Release Mechanism of Fuel-N into NOx and N2O Precursors during Pyrolysis of Rice Straw
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
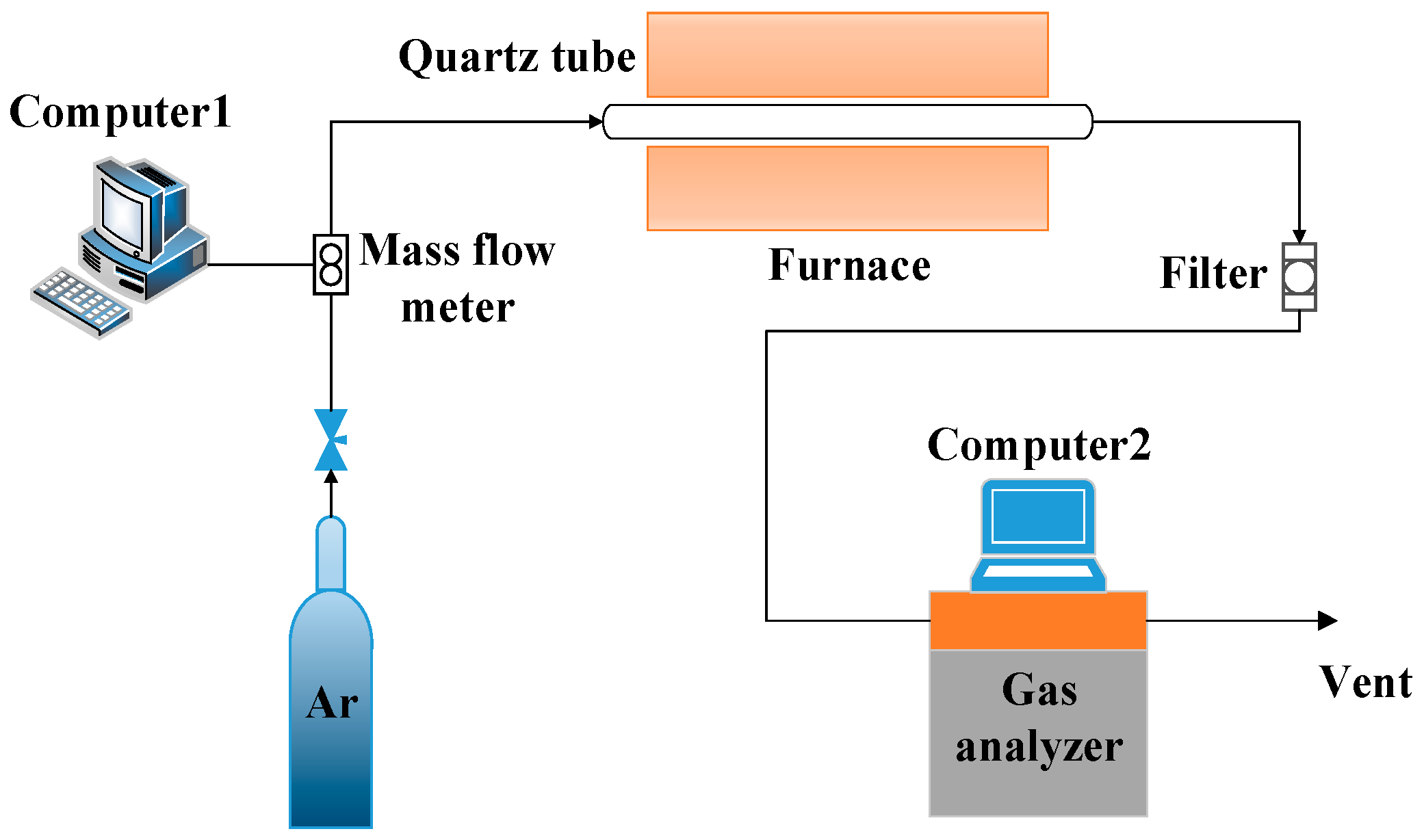
2.2. Experimental Methods
3. Results and Discussion
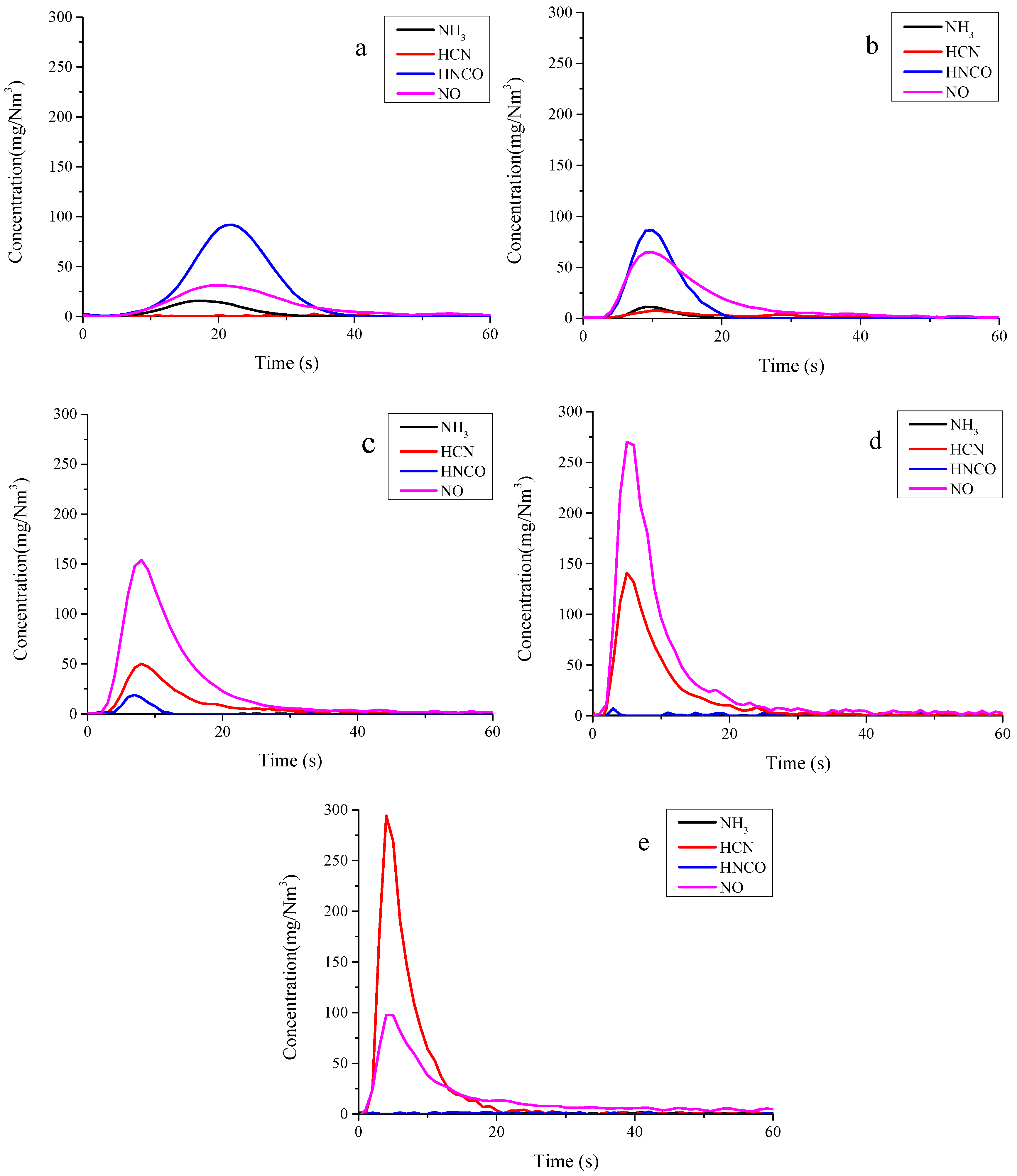
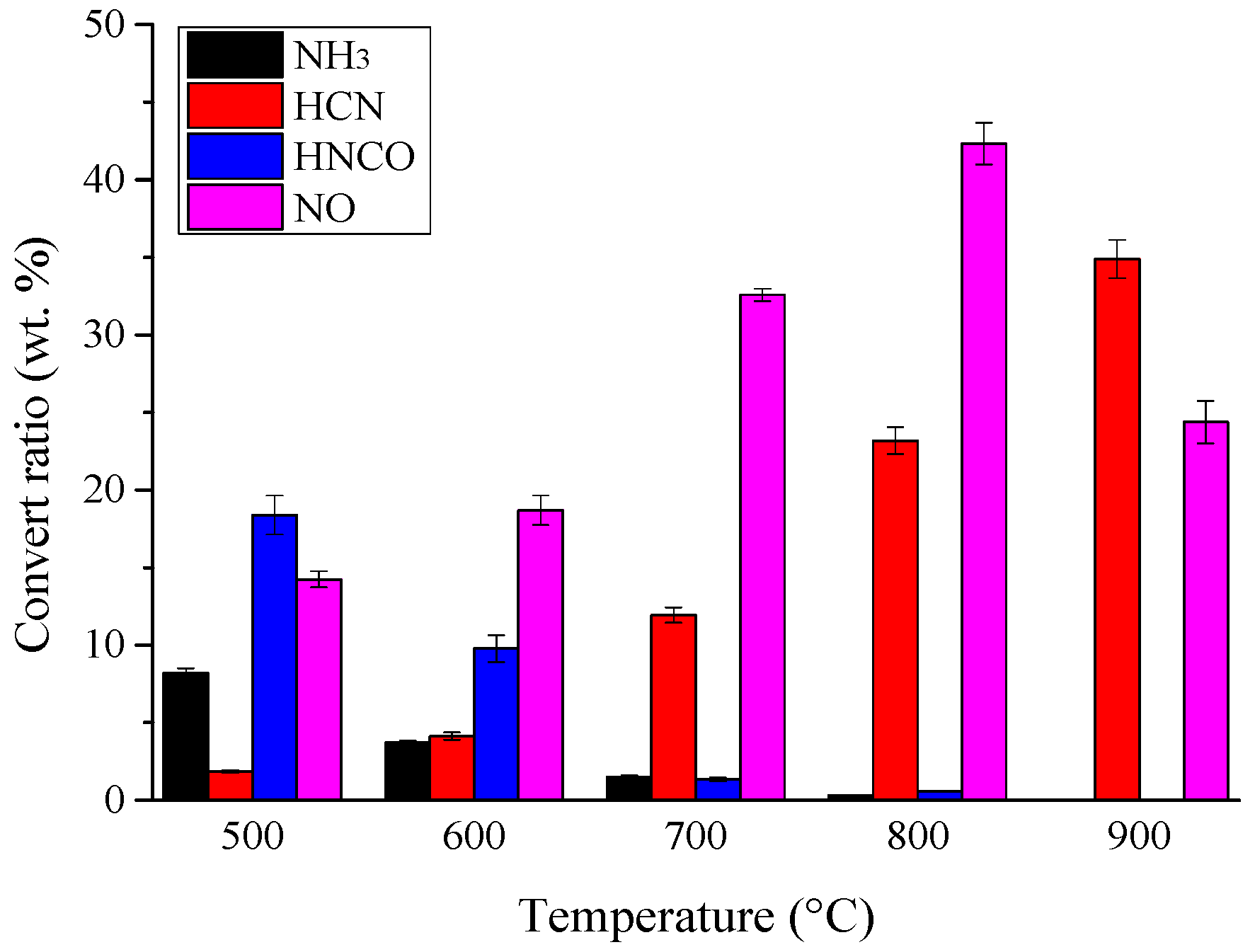
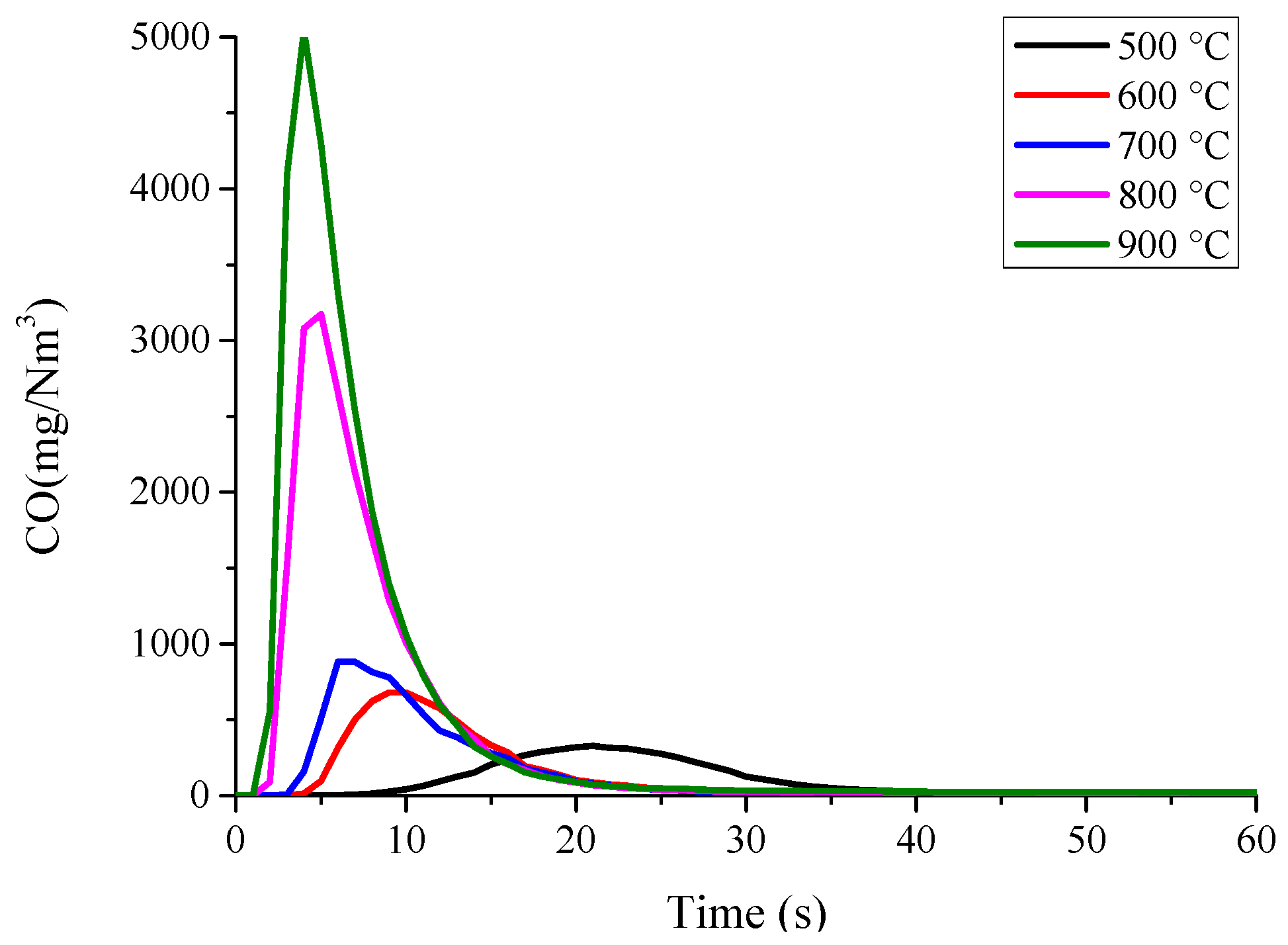
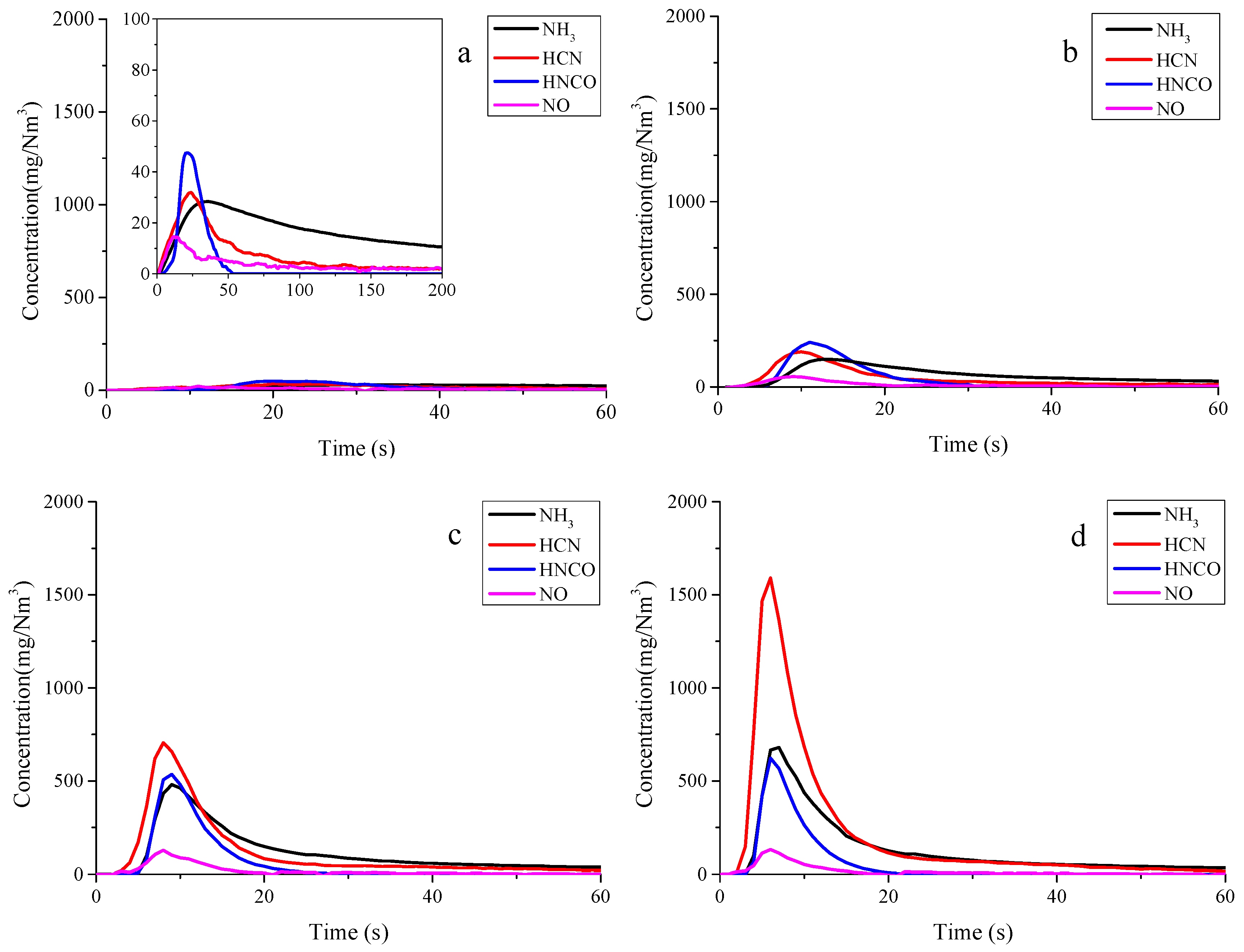
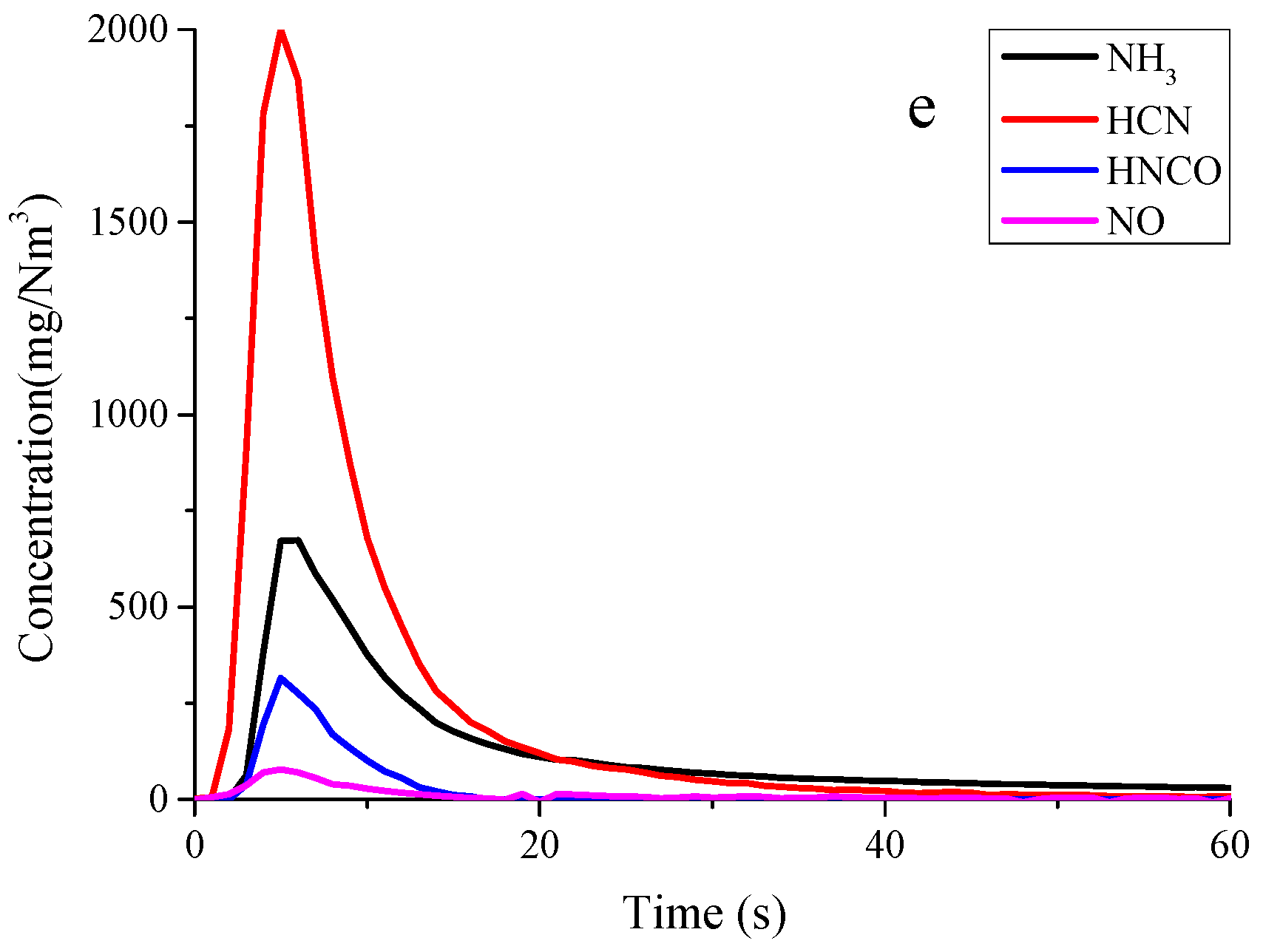
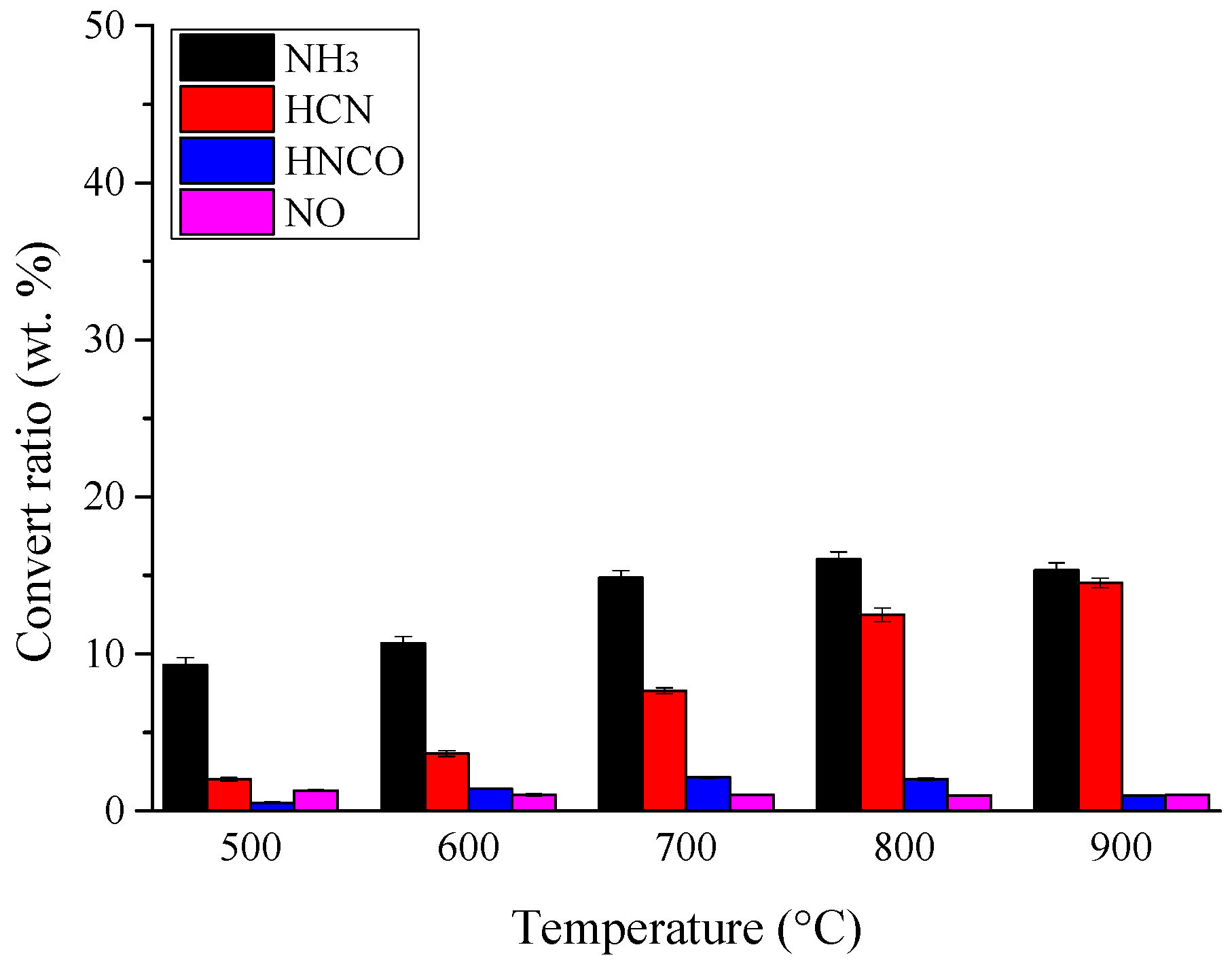
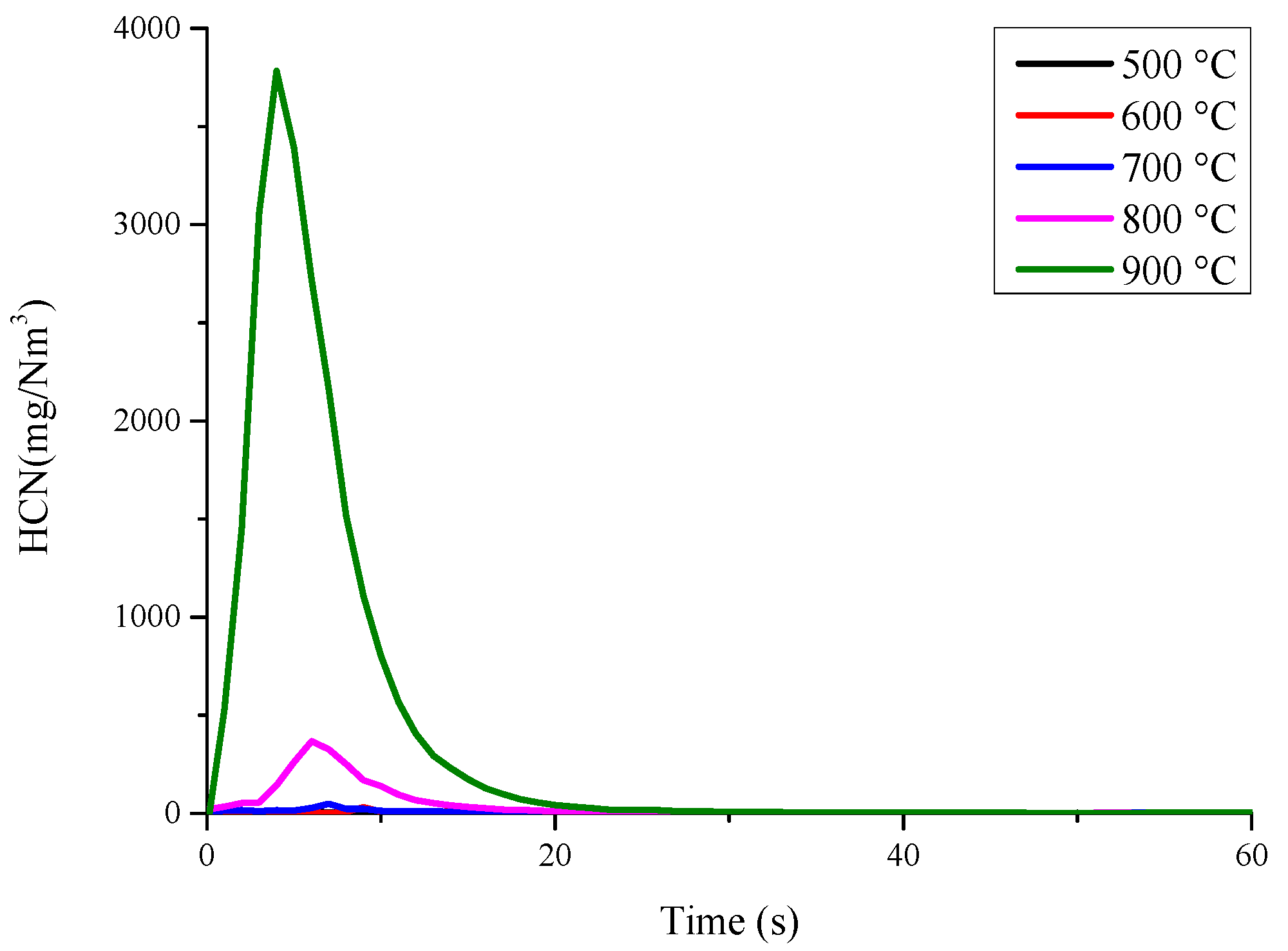
3.1. Nitrogen Release during Pyrolysis of Rice Straw
3.2. Nitrogen Release during Pyrolysis of Nitrogen Modeling Compounds
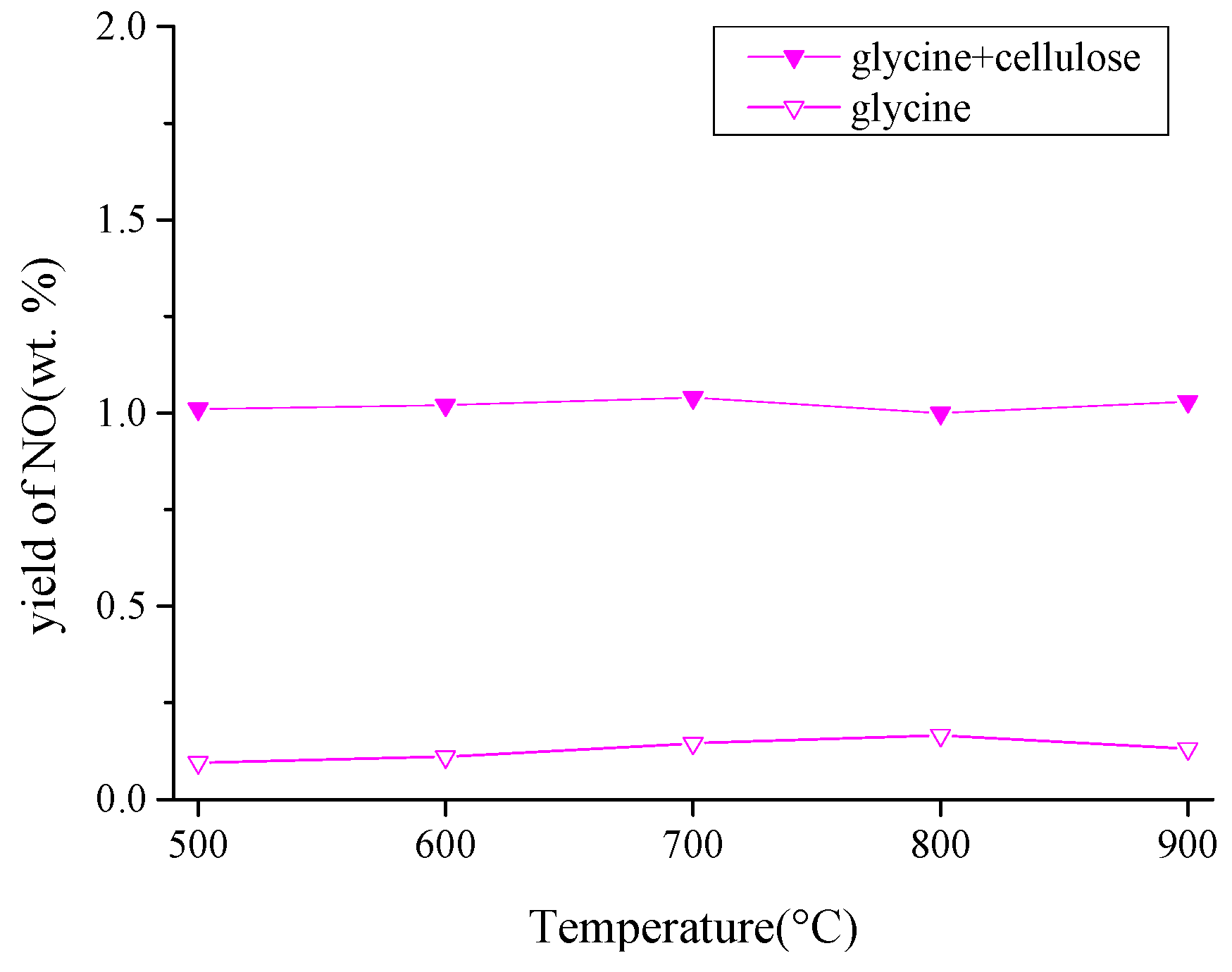
3.2.1. Pyrolysis of Glycine
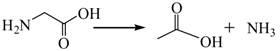
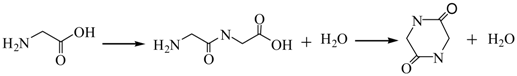
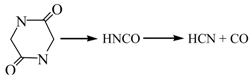
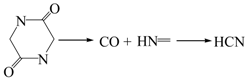
3.2.2. Pyrolysis of Pyrrole
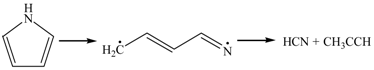
3.2.3. Effect of –OH on the Formation of NO
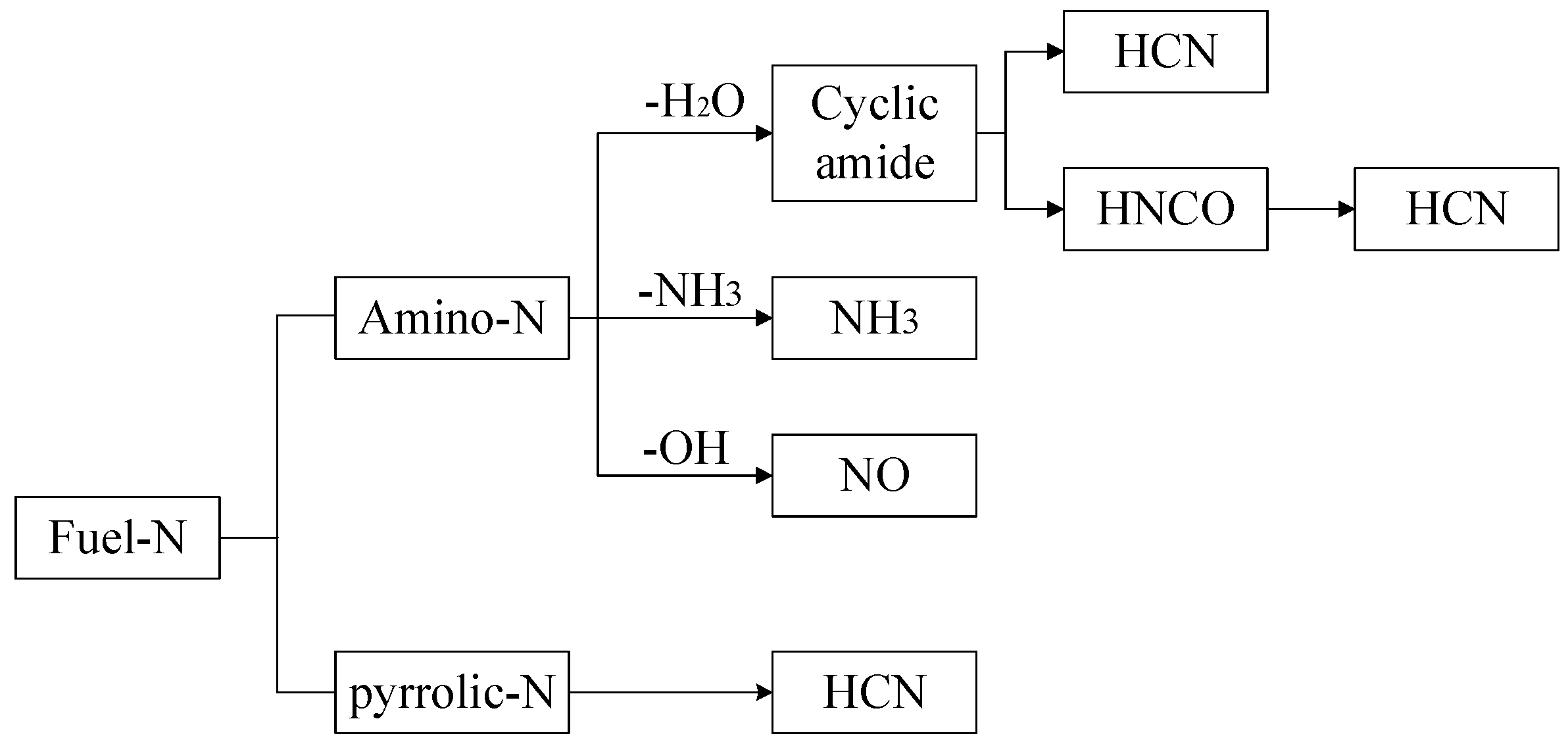
3.3. Mechanism of Nitrogen Release during Rice Straw Pyrolysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tripathi, M.; Sahu, J.N.; Ganesan, P. Effect of process parameters on production of biochar from biomass waste through pyrolysis. Renew. Sustain. Energy Rev. 2016, 55, 467–481. [Google Scholar] [CrossRef]
- Chen, H.; Si, Y.; Chen, Y.; Yang, H.; Chen, D.; Chen, W. NOx precursors from biomass pyrolysis: Distribution of amino acids in biomass and Tar-N during devolatilization using model compounds. Fuel 2017, 187, 367–375. [Google Scholar] [CrossRef]
