Improvement of Organosolv Fractionation Performance for Rice Husk through a Low Acid-Catalyzation
Abstract
:1. Introduction
2. Results and Discussion
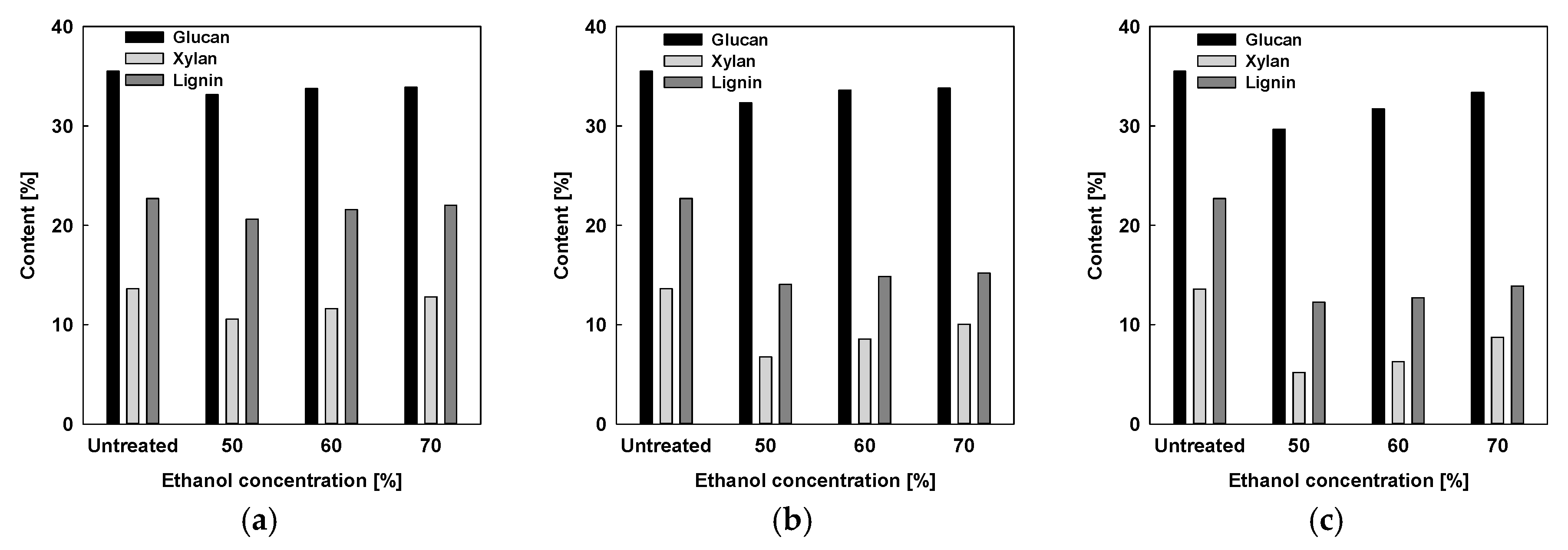
2.1. Organosolv Fractionation of RH
2.1.1. Organosolv Fractionation with Reaction Temperature and EtOH Concentration
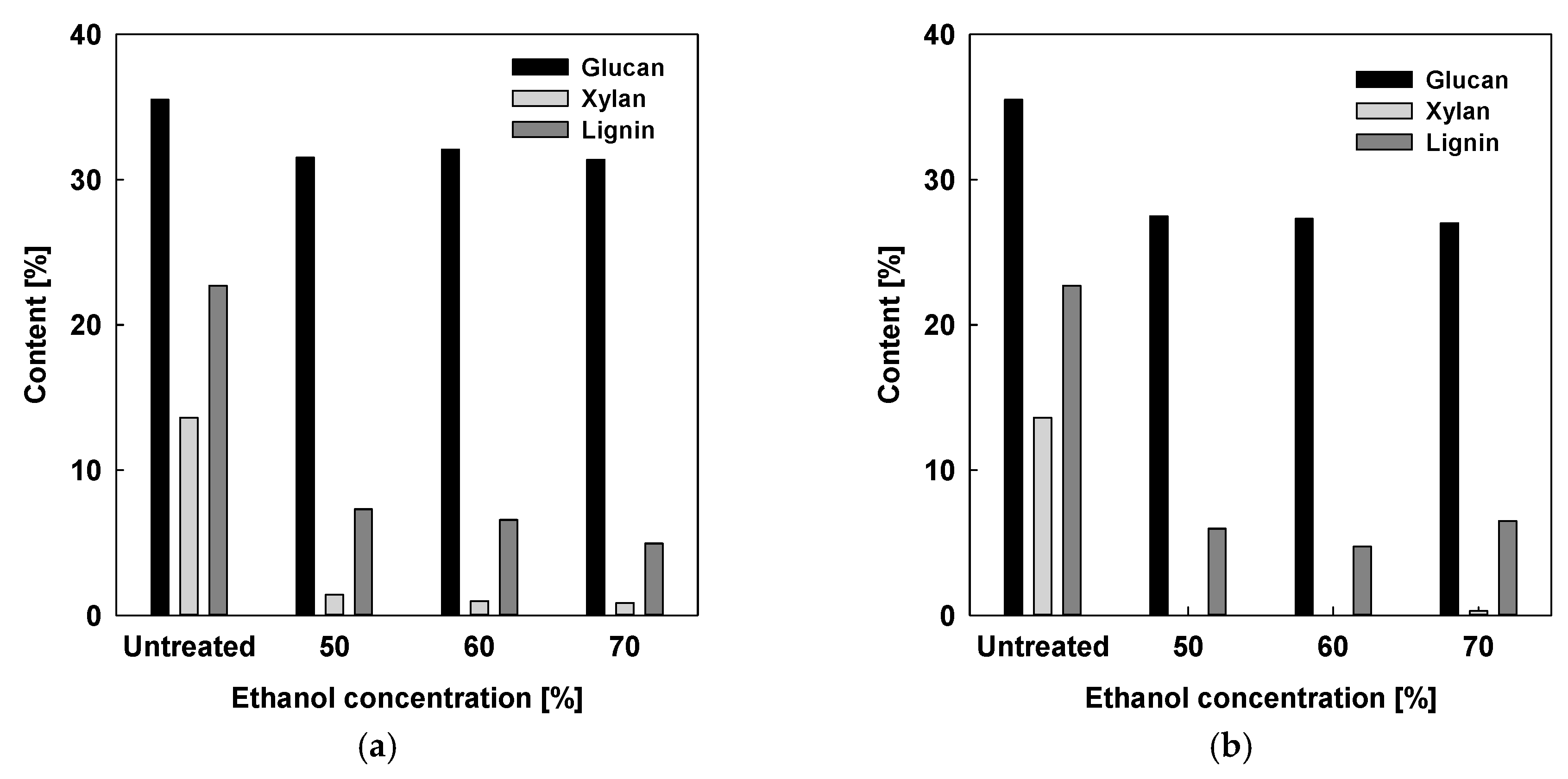
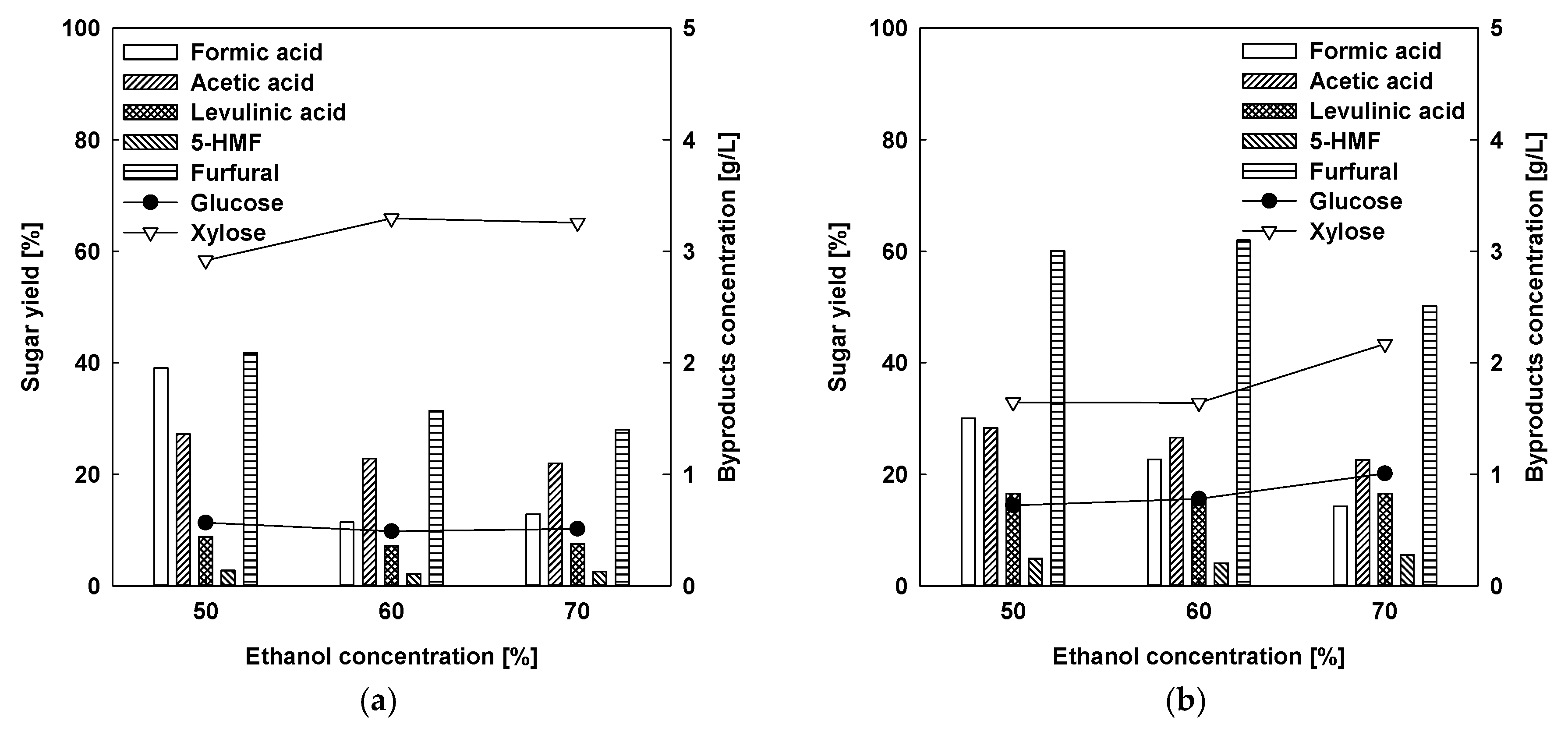
