2. Results
As an extraction procedure with a defined solvent system always has a different selectivity for different lipid classes (neutral lipids, polar lipids, glycolipids, polyunsaturated fatty acids…) there is no possibility to determine an absolute lipid content in general [
17], it can only be compared when applying the same extraction method. Therefore, an extraction process always only extracts a subset of a theoretical total lipid content. For this study, the algae samples were processed by three different ways (
Figure 1), which always include the same last extraction step with n-hexane. First was direct extraction of the samples without any pre-treatment. This was used as a base line and to determine if direct extraction without pre-treatment is a feasible way. Second was the extraction with prior acidic hydrolysis as it is often done for determination of the lipid fraction of any kind of biomass. This is a mature pre-treatment process. Third is the extraction with prior hydrothermal carbonization as a novel pre-treatment process. Hydrothermal treatment was always conducted for 90 min at 200 °C.
After conducting the different pre-treatment (no pre-treatment, acidic hydrolysis, hydrothermal treatment) and extraction on the different microalgae species, the microalgae and the products were analysed.
The elemental composition of the different algae species (
Table 1) shows that there are similarities concerning Nitrogen content (always above 9 wt %) but some variance for the Carbon content, especially for
N. oculata. In general, the elemental composition is in a normal range.
The greatest variance between the different algae species lies in the ash content with 4.58 wt % to 23.10 wt %, originating from several ash-forming elements contained inside the algal cell. The high ash content of
N. oculata can be explained by its growth medium (salt water). Main fractions of these are alkali metals like Potassium and Sodium. These are known for their catalytic impact [
18,
19,
20,
21] and might change the proportion of liquefied and solid products and thereby the yield of extractable lipids from the HTC char. The influence of this for the given experiments remains a subject to further research.
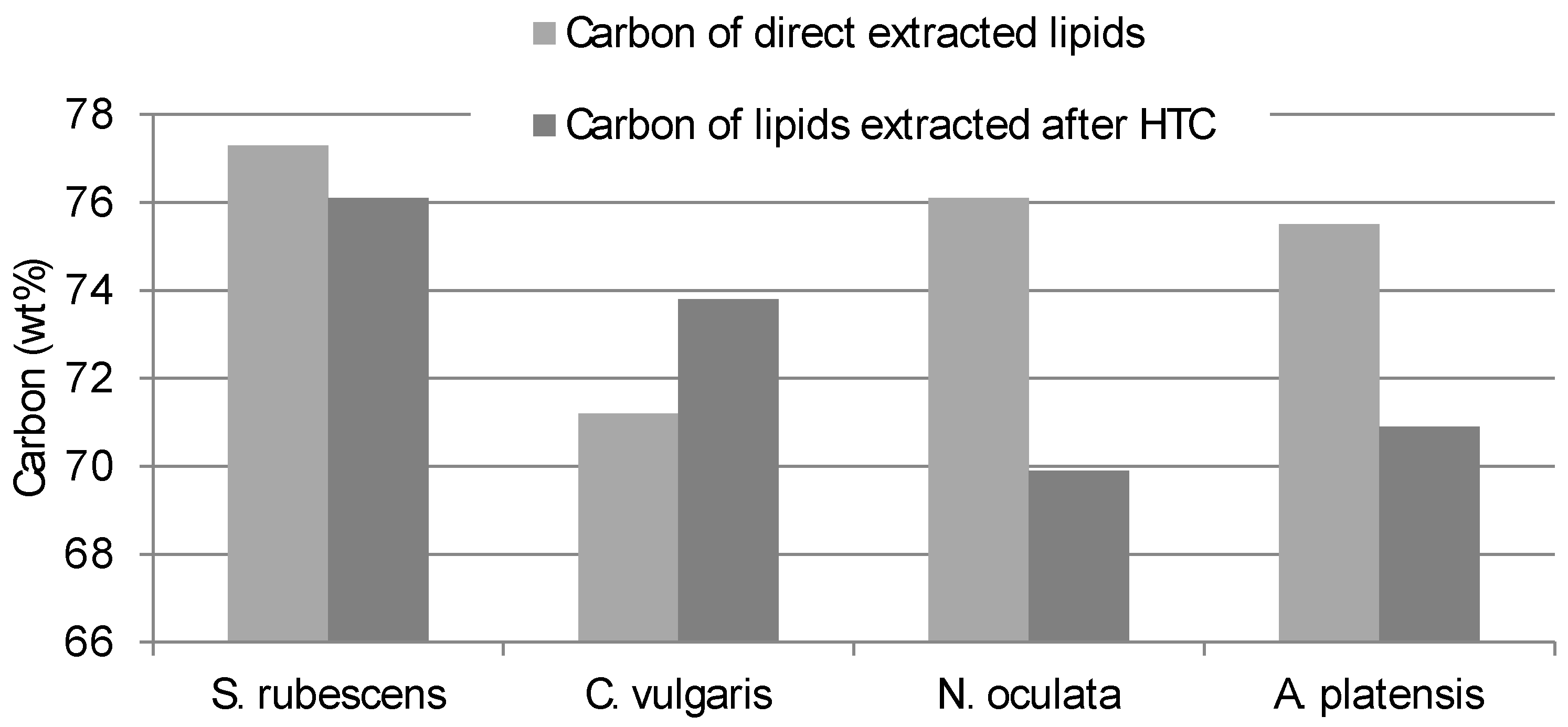
