Abstract
Biogas is an alternative source of energy for fossil fuels. In the process of transforming organic materials into biogas significant amounts of valuable digestate are produced. In order to make the whole process sustainable digestate should be utilized this is a constraining factor in the development of the biogas industry. Consequently, there is an on-going search for new technologies to process digestate, allowing to broaden the range of possible ways of digestate utilization. One of such possibilities is technology of nitrogen (N) and phosphorus (P) recovery from the anaerobic digestate. In this study results of physicochemical analysis of materials flowing through the farm-scale bio-refinery producing struvite (STR) and ammonium sulphate (AS) are presented. Struvite was precipitated from the liquid fraction of digestate (LFDS). Ammonia was bound by sulphuric acid resulting in obtaining ammonium sulphate. The STR obtained was of medium purity and contained other macronutrients and micronutrients that further enhanced its agronomic value. The P recovery effectiveness, counted as the difference between the Ptot content in the material before and after STR precipitation was 43.8%. The AS was characterized by relatively low Ntot and Stot content. The Ntot recovery efficiency reached 43.2%. The study showed that struvite precipitation and ammonia stripping technologies can be used for processing digestate however, the processes efficiency should be improved.
1. Introduction
The progressing concentration of animal production causes a growing problem of environmental pollution. The problem is so serious that in some European countries systemic solutions began to be introduced forcing farmers to implement technologies aimed at the export of nitrogen and phosphorus (P) from farms [1]. This is exemplified by Dutch agriculture [2]. Due to a large excess of phosphorus occurring in farms with intensive livestock production, the Dutch government introduced legislation to limit the amount of phosphate used in agriculture. Farmers are eligible for state-founded payments when they exported phosphorus from their farm.
Proper allocation of nitrogen (N) and P from manure between areas with excess of the components and those where their deficits are supplemented with mineral fertilizers will provide measurable benefits in the form of limiting water eutrophication and reduction of N2O emissions from the soil [3]. The closing of the N and P cycle by recovering and recycling can be implemented through farm expansion and creating a system of the organic materials treatment consisting in the struvite (STR) (MgNH4PO4·6H2O) precipitation and subsequent stripping of ammonia. The obtained products: STR and ammonium sulphate (AS) can be used as substitutes for currently used artificial fertilizers which corresponds with the idea of the circular bioeconomy. One of its objectives is to increase efficiency of natural resources use and to decrease the dependence European agriculture on imports of raw materials necessary for fertilizer production [4]. This implies a necessity to search for new N and P sources for fertilizer use [5].
Nowadays anaerobic digestion has become an increasingly important source of renewable energy. During the anaerobic digestion, organic compounds are subject to degradation, leading to the accumulation of mineral forms of N and P in the remaining digestate [6,7,8]. Digestate is usually subject to solid-liquid separation [9]. The solid fraction is applied on fields managing large amounts of liquid fraction, which contains 3–5% dry matter is difficult to achieve [10]. Further processing of the liquid fraction of digestate permits a recovery of nutrients. They can be exported from the farms over large distances to places where the soil is deficient in these nutrients [11,12]. This allows for obtaining additional income from the sale of the obtained product [13]. It is possible to apply several technologies of the digestate liquid fraction processing, such as struvite precipitation, ammonia stripping, evaporation, or membrane separation, resulting in the nutrient recovery and production of