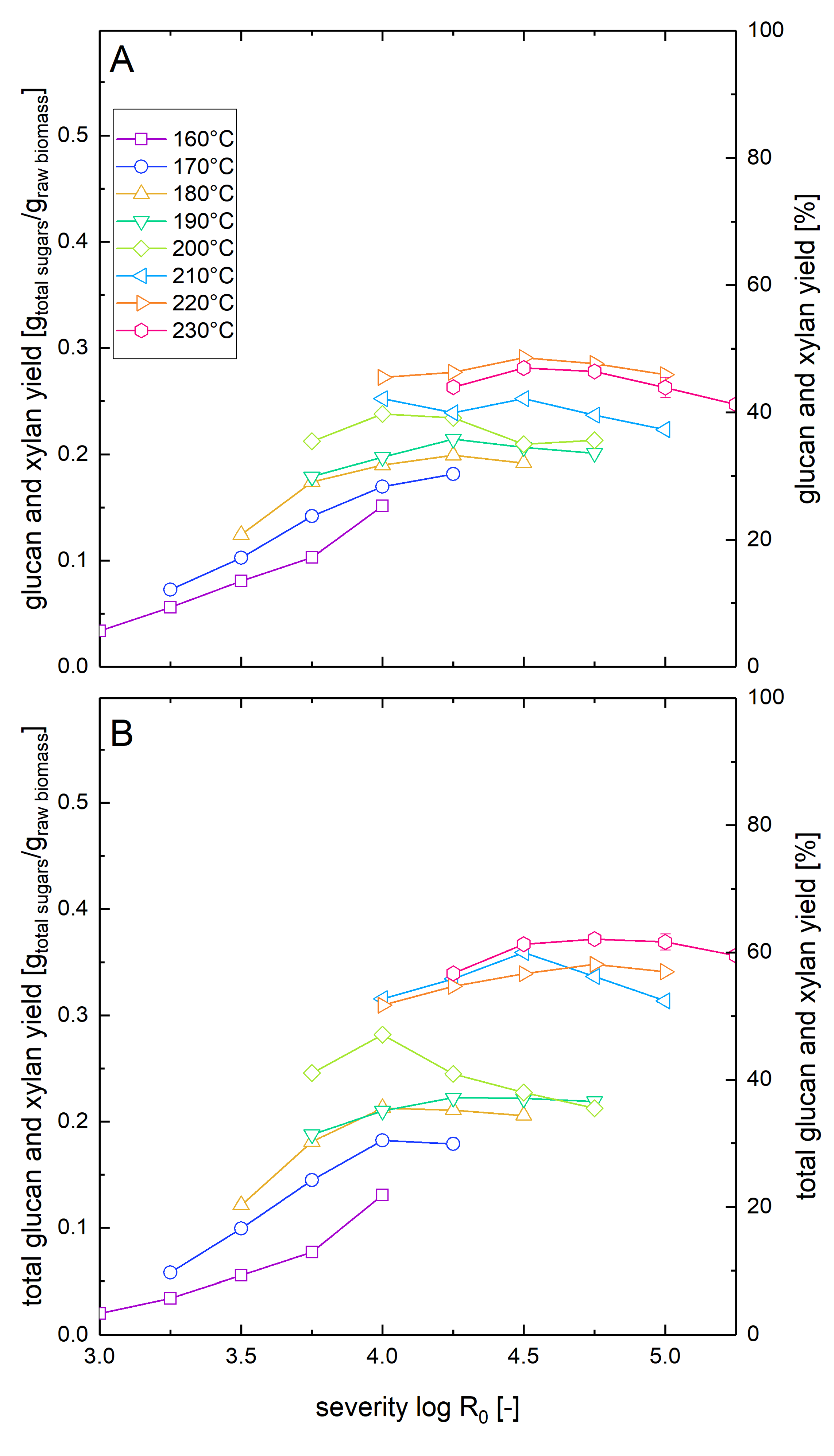
The sugar yields after pretreatment and enzymatic hydrolysis in this series of experiments were calculated by adding up the amount sugars released into the prehydrolyzate (analyzed by posthydrolysis) and the amount of sugars solubilized by enzymatic hydrolysis. Yields are reported as xylan and glucan recoveries which are obtained by converting glucose and xylose concentrations to the corresponding glucan and xylan amounts using the stochiometric conversion factors of 0.9 and 0.88, respectively. The results of this experimental series are displayed in Figures 2B, 3B, 4B and 5.
2.4.2. Whole Slurry Enzymatic Hydrolysis
Whole slurry enzymatic hydrolysis was performed as described in Section 2.4.1, but also prehydrolyzate was added to the reaction mixture. To determine the volume of prehydrolyzate to be added, an additional conversion factor relating the mass of the raw beechwood to the volume of the prehydrolyzate was utilized.
The sugar yields after pretreatment and enzymatic hydrolysis in this series of experiments were calculated based on the amount of glucose and xylose measured in the supernatant of enzymatic hydrolysis reaction and are reported as xylan and glucan recoveries. Results of this experimental series are displayed in Figures 2A, 3A, 4A and 5.
All enzymatic hydrolysis experiments were performed in triplicates and in Section 3, the mean values are shown. The average standard deviation of the mean was 1.7% and the maximal standard deviation was 6.0%. For a better readability, the error bars are not included in the figures.
2.5. Analytical Methods
Raw and pretreated biomass was analyzed for its glucan, xylan and acid insoluble lignin content, and soluble oligomeric sugars in the prehydrolyzate were quantified by posthydrolysis with 4% H2SO4 as reported by the National Renewable Energy Laboratory (Golden, CO, USA). All compositional analysis runs were performed in triplicates and reported are the mean values. For xylan the mean standard deviation over all samples was 0.25% and the maximal standard deviation was 2.2%. For a better readability, the corresponding error bars are not included in the figures.
Glucose, xylose and cellobiose in the liquid fractions were quantified by high performance liquid chromatography (Waters 2695 Separation Module, Waters Corporation, Milford, CT, USA) with refractive index detection (Waters 410) using an Aminex HPX-87H column (Bio-Rad, Hercules, LA, USA) at 60 °C and 5 mmol/L H2SO4 as a mobile phase flowing with 0.6 mL/min.
