Possible Interactions and Interferences of Copper, Chromium, and Arsenic during the Gasification of Contaminated Waste Wood †
Abstract
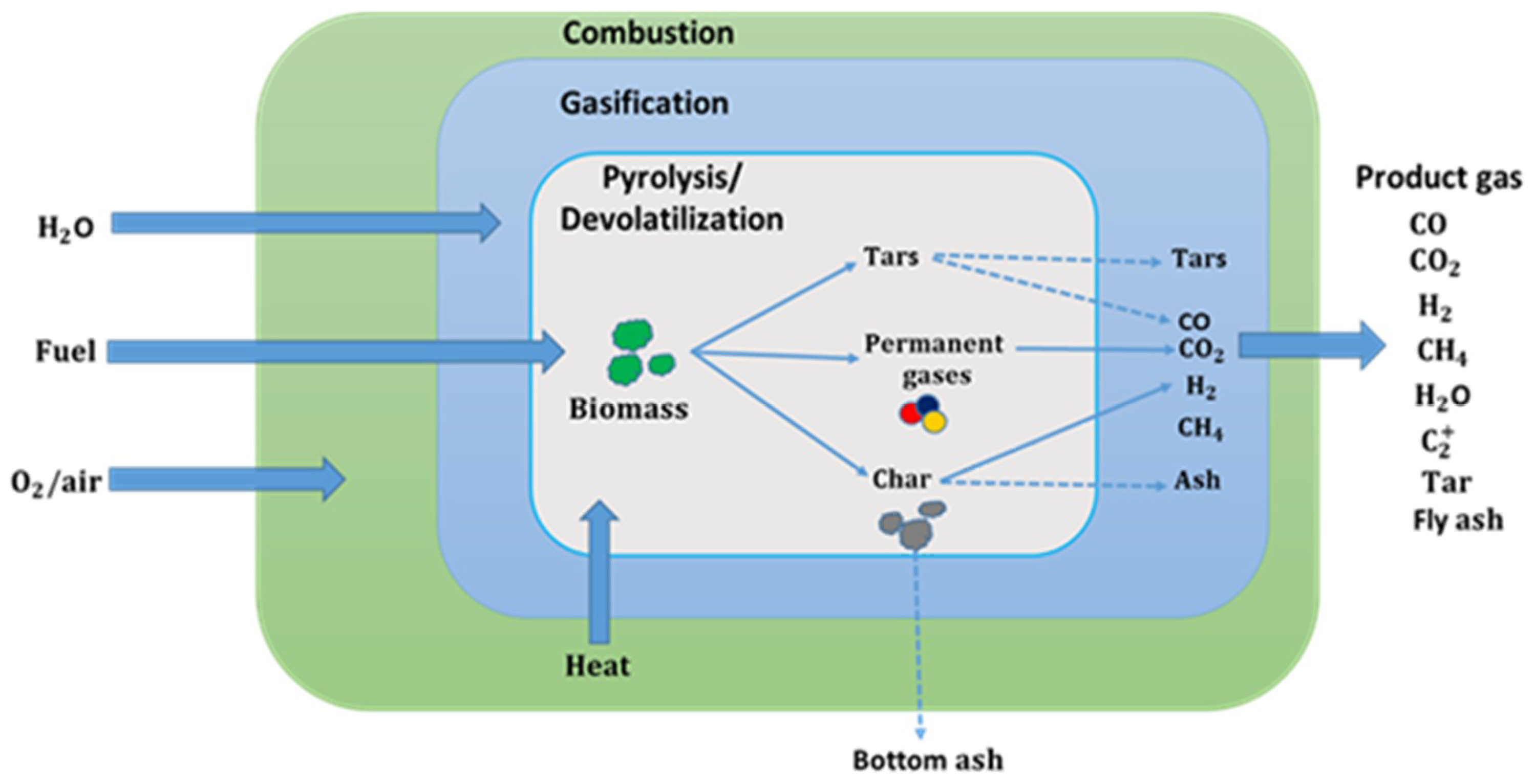
1. Introduction
2. Thermodynamic Equilibrium Modelling
3. Results and Discussion
3.1. Under Partial Combustion Reaction (PCR)
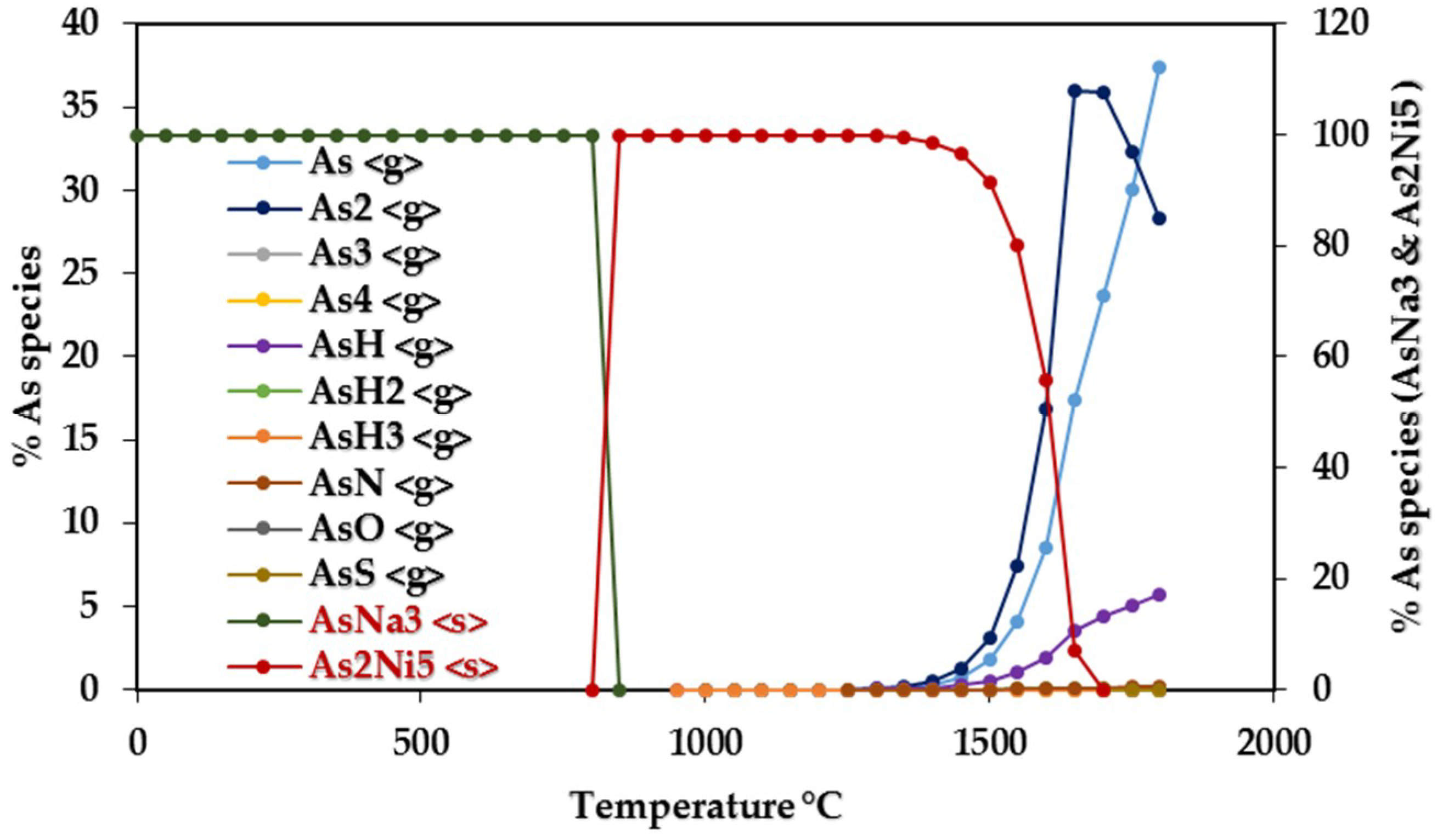
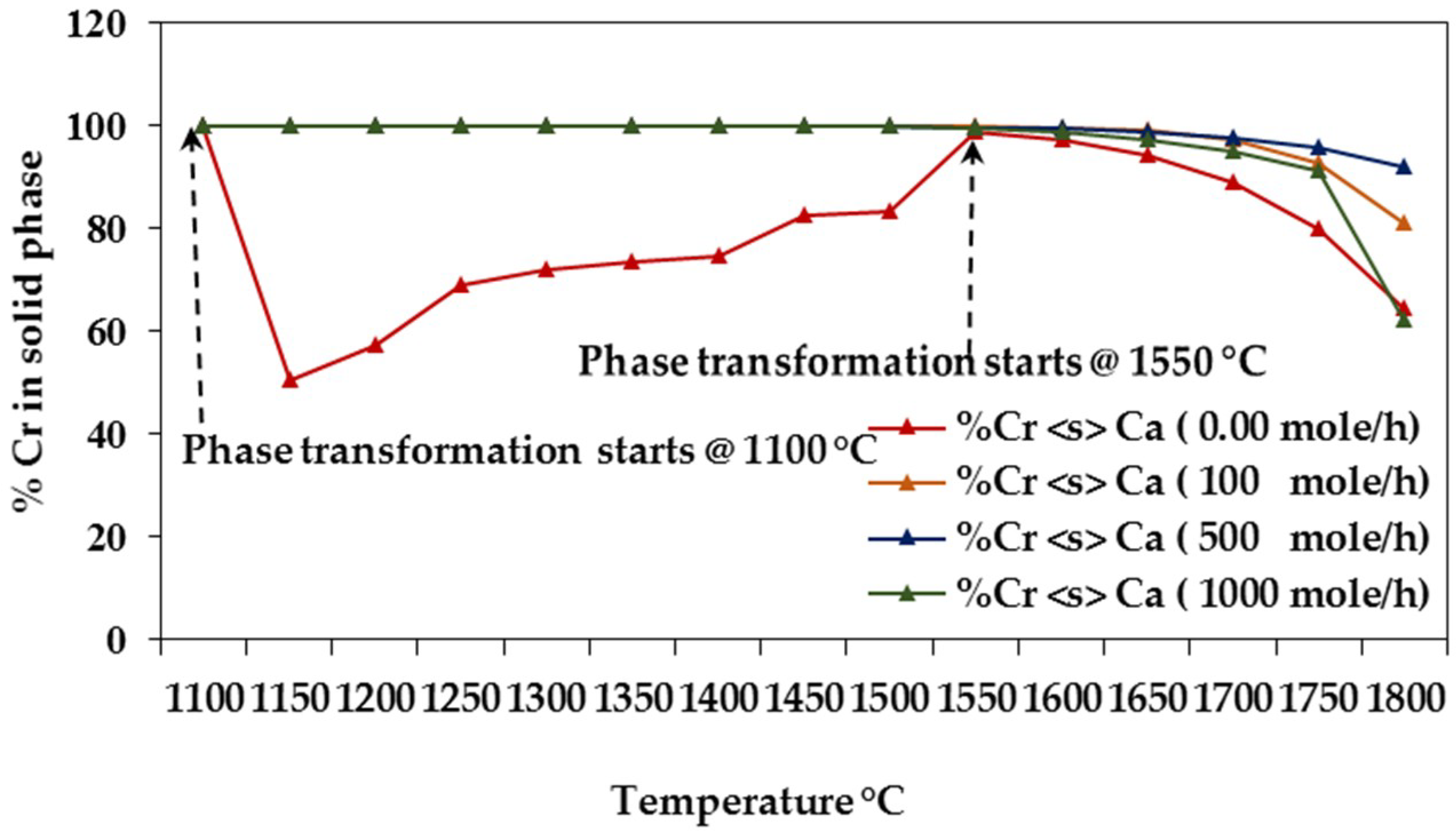
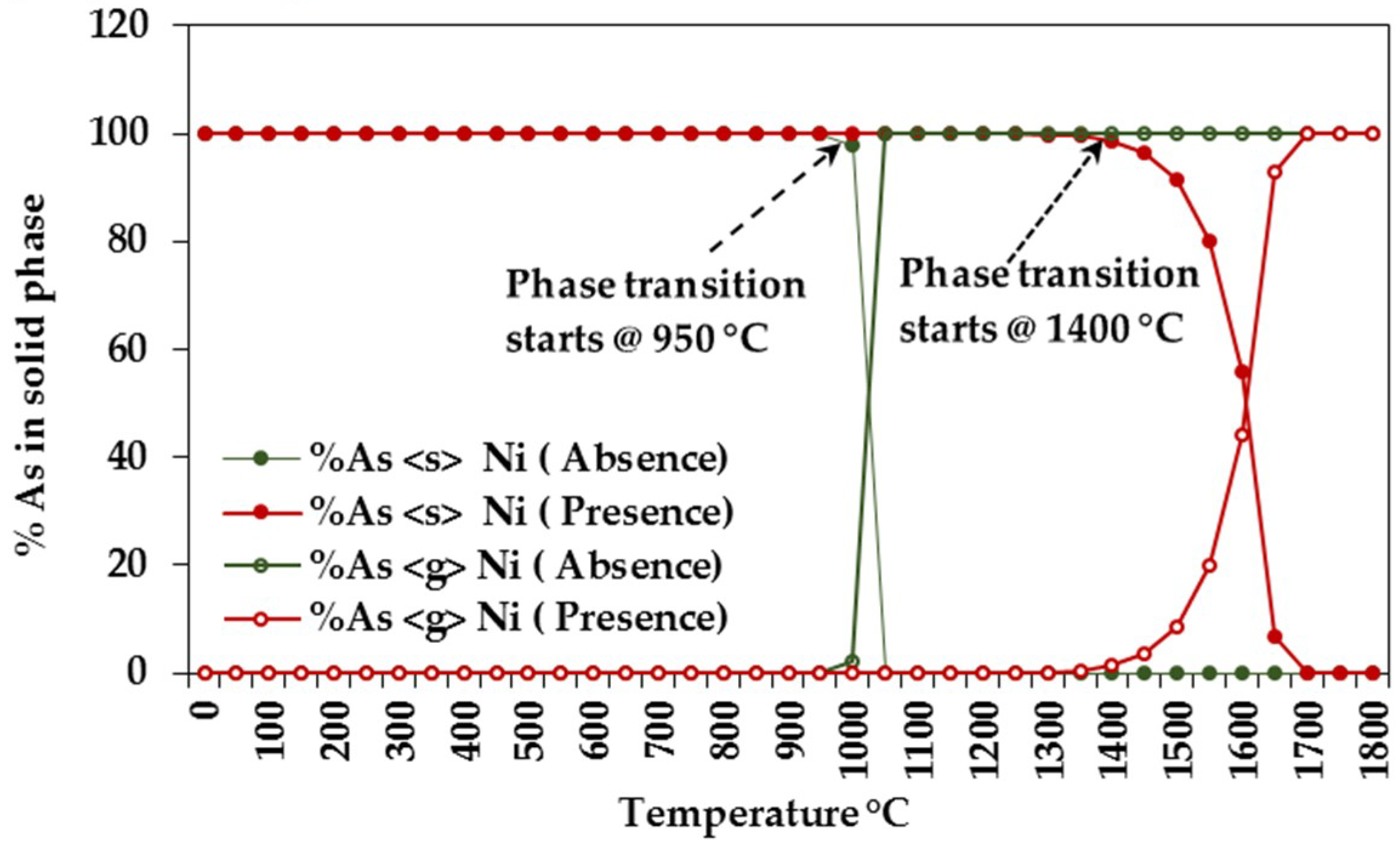
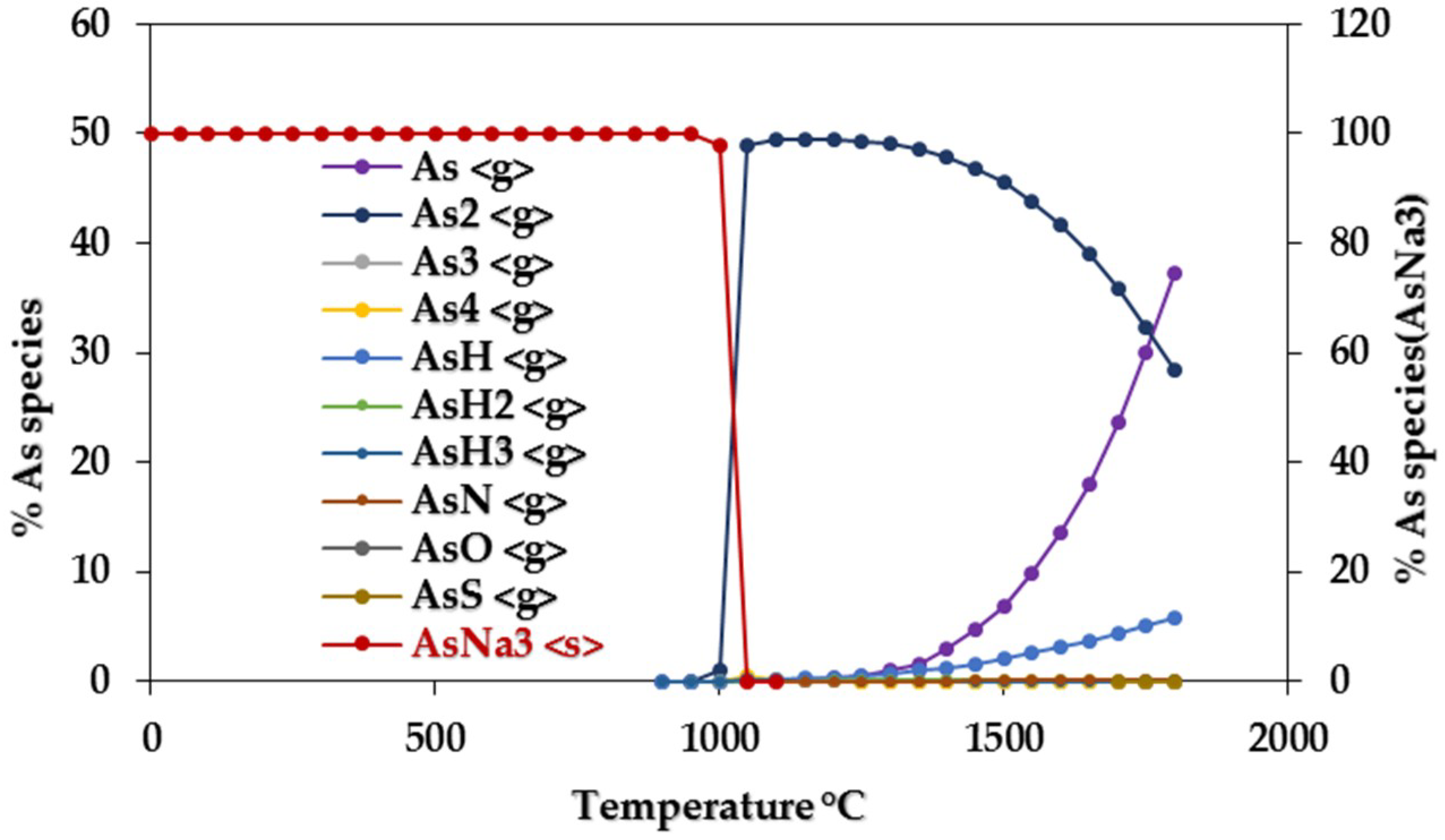
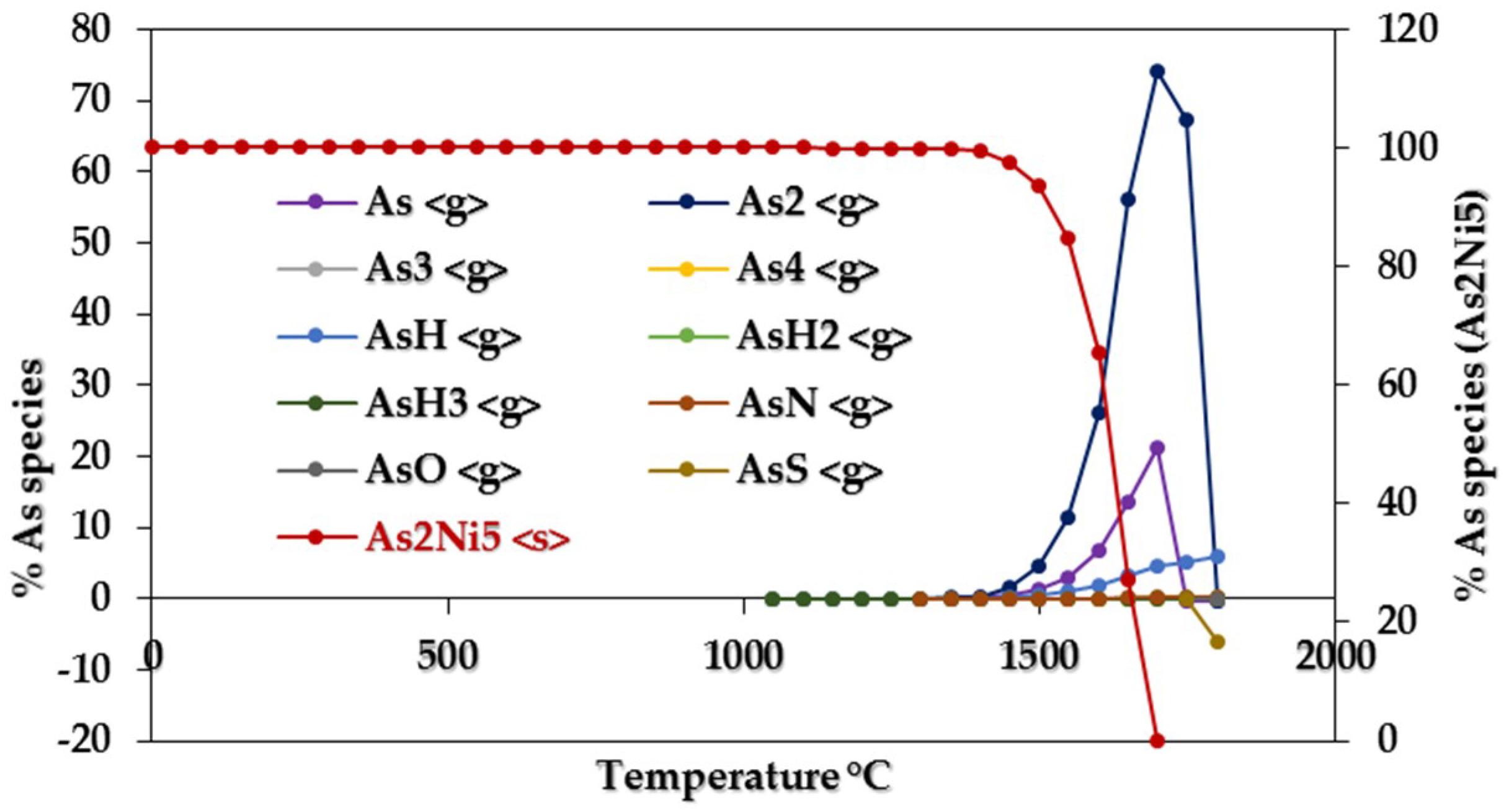
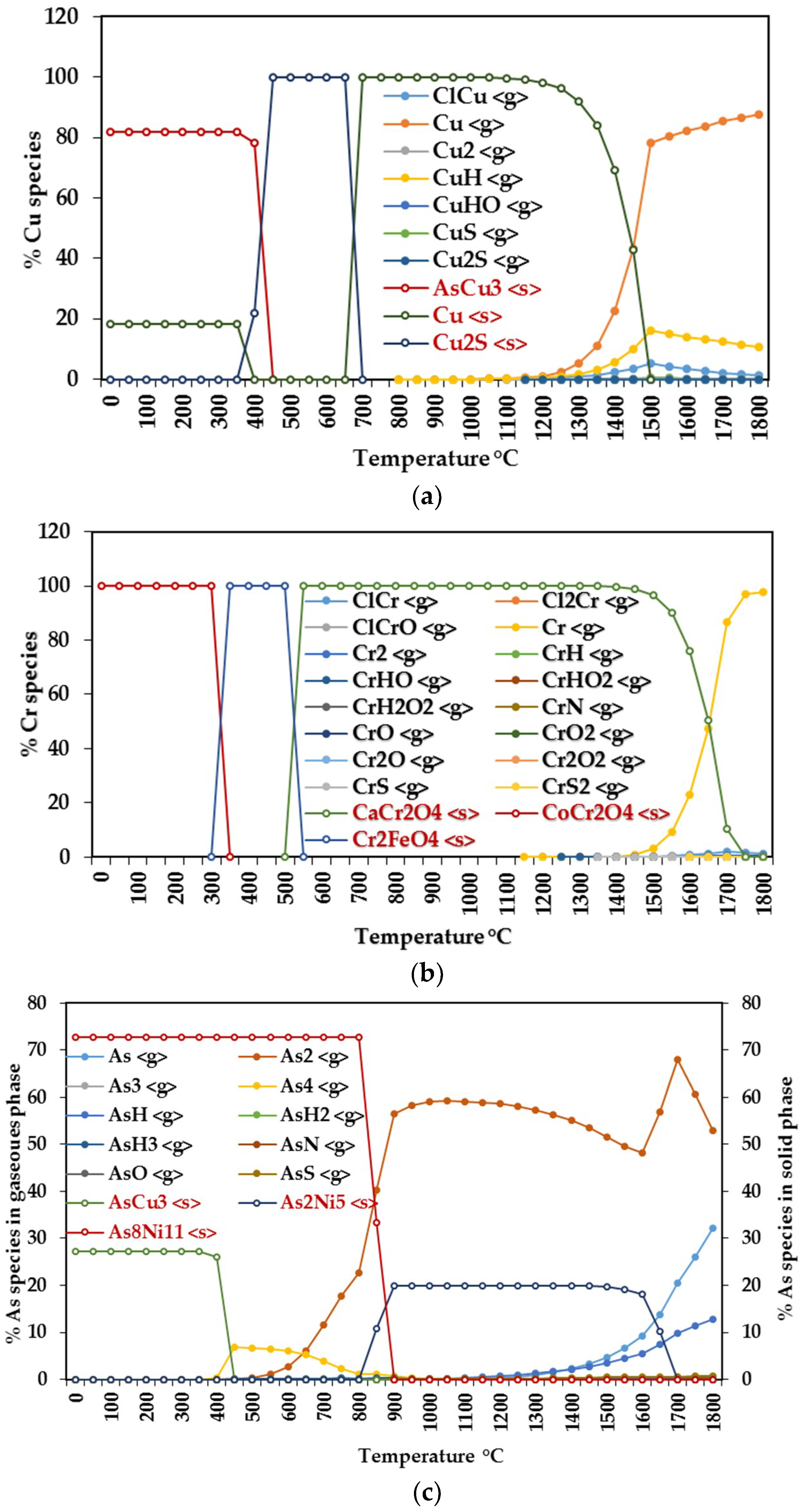
3.1.1. Equilibrium Composition of CCA-Elements
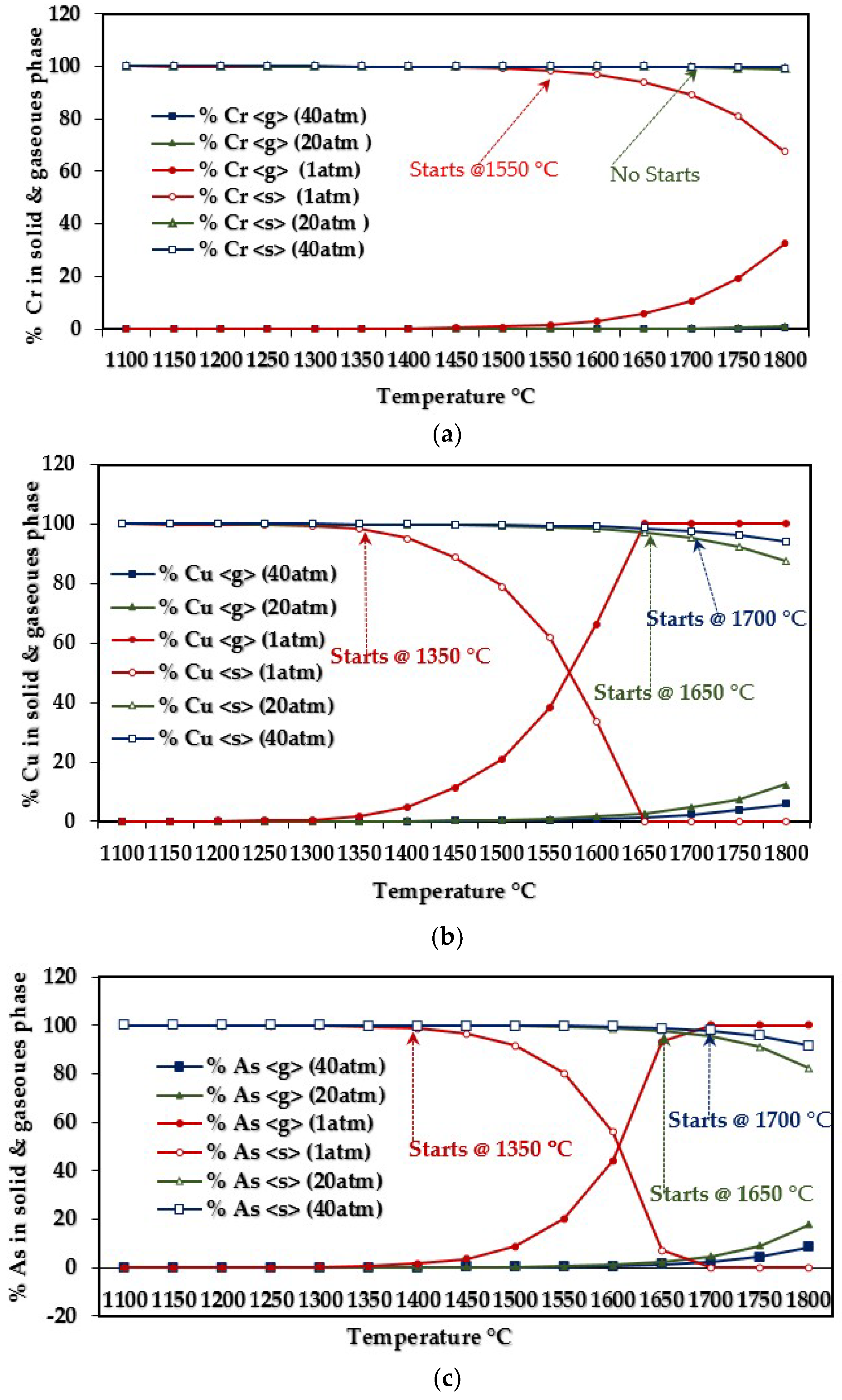
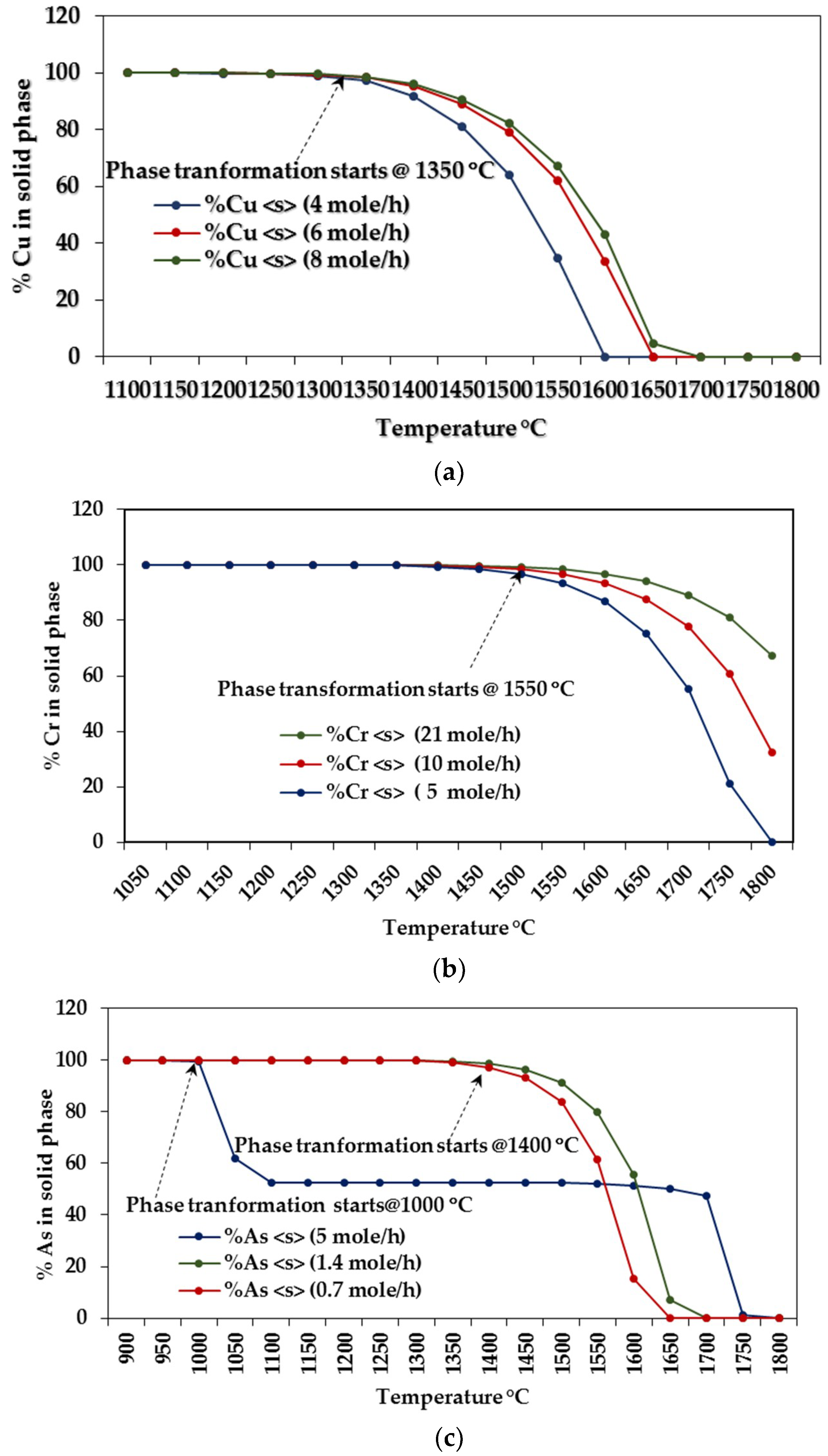
3.1.2. Influence of Elemental Contents
Influences on CCA-elements
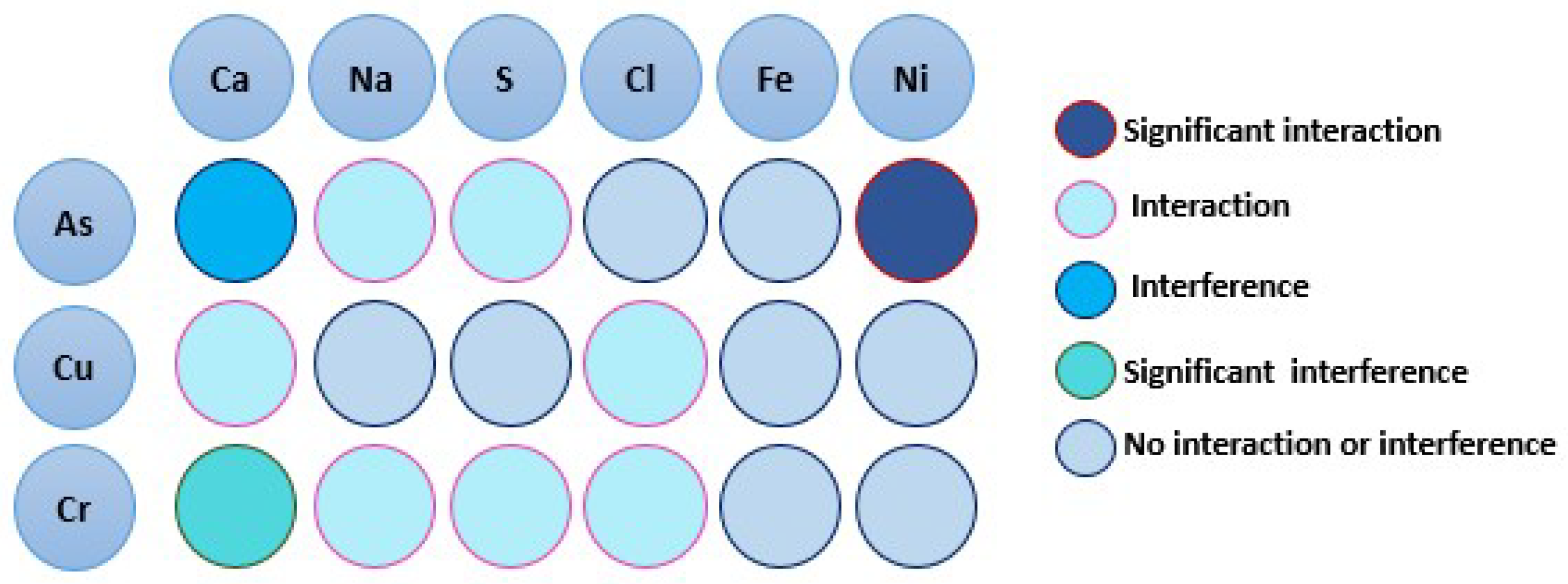
Influences of Ca, Na, S, Cl, Fe, and Ni on CCA-element
Assessing CCA-elements Reactivity
3.2. Under Boudouard Reaction (BR)
4. Conclusion
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Edo, M.; Björn, E.; Persson, P.E.; Jansson, S. Assessment of chemical and material contamination in waste wood fuels-A case study ranging over nine years. Waste Manag. 2016, 49, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.; Kearley, V. Role of wood waste as source of biomass fuel in the UK. Energy Mater. 2009, 4, 162–165. [Google Scholar] [CrossRef]
- Department for Environment, Food and Rural Affairs. Wood Waste: A Short Review of Recent Research; Department for Environment, Food and Rural Affairs: London, UK, 2012.
- Vassilev, S.V.; Baxter, D.; Andersen, L.K.; Vassileva, C.G. An overview of the chemical composition of biomass. Fuel 2010, 89, 913–933. [Google Scholar] [CrossRef]