- Shan, F.; Lin, Q.; Zhou, K.; Wu, Y.; Fu, W.; Zhang, P.; Song, L.; Shao, C.; Yi, B. An experimental study of ignition and combustion of single biomass pellets in air and oxy-fuel. Fuel 2017, 188, 277–284. [Google Scholar] [CrossRef]
- Wang, S.; Hu, Y.; Wang, Q.; Xu, S.; Lin, X.; Ji, H.; Zhang, Z. TG-FTIR-MS analysis of the pyrolysis of blended seaweed and rice husk. J. Therm. Anal. Calorim. 2016, 126, 1689–1702. [Google Scholar] [CrossRef]
- Houshfar, E.; Løvås, T.; Skreiberg, Ø. Experimental Investigation on NOx Reduction by Primary Measures in Biomass Combustion: Straw, Peat, Sewage Sludge, Forest Residues and Wood Pellets. Energies 2012, 5, 270–290. [Google Scholar] [CrossRef]
- Molina-Moreno, V.; Leyva-Díaz, J.; Sánchez-Molina, J. Pellet as a Technological Nutrient within the Circular Economy Model: Comparative Analysis of Combustion Efficiency and CO and NOx Emissions for Pellets from Olive and Almond Trees. Energies 2016, 9, 777. [Google Scholar] [CrossRef]
- Yang, X.; Wang, H.; Strong, P.J.; Xu, S.; Liu, S.; Lu, K.; Sheng, K.; Guo, J.; Che, L.; He, L.; et al. Thermal Properties of Biochars Derived from Waste Biomass Generated by Agricultural and Forestry Sectors. Energies 2017, 10, 469. [Google Scholar] [CrossRef]
- Gao, P.; Xue, L.; Lu, Q.; Dong, C. Effects of Alkali and Alkaline Earth Metals on N-Containing Species Release during Rice Straw Pyrolysis. Energies 2015, 8, 13021–13032. [Google Scholar] [CrossRef]
