Nitric Acid Pretreatment of Jerusalem Artichoke Stalks for Enzymatic Saccharification and Bioethanol Production
Abstract
1. Introduction
2. Results and Discussions
2.1. Chemical Composition of Raw and Pretreated Jerusalem Artichoke Stalks
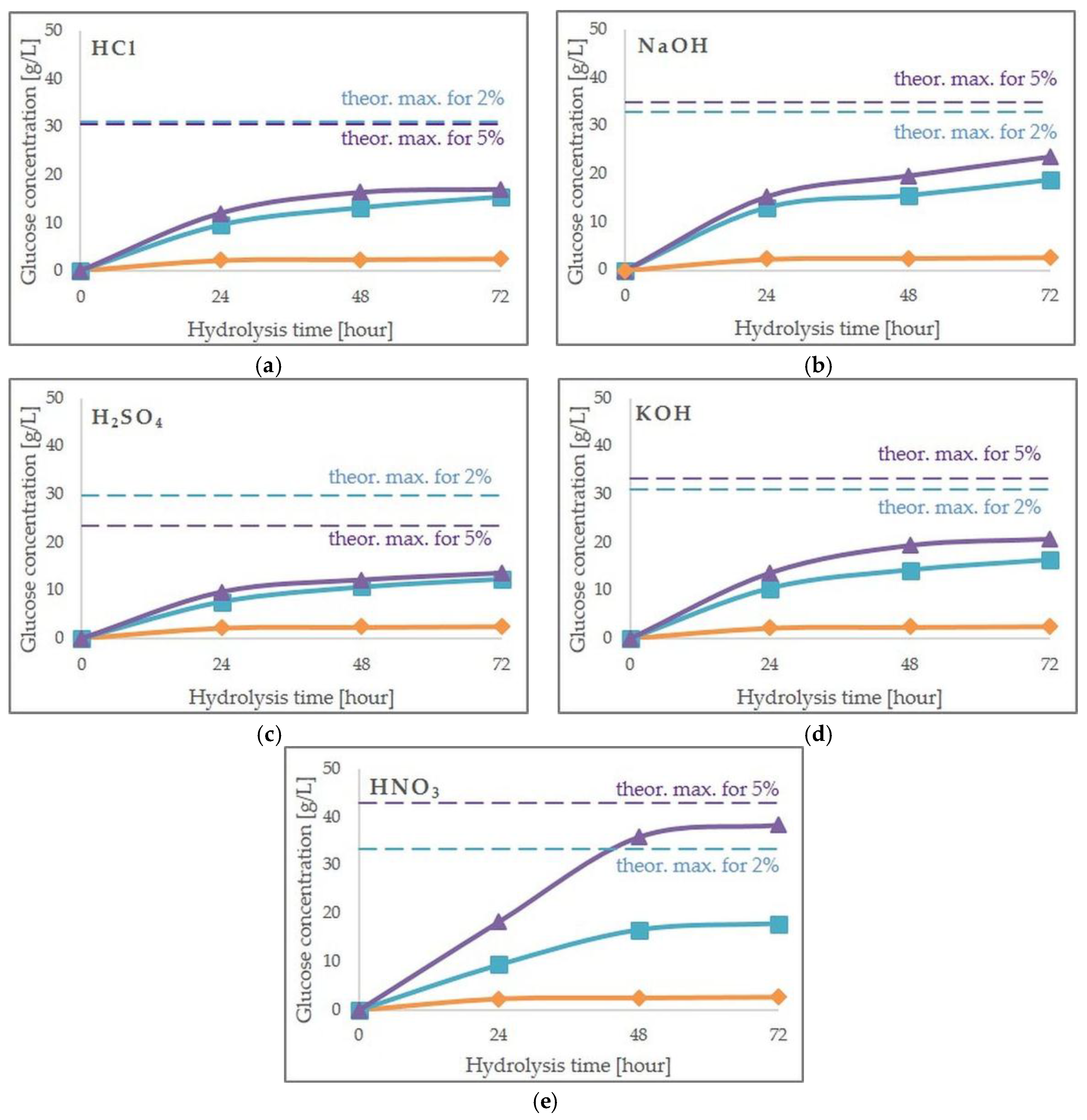
2.2. Effect of Pretreatment on Enzymatic Hydrolysis
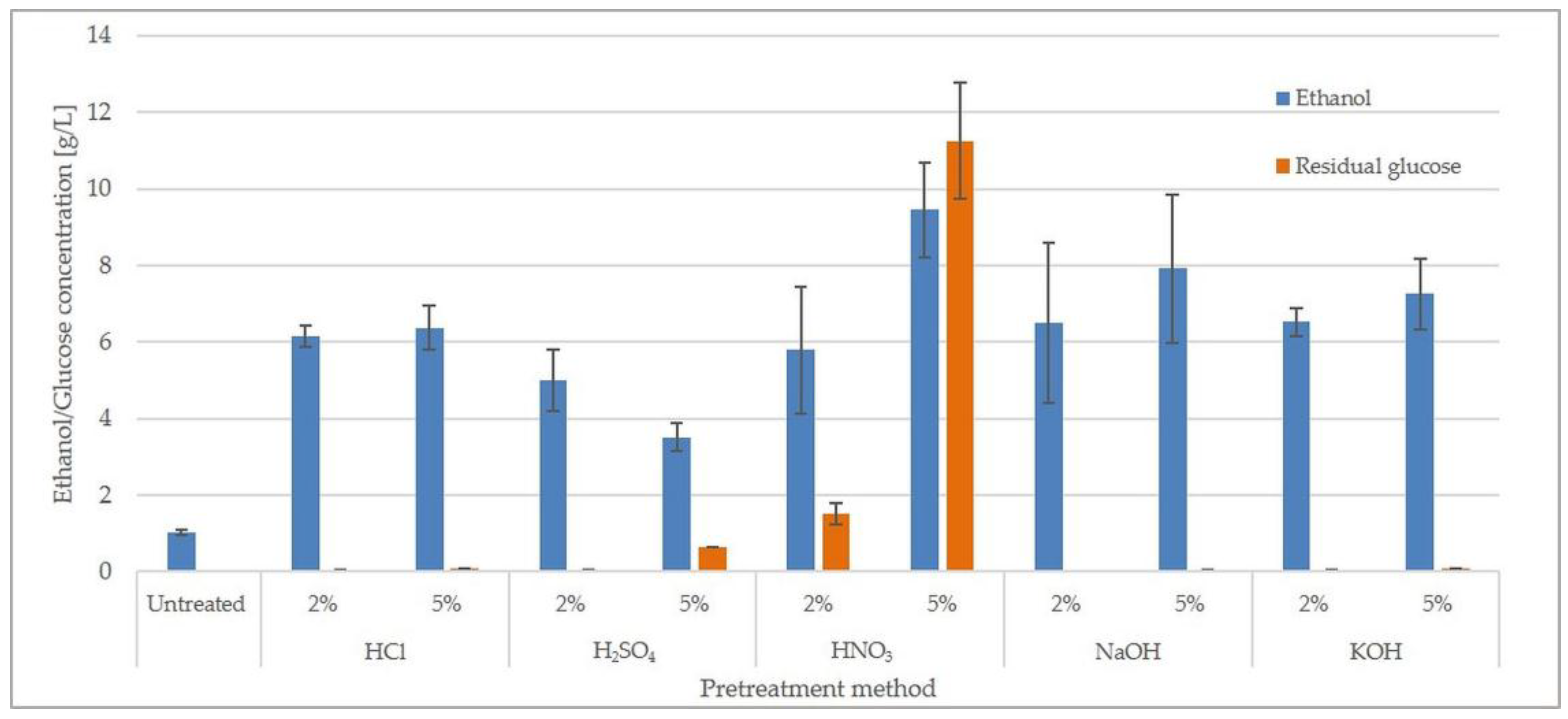
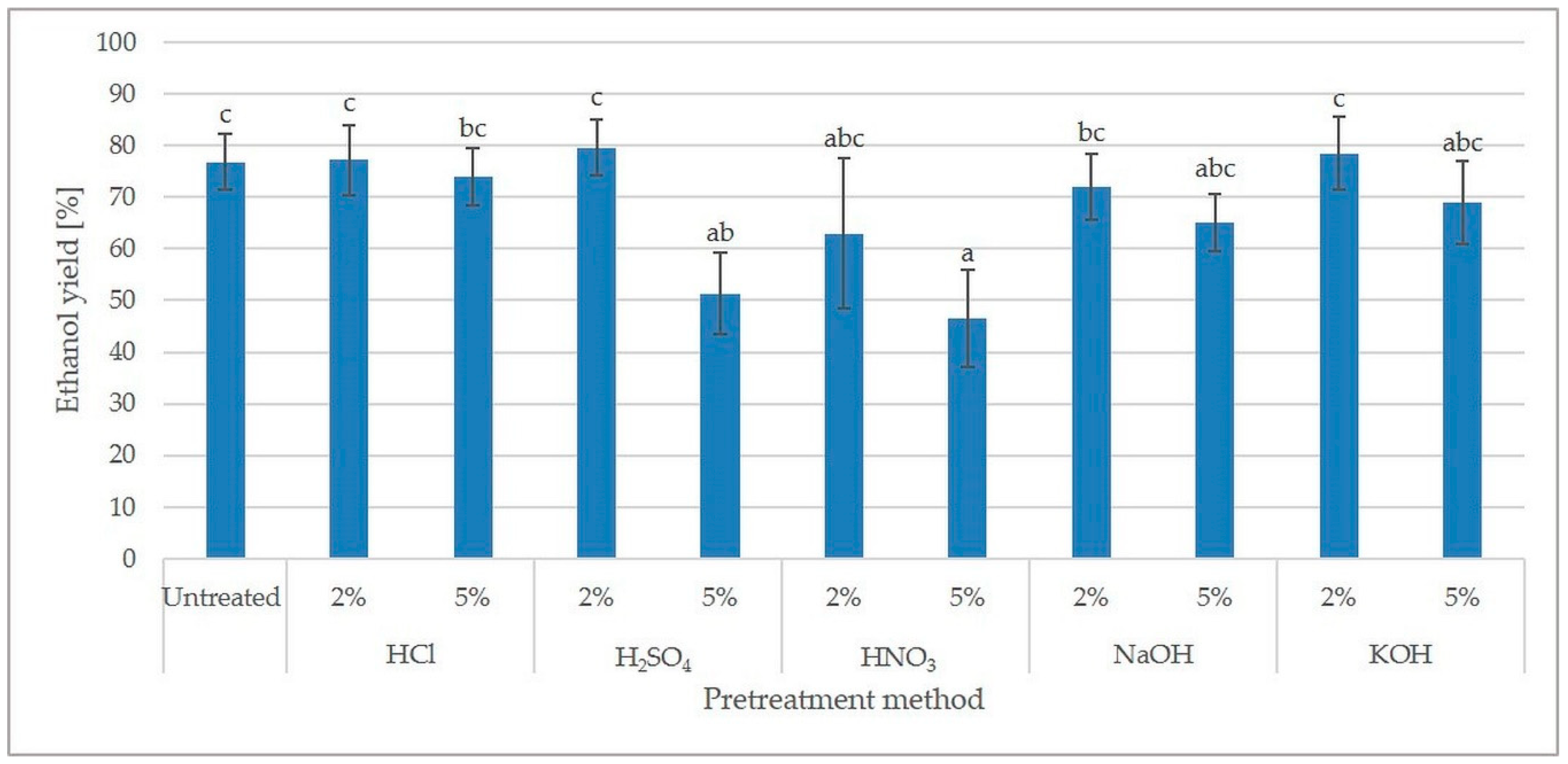
2.3. Concentration of Inhibitors and Fermentation of Hydrolysates
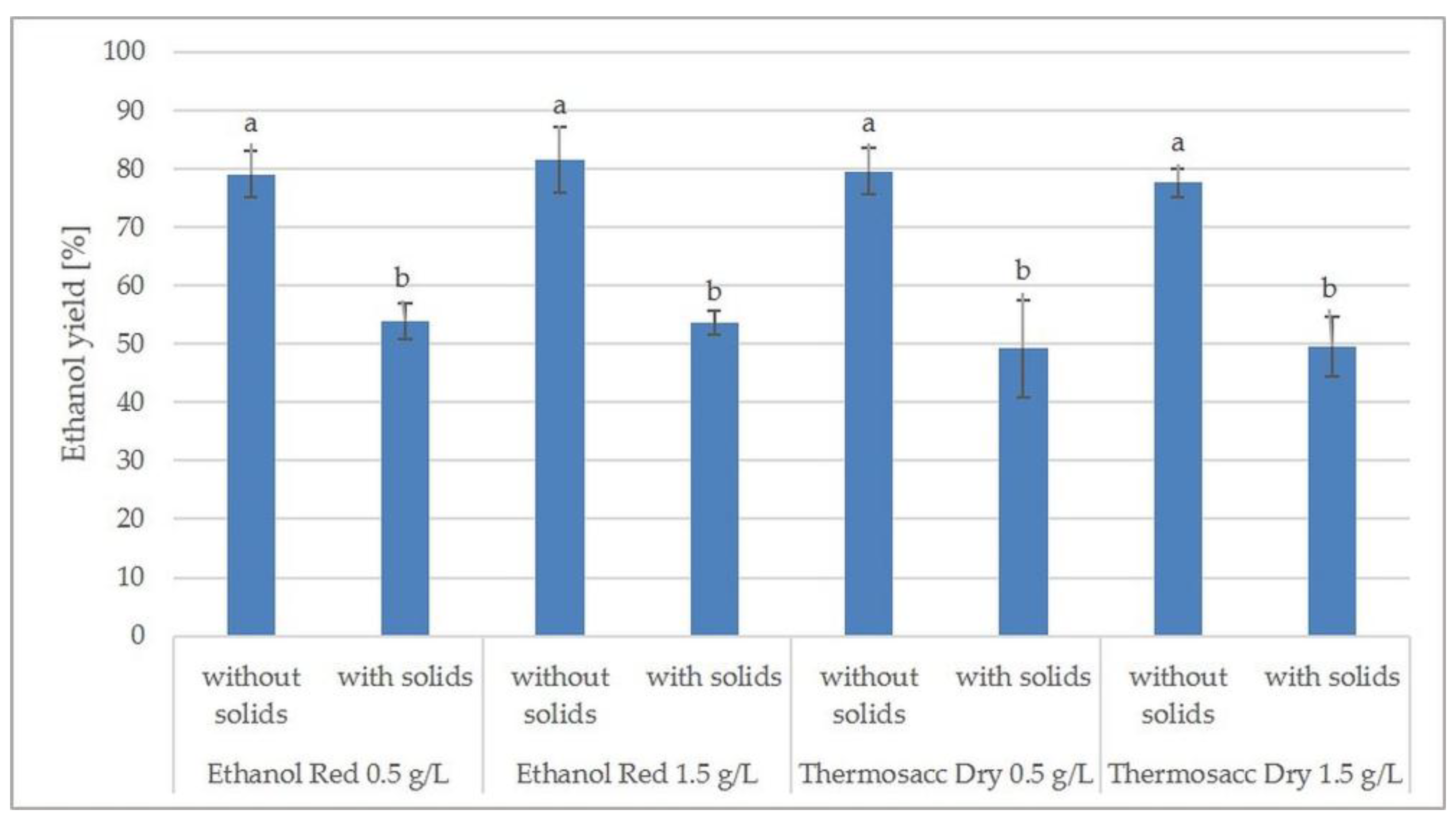
Fermentation of Nitric Acid Pretreated Hydrolysates—The Effect of Yeast Strain, Inoculum Size and Presence of Solids Residues
3. Materials and Methods
3.1. Materials
3.1.1. Plant Biomass
3.1.2. Enzymes and Chemicals
3.1.3. Yeasts
3.2. Pretreatment
3.3. Enzymatic Hydrolysis and Fermentation
3.4. Analysis and Calculations
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Eyidogan, M.; Kilic, F.C.; Kaya, D.; Coban, V.; Cagman, S. Investigation of Organic Rankine Cycle (ORC) technologies in Turkey from the technical and economic point of view. Renew. Sustain. Energy Rev. 2016, 58, 885–895. [Google Scholar] [CrossRef]
- Zecchina, A. Energy sources and carbon dioxide waste. Rend. Fis. Acc. Lincei 2014, 25, 113–117. [Google Scholar] [CrossRef]
- Rastogi, M.; Shrivastava, S. Recent advances in second generation bioethanol production: An insight to pretreatment, saccharification and fermentation processes. Renew. Sustain. Energ. Rev. 2017, 80, 330–340. [Google Scholar] [CrossRef]
- Wilson, P.; Glithero, N.J.; Ramsden, S.J. Prospects for dedicated energy crop production and attitudes towards agricultural straw use: The case of livestock farmers. Energy Policy 2014, 74, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Long, X.H.; Shao, H.B.; Liu, L.; Liu, L.P.; Liu, Z.P. Jerusalem artichoke: A sustainable biomass feedstock for biorefinery. Renew. Sustain. Energ. Rev. 2016, 54, 1382–1388. [Google Scholar] [CrossRef]
- Yang, L.; He, Q.S.; Corscadden, K.; Udenigwe, C.C. The prospects of Jerusalem artichoke in functional food ingredients and bioenergy production. Biotechnol. Rep. 2015, 5, 77–88. [Google Scholar] [CrossRef] [PubMed]
- Negro, M.J.; Ballesteros, I.; Manzanares, P.; Oliva, J.M.; Sáez, F.; Ballesteros, M. Inulin containing biomass for ethanol production: Carbohydrate extraction and ethanol fermentation. Appl. Biochem. Biotechnol. 2006, 132, 922–932. [Google Scholar] [CrossRef]
- Panchev, I.; Delchev, N.; Kovacheva, D.; Slavov, A. Physicochemical characteristics of inulins obtained from Jerusalem artichoke (Helianthus tuberosus L.). Eur. Food Res. Technol. 2011, 233, 889–896. [Google Scholar] [CrossRef]
