Catalytic Hydrothermal Liquefaction of Food Waste Using CeZrOx
Abstract
:1. Introduction
2. Results
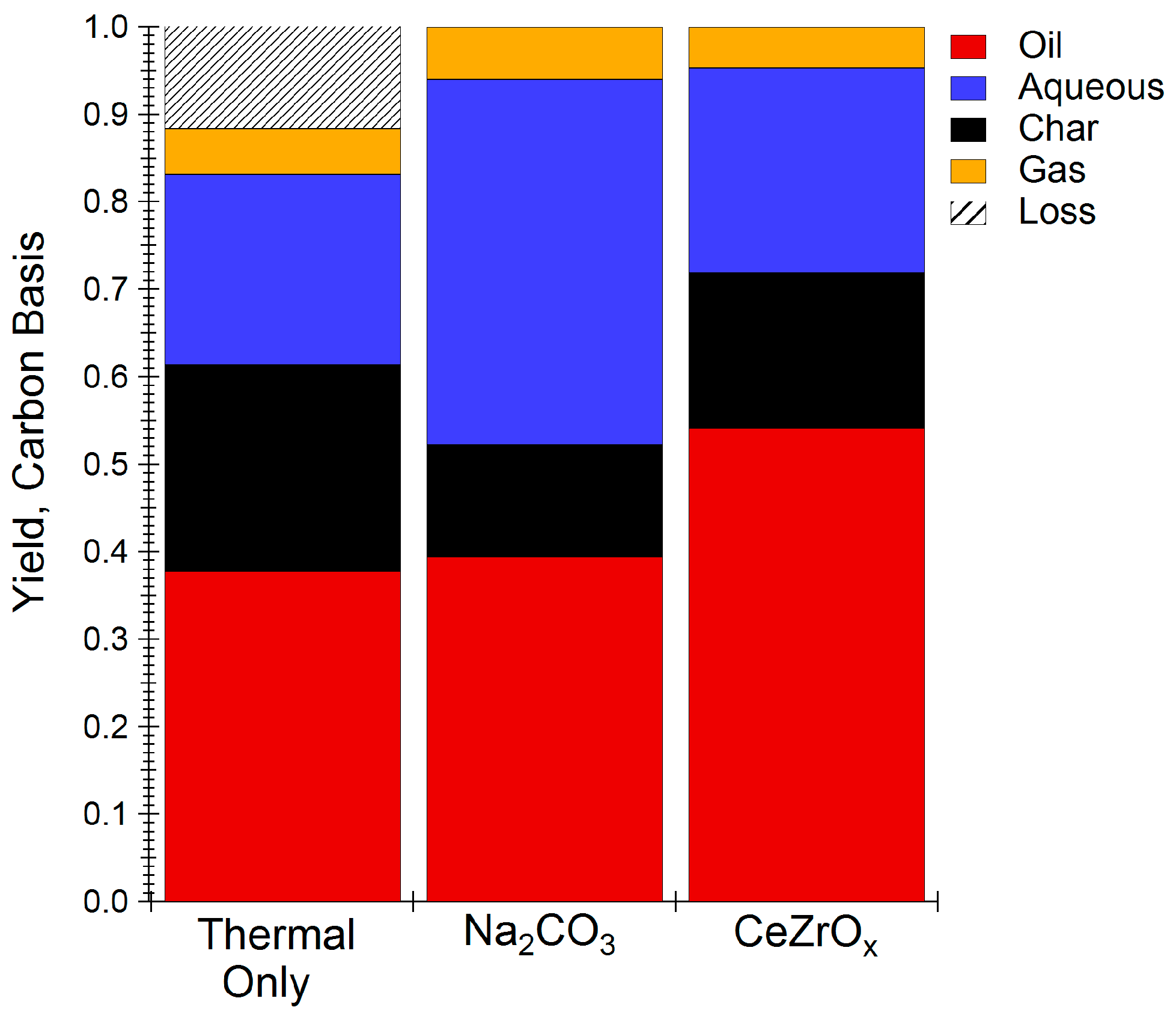
2.1. Hydrothermal Liquefaction (HTL) or Food Waste
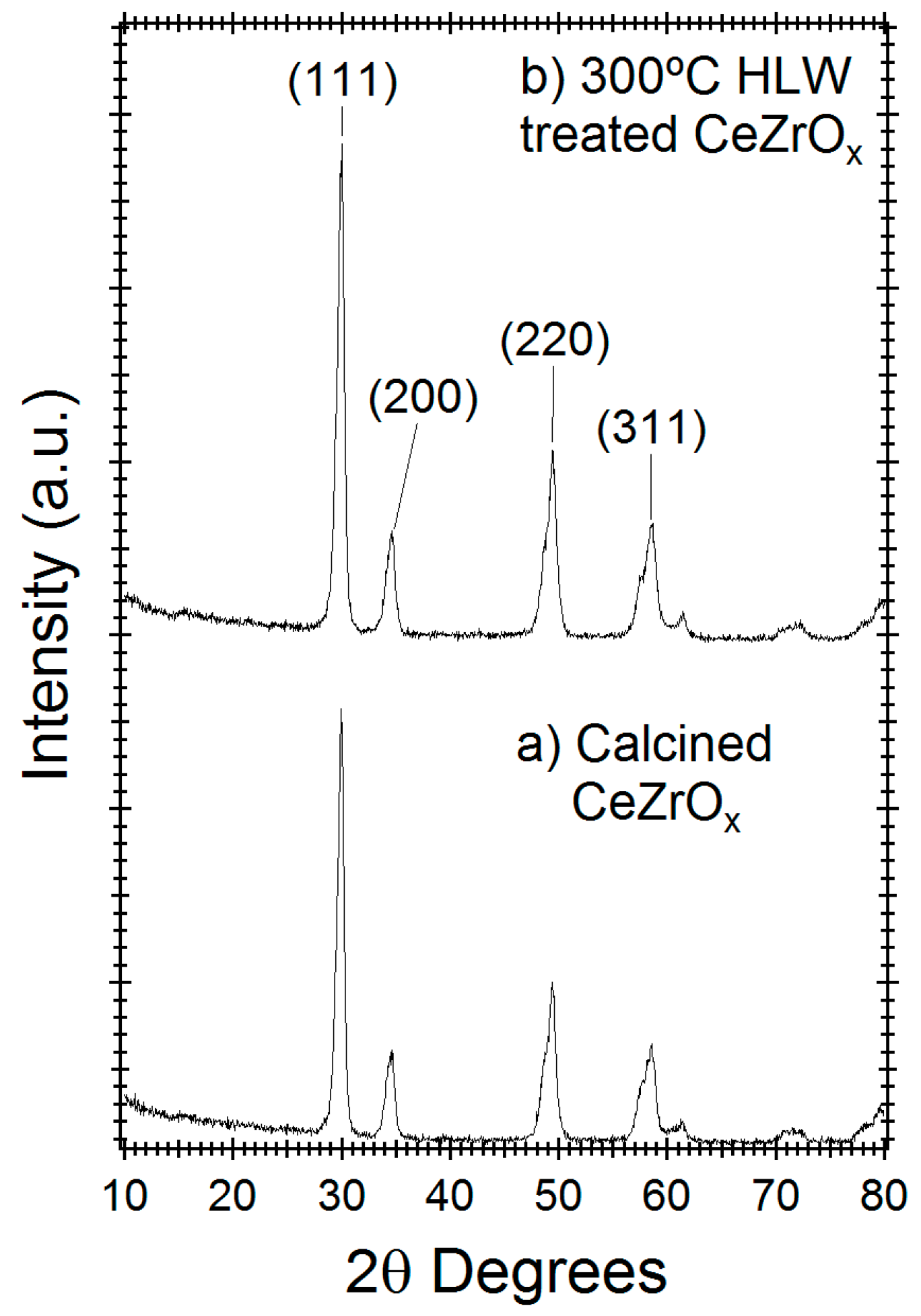
