3.1. Parameter Values Declared by the Manufacturer in Reference to the Parameter Values Measured in the Laboratory
Figure 1,
Figure 2,
Figure 3,
Figure 4 and
Figure 5 present the distributions of values of the tested parameters: kinematic viscosity at 40 °C, 100 °C, −20 °C and 130 °C as well as the viscosity index (VI). With the beginning kinematic viscosity at 40 °C, kinematic viscosity at 100 °C and the VI (
Figure 1,
Figure 2 and
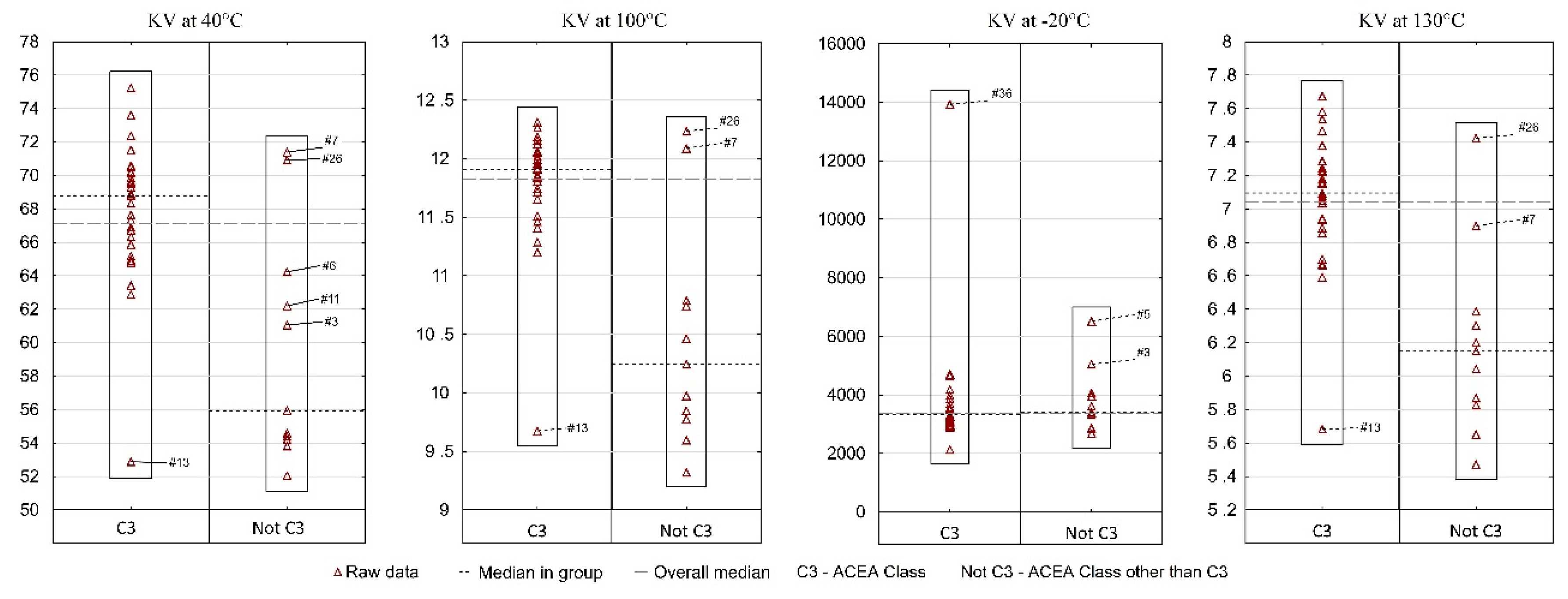
Figure 3) are analyzed as they are among the information provided by the manufactures. For those three parameters the results are compiled in such a way that on one side (on the left) there is information taken from the manufacturers’ labels/product data sheets, while on the other (on the right) there are results obtained from laboratory tests. To facilitate an easy interpretation of the results the data were divided into the clusters of oils which were the most similar to each other in terms of the analyzed parameter. The sample numbers (sample ID,
Table 2) for which the manufacturer’s information on the viscosity level was not found were also included in the chart containing the manufacturers’ declared parameter values. Samples circled in red showed a difference of more than 5%, while samples marked with a black dotted circle at a level of 3–5%.
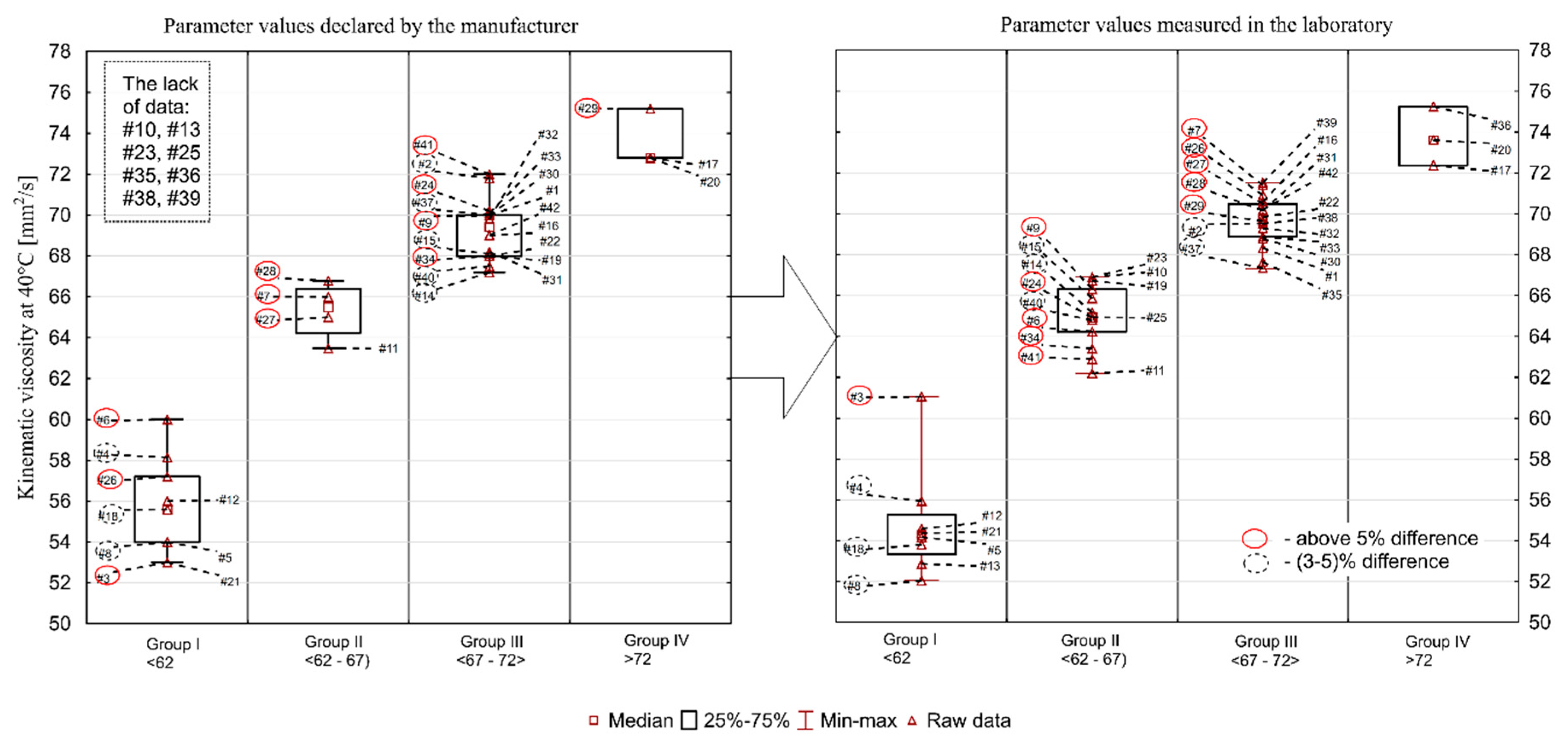
Kinematic viscosity at 40 °C (declared by manufacturer/measured in the laboratory) was in the range of 53.0–75.2/52.0–75.3 mm
2/s. The average value was 65.6/65.7 mm
2/s, while the median was 68.0/67.1 mm
2/s. The standard deviation was 6.45/6.04 mm
2/s and the coefficient of variation was 9.8/9.2%. According to the current SAE J300_2015 standard, there is no min/max indication for kinematic viscosity at 40 °C. However, the majority of manufacturers declare viscosity values for this temperature in their safety data sheets. The summary of data declared by the manufacturers and the values determined in the laboratory tests are presented in
Figure 1. A significant number of oils deviates from the values declared by the manufacturers. Thirteen oil samples had a higher value than the one declared, whereas 21 had a lower value (more details are given below). In eight cases the manufacturer’s declaration could not be found. Discrepancies at the level of up to 20% found between individual oils mean that the engine oil selected by a consumer may not fulfil the expected functions.