- Roberfroid, M.B. Functional food: Concepts and application to inulin and oligofructose. Br. J. Nutr. 2002, 87, S139–S143. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.Z.; Zou, S.M.; He, M.L.; Wang, C.H. Bioethanol production from the dry powder of Jerusalem artichoke tubers by recombinant Saccharomyces cerevisiae in simultaneous saccharification and fermentation. J. Ind. Microbiol. Biotechnol. 2015, 42, 543–551. [Google Scholar] [CrossRef] [PubMed]
- Matías, J.; Encinar, J.M.; González, J.; González, J.F. Optimisation of ethanol fermentation of Jerusalem artichoke tuber juice using simple technology for a decentralised and sustainable ethanol production. Energy Sustain. Dev. 2015, 25, 34–39. [Google Scholar] [CrossRef]
- Guan, W.; Xu, G.; Duan, J.; Shi, S. Acetone–Butanol–Ethanol Production from Fermentation of Hot-Water-Extracted Hemicellulose Hydrolysate of Pulping Woods. Ind. Eng. Chem. Res. 2018, 57, 775–783. [Google Scholar] [CrossRef]
- Braun, M.; Antonietti, M. A continuous flow process for the production of 2,5-dimethylfuran from fructose using (non-noble metal based) heterogeneous catalysis. Green Chem. 2017, 19, 3813–3819. [Google Scholar] [CrossRef]
- Hailong, C.; Qishun, L.; Shuguang, L.; Zhongbao, Z.; Yuguang, D. Helianthus tuberosus—A good kind of biomass source for dimethylfuran production. J. Biotechnol. 2008, 136, 271–272. [Google Scholar] [CrossRef]
- Sarchami, T.; Rehmann, L. Optimizing Acid Hydrolysis of Jerusalem Artichoke-Derived Inulin for Fermentative Butanol Production. Bioenergy Res. 2015, 8, 1148. [Google Scholar] [CrossRef]
- Kumar, P.; Barrett, D.M.; Delwiche, M.J.; Stroeve, P. Methods for pretreatment of lignocellulosic biomass for efficient hydrolysis and biofuel production. Ind. Eng. Chem. Res. 2009, 48, 3713–3729. [Google Scholar] [CrossRef]
- Xiao, W.; Yin, W.; Xia, S.; Ma, P. The study of factors affecting the enzymatic hydrolysis of cellulose after ionic liquid pretreatment. Carbohydr. Polym. 2012, 87, 2019–2023. [Google Scholar] [CrossRef]
- Kim, S.; Park, J.M.; Kim, C.H. Ethanol production using whole plant biomass of Jerusalem artichoke by Kluyveromyces marxianus CBS1555. Appl. Biochem. Biotechnol. 2013, 169, 1531–1545. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, C.H. Evaluation of whole Jerusalem artichoke (Helianthus tuberosus L.) for consolidated bioprocessing ethanol production. Renew. Energy 2014, 65, 83–91. [Google Scholar] [CrossRef]
- Khatun, M.M.; Li, Y.H.; Liu, C.G.; Zhao, X.Q.; Bai, F.W. Fed-batch saccharification and ethanol fermentation of Jerusalem artichoke stalks by an inulinase producing Saccharomyces cerevisiae MK01. RSC Adv. 2015, 5, 107112–107118. [Google Scholar] [CrossRef]
- Song, Y.; Wi, S.G.; Kim, H.M.; Bae, H.-J. Cellulosic bioethanol production from Jerusalem artichoke (Helianthus Tuberosus L.) using hydrogen peroxide-acetic acid (HPAC) pretreatment. Bioresour. Technol. 2016, 214, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Wyman, C. Pretreatment: The key to Unlocking Low-Cost Cellulosic Ethanol. Biofuels Bioprod. Biorefin. 2008, 2, 26–40. [Google Scholar] [CrossRef]
