Flexible, Heat-Resistant, and Flame-Retardant Glass Fiber Nonwoven/Glass Platelet Composite Separator for Lithium-Ion Batteries
Abstract
1. Introduction
2. Results
2.1. Physical Properties
2.1.1. Morphology
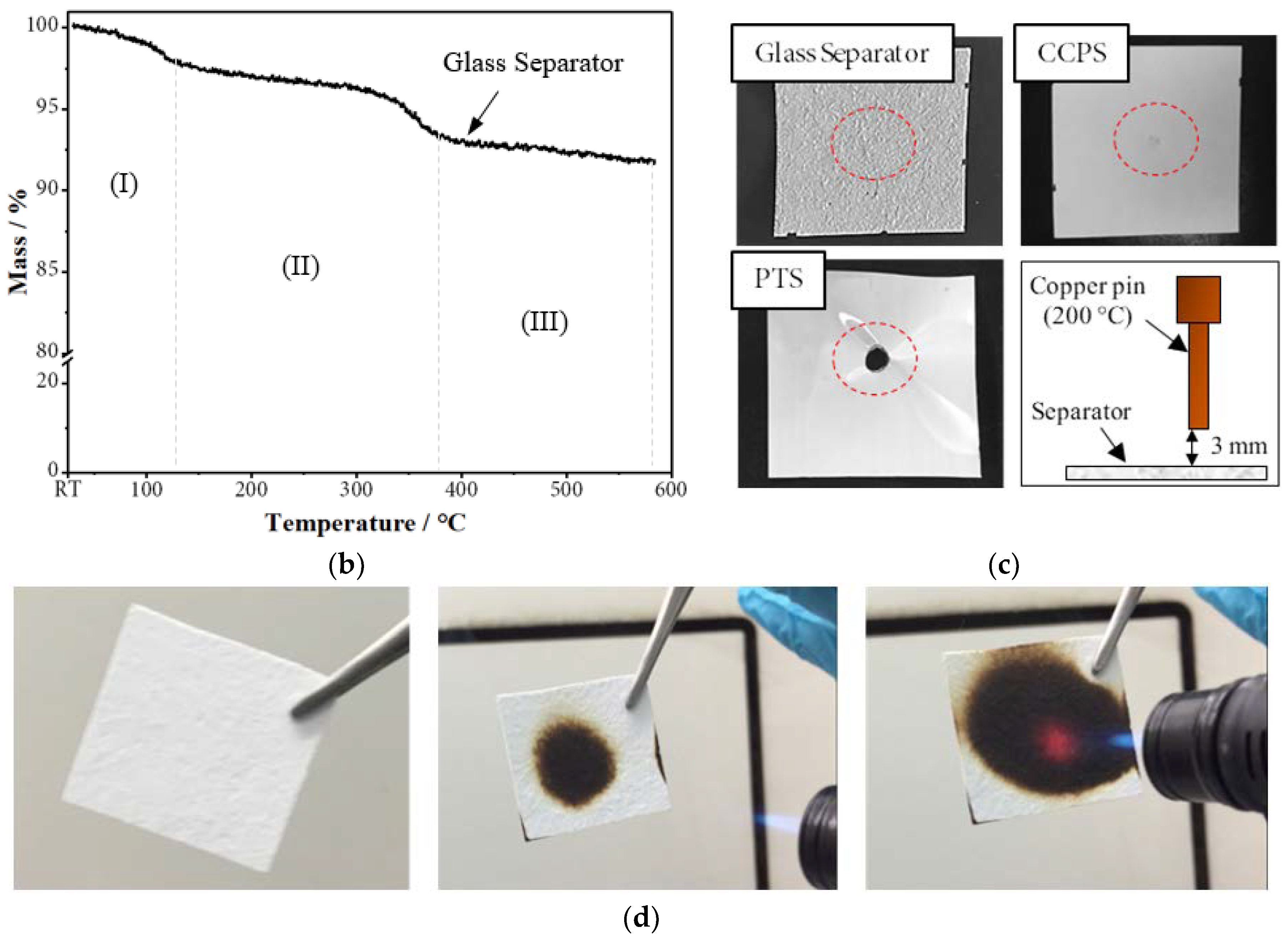
2.1.2. Thermal Stability
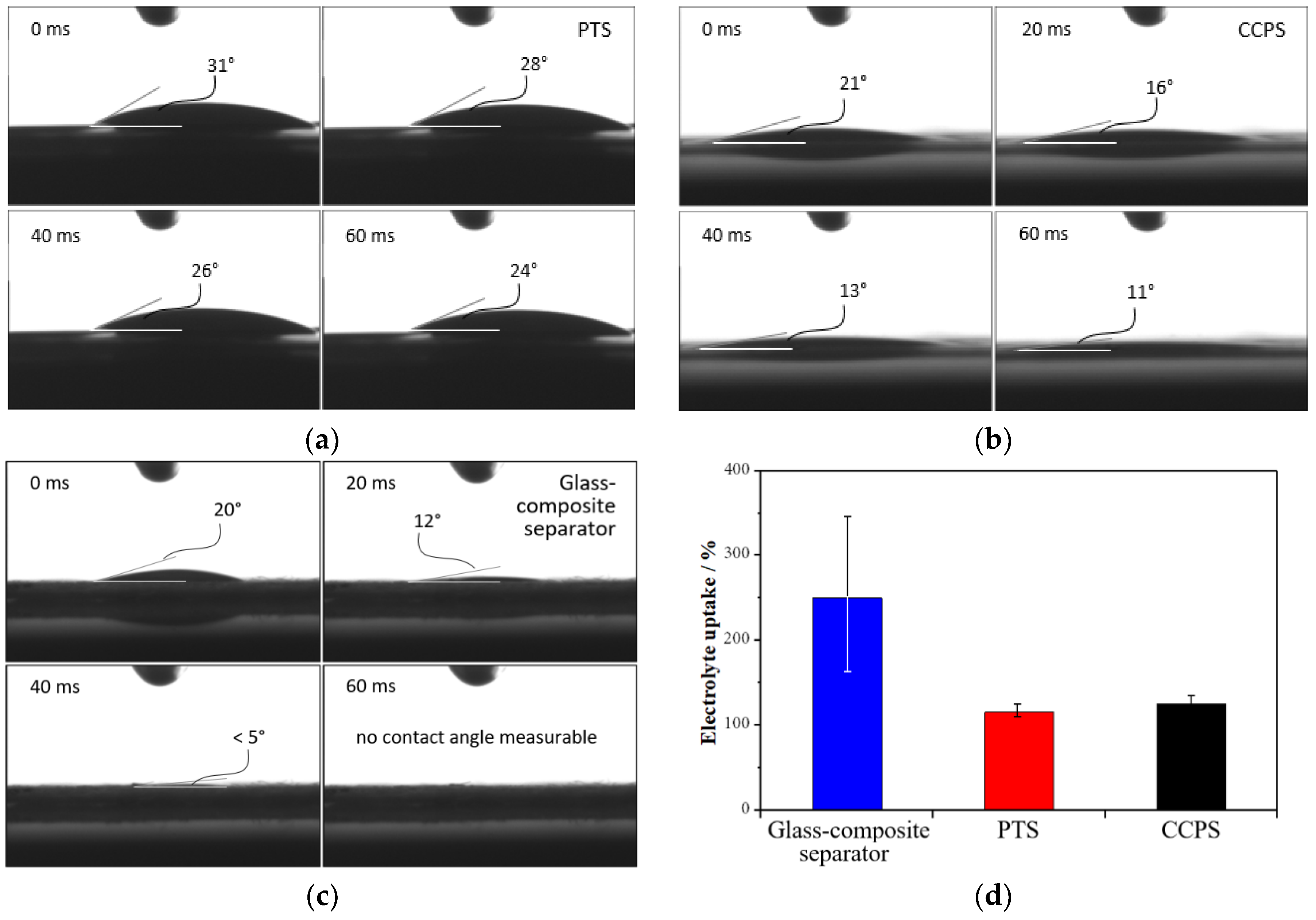
2.1.3. Wettability
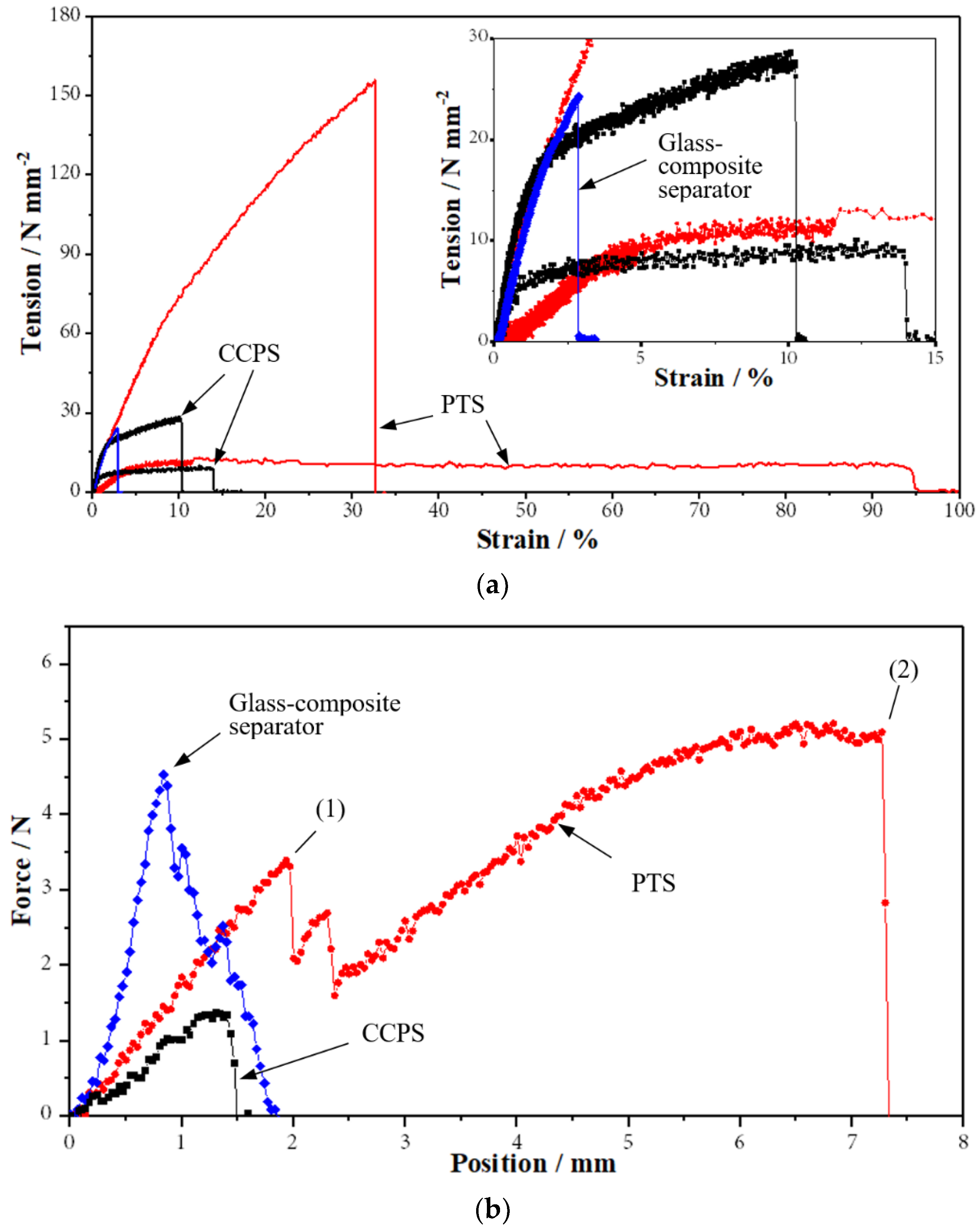
2.1.4. Mechanical Stability
2.2. Electrochemical Properties
3. Materials and Methods
3.1. Materials
3.2. Preparation of a Glass Platelet—Glass Fiber Nonwoven Composite Separator
3.3. Characterization
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lu, L.; Han, X.; Li, J.; Hua, J.; Ouyang, M. A review on the key issues for lithium-ion battery management in electric vehicles. J. Power Sources 2013, 226, 272–288. [Google Scholar] [CrossRef]
- Park, J.-K. (Ed.) Principles and Applications of Lithium Secondary Batteries; Wiley-VCH: Weinheim, Germany, 2012; ISBN 978-3-52-733151-2. [Google Scholar] [CrossRef]
- Linden, D.; Reddy, T.B. (Eds.) Linden’s Handbook of Batteries, 4th ed.; McGraw-Hill: New York, NY, USA, 2011; ISBN 978-0-07-162421-3. [Google Scholar]
- Cairns, E.J.; Albertus, P. Batteries for electric and hybrid-electric vehicles. Annu. Rev. Chem. Biomol. Eng. 2010, 1, 299–320. [Google Scholar] [CrossRef] [PubMed]
- Gerssen-Gondelach, S.J.; Faaij, A.P.C. Performance of batteries for electric vehicles on short and longer term. J. Power Sources 2012, 212, 111–129. [Google Scholar] [CrossRef]
- Abada, S.; Marlair, G.; Lecocq, A.; Petit, M.; Sauvant-Moynot, V.; Huet, F. Safety focused modeling of lithium-ion batteries, A review. J. Power Sources 2016, 306, 178–192. [Google Scholar] [CrossRef]
- Nazri, G.-A.; Pistoia, G. (Eds.) Lithium Batteries: Science and Technology, 1st ed.; Springer: New York, NY, USA, 2009; ISBN 978-0-38-792675-9. [Google Scholar]