2.1.2. Acid-Catalyzed Organosolv Fractionation for Xylose and Lignin Extraction
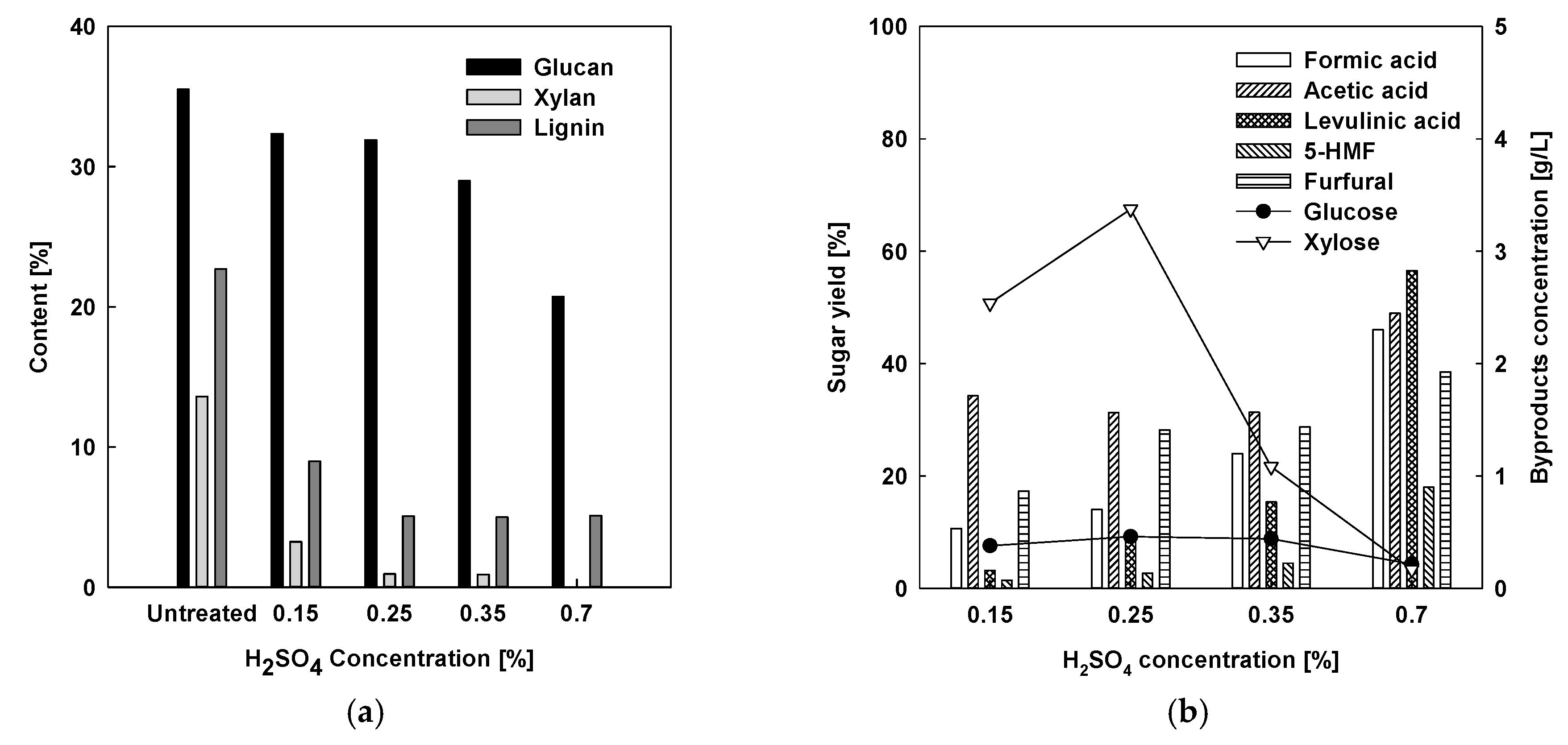
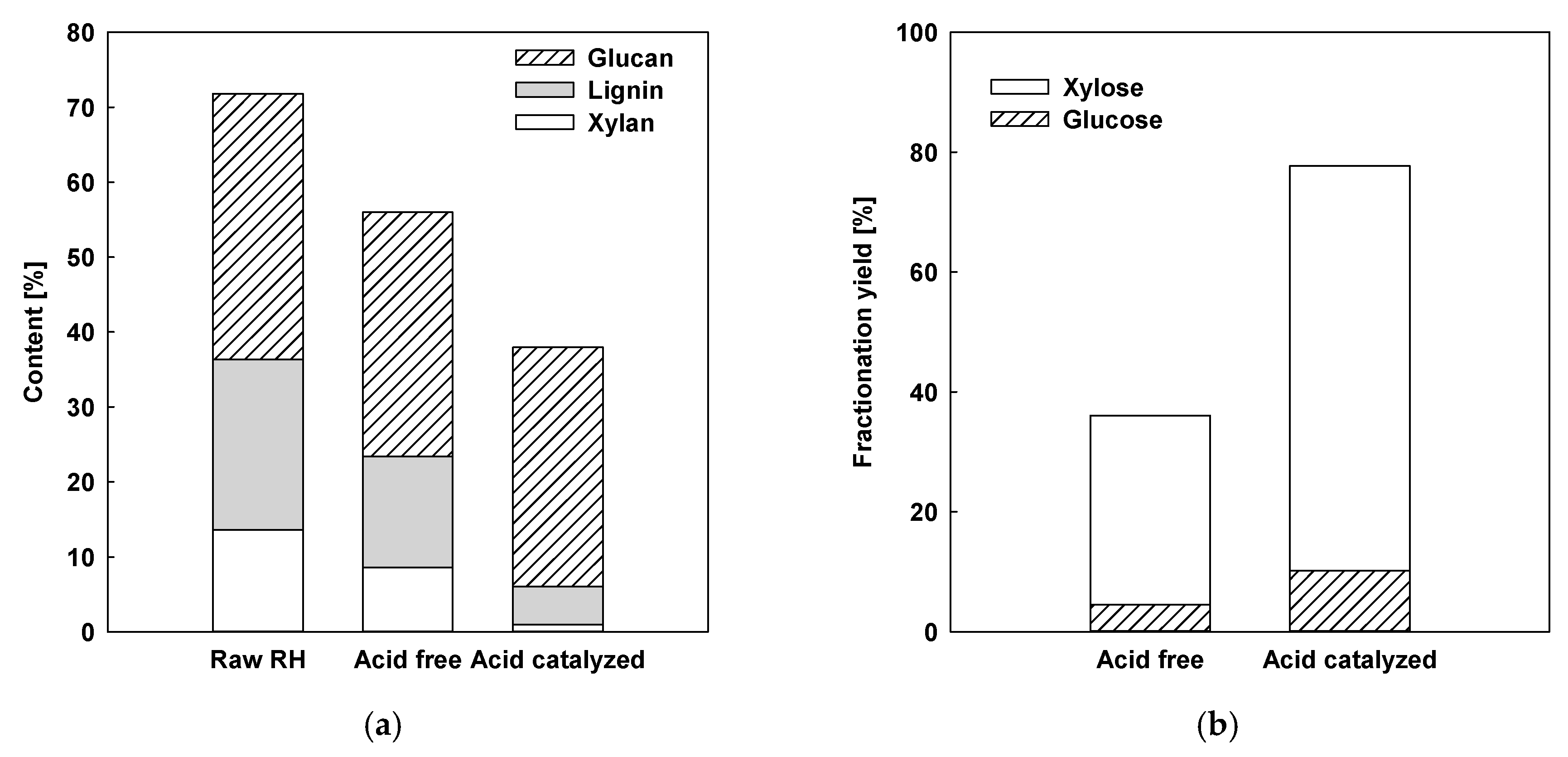
2.1.3. Comparison with Acid-Free and Acid-Catalyzed Organosolv Fractionation
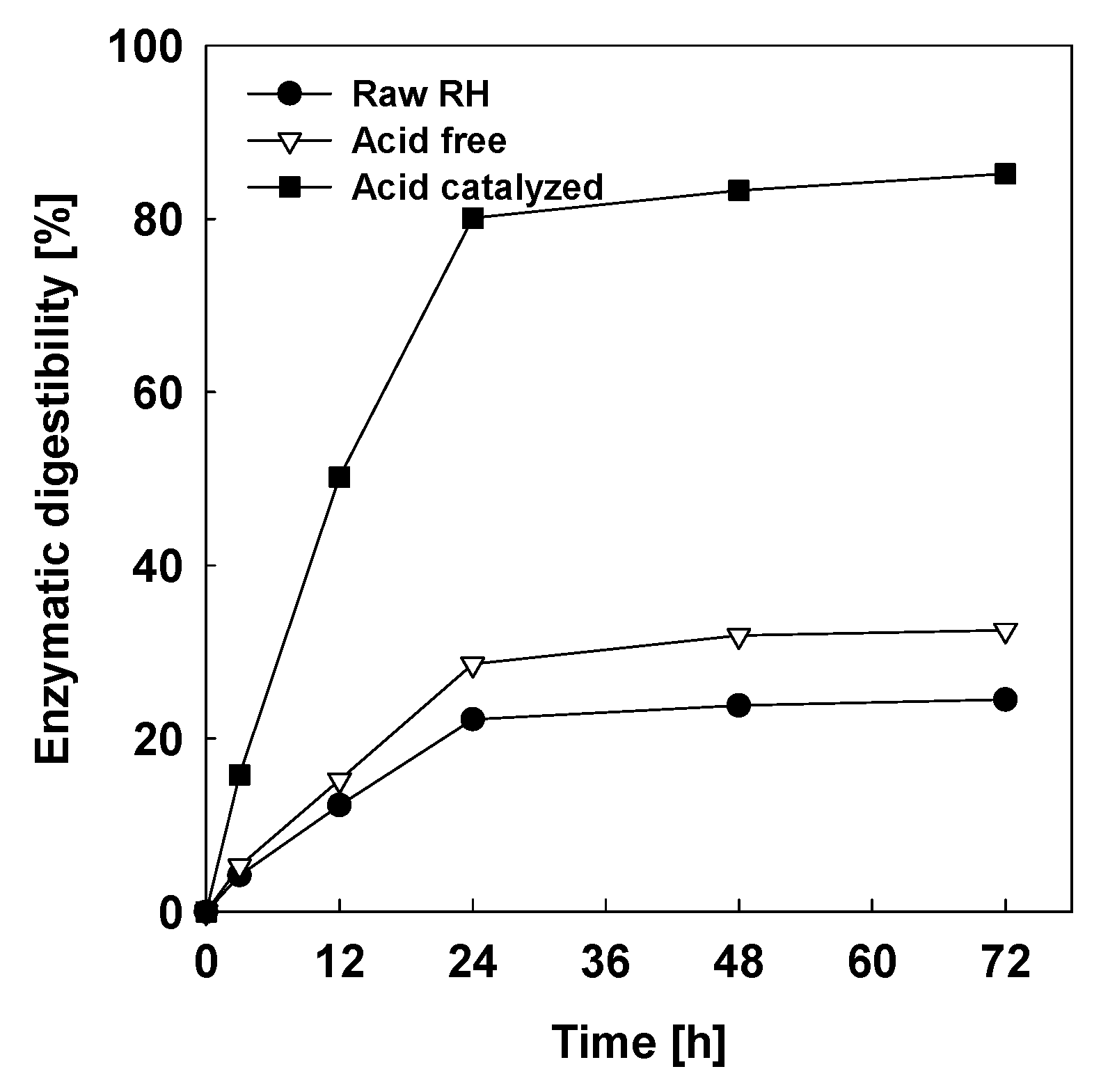
2.1.4. Enzymatic Hydrolysis Tests
2.2. Chemical Characteristics of Organosolv-Fractionated Lignin
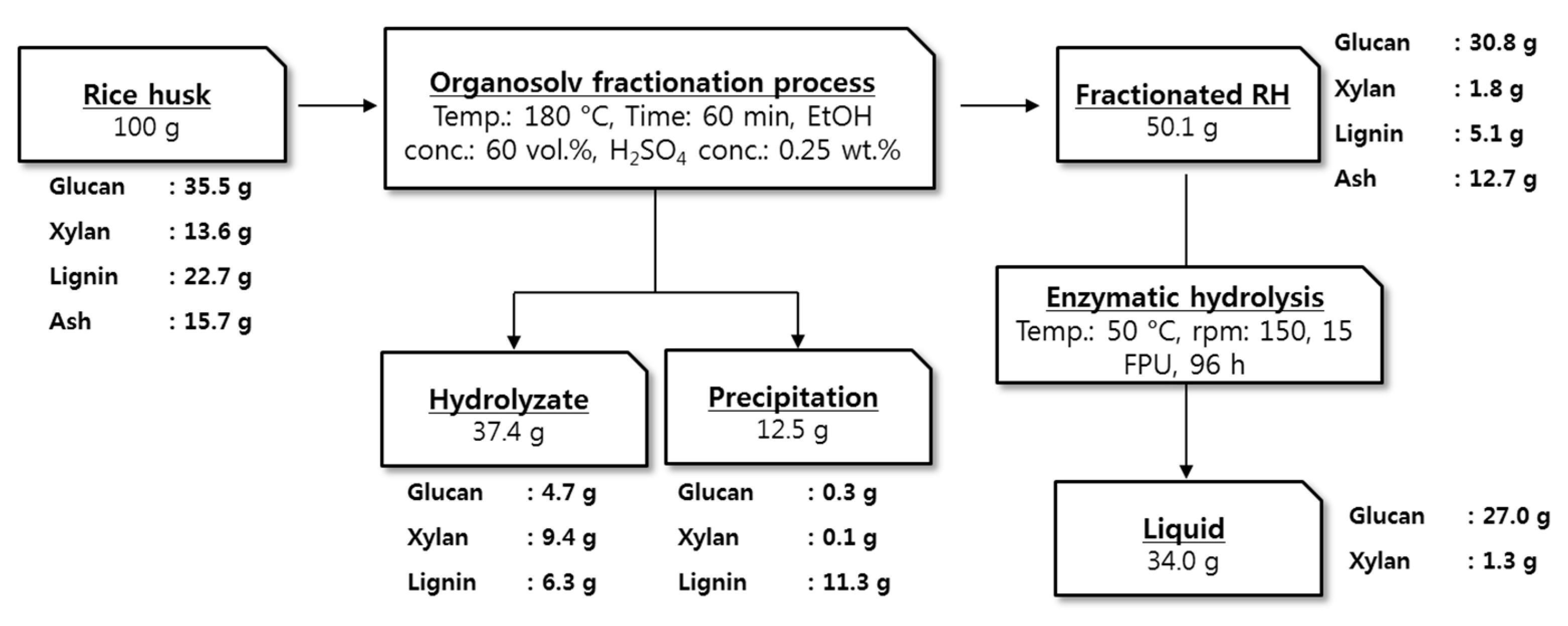