- Kamińska-Pietrzak, N.; Smoliński, A. Selected Environmental Aspects of Gasification and Co-Gasification of Various Types of Waste. J. Sustain. Min. 2013, 12, 6–13. [Google Scholar] [CrossRef]
- Couto, N.; Rouboa, A.; Silva, V.; Monteiro, E.; Bouziane, K. Influence of the biomass gasification processes on the final composition of syngas. Energy Procedia 2013, 36, 596–606. [Google Scholar] [CrossRef]
- Pandey, A.; Bhaskar, T.; Stöcker, M.; Sukumaran, R. Recent Advances in ThermoChemical Conversion of Biomass; Elsevier: New York, NY, USA, 2015. [Google Scholar]
- Gomez-Barea, A.; Leckner, B. Modeling of biomass gasification in fluidized bed. Prog. Energy Combust. Sci. 2010, 36, 444–509. [Google Scholar] [CrossRef]
- James, R.; Arthur, M.; Yuan, W.; Boyette, M.D. The effect of biomass physical properties on top-lit updraft gasification of woodchips. Energies 2016, 9, 283. [Google Scholar] [CrossRef]
- González-Vázquez, M.P.; García, R.; Pevida, C.; Rubiera, F. Optimization of a bubbling fluidized bed plant for low-temperature gasification of biomass. Energies 2017, 10, 306. [Google Scholar] [CrossRef]
- Tanger, P.; Field, J.L.; Jahn, C.E.; DeFoort, M.W.; Leach, J.E. Biomass for thermochemical conversion: targets and challenges. Front. Plant Sci. 2013, 4, 218. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, M.; Sahu, J.N.; Ganesan, P. Effect of process parameters on production of biochar from biomass waste through pyrolysis: A review. Renew. Sustain. Energy Rev. 2016, 55, 467–481. [Google Scholar] [CrossRef]