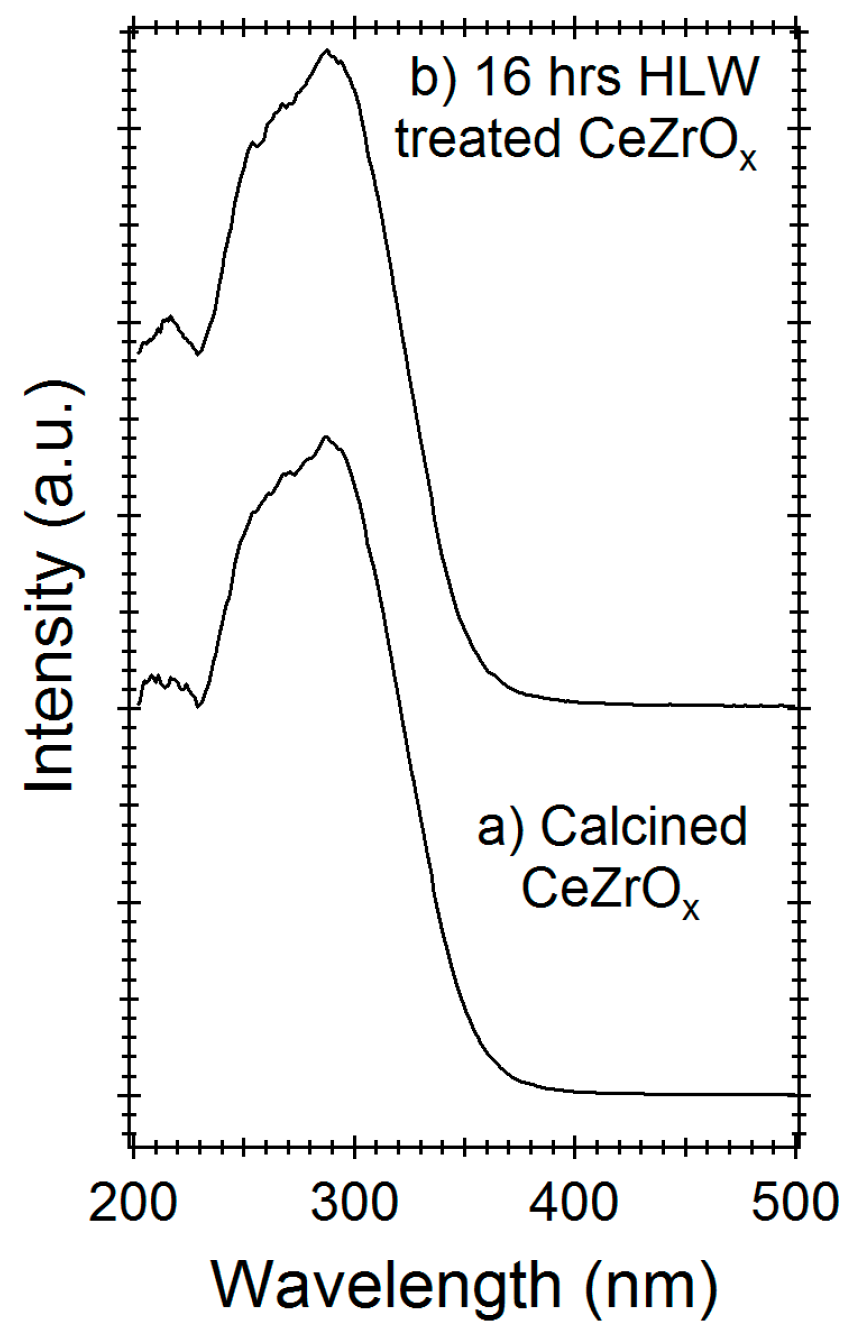
2.2. Hydrothermal Stability of CeZrOx Catalyst
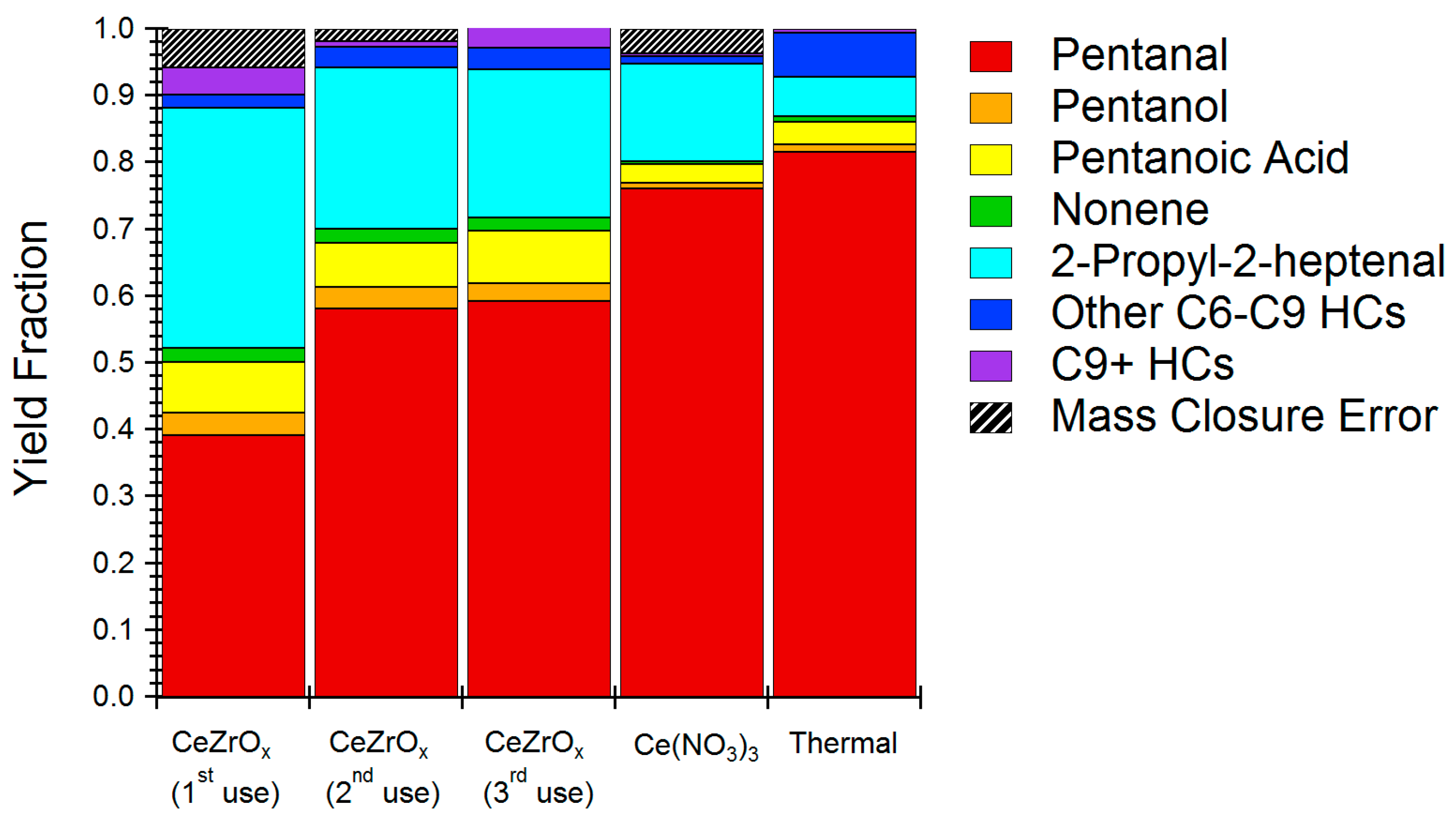
2.3. Model Chemistries for HTL Reaction
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Food Waste HTL Reactions
4.3. Food Waste HTL Product Analysis
4.4. Model Food Waste HTL Reactions
4.5. Hydrothermal Stability of CeZrOx
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- U.S. Energy Information Administration. International Energy Outlook 2016 Chapter 8: Transportation Sector Energy Consumption; U.S. Energy Information Administration: Washington, DC, USA, 2016.
- Environmental Protection Agency. U.S. Energy Independence and Security Act; Environmental Protection Agency: Washington, DC, USA, 2007; p. 882.
