Long-Term Evaluation of Mesophilic Semi-Continuous Anaerobic Digestion of Olive Mill Solid Waste Pretreated with Steam-Explosion
Abstract
1. Introduction
2. Materials and Methods
2.1. Substrate and Inoculum
2.2. Anaerobic Digestion Experimental Procedure
2.3. Chemical Analyses
2.4. Microbial Analysis
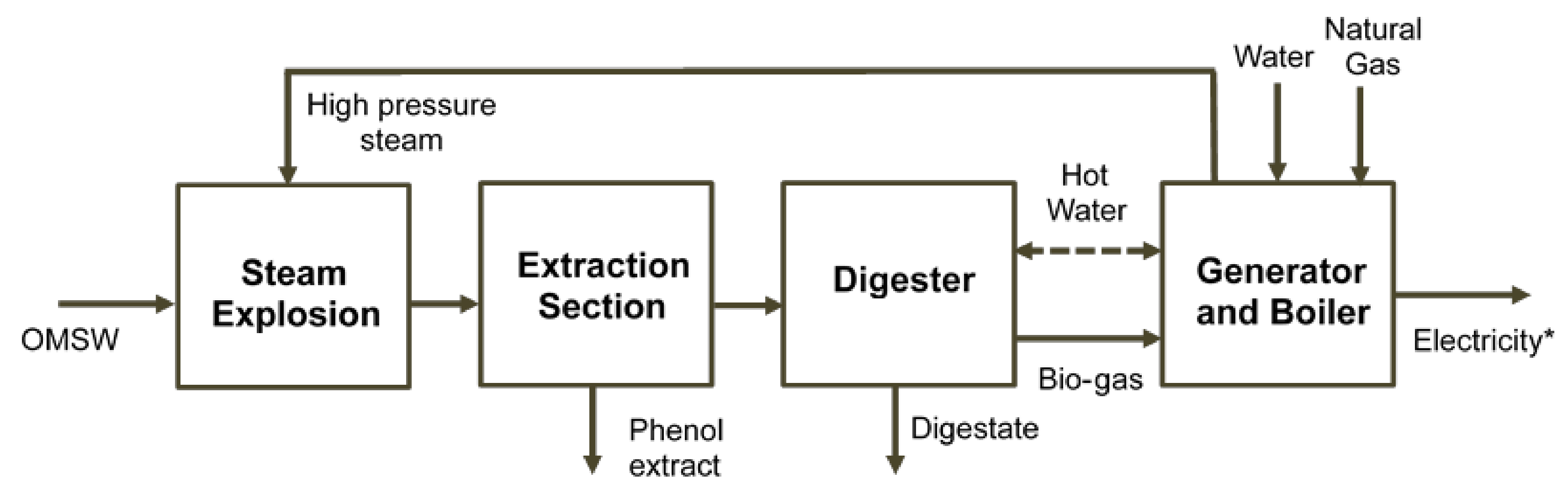
2.5. Economic Assessment
3. Results and Discussion
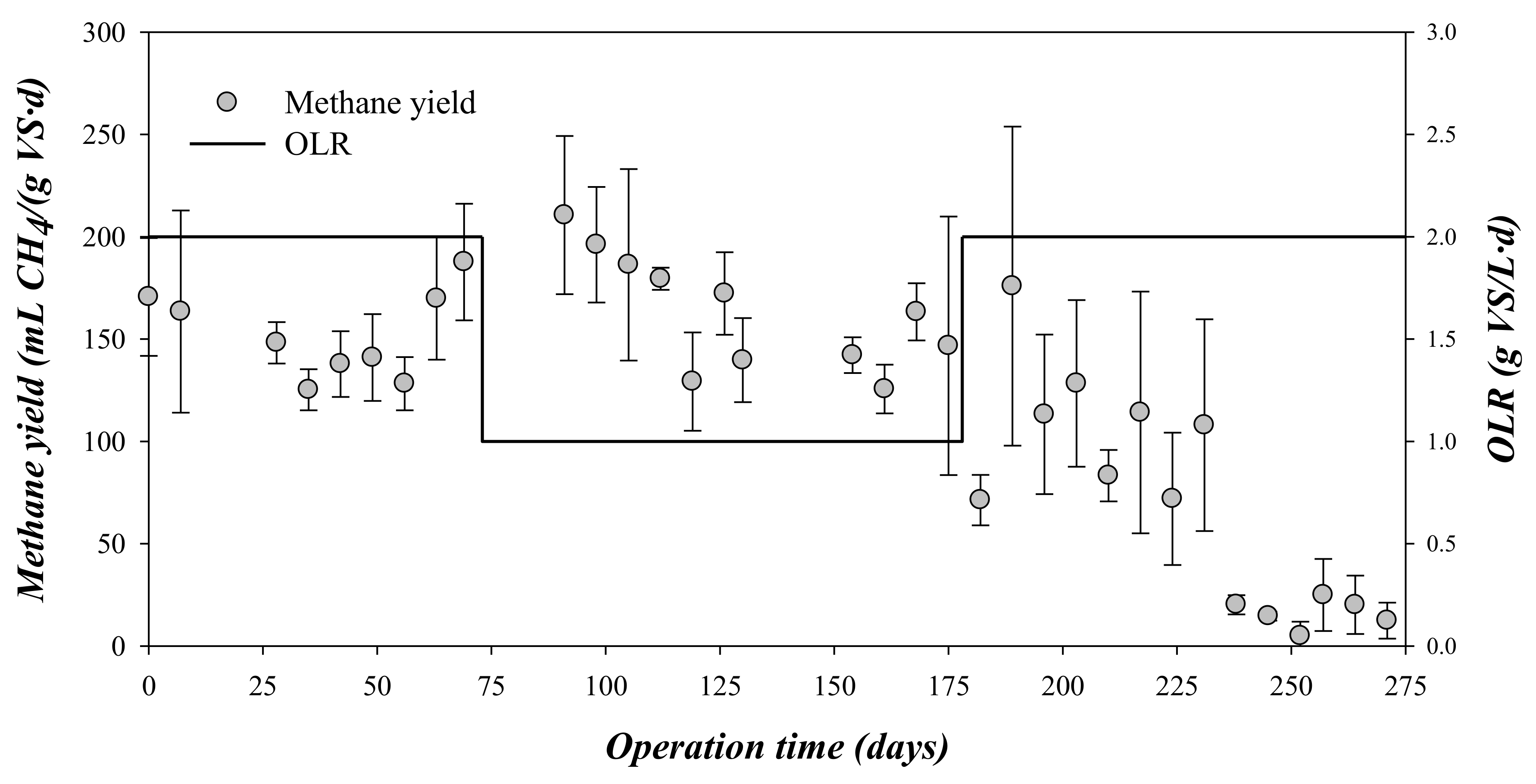
3.1. Methane Production at the Different Experimental Phases
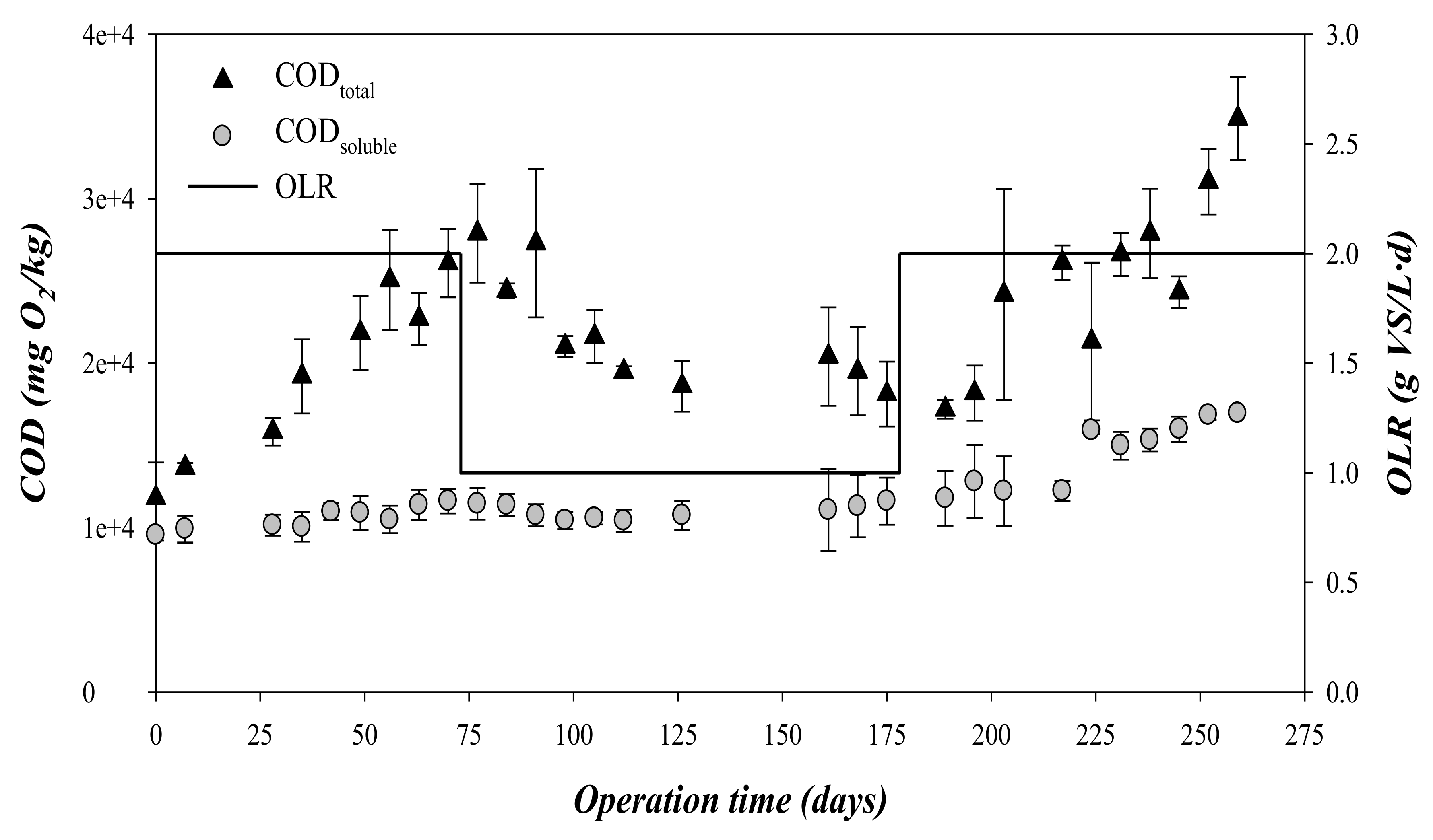
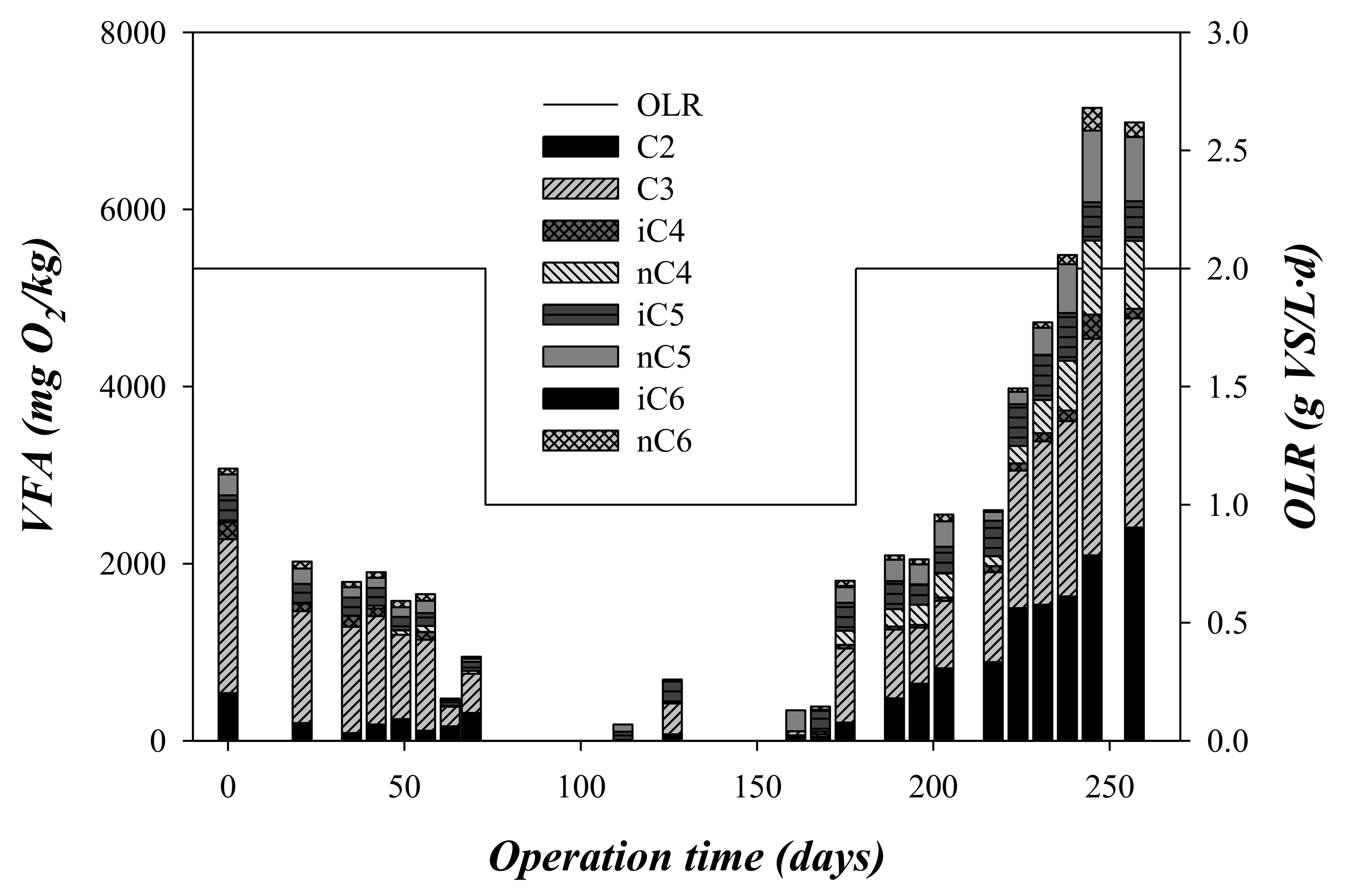
3.2. Chemical Oxygen Demand and Volatile Fatty Acid Concentration throughout the Experimental Time
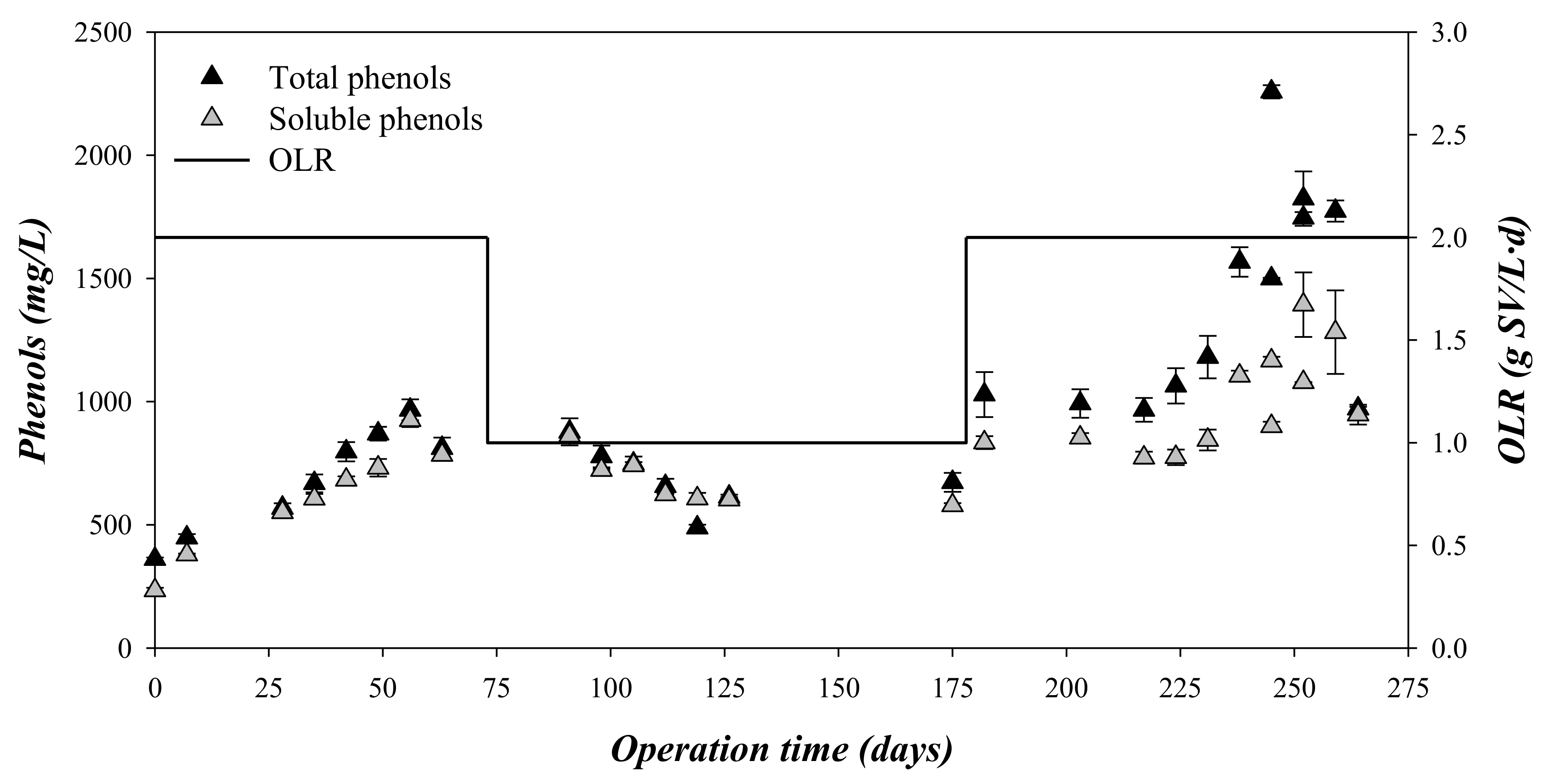
3.3. Composition and Concentration of the Phenolic Compounds
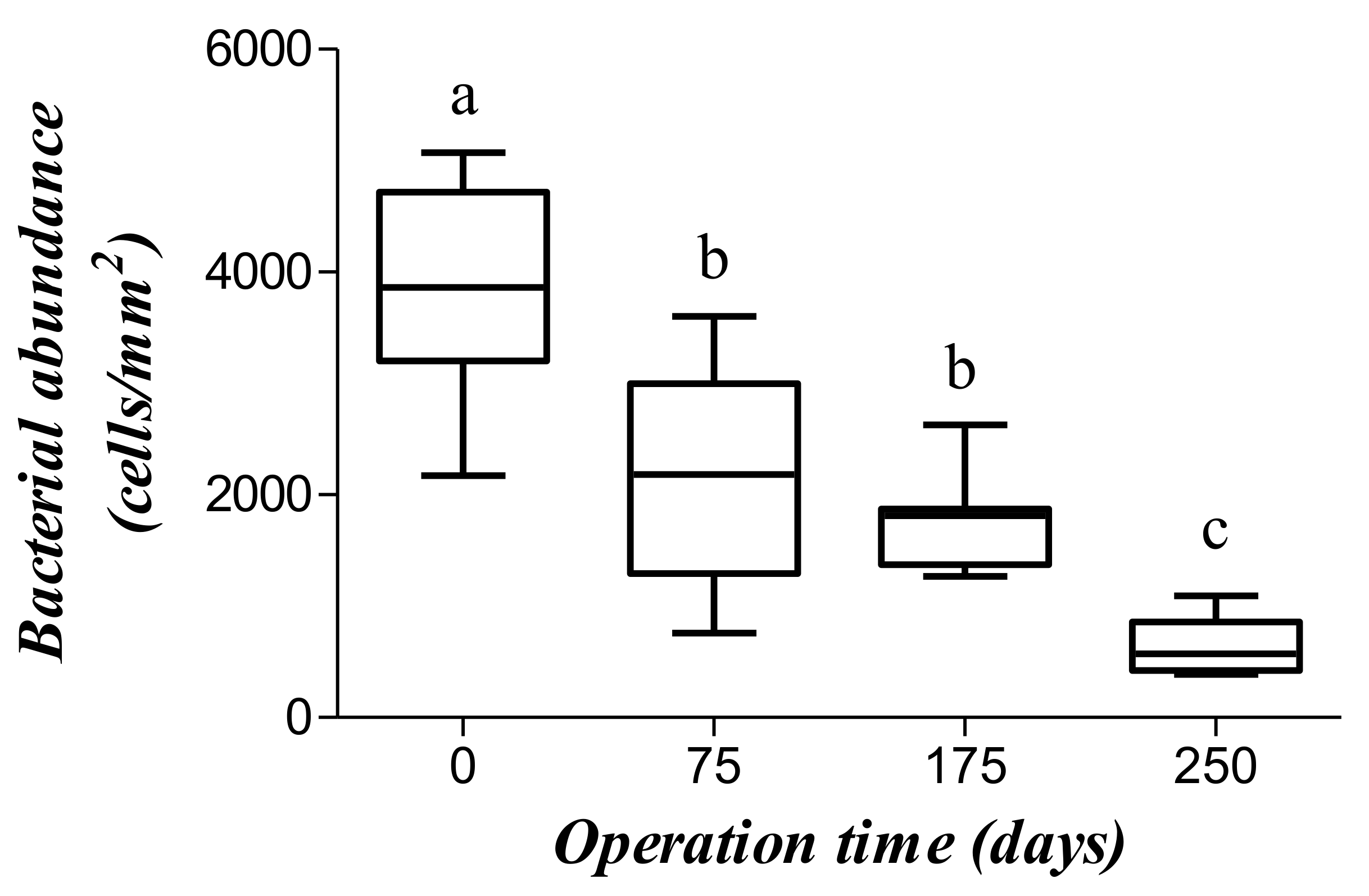
3.4. Microbiology Assessment
3.5. Economic Assessment