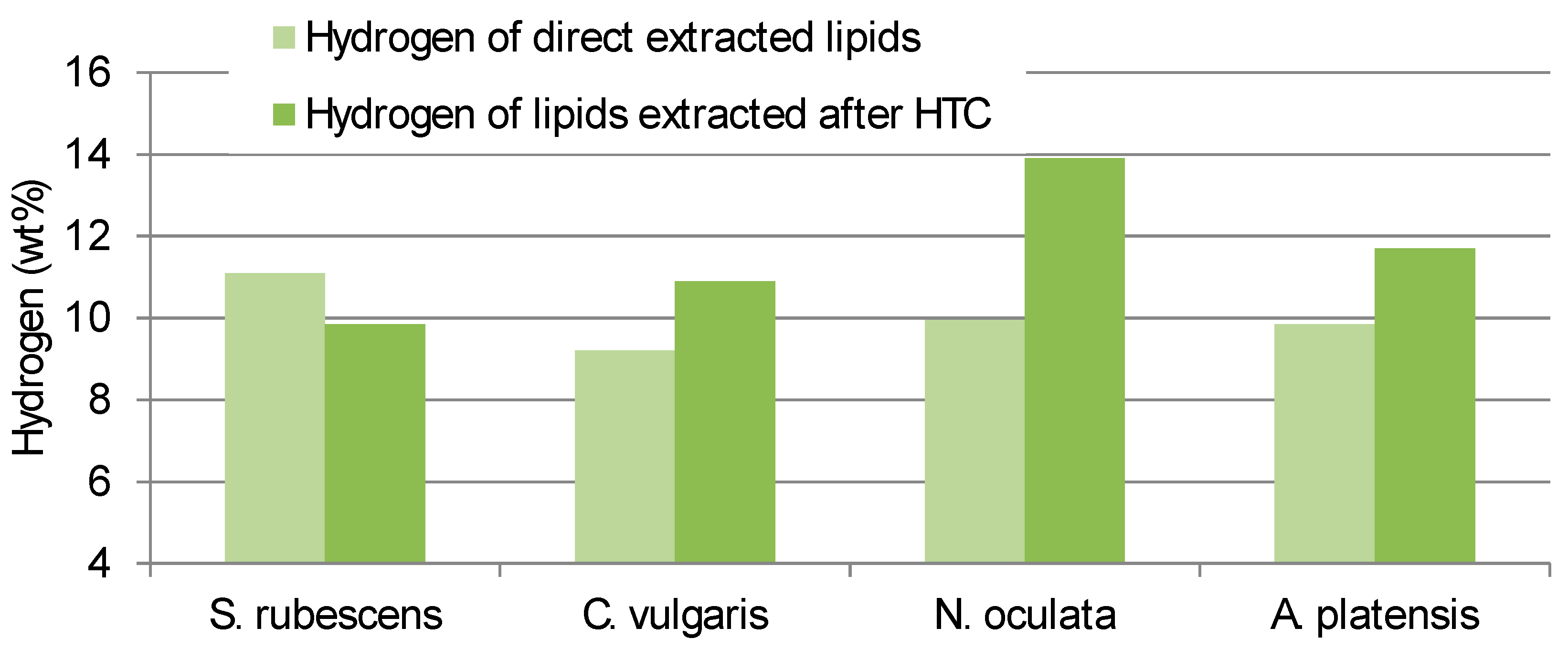
The Carbon and Hydrogen content of the lipid obtained from direct extraction consistently differed from the contents of the lipid extracted from the HTC-char (
Figure 2 and
Figure 3). Interestingly
S. rubescens has the lowest differences between direct extracted and HTC pre-treated extracts.
Therefore, not only the quantity of the lipid yield differs but also the quality of the extract is different between the pre-treated microalgae and the not pre-treated.
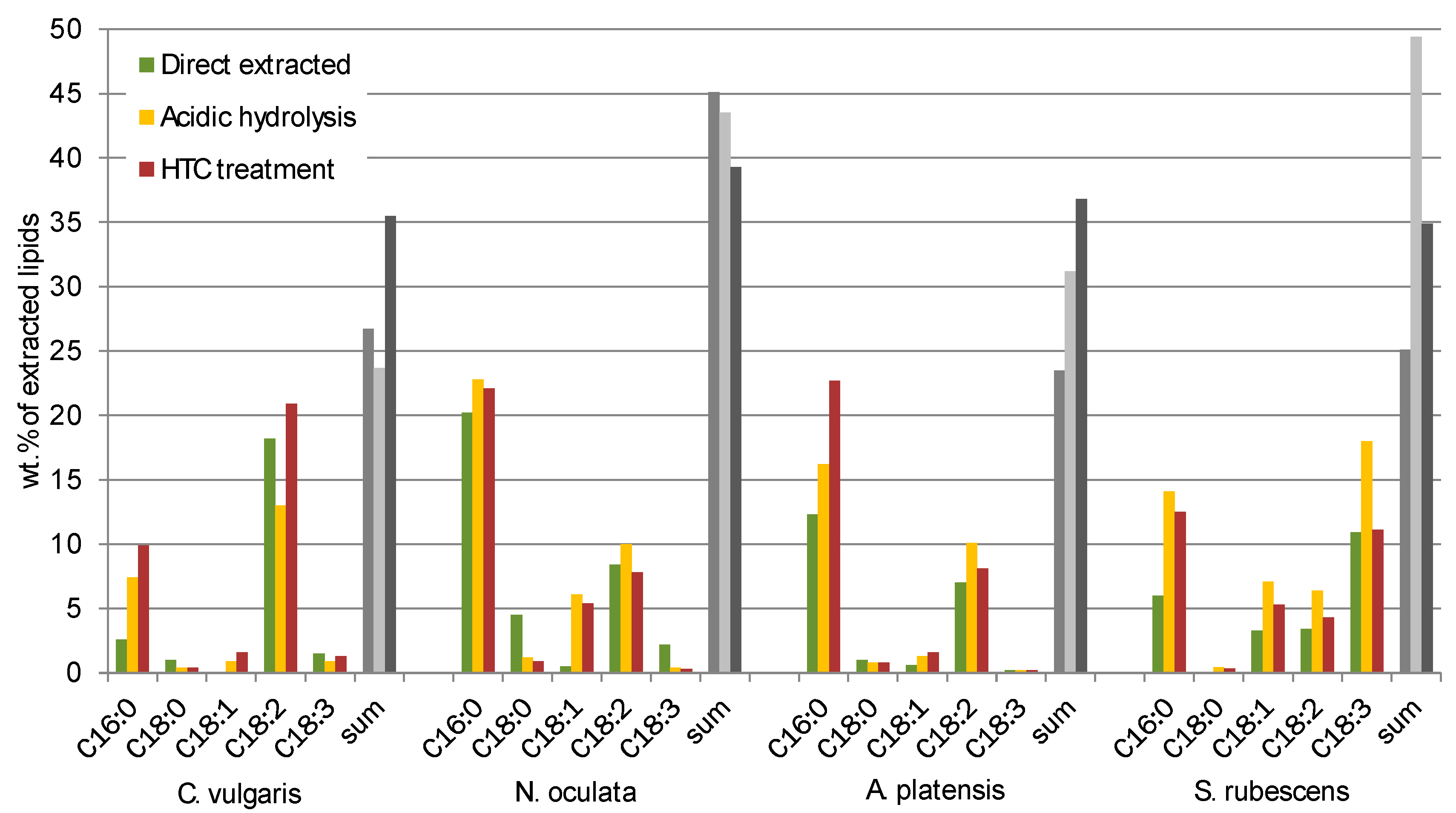
A correlation with the fatty acid distribution analysed by GC-MS could not be found but the low sums of recognized fatty acids (between 25 wt % and 45 wt %, see Figure 6) indicate that there are a lot of extracted substances which might influence the elemental composition.
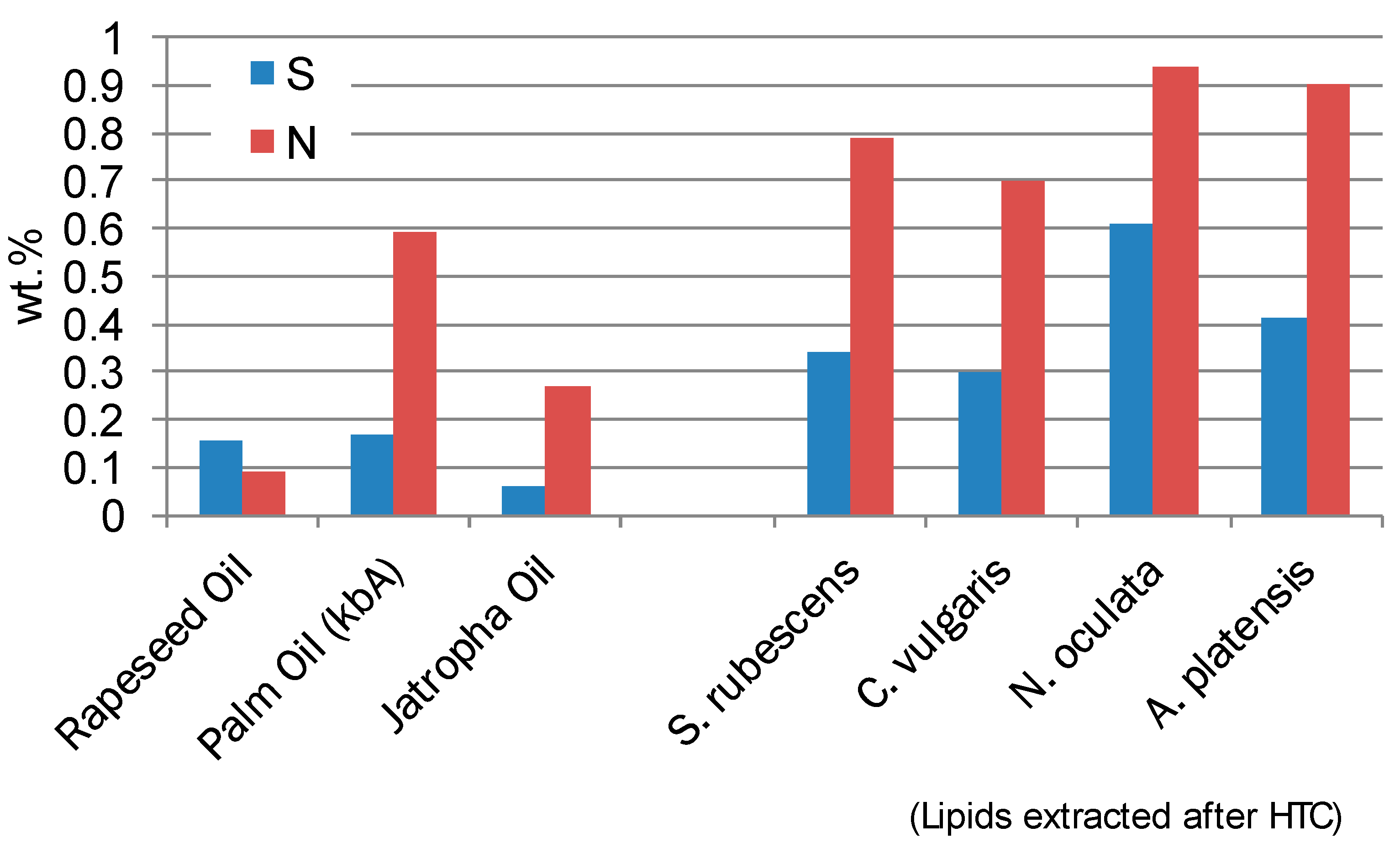
The content of Sulphur and Nitrogen is always higher in comparison to other vegetable oils (
Figure 4). This is due to the relative high Nitrogen and Sulphur content of the microalgae and the incorporation of these elements into lipid structures of the cell walls.