- Ren, X.; Sun, R.; Meng, X.; Vorobiev, N.; Schiemann, M.; Levendis, Y.A. Carbon, sulfur and nitrogen oxide emissions from combustion of pulverized raw and torrefied biomass. Fuel 2017, 188, 310–323. [Google Scholar] [CrossRef]
- Ren, Q.; Zhao, C. Evolution of fuel-N in gas phase during biomass pyrolysis. Renew. Sustain. Energy Rev. 2015, 50, 408–418. [Google Scholar] [CrossRef]
- Ghouma, I.; Jeguirim, M.; Sager, U.; Limousy, L.; Bennici, S.; Däuber, E.; Asbach, C.; Ligotski, R.; Schmidt, F.; Ouederni, A. The Potential of Activated Carbon Made of Agro-Industrial Residues in NOx Immissions Abatement. Energies 2017, 10, 1508. [Google Scholar] [CrossRef]
- Glarborg, P.; Jensen, A.D.; Johnsson, J.E. Fuel nitrogen conversion in solid fuel fired systems. Prog. Energy Combust. Sci. 2003, 29, 89–113. [Google Scholar] [CrossRef]
- Chen, H.; Wang, Y.; Xu, G.; Yoshikawa, K. Fuel-N Evolution during the Pyrolysis of Industrial Biomass Wastes with High Nitrogen Content. Energies 2012, 5, 5418–5438. [Google Scholar] [CrossRef]
- Yuan, S.; Zhou, Z.J.; Li, J.; Chen, X.L.; Wang, F.C. HCN and NH3 (NOx precursors) released under rapid pyrolysis of biomass/coal blends. J. Anal. Appl. Pyrolysis 2011, 92, 463–469. [Google Scholar] [CrossRef]
- Ren, Q.; Zhao, C.; Wu, X.; Liang, C.; Chen, X.; Shen, J.; Wang, Z. Formation of NOx precursors during wheat straw pyrolysis and gasification with O2 and CO2. Fuel 2010, 89, 1064–1069. [Google Scholar] [CrossRef]