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mahmoud, A.E.; Fathy, S.A.; Ali, M.M.; Ezz, M.K.; Mohammed, A.T. Antioxidant and anticancer efficacy of therapeutic bioactive compounds from fermented olive waste. Grasas y Aceites 2018, 69, 266. [Google Scholar] [CrossRef]
- Tortosa, G.; Alburquerque, J.A.; Bedmar, E.J.; Ait-Baddi, G.; Cegarra, J. Strategies to produce commercial liquid organic fertilisers from “alperujo” composts. J. Clean. Prod. 2014, 82, 37–44. [Google Scholar] [CrossRef]
- Serrano, A.; Fermoso, F.G.; Alonso-Fariñas, B.; Rodríguez-Gutierrez, G.; Fernandez-Bolaños, J.; Borja, R. Olive mill solid waste biorefinery: High-temperature thermal pre-treatment for phenol recovery and biomethanization. J. Clean. Prod. 2017, 148, 314–323. [Google Scholar] [CrossRef]
- Rodríguez-Gutiérrez, G.; Lama-Muñoz, A.; Rubio-Senent, F.; Fernández-Bolaños, J.; Ruiz-Méndez, M.V. New Olive-Pomace Oil Improved by Hydrothermal Pre-Treatments; INTECH Open Access Publisher: London, UK, 2012. [Google Scholar]