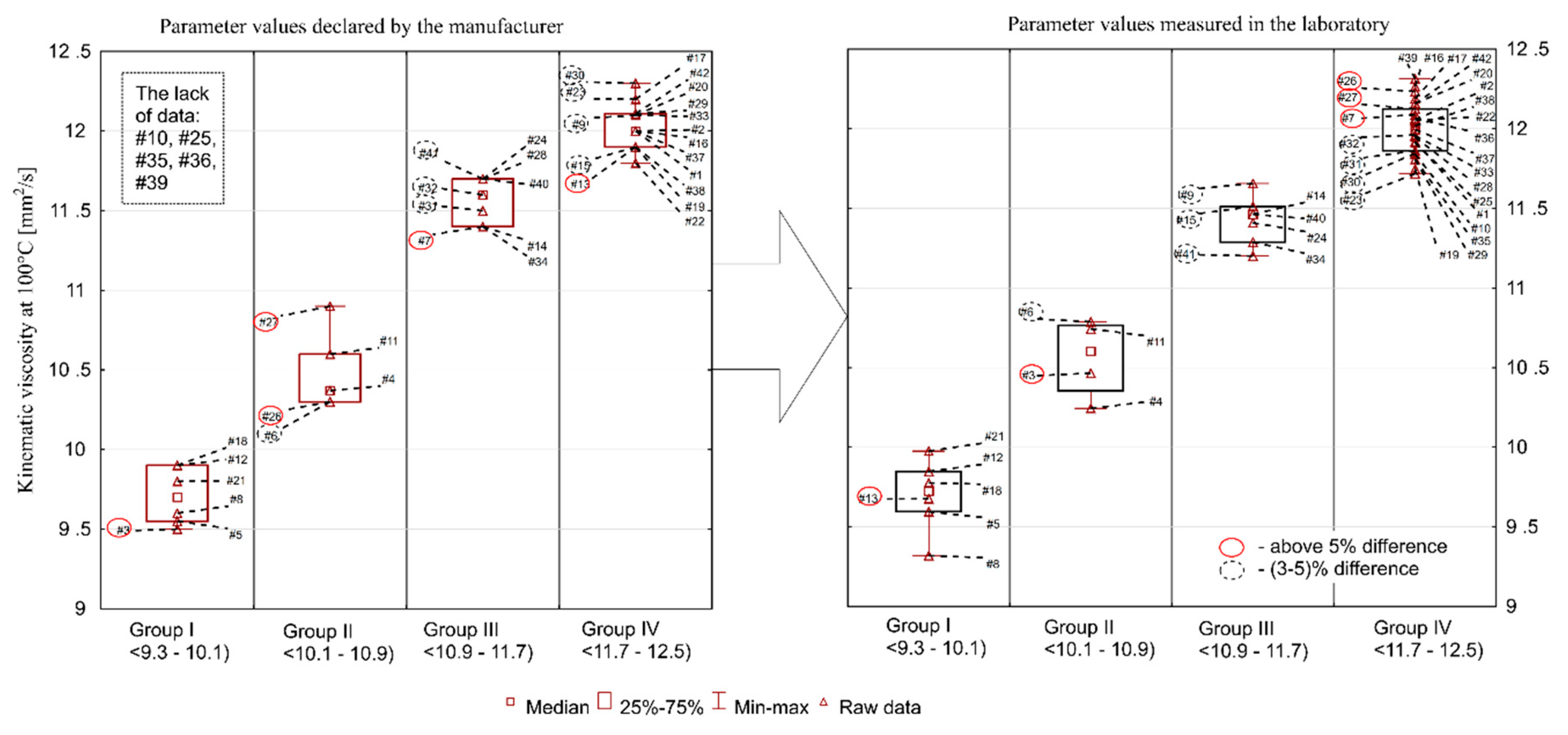
The aforementioned SAE J300 classification contains, however, guidelines (nominal range) for kinematic viscosity measured at 100 °C, and for oils of class 5W-30 it is a range of 9.3–12.5 mm
2/s. It should be stated that all results of the laboratory testing fell within this range, because the min-max (declared by manufacturer/measured in the laboratory) range was 9.5–12.3/9.3–12.3 mm
2/s. The average value was 11.4 mm
2/s (for both), median 11.7 mm
2/s (for both), standard deviation 0.9 mm
2/s (for both), while the coefficient of variation was at 8%/7%. Similar to viscosity at 40 °C, a significant number of manufacturers declare viscosity at 100 °C in their product data sheets (only in five cases there was no such information available). A comparison of manufacturers’ declarations and the measured values in the laboratory is shown in
Figure 2. As in the case of viscosity at 40 °C, the summary shows that a significant number of oils deviated from the values declared by the manufacturers: 18 oils had a higher value than the declared one, and 19 had a lower value (exact percentage difference are given below).
The largest discrepancies for kinematic viscosity at 100 °C were found for the following samples: #26 (measured value was 19% higher than the declared value), #27 (11% difference), #3 (10%), #7 (6%), #6 (4.7%), and #41 (4.3%). The situation was similar with regard to the discrepancies in kinematic viscosity measured at 40 °C. These were: #26 (24%), #27 (9%), #3 (15%), #7 (8%), #6 (7%), and #41 (13%), respectively. Viscosity may increase or decrease during use. An increase in viscosity is associated with progressive oxidation processes at elevated temperatures, and a decrease can be a consequence of oil dilution with fuel and/or shear polymers being added as oil viscosity modifiers. This means that a consumer choosing an oil from group I (#5, #8, #12, #13, #18, #21) may have to face its rapid exceedance in recommended viscosity grade, especially if a decrease in oil viscosity is observed during the engine operation. The situation is similar with oils from group IV, whose values are close to the upper limit value of 12.5 mm
2/s. They may exceed the limit as a result of an increase in engine oil viscosity, e.g., due to oxidation processes. As it was shown by [
27], the kinematic viscosity of engine oils used in “harsh” conditions increases by an average of about 10% after being in operation for 10,000 km. It was also indicated that the nature of viscosity changes depends on the oil formulation, i.e., the composition and technology used by the manufacturer.