- Vervaeke, P.; Tack, F.M.G.; Navez, F.; Martin, J.; Verloo, M.G.; Lust, N. Fate of heavy metals during fixed bed downdraft gasification of willow wood harvested from contaminated sites. Biomass Bioenergy 2006, 30, 58–65. [Google Scholar] [CrossRef]
- Šyc, M.; Pohořelý, M.; Jeremiáš, M.; Vosecký, M.; Kameníková, P.; Skoblia, S.; Svoboda, K.; Punčochář, M. Behavior of heavy metals in steam fluidized bed gasification of contaminated biomass. Energy Fuels 2011, 25, 2284–2291. [Google Scholar] [CrossRef]
- Tafur-Marinos, J.A.; Ginepro, M.; Pastero, L.; Torazzo, A.; Paschetta, E.; Fabbri, D.; Zelano, V. Comparison of inorganic constituents in bottom and fly residues from pelletised wood pyro-gasification. Fuel 2014, 119, 157–162. [Google Scholar] [CrossRef]
- Zhou, X.; Liu, W.; Zhang, P.; Wu, W. Study on Heavy Metals Conversion Characteristics During Refused Derived Fuel Gasification Process. Procedia Environ. Sci. 2016, 31, 514–519. [Google Scholar] [CrossRef]
- Wu, M.H.; Lin, C.L.; Zeng, W.Y. Effect of waste incineration and gasification processes on heavy metal distribution. Fuel Process. Technol. 2014, 125, 67–72. [Google Scholar] [CrossRef]
- Jiang, Y.; Ameh, A.; Lei, M.; Duan, L.; Longhurst, P. Solid–gaseous phase transformation of elemental contaminants during the gasification of biomass. Sci. Total Environ. 2016, 563–564, 724–730. [Google Scholar] [CrossRef] [PubMed]
- Froment, K.; Defoort, F.; Bertrand, C.; Seiler, J.M.; Berjonneau, J.; Poirier, J. Thermodynamic equilibrium calculations of the volatilization and condensation of inorganics during wood gasification. Fuel 2013, 107, 269–281. [Google Scholar] [CrossRef]