- García, A.; Rodríguez-Juan, E.; Rodríguez-Gutiérrez, G.; Rios, J.J.; Fernández-Bolaños, J. Extraction of phenolic compounds from virgin olive oil by deep eutectic solvents (DESs). Food Chem. 2016, 197, 554–561. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, G.; Lama, A.; Rodríguez, R.; Jiménez, A.; Guillén, R.; Fernández-Bolaños, J. Olive stone an attractive source of bioactive and valuable compounds. Bioresour. Technol. 2008, 99, 5261–5269. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Senent, F.; Rodríguez-Gutíerrez, G.; Lama-Muñoz, A.; Fernández-Bolaños, J. New phenolic compounds hydrothermally extracted from the olive oil byproduct alperujo and their antioxidative activities. J. Agric. Food Chem. 2012, 60, 1175–1186. [Google Scholar] [CrossRef] [PubMed]
- Hendriks, A.T.W.M.; Zeeman, G. Pretreatments to enhance the digestibility of lignocellulosic biomass. Bioresour. Technol. 2009, 100, 10–18. [Google Scholar] [CrossRef]
- Serrano, A.; Fermoso, F.G.; Alonso-Fariñas, B.; Rodríguez-Gutierrez, G.; Fernandez-Bolaños, J.; Borja, R. Phenols recovery after steam explosion of Olive Mill Solid Waste and its influence on a subsequent biomethanization process. Bioresour. Technol. 2017, 243, 169–178. [Google Scholar] [CrossRef]
- Christoforou, E.; Fokaides, P.A. A review of olive mill solid wastes to energy utilization techniques. Waste Manag. 2016, 49, 346–363. [Google Scholar] [CrossRef]
- Al-Mallahi, J.; Furuichi, T.; Ishii, K. Appropriate conditions for applying NaOH-pretreated two-phase olive milling waste for codigestion with food waste to enhance biogas production. Waste Manag. 2016, 48 (Suppl. C), 430–439. [Google Scholar] [CrossRef]
- Stoyanova, E.; Lundaa, T.; Bochmann, G.; Fuchs, W. Overcoming the bottlenecks of anaerobic digestion of olive mill solid waste by two-stage fermentation. Environ. Technol. 2017, 38, 394–405. [Google Scholar] [CrossRef] [PubMed]