- Davis, R.; Tao, L.; Tan, E.C.D.; Biddy, M.J.; Beckham, G.T.; Scarlata, C.; Jacobson, J.; Cafferty, K.; Ross, J.; Lukas, J.; et al. Process Design and Economics for the Conversion of Lignocellulosic Biomass to Hydrocarbons: Dilute-Acid and Enzymatic Deconstruction of Biomass to Sugars and Biological Conversion of Sugars to Hydrocarbons; National Renewable Energy Laboratory: Golden, CO, USA, 2013.
- Pavlovič, I.; Knez, Ž.; Škerget, M. Hydrothermal reactions of agricultural and food processing wastes in sub- and supercritical water: A review of fundamentals, mechanisms, and state of research. J. Agric. Food Chem. 2013, 61, 8003–8025. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Energy. Biofuels and Bioproducts from Wet and Gaseous Waste Streams: Challenges and Opportunities; U.S. Department of Energy: Washington, DC, USA, 2017.
- Déniel, M.; Haarlemmer, G.; Roubaud, A.; Weiss-Hortala, E.; Fages, J. Energy valorisation of food processing residues and model compounds by hydrothermal liquefaction. Renew. Sustain. Energy Rev. 2016, 54, 1632–1652. [Google Scholar] [CrossRef]
- Appels, L.; Lauwers, J.; Degrve, J.; Helsen, L.; Lievens, B.; Willems, K.; Van Impe, J.; Dewil, R. Anaerobic digestion in global bio-energy production: Potential and research challenges. Renew. Sustain. Energy Rev. 2011, 15, 4295–4301. [Google Scholar] [CrossRef]
- Huber, G.W.; Iborra, S.; Corma, A. Synthesis of transportation fuels from biomass: Chemistry, catalysts, and engineering. Chem. Rev. 2006, 106, 4044–4098. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, J.; Saidina Amin, N. A review on operating parameters for optimum liquid oil yield in biomass pyrolysis. Renew. Sustain. Energy Rev. 2012, 16, 5101–5109. [Google Scholar] [CrossRef]
- Zhu, Y.; Biddy, M.J.; Jones, S.B.; Elliott, D.C.; Schmidt, A.J. Techno-economic analysis of liquid fuel production from woody biomass via hydrothermal liquefaction (HTL) and upgrading. Appl. Energy 2014, 129, 384–394. [Google Scholar] [CrossRef]
- Yan, W.H.; Duan, P.G.; Wang, F.; Xu, Y.P. Composition of the bio-oil from the hydrothermal liquefaction of duckweed and the influence of the extraction solvents. Fuel 2016, 185, 229–235. [Google Scholar] [CrossRef]
- Posmanik, R.; Cantero, D.A.; Malkani, A.; Sills, D.L.; Tester, J.W. Biomass conversion to bio-oil using sub-critical water: Study of model compounds for food processing waste. J. Supercrit. Fluids 2017, 119, 26–35. [Google Scholar] [CrossRef]
- Elliott, D.C.; Hart, T.R.; Neuenschwander, G.G. Chemical processing in high-pressure aqueous environments. 8. Improved catalysts for hydrothermal gasification. Ind. Eng. Chem. Res. 2006, 45, 3776–3781. [Google Scholar] [CrossRef]
- Zhu, Y.; Albrecht, K.O.; Elliott, D.C.; Hallen, R.T.; Jones, S.B. Development of hydrothermal liquefaction and upgrading technologies for lipid-extracted algae conversion to liquid fuels. Algal Res. 2013, 2, 455–464. [Google Scholar] [CrossRef]
- Zhang, Y. Hydrothermal Liquefaction to Convert Biomass into Crude Oil; Blackwell Publishing: Hoboken, NJ, USA, 2010; ISBN 9780813802527. [Google Scholar]