- Yuan, S.; Chen, X.L.; Li, W.F.; Liu, H.F.; Wang, F.C. Nitrogen conversion under rapid pyrolysis of two types of aquatic biomass and corresponding blends with coal. Bioresour. Technol. 2011, 102, 10124–10130. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.; Zhou, Z.J.; Li, J.; Chen, X.L.; Wang, F.C. HCN and NH3 Released from Biomass and Soybean Cake under Rapid Pyrolysis. Energy Fuels 2010, 24, 6166–6171. [Google Scholar] [CrossRef]
- Tian, F.J.; Yu, J.; McKenzie, L.J.; Hayashi, J.I.; Li, C.Z. Conversion of Fuel-N into HCN and NH3 during the Pyrolysis and Gasification in Steam: A Comparative Study of Coal and Biomass. Energy Fuels 2007, 21, 517–521. [Google Scholar] [CrossRef]
- Tian, F.J.; Yu, J.L.; Mckenzie, L.J.; Hayashi, J.I.; Chiba, T.; Li, C.Z. Formation of NO precursors during the pyrolysis of coal and biomass. Part VII. Pyrolysis and gasification of cane trash with steam. Fuel 2005, 84, 371–376. [Google Scholar] [CrossRef]
- Chang, L.; Xie, Z.; Xie, K.C.; Pratt, K.C.; Hayashi, J.I.; Chiba, T.; Li, C.Z. Formation of NOx precursors during the pyrolysis of coal and biomass. Part VI. Effects of gas atmosphere on the formation of NH3 and HCN. Fuel 2003, 82, 1159–1166. [Google Scholar] [CrossRef]
- Tan, L.L.; Li, C. Formation of NOx and SOx precursors during the pyrolysis of coal and biomass. Part I. Effects of reactor configuration on the determined yields of HCN and NH3 during pyrolysis. Fuel 2000, 79, 1883–1889. [Google Scholar] [CrossRef]
- Zhan, H.; Yin, X.; Huang, Y.; Yuan, H.; Xie, J.; Wu, C.; Shen, Z.; Cao, J. Comparisons of Formation Characteristics of NOx Precursors during Pyrolysis of Lignocellulosic Industrial Biomass Wastes. Energy Fuels 2017, 31, 9557–9567. [Google Scholar] [CrossRef]
