Boosting the Power-Generation Performance of Micro-Sized Al-H2O2 Fuel Cells by Using Silver Nanowires as the Cathode
Abstract
:1. Introduction
2. Results and Discussions
2.1. Characterization of Electrode Surface
2.2. Ag NW//CC Electrode Performance Test
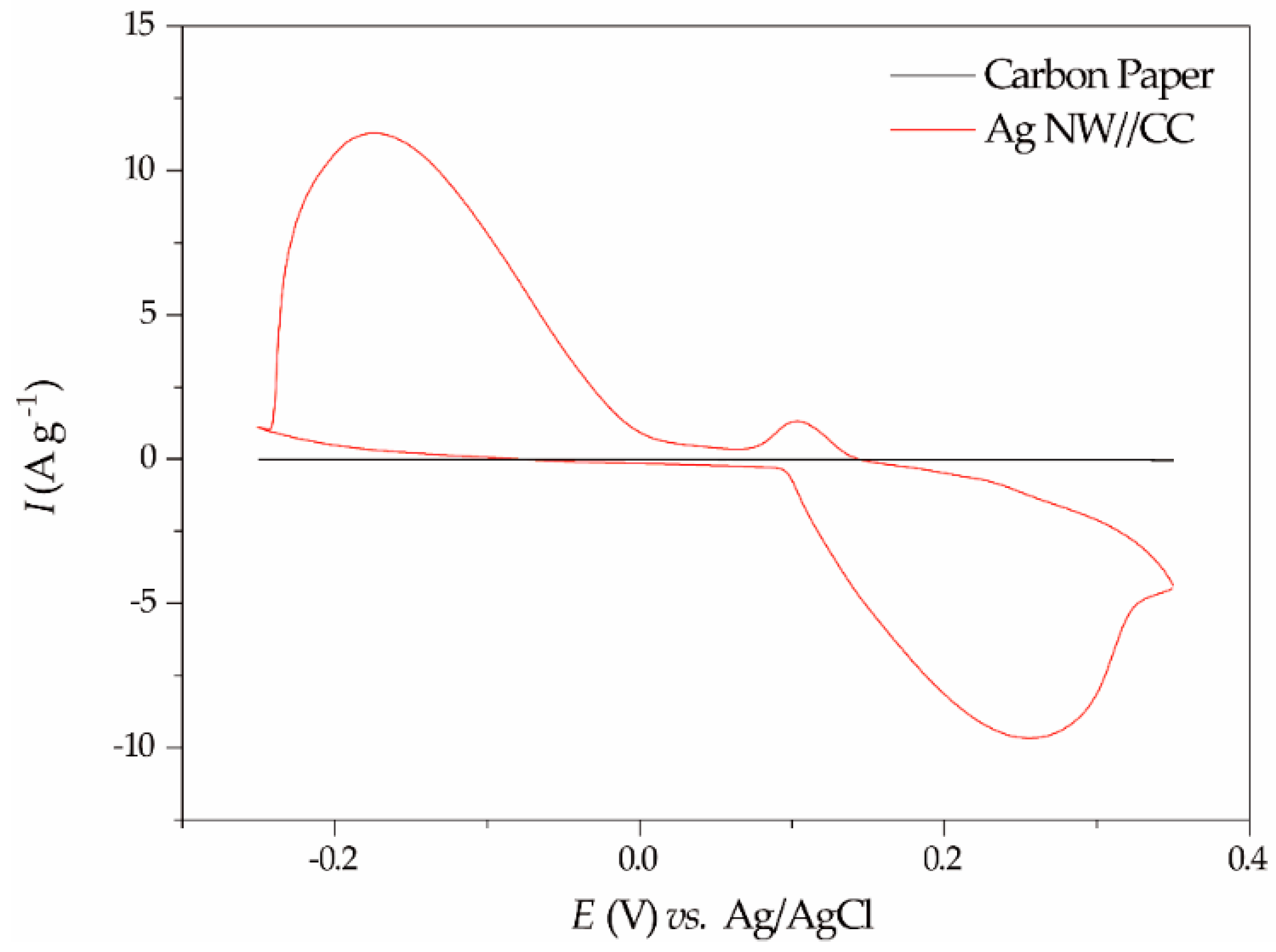
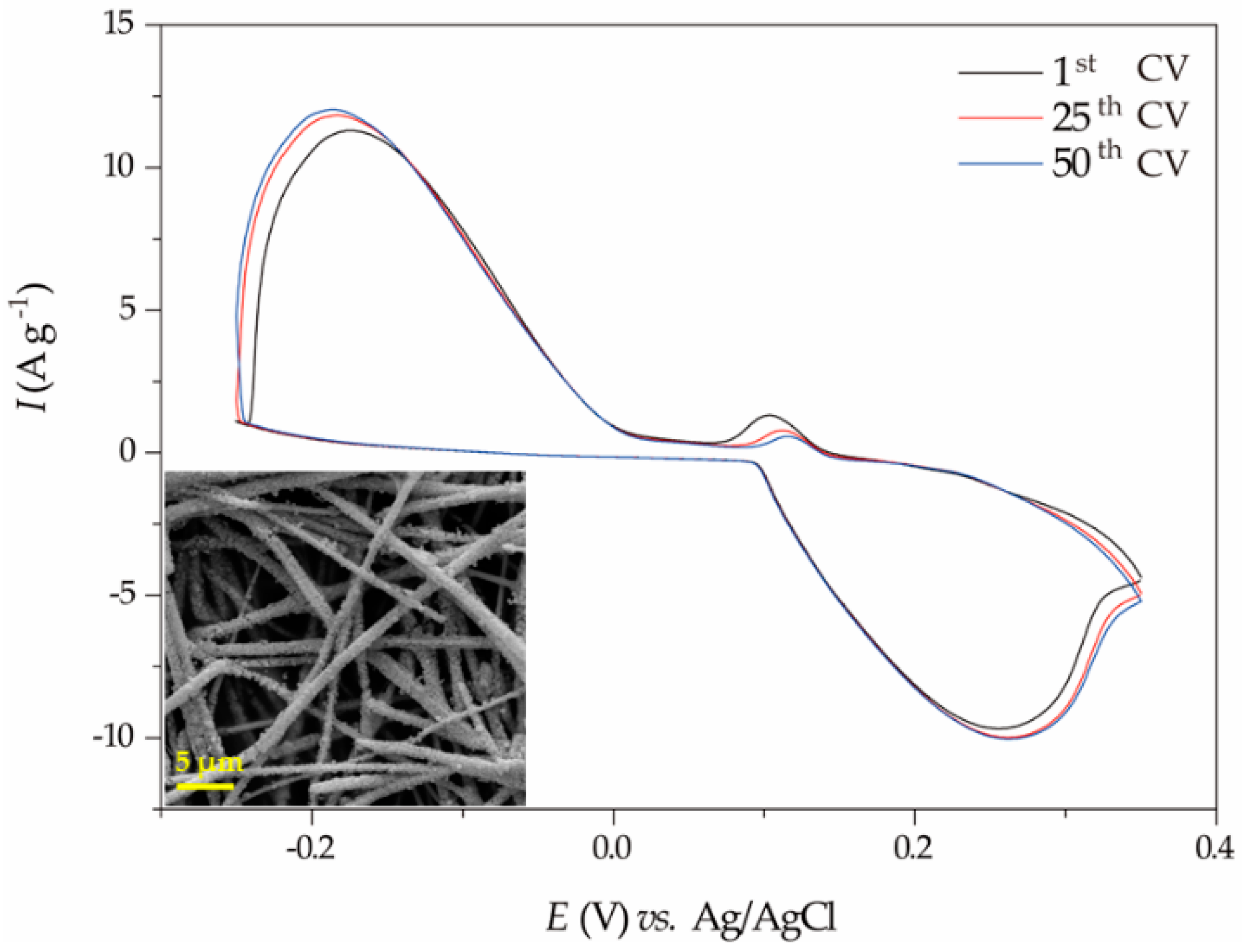
2.2.1. Cyclic Voltammetry Performance of the Ag NW//CC Electrode
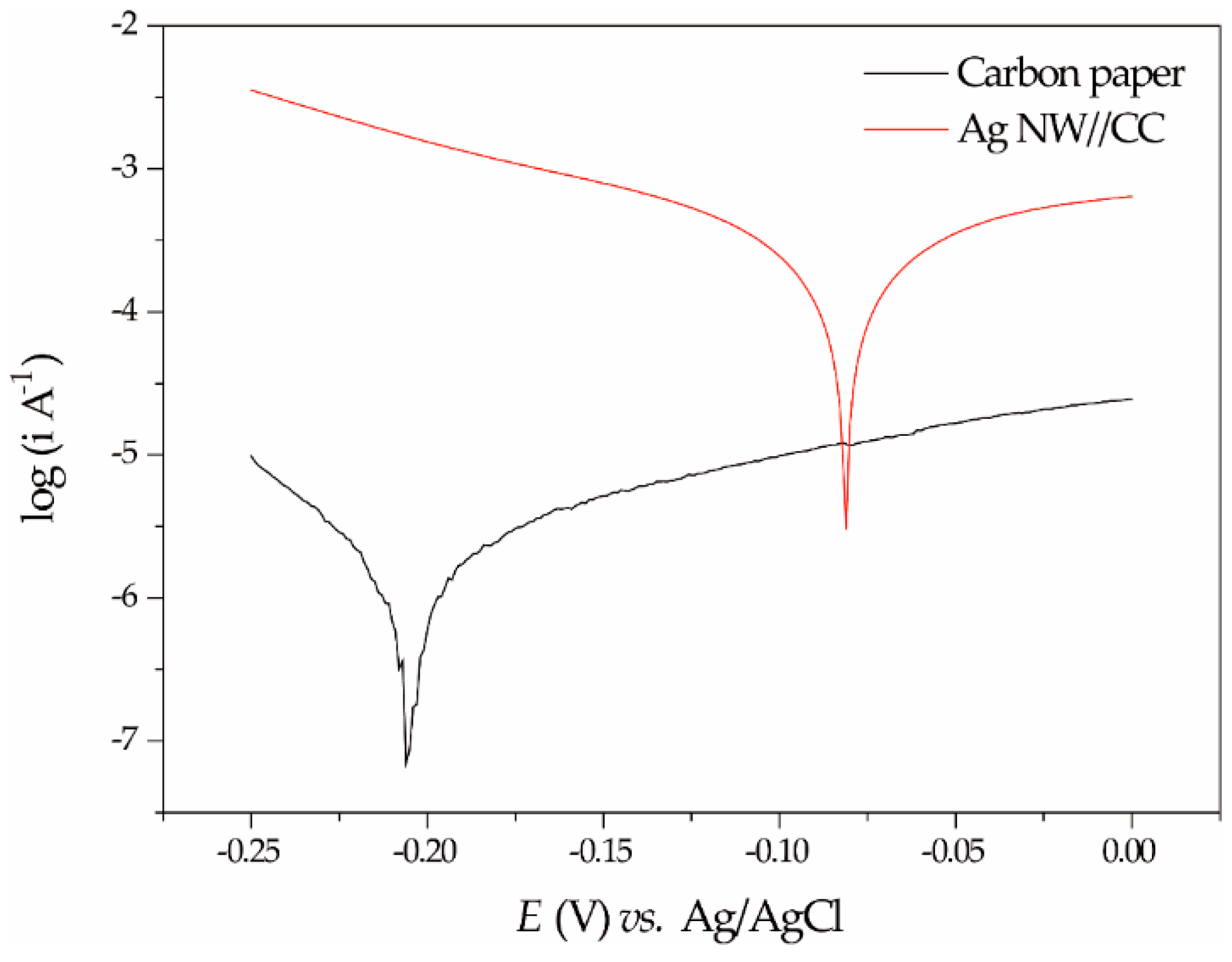
2.2.2. Tafel Polarization of the Ag NW//CC Electrode
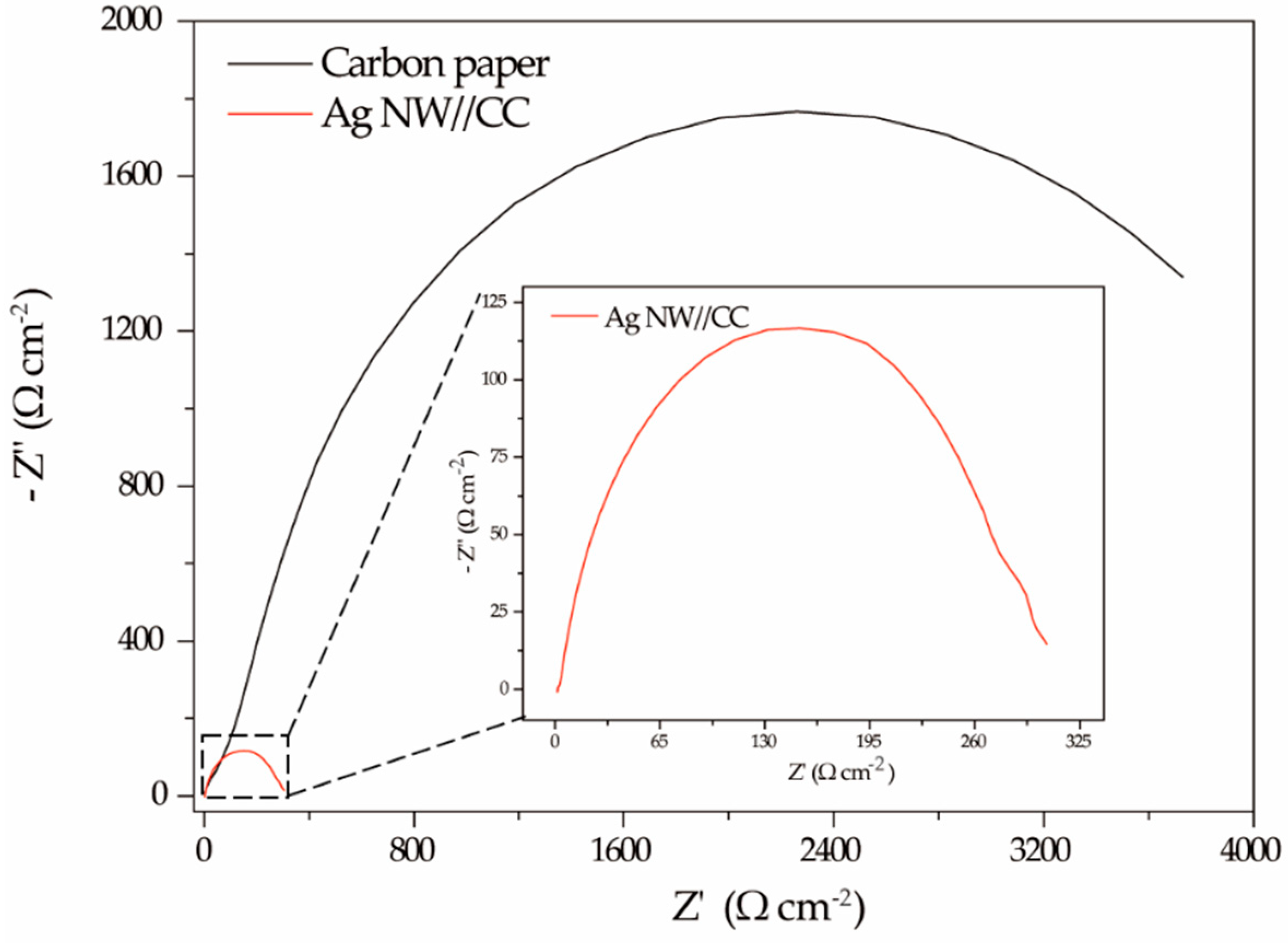
2.2.3. Characterization of Electrode Resistance
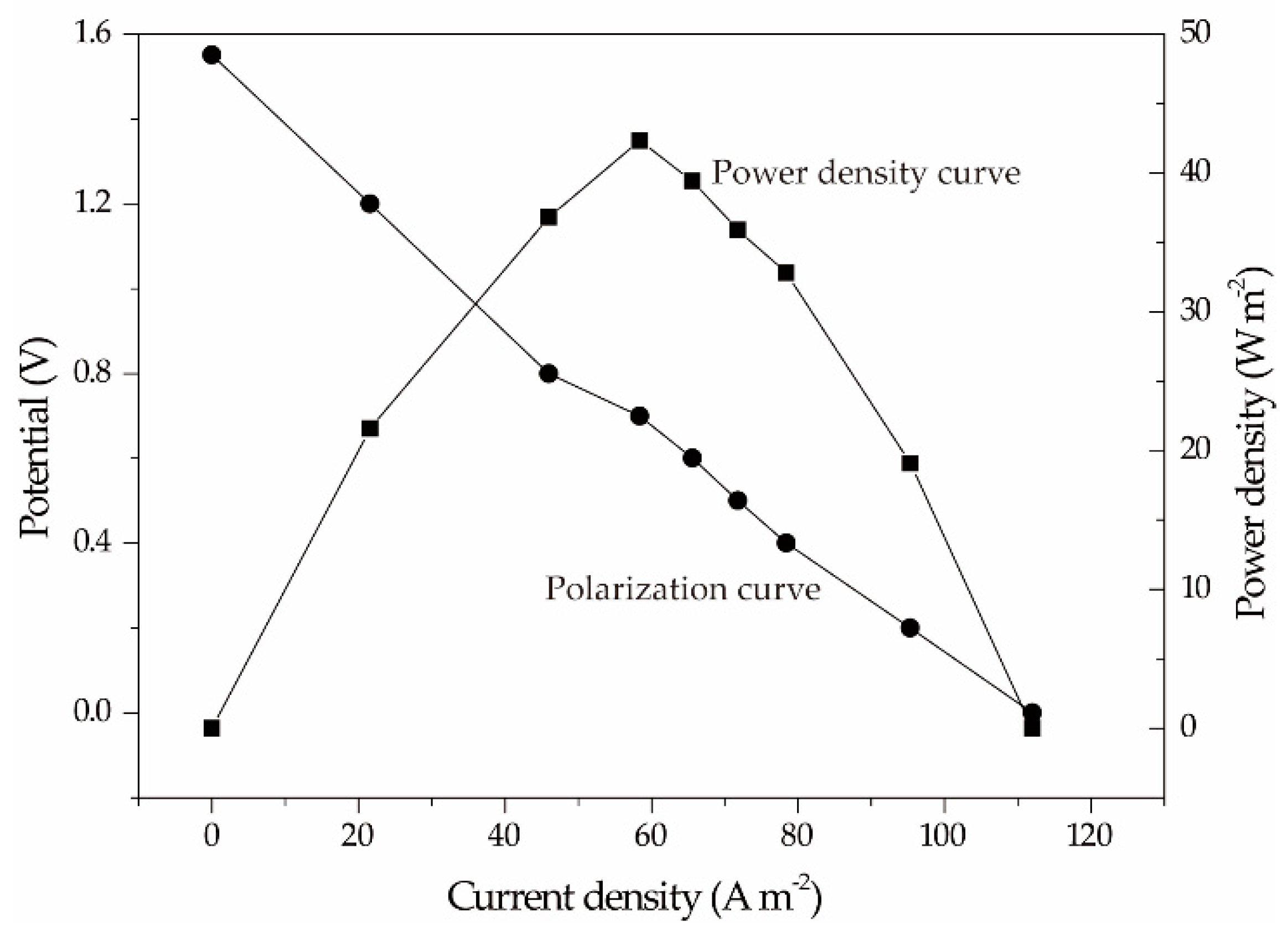
2.3. Cell Performance Test of the Micro-Sized Al-H2O2 FC
3. Materials and Methods
3.1. Synthesis and Purification of Ag NW Solution
3.2. Electrode Synthesis
3.3. Analysis of Electrode
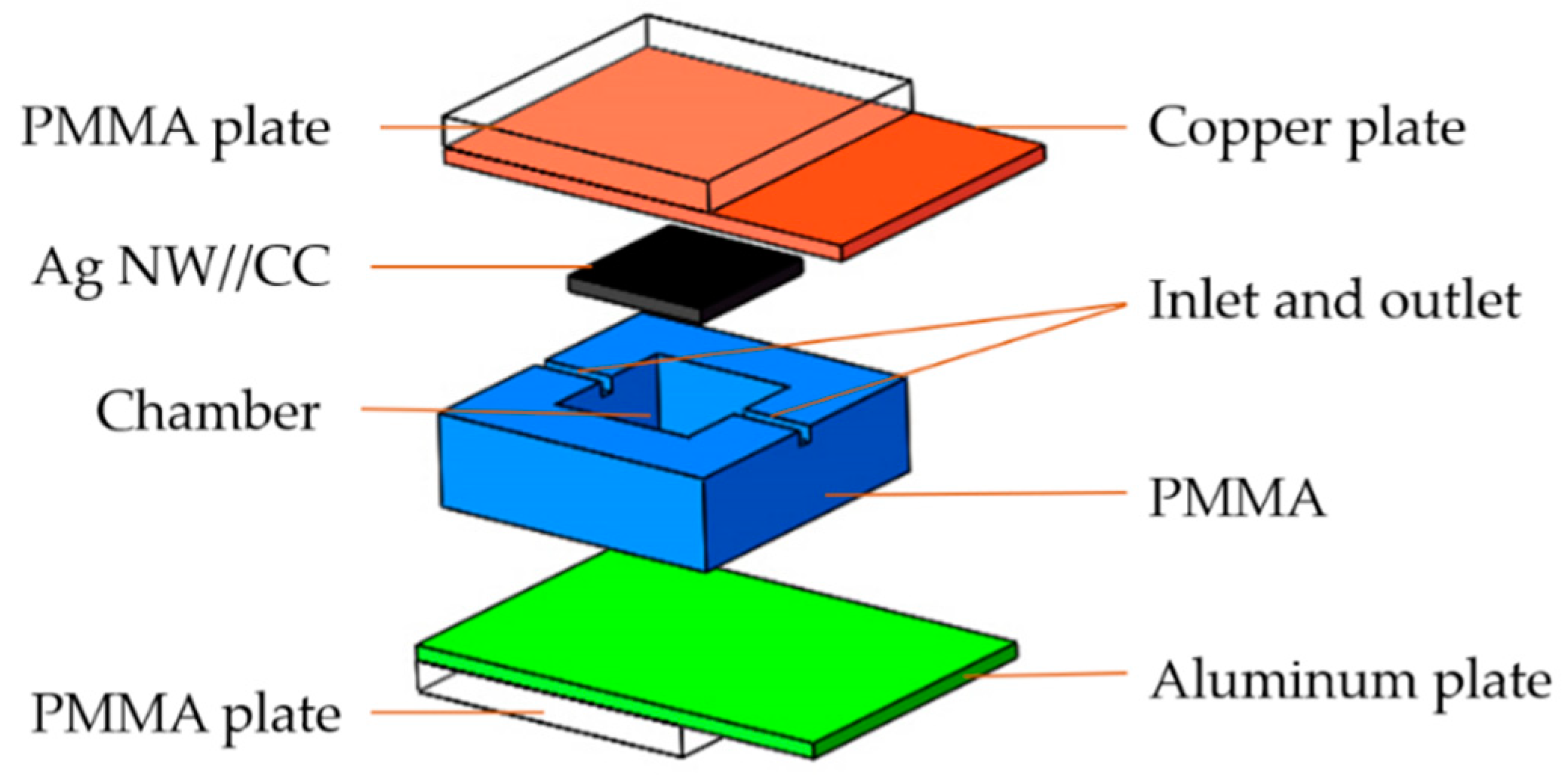
3.4. Fuel Cell Set-Up
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bahrebar, S.; Blaabjerg, F.; Wang, H.; Vafamand, N.; Khooban, M.-H.; Rastayesh, S.; Zhou, D. A novel type-2 fuzzy logic for improved risk analysis of proton exchange membrane fuel cells in marine power systems application. Energies 2018, 11, 721. [Google Scholar] [CrossRef]
- Yang, W.; Yang, S.; Sun, W.; Sun, G.; Xin, Q. Nanostructured silver catalyzed nickel foam cathode for an aluminum–hydrogen peroxide fuel cell. J. Power Source 2006, 160, 1424. [Google Scholar] [CrossRef]