2.3. Overall Fractionation Yield and Total Mass Balance
3. Materials and Methods
3.1. Materials
3.2. Experimental Setup and Operation
3.3. Enzymatic Digestibility Tests
3.4. Composition Analysis of Raw and Fractionated RH
3.5. Chemical Characterization of Organosolv Fractionated Lignin
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lim, J.S.; Manan, Z.A.; Alwi, S.R.W.; Hashim, H. A review on utilisation of biomass from rice industry as a source of renewable energy. Renew. Sustain. Energy Rev. 2012, 16, 3084–3094. [Google Scholar] [CrossRef]
- Zhang, H.; Ding, X.; Chen, X.; Ma, Y.; Wang, Z.; Zhao, X. A new method of utilizing rice husk: Consecutively preparing d-xylose, organosolv lignin, ethanol and amorphous superfine silica. J. Hazard. Mater. 2015, 291, 65–73. [Google Scholar] [CrossRef]
- Soltani, N.; Bahrami, A.; Pech-Canul, M.I.; González, L.A. Review on the physicochemical treatments of rice husk for production of advanced materials. Chem. Eng. J. 2015, 264, 899–935. [Google Scholar] [CrossRef]
- Menya, E.; Olupot, P.W.; Storz, H.; Lubwama, M.; Kiros, Y. Production and performance of activated carbon from rice husks for removal of natural organic matter from water: A review. Chem. Eng. Res. Des. 2018, 129, 271–296. [Google Scholar] [CrossRef]
- Kim, T.H.; Kim, T.H. Consecutive Recovery of Non-Structural Sugars and Xylooligomers from Corn Stover using Hot Water and Acidified Calcium Chloride. BioResources 2018, 13, 7294–7309. [Google Scholar] [CrossRef]
- International Energy Agency. Available online: https://www.ieabioenergy.com/publications/bio-based-chemicals-value-added-products-from-biorefineries/ (accessed on 14 March 2019).
