Autoignition Behavior of an Ethanol-Methylcellulose Gel Droplet in a Hot Environment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethanol Gel Preparation
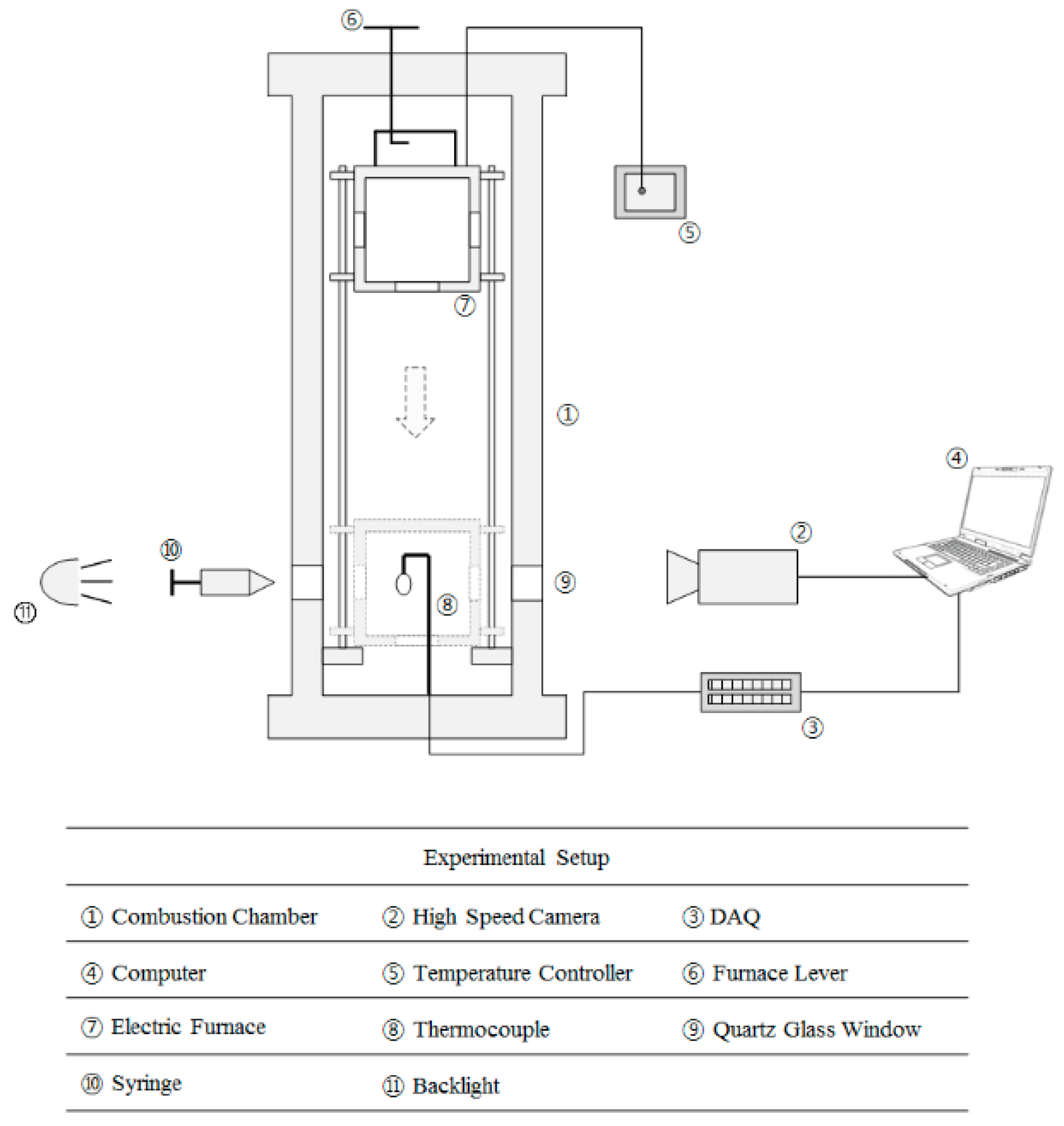
2.2. Combustion Chamber
2.3. High-Speed Camera
2.4. Ignition Delay and Average Burning Rate
2.5. Experimental Condition
3. Results and Discussion
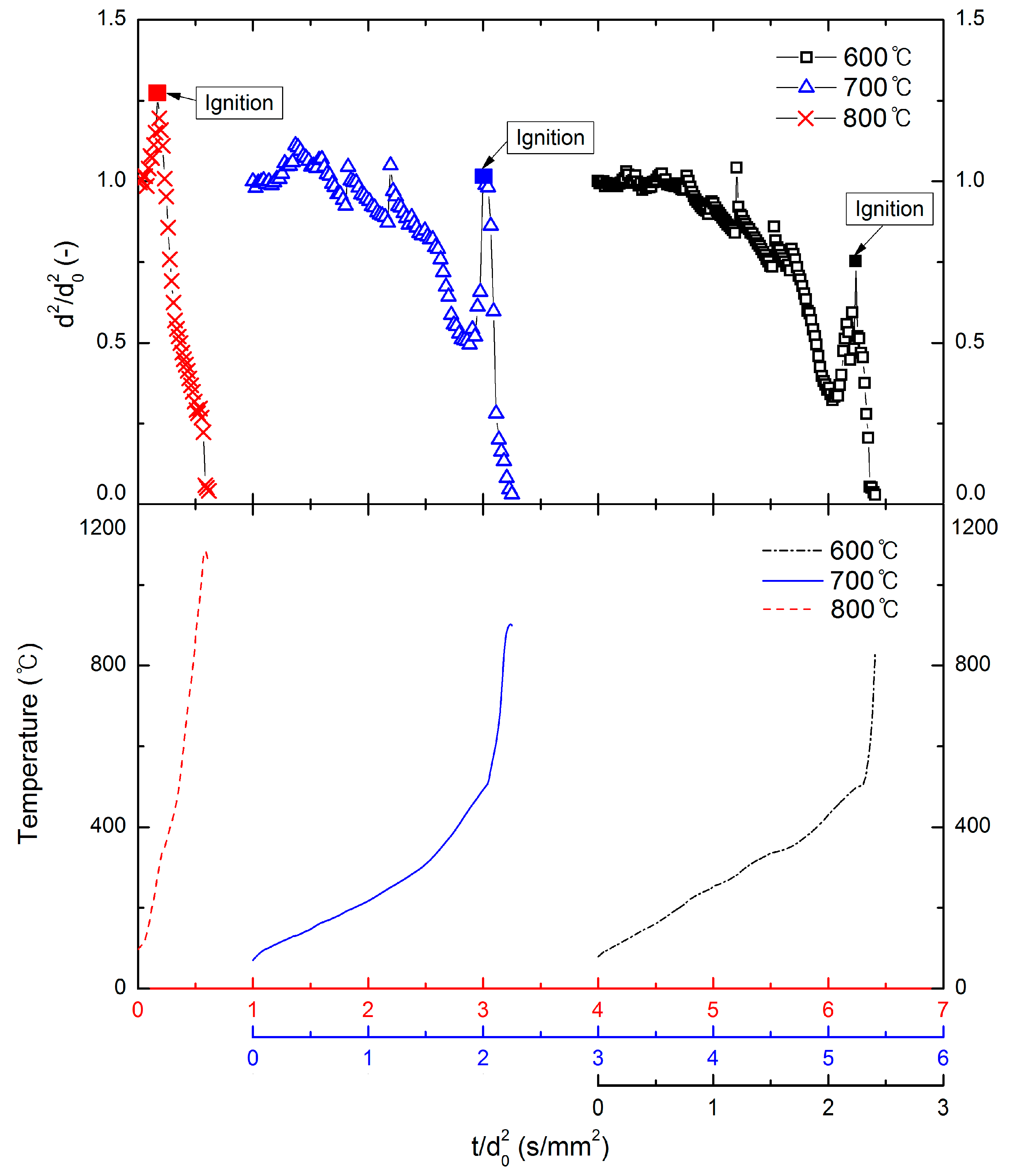
3.1. Ignition and Combustion Behavior of Ethanol Gel
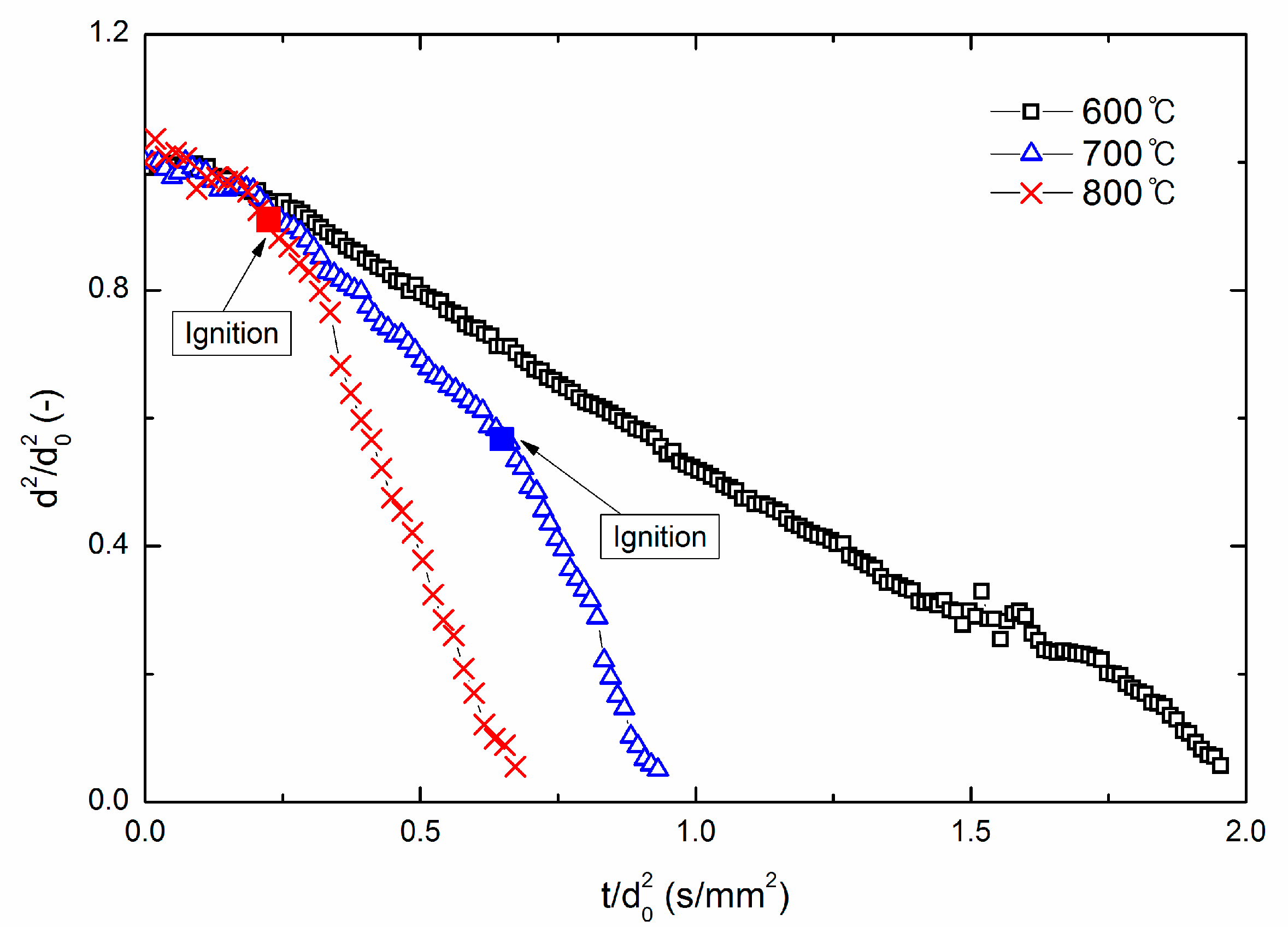
3.2. Droplet Diameter Variation for Various Ambient Temperatures
3.3. IgnitionDdelay and Average Burning Rate
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Teipel, U.; Forter-Barth, U. Rheological behavior of nitromethane gelled with nanoparticles. J. Propul. Power 2005, 21, 40–43. [Google Scholar] [CrossRef]
- Natan, B.; Rahimi, S. The status of gel propellants in year 2000. Int. J. Energ. Mater. Chem. Propul. 2002, 5, 172–194. [Google Scholar] [CrossRef]
- Hodge, K.; Crofoot, T.; Nelson, S. Gelled propellants for tactical missile applications. In Proceedings of the 35th AIAA/ASME/SAE/ASEE Joint Propulsion Conference & Exhibit, Los Angeles, CA, USA, 20–23 June 1999. AIAA-99-2976. [Google Scholar]
- Rapp, D.; Zurawski, R. Characterization of aluminum/rp-1 gel propellant properties. In Proceedings of the 24th AIAA/ASME/SAE/ASEE Joint Propulsion Conference, Boston, MA, USA, 11–13 July 1988. AIAA-88-2821. [Google Scholar]
- Varma, M.; Gupta, B.; Pandey, M. Formulation & storage studies on hydrazine-based gelled propellants. Def. Sci. J. 1996, 46, 435–442. [Google Scholar]
- Santos, P.; Carignano, M.; Campanella, O. Qualitative study of thixotropy in gelled hydrocarbon fuels. Eng. Lett. 2011, 19, 13–19. [Google Scholar]