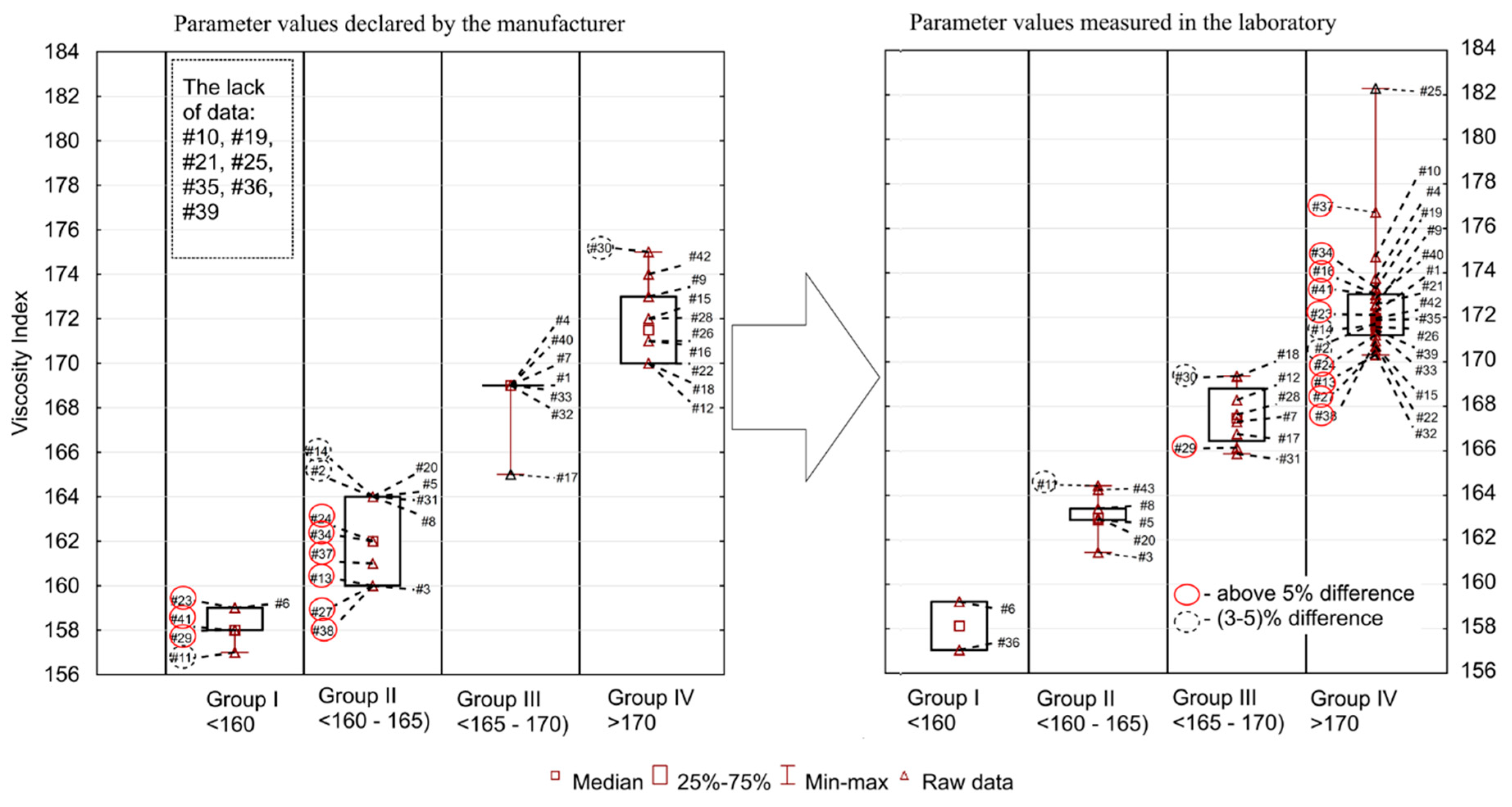
The viscosity index (VI) is a dimensionless quantity characterizing the variability of oil viscosity changes as a function of temperature changes. The viscosity indexes of modern lubricating oils fall between 85 and 250. High numbers mean smaller changes in viscosity, whereas low values mean relatively large changes, which are not favorable for engine operation. The VI can be increased by adding special additives (high-molecular polymers) to oils. In recent years, the following polymers have been used most frequently: polymethacrylates, polyalkylstyrenes and polyisobutylenes. During operation, an increase or decrease in the VI may occur. The reduction of the oil viscosity index is the result of the shear of viscositors, oxidation or degradation under the influence of water, while the increase is connected to the progressive oxidation processes at elevated temperature. The comparison of manufacturers’ declarations (no information for seven oils) and the values measured in laboratory tests are shown in
Figure 3. The range of obtained VI values was relatively wide. The minimum declared/measured value was 157 (in both), whereas the maximum was 175/182, the average (166/170) and median (164/171) were at different level. The coefficient of variation was 3.1%/2.8%, while the standard deviation was 5.2/4.7. Among all oils tested, 24 had a higher value than declared and 11 had lower. Different conclusions than in the case of the KV 40 and KV 100 were obtained for the VI. Here, the largest discrepancies were found in samples #37 and #41 (10%), #34 and #27 (7%), #24 (6%), and in #29, #2, #11 (5%).
3.2. Kinematic Viscosity at −20 °C and 130 °C
In addition to the comparison of the viscosity parameters provided by the manufacturers or resulting from the SAE J300 standard with the values from laboratory tests, it was also decided to explore the KV behavior at the sub-zero and above 100 °C. The kinematic viscosity measured at −20 °C is indirectly related to another very important parameter—structural viscosity (measured in the CCS apparatus—Cold Cranking Simulator), which determines engine starting ability at low temperatures [
16,
28]. A lower value of such viscosity corresponds to an easier start of the engine at low temperatures and means less load being put on the car battery.
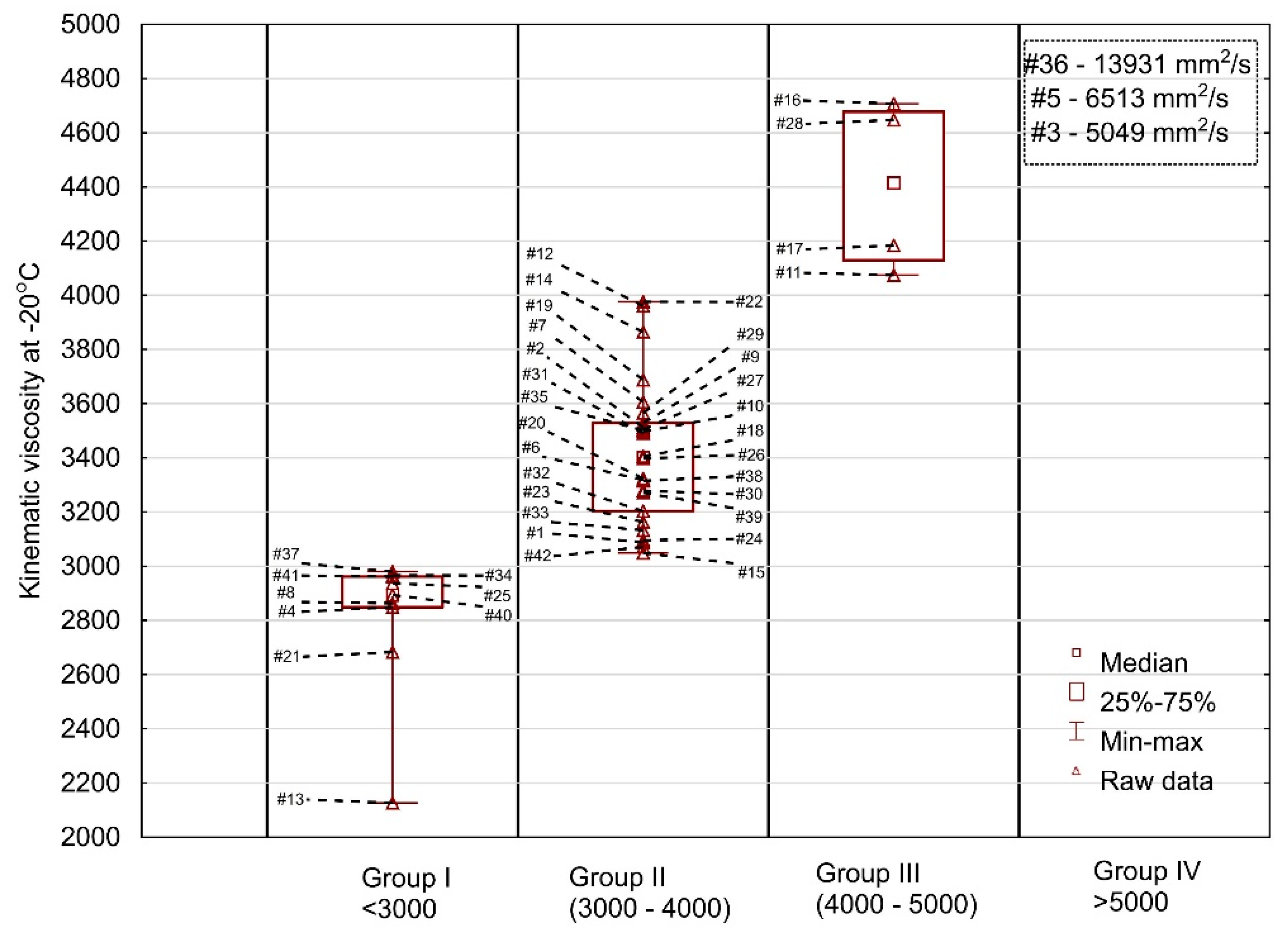
The results of the test at −20 °C are shown in
Figure 4. The oil samples tested were divided into four groups—nine oils in group I (<3000 mm
2/s), 26 oils in group II (3000–4000 mm
2/s) and four oils (#16, #28, #17, #11) in group III (>4000 mm
2/s) and in group IV—oils were characterized by surprisingly high kinematic viscosity values (#3, #5 and #36) of 5049, 6513 and 13,931 mm
2/s, respectively. A very large dispersion of the measurements was found mainly due to the sample #36 (the spread was on the level of 11,804 mm
2/s). The second most extreme oil had a significantly lower viscosity readout (6513 mm
2/s). The probability of selecting samples #3, #5 or #36 at random is 7%, nevertheless it is non-zero and may take place in practice causing problems for the user of such oil when starting the engine at extremely low temperatures. At the same time, it is worth to mention the oil #13 which was characterized by relatively low viscosity. Considering these two outlying oils, #13 and #36, it can be stated that when buying oil of the same viscosity grade, consumers may be faced with completely different oil behavior at low temperatures.