The heating value of all extracted lipids (as far as it was measured) varied in a small range from 37.6 to 39.0 MJ/kg, which is in accordance with standard values for vegetable oils.
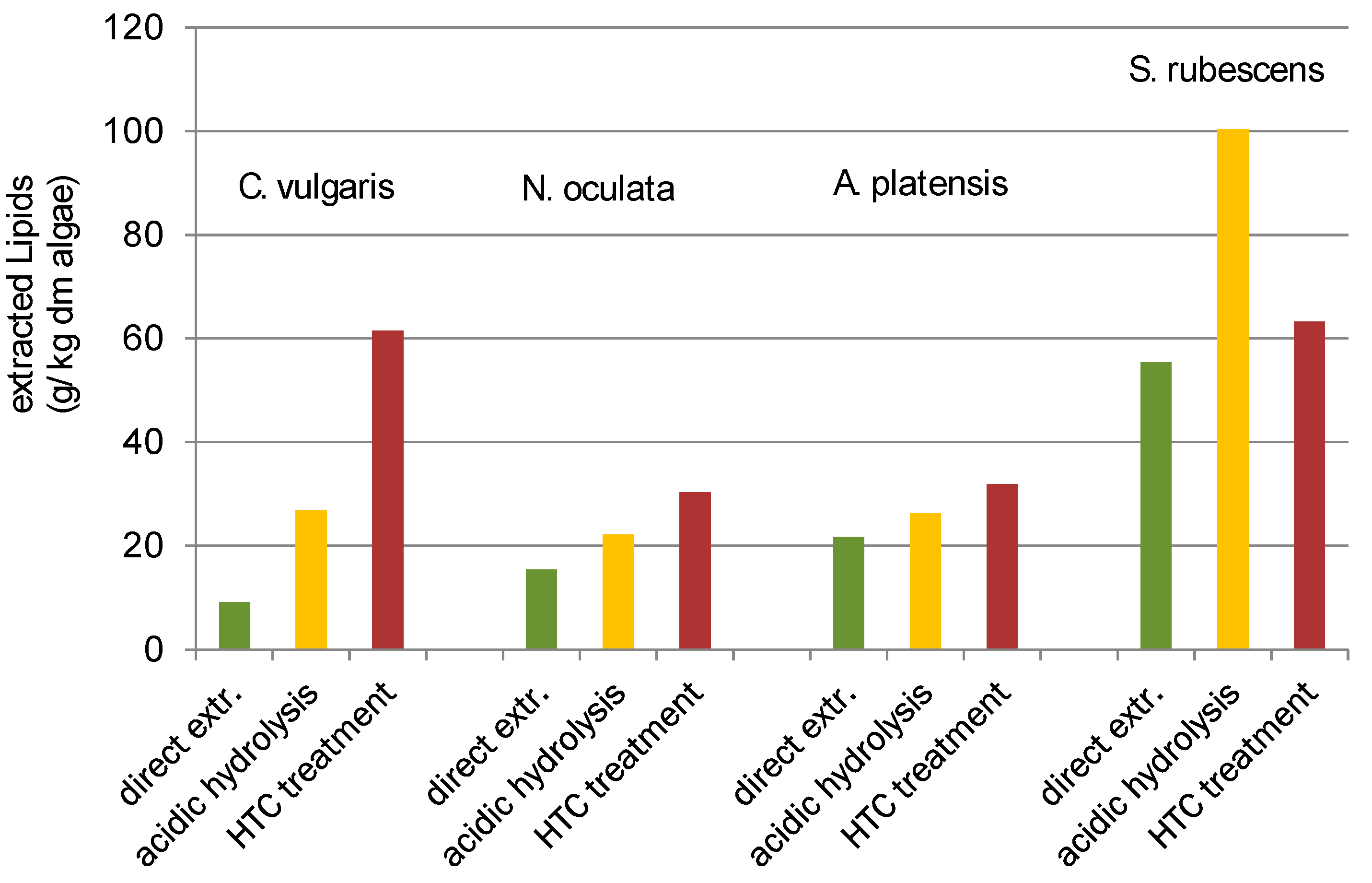
Besides the higher HTC char yield of
S. rubescens and
C. vulgaris (
Table 1), and the proportionate variation of the lipid yield according to the treatment procedures (direct, acidic hydrolysis, HTC treatment did change (
Figure 5).
For
C. vulgaris,
N. oculata and
A. platensis the yield changes between the different pre-treatment methods is the same (
Figure 5). Direct extraction gives the lowest yield, acidic hydrolysis improves the yield and the HTC pre-treatment again improves the yield in comparison to acidic hydrolysis. For
C. vulgaris the yield was more than six times higher with HTC then direct extracted. For
N. oculata the yield doubled and for
A. platensis it increased for 50%.
S. rubescens did show a different behaviour then the other species. Acidic hydrolysis gave the highest result for
S. rubescens. The yield from the HTC treatment did slightly improve the yield of the direct extraction (~14%) but the yield of acidic hydrolysis did nearly double the yield.