- Dennis, J.D.; Kubal, T.D.; Campanella, O.; Son, S.F.; Pourpoint, T.L. Rheological characterization of monomethylhydrazine gels. J. Propul. Power 2013, 29, 313–320. [Google Scholar] [CrossRef]
- Mansour, A.; Chigier, N. Air-blast atomization of non-newtonian liquids. J. Non-Newtonian Fluid Mech. 1995, 58, 161–194. [Google Scholar] [CrossRef]
- Lee, I.; Koo, J. Break-up characteristics of gelled propellant simulants with various gelling agent contents. J. Therm. Sci. 2010, 19, 545–552. [Google Scholar] [CrossRef]
- Chojnacki, K.; Feikema, D. Atomization studies of gelled liquids. In Proceedings of the 30th AIAA/ASME/SAE/ASEE Joint Propulsion Conference, Indianapolis, IN, USA, 27–29 June 1994. AIAA-94-2773. [Google Scholar]
- Heislbetz, B.; Madlener, K.; Ciezki, H. Breakup characteristics of a newtonian liquid sheet formed by a doublet impinging jet injector. In Proceedings of the 43rd AIAA/ASME/SAE/ASEE Joint Propulsion Conference & Exhibit, Cincinnati, OH, USA, 8–11 July 2007. AIAA-2007-5694. [Google Scholar]
- Green, J.; Rapp, D.; Roncace, J. Flow visualization of a rocket injector spray using gelled propellant simulants. In Proceedings of the 27th AIAA/ASME/SAE/ASEE Joint Propulsion Conference, Sacramento, CA, USA, 24–27 June 1991. AIAA-91-2198. [Google Scholar]
- Mallory, J.; DeFini, S.J.; Sojka, P. Formulation of gelled propellant simulants. In Proceedings of the 46th AIAA/ASME/SAE/ASEE Joint Propulsion Conference & Exhibit, Nashville, TN, USA, 25–28 July 2010. AIAA-2010-7142. [Google Scholar]
- Rahimi, S.; Natan, B. Numerical solution of the flow of power-law gel propellants in converging injectors. Propellants Explos. Pyrotech. 2000, 25, 203–212. [Google Scholar] [CrossRef]
- Liu, Z.; Wu, J.; Zhen, H.; Hu, X. Numerical simulation on head-on binary collision of gel propellant droplets. Energies 2013, 6, 204–219. [Google Scholar] [CrossRef]