nutrient-rich products [14,15,16,17]. Struvite precipitation and ammonia stripping are promising methods for recovering the nitrogen and phosphorus from the liquid fractions of digestate. However, these processes are still at an experimental level [18]. According to Rahaman et al. [19], equi-molar concentration (1:1:1) of magnesium (Mg), phosphorus (P) and ammonium (NH4+) with alkaline (optimal pH value 8.5–9.0) are required to precipitate struvite. Depending on the chemical reaction conditions, struvite may be formed at reactants ratios different from the optimal molar ones [20,21,22]. Czajkowska and Siwiec [23] proved that at pH ≤ 7.5 struvite precipitates from a solution at the concentration of substrates: ≥300 mg PO43−·kg−1, ≥250 mg NH4+·kg−1 and ≥111.5 mg Mg·kg−1. Results of the cited research indicate that increase in phosphate concentration up to 1000 mg PO43−·kg−1 allows crystallization of struvite with a lower content of other substrates (49.4 mg Mg·kg−1 and 100 mg NH4+·kg−1). However, a decrease in the content of one substrate must be accompanied by an increase in the concentration of the other substrate. At pH 8.0, struvite does not crystallize from the solution at a concentration of 100 mg PO43−·kg−1, <200 mg NH4+·kg−1 and <113 mg Mg·kg−1. However, in the pH range 8.5–9.0, the ion concentrations that do not allow crystallization from the solution are as follows: 200 mg PO43−·kg−1, <100 mg NH4+·kg−1 and <48 mg Mg·kg−1 [23]. A potentially high content of competing elements can inhibit struvite crystallization. The content of potassium (K) [18], calcium (Ca) [17], and organic compounds [24] could decrease the N and P removal efficiency from the liquid fraction of digestate. The ammonia stripping process depends on pH, temperature and mass transfer area. The process is typically conducted at a temperature higher than 20 °C and pH above 9 [25]. Guštinet et al. [26] reached high efficiency of N removal (approximately 93%) from liquid fraction of digestate at pH 10.5 and temperature of 50 °C. The remaining material could still contain nutrients. However, it can be used in the agriculture after pH neutralization [25]. An alternative method is cultivation of duckweed. Oron et al. [27] used duckweed for domestic wastewater treatment to recycle the nutrients. Additionally production of duckweed is aimed at the recovery of nutrients, especially ammonium and phosphorus, from swine lagoon water [28]. Duckweed is well known for its high productivity and high protein content, ranging between 15% and 45% of the dry mass [29,30]. Considering the above, production of duckweed can substitute imported fodders and therefore limit the inflow of another portion of nitrogen and phosphorus within protein-rich fodders to the farm.
Thus far N and P has been recovered mainly from municipal sludge digester effluent and swine wastewater [17]. Moreover, the growing problem of point accumulation of these nutrients in digestate from biogas plants encourages the undertaking of research aiming at N and P recovery.
The aim of the study was to assess the possibility of using digestate processing technology in biogas plants, designed for N and P recovery and production of innovative substances that could be transported over long distances and used as mineral fertilizers.
2. Materials and Methods
2.1. The Farm Biogas Plant Scheme
The bio-refinery was located at the Experimental Dairy Farm ‘De Marke’ in Hengelo (Gld), the Netherlands (52°03″N, 6°18″E). In the bio-refinery (Figure 1), cattle slurry (S) was subject to anaerobic digestion (1), and the obtained biogas was used to produce energy (2). The CHP (Combined Heat and Power) installation consisted of a gas engine (MAN E0824 E302) and generator. The electrical capacity is 36 kWe and thermal capacity is 51 kWth. The produced heat is being used to maintain the temperature of the digestion process. Electricity is being used to cover the own electricity consumption at the Experimental Dairy Farm De Marke and the surplus is being sold over the public electricity grid.
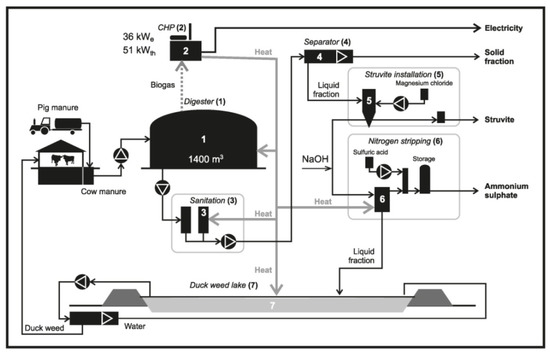
Figure 1.
Scheme of the integrate bio-refinery system on farm scale.