- Dow, E.G.; Bessette, R.R.; Seeback, G.L.; Marsh-Orndorff, C.; Meunier, H.; VanZee, J.; Medeiros, M.G. Enhanced electrochemical performance in the development of the aluminum/hydrogen peroxide semi-fuel cell. J. Power Sources 1997, 65, 207–212. [Google Scholar] [CrossRef]
- Deuchars, G.D.; Hill, J.R.; Stannard, J.H.; Stockburger, D.C. Aluminum-hydrogen peroxide power system for an unmanned underwater vehicle. Oceans 1993, 152, 158–165. [Google Scholar]
- Mendez, A.; Leo, T.; Herreros, M. Current state of technology of fuel cell power systems for autonomous underwater vehicles. Energies 2014, 7, 4676–4693. [Google Scholar] [CrossRef]
- Luo, N.; Miley, G.H.; Gimlin, R.J.; Burton, R.L.; Rusek, J.; Holcomb, F. Hydrogen-peroxide-based fuel cells for space power systems. J. Propul. Power 2012, 24, 583–589. [Google Scholar] [CrossRef]
- Debe, M.K. Electrocatalyst approaches and challenges for automotive fuel cells. Nature 2012, 486, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Mousavi Shaegh, S.A.; Nguyen, N.-T.; Mousavi Ehteshami, S.M.; Chan, S.H. A membraneless hydrogen peroxide fuel cell using prussian blue as cathode material. Energy Environ. Sci. 2012, 5, 8225. [Google Scholar] [CrossRef]