3. Results and Discussion
3.1. Xylan Mass Balance after Pretreatment
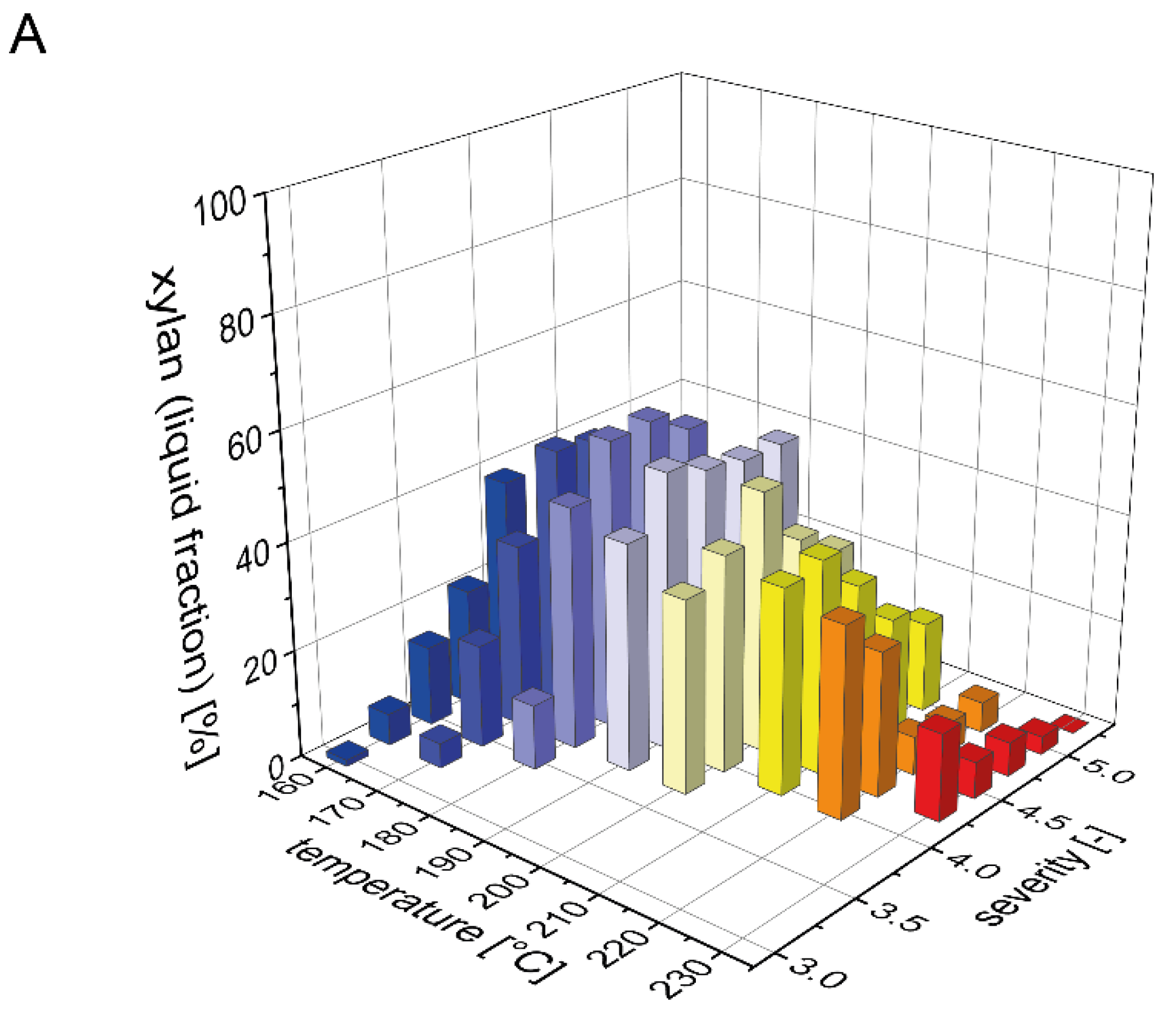
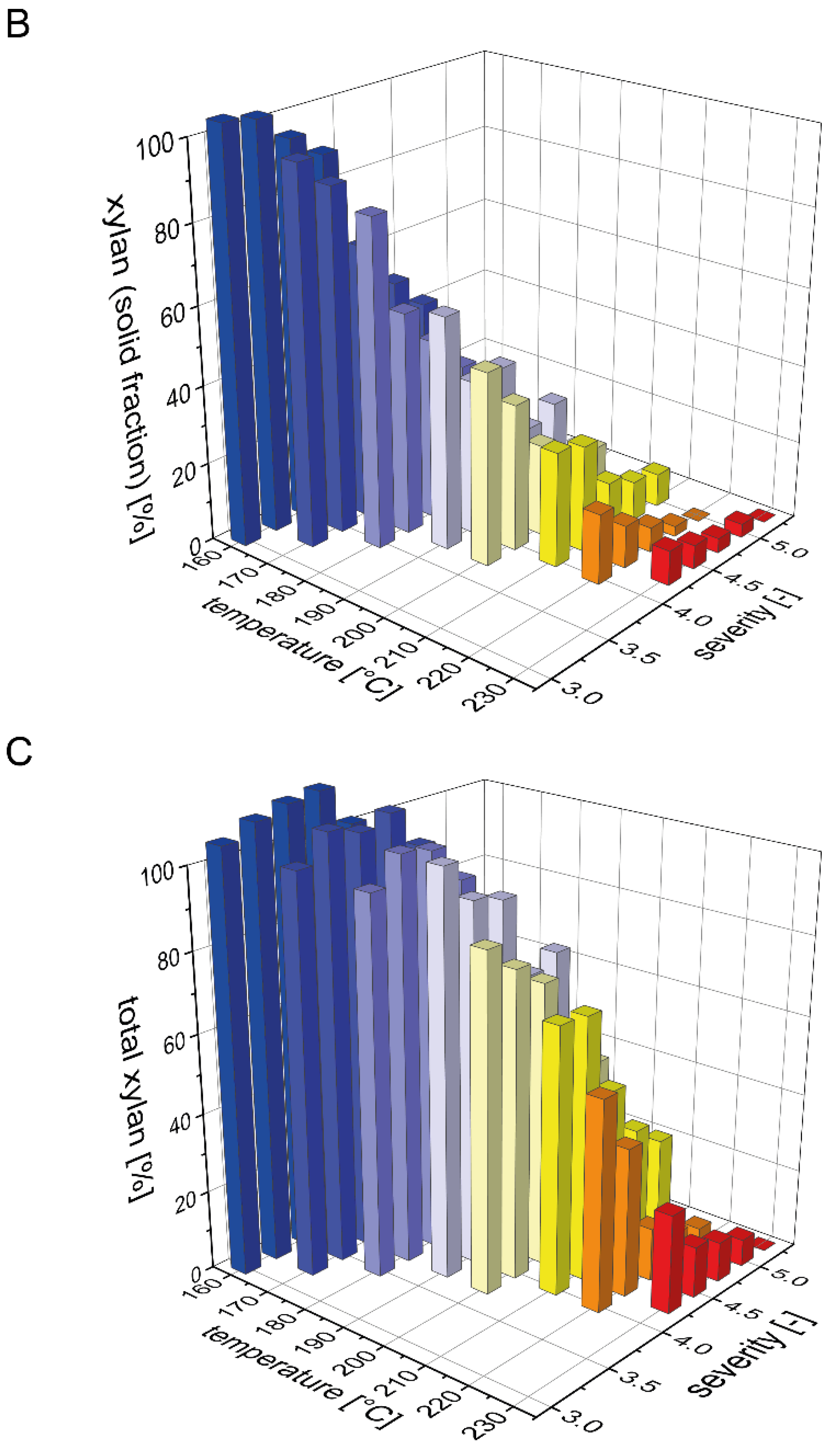
The beechwood used in this study contained 19% xylan, which is the second largest carbohydrate fraction after glucan. It is well known that xylan is more easily solubilized than glucan during acid and water only based pretreatments, but that it is also prone to decomposition to furfural [36]. To determine a mass balance for xylan (calculated in the following as the sum of xylan, xylooligomers and xylose) after steam pretreatment, we analyzed the distribution of xylan between the pretreated solids and the prehydrolyzate (Figure 1). The xylan content of the solid fraction after pretreatment strongly decreased with rising temperature or severity of the pretreatment process (Figure 1B). For the highest pretreatment severities at 220 and 230 °C, xylan was completely solubilized as no residual xylan was detected in the solid fraction. In contrast, during the pretreatments with low severities at temperatures of 160 and 170 °C, the xylan content in the pretreated biomass was identical to the raw biomass. Correspondingly, the xylan recovery in the liquid fraction was very low at severities below 3.75 and then steeply increased with increasing severity until reaching a maximum value of 54% at a severity of 4.25 and a pretreatment temperature of 180 °C (Figure 1A). At pretreatment severities above 4.5, the xylan yield in the liquid fraction decreased due to the partial degradation of xylose to furfural. At 230 °C and a severity of 5.25, xylan was completely degraded. As detailed in the companion publication, most of the solubilized xylan in the liquid fraction was present as xylooligomers and not as monomeric xylose. Overall, the total amount of xylan that could be recovered after pretreatment steadily decreased with increasing severity, but also with increasing temperature at a constant severity (Figure 1C). At the condition where the maximal amount of xylan was found in the liquid phase, the total xylan recovery amounted to 91%.