The remaining digestate (DS) after sanitation (3) was separated into the solid (SFDS) and liquid fractions (LFDS) (4). Digestate was hygienized in order to reduce the amount of pathogens and to prevent infections. In this case the standard method of sanitation was used—pasteurization, which involves keeping the material at 70 °C for at least 60 min. The first stage of LFDS processing was struvite (STR) precipitation (5). MgCl2 was added to the LFDS as a source of magnesium. Moreover, the pH of LFDS was adjusted to 9 by means of continuous 5N NaOH addition. At the second stage, ammonium sulphate (AS) was produced at 70 °C in the ammonia stripping installation: H2SO4 vessel, stripping tower and reactor (6). in order to increase the N recovery efficiency from the liquid fraction of digestate. (6). The liquid fraction after STR precipitation was alkalised by means of 5N NaOH, reaching a pH of 10.5. In such conditions, the released ammonium was bound by sulphuric acid resulting in obtaining AS. At the third stage, the effluent (EFL) after ammonia stripping was directed to the duckweed pond, where duckweed was cultivated (7). The produced duckweed served as fodder for cattle on the farm. The concept of our installation assumes that the duckweed will be kept in the pond and will absorb the nutrients until they reach a level that is acceptable for dumping the pond water to the surface waters.
2.2. Analytical Methods
The samples were collected in 2017. The chemical analysis of the S, DS, LFDS, SFDS, EFL, STR and AS was carried out in triplicate. The S, DS, LFDS, SFDS and EFL samples were dried out at 60°C using drier PREMED (Marki, Poland) to estimate Total Solids content (TS). The Volatile Solids (VS) content was measured in accordance with the Polish Standard PN-Z-15011-3:2001 after roasting samples at 550 °C in a muffle furnace model FCF 12SHM (Czylok, Jastrzębie Zdrój, Poland). The pH value was measured by the potentiometric method using a pH meter (Schott, Mainz, Germany). The total N content (Ntot) was measured by means of the Kjeldahl method using a Vapodest analyzer model VAP 30 (Gerhardt, Bonn, Germany). The NH4+-N content was measured using the Skalar San Plus analyzer (Breda, Netherlands), after wet basis samples extraction in 0.01 mol·dm−3 CaCl2 with sample/extractant ratio of 1:10. The total phosphorus (Ptot), potassium (Ktot), magnesium (Mgtot), calcium (Catot), sodium (Natot), zinc (Zn), manganese (Mn), copper (Cu) and iron (Fe) content were determined after digestion in HNO3 and HClO4 acids using a Model DK 20 digestion unit (VELP Scientifica, Usmate, Italy). Ktot, Mgtot, Catot, Natot, Zn, Mn, Cu and Fe were measured by AAS (Atomic Absorption Spectrometry) method using a spectrometer SOLAAR (Thermo Elemental, Cambridge, UK). The total content of sulphur (Stot) was measured by inductively coupled plasma atomic emission spectrometry (ICP-AES) (IRYS Advantage ThermoElementar, Cambridge, UK). Ptot was determined by the molybdenum-blue ascorbic method using the spectrophotometer Genesys 10 UV-VIS (Thermo Electron Corporation, Madison, USA). Water-soluble forms of Pws, Mgws and Caws were determined in water/the samples solution with ratio of 1:100. The crystal morphology of the STR deposit was observed using an optical polarization microscope (Model MN-800, POL OPTA-TECH) with a 5MP digital camera (OPTA-TECH) and Opta View software. X-ray Diffraction (XRD) measurements were performed on a diffractometer by PANalyticalEmpyreanin Bragg-Brentano geometry equipped with a PIXcel3D detector. The measurements were performed on a Cu lamp with parameters I = 40 mA and U = 40 kV. The analysis of the diffractogram was performed in HighScorePlus software.
2.3. Statistical Analysis
Statistical analysis was carried out using the Statistica PL 13.0 software (Tulsa, USA). The data were subjected to ANOVA and the differences between the means were determined according to Tukey’s test (P < 0.05).
3. Results and Discussion
The total content of studied macronutrients was similar in raw (S) and digestated slurry (DS) (Table 1). After anaerobic digestion the relation between total and water soluble forms of nutrients changed, especially concerning P. In the S the content of Pws was 41.6 mg·kg−1 WB, whereas in the DS it was 212.6 mg·kg−1 WB. It constituted of 10.6% and 56.8% of Ptot, respectively. After mechanical DS separation, the remaining SFDS contained a significantly higher content of Ntot, Ptot, Ktot, Mgtot and Catot compared to the LFSD. However, the contribution of water-soluble nutrients in total forms was higher in the LFDS (Table 1).