- Brodrecht, D.J.; Rusek, J.J. Aluminum–hydrogen peroxide fuel-cell studies. Appl. Energy 2003, 74, 113–124. [Google Scholar] [CrossRef]
- Bessette, R.R.; Cichon, J.M.; Dischert, D.W.; Dow, E.G. A study of cathode catalysis for the aluminium/hydrogen peroxide semi-fuel cell. J. Power Sources 1999, 80, 248–253. [Google Scholar] [CrossRef]
- Asadnia, M.; Mousavi Ehteshami, S.M.; Chan, S.H.; Warkiani, M.E. Development of a fiber-based membraneless hydrogen peroxide fuel cell. RSC Adv. 2017, 7, 40755–40760. [Google Scholar] [CrossRef]
- Marsh, C.L.; Seebach, G.L.; Vanzee, J.W.; Bessette, R.R.; Meunier, H.G. Preparation of an electrocatalytic cathode for an aluminum-hydrogen peroxide battery. U.S. Patent No. 5,296,429, 22 March 1994. [Google Scholar]
- Patrissi, C.; Bessette, R.R.; Kim, Y.; Schumacher, R.C. Fabrication and rate performance of a microfiber cathode in a mg–H2O2 flowing electrolyte semi-fuel cell. J. Electrochem. Soc. 2008, 155, B558–B562. [Google Scholar] [CrossRef]
- Honda, M.; Kodera, T.; Kita, H. On the electrochemical behavior of H2O2 at ag in alkaline solution. Electrochim. Acta 1983, 28, 727–733. [Google Scholar] [CrossRef]
- Tilak, B.V.; Perkins, R.S.; Kozlowska, H.A.; Conway, B.E. Impedance and formation characteristics of electrolytically generated silver oxides—Formation and reduction of surface oxides and the role of dissolution processes. Electrochim. Acta 1972, 17, 1447–1469. [Google Scholar] [CrossRef]