- Serrano, A.; Fermoso, F.G.; Alonso-Fariñas, B.; Rodríguez-Gutiérrez, G.; López, S.; Fernandez-Bolaños, J.; Borja, R. Performance evaluation of mesophilic semi-continuous anaerobic digestion of high-temperature thermally pre-treated olive mill solid waste. Waste Manag. 2019, 87, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Borja, R.; Alba, J.; Banks, C.J. Impact of the main phenolic compounds of olive mill wastewater (OMW) on the kinetics of acetoclastic methanogenesis. Process Biochem. 1997, 32, 121–133. [Google Scholar] [CrossRef]
- Kourmentza, C.; Koutra, E.; Venetsaneas, N.; Kornaros, M. Integrated Biorefinery Approach for the Valorization of Olive Mill Waste Streams towards Sustainable Biofuels and Bio-Based Products. In Microbial Applications Vol.1: Bioremediation and Bioenergy; Kalia, V.C., Kumar, P., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 211–238. [Google Scholar]
- Ghasimi, D.S.M.; Aboudi, K.; de Kreuk, M.; Zandvoort, M.H.; van Lier, J.B. Impact of lignocellulosic-waste intermediates on hydrolysis and methanogenesis under thermophilic and mesophilic conditions. Chem. Eng. J. 2016, 295, 181–191. [Google Scholar] [CrossRef]
- American Public Health Association, American Water Works Association, The Water Environment Federation. Standard Methods for the Examination of Water and Wastewater; APHA-AWWA-WEF: Washington, DC, USA, 2005. [Google Scholar]
- Siritantikorn, S.; Jintaworn, S.; Noisakran, S.; Suputtamongkol, Y.; Paris, D.H.; Blacksell, S.D. Application of ImageJ program to the enumeration of Orientia tsutsugamushi organisms cultured in vitro. Trans. R. Soc. Trop. Med. Hyg. 2012, 106, 632–635. [Google Scholar] [CrossRef] [PubMed]
- Ciriminna, R.; Meneguzzo, F.; Fidalgo, A.; Ilharco, L.M.; Pagliaro, M. Extraction, benefits and valorization of olive polyphenols. Eur. J. Lipid Sci. Technol. 2016, 118, 503–511. [Google Scholar] [CrossRef]
- EUROSTAT. Available online: https://ec.europa.eu/eurostat/web/main/home (accessed on 27 March 2019).
- endesaonline.es. Available online: www.endesaonline.es (accessed on 20 March 2019).