- Chen, H.; Liu, J.; Chang, X.; Chen, D.; Xue, Y.; Liu, P.; Lin, H.; Han, S. A review on the pretreatment of lignocellulose for high-value chemicals. Fuel Process. Technol. 2017, 160, 196–206. [Google Scholar] [CrossRef]
- Guo, Y.; Zhou, J.; Wen, J.; Sun, G.; Sun, Y. Structural transformations of triploid of Populus tomentosa Carr. lignin during auto-catalyzed ethanol organosolv pretreatment. Ind. Crops Prod. 2015, 76, 522–529. [Google Scholar] [CrossRef]
- Zhao, X.; Cheng, K.; Liu, D. Organosolv pretreatment of lignocellulosic biomass for enzymatic hydrolysis. Appl. Microbiol. Biotechnol. 2009, 82, 815. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Pei, Z.; Wang, D. Organic solvent pretreatment of lignocellulosic biomass for biofuels and biochemicals: A review. Bioresour. Technol. 2016, 199, 21–33. [Google Scholar] [CrossRef]
- Lora, J.H.; Glasser, W.G. Recent industrial applications of lignin: A sustainable alternative to nonrenewable materials. J. Polym. Environ. 2002, 10, 39–48. [Google Scholar] [CrossRef]
- Teramoto, Y.; Tanaka, N.; Lee, S.H.; Endo, T. Pretreatment of eucalyptus wood chips for enzymatic saccharification using combined sulfuric acid-free ethanol cooking and ball milling. Biotechnol. Bioeng. 2008, 99, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.H.; Im, D.J.; Oh, K.K. Effects of Organosolv Pretreatment Using Temperature-Controlled Bench-Scale Ball Milling on Enzymatic Saccharification of Miscanthus × giganteus. Energies 2018, 11, 2657. [Google Scholar] [CrossRef]
- Pan, X.; Kadla, J.F.; Ehara, K.; Gilkes, N.; Saddler, J.N. Organosolv ethanol lignin from hybrid poplar as a radical scavenger: Relationship between lignin structure, extraction conditions, and antioxidant activity. J. Agric. Food Chem. 2006, 54, 5806–5813. [Google Scholar] [CrossRef] [PubMed]
- Weinwurm, F.; Turk, T.; Denner, J.; Whitmore, K.; Friedl, A. Combined liquid hot water and ethanol organosolv treatment of wheat straw for extraction and reaction modeling. J. Clean. Prod. 2017, 165, 1473–1484. [Google Scholar] [CrossRef]
- Kim, T.H.; Jeon, Y.J.; Oh, K.K.; Kim, T.H. Production of furfural and cellulose from barley straw using acidified zinc chloride. Korean J. Chem. Eng. 2013, 30, 1339–1346. [Google Scholar] [CrossRef]
- Pan, X.; Gilkes, N.; Kadla, J.; Pye, K.; Saka, S.; Gregg, D.; Ehara, K.; Xie, D.; Lam, D.; Saddler, J. Bioconversion of hybrid poplar to ethanol and co-products using an organosolv fractionation process: Optimization of process yields. Biotechnol. Bioeng. 2006, 94, 851–861. [Google Scholar] [CrossRef]
- Agarwal, A.; Rana, M.; Park, J.H. Advancement in technologies for the depolymerization of lignin. Fuel Process. Technol. 2018, 181, 115–132. [Google Scholar] [CrossRef]
- Yáñez-S, M.; Matsuhiro, B.; Nunez, C.; Pan, S.; Hubbell, C.A.; Sannigrahi, P.; Ragauskas, A.J. Physicochemical characterization of ethanol organosolv lignin (EOL) from Eucalyptus globulus: Effect of extraction conditions on the molecular structure. Polym. Degrad. Stab. 2014, 110, 184–194. [Google Scholar] [CrossRef]
- Domínguez-Robles, J.; Tamminen, T.; Liitiä, T.; Peresin, M.S.; Rodríguez, A.; Jääskeläinen, A.S. Aqueous acetone fractionation of kraft, organosolv and soda lignins. Int. J. Biol. Macromol. 2018, 106, 979–987. [Google Scholar] [CrossRef]
- El Mansouri, N.E.; Salvadó, J. Structural characterization of technical lignins for the production of adhesives: Application to lignosulfonate, kraft, soda-anthraquinone, organosolv and ethanol process lignins. Ind. Crops Prod. 2006, 24, 8–16. [Google Scholar] [CrossRef]
- Fahmi, R.; Bridgwater, A.V.; Donnison, I.; Yates, N.; Jones, J.M. The effect of lignin and inorganic species in biomass on pyrolysis oil yields, quality and stability. Fuel 2008, 87, 1230–1240. [Google Scholar] [CrossRef]
- Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, A.; Sluiter, J.; Tmpleton, D. Preparation of Samples for Compositional Analysis; NREL/TP-510-42620; National Renewable Energy Laboratory: Golden, CO, USA, 2012.