- Mosier, N.; Wyman, C.; Dale, B.; Elander, R.; Lee, Y.Y.; Holtzapple, M.; Ladisch, M. Features of promising technologies for pretreatment of lignocellulosic biomass. Bioresour. Technol. 2005, 96, 673–686. [Google Scholar] [CrossRef]
- Sannigrahi, P.; Kim, D.H.; Jung, S.; Ragauskas, A. Pseudo-lignin and pretreatment chemistry. Energy Environ. Sci. 2011, 4, 1306–1310. [Google Scholar] [CrossRef]
- Digman, M.F.; Shinners, K.J.; Casler, M.D. Optimizing on-farm pretreatment of perennial grasses for fuel ethanol production. Bioresour. Technol. 2010, 101, 5305–5314. [Google Scholar] [CrossRef] [PubMed]
- Karapatsia, A.; Pappas, I.; Penloglou, G.; Kotrotsiou, O.; Kiparissides, C. Optimization of dilute acid pretreatment and enzymatic hydrolysis of Phalaris aquatica L. lignocellulosic biomass in batch and fed-batch processes. Bioenergy Res. 2017, 10, 225–236. [Google Scholar] [CrossRef]
- Deshavath, N.N.; Dasu, V.V.; Goud, V.V.; Rao, P.S. Development of dilute sulfuric acid pretreatment method for the enhancement of xylose fermentability. Biocatal. Agric. Biotechnol. 2017, 11, 224–230. [Google Scholar] [CrossRef]
- Rodíguez-Chong, A.; Ramírez, J.A.; Garrote, G.; Vázquez, M. Hydrolysis of sugar cane bagasse using nitric acid: A kinetic assessment. J. Food Eng. 2004, 61, 143–152. [Google Scholar] [CrossRef]
- Zhang, R.; Lu, X.; Sun, Y.; Wang, X.; Zhang, S. Modeling and optimization of dilute nitric acid hydrolysis on corn stover. J. Chem. Technol. Biotechnol. 2011, 86, 306–314. [Google Scholar] [CrossRef]
- Kim, I.; Lee, B.; Park, J.; Choi, S.; Han, J. Effect of nitric acid on pretreatment and fermentation for enhancing ethanol production of rice straw. Carbohydr. Polym. 2014, 99, 563–567. [Google Scholar] [CrossRef] [PubMed]
- Maurya, D.P.; Singla, A.; Negi, S. An overview of key pretreatment processes for biological conversion of lignocellulosic biomass to bioethanol. 3 Biotech 2015, 5, 597–609. [Google Scholar] [CrossRef] [PubMed]
- Guan, W.; Shi, S.; Blersch, D. Effects of Tween 80 on fermentative butanol production from alkali-pretreated switchgrass. Biochem. Eng. J. 2018, 135, 61–70. [Google Scholar] [CrossRef]
- Umagiliyage, A.L.; Choudhary, R.; Liang, Y.; Haddock, J.; Watson, D.G. Laboratory scale optimization of alkali pretreatment for improving enzymatic hydrolysis of sweet sorghum bagasse. Ind. Crops Prod. 2015, 74, 977–986. [Google Scholar] [CrossRef]
- Berłowska, J.; Pielech-Przybylska, K.; Balcerek, M.; Dziekońska-Kubczak, U.; Patelski, P.; Dziugan, P.; Kręgiel, D. Simultaneous saccharification and fermentation of sugar beet pulp for efficient bioethanol production. BioMed Res. Int. 2016, 2016, 3154929. [Google Scholar] [CrossRef]
- Gonzales, R.R.; Sivagurunathan, P.; Kim, S.H. Effect of severity on dilute acid pretreatment of lignocellulosic biomass and the following hydrogen fermentation. Int. J. Hydrogen Energy 2016, 41, 21678–21684. [Google Scholar] [CrossRef]