Table 1.
The physicochemical properties of organic materials flowing through the farm scale bio-refinery.
The chemical composition of LFDS was not optimal for struvite precipitation. In the LFSD, the content of NH4-N was 3000.0 mg·kg−1 WB, Pws 183.0 mg·kg−1 WB and Mgws 239.4 mg·kg−1 WB (Table 1). According to Kataki et al. [31] and Jia et al. [32], the highest effectiveness of struvite precipitation is achieved in solutions where pH is 9.0, whereas the pH of LFSD was 7.8; thus in order to increase the pH NaOH, was added. STR crystals obtained in the bio-refinery were very small. Their linear dimensions reached 1400 µm (Figure 2). They were colored brown, which indicated the presence of organic pollutants. The content of organic matter in the STR was estimated for 51.3 g VS kg−1. Additionally Achat et al. [33] found the presence of organic matter between 19.3 and 165.0 g·kg−1 in recycled P-products.

Figure 2.
Microscopic image of the struvite precipitated in the farm scale bio-refinery (STR).
The applied X-ray Diffraction (XRD) analysis allowed for the determination of the type of crystalline phases occurring in the sample (Figure 3). The analyzed sample is characterized by well-formed crystallographic phases. The presence of the struvite crystallographic phase is clearly evidenced by a series of characteristic diffraction lines with the highest intensities at 2θ 15.00 (hkl 110), 15.81 (hkl 020), 16.47 (hkl 011), 20.85 (100% intensity, hkl 111), 21.45 (hkl 021) and 30.60 (hkl 211). In addition to struvite, monohydroxycalcite (CaCO3·H2O) and quartz (SiO2) were found.
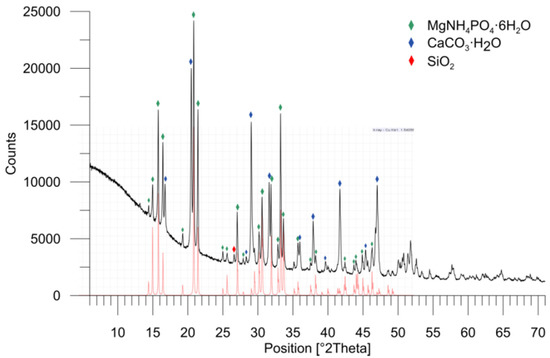
Figure 3.
X-ray diffraction pattern of the product from the bio-refinery with struvite (STR) evidence: struvite (MgNH4PO4·6H2O)-green, monohydroxycalcite (CaCO3·H2O)-blue, and quartz (SiO2)-red; the reference diffractogram of struvite is shown below the diffractogram of the analyzed sample.
Organic and mineral pollutants detected in the STR resulted in a low content of main nutrients (23.1 g Ntot·kg−1 WB, 39.4 g Ptot·kg−1 WB and 32.3 g Mgtot·kg−1 WB) (Figure 4 and Figure 5). Much higher nutrient content occurs in the struvite fertilizer Crystal Green®, Ostara: 50.0 g Ntot·kg−1; 122.0 g Ptot·kg−1, and 100.0 g Mgtot·kg−1, produced from sewage sludge sediment [34,35]. However, struvite composition depends on different factors; particularly, on the characteristics of raw materials [36]. According to Kataki et al. [31], struvite from yeast anaerobic effluent contained 35 g N·kg−1, 108 g P·kg−1 and 79.7 g Mg·kg−1, from municipal wastewater 29 g N·kg−1, 91 g P·kg−1 and 99 g Mg·kg−1 and from human urine 29 g N·kg−1 and 71 g P·kg−1.
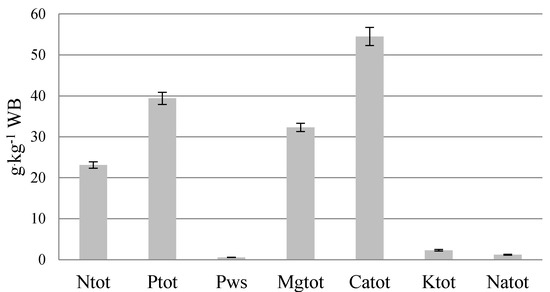
Figure 4.