- Shu, C.; Wang, E.; Jiang, L.; Tang, Q.; Sun, G. Studies on palladium coated titanium foams cathode for mg–H2O2 fuel cells. J. Power Sources 2012, 208, 159–164. [Google Scholar] [CrossRef]
- Lue, S.; Liu, N.-Y.; Rajesh Kumar, S.; Tseng, K.; Wang, B.-Y.; Leung, C.-H. Experimental and one-dimensional mathematical modeling of different operating parameters in direct formic acid fuel cells. Energies 2017, 10, 1972. [Google Scholar] [CrossRef]
- Oliveira, F.M.D.; Guedes, T.D.J.; Lima, A.B.; Silva, L.M.D.; Santos, W.T.P.D. Alternative method to obtain the tafel plot for simple electrode reactions using batch injection analysis coupled with multiple-pulse amperometric detection. Electrochim. Acta 2017, 242, 180–186. [Google Scholar] [CrossRef]
- Sharma, S.; Zhang, K.; Gupta, G.; Santamaria, D. Exploring pani-tin nanoparticle coatings in a pefc environment: Enhancing corrosion resistance and conductivity of stainless steel bipolar plates. Energies 2017, 10, 1152. [Google Scholar] [CrossRef]
- Hu, C.; Xiao, Y.; Zhao, Y.; Chen, N.; Zhang, Z.; Cao, M.; Qu, L. Highly nitrogen-doped carbon capsules: Scalable preparation and high-performance applications in fuel cells and lithium ion batteries. Nanoscale 2013, 5, 2726–2733. [Google Scholar] [CrossRef] [PubMed]