Another parameter that is not declared by manufacturers, although an important one from the point of view of engine operation, is the kinematic viscosity at 130 °C. The measurements at 130 °C are useful for the description of oil viscosity behavior at high temperatures. Preferably the change in viscosity when going from 100 °C to 130 °C should be small as this indicates oils with good performance properties.
The results of the test at 130 °C are shown in
Figure 5. The min–max range was 5.5–7.7 mm
2/s. The average value was 6.8 mm
2/s, median 7.0 mm
2/s, standard deviation 0.6 mm
2/s, and the coefficient of variation was at 8.4%. Comparing the viscosity at 130 °C to the viscosity at 100 °C, it was discovered that the oils showed a decrease in their kinematic viscosity of 37–44% (average 40%). The lowest change was recorded for oil #32 and the highest for oil #22. At the same time, it should be stated that in the group of oils with the lowest viscosity change there were both low and high viscosity oils at 100 °C, which means that high viscosity at 100 °C does not necessarily entail that the oil will maintain high viscosity at 130 °C. This indicates the differences in the formulation of individual oils and the content of viscosity modifiers.
For two extreme results (#21 and #32) additional HTHS measurements (temperature ~150 °C and shear rate ~10
6 s
−1) were performed. The lowest result was obtained for sample 8, while the volume of the oil sample necessary to perform the HTHS analysis resulted in sample 21 being analyzed, of which there was sufficient amount (all additional analyses absorbed large amounts of test material) to perform the analysis. The results were as follows: sample 21—3.02 mPa·s and sample 32—3.52 mPa·s. In his study [
29], Wolak gives HTHS results of five different fresh engine oils and none of them were as low (3.02)/high (3.52) as those selected as bottom (extreme lowest value) and top (extreme highest value) oils in the current work. In the quoted article, the minimum HTHS was 3.1, while the maximum HTHS was 3.48. This means that with a high degree of probability the selected oils, despite the relatively high HTHS range, meet the requirements of SAE J300, which is defined as minimum 2.6 mPa·s. Interestingly enough, oil samples with the highest VI (oils #37 and #25) were not found in the group of oils with the lowest viscosity change in the temperature range 100–130 °C, obtaining a value of 40%. At the same time, the oil with the highest change of 44% (oil #32) had a value of VI = 170. These results show that the information on the viscosity index alone does not give complete information about the change in oil viscosity above 100 °C. It should also be considered that modern cars come with the ever-smaller capacities of the oil system, which reduces the possibility of heat dissipation. With high long-term engine loads the oil temperature may increase, which might cause excessive reduction of viscosity and the loss of proper lubrication.
Figure 6 presents the results of the KV by ACEA performance class. A vast majority (31 samples) meet the ACEA C3 performance class and constitute one reference group. All other samples that belong to a different performance class than C3 were included in the second group.
The analysis of kinematic viscosity at positive temperatures indicates that in each of the three cases (40 °C, 100 °C, 130 °C) the situation was similar. The median KV for group I (ACEA C3) is significantly higher than for group II (ACEA class other than C3). On the other hand, in both groups at least one sample can be indicated which differs significantly from the others in terms of KV values—for group I it is sample #13 and for group II—samples #7 and #26. Therefore, in the same viscosity class and in the same performance class, a consumer can, with little, yet not zero probability, find an oil which will differ significantly from most other oils. A different situation occurred for KV at a temperature lower than 0—the median in both groups was at a similar level, while three samples (#36, #3, #5) were indicated as significantly different from the remaining oils. The result for sample #36 is particularly worrying. The research carried out by the authors previously [
16] showed that in −30 °C, too high kinematic viscosities may indicate not meeting the requirements of SAE J300 classification in sub-zero temperature parameters (CCS). As a result, it was decided that CCS measurements should be performed for two extreme samples (#13 and #36). The results were as follows: sample #13—CCS 3470 mPa·s, sample #36—CCS 11,000 mPa·s (the range of results was 7530 mPa·s). It may be recalled that KV at −20 °C was 2127 mm
2/s and 13,931 mm
2/s, respectively (the spread was on the level of 11,804 mm
2/s). Thus, as a result of a temperature decrease of −10 °C (CCS was measured at −30 °C) and the introduction of the shear rate, the gap between the highest and lowest viscosity values decreased, but still was large. This also confirmed that sample #36 did not meet the requirements of SAE J300, which assumes a maximum structural viscosity value of 6600 mPa·s.
In the paper all fluids tested met the SAE J300 100C viscosity specifications for SAE 5W-30 oils. However, when marketers blend oils to meet the SAE J300 5W-30 specifications they also must meet viscosity specifications at −30 °C and high shear rates and 150 °C and high shear rates. The viscosity measurements made by the authors at low temperature (−20 °C) and high temperature (130 °C) are at low shear rates. This difference in shear rates is significant since the 5W-30 oils contain polymers. The differences between the viscosities of the 5W-30 oils at high shear rates would be more critical than the low shear rate viscosities. Oils are not developed based on the low shear rate viscosities at −20 °C and 130 °C so there is no expectation that the 5W-30 oils should have similar viscosities at these temperatures and low shear rates. Partial testing, which was to verify the relationship between the viscosities measured at different shear rates is not a perfect solution, however, it is able to satisfactorily confirm the relationships that occur between the tested oils.
3.3. Viscosity Curves in the Temperature Range from −20 °C to 130 °C
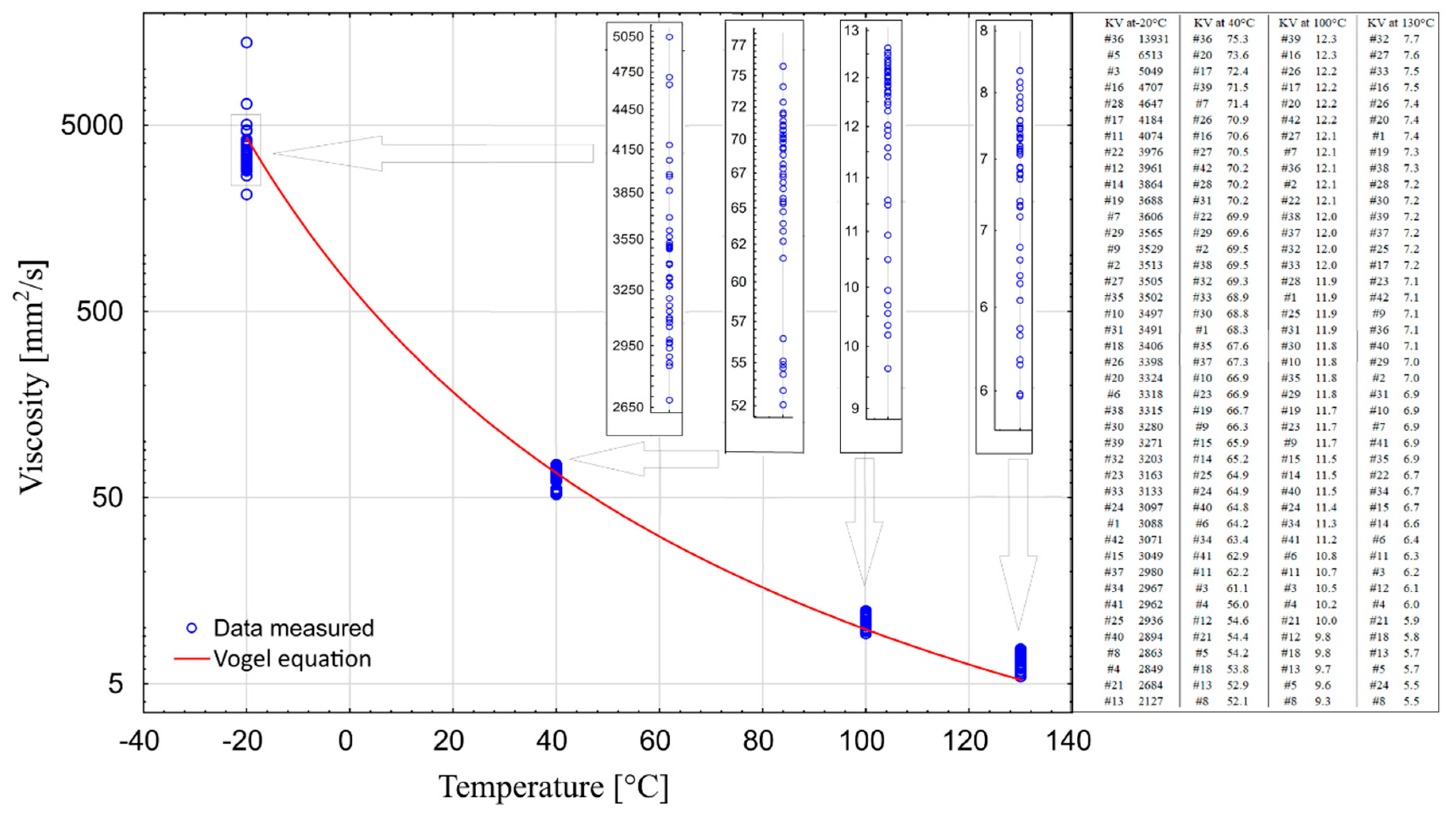
The combined results of the performed measurements were summarized in one chart (
Figure 7). This made it possible to assess the operational performance of all tested oils. For operational reasons, considering the specificity of the climate in which a car is used, the optimal solution is to use engine oils demonstrating the smallest variation in viscosity in the temperature range from −20 °C to 130 °C, and at the same time having the lowest possible viscosity at −20 °C.