- McIntosh, S.; Vancov, T. Enhanced enzyme saccharification of Sorghum bicolor straw using dilute alkali pretreatment. Bioresour. Technol. 2010, 101, 6718–6727. [Google Scholar] [CrossRef] [PubMed]
- Sluiter, A.; Hames, B.; Hyman, D.; Payne, C.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D. Determination of Total Solids in Biomass and Total Dissolved Solids in Liquid Process Samples; Technical Report No. NREL/TP-510-42621; NREL: Golden, CO, USA, 2008.
- Kurschner, K.; Hoffer, A. Cellulose and cellulose derivatives. Fresenius J. Anal. Chem. 1993, 92, 145–154. [Google Scholar] [CrossRef]
- Arasimovich, V.V.; Ermakov, A.I. Measurement of the total content of hemielluloses. In Methods for Biochemical Studies of Plants; Ermakov, A.I., Ed.; Agropromizdat: Saint Petersburg, Russia, 1987; pp. 164–165. [Google Scholar]
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D.; Crocker, D. Determination of Structural Carbohydrates and Lignin in Biomass; Technical Report No. NREL/TP-510-42618; NREL: Golden, CO, USA, 2012.
- Ma, S.; Wang, H.; Wang, Y.; Bu, H.; Bai, J. Bio-hydrogen production from cornstalk wastes by orthogonal design method. Renew. Energy 2011, 36, 709–713. [Google Scholar] [CrossRef]
- Imman, S.; Arnthong, J.; Burapatana, V.; Champreda, V.; Laosiripojana, N. Fractionation of rice straw by a single-step solvothermal process: Effects of solvents, acid promoters, and microwave treatment. Renew. Energy 2015, 83, 663–673. [Google Scholar] [CrossRef]
- Valdez-Vazquez, I.; Pérez-Rangel, M.; Tapia, A.; Buitrón, G.; Molina, C.; Hernández, G.; Amaya-Delgado, L. Hydrogen and butanol production from native wheat straw by synthetic microbial consortia integrated by species of Enterococcus and Clostridium. Fuel 2015, 159, 214–222. [Google Scholar] [CrossRef]
- Hendriks, A.T.W.M.; Zeeman, G. Pretreatments to enhance the digestibility of lignocellulosic biomass. Bioresour. Technol. 2009, 100, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Jönsson, L.J.; Martín, C. Pretreatment of lignocellulose: formation of inhibitory by-products and strategies for minimizing their effects. Bioresour. Technol. 2016, 199, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Herrera, A.; Tellez-Luis, S.J.; Gonzalez-Cabriales, J.J.; Ramírez, J.A.; Vazquez, M. Effect of the hydrochloric acid concentration on the hydrolysis of sorghum straw at atmospheric pressure. J. Food Eng. 2004, 63, 103–109. [Google Scholar] [CrossRef]
- Chang, V.S.; Holtzapple, M.T. Fundamental factors affecting biomass enzymatic reactivity. Appl. Biochem. Biotechnol. 2000, 84–86, 5–37. [Google Scholar] [CrossRef]