Macronutrients content in STR from the farm scale bio-refinery plant.
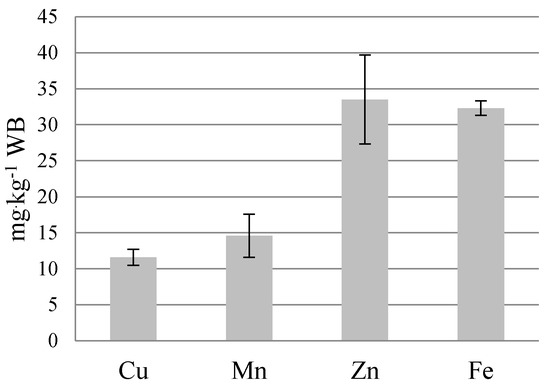
Figure 5.
Micronutrients content in STR from the farm scale bio-refinery.
The P recovery efficiency, calculated as the difference between the Ptot content in the LFSD and the EFL, reached 43.8%. Low P recovery efficiency could be caused by the Ca, K, Na, Cu, Mn and Zn content in the LFSD (Table 1) as these elements are considered to be STR precipitation inhibitors [18]. Moreover, the total solids in the LFSD could hinder STR precipitation. Taddeo et al. [18] proved a strong correlation between the content of total solids in raw materials used to struvite precipitation and process efficiency.
The total ammonia nitrogen (sum of NH3 and NH4+), which remained in the effluent after STR precipitation, was bound to (NH4)2SO4 by sulphuric acid in the ammonia stripping process. The obtained AS was characterized by a low content of Ntot (14.1 g·kg−1 WB) and Stot (16.8 g·kg−1 WB). The content of pure (NH4)2SO4 was only 66.0 g·kg WB (Table 2). In the research conducted by Laureni et al. [37], the ammonium sulphate solution obtained from pig manure contained 400.0–600.0 g·kg−1 (NH4)2SO4. A low AS pH (2.1) can affect the reduction of its fertilizer value. It is possible to increase AS pH value using CaCO3 [38] or caustic magnesite [39]. The N recovery efficiency, calculated as the difference between the Ntot content in the LFSD and the EFL, was 43.2%. Depending on flow rate and alkalinity, the total ammonia removal from the digestate might reached 42–80% [40].

Table 2.
Composition of ammonium sulphate (AS) from the farm scale bio-refinery.
4. Conclusion
The disadvantage of biogas production being a renewable energy source is the remaining significant amount of digestate. Because of its constraints, limiting potential use of digestate as a natural fertilizer spread on agricultural land, there is a search for technologies allowing further treatments of digestate to make exporting beyond farms where biogas is processed to energy possible. The digestate can be a substrate for the production of valuable mineral fertilizers. Considering problems of exhaustion of phosphate rock reserve the construction of a bio-refinery recovering N and P as struvite and ammonium sulphate from digestate seems to be fully justified—both may substitute commercial N-P or N fertilizers. The results indicate that the products obtained in the bio-refinery contain smaller amounts of nutrients compared to mineral fertilizers that reduce their fertilizing value. However, besides the main nutrients (N, P, Mg), recovered, struvite contains organic matter and other macro- and micronutrients important for plant growth (K, Ca, Na, Cu, Mn, Zn, Fe) that increase fertilizing value of struvite. Biogas plants can be transformed into a bio-refinery using innovative processes; however, it is necessary to improve nutrient recovery efficiency. Further vegetation experiments are required in order to determine the fertilizer value and optimal doses and methods of application of these recycled products considering their specific properties.
Author Contributions
Conceptualization, G.W.P.v.P., R.L.C., M.S., and E.S.; Methodology, G.W.P.v.P., R.L.C., M.S., E.S., M.B. and M.K.; Formal Analysis, M.S., E.S., A.W. and T.S.; Investigation, M.S., E.S., and M.B.; Writing-Original Draft Preparation, M.S.; Writing-Review & Editing, T.S., E.S. and A.W.; Visualization, M.S.; Project Administration, G.W.P.v.P., R.L.C. and A.W.