To illustrate the course of kinematic viscosity curves from temperature, the Vogel equation was used in the form:
where:
η—dynamic viscosity,
a, b, c—constant values,
t0—temperature.
The empirical values of coefficients were taken from [
30] for Group IV oil—PAO—(
a—0.04839,
b—1193.38,
c—−124.72). Based on the obtained values, the value of kinematic viscosity was calculated assuming a linear variation of density with temperature.
It is worth noting that sample #16 is the only one that can be found at each measured temperature in the group of 10 oils with the highest values. The samples #17 and #36 could also be put on this list, if viscosity measurements at 130 °C were not included. On the other hand, samples #4, #8, #13, and #21 can be found in the group of 10 oils with the lowest viscosity values at each of the four temperatures. In temperatures above zero, sample #5 is one of the three samples with the lowest viscosity, whereas in temperatures below zero (−20 °C) it is the sample with the second highest viscosity level. The situation is similar in case of sample #3. These two samples are very different from sample #36, which exhibits one of the highest levels of viscosity over the entire temperature range. Sample #12 should also be distinguished as having one of the lowest viscosity values at temperatures above zero, and one of the highest at temperatures below zero.
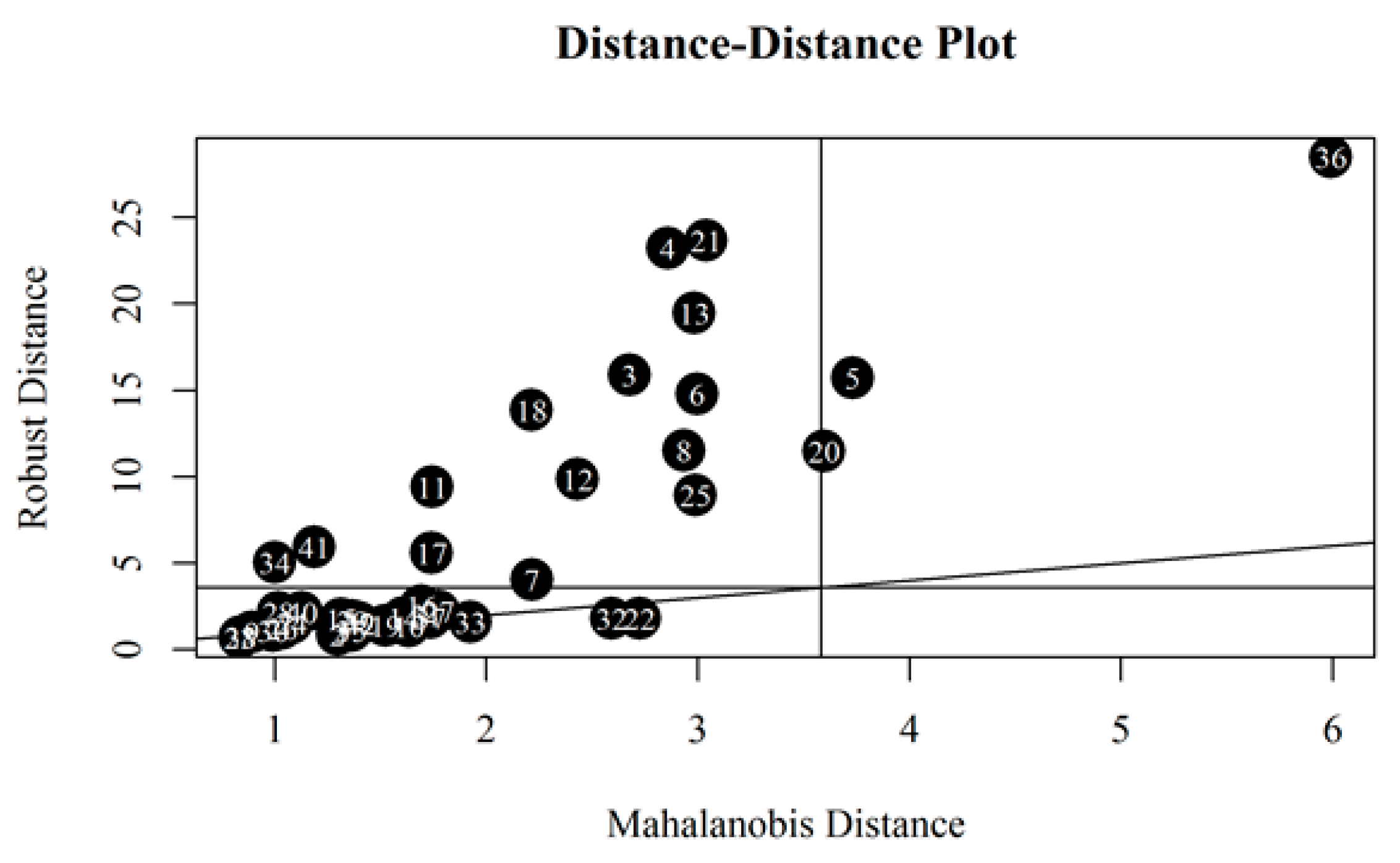
An attempt was made to discover potential outliers based on all collected data. The standard MD (Mahalanobis Distance) was placed on the horizontal axis and the robust MD was placed on the vertical axis (
Figure 8). The vertical and horizontal lines around 3.5 are a square root of 97.5% quantile of the chi-square distribution with five degrees of freedom. Robust Mahalanobis distances identified 16 outliers as oils above horizontal line can be considered as such. These were the following oils (ordered from the largest to the smallest MD): #36, 21, 4, 13, 3, 5, 6, 18, 8, 20, 12, 11, 25, 41, 17 and 34. The MD revealed similar samples that had been also flagged in the previous paragraphs. Oil samples #4, #21, #13, #8 are outliers due to their small KV values, as described earlier. Sample #6 is outlying due to the lowest VI value. In turn, sample #25 because of the highest VI value. Sample #3 has been discussed before (high viscosity values at temperatures below zero and low viscosity values at temperatures above zero). However, for samples #11 and #41 it is not clear why they were flagged as potential outliers.