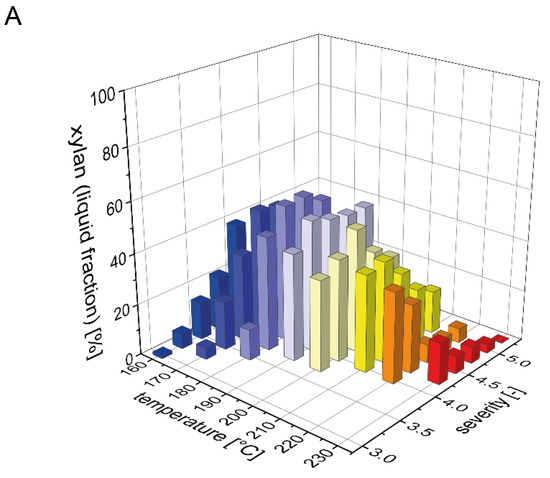
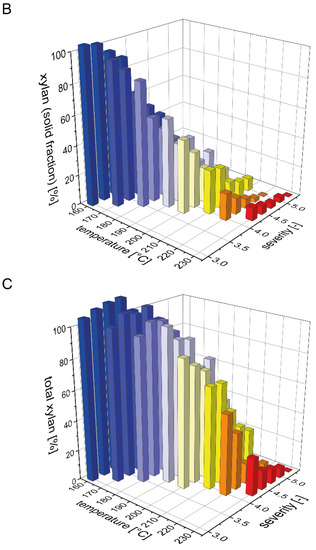
Figure 1.
Mass balance for xylan after pretreatment. The recovery of xylan (A) in the prehydrolyzate in the form of xylose and soluble xylooligomers and (B) in the solids after steam explosion pretreatment. (C) The resulting total xylan recovery based on the composition of the raw material is shown as a function of pretreatment temperature and severity.
3.2. Xylan Recovery after Steam Explosion Pretreatment and Enzymatic Hydrolysis at Low Solid Loading
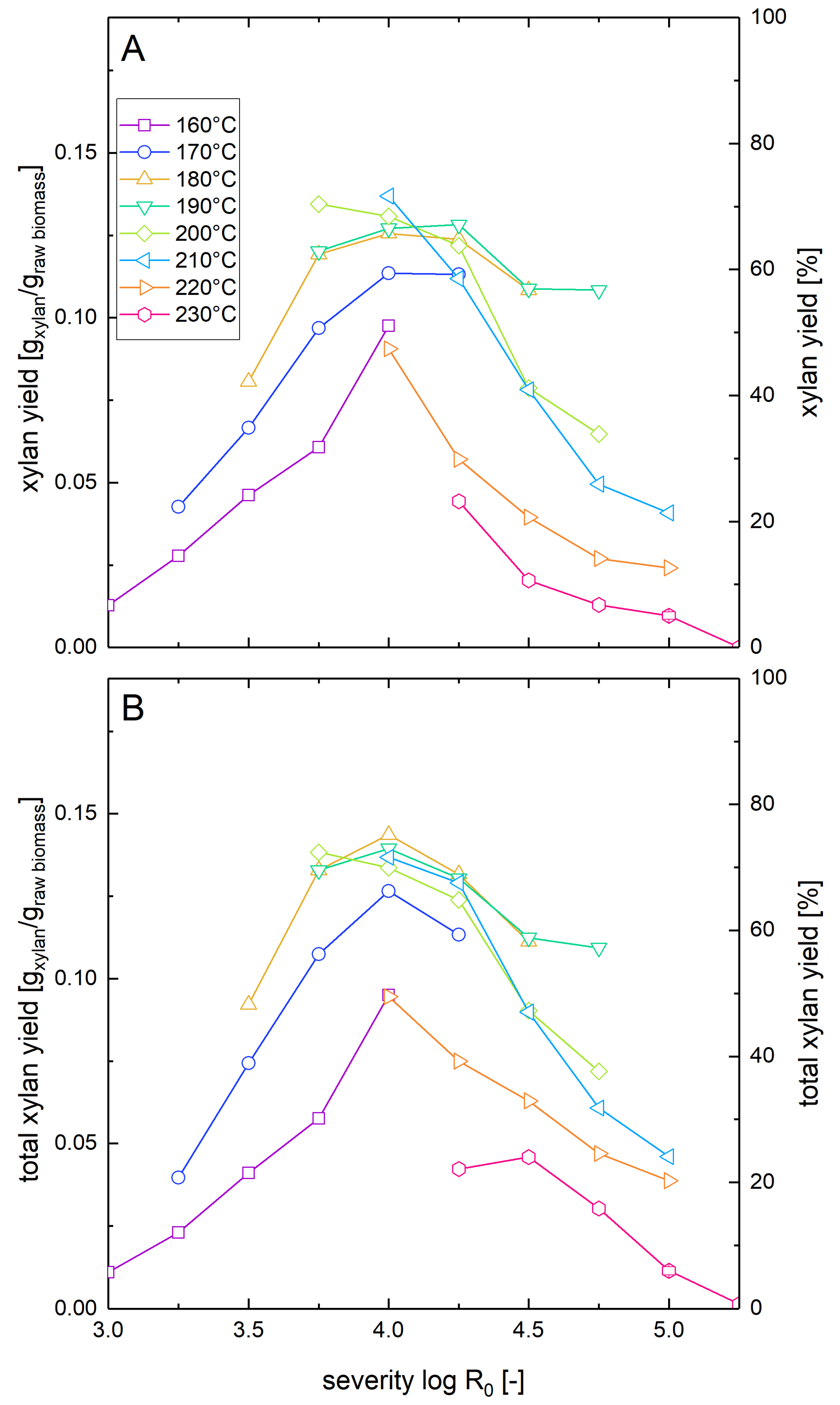
The xylan mass balance presented in Section 3.1 represents the maximal possible amount of xylan that can be recovered in the liquid phase after combined pretreatment and enzymatic hydrolysis, but it does not show how much of the xylan is actually available in the form of solubilized sugars for fermentation after pretreatment and enzymatic hydrolysis. Thus, the washed pretreated solids as well as the whole biomass slurry were subjected to enzymatic hydrolysis at a glucan loading of 1%, and xylose concentrations after seven days were measured. For the whole slurry experiments, this number corresponds to the total xylose yield after pretreatment and enzymatic hydrolysis (Figure 2A). These results were compared with the sum of the xylose release in enzymatic hydrolysis of washed solids and the total amount of xylose and xylooligomers determined after posthydrolysis of the liquid fraction (Figure 2B). It was found that there were only minor deviations of the solubilized xylan yields between the two kinds of experiments. This shows that the xylooligomers in the prehydrolyzate were converted almost quantitatively to xylose monomers by the enzymes in the whole slurry experiments. The maximum values of xylan solubilization and recoveries were 72% for whole slurry enzymatic hydrolysis at a severity of 4.0 at 210 °C and 75% for combined posthydrolysis and washed solid enzymatic hydrolysis at a severity of 4.0 at 180 °C.