Fatty acids (FA) ranging from C14 to C22 contained in the extracted lipids were measured by GC-MS. In
Figure 6 the main FA fractions (C16:0, C18:0, C18:1, C18:3) of the different extracts of
C. vulgaris,
N. oculata,
A. platensis and
S. rubescens are displayed.
Besides the differences in the total quantity (sum) of recognized fatty acids (from 25 wt % to 45 wt %) the comparison of the different algae showed great differences in the FA profiles. Nonetheless the qualitative composition of the fatty acids is according to the one usually found in the Chlorella strain [
22]. Also for
N. oculata [
23] and
A. platensis [
24] the composition of fatty acids resembles the known composition. The FA profile of the
S. rubescens samples is in accordance with FA profiles published in literature [
25].
Regarding the differences of the FA profiles of the different disintegration processes, for C. vulgaris and A. platensis the FA C16:0 has a higher selectivity when utilizing acidic hydrolysis and HTC treatment but for the rest of the FA it is hard to find a correlation. For N. oculata the yield of FA C18:0 is reduced for acidic hydrolysis and HTC in comparison to direct extraction, meanwhile the yield of FA C18:1 increased.
3. Discussion
The analysis of the different lipid yields shows that there are differences between the used species. These differences originate from the type of biomass. The degree of disruption of the cell wall has a great influence on the yield of the extraction. Therefore, the differences in the behaviour of the used species under different pre-treatments need to originate from the morphology of the microalgae. The cell wall of microalgae can be composed of many different molecules. The structural components can (or may not) include crystalline or amorphous cellulose, hemicellulose, laminarin, sulphated polysaccharides (agar, carrageenan, alginate, fucoidan or ulvan), glycoproteins, CaCO
3, (silica) [
26]. In the literature, several experiments found a correlation of degree of disruption and cell wall composition.
In [
27]
Scenedesmus sp. were thermally treated at 70 °C and 90 °C for up to one hour (at no pressure). Anaerobic digestion tests showed, that the methane yield doubled for 90 °C treated algae. In Reference [
26], for
Chlorella sp. and
Nannochloropsis sp., the cell walls kept intact when hydrothermally treated at 120 °C for 30 min. The treatment also had no effect on
T. weissflogi, which has a silica based cell wall. In contrast to that, the species
Pavlova_cf sp. and
Tetraselmis sp. were solubilized into lower molecular weight components. These species have a glycoprotein based cell wall. This shows that even at much lower temperatures then used for this work, a rupture of the cell wall can be achieved when it is of the “right” components.
In Reference [
28], the three microalgal species
Scenedesmus obliquus,
Chlorella sorokiniana and
Arthrospira maxima were tested at five temperatures (105 °C, 120 °C, 145 °C, 155 °C, 165 °C) with two kinds of processes ((hydro-)thermal degradation and (hydro-)thermal hydrolysis with steam injection). It was concluded that the temperature was identified as the main driver of the cell wall breakage [
28]. It was also concluded that, for single cell algae that have carbohydrates polymers/matrix and acetolysis resistance biopolymers (algaenan) included in their cell walls (
Scenedesmus obliquus,
Chlorella sorokiniana), the rapid change of temperature/pressure caused by steam injection was only effective at pressures and temperatures higher than 4 bar and 150 °C respectively, while for cellulose free filamentous algae (
Arthrospira maxima), lower temperature/pressure combinations were sufficient to produce cell damage [
28]. This is generally in accordance with the results of this work, as
S. rubescens,
C. vulgaris and
N. oculata (which all have an algaenan layer) have a higher increase of the lipid yield from direct to pre-treated extraction in contrast to
A. platensis, which only has a small increase as the cell wall is already ruptured by freeze drying.
In Reference [
29], for the hydrothermal treatment (HTT) of
Desmodesmus sp. from 175 °C to around 250 °C and short reaction time, hydrolysis into smaller fragments (soluble in water) dominated, as indicated by the increase in water-soluble organics and the low oil yields obtained. In the 250 °C to 375 °C range, oil production—biocrude—increased and reached a maximum, while the organic content in the water decreased. After the hydrothermal treatment, the algae liquids and solids were extracted with DCM (Dichloromethane). The quality of the extract did change over the temperature range of hydrothermal treatment [
29]. This is also in accordance with the results of this work. The elemental analysis and also the GC-MS analysis of the extracted lipids showed that the quality of the extracts changes with the utilized pre-treatment. Reasons for this can be on the one hand side the different kind of damage to the cell wall, which leads to different extractable membrane lipids, whereas other lipids keep connected to the debris of the cell wall. On the other hand, there might already be chemical alterations of the lipids, changing the FA profile, induced by the different pre-treatment methods. In [
29] it is noted, that at elevated temperature not only more and different lipids are extracted due to a cell wall breakage but also the lipids are already degraded, esterified and hydrolysed because of the increased temperature.
It is remarkable that also in the conducted experiments of [
29] there was a high increase in oil yield between 225 °C and 250 °C. It was anticipated that this could be related to a change in morphology of the algal cells, considering that intracellular compounds are more accessible when the structure of the cell wall is broken. This was confirmed by a visual inspection of the cell walls. After a hydrothermal treatment at 225 °C the cell wall still was visually unbroken but at a temperature of 250 °C and higher no individual cells were recognizable and only a compact mass of cell debris was visible. The authors conclude that this thermal resistance could be related to the presence of biomarcomolecules named algaenans.