3.4. Results of FTIR Analysis
In order to explain the obtained differences in viscosities of individual oils, their FTIR spectra were measured [
15,
31,
32]. It was found that the carbonyl stretching vibration band C=O (~1700–1800 cm
−1) in the hydrocarbon fraction is relatively low. In turn, the absorption bands of various additives in the same region, e.g., antioxidants, water, soot, etc. are quite intense. The fact that a stretching band can be observed in this region (1700–1800 cm
−1) in the hydrocarbon fraction of engine oils may also indicate the presence of various types of oxidation products (such as aldehydes, ketones, carboxylic acids) or even some oxidation inhibitors (such as imides). Oftentimes, poor absorption of some oils in the regions of ~1750 cm
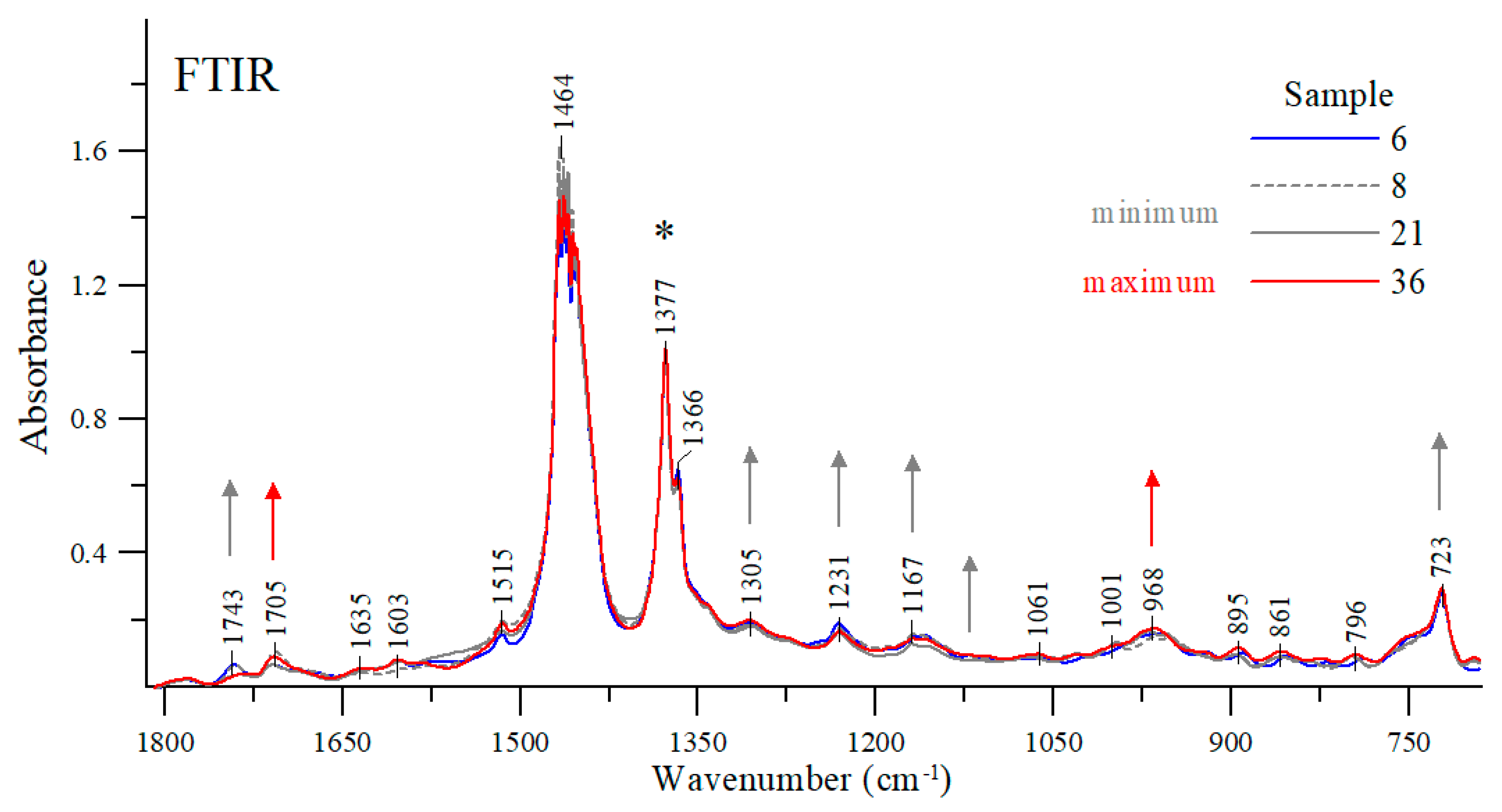
−1 wavenumber can be attributed to the presence of small amounts (usually less than 1%) of depressants (such as polymers with carbonyl groups). The spectra were measured for all of the oils tested. However, to simplify the analysis, only those oils were selected for further study whose viscosity was significantly different from other oil samples (e.g., #8, #21, #36) or represented average values (such as oil sample #6). All of the spectra were normalized at a maximum of about 1377 cm
−1 in order to facilitate the analysis (
Figure 9).
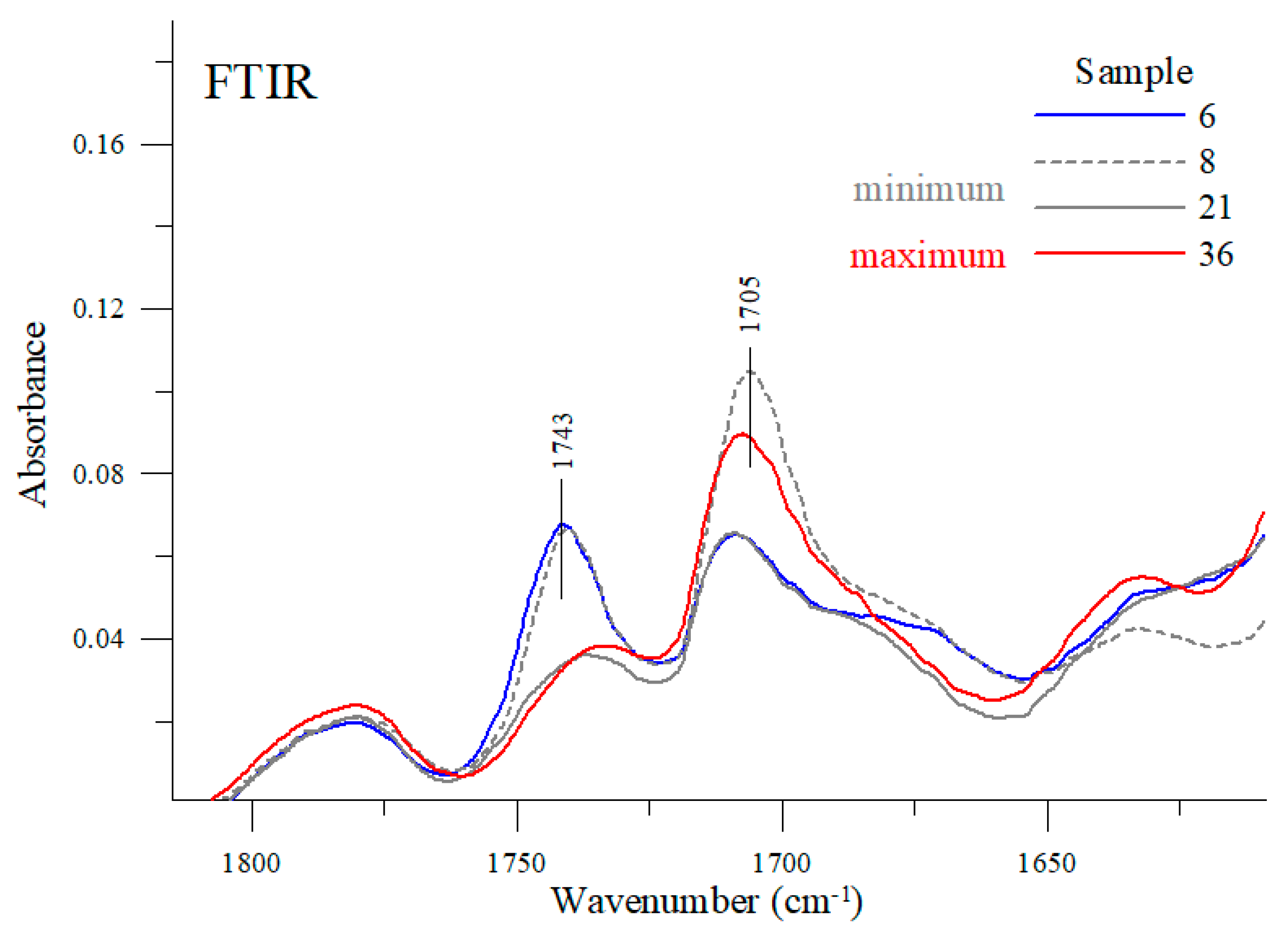
At the wavenumber of 1743 cm
−1 (and 1705 cm
−1), carbonyl stretching vibration bands C=O can be detected [
33,
34]. Depending on the engine oil, the intensity ratio of these bands varies considerably. For example, the stretching bands of the samples #8, #6 or #21 had their maxima at a higher wavenumber; whereas for sample #36 the stretching band with a maximum at about 1743 cm
−1 lost its intensity in favor of a sharp increase in the band with a maximum at about 1705 cm
−1. As shown earlier, oil samples #8 and #21 have the lowest viscosity while sample #36 has the highest viscosity among the samples tested. For comparison purposes, sample #6 exhibits one of the average viscosity values as measured in the study. Vibrations with a maximum at about 1705 cm
−1 can be attributed to the formation of a hydrogen bond between the groups C=O⋯H-O- [
33,
34,
35]. In the case of the samples tested, a decrease in the intensity of this band in favor of an increase in the intensity of the band with a maximum at about 1743 cm
−1 is clearly associated with changes in the viscosity of the oils tested. The more exact regions of these two bands are shown in
Figure 10.