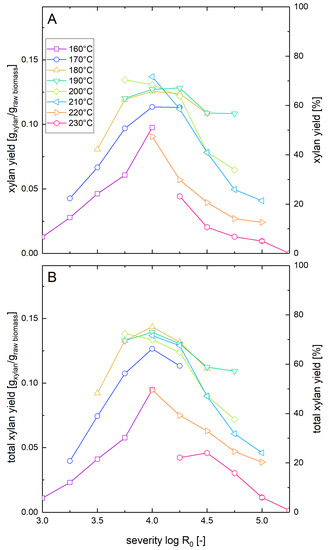
Figure 2.
Solubilization of xylan after pretreatment and low solid enzymatic hydrolysis. The solubilization and recovery of xylan as xylose or soluble xylooligomers after pretreatment and enzymatic hydrolysis was determined (A) after whole slurry enzymatic hydrolysis or (B) after combined enzymatic hydrolysis of washed solids and separate acid hydrolysis of the prehydrolyzate as a function of the pretreatment severity. A glucan loading of 1% based on the unpretreated biomass was applied in enzymatic hydrolysis experiments.
3.3. Glucose Yields after Pretreatment and Enzymatic Hydrolysis at Low Solid Loading
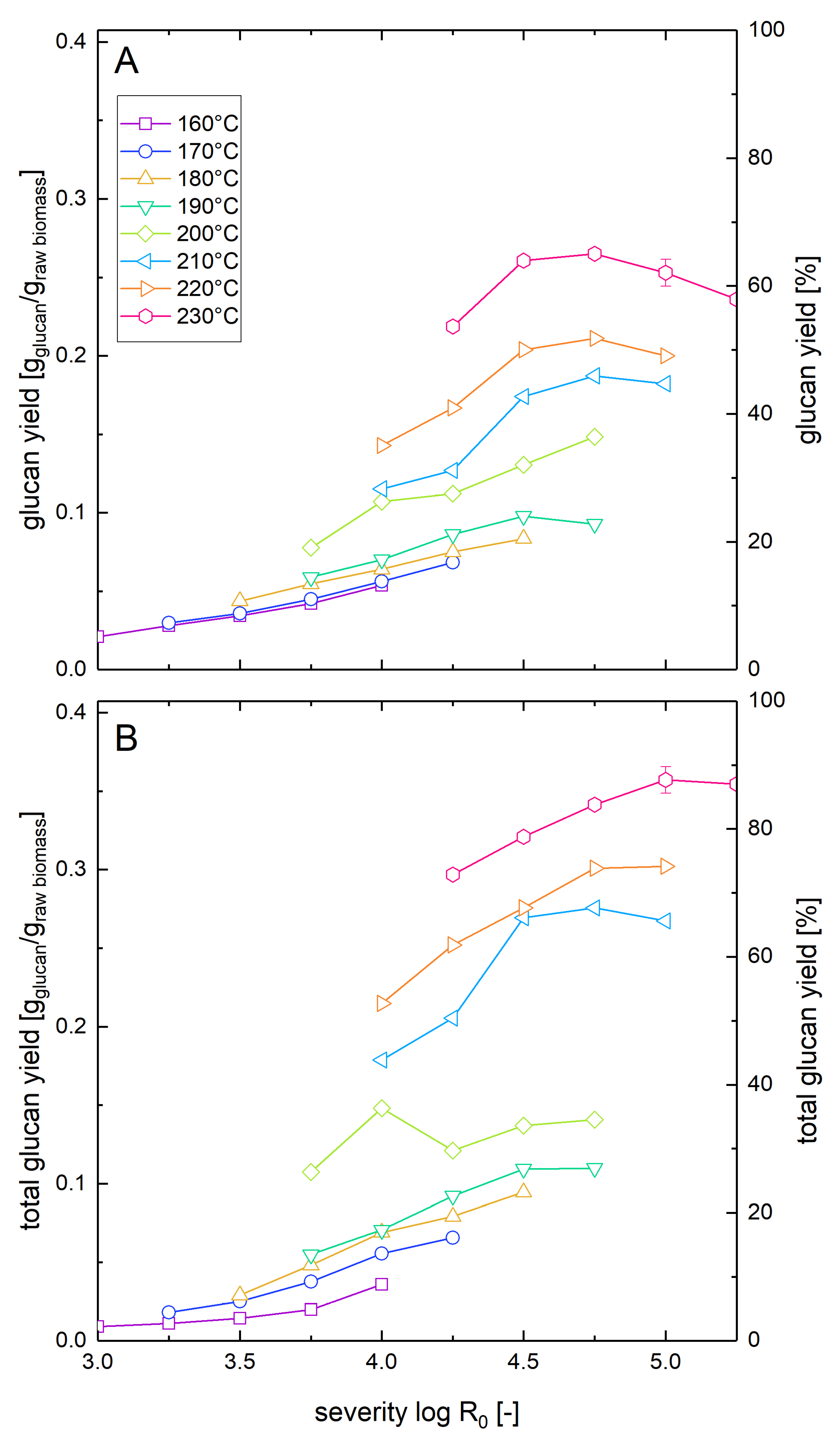
Similar to the measurement of the xylose yields after pretreatment and enzymatic hydrolysis described in Section 3.2, we also determined the corresponding glucose yields (Figure 3). At low pretreatment severities and temperatures, the glucose yields were similar for both whole slurry and washed solids enzymatic hydrolysis. However, with increasing pretreatment severity, the yield difference between the two types of experiments increased. If enzymatic hydrolysis was performed with washed solids, the maximal glucose yield was 88% and was reached at a temperature of 230 °C and a severity of 5.0. For comparison, Nitsos et al. [41] observed a maximal glucose yield of 58% in enzymatic hydrolysis of washed hot water pretreated beechwood at a pretreatment temperature of 190 °C for 15 min (log R0 = 3.81). When in the same study, the pretreatment temperature was raised to 220 °C while keeping the duration constant (log R0 = 4.69), the glucose yields dropped to 31% [37]. The differences to the steam explosion pretreatment method reported in this manuscript are the slower heating rate of 7 °C/min (versus approximately 230 °C/min) and the absence of the explosive decompression. Explosive decompression has been shown to reduce the particle size and alter the microstructure of the biomass which leads to enhanced glucose yields in enzymatic hydrolysis [39,45].