- Hansson, K.M.; Samuelsson, J.; Tullin, C.; Åmand, L.E. Formation of HNCO, HCN, and NH3 from the pyrolysis of bark and nitrogen-containing model compounds. Combust. Flame 2004, 137, 265–277. [Google Scholar] [CrossRef]
- Zhu, X.; Yang, S.; Wang, L.; Liu, Y.; Qian, F.; Yao, W.; Zhang, S.; Chen, J. Tracking the conversion of nitrogen during pyrolysis of antibiotic mycelial fermentation residues using XPS and TG-FTIR-MS technology. Environ. Pollut. 2016, 211, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Si, J.; Tan, H.; Ma, L.; Pourkashanian, M.; Xu, T. Nitrogen, Sulfur, and Chlorine Transformations during the Pyrolysis of Straw. Energy Fuels 2010, 24, 5215–5221. [Google Scholar] [CrossRef]
- De Jong, W.; Di Nola, G.; Venneker, B.C.; Spliethoff, H.; Wójtowicz, M.A. TG-FTIR pyrolysis of coal and secondary biomass fuels: Determination of pyrolysis kinetic parameters for main species and NOx precursors. Fuel 2007, 86, 2367–2376. [Google Scholar] [CrossRef]
- Becidan, M.; Skreiberg, Ø.; Hustad, J.E. NOx and N2O Precursors (NH3 and HCN) in Pyrolysis of Biomass Residues. Energy Fuels 2007, 21, 1173–1180. [Google Scholar] [CrossRef]
- Ren, Q.; Zhao, C. Effect of mineral matter on the formation of NOx precursors during biomass pyrolysis. J. Anal. Appl. Pyrolysis 2009, 85, 447–453. [Google Scholar] [CrossRef]
- Ren, Q.; Zhao, C. NOx and N2O Precursors from Biomass Pyrolysis: Role of Cellulose, Hemicellulose and Lignin. Environ. Sci. Technol. 2013, 47, 8955–8961. [Google Scholar] [CrossRef] [PubMed]