- Kim, D.; Orrego, D.; Ximenes, E.A.; Ladisch, M.R. Cellulose conversion of corn pericarp without pretreatment. Bioresour. Technol. 2017, 245, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Ximenes, E.; Kim, Y.; Mosier, N.; Dien, B.; Ladisch, M. Inhibition of cellulases by phenols. Enzym. Microb. Technol. 2010, 46, 170–176. [Google Scholar] [CrossRef]
- Ximenes, E.; Kim, Y.; Mosier, N.; Dien, B.; Ladisch, M. Deactivation of cellulases by phenols. Enzym. Microb. Technol. 2011, 48, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Martín, C.; García, A.; Schreiber, A.; Puls, J.; Saake, B. Combination of water extraction with dilute-sulphuric acid pretreatment for enhancing the enzymatic hydrolysis of Jatropha curcas shells. Ind. Crop. Prod. 2015, 64, 233–241. [Google Scholar] [CrossRef]
- Cheng, Y.S.; Zheng, Y.; Yu, C.W.; Dooley, T.M.; Jenkins, B.M.; Gheynst, J.S.V. Evaluation of high solids alkaline pretreatment of rice straw. Appl. Biochem. Biotechnol. 2010, 162, 1768–1784. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.M.; El-Zawawy, W.K.; Abdel-Fattah, Y.R.; Soliman, N.A.; Agblevor, F.A. Comparison of alkaline pulping with steam explosion for glucose production from rice straw. Carbohydr. Polym. 2011, 83, 720–726. [Google Scholar] [CrossRef]
- Wang, Z.; Keshwani, D.R.; Redding, A.P.; Cheng, J.J. Sodium hydroxide pretreatment and enzymatic hydrolysis of coastal Bermuda grass. Bioresour. Technol. 2010, 101, 3583–3585. [Google Scholar] [CrossRef] [PubMed]
- Jönsson, L.J.; Björn, A.; Nilvebrant, N.O. Bioconversion of Lignocellulose: Inhibitors and Detoxification. Biotechnol. Biofuels 2013, 6, 16. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.; Palmqvist, E.; Hahn-Hägerdal, B.; Tengborg, C.; Stenberg, K.; Zacchi, G.; Nilvebrant, N.O. The generation of fermentation inhibitors during dilute acid hydrolysis of softwood. Enzym. Microb. Technol. 1999, 24, 151–159. [Google Scholar] [CrossRef]
- Farías-Sánchez, J.C.; Velázquez-Valadez, U.; Pineda-Pimentel, M.G.; López-Miranda, J.; Castro-Montoya, A.J.; Carrillo-Parra, A.; Vargas-Santillán, A.; Rutiaga-Quiñones, J.G. Simultaneous Saccharification and Fermentation of Pine Sawdust (Pinus pseudostrobus L.) Pretreated with Nitric Acid and Sodium Hydroxide for Bioethanol Production. Bioresources 2016, 12, 1052–1063. [Google Scholar] [CrossRef]
- Luo, C.; Brink, D.L.; Blanch, H.W. Identification of potential fermentation inhibitors in conversion of hybrid poplar hydrolysate to ethanol. Biomass Bioenergy 2002, 22, 125–138. [Google Scholar] [CrossRef]
- Modelska, M.; Berlowska, J.; Kregiel, D.; Cieciura, W.; Antolak, H.; Tomaszewska, J.; Binczarski, M.; Szubiakiewicz, E.; Witonska, I.A. Concept for Recycling Waste Biomass from the Sugar Industry for Chemical and Biotechnological Purposes. Molecules 2017, 22, 1544. [Google Scholar] [CrossRef] [PubMed]
- Adeboye, P.T.; Bettiga, M.; Olsson, L. The chemical nature of phenolic compounds determines their toxicity and induces distinct physiological responses in Saccharomyces cerevisiae in lignocellulose hydrolysates. AMB Express 2014, 4, 46. [Google Scholar] [CrossRef] [PubMed]