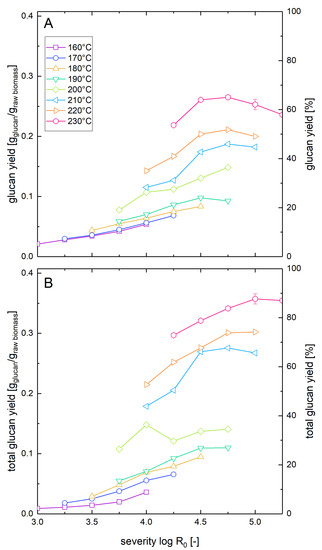
Figure 3.
Solubilization and recovery of glucan after pretreatment and low solid enzymatic hydrolysis. The solubilization and recovery of glucan as glucose or soluble glucose oligomers after pretreatment and enzymatic hydrolysis was determined (A) after whole slurry enzymatic hydrolysis or (B) after combined enzymatic hydrolysis of washed solids and separate acid hydrolysis of the prehydrolyzate as a function of the pretreatment severity. A glucan loading of 1% based on the unpretreated biomass was applied in enzymatic hydrolysis experiments.
In the whole slurry experiments, the highest glucose yield was 65%, corresponding to a relative reduction of 26% compared to the washed solids enzymatic hydrolysis (Table 2). This maximal value was reached at a slightly lower severity of 4.75 but at an identical pretreatment temperature of 230 °C. The differences between both experimental types show the influence of the inhibitory compounds derived from the pretreatment process in the whole slurry liquor that is investigated in more detail in the companion paper. The negative influence of the inhibitors was also shown by the decline of the yield at higher severities than 4.75 at 230 °C occurring only in the whole slurry enzymatic hydrolysis experiments. The yield reduction in whole slurry enzymatic hydrolysis of pretreated beech was more severe than the predicted yield reduction of about 5% during enzymatic hydrolysis of Avicel in the presence of prehydrolyzates at the same concentration level (see Part 2 of this paper series). The substrate might also influence the amount of inhibition, e.g., by lowering the amount of available enzymes by irreversible adsorption onto lignin or by the requirement of different types of enzymes that are inhibited to different extents.

Table 2.
Summary of the maximal sugar yields after steam explosion pretreatment and enzymatic hydrolysis. Shown are the maximal sugar yields after steam explosion pretreatment and enzymatic hydrolysis of washed solids or whole biomass slurry at two different glucan loadings.
Both experimental series clearly showed that the pretreatment temperature has a higher influence on the digestibility of the substrate than predicted by the severity factor which combines the pretreatment duration and temperature in one number (Equation (1)) as the glucose yields at a constant severity increased with increasing pretreatment temperature. This observation was also made by others and, e.g., Kim et al. therefore altered the calculation of the severity factor by lowering the empirical factor of 14.75 to 4.6 to be able to correlate the glucose yield in enzymatic hydrolysis with the pretreatment severity [46].
3.4. Total Sugar Yields after Steam Explosion Pretreatment and Enzymatic Hydrolysis at Low Solid Loading
Based on the data presented above, we calculated the total sugar yields that could be reached after steam explosion pretreatment and enzymatic hydrolysis (Figure 4). Due to the different optimal conditions for maximum glucan and xylan recovery, the maximum total sugar yields were lower than the maximum yields achieved for one type of sugar (see Table 2). At high severities, glucan recovery is maximal, but xylan recovery is very small due to thermal degradation. At medium severities, high amounts of xylan can be recovered as soluble molecules, but the enzymatic digestibility of the glucan fraction is modest. The maximum total sugar yield for combined washed solids enzymatic hydrolysis and acid hydrolysis of the prehydrolyzate amounted to 62% at a severity of 4.75 at 230 °C. For whole slurry enzymatic hydrolysis, the maximum total sugar yield is 49% reached at a severity of 4.5 at a temperature of 220 °C.
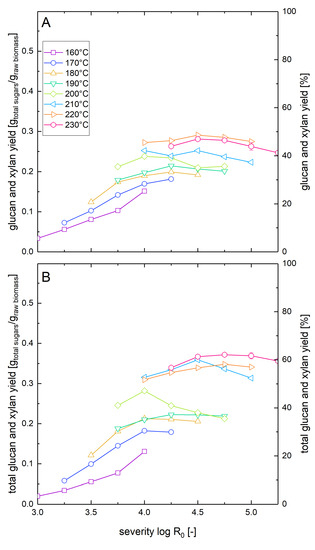
Figure 4.
Total sugar yields after pretreatment and low solid enzymatic hydrolysis. The solubilization and recovery of glucan and xylan as soluble molecules after pretreatment and enzymatic hydrolysis was determined (A) after whole slurry enzymatic hydrolysis or (B) after combined enzymatic hydrolysis of washed solids and separate acid hydrolysis of the prehydrolyzate as a function of the pretreatment severity. A glucan loading of 1% based on the unpretreated biomass was applied in enzymatic hydrolysis experiments.
3.5. Optimization of Steam Explosion Pretreatment Conditions for High Solids Enzymatic Hydrolysis
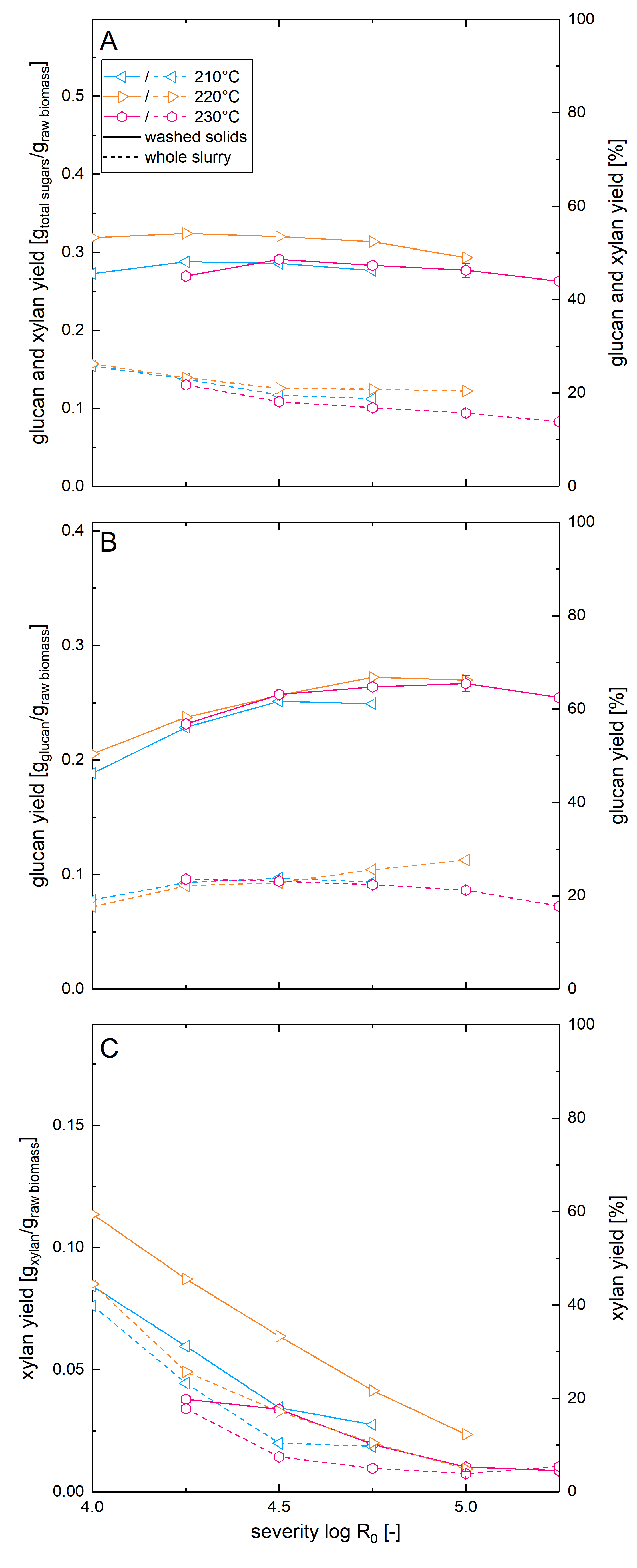
To examine the influence of the solid loading in enzymatic hydrolysis on the results of the optimization of the steam explosion pretreatment conditions, the materials pretreated at temperatures of 210–230 °C were enzymatically hydrolyzed also at a glucan loading of 5% (Figure 5). Analogous to the above described experiments, the whole pretreatment slurry as well as the washed solids were used.