- Ross, A.B.; Biller, P.; Kubacki, M.L.; Li, H.; Lea-Langton, A.; Jones, J.M. Hydrothermal processing of microalgae using alkali and organic acids. Fuel 2010, 89, 2234–2243. [Google Scholar] [CrossRef]
- Yang, Y.F.; Feng, C.P.; Inamori, Y.; Maekawa, T. Analysis of energy conversion characteristics in liquefaction of algae. Resour. Conserv. Recycl. 2004, 43, 21–33. [Google Scholar] [CrossRef]
- Beckers, J.; Rothenberg, G. Sustainable selective oxidations using ceria-based materials. Green Chem. 2010, 12, 939. [Google Scholar] [CrossRef]
- Gärtner, C.A.; Serrano-Ruiz, J.C.; Braden, D.J.; Dumesic, J.A. Catalytic upgrading of bio-oils by ketonization. ChemSusChem 2009, 2, 1121–1124. [Google Scholar] [CrossRef] [PubMed]
- Gaertner, C.A.; Serrano-Ruiz, J.C.; Braden, D.J.; Dumesic, J.A. Catalytic coupling of carboxylic acids by ketonization as a processing step in biomass conversion. J. Catal. 2009, 266, 71–78. [Google Scholar] [CrossRef]
- Gaertner, C.A.; Serrano-Ruiz, J.C.; Braden, D.J.; Dumesic, J.A. Ketonization reactions of carboxylic acids and esters over ceria-zirconia as biomass-upgrading processes. Ind. Eng. Chem. Res. 2010, 49, 6027–6033. [Google Scholar] [CrossRef]
- Zastrow, D.J.; Jennings, P.A. Hydrothermal liquefaction of food waste and model food. In Proceedings of the 2013 AIChE Annual Meeting Online Proceedings, San Francisco, CA, USA, 3–8 November 2013; pp. 1–9. [Google Scholar]
- Welcome to the USDA Food Composition Database. Available online: https://ndb.nal.usda.gov/ndb/ (accessed on 14 September 2017).
- Xiong, H.; Pham, H.N.; Datye, A.K. Hydrothermally stable heterogeneous catalysts for conversion of biorenewables. Green Chem. 2014, 16, 4627–4643. [Google Scholar] [CrossRef]
- Gollakota, A.R.K.; Kishore, N.; Gu, S. A review on hydrothermal liquefaction of biomass. Renew. Sustain. Energy Rev. 2018, 81, 1378–1392. [Google Scholar] [CrossRef]
- Sánchez Escribano, V.; Fernández López, E.; Panizza, M.; Resini, C.; Gallardo Amores, J.M.; Busca, G. Characterization of cubic ceria-zirconia powders by X-ray diffraction and vibrational and electronic spectroscopy. Solid State Sci. 2003, 5, 1369–1376. [Google Scholar] [CrossRef]
- Damyanova, S.; Pawelec, B.; Arishtirova, K.; Huerta, M.V.M.; Fierro, J.L.G. Study of the surface and redox properties of ceria-zirconia oxides. Appl. Catal. A Gen. 2008, 337, 86–96. [Google Scholar] [CrossRef]
- Besson, M.; Gallezot, P. Deactivation of metal catalysts in liquid phase organic reactions. Catal. Today 2003, 81, 547–559. [Google Scholar] [CrossRef]
- Kumar, R.; Enjamuri, N.; Shah, S.; Al-Fatesh, A.S.; Bravo-Suárez, J.J.; Chowdhury, B. Ketonization of oxygenated hydrocarbons on metal oxide based catalysts. Catal. Today 2017, 302, 16–49. [Google Scholar] [CrossRef]
- Postole, G.; Chowdhury, B.; Karmakar, B.; Pinki, K.; Banerji, J.; Auroux, A. Knoevenagel condensation reaction over acid-base bifunctional nanocrystalline CexZr1−xO2 solid solutions. J. Catal. 2010, 269, 110–121. [Google Scholar] [CrossRef]