- Tutt, M.; Kikas, T.; Olt, J. Influence of different pretreatment methods on bioethanol production from wheat straw. Agron. Res. 2012, S1, 269–276. [Google Scholar]
- Azhar, S.H.M.; Abdulla, R.; Jambo, S.A.; Marbawi, H.; Gansau, J.A.; Faik, A.A.M.; Rodrigues, K.F. Yeasts in sustainable bioethanol production: A review. Biochem. Biophys. Rep. 2017, 10, 52–61. [Google Scholar] [CrossRef]
- Häggström, C.; Rova, U.; Brandberg, T.; Hodge, D. Integration of Ethanol Fermentation with Second Generation Biofuels Technologies. In Biorefineries: Integrated Biochemical Processes for Liquid Biofuels; Qureshi, N., Hodge, D., Vertès, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; Chapter 8; pp. 161–187. ISBN 978-0-444-59498-3. [Google Scholar]
- Monot, F.; Duplan, J.L.; Alazard-Toux, N.; His, S. Biofuels. In Renewable Energies; Sabonnadière, J.C., Ed.; ISTE Ltd.: London, UK; John Wiley & Sons, Inc.: Hoboken, NY, USA, 2009; Chapter 10; pp. 329–395. ISBN 9781848211353. [Google Scholar]
- Cannella, D.; Jørgensen, H. Do new cellulolytic enzyme preparations affect the industrial strategies for high solids lignocellulosic ethanol production? Biotechnol. Bioeng. 2014, 111, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Balcerek, M.; Pielech-Przybylska, K.; Dziekońska-Kubczak, U.; Patelski, P.; Strąk, E. Fermentation results and chemical composition of agricultural distillates obtained from rye and barley grains and the corresponding malts as a source of amylolytic enzymes and starch. Molecules 2016, 21, 1320. [Google Scholar] [CrossRef] [PubMed]
- Antolak, H.; Czyzowska, A.; Sakač, M.; Mišan, A.; Đuragić, O.; Kregiel, D. Phenolic compounds contained in little-known wild fruits as antiadhesive agents against the beverage-spoiling bacteria Asaia spp. Molecules 2017, 22, 1256. [Google Scholar] [CrossRef] [PubMed]
| Pretreatment Method | Solid Recovery (%) | Cellulose | Hemicellulose | Lignin |
|---|---|---|---|---|
| (% d.m.) | ||||
| 2% HCl | 74.75 ± 2.13e 1 | 56.03 ± 1.37bcd | 5.41 ± 0.41bcd | 23.66 ± 1.66bcd |
| 5% HCl | 59.51 ± 2.01ab | 54.53 ± 1.99bc | 2.31 ± 0.22abc | 25.74 ± 1.20d |
| 2% H2SO4 | 73.52 ± 1.82d | 53.09 ± 2.46b | 4.93 ± 1.02bcd | 24.98 ± 2.35cd |
| 5% H2SO4 | 60.27 ± 1.98ab | 42.16 ± 1.80a | 1.91 ± 0.23ab | 27.41 ± 1.47d |
| 2% HNO3 | 56.62 ± 1.18a | 60.31 ± 1.67cde | 5.78 ± 0.27cd | 22.96 ± 1.09bcd |
| 5% HNO3 | 66.27 ± 1.42c | 77.27 ± 1.32f | 1.31 ± 0.16a | 20.70 ± 2.02bc |
| 2% NaOH | 74.83 ± 2.65d | 59.30 ± 1.97cde | 9.64 ± 0.66e | 6.47 ± 1.22a |
| 5% NaOH | 66.29 ± 2.03c | 62.75 ± 2.34e | 8.17 ± 0.70de | 4.77 ± 0.36a |
| 2% KOH | 72.48 ± 1.66d | 56.28 ± 1.98bcd | 10.43 ± 1.50e | 6.90 ± 0.93a |
| 5% KOH | 64.86 ± 2.02bc | 59.98 ± 2.35de | 8.70 ± 0.61de | 5.23 ± 0.64a |