- Kunin, A.; Natan, B.; Greenberg, J.B. Theoretical model of the transient combustion of organic-gellant-based gel fuel droplets. J. Propul. Power 2010, 26, 765–771. [Google Scholar] [CrossRef]
- Mishra, D.P.; Patyal, A.; Padhwal, M. Effects of gellant concentration on the burning and flame structure of organic gel propellant droplets. Fuel 2011, 90, 1805–1810. [Google Scholar] [CrossRef]
- Gupta, B.L.; Varma, M. Ignition and combustion studies on metallized UDMH-RFNA bipropellant system. Indian J. Eng. Mater. Sci. 1999, 6, 13–21. [Google Scholar]
- Liu, Z.; Hu, X.; He, Z.; Wu, J. Experimental study on the combustion and microexplosion of freely falling gelled unsymmetrical dimethylhydrazine (UDMH) fuel droplets. Energies 2012, 5, 3126–3136. [Google Scholar] [CrossRef]
- Bar-or, D.; Natan, B. The effect of ambient conditions on the burning rate of gel fuel droplets. Propellants Explos. Pyrotech. 2013, 38, 199–203. [Google Scholar] [CrossRef]
- Arnold, R.; Anderson, W. Droplet burning of JP-8/silica gels. In Proceedings of the 48th AIAA Aerospace Sciences Meeting Including the New Horizons Forum and Aerospace Exposition, Orlando, FL, USA, 4–7 January 2010. AIAA-2010-421. [Google Scholar]
- Solomon, Y.; Natan, B.; Cohen, Y. Combustion of gel fuels based on organic gellants. Combust. Flame 2009, 156, 261–268. [Google Scholar] [CrossRef]
- Negri, M.; Ciezki, H.K. Combustion of gelled propellants containing microsized and nanosized aluminum particles. J. Propul. Power 2014, 31, 400–407. [Google Scholar] [CrossRef]
- Kirchberger, C.; Ciezki, H.; Kröger, P. Realization and run-in of a gel combustion chamber with optical access. In Proceedings of the 7th European conference for Aeronautics and Space Sciences(EUCASS2017), Milan, Italy, 3–6 July 2017. [Google Scholar]