- Saber, M.; Golzary, A.; Hosseinpour, M.; Takahashi, F.; Yoshikawa, K. Catalytic hydrothermal liquefaction of microalgae using nanocatalyst. Appl. Energy 2016, 183, 566–576. [Google Scholar] [CrossRef]
- Robin, T.; Jones, J.M.; Ross, A.B. Catalytic hydrothermal processing of lipids using metal doped zeolites. Biomass Bioenergy 2017, 98, 26–36. [Google Scholar] [CrossRef]
- Nazari, L.; Yuan, Z.; Souzanchi, S.; Ray, M.B.; Xu, C. Hydrothermal liquefaction of woody biomass in hot-compressed water: Catalyst screening and comprehensive characterization of bio-crude oils. Fuel 2015, 162, 74–83. [Google Scholar] [CrossRef]
- Duan, P.; Savage, P.E. Hydrothermal liquefaction of a microalga with heterogeneous catalysts. Ind. Eng. Chem. Res. 2011, 50, 52–61. [Google Scholar] [CrossRef]
- Jena, U.; Das, K.C.; Kastner, J.R. Comparison of the effects of Na2CO3, Ca3(PO4)2, and NiO catalysts on the thermochemical liquefaction of microalga Spirulina platensis. Appl. Energy 2012, 98, 368–375. [Google Scholar] [CrossRef]
- Chen, D.; Ma, Q.; Wei, L.; Li, N.; Shen, Q.; Tian, W. Catalytic hydroliquefaction of rice straw for bio-oil production using Ni/CeO2 catalysts. J. Anal. Appl. Pyrolysis 2018, 130, 169–180. [Google Scholar] [CrossRef]
- Shakya, R.; Whelen, J.; Adhikari, S.; Mahadevan, R.; Neupane, S. Effect of temperature and Na2CO3 catalyst on hydrothermal liquefaction of algae. Algal Res. 2015, 12, 80–90. [Google Scholar] [CrossRef]
- Shuping, Z.; Yulong, W.; Mingde, Y.; Kaleem, I.; Chun, L.; Tong, J. Production and characterization of bio-oil from hydrothermal liquefaction of microalgae Dunaliella tertiolecta cake. Energy 2010, 35, 5406–5411. [Google Scholar] [CrossRef]
- Yang, W.; Li, X.; Liu, S.; Feng, L. Direct hydrothermal liquefaction of undried macroalgae Enteromorpha prolifera using acid catalysts. Energy Convers. Manag. 2014, 87, 938–945. [Google Scholar] [CrossRef]
- Elliott, D.C.; Biller, P.; Ross, A.B.; Schmidt, A.J.; Jones, S.B. Hydrothermal liquefaction of biomass: Developments from batch to continuous process. Bioresour. Technol. 2015, 178, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Von Keitz, M.; Valentas, K. Thermal effects on hydrothermal biomass liquefaction. Appl. Biochem. Biotechnol. 2008, 147, 143–150. [Google Scholar] [CrossRef] [PubMed]
| Food Item | Feedstock Percent (Dry Basis) | Feedstock Composition and Heating Values | Value [% or MJ/kg] |
|---|---|---|---|
| American Cheese | 12.8 | Moisture [%] | 73.0 |
| Canned Chicken | 14.9 | Protein [%] | 4.8 |
| Instant Potatoes | 10.6 | Lipids [%] | 5.9 |
| Green Beans | 14.9 | Carbohydrates [%] | 15.9 |
| White Rice | 19.1 | Ash [%] | 0.3 |
| Apple Sauce | 22.3 | HHV, bone dry [MJ/kg] | 24.6 |
| Butter | 5.4 | HHV, wet [MJ/kg] | 6.5 |
| Catalyst | C Content [%] | H Content [%] | O Content [%] | N Content [%] | Moisture Content [%] | HHV 1 [MJ/kg] | Energy Recovery [%] | HTL Water TOC [ppm] |
|---|---|---|---|---|---|---|---|---|
| Food Waste 2 | 58.3 | 10.3 | 29.3 | 2.0 | N/A | 24.6 | N/A | N/A |
| Thermal | 79.0 | 10.3 | 6.3 | 4.4 | 10.5 | 35.6 | 27.6 | 13,800 |