| Pretreatment Method | Cellobiose | Glucose | Xylose | Arabinose | Hemicellulose Solubilization Rate (%) | Cellulose Solubilization Rate (%) |
|---|---|---|---|---|---|---|
| (g/L) | ||||||
| 2% HCl | 0.41 ± 0.04bcd 1 | 2.20 ± 0.19a | 9.38 ± 0.81b | 0.53 ± 0.05ab | 68.76 ± 5.92b | 5.64 ± 0.49a |
| 5% HCl | 0.61 ± 0.12de | 2.73 ± 0.53a | 11.96 ± 1.32bc | 0.84 ± 0.16c | 87.70 ± 7.04bc | 7.02 ± 1.36a |
| 2% H2SO4 | 0.64 ± 0.09e | 2.63 ± 0.39a | 10.84 ± 1.21bc | 0.47 ± 0.07ab | 79.52 ± 6.72bc | 6.76 ± 1.00a |
| 5% H2SO4 | 0.54 ± 0.06cde | 2.80 ± 0.30a | 11.35 ± 0.90bc | 0.68 ± 0.07bc | 83.23 ± 5.81bc | 7.19 ± 0.76a |
| 2% HNO3 | 0.09 ± 0.01a | 2.45 ± 0.22a | 9.41 ± 0.85b | 0.55 ± 005ab | 69.01 ± 6.26b | 6.30 ± 0.57a |
| 5% HNO3 | 1.12 ± 0.10f | 5.70 ± 0.52b | 13.20 ± 1.19c | 1.29 ± 0.12d | 96.77 ± 8.75c | 14.66 ± 1.33b |
| 2% NaOH | 0.42 ± 0.05bcd | 2.95 ± 0.36a | n.d. 2 | 0.62 ± 0.07abc | 0.00 ± 0.00a | 7.60 ± 0.92a |
| 5% NaOH | 0.23 ± 0.01ab | 2.12 ± 0.07a | 0.29 ± 0.01a | 0.56 ± 0.02ab | 2.15 ± 0.07a | 5.45 ± 0.17a |
| 2% KOH | 0.37 ± 0.03bc | 2.51 ± 0.18a | 0.13 ± 0.01a | 0.43 ± 0.03a | 0.95 ± 0.07a | 6.45 ± 0.47a |
| 5% KOH | 0.30 ± 0.02abc | 2.99 ± 0.17a | 0.31 ± 0.02a | 0.52 ± 0.03abc | 2.26 ± 0.13a | 7.69 ± 0.44a |
| Pretreatment Method | Formic Acid | Acetic Acid | TPC 1 | HMF 2 | Furfural |
|---|---|---|---|---|---|
| (g/L) | (mg/L) | ||||
| 2% HCl | 0.19 ± 0.02ab 3 | 1.16 ± 0.10a | 0.69 ± 0.06b | 201.61 ± 17.34c | 95.81 ± 8.24a |
| 5% HCl | 0.32 ± 0.06abc | 2.92 ± 0.57b | 0.55 ± 0.05a | 136.62 ± 26.55bc | 523.79 ± 81.79c |
| 2% H2SO4 | 0.13 ± 0.02a | 2.80 ± 0.41b | 0.87 ± 0.12c | 739.26 ± 108.93d | 95.49 ± 14.07a |
| 5% H2SO4 | 0.36 ± 0.04bc | 3.25 ± 0.34bc | 1.08 ± 0.08de | 220.65 ± 23.36c | 293.09 ± 31.03b |
| 2% HNO3 | 0.48 ± 0.04c | 1.76 ± 0.16a | 0.51 ± 0.06a | 153.95 ± 13.97bc | 8.33 ± 0.75a |
| 5% HNO3 | 1.18 ± 0.11d | 3.19 ± 0.29bc | 0.46 ± 0.04a | 14.00 ± 1.26a | 41.63 ± 3.76a |
| 2% NaOH | 1.16 ± 0.14d | 3.42 ± 0.41bc | 1.39 ± 0.11f | 1.42 ± 0.17a | 0.98 ± 0.12a |
| 5% NaOH | 2.86 ± 0.09f | 5.02 ± 0.16de | 1.12 ± 0.05e | 2.46 ± 0.07a | 16.43 ± 0.52a |
| 2% KOH | 0.97 ± 0.07d | 4.07 ± 0.30cd | 1.23 ± 0.10e | 1.04 ± 0.07a | 1.94 ± 0.08a |
| 5% KOH | 1.97 ± 0.11e | 6.05 ± 0.34e | 1.25 ± 0.11ef | 2.92 ± 0.17a | 10.17 ± 0.57a |
| Pretreatment Method | Cellobiose | Glucose | Xylose | Arabinose | Glucose Yield (%) |
|---|---|---|---|---|---|
| (g/L) | |||||
| Untreated | 0.01 ± 0.001a 1 | 2.62 ± 0.36a | 1.42 ± 0.28ab | 0.05 ± 0.01c | 13.27 ± 1.21a |
| 2% HCl | 0.05 ± 0.01ab | 15.70 ± 1.84b | 1.53 ± 0.45ab | 0.02 ± 0.00ab | 50.81 ± 1.71c |
| 5% HCl | 0.05 ± 0.02ab | 16.99 ± 2.50bc | 1.54 ± 0.16ab | 0.01 ± 0.01a | 55.57 ± 1.81cde |
| 2% H2SO4 | 0.02 ± 0.01a | 12.47 ± 0.88b | 1.90 ± 0.29ab | n.d. 2 | 41.64 ± 1.48b |
| 5% H2SO4 | 0.04 ± 0.01ab | 13.55 ± 1.99b | 1.28 ± 0.14a | 0.01 ± 0.00a | 57.64 ± 1.55de |