- Loveridge, M.; Remy, G.; Kourra, N.; Genieser, R.; Barai, A.; Lain, M.; Guo, Y.; Amor-Segan, M.; Williams, M.; Amietszajew, T.; et al. Looking Deeper into the Galaxy (Note 7). Batteries 2018, 4, 3. [Google Scholar] [CrossRef]
- Huang, X. Separator technologies for lithium-ion batteries. J. Solid State Electrochem. 2011, 15, 649–662. [Google Scholar] [CrossRef]
- Yang, M.; Hou, J. Membranes in lithium ion batteries. Membranes 2012, 2, 367–383. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Zhang, P.; Chen, L.; Yang, P.; Zhao, J. Effect of a thin ceramic-coating layer on thermal and electrochemical properties of polyethylene separator for lithium-ion batteries. J. Power Sources 2014, 270, 547–553. [Google Scholar] [CrossRef]
- Roth, E.P.; Doughty, D.H.; Pile, D.L. Effects of separator breakdown on abuse response of 18,650 Li-ion cells. J. Power Sources 2007, 174, 579–583. [Google Scholar] [CrossRef]
- Kim, J.Y.; Lim, D.Y. Surface-Modified Membrane as a Separator for Lithium-Ion Polymer Battery. Energies 2010, 3, 866–885. [Google Scholar] [CrossRef]
- Huang, F.; Liu, W.; Li, P.; Ning, J.; Wei, Q. Electrochemical Properties of LLTO/Fluoropolymer-Shell Cellulose-Core Fibrous Membrane for Separator of High Performance Lithium-Ion Battery. Materials 2016, 9, 75. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.-A.; Kim, S.H.; Kim, D.-W. Enhancement of thermal stability and cycling performance in lithium-ion cells through the use of ceramic-coated separators. J. Power Sources 2010, 195, 6192–6196. [Google Scholar] [CrossRef]
- Shi, C.; Dai, J.; Li, C.; Shen, X.; Peng, L.; Zhang, P.; Wu, D.; Sun, D.; Zhao, J. A Modified Ceramic-Coating Separator with High-Temperature Stability for Lithium-Ion Battery. Polymers 2017, 9, 159. [Google Scholar] [CrossRef]