- Barsoukov, E.; Macdonald, J.R. Impedance spectroscopy: Theory, experiment, and applications. J. Hosp. Palliat. Nurs. 2005, 4, 206–207. [Google Scholar]
- León, C.P.D.; Walsh, F.C.; Rose, A.; Lakeman, J.B.; Browning, D.J.; Reeve, R.W. A direct borohydride--acid peroxide fuel cell. J. Power Sources 2007, 164, 441–448. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, Y.; Wu, Z.; Leung, D.Y.C. A direct urea microfluidic fuel cell with flow-through ni-supported-carbon- nanotube-coated sponge as porous electrode. J. Power Sources 2017, 363, 61–69. [Google Scholar] [CrossRef]
- Ma, J.; Habrioux, A.; Morais, C.; Alonso-Vante, N. Electronic modification of Pt via Ti and Se as tolerant cathodes in air-breathing methanol microfluidic fuel cells. Phys. Chem. Chem. Phys. 2014, 16, 13820–13826. [Google Scholar] [CrossRef] [PubMed]
- Esquivel, J.P.; Del Campo, F.J.; Gómez de la Fuente, J.L.; Rojas, S.; Sabaté, N. Microfluidic fuel cells on paper: Meeting the power needs of next generation lateral flow devices. Energy Environ. Sci. 2014, 7, 1744–1749. [Google Scholar] [CrossRef]
- LóPez-González, l.B.; Dector, A.; Cuevas-Muňiz, F.M.; Arjona, N.; Cruz-Madrid, C.; Arana-Cuenca, A.; Guerra-Balcázar, z.M.; Arriaga, L.G.; Ledesma-García A, J. Hybrid microfluidic fuel cell based on laccase/c and auag/c electrodes. Biosens. Bioelectron. 