For
C. vulgaris and
A. platensis in a continuous reactor [
30] it was also shown that at 250 °C with 3 min residence time the structure of Chlorella cell wall was still given but to a great extent not functional anymore. At temperatures of 275 °C and 300 °C the structure of the cell wall itself was destroyed.
All these results lead to the conclusion that the compounds of the cell wall play a major role for the disintegration of it. As the focus of this work was not the biology of microalgae, there were no examinations of the cell wall of the microalgae used. Also in literature only little information is available for most species [
31], the determination is complicated and literature shows that over the last 30 years assumptions on what strain or class specific microalgae cell walls are composed of, had to be revised sometimes [
32]. Nonetheless, there can be found class and strain specific definitions of algal cell wall structures. In
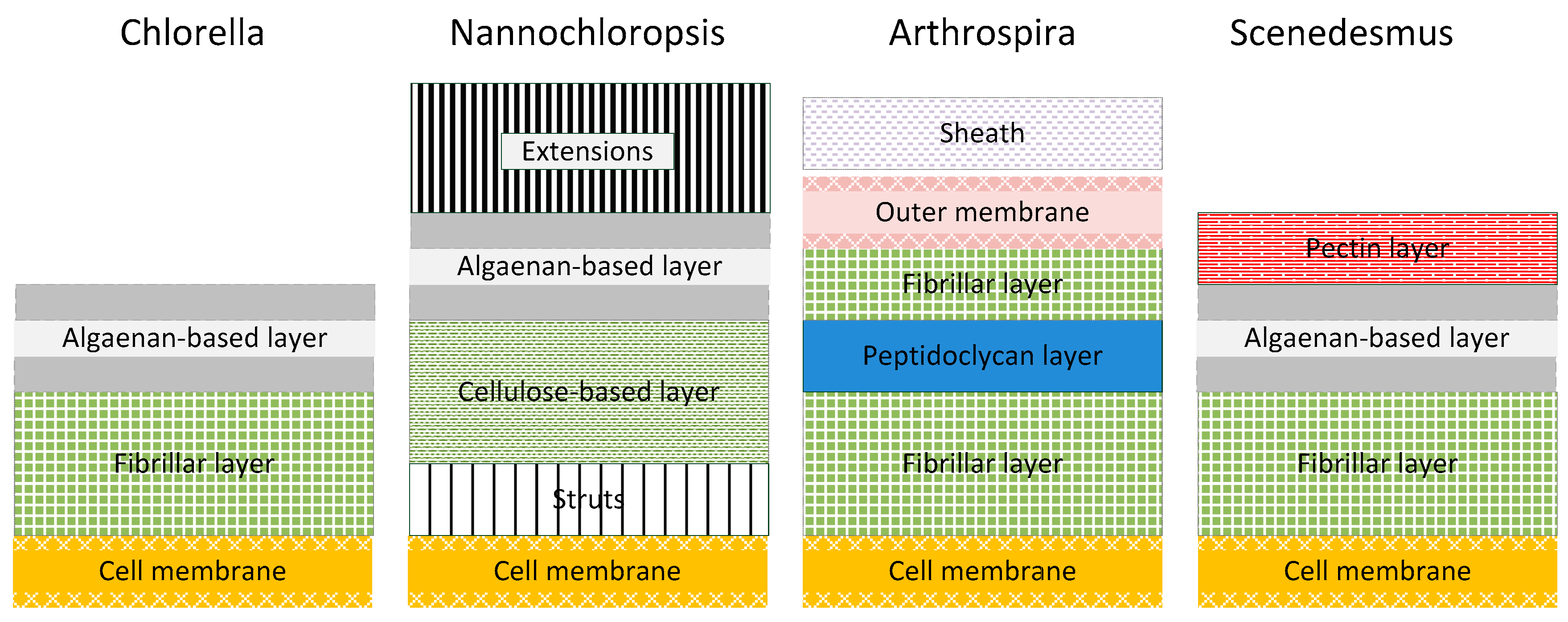
Figure 7 the cell wall structure of the four strains according to the examined species is schematically described (modified from [
33]).
In general, the inner cell wall layer is composed of a rigid microfibrillar structure embedded into a continuous matrix. This layer has a high cellulose content and chitin-like glycan is a predominant amino sugar in the cell wall. Other components embedded within the cell wall matrix are composed of uronic acids, rhamnose, arabinose, fucose, xylose, mannose, galactose, glucose and pectin. The outer cell wall of different species may include a trilaminar algaenan or form a thin homogeneous monolayer [
33].
Chlorella cell walls are generally known for their stability. They have a cell wall composed of fibrillar polysaccharides (cellulose/hemicellulose/pectin) and glucosamine (Chitin; Cellulose and chitin are very similar: cellulose is poly-ß(1,4)-
d-glucose and chitin is its 2-acetamido derivative [
32].
Nannochloropsis species possess a thick and multilayered cell wall composed of polysaccharides and also has non-hydrolysable biopolymers—algaenan-included [
34]. The inner layer of the cell wall is porous with a slender fibrous substructure and struts connecting the cellulose-based layer to the cell membrane. This layer is primarily composed of cellulose and glucose; amino acids represent an integral cell wall constituent. Small amounts of other sugars (rhamnose, mannose, ribose, xylose, fucose and galactose) are present. Algaenan layers comprise a thin trilaminar sheath in the cell periphery [
33].
Arthrospira cell walls have four layers. The peptidoglycan layer—also known as murein—provides rigidity and is located between two fibrillary layers. The outer membrane is tightly connected with the peptidoglycan layer and is covered with a sheath of acidic polysaccharides [
33].