- Zhai, Y.; Xiao, K.; Yu, J.; Ding, B. Fabrication of hierarchical structured SiO2/polyetherimide-polyurethane nanofibrous separators with high performance for lithium ion batteries. Electrochim. Acta 2015, 154, 219–226. [Google Scholar] [CrossRef]
- Yeon, D.; Lee, Y.; Ryou, M.-H.; Lee, Y.M. New flame-retardant composite separators based on metal hydroxides for lithium-ion batteries. Electrochim. Acta 2015, 157, 282–289. [Google Scholar] [CrossRef]
- Yanilmaz, M.; Zhang, X. Polymethylmethacrylate/Polyacrylonitrile Membranes via Centrifugal Spinning as Separator in Li-Ion Batteries. Polymers 2015, 7, 629–643. [Google Scholar] [CrossRef]
- Yanilmaz, M.; Dirican, M.; Zhang, X. Evaluation of electrospun SiO2/nylon 6,6 nanofiber membranes as a thermally-stable separator for lithium-ion batteries. Electrochim. Acta 2014, 133, 501–508. [Google Scholar] [CrossRef]
- Shi, C.; Dai, J.; Huang, S.; Li, C.; Shen, X.; Zhang, P.; Wu, D.; Sun, D.; Zhao, J. A simple method to prepare a polydopamine modified core-shell structure composite separator for application in high-safety lithium-ion batteries. J. Membr. Sci. 2016, 518, 168–177. [Google Scholar] [CrossRef]
- Lee, J.; Lee, C.-L.; Park, K.; Kim, I.-D. Synthesis of an Al2O3-coated polyimide nanofiber mat and its electrochemical characteristics as a separator for lithium ion batteries. J. Power Sources 2014, 248, 1211–1217. [Google Scholar] [CrossRef]
- Jiang, W.; Liu, Z.; Kong, Q.; Yao, J.; Zhang, C.; Han, P.; Cui, G. A high temperature operating nanofibrous polyimide separator in Li-ion battery. Solid State Ion. 2013, 232, 44–48. [Google Scholar] [CrossRef]
- Song, J.; Ryou, M.-H.; Son, B.; Lee, J.-N.; Lee, D.J.; Lee, Y.M.; Choi, J.W.; Park, J.-K. Co-polyimide-coated polyethylene separators for enhanced thermal stability of lithium ion batteries. Electrochim. Acta 2012, 85, 524–530. [Google Scholar] [CrossRef]
- Miao, Y.-E.; Zhu, G.-N.; Hou, H.; Xia, Y.-Y.; Liu, T. Electrospun polyimide nanofiber-based nonwoven separators for lithium-ion batteries. J. Power Sources 2013, 226, 82–86. [Google Scholar] [CrossRef]