2014, 62, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Jindal, A.; Basu, S.; Chauhan, N.; Ukai, T.; Kumar, D.S.; Samudhyatha, K.T. Application of electrospun cnx nanofibers as cathode in microfluidic fuel cell. J. Power Sources 2017, 342, 165–174. [Google Scholar] [CrossRef]
- Falcão, D.; Silva, R.; Rangel, C.; Pinto, A. Performance of an Active Micro Direct Methanol Fuel Cell Using Reduced Catalyst Loading MEAs. Energies 2017, 10, 1683. [Google Scholar] [CrossRef]
- Sun, Y.; Gates, B.; Brian Mayers, A.; Xia, Y. Crystalline silver nanowires by soft solution processing. Nano Lett. 2002, 2, 165–168. [Google Scholar] [CrossRef]
- Sun, Y.; Yin, Y.; Mayers, B.T.; Herricks, T.; Xia, Y. Uniform silver nanowires synthesis by reducing AgNO3 with ethylene glycol in the presence of seeds and poly(vinyl pyrrolidone). Chem. Mater. 2002, 14, 4736–4745. [Google Scholar]
- Coskun, S.; Aksoy, B.; Unalan, H.E. Polyol synthesis of silver nanowires: An extensive parametric study. Cryst. Growth Des. 2011, 11, 4963–4969. [Google Scholar] [CrossRef]
- Li, B.; Ye, S.; Stewart, I.E.; Alvarez, S.; Wiley, B.J. Synthesis and purification of silver nanowires to make conducting films with a transmittance of 99%. Nano Lett. 2015, 15, 6722. [Google Scholar] [CrossRef] [PubMed]