Funding
This research was funded by the National Centre for Research and Development. Contracts No: BIOENERGY/Ct°Cfarming/02/2016 and BIOENERGY/Ct°Cfarming/03/2016. Project ”Cradle to cattle farming”.
Acknowledgments
The authors are grateful to Cornelissen Consulting Services Services BV, ProfiNutrients BV, and Pieter Teeuwen for the valuable collaboration during this project.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| TS | Total Solids |
| WB | Wet Basis |
| S | Slurry |
| DS | Digested slurry |
| SFDS | Solid fraction of digested slurry |
| LFDS | Liquid fraction of digested slurry |
| EFL | Effluent after struvite precipitation and ammonia stripping |
| STR | Struvite from bio-refinery |
| AS | Ammonium sulphate |
References
- Amery, F.; Schoumans, O.F. Agricultural Phosphorus Legislation in Europe. Available online: https://pure.ilvo.be/portal/files/2640562/Phosphorus_legislation_Europe.pdf. (accessed on 12 September 2018).
- Rozemeijer, J.C.; Klein, J.; Broers, H.P.; van Tol-Leenders, T.P.; Van Der Grift, B. Water quality status and trends in agriculture-dominated headwaters; a national monitoring network for assessing the effectiveness of national and European manure legislation in The Netherland. Environ. Monit. Assess. 2014, 186, 8981–8995. [Google Scholar] [CrossRef] [PubMed]
- Sosulski, T.; Szara, E.; Szymańska, M.; Stępień, W. N2O emission and nitrogen and carbon leaching from the soil in relation to long-term and current mineral and organic fertilization-a laboratory study. Plant Soil Environ. 2017, 63, 97–104. [Google Scholar] [CrossRef]
- Circular Economy Package Proposal for a Regulation of the European Parliament and of The Council Laying Down Rules on the Making Available on the Market of CE Marked Fertilizing Products and Amending Regulations (EC) No 1069/2009 and (EC) No 1107/2009. Available online: https:// ec.europa.eu/transparency/regdoc/rep/1/2016/EN/1-2016-157-EN-F1-1.PDF (accessed on 11 November 2018).
- Chien, S.H.; Prochnow, L.I.; Tu, S.; Snyder, C.S. Agronomic and environmental aspects of phosphate fertilizers varying in source solubility: an update review. Nutr. Cycl. Agroecosys. 2010, 89, 229–255. [Google Scholar] [CrossRef]
- Lošák, T.; Hlušek, J.; Válka, T.; Elbl, J.; Vítěz, T.; Bělíková, H.; Von Bennewitz, E. The effect of fertilisation with digestate on kohlrabi yields and quality. Plant Soil Environ. 2016, 62, 274–278. [Google Scholar] [CrossRef]
- Szymańska, M.; Szara, E.; Sosulski, T.; Wrzosek-Jakubowska, J.; Gworek, B. Changes in the chemical properties of solid fraction of digestate from biogas plant during co-composting. Przem. Chem. 2016, 95, 577–580. (In Polish) [Google Scholar] [CrossRef]
- Tampio, E.; Salo, T.; Rintala, J. Agronomic characteristic of five different urban waste digestates. J. Environ. Manage. 2016, 169, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Szymańska, M.; Nowaczewska, D.; Świerżewska, E.; Wrzosek-Jakubowska, J.; Gworek, B. An attempt to assess physicochemical properties of soil fertilized with fresh and treated digestate from biogas plant. Przem. Chem. 2016, 95, 572–576. (In Polish) [Google Scholar] [CrossRef]
- Szymańska, M.; Sosulski, T.; Szara, E.; Pilarski, K. Conversion and properties of anaerobic digestate from biogas production. Przem. Chem. 2015, 94/8, 419–1423. (In Polish) [Google Scholar] [CrossRef]
- Rehl, T.; Müller, J. Life cycle assessment of biogas digestate processing technologies. Resour. Conserv. Recycl. 2011, 56, 92–104. [Google Scholar] [CrossRef]