- Smith, S.A.; Williams, B.P.; Joo, Y.L. Effect of polymer and ceramic morphology on the material and electrochemical properties of electrospun PAN/polymer derived ceramic composite nanofiber membranes for lithium ion battery separators. Annu. Rev. Chem. Biomol. Eng. 2017, 526, 315–322. [Google Scholar] [CrossRef]
- Khan, W.S.; Asmatulu, R.; Rodriguez, V.; Ceylan, M. Enhancing thermal and ionic conductivities of electrospun PAN and PMMA nanofibers by graphene nanoflake additions for battery-separator applications. Int. J. Energy Res. 2014, 38, 2044–2051. [Google Scholar] [CrossRef]
- Yanilmaz, M.; Lu, Y.; Li, Y.; Zhang, X. SiO2/polyacrylonitrile membranes via centrifugal spinning as a separator for Li-ion batteries. J. Power Sources 2015, 273, 1114–1119. [Google Scholar] [CrossRef]
- Lee, J.H.; Manuel, J.; Choi, H.; Park, W.H.; Ahn, J.-H. Partially oxidized polyacrylonitrile nanofibrous membrane as a thermally stable separator for lithium ion batteries. Polymer 2015, 68, 335–343. [Google Scholar] [CrossRef]
- Wang, Q.; Yu, Y.; Ma, J.; Zhang, N.; Zhang, J.; Liu, Z.; Cui, G. Electrospun melamine resin-based multifunctional nonwoven membrane for lithium ion batteries at the elevated temperatures. J. Power Sources 2016, 327, 196–203. [Google Scholar] [CrossRef]
- Carvalho, D.V.; Loeffler, N.; Kim, G.-T.; Passerini, S. High Temperature Stable Separator for Lithium Batteries Based on SiO2 and Hydroxypropyl Guar Gum. Membranes 2015, 5, 632–645. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Pan, W.; Liu, H.; Gong, J.; Wu, H. Glass fiber fabric mat as the separator for lithium-ion battery with high safety performance. Ionics 2015, 21, 3135–3139. [Google Scholar] [CrossRef]