- Mayousse, C.; Celle, C.; Moreau, E.; Mainguet, J.F.; Carella, A.; Simonato, J.P. Improvements in purification of silver nanowires by decantation and fabrication of flexible transparent electrodes: Application to capacitive touch sensors. Nanotechnology 2013, 24, 215501. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.; Xiao, S.; Li, Z.; Li, Y.B.; Kobayashi, H.; Li, J.; Yang, Y.; Liao, Q.; Zhu, X.; He, X.F.; et al. Hybrid solar-to-methane conversion system with a Faradaic efficiency of up to 96%. Nano Energy 2018, 53, 232–239. [Google Scholar] [CrossRef]

| Anode Catalyst | Cathode Catalyst | Electrolyte | Imax-areal (A·m−2) | Pmax-areal (W·m−2) | Ref. |
|---|---|---|---|---|---|
| aluminum | Ag NW | 0.1 M H2O2 & 1 M NaOH & 20 g·L−1 NaCl | 112 | 43 | This work |
| silver | Prussian blue | 0.1 M HCl & 0.5 M H2O2 | 57 a | 8 b | [8] |
| nickel | Prussian blue | 0.1 M HCl & 0.5 M H2O2 | 101 a | 15 b | [8] |
| Ni/carbon nanotube sponge | Pt/C | Anolyte: 3 M urea in 3 M KOH; catholyte: 1.5 M H2SO4 | 230 b | 39 b | [23] |
| Pt-Ru/C | PtxSey/C (x/y = 5:1) | 5 M methanol & 0.5 M H2SO4 | 409 a | 30 a | [24] |
| aluminum | Prussian blue | 0.1 M HCl & 0.5 M H2O2 | 45 a | 10 b | [11] |
| Pt-Ru on Au COP | Pt/CC (40 wt %) | 4 M methanol & 1 M NaOH | 155 a | 32 a | [25] |
| Au-Ag/C | Vulcan XC-72 | Laccase/glucose | 19 a | 5 b | [26] |