Along with the changes in band intensity at around 1743 cm
−1 and 1705 cm
−1 (
Figure 9 and
Figure 10), additional ones are observed for selected samples in other spectral ranges, also interpreted in the context of changes in their viscosity [
33,
34,
35]. The region with a maximum at about 968 cm
−1 represents various stretching vibrations such as ν(C-H)
s or ν(C-C), increasing their intensity as the intensity of the band goes up to a maximum at about 1705 cm
−1. Considering this band together with the detected changes in the intensity of the previously discussed regions in which the samples exhibited a higher viscosity value leads to an obvious conclusion that an increase in its intensity is yet another factor confirming earlier observations. However, as the bands go up with a maximum at around 1743 cm
−1, the other ones in the following regions: 1305, 1231, 1167 and 1109 and 723 cm
−1 also increase in intensity (
Figure 9), perfectly matching the samples for which the lowest viscosity values were observed.
3.5. The Chromatographic Analysis
The rheological properties at temperatures above and below zero largely depend on the type of base oils used, which make up from 85% to 90% of the engine oil composition in total. Following the manufacturers’ declarations, the group of oils tested should contain base oils obtained by polymerization, polycondensation, chemical synthesis or catalytic hydrocracking. One of the methods to assess the composition of engine oil is the analysis of true boiling points, determined by gas chromatography with the use of an SIMDIS apparatus [
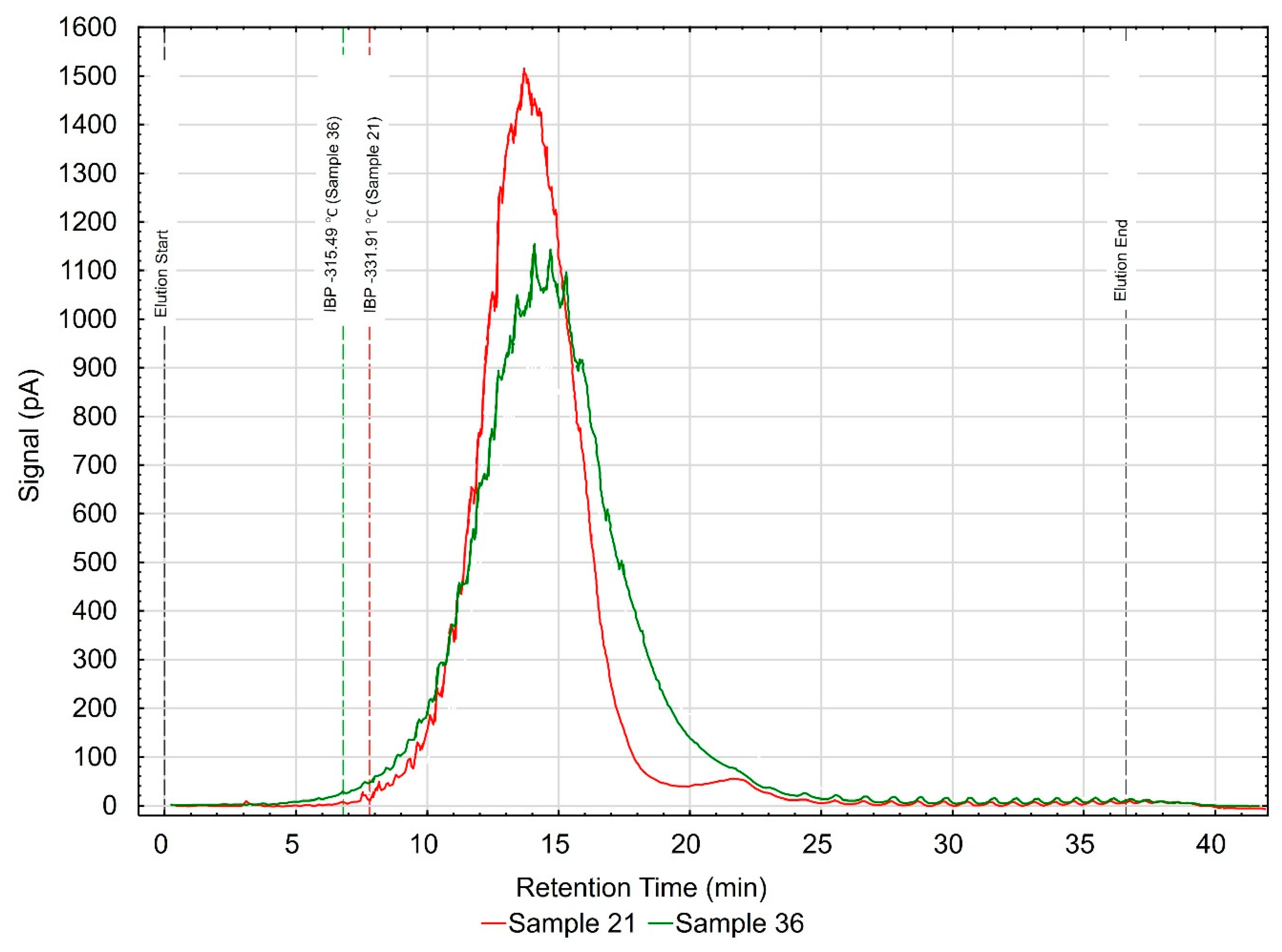
36]. The analysis of the results of rheological property tests made it possible to select two samples marked #21 (representing low-viscosity samples at −20 °C) and #36 (representing high-viscosity samples at −20 °C) from a group of 42 oils. The results of chromatographic tests are depicted in
Figure 11, showing chromatograms of the samples tested in the characteristics of “retention time—signal size.”
Visible differences in the obtained chromatograms have led to the assumption that the compositions of the base oils used in the manufacturing of the engine oils tested visibly vary. A symmetrical shape of the curve representing sample #21 indicates the use of a thermal hydrocracking oil. This sample also contains fewer ingredients with high boiling points. In turn, sample #36 may contain admixtures of various types of base oils (Group II and Group III—API base oil) in its composition. This is especially visible through clear peaks corresponding to components (poly-alpha-olefins—PAOs) of a different chemical nature in comparison to other components [
36,
37,
38]. A different slope of the graphs in terms of signal to retention time observed in the range from 15 to 20 min (sample #36), and a much higher signal level (sample #36) in the retention time range from 15 to 25 min, confirm that there are significant differences in the composition of the engine oils tested [
39]. The base oils used to compose the final product have a discernible impact on the rheological properties of engine oils, especially at subzero temperatures. Extra-normative results were obtained for sample #36 in structural viscosity at −30 °C, which might have been caused by incorporating into its composition some mineral base oils with much worse properties and which have higher boiling points, as can be observed in the analyzed chromatogram.
3.6. Oil’s Energy Efficiency
Out of 42 studied oils, a significant group (eight oils, 19%) was marked by the manufacturers as the energy-saving oils (Among the 42 oils, the sample #14 has an indication ILSAC GF- 5, the samples #5 and #8 have ILSAC GF-4, while the samples #11, #12, #18, #21, #42 have a manufacturer’s designation as “fuel economy”). An energy-saving motor oil is an oil with a reduced HTHS (High Temperature High Shear-rate viscosity), usually below 3.5 mPa·s. One of the results of energy-saving is the reduction in fuel consumption. This is the cost most affecting the total cost of car operation. Reducing fuel consumption also significantly reduces atmospheric pollution. As this is an important engine oil segment, additional details are provided.