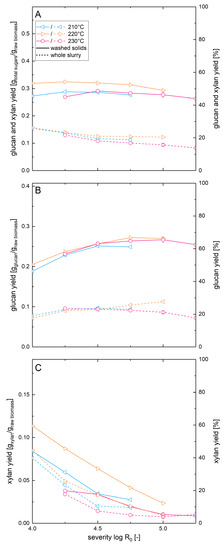
Figure 5.
Solubilization and recovery of glucan and xylan after pretreatment and high solid enzymatic hydrolysis. Shown are the recoveries of (A) total sugars, (B) glucan and (C) xylan as soluble molecules after steam explosion pretreatment and whole slurry enzymatic hydrolysis (dashed line) or combined enzymatic hydrolysis of washed solids and acid hydrolysis of the prehydrolyzate (solid line). A glucan loading of 5% was applied.
In this experimental series, the highest total xylan recovery of 59% was found when posthydrolysis of the liquid fraction was combined with enzymatic hydrolysis of washed solids pretreated at 220 °C at a severity of 4.0. In comparison to this, the xylose yield of the whole slurry experiment for the same condition was 44%, corresponding to a yield reduction of 25%. Analogous to the pretreatment optimization for low solids enzymatic hydrolysis, the xylan recovery was decreasing with increasing pretreatment severities. The glucose yields in high solids enzymatic hydrolysis of washed pretreated biomass showed much less variation over the range of severities tested than observed in the low solids enzymatic hydrolysis. At high solids concentration, the glucose yields varied between 47% and 67%, while at low solids enzymatic hydrolysis values of 43–88% were observed at similar conditions. In contrast to the low solids experiments, almost identical glucose yields could be achieved when hydrolyzing high concentrations of beechwood pretreated at a given severity independently of the pretreatment temperature. The highest glucose yield of 67% was achieved at a severity of 4.75 and a pretreatment temperature of 220 °C. It was not surprising that the glucose yield in enzymatic hydrolysis dropped at higher solids concentration, as this effect is described in the literature and is termed the solids effect [47,48]. The suggested causes for this phenomenon are lower cellulase adsorption due to increased sugar concentrations [49], product inhibition, inadequate mixing and availability of free water. However, to the best of our knowledge, there are only few publications, where the solids effect is quantified and compared for differently pretreated materials. Martins et al. [50] reported a higher reduction of glucose yield upon increasing the solids concentration for sugar cane bagasse pretreated with hypochlorite compared to material pretreated with dilute sulfuric acid or alkaline hydrogen peroxide. Cara et al. demonstrated that delignification of steam pretreated olive tree biomass was able to increase glucose yields in enzymatic hydrolysis at solid concentrations between 2% and 10% but not at solid concentrations of 20–30% [51]. This confirms our results that the solids effect can mask the enhanced reactivity of biomass pretreated at more optimal conditions observed at low solids enzymatic hydrolysis.
When using whole pretreatment slurry at high solids concentration, the glucose yield dropped considerably compared to the washed solids experiment and values between only 18% and 28% were found. The yield reduction due to the presence of the different inhibitors was, at 58% (when comparing the individual yields at optimal conditions), much higher in the high solids concentration experiments compared to the low solids whole slurry enzymatic hydrolysis. Thus, the strength of the yield reducing effect of the pretreatment inhibitors depends on their concentration, which is discussed in more detail in the companion paper.
Finally, the total sugar yields we calculated for the high solids concentration experiments are in the range of 44–54% when using washed solids for enzymatic hydrolysis and are in the range of 13–26% when using the whole pretreatment slurry experiment. When using washed solids, the highest total sugar yield was reached at a severity of 4.25 and a pretreatment temperature of 220 °C. For whole slurry enzymatic hydrolysis, the highest total sugar yield was reached with a pretreatment severity of 4.0 at the same pretreatment temperature.
4. Summary and Conclusions
We showed in this study that the results of a pretreatment optimization for beechwood depend on the chosen enzymatic hydrolysis conditions. For enzymatic hydrolysis at a glucan loading of 1%, the glucose yields increased with increasing pretreatment temperature, even at identical severities, and maximal values were reached at a pretreatment temperature of 230 °C. When whole slurry pretreated beechwood instead of the washed pretreated solids was enzymatically hydrolyzed at low solid loading, the optimal severity for maximal glucose yield shifted from 5.0 to 4.75 and glucose yields were generally lower. At elevated solid loading in the enzymatic hydrolysis reaction, the observed glucose yields were virtually identical for the tested pretreatment temperatures (210–230 °C) at a given severity. Thus, the extent of the solids effect varies for different pretreatment conditions and the enhanced reactivity of beechwood pretreated at 230 °C observed at low solids enzymatic hydrolysis was presumably masked by product inhibition at high solid loadings. Further work will explore whether this is the case also for simultaneous saccharification and fermentation.
Author Contributions
Conceptualization, S.B. and M.S.; methodology, A.A., R.B., S.B. and T.Z.; formal analysis, A.A., R.B., S.B. and T.Z.; investigation, A.A., R.B., S.B., M.H.S. and T.Z.; writing—original draft preparation, R.B. and S.B.; writing—review and editing, A.A., R.B., S.B., T.Z. and M.H.S.; visualization, R.B. and S.B.; supervision, M.H.S.; project administration, M.H.S.; and funding acquisition, A.A., S.B., T.Z. and M.H.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Swiss National Science Foundation in the framework of the National Research Program 66 “Resource wood” (grant number 406640_136712) and by a research exchange project from the Polish National Centre for Research and Development “Program for improvement of didactics of WULS-SGGW in the field of acquiring plant raw materials for energy in the context of the objectives of the Europe 2020 Strategy”, which was financed under the Human Capital Operational Program (UDA-POKL-04.03.00-00-042/12-00).