- Harada, T.; Watanabe, H.; Suzuki, Y.; Kamata, H.; Matsushita, Y.; Aoki, H.; Miura, T. A numerical investigation of evaporation characteristics of a fuel droplet suspended from a thermocouple. Int. J. Heat Mass Transf. 2011, 54, 649–655. [Google Scholar] [CrossRef]
- Khan, Q.S.; Baek, S.W.; Ghassemi, H. On the autoignition and combustion characteristics of kerosene droplets at elevated pressure and temperature. Combust. Sci. Technol. 2007, 179, 2437–2451. [Google Scholar] [CrossRef]
- Ghassemi, H.; Baek, S.W.; Khan, Q.S. Experimental study on binary droplet evaporation at elevated pressures and temperatures. Combust. Sci. Technol. 2006, 178, 1031–1053. [Google Scholar] [CrossRef]
- Javed, I.; Baek, S.W.; Waheed, K. Evaporation characteristics of heptane droplets with the addition of aluminum nanoparticles at elevated temperatures. Combust. Flame 2013, 160, 170–183. [Google Scholar] [CrossRef]
- Kang, H.; Won, J.; Baek, S.W.; Kwon, S. Autoignition and combustion characteristics of sodium borohydride-based non-toxic hypergolic fuel droplet at elevated temperatures. Combust. Flame 2017, 181, 149–156. [Google Scholar] [CrossRef]
- Nachmoni, G.; Natan, B. Combustion characteristics of gel fuels. Combust. Sci. Technol. 2000, 156, 139–157. [Google Scholar] [CrossRef]
- Rah, S.-C.; Sarofim, A.F.; Beer, J.M. Ignition and combustion of liquid fuel droplets part II: Ignition studies. Combust. Sci. Technol. 1986, 49, 169–184. [Google Scholar] [CrossRef]
- Qate, M.; Pourabdian, M.; Javareshkian, A.; Farzbod, A. A review of ignition delay and combustion characteristics of biodiesel fueled diesel engine. Appl. Mech. Mater. 2013, 390, 333–337. [Google Scholar]