Scenedesmus species generally have a rigid cell wall constituted of glucose, mannose and galactose, [
35]. It consists of three layers: an inner cellulosic layer delimiting individual cells, a thin middle algaenan-based layer and an outer pectic layer joining the cells into coenobium. Based on scanning microscopy, pectic layers consist of a hexagonal network of electron-dense material on the surface and a system of tubules radiating out from the middle layer [
33].
Three of the four investigated species therefore have an algaenan layer. Many microalgae of the chlorophyte division (green algae) have this layer of biomacromolecules incorporated in their cell wall. There is still discussion upon the exact structure and carbon chain length of algaenan [
36] but it is clear that algaenan is a recalcitrant substance that is insoluble and non-hydrolysable. It is supposed to be the part of the aquatic biomass which is preserved in sediments and converted into fossil oil as it matures over geological time frames [
37]. Algaenan consists of highly resistant aliphatic polymers [
31] and therefore a high effort is required for cell wall disruption. In Reference [
38], a very low conversion by means of temperatures between 200 °C and 300 °C (for 9 h) of algaenan isolated from
Botryococcus braunii was reported. In [
37] a
Scenedesmus species and also its extracted algaenan were hydrothermally treated. They showed that the algaenan polymethylenic structure can resist cracking at temperatures up to 310 °C. Therefore, algaenan seems to have a particular resistance to temperature. Although these biomolecules all are named algaenan, they can be of several different structures. For
Nannochloropsis and others they are comprised of linear C22 to C34 even numbered carbon chains with functional groups cross-linking the monomers with ether and ester bonds. For a specific
Scenedesmus species (
S. communis) it is assumed to consist of very long-chain (up to C120) monomeric (di)carboxylic acids [
33]. This makes
Scenedesmus special in comparison to the other strains with algaenan incorporated.
In general, its existence and its composition seem to be the explanation for the different extraction yields of the different algae species at different pre-treatments. The microalgae with no algaenan (Arthrospira) showed a good extraction yield already with no pre-treatment and the increase of lipid yield after the different pre-treatments was lower than for the other species. The difference in the algaenan of Scenedesmus communis might be a hint towards the different extraction behaviour of S. rubescens in contrast to C. vulgaris and N. oculata.
As Scenedesmus also is the only strain which has a pectin layer on the outside, pectin as a part of the cell wall also was further investigated.
As pectin is a product for the food industry which is used for example as a gelling agent it is quite well examined as a raw material. Usually it is extracted from the remainders of citrus fruits or apples.
In an overview on pectin [
39] the following procedure for pectin extraction from citrus and apple remainders can be found: “
Independently of the raw material, however, the current manufacturing process is based on extraction via acid hydrolysis in hot water. Careful processing is needed with regard to both the pH and hydrolysis time, as polymeric degradation can otherwise rapidly take place. In detail, manufacturers use a dilute mineral acid (
HCI or HNO3 or H2SO4)
between 50 °C and 100 °C and at pH 2–3 for several hours to solubilize the protopectin.” If pectin is one of the cell wall components which makes the cell wall rigid, the acidic hydrolysis extraction process is a good explanation for the very good extraction results for acidic hydrolysed
S. rubescens in contrast to the hydrothermal treatment.
Concerning the hydrothermal treatment only a few papers are published. (On the influence of pectin on the stability of a cell wall nothing is known to the author). Pectin was hydrothermally treated in Reference [
40], the focus was on HTC to achieve nanoporous carbon. Raw pectin from citrus fruits was utilized as feed. It was heated to 230 °C for two hours. No more temperatures were tested. At this temperature, the original fibrous structure disappeared and smooth and quite regular clustered microspheres having a diameter around 7 µm were formed. Therefore, a pectin layer of a cell wall might have already been ruptured. In [
41] HTC also was used to produce nanoporous material from pectin. Although several temperatures were tested (200 °C, 400 °C, 600 °C) there was no assumption on its degradation. An acid-free micro-wave assisted hydrothermal extraction of pectin from mango peel waste was investigated in [
42]. It was compared to a conventional acid assisted extraction. The optimum yield of 11.63 wt % was achieved at 110 °C. Temperatures from 120 to 200 in 20° steps were also conducted. At 120 °C (10.37 wt %), 140 °C (11.22 wt %) and 160 °C (9.23 wt %) the yield was still quite high. But at 180 °C the yield dropped to 0.65 wt % (and 0.16 wt % at 200 °C). It is concluded that this is the point the degradation process of pectin becomes dominant and faster than the extraction.
Therefore, although S. rubescens was the species with the most differing behaviour and was the only one with a pectin layer, the investigation showed, that the thermal and acid stability of pectin is too low to have an influence on the rigidity of the cell wall.
4. Materials and Methods
4.1. Material
The species used in this study;
Scenedesmus rubescens (
S. rubescens),
Chlorella vulgaris (
C. vulgaris),
Nannochloropsis oculata (
N. oculata) and
Arthrospira platensis (
A. platensis, the platensis species was classified as genus Spirulina in the past. Now it is classified as
Arthrospira [
43]); were kindly provided by the IGV GmbH, Germany.
S. rubescens and
C. vulgaris are green microalgae which grow in freshwater.
N. oculata belongs to the class of Eustigmatophyceae, which have only chlorophyll a and can grow both in fresh and salt water.
A. platensis is a blue microalga or a cyanobacteria, which grows in saltwater. About 1 to 2 kg (dw) of each species biomass were obtained. The biomass was freeze dried after harvesting. This was conducted at the IGV GmbH. The freeze-dried biomass had a residual water content of approx. 6 wt %. Like this the microalgae can be stored at room temperature for several months.