- Orive, M.; Cebrián, M.; Zufía, J. Techno-economic anaerobic co-digestion feasibility study for two-phase olive oil mill pomace and pig slurry. Renew. Energy 2016, 97, 532–540. [Google Scholar] [CrossRef]
- Ortega, L.; Husser, C.; Barrington, S.; Guiot, S.R. Evaluating limiting steps of anaerobic degradation of food waste based on methane production tests. Water Sci. Technol. 2008, 57, 419–422. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Y.; Wang, J.; Meng, L. Effects of volatile fatty acid concentrations on methane yield and methanogenic bacteria. Biomass Bioenergy 2009, 33, 848–853. [Google Scholar] [CrossRef]
- Rincón, B.; Rodríguez-Gutiérrez, G.; Bujalance, L.; Fernández-Bolaños, J.; Borja, R. Influence of a steam-explosion pre-treatment on the methane yield and kinetics of anaerobic digestion of two-phase olive mil solid waste or alperujo. Process Saf. Environ. 2016, 102, 361–369. [Google Scholar] [CrossRef]
- Liu, C.; Yuan, X.; Zeng, G.; Li, W.; Li, J. Prediction of methane yield at optimum pH for anaerobic digestion of organic fraction of municipal solid waste. Bioresour. Technol. 2008, 99, 882–888. [Google Scholar] [CrossRef] [PubMed]
- Borja, R. Biogas Production. In Comprehensive Biotechnology, 2nd. ed; Elsevier: Amsterdam, The Netherlands, 2011; Volume 2, pp. 785–798. [Google Scholar]
- Battista, F.; Fino, D.; Erriquens, F.; Mancini, G.; Ruggeri, B. Scaled-up experimental biogas production from two agro-food waste mixtures having high inhibitory compound concentrations. Renew. Energy 2015, 81 (Suppl. C), 71–77. [Google Scholar] [CrossRef]
- Carrere, H.; Antonopoulou, G.; Affes, R.; Passos, F.; Battimelli, A.; Lyberatos, G.; Ferrer, I. Review of feedstock pretreatment strategies for improved anaerobic digestion: From lab-scale research to full-scale application. Bioresour. Technol. 2016, 199, 386–397. [Google Scholar] [CrossRef]
- Theuerl, S.; Klang, J.; Prochnow, A. Process disturbances in agricultural biogas production—Causes, mechanisms and effects on the biogas microbiome: A review. Energies 2019, 12, 365. [Google Scholar] [CrossRef]
- Alcántara-Hernández, R.J.; Taş, N.; Carlos-Pinedo, S.; Durán-Moreno, A.; Falcón, L.I. Microbial dynamics in anaerobic digestion reactors for treating organic urban residues during the start-up process. Lett. Appl. Microbiol. 2017, 64, 438–445. [Google Scholar] [CrossRef] [PubMed]

| TS | g/L | 72.7 ± 0.8 |
| VS | g/L | 68.4 ± 0.8 |
| COD | g/L | 80.6 ± 2.6 |
| sCOD | g/L | 47.5 ± 0.3 |
| pH | 4.49 ± 0.05 | |
| Alkalinity | mg CaCo3/L | - |
| Acetic acid | mg/L | 1802 ± 8 |
| Total Phenols | mg Gallic Acid/L | 4400 ± 149 |
| Soluble Phenols | mg Gallic Acid/L | 1373 ± 66 |
| 3,4-Dihydroxyphenylglycol | mg/L | 22.0 ± 2.5 |
| Hydroxymethylfurfural | mg/L | 85.1 ± 6.1 |
| Hydroxytyrosol | mg/L | 513.8 ± 25.4 |
| Tyrosol | mg/L | 92.9 ± 14.1 |
| Homovanillic acid | mg/L | 1.16 ± 0.09 |
| Vanillic acid | mg/L | 7.7 ± 0.9 |
| Vanillin | mg/L | 188.7 ± 27.8 |
| 4-Ethylphenol | mg/L | n.d. |
| Experimental Stage | Duration | Feeded Substrate | OLR (gVS/(L·d)) |
|---|---|---|---|
| Bio-stimulation | 15 days | SS | 0.5–1 |
| Adaptation | 15 days | SS:OMSW 75:25, in VS | 2 |
| 15 days | SS:OMSW 50:50, in VS | 2 | |
| 15 days | SS:OMSW 25:75, in VS | 2 | |
| Stage 1 | 75 days (days 0–75) | OMSW | 2 |
| Stage 2 | 100 days (days 75–175) | OMSW | 1 |
| Stage 3 | 100 days (days 175–275) | OMSW | 2 |
| Stage 1 | Stage 2 | Stage 3 | ||
|---|---|---|---|---|