Acknowledgments
The authors acknowledge the co-financing of the steam gun through Bern University of Applied Sciences, School of Agricultural, Forest and Food Sciences (HAFL) and thank Robert Shahab for his valuable help in generating the figures.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lynd, L.R.; Liang, X.; Biddy, M.J.; Allee, A.; Cai, H.; Foust, T.; Himmel, M.E.; Laser, M.S.; Wang, M.; Wyman, C.E. Cellulosic ethanol: Status and innovation. Curr. Opin. Biotechnol. 2017, 45, 202–211. [Google Scholar] [CrossRef]
- Lynd, L.R.; Laser, M.S.; Bransby, D.; Dale, B.E.; Davison, B.; Hamilton, R.; Himmel, M.; Keller, M.; McMillan, J.D.; Sheehan, J.; et al. How biotech can transform biofuels. Nat. Biotechnol. 2008, 26, 169–172. [Google Scholar] [CrossRef] [PubMed]
- Limayem, A.; Ricke, S.C. Lignocellulosic biomass for bioethanol production: Current perspectives, potential issues and future prospects. Prog. Energy Combust. Sci. 2012, 38, 449–467. [Google Scholar] [CrossRef]
- Cioldi, F.; Keller, M.; Rösler, E.; Speich, S.; Brändli, U.-B.; Vidondo, B.; Abegg, M.; Meile, R.; Huber, M.; Traub, B.; et al. Schweizerisches Landesforstinventar—Ergebnistabelle Nr. 177425; Eidgenössische Forschungsanstalt WSL: Birmensdorf, Switzerland, 2014. [Google Scholar]
- Himmel, M.E.; Ding, S.-Y.; Johnson, D.K.; Adney, W.S.; Nimlos, M.R.; Brady, J.W.; Foust, T.D. Biomass recalcitrance: Engineering plants and enzymes for biofuels production. Science 2007, 315, 804–807. [Google Scholar] [CrossRef]
- Lynd, L.R.; Wyman, C.E.; Gerngross, T.U. Biocommodity engineering. Biotechnol. Prog. 1999, 15, 777–793. [Google Scholar] [CrossRef] [PubMed]
- Mosier, N.; Wyman, C.E.; Dale, B.; Elander, R.; Lee, Y.Y.; Holtzapple, M.; Ladisch, M. Features of promising technologies for pretreatment of lignocellulosic biomass. Bioresour. Technol. 2005, 96, 673–686. [Google Scholar] [CrossRef] [PubMed]
- Wyman, C.E.; Dale, B.E.; Balan, V.; Elander, R.T.; Holtzapple, M.T.; Ramirez, R.S.; Ladisch, M.R.; Mosier, N.S.; Lee, Y.Y.; Gupta, R.; et al. Comparative performance of leading pretreatment technologies for biological conversion of corn stover, poplar wood, and switchgrass to sugars. In Aqueous Pretreatment of Plant Biomass for Biological and Chemical Conversion to Fuels and Chemicals; Wyman, C.E., Ed.; John Wiley & Sons Inc.: Chichester, West Sussex, UK, 2013; pp. 239–259. ISBN 9780470975831. [Google Scholar]
- Wyman, C.E.; Balan, V.; Dale, B.E.; Elander, R.T.; Falls, M.; Hames, B.; Holtzapple, M.T.; Ladisch, M.R.; Lee, Y.Y.; Mosier, N.; et al. Comparative data on effects of leading pretreatments and enzyme loadings and formulations on sugar yields from different switchgrass sources. Bioresour. Technol. 2011, 102, 11052–11062. [Google Scholar] [CrossRef] [PubMed]
- Hendriks, A.T.W.M.; Zeeman, G. Pretreatments to enhance the digestibility of lignocellulosic biomass. Bioresour. Technol. 2009, 100, 10–18. [Google Scholar] [CrossRef]
- Yang, B.; Wyman, C.E. Pretreatment: The key to unlocking low-cost cellulosic ethanol. Biofuels Bioprod. Bioref. 2008, 2, 26–40. [Google Scholar] [CrossRef]
- Holtzapple, M.T.; Humphrey, A.E.; Taylor, J.D. Energy requirements for the size reduction of poplar and aspen wood. Biotechnol. Bioeng. 1989, 33, 207–210. [Google Scholar] [CrossRef]
- Vidal, B.C.; Dien, B.S.; Ting, K.C.; Singh, V. Influence of feedstock particle size on lignocellulose conversion—A review. Appl. Biochem. Biotechnol. 2011, 164, 1405–1421. [Google Scholar] [CrossRef] [PubMed]
- Cara, C.; Ruiz, E.; Ballesteros, M.; Manzanares, P.; Negro, M.J.; Castro, E. Production of fuel ethanol from steam-explosion pretreated olive tree pruning. Fuel 2008, 87, 692–700. [Google Scholar] [CrossRef]
- Romaní, A.; Garrote, G.; Ballesteros, I.; Ballesteros, M. Second generation bioethanol from steam exploded Eucalyptus globulus wood. Fuel 2013, 111, 66–74. [Google Scholar] [CrossRef]
- Park, J.Y.; Kang, M.; Kim, J.S.; Lee, J.P.; Choi, W.I.; Lee, J.S. Enhancement of enzymatic digestibility of Eucalyptus grandis pretreated by NaOH catalyzed steam explosion. Bioresour. Technol. 2012, 123, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Cantarella, M.; Cantarella, L.; Gallifuoco, A.; Spera, A.; Alfani, F. Effect of inhibitors released during steam-explosion treatment of poplar wood on subsequent enzymatic hydrolysis and SSF. Biotechnol. Prog. 2004, 20, 200–206. [Google Scholar] [CrossRef]
- Sipos, B.; Réczey, J.; Somorai, Z.; Kádár, Z.; Dienes, D.; Réczey, K. Sweet sorghum as feedstock for ethanol production: Enzymatic hydrolysis of steam-pretreated bagasse. Appl. Biochem. Biotechnol. 2009, 153, 151–162. [Google Scholar] [CrossRef]
- Petersen, M.Ø.; Larsen, J.; Thomsen, M.H. Optimization of hydrothermal pretreatment of wheat straw for production of bioethanol at low water consumption without addition of chemicals. Biomass Bioenerg. 2009, 33, 834–840. [Google Scholar] [CrossRef]
- Alvira, P.; Negro, M.J.; Ballesteros, I.; González, A.; Ballesteros, M. Steam explosion for wheat straw pretreatment for sugars production. Bioethanol 2016, 2, 66–75. [Google Scholar] [CrossRef]
- Galbe, M.; Wallberg, O. Pretreatment for biorefineries: A review of common methods for efficient utilisation of lignocellulosic materials. Biotechnol. Biofuels 2019, 12, 294. [Google Scholar] [CrossRef]
- Galbe, M.; Zacchi, G. Pretreatment of lignocellulosic materials for efficient bioethanol production. Adv. Biochem. Eng. Biotechnol. 2007, 108, 41–65. [Google Scholar] [CrossRef]
- Chen, H.; Qiu, W. Key technologies for bioethanol production from lignocellulose. Biotechnol. Adv. 2010, 28, 556–562. [Google Scholar] [CrossRef]
- Asada, C.; Sasaki, C.; Hirano, T.; Nakamura, Y. Chemical characteristics and enzymatic saccharification of lignocellulosic biomass treated using high-temperature saturated steam: Comparison of softwood and hardwood. Bioresour. Technol. 2015, 182, 245–250. [Google Scholar] [CrossRef]