- Kramb, J.; Konttinen, J.; Backman, R.; Salo, K.; Roberts, M. Elimination of arsenic-containing emissions from gasification of chromated copper arsenate wood. Fuel 2016, 181, 319–324. [Google Scholar] [CrossRef]
- Thompson, D.; Argent, B.B. Prediction of the distribution of trace elements between the product streams of the Prenflo gasifier and comparison with reported data. Fuel 2002, 81, 555–570. [Google Scholar] [CrossRef]
- Kilgallon, P.J.; Simms, N.J.; Oakey, J.E. Fate of trace contaminants from biomass fuels in gasification systems. Mater. Adv. Power Eng. 2002, 21, 903–912. [Google Scholar]
- Reed, G.P.; Dugwell, D.R.; Kandiyoti, R. Control of trace elements in a gasifier hot gas filter: A comparison with predictions from a thermodynamic equilibrium model. Energy Fuels 2001, 15, 1480–1487. [Google Scholar] [CrossRef]
- Liu, S.; Wang, Y.; Yu, L.; Oakey, J. Thermodynamic equilibrium study of trace element transformation during underground coal gasification. Fuel Process. Technol. 2006, 87, 209–215. [Google Scholar] [CrossRef]
- Bradshaw, A.; Simms, N.J.; Nicholls, J.R. Passage of trace metal contaminants through hot gas paths of gas turbines burning biomass and waste-fuels. Fuel 2008, 87, 3529–3536. [Google Scholar] [CrossRef]
- Svoboda, K.; Leitner, J.; Havlica, J.; Hartman, M.; Pohořelý, M.; Brynda, J.; Šyc, M.; Chyou, Y.P.; Chen, P.C. Thermodynamic aspects of gasification derived syngas desulfurization, removal of hydrogen halides and regeneration of spent sorbents based on La2O3/La2O2CO3 and cerium oxides. Fuel 2017, 197, 277–289. [Google Scholar] [CrossRef]
- Contreras, M.L.; Arostegui, J.M.; Armesto, L. Arsenic interactions during co-combustion processes based on thermodynamic equilibrium calculations. Fuel 2009, 88, 539–546. [Google Scholar] [CrossRef]