| OLR | gVS/(L*d) | 2 | 1 | 2 |
| Days | 0–75 | 75–175 | 175–275 | |
| Methane production rate | mL CH4/(gVS*d) | 152 ± 21 | 163 ± 28 | 85 ± 59 (from 175 to 8) |
| Methane production rate | gCOD-CH4/(gCOD*d) | 0.369 ± 0.049 | 0.395 ± 0.066 | 0.167 ± 0.126 (from 0.345 to 0.030) |
| pH | 7.81 ± 0.34 | 7.39 ± 0.41 | 6.73 ± 0.58 | |
| Alkalinity | mg CaCO3/L | 3033 ± 894 | 1869 ± 694 | 2704 ± 902 |
| CODt | mgO2/L | 19290 ± 5780 | 21220 ± 3760 | 24890 ± 5500 |
| sCOD | mgO2/L | 4045 ± 1175 | 4010 ± 830 | 8613 ± 2733 (from 4350 to 11765) |
| Total phenols | mg Gallic Acid/L | 686 ± 199 | 697 ± 112 | 1406 ± 413 |
| Soluble phenols | mg Gallic Acid/L | 612 ± 208 | 676 ± 93 | 996 ± 197 |
| VFA | mgO2/L | 1682 ± 718 | 683 ± 586 | 4180 ± 1907 |
| C2 | mgO2/L | 230 ± 135 | 80 ± 67 | 1334 ± 629 |
| C3 | mgO2/L | 1010 ± 450 | 251 ± 318 | 1486 ± 670 |
| iC4 | mgO2/L | 71 ± 64 | 16 ± 17 | 94 ± 70 |
| nC4 | mgO2/L | 24 ± 24 | 32 ± 64 | 294 ± 250 |
| iC5 | mgO2/L | 172 ± 76 | 180 ± 117 | 406 ± 97 |
| nC5 | mgO2/L | 119 ± 64 | 99 ± 95 | 376 ± 242 |
| iC6 | mgO2/L | < D.L. | 5 ± 6 | <D.L. |
| nC6 | mgO2/L | 57 ± 22 | 18 ± 24 | 92 ± 71 |
| Experimental Stage | Start | End Stage 1 | End Stage 2 | End Stage 3 |
|---|---|---|---|---|
| Day | 0 | 63 | 175 | 275 |
| OLR (gVS/(L*d)) | - | 2 | 1 | 2 |
| 3,4-Dihydroxyphenylglycol | 0.35 ± 0.04 | 0.08 ± 0.01 | Traces | 0.62 ± 0.05 |
| Hydroxymethylfurfural | n.d. | n.d. | n.d. | n.d. |
| Hydroxytyrosol | 3.90 ± 0.17 | 3.49 ± 0.05 | 3.87 ± 0.55 | 10.3 ± 2.52 |
| Tyrosol | n.d. | n.d. | n.d. | 9.9 ± 0.4 |
| Homovanillic acid | n.d. | n.d. | n.d. | n.d. |
| Vanillic acid | n.d. | n.d. | n.d. | 1.3 ± 0.4 |
| Vanillin | 48.1 ± 6.9 | 30.8 ± 11.8 | 25.6 ± 2.1 | 149 ± 55 |
| 4-Ethylphenol | 23.2 ± 2.9 | 141 ± 33 | 108 ± 6 | 136 ± 15 |
| Methane Production (Nm3/year) | Initial Investment Cost (€) | Incoming (€/year) | Annual Costs (€/year) a | NPV (M€) | IRR (%) | Payback Period (years) | Limit Phenol Price (€/kg) b |
|---|---|---|---|---|---|---|---|
| 2,951,115 | 28,367,480 | 330,066,600 | 28,277,122 | 3.9·103 | 1011 | 1 | 42.2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serrano, A.; Fermoso, F.G.; Alonso-Fariñas, B.; Rodríguez-Gutiérrez, G.; López, S.; Fernandez-Bolaños, J.; Borja, R. Long-Term Evaluation of Mesophilic Semi-Continuous Anaerobic Digestion of Olive Mill Solid Waste Pretreated with Steam-Explosion. Energies 2019, 12, 2222. https://doi.org/10.3390/en12112222
Serrano A, Fermoso FG, Alonso-Fariñas B, Rodríguez-Gutiérrez G, López S, Fernandez-Bolaños J, Borja R. Long-Term Evaluation of Mesophilic Semi-Continuous Anaerobic Digestion of Olive Mill Solid Waste Pretreated with Steam-Explosion. Energies. 2019; 12(11):2222. https://doi.org/10.3390/en12112222
Chicago/Turabian StyleSerrano, Antonio, Fernando G. Fermoso, Bernabé Alonso-Fariñas, Guillermo Rodríguez-Gutiérrez, Sergio López, Juan Fernandez-Bolaños, and Rafael Borja. 2019. "Long-Term Evaluation of Mesophilic Semi-Continuous Anaerobic Digestion of Olive Mill Solid Waste Pretreated with Steam-Explosion" Energies 12, no. 11: 2222. https://doi.org/10.3390/en12112222
APA StyleSerrano, A., Fermoso, F. G., Alonso-Fariñas, B., Rodríguez-Gutiérrez, G., López, S., Fernandez-Bolaños, J., & Borja, R. (2019). Long-Term Evaluation of Mesophilic Semi-Continuous Anaerobic Digestion of Olive Mill Solid Waste Pretreated with Steam-Explosion. Energies, 12(11), 2222. https://doi.org/10.3390/en12112222