- Jung, Y.-C.; Kim, S.-K.; Kim, M.-S.; Lee, J.-H.; Han, M.-S.; Kim, D.-H.; Shin, W.-C.; Ue, M.; Kim, D.-W. Ceramic separators based on Li+-conducting inorganic electrolyte for high-performance lithium-ion batteries with enhanced safety. J. Power Sources 2015, 293, 675–683. [Google Scholar] [CrossRef]
- Brigham, S.J.; Watkinson, C. Understanding and use of glass flake. Paint Coat. Ind. 2009, 25, 22–24. [Google Scholar]
- Anselmann, R.; Ambrosius, K.; Mathias, M. Effect Pigments Based on Coated Glass Flakes. U.S. Patent 7,226, 503, 5 June 2007. [Google Scholar]
- Koronis, G.; Silva, A.; Fontul, M. Green composites, a review of adequate materials for automotive applications. Composites Part B 2013, 44, 120–127. [Google Scholar] [CrossRef]
- Kaynakli, O. A review of the economical and optimum thermal insulation thickness for building applications. Renew. Sustain. Energy Rev. 2012, 16, 415–425. [Google Scholar] [CrossRef]
- Aleksic, B.; Bailly, S.; Draghi, M.; Pestka, J.J.; Oswald, I.P.; Robine, E.; Bailly, J.D.; Lacroix, M.Z. Production of four macrocyclic trichothecenes by Stachybotrys chartarum during its development on different building materials as measured by UPLC-MS/MS. Build. Environ. 2016, 106, 265–273. [Google Scholar] [CrossRef]
- Kumar, V.; Rao, P.K.; Rawal, A. Amplification of electrolyte uptake in the absorptive glass mat (AGM) separator for valve regulated lead acid (VRLA) batteries. J. Power Sources 2017, 341, 19–26. [Google Scholar] [CrossRef]
- Zhu, J.; Yanilmaz, M.; Fu, K.; Chen, C.; Lu, Y.; Ge, Y.; Kim, D.; Zhang, X. Understanding glass fiber membrane used as a novel separator for lithium–sulfur batteries. J. Membr. Sci. 2016, 504, 89–96. [Google Scholar] [CrossRef]
- Schadeck, U.; Kyrgyzbaev, K.; Gerdes, T.; Willert-Porada, M.; Moos, R. Porous and non-porous micrometer-sized glass platelets as separators for lithium-ion batteries. J. Membr. Sci. 2018, 550, 518–525. [Google Scholar] [CrossRef]