| 5% Na2CO3 | 77.6 | 10.2 | 8.4 | 3.8 | 11.7 | 24.2 | 21.3 | 24,200 |
| 5% CeZrOx | 80.8 | 10.1 | 4.7 | 4.5 | 10.1 | 31.2 | 38.8 | 12,500 |
| Model Compounds | Alcohol | Carboxylic Acid | Ketone | Aldehyde |
|---|---|---|---|---|
| Alcohol (Isobutanol) | No Product | |||
| Carboxylic Acid (Propionic Acid) | No Product | No Product | ||
| Ketone (3-Pentanone) | Trace Product | Trace Product | Trace Product | |
| Aldehyde (Pentanal) | Slightly Reactive | Slightly Reactive | Slightly Reactive | Reactive |
| Feedstock | Temperature (°C)/Catalyst Loading (Dry Feed Basis) | Catalyst | Energy Recovery | Oil Yield Improvement | Oil HHV Improvement |
|---|---|---|---|---|---|
| Food Waste | 300 °C/33% | CeZrOx | 38.8 | 1.59 | 0.88 |
| Na2CO3 | 21.3 | 1.12 | 0.68 | ||
| Vegetable Oil [32] | 350 °C/16% | Cr-ZSM5 | 75.8 | 0.81 | 1.01 |
| Co-ZSM5 | 70.0 | 0.73 | 1.04 | ||
| H-ZSM5 | 77.4 | 0.79 | 1.06 | ||
| Rice Straw [36] | 290 °C/5% | Ni/CeO2 | 81.6 | 1.39 | 1.22 |
| Sawdust [33] | 300 °C/20% | Hydrotalcite | 52.3 | 1.82 | 0.84 |
| MgO | 48.6 | 1.60 | 0.93 | ||
| Colemanite | 57.1 | 1.92 | 0.92 | ||
| Spirunella Algae [35] | 350 °C/20% | NiO | 56.5 | 0.76 | 1.08 |
| Microalgae [31] | 250 °C/50% | Nano-Ni/SiO2 | 28.9 | 1.49 | 1.05 |
| Zeolite | 27.8 | 1.42 | 1.06 |
| Feedstock | Catalyst | Temperature (°C)/Residence Time (h) | Lifetime Energy Yield [MJoil/gcat] | Ref. | |
|---|---|---|---|---|---|
| 1 Reuse | Expected Reuse [# of Reuses] | ||||
| Food Waste | CeZrOx | 300 °C/1 h | 0.242 | 0.73 [3×] | Our work |
| CeZrOx | 300 °C/1 h | 0.242 | 2.42 [10×] | Our work | |
| CeZrOx | 300 °C/1 h | 0.242 | 24.2 [100×] | Our work | |
| CeZrOx | 300 °C/1 h | 0.242 | 39.9 [165×] | Our work | |
| Na2CO3 | 300 °C/1 h | 0.103 | 0.21 [2×] | Our work | |
| Algae 1 | Na2CO3 | 300–360 °C/0.5–1 h | 0.0061–0.362 | 0.007–0.37 [2×] | [16,17,35,37,38] |
| KOH | 300 °C/1 h | 0.010–0.014 | 0.020–0.028 [2×] | [18] | |
| Acetic Acid | 290–300 °C/0.33–1 h | 0.007–0.398 | 0.015–0.80 [2×] | [18,39] | |
| Formic Acid | 300 °C/1 h | 0.012–0.019 | 0.024–0.038 [2×] | [18] | |
| Ca3(PO4)2 | 350 °C/1 h | 0.250 | 0.5 [2×] | [38] | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maag, A.R.; Paulsen, A.D.; Amundsen, T.J.; Yelvington, P.E.; Tompsett, G.A.; Timko, M.T. Catalytic Hydrothermal Liquefaction of Food Waste Using CeZrOx. Energies 2018, 11, 564. https://doi.org/10.3390/en11030564
Maag AR, Paulsen AD, Amundsen TJ, Yelvington PE, Tompsett GA, Timko MT. Catalytic Hydrothermal Liquefaction of Food Waste Using CeZrOx. Energies. 2018; 11(3):564. https://doi.org/10.3390/en11030564
Chicago/Turabian StyleMaag, Alex R., Alex D. Paulsen, Ted J. Amundsen, Paul E. Yelvington, Geoffrey A. Tompsett, and Michael T. Timko. 2018. "Catalytic Hydrothermal Liquefaction of Food Waste Using CeZrOx" Energies 11, no. 3: 564. https://doi.org/10.3390/en11030564
APA StyleMaag, A. R., Paulsen, A. D., Amundsen, T. J., Yelvington, P. E., Tompsett, G. A., & Timko, M. T. (2018). Catalytic Hydrothermal Liquefaction of Food Waste Using CeZrOx. Energies, 11(3), 564. https://doi.org/10.3390/en11030564