- Yan, R.; Gauthier, D.; Flamant, G. Possible interactions between As, Se, and Hg during coal combustion. Combust. Flame 2000, 120, 49–60. [Google Scholar] [CrossRef]
- Energy Research Centre of the Netherlands. Available online: https://www.ecn.nl/phyllis2/ (accessed on 20 March 2017).
- Prabir, B. Biomass Gasification, Pyrolysis, and Torrefaction Practical Design and Theory, 2nd ed; Academic Press: Cambridge, MA, USA, 30 July 2013. [Google Scholar]
| Species | As | Cr | Cu |
|---|---|---|---|
| Gaseous | As4O10, AsS, As4S4AsH2, AsH3, As4O8, As, As2, AsN, AsO, AsO2, AsCl3, AsH, As4O6, As4O7, As3, As4O9. | ClCr, CrHO, CrHO2CrHO3, CrH2O2, CrH2O3, Cl4Cr, Cl5Cr, Cl6Cr, ClCrO, CrH2O4, CrH3O3, Cl4CrO, Cr, Cr2, CrH, CrH3O4, CrH4O4, Cl2Cr, Cl3Cr, CrO2, CrO3, Cr2O, ClCrO2, Cl2CrO, Cl2CrO2, Cl3CrO, CrS2, CrH4O5, CrN, CrO, Cr2O2, Cr2O3, CrS. | Cl5Cu5, Cu, Cl3Cu3, Cl4Cu2, CuO, CuS, ClCu, Cu2S, Cu2, CuH, CuHO, Cl2Cu, Cl2Cu2, Cl4Cu4. |
| Liquid | AsCL3, As4S4. | - | - |
| Solid | As, As2Cu3O8, As2Fe3O, As2S2, AsNa3, AsCu3, As2Ca3O8, As2Co3O8, AsNa3O4, AsNi, As2Ni5, As8Ni11, AsCrO4, As2Cr3O8, AsS, AsCu3O4, As2Ni3O8, As2O5, As2S3, AsFeO4. | AsCrO4, Cl3Cr, CoCr2O4, Cr, Cr2O12S3, CrS, CaCrO3, CaCr2O4, Cl2Cr, Cr2O3, Cr5O12, Cr8O21, CrNa2O4, Cr2Na2O4, As2Cr3O8, C2Cr3, C3Cr7, C6Cr23, C6CrO6, Cr2FeO4, CrN, Cr2N, Cr2NiO4, CrO2, CrO3. | CuH6O7S, CuH10O9S, CuO, Cu2O, CuO4S, Cu2O4S, Cu2O5S, CuS, Cu2S, AsCu3, AsCu3O4, As2Cu3O8, CCuN, CCuO3, CaCu, Ca2CuO3, ClCu, Cl2Cu, Cu, CuFeO2, CuFe2O4, Cu2Fe2O4, CuFeS2, Cu5FeS4, CaCu5, Ca2Cu, CaCu2O3, CuH2O2, CuH2O5S. |