- Ren, Q.; Zhao, C. NOx and N2O precursors (NH3 and HCN) from biomass pyrolysis: Interaction between amino acid and mineral matter. Appl. Energy 2013, 112, 170–174. [Google Scholar] [CrossRef]
- Ren, Q.; Zhao, C. NOx and N2O Precursors from Biomass Pyrolysis: Nitrogen Transformation from Amino Acid. Environ. Sci. Technol. 2012, 46, 4236–4240. [Google Scholar] [CrossRef] [PubMed]
- Jie, L.; Yuwen, L.; Jingyan, S.; Zhiyong, W.; Ling, H.; Xi, Y.; Cunxin, W. The investigation of thermal decomposition pathways of phenylalanine and tyrosine by TG–FTIR. Thermochim. Acta 2008, 467, 20–29. [Google Scholar] [CrossRef]
- Li, J.; Wang, Z.; Yang, X.; Hu, L.; Liu, Y.; Wang, C. Evaluate the pyrolysis pathway of glycine and glycylglycine by TG–FTIR. J. Anal. Appl. Pyrolysis 2007, 80, 247–253. [Google Scholar] [CrossRef]
- Titantah, J.T.; Lamoen, D. Carbon and nitrogen 1s energy levels in amorphous carbon nitride systems: XPS interpretation using first-principles. Diam. Relat. Mater. 2007, 16, 581–588. [Google Scholar] [CrossRef]
- Schmiers, H.; Friebel, J.; Streubel, P.; Hesse, R.; Köpsel, R. Change of chemical bonding of nitrogen of polymeric N-heterocyclic compounds during pyrolysis. Carbon 1999, 37, 1965–1978. [Google Scholar] [CrossRef]
- Jansen, R.J.J.; van Bekkum, H. XPS of nitrogen-containing functional groups on activated carbon. Carbon 1995, 33, 1021–1027. [Google Scholar] [CrossRef]
- Deng, L.; Jin, X.; Zhang, Y.; Che, D. Release of nitrogen oxides during combustion of model coals. Fuel 2016, 175, 217–224. [Google Scholar] [CrossRef]