- Pazitny, A.; Russ, A.; Bohacek, S.; Stankovska, M.; Ihnat, V.; Suty, S. Various lignocellulosic raw materials pretreatment processes utilizable for increasing holocellulose accessibility for hydrolytic enzymes part II. Effect of steam explosion temperature on beech enzymatic hydrolysis. Wood Res. 2019, 64, 437–447. [Google Scholar]
- Ramos, L.P. The chemistry involved in the steam treatment of lignocellulosic materials. Química Nova 2003, 26, 863–871. [Google Scholar] [CrossRef]
- Yang, B.; Wyman, C.E. Effect of xylan and lignin removal by batch and flowthrough pretreatment on the enzymatic digestibility of corn stover cellulose. Biotechnol. Bioeng. 2004, 86, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, T.A.; Wyman, C.E. Combined sugar yields for dilute sulfuric acid pretreatment of corn stover followed by enzymatic hydrolysis of the remaining solids. Bioresour. Technol. 2005, 96, 1967–1977. [Google Scholar] [CrossRef]
- Overend, R.P.; Chornet, E.; Gascoigne, J.a. Fractionation of lignocellulosics by steam-aqueous pretreatments [and discussion]. Philos. Trans. R. Soc. A 1987, 321, 523–536. [Google Scholar] [CrossRef]
- Negro, M.J.; Manzanares, P.; Ballesteros, I.; Oliva, J.M.; Cabañas, A.; Ballesteros, M. Hydrothermal Pretreatment Conditions to Enhance Ethanol Production from Poplar Biomass. Appl. Biochem. Biotechnol. 2003, 105, 87–100. [Google Scholar] [CrossRef]
- Ballesteros, I.; Oliva, J.M.; Navarro, A.A.; González, A.; Carrasco, J.; Ballesteros, M. Effect of chip size on steam explosion pretreatment of softwood. Appl. Biochem. Biotechnol. 2000, 84–86, 97–110. [Google Scholar] [CrossRef]
- Liu, Z.-H.; Qin, L.; Pang, F.; Jin, M.-J.; Li, B.-Z.; Kang, Y.; Dale, B.E.; Yuan, Y.-J. Effects of biomass particle size on steam explosion pretreatment performance for improving the enzyme digestibility of corn stover. Ind. Crops Prod. 2013, 44, 176–184. [Google Scholar] [CrossRef]
- Wyman, C.E.; Dale, B.E.; Elander, R.T.; Holtzapple, M.; Ladisch, M.R.; Lee, Y.Y. Comparative sugar recovery data from laboratory scale application of leading pretreatment technologies to corn stover. Bioresour. Technol. 2005, 96, 2026–2032. [Google Scholar] [CrossRef]
- Wyman, C.E.; Dale, B.E.; Elander, R.T.; Holtzapple, M.; Ladisch, M.R.; Lee, Y.Y.; Mitchinson, C.; Saddler, J.N. Comparative sugar recovery and fermentation data following pretreatment of poplar wood by leading technologies. Biotechnol. Prog. 2009, 25, 333–339. [Google Scholar] [CrossRef]
- Garlock, R.J.; Balan, V.; Dale, B.E.; Pallapolu, V.R.; Lee, Y.Y.; Kim, Y.; Mosier, N.S.; Ladisch, M.R.; Holtzapple, M.T.; Falls, M.; et al. Comparative material balances around pretreatment technologies for the conversion of switchgrass to soluble sugars. Bioresour. Technol. 2011, 102, 11063–11071. [Google Scholar] [CrossRef] [PubMed]
- Vane, L.M. Separation technologies for the recovery and dehydration of alcohols from fermentation broths. Biofuels Bioprod. Bioref. 2008, 2, 553–588. [Google Scholar] [CrossRef]
- McMillan, J.D.; Jennings, E.W.; Mohagheghi, A.; Zuccarello, M. Comparative performance of precommercial cellulases hydrolyzing pretreated corn stover. Biotechnol. Biofuels 2011, 4, 29. [Google Scholar] [CrossRef] [PubMed]
- Geddes, C.C.; Nieves, I.U.; Ingram, L.O. Advances in ethanol production. Curr. Opin. Biotechnol. 2011, 22, 312–319. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pielhop, T.; Amgarten, J.; von Rohr, P.R.; Studer, M.H. Steam explosion pretreatment of softwood: The effect of the explosive decompression on enzymatic digestibility. Biotechnol. Biofuels 2016, 9, 152. [Google Scholar] [CrossRef] [PubMed]
- Schütt, F.; Puls, J.; Saake, B. Optimization of steam pretreatment conditions for enzymatic hydrolysis of poplar wood. Holzforschung 2011, 65. [Google Scholar] [CrossRef]
- Nitsos, C.K.; Matis, K.A.; Triantafyllidis, K.S. Optimization of hydrothermal pretreatment of lignocellulosic biomass in the bioethanol production process. ChemSusChem 2013, 6, 110–122. [Google Scholar] [CrossRef]
- Sassner, P.; Galbe, M.; Zacchi, G. Steam pretreatment of Salix with and without SO2 impregnation for production of bioethanol. Appl. Biochem. Biotechnol. 2005, 121, 1101–1117. [Google Scholar] [CrossRef]
- Romaní, A.; Garrote, G.; Alonso, J.L.; Parajó, J.C. Bioethanol production from hydrothermally pretreated Eucalyptus globulus wood. Bioresour. Technol. 2010, 101, 8706–8712. [Google Scholar] [CrossRef] [PubMed]
- Ghose, T.K. Measurement of cellulase activities. Pure Appl. Chem. 1987, 59, 257–268. [Google Scholar] [CrossRef]
- Muzamal, M.; Arnling Bååth, J.; Olsson, L.; Rasmuson, A.S. Contribution of structural modification to enhanced enzymatic hydrolysis and 3-D structural analysis of steam-exploded wood using X-ray tomography. BioResources 2016, 11. [Google Scholar] [CrossRef]
- Kim, Y.; Kreke, T.; Mosier, N.S.; Ladisch, M.R. Severity factor coefficients for subcritical liquid hot water pretreatment of hardwood chips. Biotechnol. Bioeng. 2014, 111, 254–263. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, J.B.; Felby, C.; Jorgensen, H. Yield-determining factors in high-solids enzymatic hydrolysis of lignocellulose. Biotechnol. Biofuels 2009, 2, 11. [Google Scholar] [CrossRef] [PubMed]
- Modenbach, A.A.; Nokes, S.E. Enzymatic hydrolysis of biomass at high-solids loadings—A review. Biomass Bioenerg. 2013, 56, 526–544. [Google Scholar] [CrossRef]
- Kumar, R.; Wyman, C.E. An improved method to directly estimate cellulase adsorption on biomass solids. Enzyme Microb. Technol. 2008, 42, 426–433. [Google Scholar] [CrossRef]
- Martins, L.H.d.S.; Rabelo, S.C.; da Costa, A.C. Effects of the pretreatment method on high solids enzymatic hydrolysis and ethanol fermentation of the cellulosic fraction of sugarcane bagasse. Bioresour. Technol. 2015, 191, 312–321. [Google Scholar] [CrossRef]
- Cara, C.; Moya, M.; Ballesteros, I.; Negro, M.J.; González, A.; Ruiz, E. Influence of solid loading on enzymatic hydrolysis of steam exploded or liquid hot water pretreated olive tree biomass. Process. Biochem. 2007, 42, 1003–1009. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).