| Pt | carbon nitride nanofibers | Anolyte: 2.1 M HCOOH catholyte: 0.144 M KMnO4 | 97 a | 34 a | [27] |
| Pt | Pt | Anolyte: 2.1 M HCOOH catholyte: 0.144 M KMnO4 | 61 a | 30 a | [27] |
| Pt | Au | Anolyte: 2.1 M HCOOH catholyte: 0.144 M KMnO4 | 60 a | 27 a | [27] |
| Pt-Ru | Pt Black | 3 M methanol (QN3 designation) | 260 b | 39 a | [28] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, H.; Yang, Y.; Liu, T.; Chang, H. Boosting the Power-Generation Performance of Micro-Sized Al-H2O2 Fuel Cells by Using Silver Nanowires as the Cathode. Energies 2018, 11, 2316. https://doi.org/10.3390/en11092316
Zhang H, Yang Y, Liu T, Chang H. Boosting the Power-Generation Performance of Micro-Sized Al-H2O2 Fuel Cells by Using Silver Nanowires as the Cathode. Energies. 2018; 11(9):2316. https://doi.org/10.3390/en11092316
Chicago/Turabian StyleZhang, Heng, Yang Yang, Tianyu Liu, and Honglong Chang. 2018. "Boosting the Power-Generation Performance of Micro-Sized Al-H2O2 Fuel Cells by Using Silver Nanowires as the Cathode" Energies 11, no. 9: 2316. https://doi.org/10.3390/en11092316
APA StyleZhang, H., Yang, Y., Liu, T., & Chang, H. (2018). Boosting the Power-Generation Performance of Micro-Sized Al-H2O2 Fuel Cells by Using Silver Nanowires as the Cathode. Energies, 11(9), 2316. https://doi.org/10.3390/en11092316