- Sluiter, A.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D. Determination of Extractives in Biomass; NREL/TP-510-42619; National Renewable Energy Laboratory: Golden, CO, USA, 2012.
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D. Determination of Structural Carbohydrates and Lignin in Biomass; NREL/TP-510-42618; National Renewable Energy Laboratory: Golden, CO, USA, 2012.
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D. Determination of Ash in Biomass; NREL/TP-510-42622; National Renewable Energy Laboratory: Golden, CO, USA, 2008.
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D. Determination of Sugars, Byproducts, and Degradation Products in Liquid Fraction Process Samples; NREL/TP-510-42623; National Renewable Energy Laboratory: Golden, CO, USA, 2008.
- Selig, M.; Weiss, N.; Ji, Y. Enzymatic Saccharification of Lignocellulosic Biomass; NREL/TP-510-42629; National Renewable Energy Laboratory: Golden, CO, USA, 2008.

| Content | Classify | Unit | Acid-Free | Acid Catalyzed |
|---|---|---|---|---|
| Hydroxyl group | Aliphatic unit | mmol/g | 2.84 | 2.76 |
| p-Hydroxyphenyl unit | mmol/g | 0.49 | 0.50 | |
| Guaiacyl unit | mmol/g | 1.19 | 1.62 | |
| Syringyl unit | mmol/g | 0.54 | 0.56 | |
| Phenols unit | mmol/g | 2.21 | 2.68 | |
| Carbonyl unit | mmol/g | 0.04 | 0.09 | |
| Molecular weight | Mn1 | g/mol | 1296 | 1073 |
| Mw2 | g/mol | 1627 | 1422 | |
| PDI 3 | - | 1.26 | 1.33 |
| Sample | S.R. [%] | Solid [%] | Liquid [%] | EMB 1 [%] | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Glucan | Xylan | Lignin | Glucose | Xylose | P. Lignin 2 | Glucan | Xylan | Lignin | |||
| Raw RH | 100 | 35.5 | 13.6 | 22.7 | - | - | - | - | - | - | |
| Acid-Free | Fractionated | 71.7 | 46.9 | 12.0 | 20.8 | 1.6 | 4.3 | 2.5 | 99.2 | 94.7 | 76.7 |
| Fractionated 3 | 32.6 | 8.6 | 14.9 | ||||||||
| Component Retention [%] | 91.8 | 63.7 | 65.6 | - | - | - | - | - | - | ||
| Acid Catalyzed | Fractionated | 50.9 | 62.7 | 2.0 | 10.0 | 3.3 | 9.2 | 11.3 | 99.2 | 75.6 | 72.2 |
| Fractionated 3 | 31.9 | 1.0 | 5.1 | ||||||||
| Component Retention [%] | 89.9 | 7.4 | 22.5 | - | - | - | - | - | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, T.H.; Ryu, H.J.; Oh, K.K. Improvement of Organosolv Fractionation Performance for Rice Husk through a Low Acid-Catalyzation. Energies 2019, 12, 1800. https://doi.org/10.3390/en12091800
Kim TH, Ryu HJ, Oh KK. Improvement of Organosolv Fractionation Performance for Rice Husk through a Low Acid-Catalyzation. Energies. 2019; 12(9):1800. https://doi.org/10.3390/en12091800
Chicago/Turabian StyleKim, Tae Hoon, Hyun Jin Ryu, and Kyeong Keun Oh. 2019. "Improvement of Organosolv Fractionation Performance for Rice Husk through a Low Acid-Catalyzation" Energies 12, no. 9: 1800. https://doi.org/10.3390/en12091800
APA StyleKim, T. H., Ryu, H. J., & Oh, K. K. (2019). Improvement of Organosolv Fractionation Performance for Rice Husk through a Low Acid-Catalyzation. Energies, 12(9), 1800. https://doi.org/10.3390/en12091800