| 2% HNO3 | 0.06 ± 0.10ab | 17.87 ± 1.09bc | 2.56 ± 0.46bc | 0.01 ± 0.00a | 53.90 ± 1.33cd |
| 5% HNO3 | 0.21 ± 0.05b | 38.47 ± 2.97d | 2.68 ± 0.09c | n.d. | 89.32 ± 2.24g |
| 2% NaOH | 0.10 ± 0.04ab | 18.85 ± 2.88bc | 6.83 ± 0.44e | 0.04 ± 0.01c | 57.53 ± 1.67de |
| 5% NaOH | 0.10 ± 0.03ab | 23.63 ± 4.07c | 4.99 ± 0.60d | n.d. | 67.60 ± 1.51f |
| 2% KOH | 0.01 ± 0.00a | 16.33 ± 1.64b | 6.69 ± 0.68e | 0.02 ± 0.00ab | 52.14 ± 1.94c |
| 5% KOH | 0.01 ± 0.00ab | 20.67 ± 2.52bc | 8.47 ± 1.03e | 0.02 ± 0.00ab | 62.27 ± 1.68ef |
| Pretreatment Method | Formic Acid | Acetic Acid | HMF 1 | Furfural | TPC 2 |
|---|---|---|---|---|---|
| (mg/L) | |||||
| Untreated | n.d. 3 | 1.11 ± 0.09a | n.d. | n.d. | n.d. |
| 2% HCl | 6.41 ± 0.65bc 4 | 29.99 ± 3.05f | 2.87 ± 0.22ab | 7.78 ± 0.48e | 162.11 ± 19.15cde |
| 5% HCl | 8.26 ± 0.66cd | 35.04 ± 2.25g | 3.40 ± 0.38b | 6.68 ± 0.44de | 178.09 ± 13.34ef |
| 2% H2SO4 | 12.29 ± 0.66e | 12.36 ± 0.82bc | 12.29 ± 1.41d | 5.40 ± 0.42bcd | 173.83 ± 12.56de |
| 5% H2SO4 | 8.11 ± 0.55cd | 37.44 ± 1.34g | 5.26 ± 0.66c | 6.18 ± 0.72cde | 155.96 ± 15.42cd |
| 2% HNO3 | 14.60 ± 0.76f | 128.58 ± 2.98i | 2.54 ± 0.25ab | 4.53 ± 0.12b | 196.91 ± 8.27f |
| 5% HNO3 | 9.48 ± 1.04d | 68.76 ± 2.97h | n.d. | 4.93 ± 0.90bc | 80.63 ± 8.11a |
| 2% NaOH | 19.62 ± 1.11g | 14.09 ± 1.27cd | n.d. | 6.39 ± 0.41cde | 128.04 ± 18.20b |
| 5% NaOH | 22.23 ± 0.72h | 8.31 ± 0.65b | n.d. | 2.83 ± 0.31a | 161.34 ± 14.13cde |
| 2% KOH | 2.57 ± 0.25a | 18.56 ± 1.08de | 2.75 ± 0.35ab | 7.15 ± 0.73e | 151.21 ± 19.11c |
| 5% KOH | 5.37 ± 0.99b | 20.32 ± 1.68e | 1.86 ± 0.24a | 6.86 ± 0.75de | 170.12 ± 17.15cde |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dziekońska-Kubczak, U.; Berłowska, J.; Dziugan, P.; Patelski, P.; Pielech-Przybylska, K.; Balcerek, M. Nitric Acid Pretreatment of Jerusalem Artichoke Stalks for Enzymatic Saccharification and Bioethanol Production. Energies 2018, 11, 2153. https://doi.org/10.3390/en11082153
Dziekońska-Kubczak U, Berłowska J, Dziugan P, Patelski P, Pielech-Przybylska K, Balcerek M. Nitric Acid Pretreatment of Jerusalem Artichoke Stalks for Enzymatic Saccharification and Bioethanol Production. Energies. 2018; 11(8):2153. https://doi.org/10.3390/en11082153
Chicago/Turabian StyleDziekońska-Kubczak, Urszula, Joanna Berłowska, Piotr Dziugan, Piotr Patelski, Katarzyna Pielech-Przybylska, and Maria Balcerek. 2018. "Nitric Acid Pretreatment of Jerusalem Artichoke Stalks for Enzymatic Saccharification and Bioethanol Production" Energies 11, no. 8: 2153. https://doi.org/10.3390/en11082153
APA StyleDziekońska-Kubczak, U., Berłowska, J., Dziugan, P., Patelski, P., Pielech-Przybylska, K., & Balcerek, M. (2018). Nitric Acid Pretreatment of Jerusalem Artichoke Stalks for Enzymatic Saccharification and Bioethanol Production. Energies, 11(8), 2153. https://doi.org/10.3390/en11082153