| Composition | Min | Mean | Max | 5% | 95% |
|---|---|---|---|---|---|
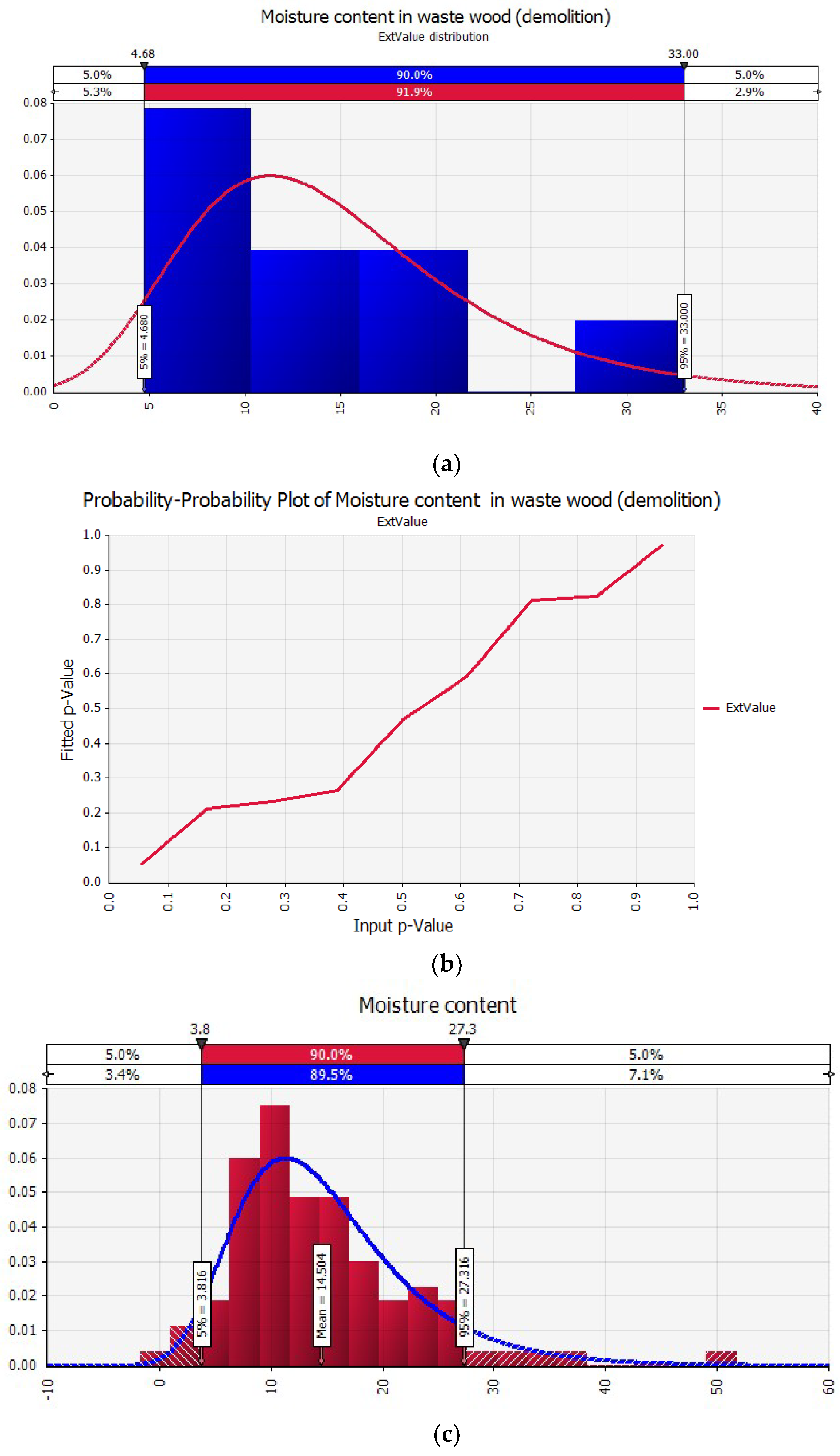
| Moisture content (Ar) (%) | 4.68 | 14.86 | 33 | 3.82 | 27.31 |
| Ash content (Ar) (%) | 0.79 | 12.09 | 70.41 | 0.843 | 48.54 |
| Carbon (C) (Dry) (%) | 43.31 | 46.55 | 49.8 | 42.84 | 50.24 |
| Hydrogen (H) (Dry) (%) | 5.26 | 5.69 | 5.93 | 5.26 | 5.95 |
| Nitrogen (N) (Dry) (%) | 0.19 | 1.06 | 1.93 | 0.069 | 2.04 |
| Oxygen (O) (Dry) (%) | 37.13 | 40.57 | 55.79 | 37.3 | 47.34 |
| Sulphur (S) (Dry) (%) | 0.03 | 0.09 | 0.18 | 0.02489 | 0.176 |
| Chlorine (Cl) (Dry) (mg/kg) | 113 | 556.49 | 1000 | 48.19 | 1077.8 |
| Copper (Cu) (Dry) (mg/kg) | 4.8 | 63.64 | 385 | 5.04 | 286.8 |
| Nickel (Ni) (Dry) (mg/kg) | 2.8 | 15.69 | 46 | 2.19 | 43.99 |
| Arsenic (As) (Dry) (mg/kg) | 1.3 | 5.5 | 13 | 1.35 | 35.45 |
| Cobalt (Co) (Dry) (mg/kg) | 0.8 | 4.88 | 25 | 84% | 21.92 |
| Chromium (Cr) (Dry) (mg/kg) | 21 | 30.2 | 37 | 21.73 | 36.87 |
| Sodium (Na) (Dry) (mg/kg) | 210 | 703.92 | 1613 | 162.0 | 1645.5 |
| Calcium (Ca) (Dry) (mg/kg) | 2200 | 4061.41 | 6537 | 2247.0 | 8959.29 |
| Iron (Fe) (Dry) (mg/kg) | 340.3 | 655.96 | 1082 | 348.16 | 2037.09 |
| Element | Concentration (mole/h) | Element | Concentration (mole/h) | Element | Concentration (mole/h) |
|---|---|---|---|---|---|
| As | 0, 0.7, 1.4, 5 | Ca | 0, 100, 250, 500, 1000 | Ni | 0, 5, 10, 20 |
| Cu | 0, 4, 6, 8 | Cl | 0, l0, 50, 100 | Na | 0, 100, 200, 500 |
| Cr | 0, 5, 10, 21 | Fe | 0, 50, 100, 500 | S | 0, 0.05, 0.1, 0.2 |
| Parameter | PCR | BR |
|---|---|---|
| Temperature range (°C) | 0–1800 | 0–1300 |
| Pressure (atm) | 1 | 1 |