- Hansson, K.; Samuelsson, J.; Åmand, L.; Tullin, C. The temperature’s influence on the selectivity between HNCO and HCN from pyrolysis of 2,5-diketopiperazine and 2-pyridone. Fuel 2003, 82, 2163–2172. [Google Scholar] [CrossRef]
- Johnsson, J.E. Formation and reduction of nitrogen oxides in fluidized-bed combustion. Fuel 1994, 73, 1398–1415. [Google Scholar] [CrossRef]
- Ren, Q.; Zhao, C.; Chen, X.; Duan, L.; Li, Y.; Ma, C. NOx and N2O precursors (NH3 and HCN) from biomass pyrolysis: Co-pyrolysis of amino acids and cellulose, hemicellulose and lignin. Proc. Combust. Inst. 2011, 33, 1715–1722. [Google Scholar] [CrossRef]
- Lifshitz, A.; Tamburu, C.; Suslensky, A. Isomerization and Decomposition of Pyrrole at Elevated Temperatures: Studies with a Single-Pulse Shock Tube. J. Chem. Phys. 1989, 93, 5802–5808. [Google Scholar] [CrossRef]
- Zhai, L.; Zhou, X.; Liu, R. A Theoretical Study of Pyrolysis Mechanisms of Pyrrole. J. Phys. Chem. A 1999, 103, 3917–3922. [Google Scholar] [CrossRef]
- Mackie, J.C.; Colket, M.B.; Nelson, P.F.; Esler, M. Shock Tube Pyrolysis of Pyrrole and Kinetic Modeling. Int. J. Chem. Kinet. 1991, 23, 733–760. [Google Scholar] [CrossRef]
- Li, C.; Tan, L.L. Formation of NOx and SOx precursors during the pyrolysis of coal and biomass. Part III. Further discussion on the formation of HCN and NH3 during pyrolysis. Fuel 2000, 79, 1899–1906. [Google Scholar] [CrossRef]
- Binod, P.; Sindhu, R.; Singhania, R.R.; Vikram, S.; Devi, L.; Nagalakshmi, S.; Kurien, N.; Sukumaran, R.K.; Pandey, A. Bioethanol production from rice straw: An overview. Bioresour. Technol. 2010, 101, 4767–4774. [Google Scholar] [CrossRef] [PubMed]