4.2. Properties
The raw material was dried in a Laboratory Drying Oven at 105 °C for 24 h to determine the water content. The water content of the HTC product was determined by headspace-Karl-Fisher-Titration (Aqua 40.0, Elektrochemie Halle GmbH, Halle, Germany) based on DIN EN 14774-1. Carbon, nitrogen, hydrogen and sulphur content were measured according to standard methods (DIN EN 15104) using an Elementar Vario Macro Cube (Elementar Analysensysteme GmbH, Hanau, Germany). C, H, N and S were reported in weight percentage on a dry basis and higher heating value (HHV) in kJ/kg on dry basis. Determination of the HHV was done by CEN method (DIN EN 14918) using a Parr oxygen bomb calorimeter 6400 (Parr Instrument (Frankfurt, Germany)). The ash content was determined by heating the dried material in a muffle furnace at 550 °C (DIN EN 14775).
4.3. Acidic Hydrolysis
For comparison and as an example for possible disintegration methods the microalgae were hydrolysed by hydrochloric acid prior to the solvent extraction. In accordance to DIN 10342:1992-09 approximately 18 g of the homogenized algae sample, 135 mL water and 65 mL hydrochloric acid (37%) were stirred thoroughly and heated slowly until boiling. Smooth boiling was maintained for 1 hour, followed by filtration and repeated rinsing of the residue and filter paper with distilled water. The filter residue was dried at 105 °C for 24 h and weighed, yielding 61% hydrolysed product.
4.4. Hydrothermal Treatment
The hydrothermal experiments were conducted in a 500 mL batch autoclave (Berghof, Highpreactor BR-500) with a data logger, a magnetic agitator and a BTC 3000 temperature controller. Temperature and pressure were recorded online. The system was stirred with 100 rpm to increase the heat transfer and avoid hot spots. Reaction temperature typically was 200 °C. Heating time was 90 min (2 K/min rate) Holding time was in general 120 min. Concentration of algae was 20 wt %, as the algae had a water content of approx. 5.5 to 7 wt % although being freeze dried, the corrected dry weight was considered when calculating the yields of HTC char and lipids. The cooling was carried out without external cooling over a period of several hours. After cooling, the resulting solid matter was separated by means of vacuum filtration and dried at 105 °C until a constant weight was attained. The gaseous phase was expanded via a gas sampling valve. The volume of the gaseous phase was measured. As the produced gas mainly consisted of CO
2 and the volume produced is low in general at these process conditions [
44] a further examination of the gaseous phase was neglected.
4.5. Extraction of Algae Oil
Extraction of the algae oil was done by soxhlet extraction of the algae sample (freeze dried or hydrolysed) or the HTC product. The solid was pulverized and homogenized in a porcelain mortar. The sample was transferred to a fat free extraction thimble and extracted continuously for 7 h. As extraction agent n-hexane was used, as hexane extraction is presently seen as the most economical method for algae extraction [
45].
4.6. Determination of Fatty Acid Profile by GC-MS
Analysis was performed on a 7890A gas chromatograph coupled with a mass-selective detector (5975C; Agilent Technologies, Santa Clara, CA, USA). The injection mode was split 1:15. A deactivated liner was inserted into the injection chamber, which was kept at 300 °C. 1 µL of the sample was injected for separation by an ionic liquid phase (1,12-Di(tripropylphosphonium)dodecane bis(trifluoromethylsulfonyl)imide; SPB-IL 60 30 m × 0.25 mm × 0.20 µm, Sigma Aldrich, Taufkirchen, Germany). The gas chromatography (GC) system was operated in programmed-temperature mode: initial temperature 160 °C, first linear ramp 1 °C min–1 until 165 °C, 15 min hold, second linear ramp 5 °C min–1 until 250 °C final temperature, 1 min hold. Data acquisition was performed on the mass-selective detector in scan mode (40–450 amu). Due to changes in laboratory workflow, some of the measurements were done on a Polyethylenglycole phase (HP-INNOWax, 30 m × 0.25 mm × 0.25 µm; Agilent Technologies) The injection mode was split 1:15, the deactivated liner was kept at a temperature of 260 °C. When using the HP-INNOWax-column the gas chromatography (GC) system was operated in programmed-temperature mode as well: initial temperature 150 °C, 0.5 min hold, linear ramp 4 °C min–1 until 260 °C final temperature, 7 min hold. Data acquisition was performed on the mass-selective detector in scan mode (40–380 amu). An external calibration was done for both of the capillary columns separately.
Samples for GC-MS determination were prepared as follows: 100 mg of the extracted algae oil was dissolved in 5 mL tert-butyl methyl ether (MTBE). 100 µL of the MTBE solution were transferred to a GC-Vial equipped with a 0.2 mL micro-insert. To this solution, 50 µL trimethylsulfonium hydroxide (TMSH) was added. The mixture was shaken shortly and measured immediately. The formed fatty acid methyl esters (FAME) were identified by mass spectral data (National Institute of Standards and Technology (NIST) 2008 Mass Spectral Library) and matching of retention times with FAME standard substances. Quantitative determination was carried out by serial dilution of Grain Fatty Acids Methyl Ester Mix (Sigma Aldrich, Taufkirchen, Germany) and applying an external linear calibration function.
All experiments were conducted only twice because of the limited amount of algae. It was not possible to obtain more microalgae of the same charge. More repetitions of the experiments by using smaller amounts for each run would have led to less products for analysing and less data. For all educts, intermediates and products, elemental composition, heating value and if possible ash content were analysed. The extracted lipid yields were measured and put in relation to the input material.