| Equivalence ratio (kg/h) | 0.2 | 0 |
| Biomass rate (m3/h) | 3.59 | 3.59 |
| Air rate (m3/h) | 0.72 | - |
| Steam rate | 1.44 | - |
| Steam/Air | 2:1 | - |
| CO2 | - | 1.19 |
| CO2/Biomass | - | 1:3 |
| Ca Concentration (mole/h) | Species | Temperature Range (°C) |
|---|---|---|
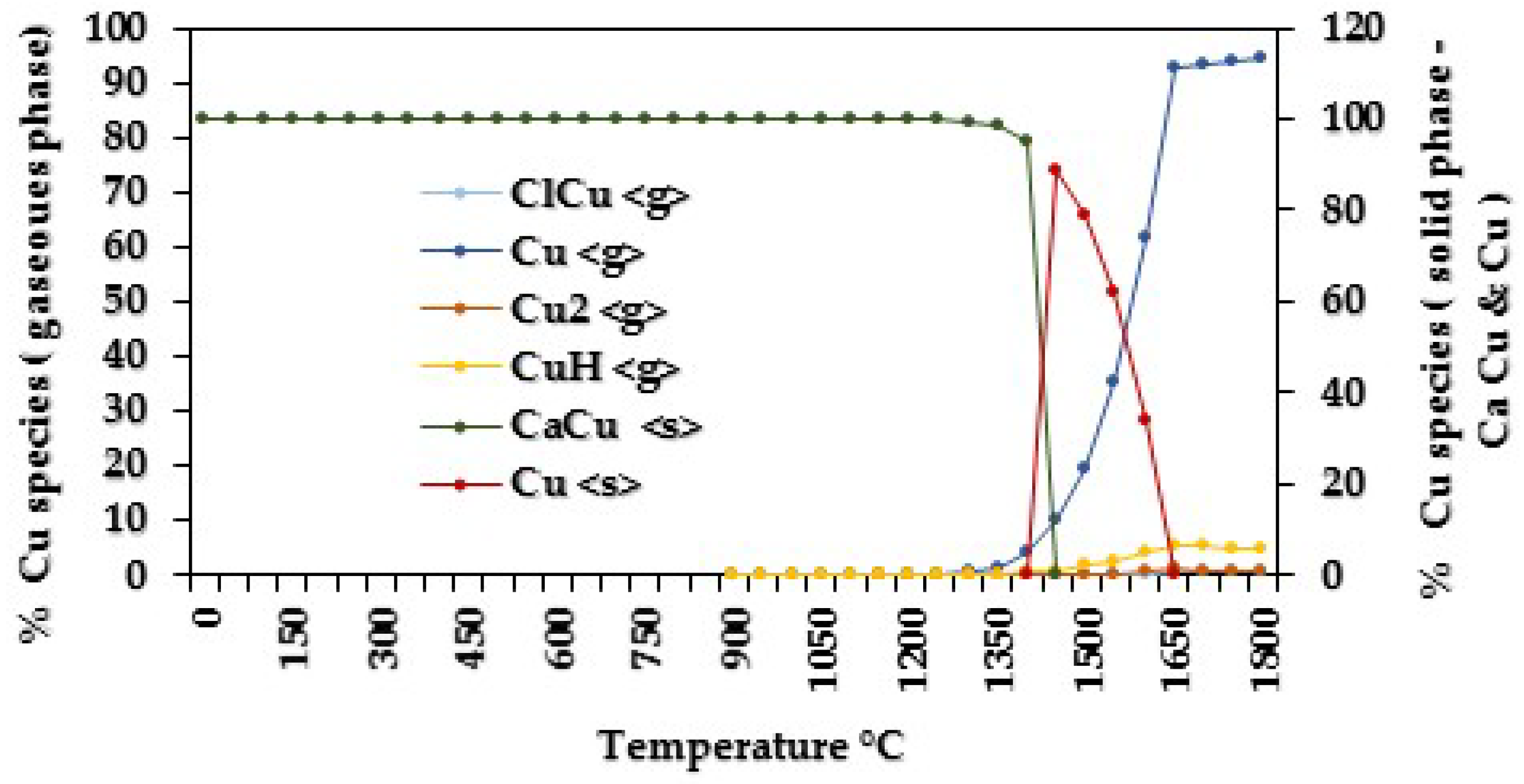
| 100 | Cu <s> | 0–800 |
| Cu <s>, Cu <g>, CuH <g> | 850–950 | |
| Cu <s>, ClCu <g>, Cu <g>, CuH <g> | 1000–1800 | |
| 250 | CaCu <s> | 0–800 |
| Cu <g>, CuH <g>, CaCu <s> | 850–1150 | |
| ClCu <g>, Cu <g>, Cu2 <g>, CuH <g>, CaCu <s> | 1350–1800 | |
| 500 | CaCu <s>, CaCu5 <s> | 0–800 |
| Cu, Cu <g>, CuH <g> | 850–950 | |
| Cu <g>, CuH <g>, CaCu <s> | 1000–1150 | |
| ClCu <g>, Cu <g>, Cu2 <g>, CuH <g>, CaCu <s> | 1250–1800 | |
| 1000 | CaCu <s> | 0–800 |
| Cu <g>, CuH <g>, CaCu <s> | 850–950 | |
| Cu <g>, CuH <g>, CaCu <s> | 1000–1150 | |
| ClCu <g>, Cu <g>, Cu2 <g>, CuH <g>, CaCu <s> | 1250–1800 |
| Ca Concentration (mole/h) | Species | Temperature Range (°C) |
|---|---|---|
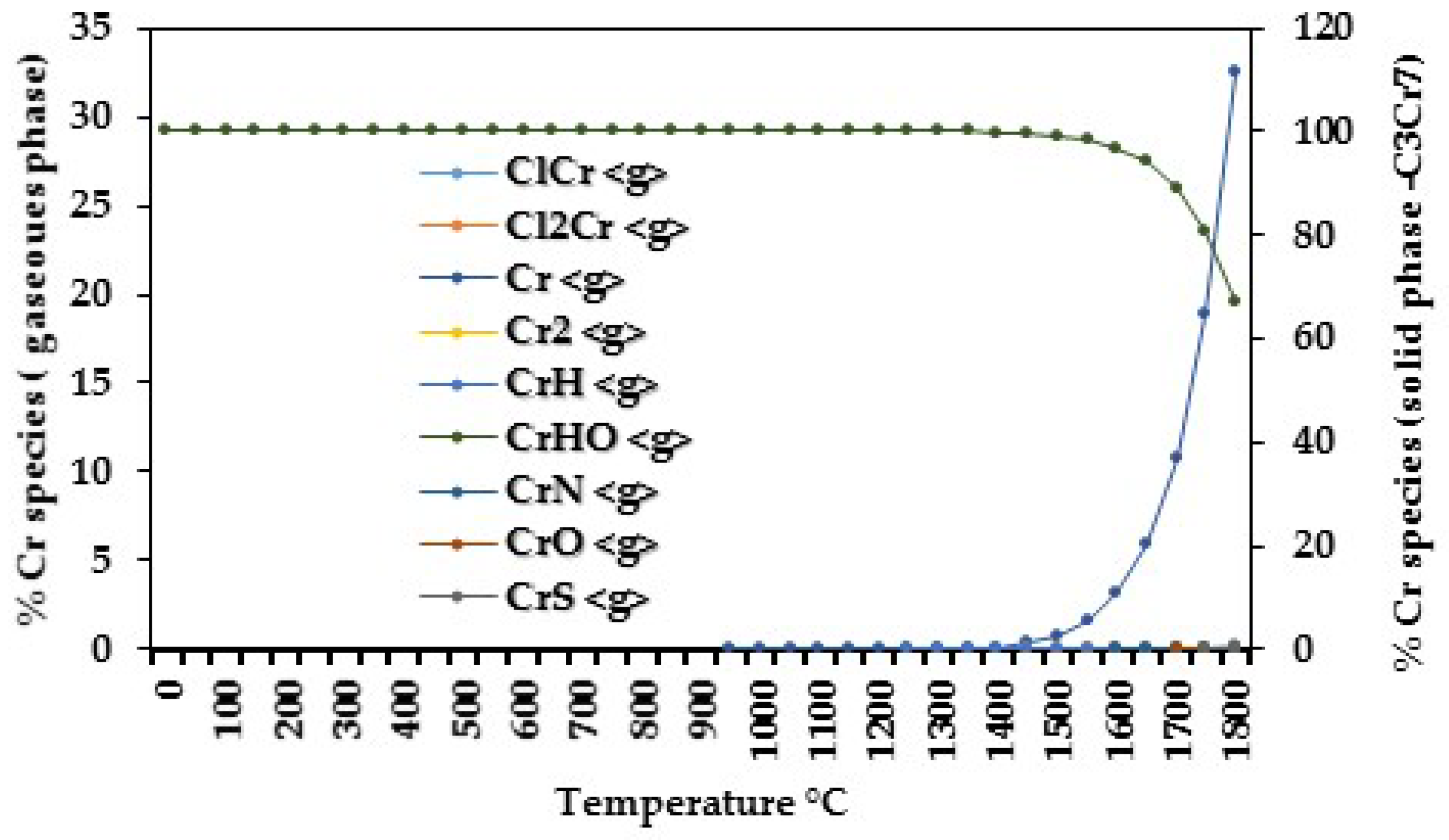
| 0 | Cr2Na2O4 <s> | 0–300 |
| C2Cr3 <s>, Cr2Na2O4 <s> | 350–400 | |
| Cr2Na2O4 <s> | 450–600 | |
| Cr <g>, C2Cr3 <s>, Cr2Na2O4 <s> | 650–1200 | |
| ClCr <g>, Cl2Cr <g>, Cr <g>, CrHO <g>, CrS <g>, C2Cr3 <s>, Cr <s>, C3Cr7 <s>, C6Cr23 <s> | 1250–1800 | |
| 100 | C2Cr3 <s>, CaCr2O4 <s>, CrN <s>, Cr2Na2O4 <s> | 0–300 |
| C2Cr3 <s>, Cr2Na2O4 <s> | 350–400 | |
| C2Cr3 <s>, CaCr2O4 <s> | 450–600 | |
| ClCr <g>, Cr <g>, C2Cr3 <s>, CaCr2O4 <s> | 650–1200 | |
| ClCr <g>, Cl2Cr <g>, Cr<g>, C2Cr3 <s> | 1250–1800 | |
| 250–500 | C2Cr3 <s> | 0–300 |
| C2Cr3 <s>, C3Cr7 <s> | 350–600 | |
| C3Cr7 <s> | 650–1200 | |