- Saha, B.C. Hemicellulose bioconversion. J. Ind. Microbiol. Biotechnol. 2003, 30, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Wen, L.; Liu, M.; Yang, T. Interaction Characteristics of Mineral Matter and Nitrogen during Sewage Sludge Pyrolysis. Energy Fuels 2016, 30, 10505–10510. [Google Scholar] [CrossRef]
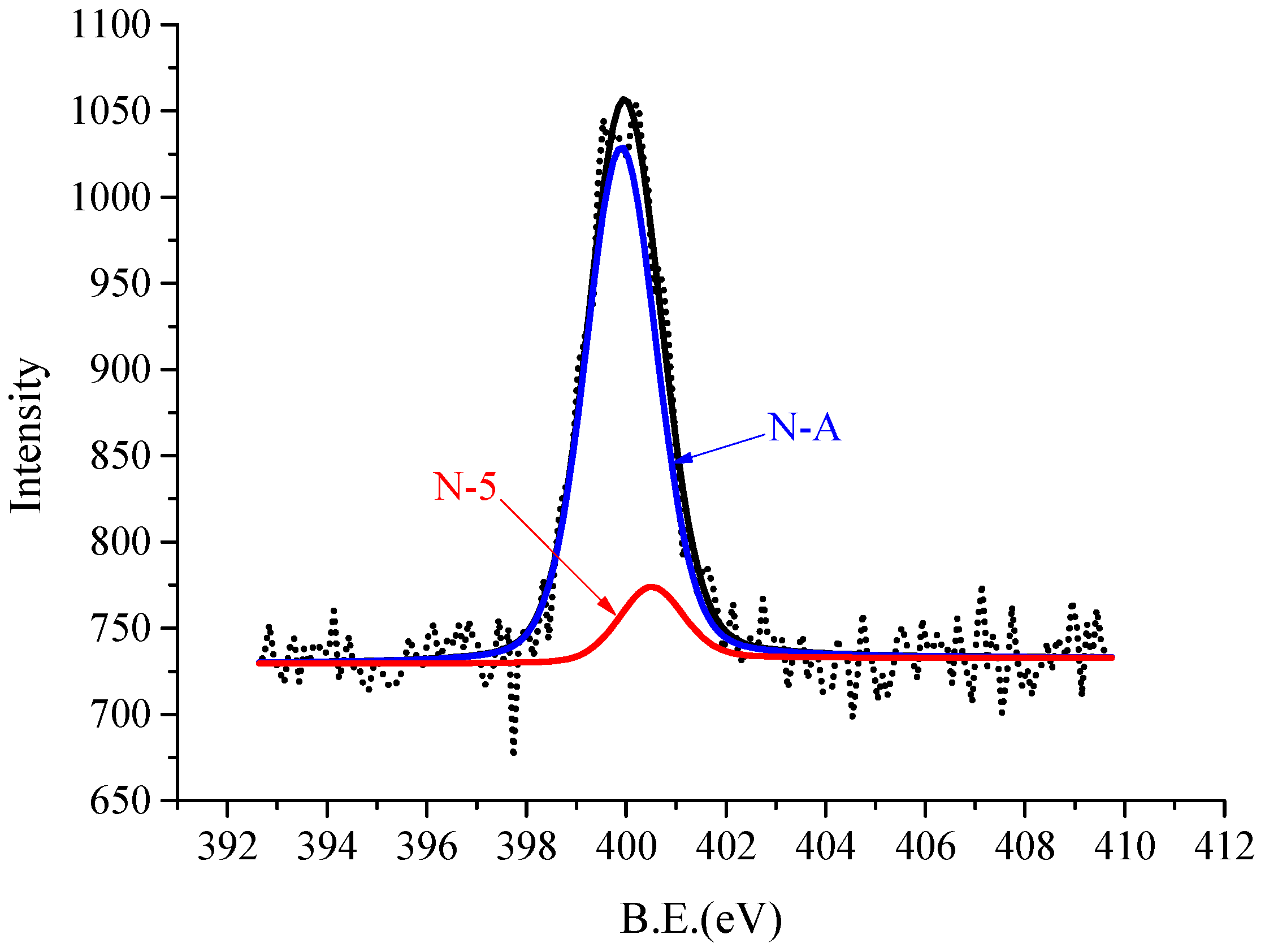
| Proximate Analysis | Ultimate Analysis | |||||||
|---|---|---|---|---|---|---|---|---|
| M a | A a | V a | FC a | C | H | N | S | O |
| 1.51 | 11.31 | 69.09 | 18.09 | 42.66 | 5.68 | 0.64 | 0.44 | 39.10 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, X.; Luo, Z.; Yu, C.; Jin, B.; Tu, H. Release Mechanism of Fuel-N into NOx and N2O Precursors during Pyrolysis of Rice Straw. Energies 2018, 11, 520. https://doi.org/10.3390/en11030520
Liu X, Luo Z, Yu C, Jin B, Tu H. Release Mechanism of Fuel-N into NOx and N2O Precursors during Pyrolysis of Rice Straw. Energies. 2018; 11(3):520. https://doi.org/10.3390/en11030520
Chicago/Turabian StyleLiu, Xiaorui, Zhongyang Luo, Chunjiang Yu, Bitao Jin, and Hanchao Tu. 2018. "Release Mechanism of Fuel-N into NOx and N2O Precursors during Pyrolysis of Rice Straw" Energies 11, no. 3: 520. https://doi.org/10.3390/en11030520
APA StyleLiu, X., Luo, Z., Yu, C., Jin, B., & Tu, H. (2018). Release Mechanism of Fuel-N into NOx and N2O Precursors during Pyrolysis of Rice Straw. Energies, 11(3), 520. https://doi.org/10.3390/en11030520