- Bolzonella, D.; Fatone, F.; Gottardo, M.; Frison, N. Nutrients recovery from anaerobic digestate of agro-waste: Techno-economic assessment of full scale applications. J. Environ. Manage. 2018, 216, 111–119. [Google Scholar] [CrossRef]
- Fuchs, W.; Drosg, B. Assessment of the state of the art of technologies for the processing of digestate residue from anaerobic digester. Water Sci. Technol. 2013, 67, 1984–1993. [Google Scholar] [CrossRef] [PubMed]
- Ek, M.; Bergström, R.; Bjurhem, J.E.; Björlenius, B.; Hellström, D. Concentration of nutrients from urine and reject water from anaerobically digested sludge. Water Sci. Technol. 2006, 54, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Chiumenti, A.; da Borso, F.; Chiumenti, R.; Teri, F.; Segantin, P. Treatment of digestate from a co-digestion biogas plant by means of vacuum evaporation: tests for process optimization and environmental sustainability. Waste Manage. 2013, 33, 1339–1344. [Google Scholar] [CrossRef] [PubMed]
- Ledda, C.; Schievano, A.; Salati, S.; Adani, F. Nitrogen and water recovery from animal slurries by a new integrated ultrafiltration, reverse osmosis and cold stripping process: A case study. Water Res. 2013, 47, 6165–6166. [Google Scholar] [CrossRef] [PubMed]
- Tao, W.; Fattah, K.P.; Huchzermeier, P. Struvite recovery from anaerobically digested dairy manure: A review of application potential and hindrances. J. Environ. Manage. 2016, 169, 46–57. [Google Scholar] [CrossRef] [PubMed]
- Taddeo, R.; Honkanen, M.; Kolppo, K.; Lepisto, R. Nutrient management via struvite precipitation and recovery from various agroindustrial wastewaters: Process feasibility and struvite quality. J. Environ. Manage. 2018, 212, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Rahaman, M.S.; Ellis, N.N.; Mavinnic, D.S. Effects of various process parameters on struvite precipitation kinetics and subsequent determination of rate constants. Water Sci. Technol. 2008, 57, 647–654. [Google Scholar] [CrossRef]
- Li, B.; Boiarkina, I.; Yu, W.; Huang, H.M.; Munir, T.; Wang, G.Q.; Young, B.R. Phosphorus recovery through struvite cristalization: Challenge for future design. Sci. Total Environ. 2019, 648, 1244–1256. [Google Scholar] [CrossRef]
- Saidou, H.; Korchef, A.; Moussa, S.B.; Amor, M.B. Struvite precipitation by the dissolved CO2 degasification technique: Impact of the airflow rate and pH. Chemosphere 2009, 74, 338–343. [Google Scholar] [CrossRef]
- Le Corre, K.S.; Valsami-Jones, E.; Hobbs, P.; Parsons, S.A. Phosphorus recovery from wastewater by struvite crystallization: a review. Crit. Rev. Env. Sci. Technol. 2009, 39, 433–477. [Google Scholar] [CrossRef]
- Czajkowska, J.; Siwiec, T. Crystalization of struvite in change conditions pH index and concentrations analysed components. Infrastructure and Ecology of Rural Areas 2011, 7, 145–154. [Google Scholar]
- Nelson, N.O.; Mikkelsen, R.L.; Hesterberg, D.L. Struvite precipitation in anaerobic swine lagoon liquid: effect of pH and Mg:P ratio and determination of rate constants. Bioresour. Technol. 2003, 89, 229–236. [Google Scholar] [CrossRef]
- Limoli, A.; Langone, M.; Andreottola, G. Ammonia removal from Raw manure digestate by means of a turbulent mixing stripping process. J. Environ. Manage. 2016, 176, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Guštin, S.; Marinšek Logar, R. Effect of pH, temperature and air flow rate on the continuous ammonia stripping of the anaerobic digestion effluent. Process Saf. Environ. Prot. 2011, 89, 61–66. [Google Scholar] [CrossRef]