| C2Cr3 <s>, ClCr <g>, Cl2Cr <g>, Cr <g>, CrH <g>, C3Cr7 <s>, CrO <g>, CrS <g>, CrN <g> | 1250–1800 | |
| 10000 | C3Cr7 <s> | 0–600 |
| Cr <g>, C3Cr7 <s> | 650–1200 | |
| ClCr <g>, Cl2Cr <g>, Cr <g>, CrH <g>, CrO <g>, CrS <g>, CrN <g>, C3Cr7 <s> | 1250–1800 |
| Elements | Solid Phase | Temperature (°C) | Gaseous Phase | Temperature (°C) |
|---|---|---|---|---|
| Cr | C3Cr7 | 0–1300 | Cr | 1000–1300 |
| CrCl | 1250–1300 | |||
| CrH | 1300 | |||
| Cu | CaCu | 0–1300 | Cu, CuH | 900–1300 |
| Cu2, ClCu | 1150–1300 | |||
| As | AsNa3 | 0–800 | As, As2, AsH, AsH2 | 1150–1300 |
| As2Ni5 | 850–1300 | AsH3 | 1150–1300 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Badri, S.B.; Jiang, Y.; Wagland, S.T. Possible Interactions and Interferences of Copper, Chromium, and Arsenic during the Gasification of Contaminated Waste Wood. Energies 2018, 11, 1966. https://doi.org/10.3390/en11081966
Al-Badri SB, Jiang Y, Wagland ST. Possible Interactions and Interferences of Copper, Chromium, and Arsenic during the Gasification of Contaminated Waste Wood. Energies. 2018; 11(8):1966. https://doi.org/10.3390/en11081966
Chicago/Turabian StyleAl-Badri, Shurooq Badri, Ying Jiang, and Stuart Thomas Wagland. 2018. "Possible Interactions and Interferences of Copper, Chromium, and Arsenic during the Gasification of Contaminated Waste Wood" Energies 11, no. 8: 1966. https://doi.org/10.3390/en11081966
APA StyleAl-Badri, S. B., Jiang, Y., & Wagland, S. T. (2018). Possible Interactions and Interferences of Copper, Chromium, and Arsenic during the Gasification of Contaminated Waste Wood. Energies, 11(8), 1966. https://doi.org/10.3390/en11081966