- Zhang, S.S. A review on the separators of liquid electrolyte Li-ion batteries. J. Power Sources 2007, 164, 351–364. [Google Scholar] [CrossRef]
- Cho, E.; Mun, J.; Chae, O.B.; Kwon, O.M.; Kim, H.-T.; Ryu, J.H.; Kim, Y.G.; Oh, S.M. Corrosion/passivation of aluminum current collector in bis(fluorosulfonyl)imide-based ionic liquid for lithium-ion batteries. Electrochem. Commun. 2012, 22, 1–3. [Google Scholar] [CrossRef]
- Zhang, S.S. A review on electrolyte additives for lithium-ion batteries. J. Power Sources 2006, 162, 1379–1394. [Google Scholar] [CrossRef]
- Kyrgyzbaev, K.; Rosin, A.; Willert-Porada, M. Influence of temperature on the thickness of ultrathin particulate glass platelets. Glass Technol. Eur. J. Glass Sci. Technol. Part A 2016, 57, 95–100. [Google Scholar] [CrossRef]






© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schadeck, U.; Kyrgyzbaev, K.; Zettl, H.; Gerdes, T.; Moos, R. Flexible, Heat-Resistant, and Flame-Retardant Glass Fiber Nonwoven/Glass Platelet Composite Separator for Lithium-Ion Batteries. Energies 2018, 11, 999. https://doi.org/10.3390/en11040999
Schadeck U, Kyrgyzbaev K, Zettl H, Gerdes T, Moos R. Flexible, Heat-Resistant, and Flame-Retardant Glass Fiber Nonwoven/Glass Platelet Composite Separator for Lithium-Ion Batteries. Energies. 2018; 11(4):999. https://doi.org/10.3390/en11040999
Chicago/Turabian StyleSchadeck, Ulrich, Kanat Kyrgyzbaev, Heiko Zettl, Thorsten Gerdes, and Ralf Moos. 2018. "Flexible, Heat-Resistant, and Flame-Retardant Glass Fiber Nonwoven/Glass Platelet Composite Separator for Lithium-Ion Batteries" Energies 11, no. 4: 999. https://doi.org/10.3390/en11040999
APA StyleSchadeck, U., Kyrgyzbaev, K., Zettl, H., Gerdes, T., & Moos, R. (2018). Flexible, Heat-Resistant, and Flame-Retardant Glass Fiber Nonwoven/Glass Platelet Composite Separator for Lithium-Ion Batteries. Energies, 11(4), 999. https://doi.org/10.3390/en11040999