The collected results for this group of oils indicate that in five cases (#5, #8, #12, #18, #21), manufacturers achieved the energy efficiency by reducing viscosity at high temperatures (KV 100 < 10 mm
2/s, both declared and measured). Another three samples (#11, #14, #42) have this viscosity above 10 mm
2/s. Especially, samples #14 and #42 do not clearly differ from the remaining oils with regard to viscosity. In their case, the energy efficiency is most likely ensured in a different way other than minimizing the viscosity of the oil. Surprisingly, samples #5 and #11 had high viscosity at −20 °C (6513 and 4074 mm
2/s, respectively), even though sample #5 was one of the two samples with the overall lowest KV 100. Sample’s #11 viscosity index (
Figure 3) declared by the manufacturer was noticeably different from the measured one. Oil #8 was in the group of 10 oils with the lowest viscosity at each of the four analyzed temperatures. Samples #18 and #21, at each of the four measured viscosity temperatures, can be found in the group of 10 oils with the lowest viscosity.
Taking the oil market perspective, the ongoing trends, reinforced by the numerous studies in this area, are driving the market towards higher performance and more fuel-efficient lubricants. It is believed that in a not-so-distant future, lower viscosity SAE 5W-30 and 5W-40 engine lubricants will be superseded by more efficient SAE 0W-20 and 0W-16 viscosity grades. The market introduction of SAE 0W-16 and 5W-16 has opened up an opportunity to improve engine oil fuel economy even further. Both of them were included in the revision of API Publication 1509 in the API SN category in 2014, but the fuel economy criteria were not provided there due to a lack of sufficient data. The improvements in fuel economy that result from the use of low friction engine oils are well known within the industry, and can be obtained mainly by reduction in oil viscosity [
40,
41,
42,
43] (through the addition of specialty chemicals like, e.g., friction modifiers). Based on the ILSAC (In 1994, the ILSAC (International Lubricant Standardization and Approval Committee) was established in the USA to introduce a new classification of energy-saving and fuel-saving oils into the scientific and information circulation, and to determine how to test and certify such products. ILSAC was created as a result of an agreement between AAM (Alliance of Automobile Manufactures) and JAMA (Japanese Automobile Manufacturers Association)). ILSAC has developed test and research procedures to identify the most fuel-saving oils. In this general sphere other organizations also exist. For example, API (American Petroleum Institute) is responsible for identifying the standards that a motor oil should meet. The API is a consensus specification voted on by all involved automotive organizations. To receive the API seal of approval, the motor oil has to comply with the SAE (Society of Automotive Engineers) oil viscosity standards. ILSAC differs from API because it also measures fuel economy, while API does not. GF-4 (Starting from 1 May 2020, ILSAC GF-6A and GF-6B will be the standard. If a car previously required ILSAC GF 1-5, it is safe to use oils formulated with the new ILSAC GF-6A standard. ILSAC GF-6B can be used to meet SAE 0W-16 recommendations.) economy classification (linked to the API SM classification), in which the same 5W-30 synthetic oil is the reference point, the following minimum fuel savings can be expected: 1.8% in SAE 0W-30 and 5W-30, 2.3% in SAE 0W-20 and 5W-20 viscosity grades. Ishikava experimentally obtained 2.8% savings for the FEI (Fuel Economy Improvement) indicator and 1.3% for the FEI2 indicator when using SAE 0W-16 oil based on readily available market GF-5 technologies [
44]. Mo confirmed that at the same viscosity grade, the vehicle fuel economy of Dexos1 engine oil is 0.36% higher than the one of API SN engine oil. The vehicle fuel economy was highly correlated with engine friction loss. Five-point-five percent engine friction loss reduction was approximately equivalent to 1% vehicle fuel economy improvement. They noted that special additives of engine oil formulation technology has further potential for fuel economy improvement [
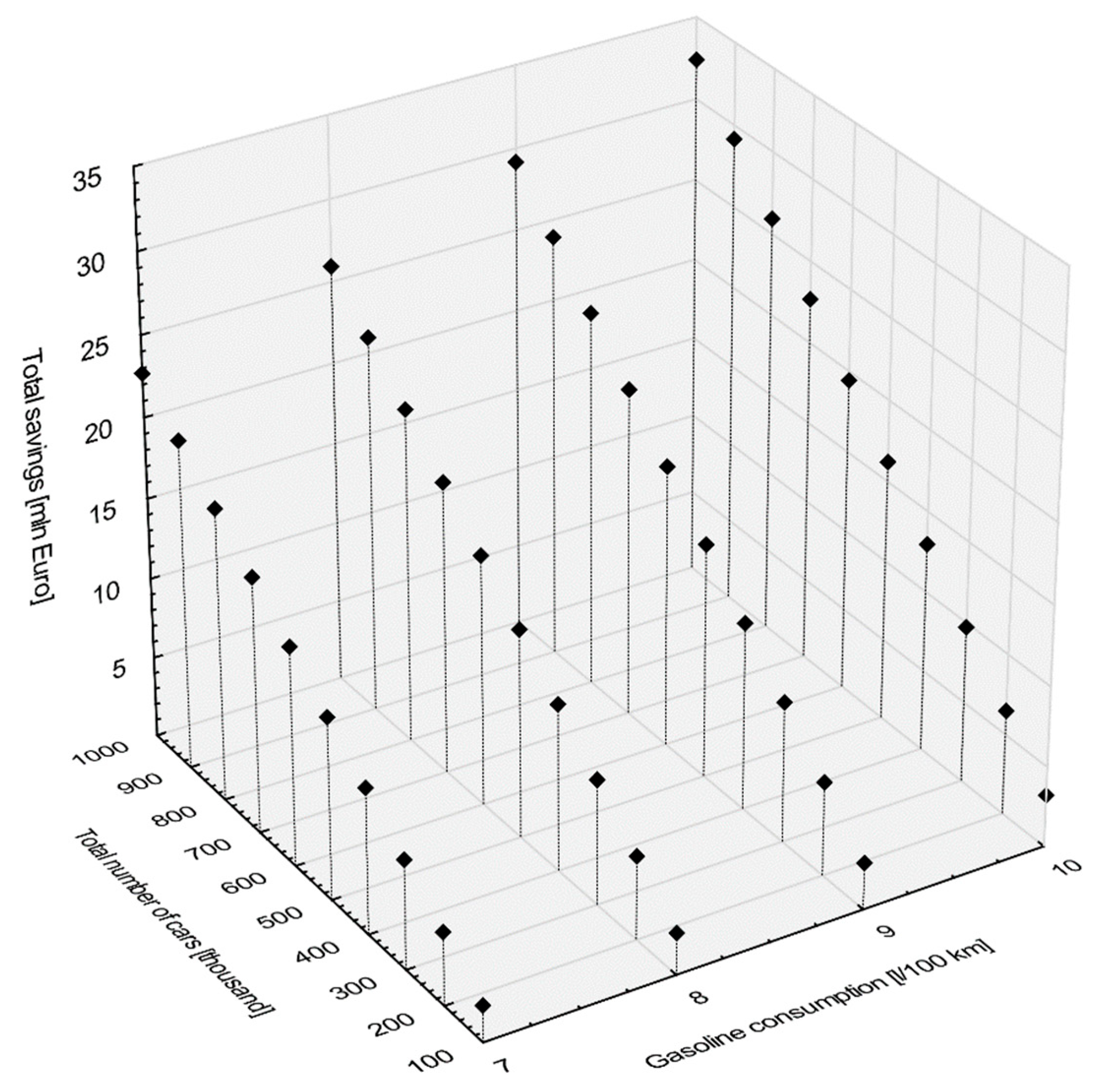
45]. Therefore, if the car manufacturer allows the use of oils with different viscosities, then from an economic and environmental point of view it is recommended to use oils with low viscosity type 0W-30, 5W-30 because such oils save more fuel than 5W-40, 10W-40 or 15W-40. Thanks to the above classifications, it can be concluded that the use of oil, e.g., 5W30 (such is the subject of analysis in the article), which underwent ILSAC testing and received GF-4 class or higher, will bring potential consumers savings in the form of reduced fuel consumption, even if compared to a similar reference synthetic oil SAE 5W-30, by at least 1.8%. To illustrate the potential upper limit of direct monetary savings for the drivers a simple hypothetical scenario was constructed. The scenario does not consider the additional cost of fuel-saving technologies. It is assumed that the price of gasoline is 1.5 Euro per liter, yearly mileage of a typical passenger car is 12,000 km and the expected reduction of gasoline consumption from baseline due to the energy efficient engine oil is equal to 1.8%. Under this scenario,
Figure 12 shows the total savings in millions of Euro per year as a function of the total number of identically utilized cars (in thousands) and the average gasoline consumption per 100 km (in liters).