- Oron, G.; de-Vegt, A.; Porath, D. Nitrogen removal and conversion by duckweed grown on wastewater. Water Res. 1988, 22, 179–184. [Google Scholar] [CrossRef]
- Cheng, J.; Bergmann, B.A.; Classen, J.J.; Stomp, A.M.; Howards, J.W. Nutrient recovery from swine lagoon water by Spirodelapunctata. Bioresour. Technol. 2002, 81, 81–85. [Google Scholar] [CrossRef]
- Landolt, E. The Family of Lemnaceae-A Monographic Study, vol 1-Biosystematic Investigations in the Family of Duckweeds (Lemnaceae), Vol 2; Veröffentlichungen des Geobotanischen Institutes of the ETH, Stiftung Rubel: Zürich, Switzerland, 1986. [Google Scholar]
- Ozengin, N.; Elmaci, A. Performance of Duckweed (Lemna minor L.) on different types of wastewater treatment. J. Environ. Biol. 2007, 28, 307–314. [Google Scholar] [PubMed]
- Kataki, S.; West, H.; Clarke, M.; Baruah, D.C. Phosphorus recovery as struvite: Recent concerns for use of seed, alternative Mg source, nitrogen conservation and fertilizer potential. Resour. Conserv. Recycl. 2016, 107, 142–156. [Google Scholar] [CrossRef]
- Jia, G.; Zhang, H.; Krampe, J.; Muster, T.; Gao, B.; Zhu, N. Applying a chemical equilibrium model for optimizing struvite precipitation for ammonium recovery from anaerobic digester effluent. J. Clean. Prod. 2017, 147, 297–305. [Google Scholar] [CrossRef]
- Achat, D.L.; Sperandio, M.; Daumer, M.L.; Santellani, A.C.; Prud`Homme, L.; Akhtar, M.; Morel, C. Plant-availability of phosphorus recycled from pig manures and dairy effluents as assessed by isotopic labeling techniques. Geoderma 2014, 232/234, 24–33. [Google Scholar] [CrossRef]
- Talboys, P.J.; Heppell, J.; Roose, T.; Healey, J.R.; Jones, D.J.; Withers, P.J.A. Struvite: a slow-release fertilizer for sustainable phosphorus management? Plant Soil 2016, 401, 109–123. [Google Scholar] [CrossRef] [PubMed]
- Degryse, F.; Baird, R.; da Silva, R.C.; McLaughlin, M.J. Dissolution rate and agronomic effectiveness of struvite fertilizers—effect of soil pH, granulation and base excess. Plant Soil 2017, 410, 139–152. [Google Scholar] [CrossRef]
- Kataki, S.; West, H.; Clarke, M.; Baruah, D.C. Phosphorus recovery as struvite from farm, municipal and industrial waste: Feedstock suitability, methods and pre-treatments. Waste Manag. 2016, 49, 437–454. [Google Scholar] [CrossRef] [PubMed]
- Laureni, M.; Palatsi, J.; Llovera, M.; Bonmati, A. Influence of pig slurry characteristics on ammonia stripping efficiencies and quality of the recovered ammonium-sulfate solution. J. Chem. Technol. Biotechnol. 2013, 88, 1654–1662. [Google Scholar] [CrossRef]
- Kandil, A.-H.T.; Cheira, M.F.; Gado, H.S.; Soliman, M.H.; Akl, H.M. Ammonium sulfate preparation from phosphogypsum waste. J. Radiat. Res. Appl. 2017, 10, 24–33. [Google Scholar] [CrossRef]
- Schab, S.; Zdunek, A.; Biskupski, A. Research for the Granulation of Ammonium Sulphate with Calcium Salt - Pilot Tests. 2015. Available online: http://www.granulacja.pl/index.php/referaty-2015/176-badania-granulacji-siarczanu-amonu-przy-uzyciu-soli-wapnia-proby-poltechniczne (accessed on 10 November 2018).
- Törnwall, E.; Pettersson, H.; Thorin, E.; Schwede, S. Post-treatment of biogas digestate-An evaluation of ammonium recovery, energy use and sanitation. Energy Procedia 2017, 142, 957–963. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).