- Wang, C.-H.; Chen, J.-T. An experimental investigation of the burning characteristics of water-oil emulsions. Int. Commun. Heat Mass Transfer 1996, 23, 823–834. [Google Scholar] [CrossRef]
- Mishra, D.; Patyal, A. Effects of initial droplet diameter and pressure on burning of atf gel propellant droplets. Fuel 2012, 95, 226–233. [Google Scholar] [CrossRef]
- Lee, A.; Law, C.K. An experimental investigation on the vaporization and combustion of methanol and ethanol droplets. Combust. Sci. Technol. 1992, 86, 253–265. [Google Scholar] [CrossRef]
- Botero, M.; Huang, Y.; Zhu, D.; Molina, A.; Law, C. Synergistic combustion of droplets of ethanol, diesel and biodiesel mixtures. Fuel 2012, 94, 342–347. [Google Scholar] [CrossRef]






| Component | Ethanol | Methylcellulose | Water |
|---|---|---|---|
| Concentration (wt %) | 75 | 10 | 15 |
| Temperature (°C) | Average Burning Rate (mm2/s) | |
|---|---|---|
| Pure Ethanol | Ethanol Gel | |
| 600 | - | 2.26 |
| 700 | 0.84 | 3.33 |
| 800 | 1.09 | 1.84 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, D.; Won, J.; Baek, S.W.; Kim, H. Autoignition Behavior of an Ethanol-Methylcellulose Gel Droplet in a Hot Environment. Energies 2018, 11, 2168. https://doi.org/10.3390/en11082168
Lee D, Won J, Baek SW, Kim H. Autoignition Behavior of an Ethanol-Methylcellulose Gel Droplet in a Hot Environment. Energies. 2018; 11(8):2168. https://doi.org/10.3390/en11082168
Chicago/Turabian StyleLee, Donggi, Jonghan Won, Seung Wook Baek, and Hyemin Kim. 2018. "Autoignition Behavior of an Ethanol-Methylcellulose Gel Droplet in a Hot Environment" Energies 11, no. 8: 2168. https://doi.org/10.3390/en11082168
APA StyleLee, D., Won, J., Baek, S. W., & Kim, H. (2018). Autoignition Behavior of an Ethanol-Methylcellulose Gel Droplet in a Hot Environment. Energies, 11(8), 2168. https://doi.org/10.3390/en11082168




