Optimisation of Second-Generation Biodiesel Production from Australian Native Stone Fruit Oil Using Response Surface Method
Abstract
1. Introduction
2. Literature Review
3. Materials and Methods
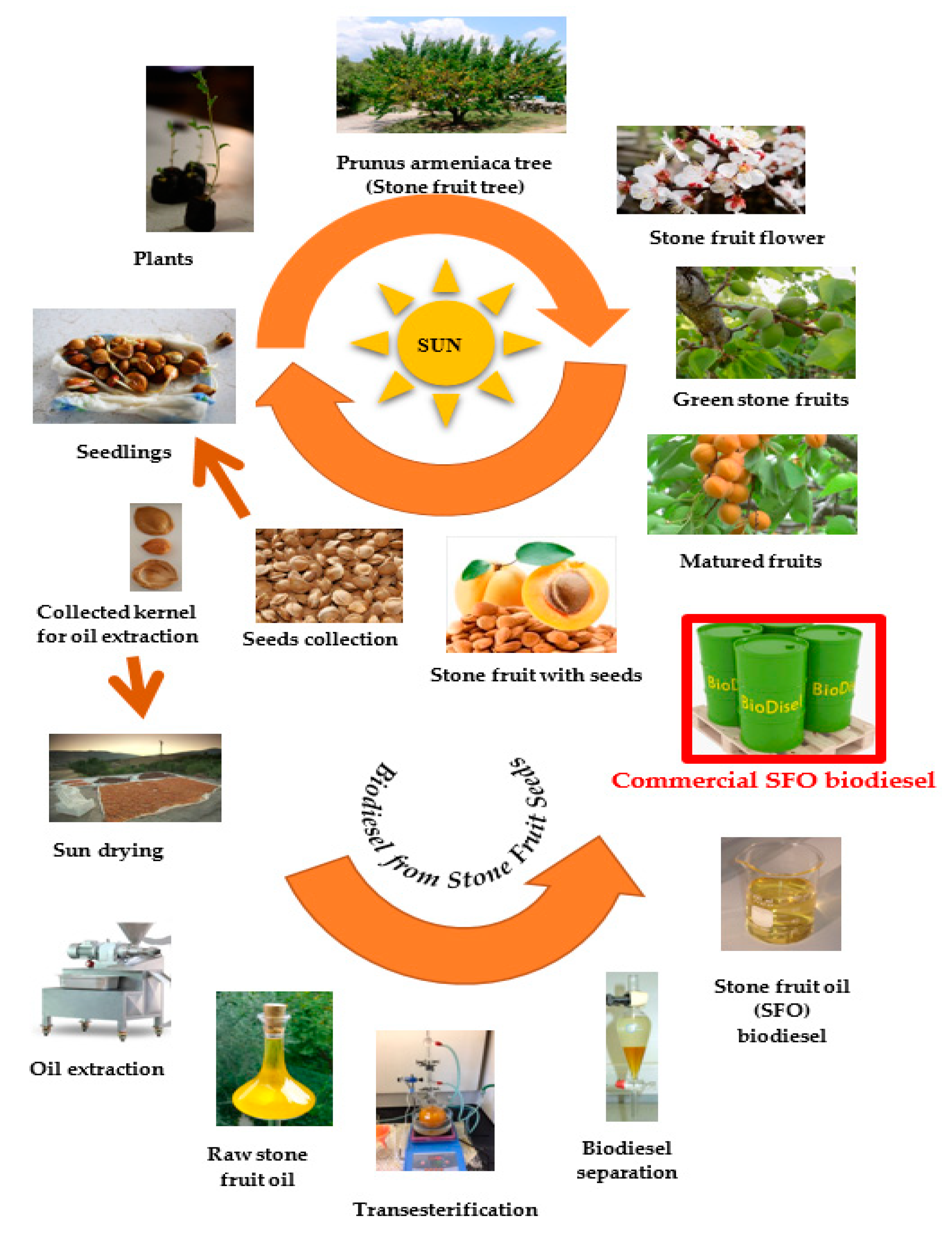
3.1. Materials
3.2. Oil Extraction
3.3. Biodiesel Production
3.4. Physicochemical Characterisation of SFO
3.4.1. Fuel Properties
3.4.2. Gas Chromatography
3.4.3. Fourier Transform Infrared (FTIR) Spectroscopy
3.5. Design of Experiments
4. Results and Discussion
4.1. Characterisation of Crude SFO
4.2. Properties and Qualities of SFO Biodiesel
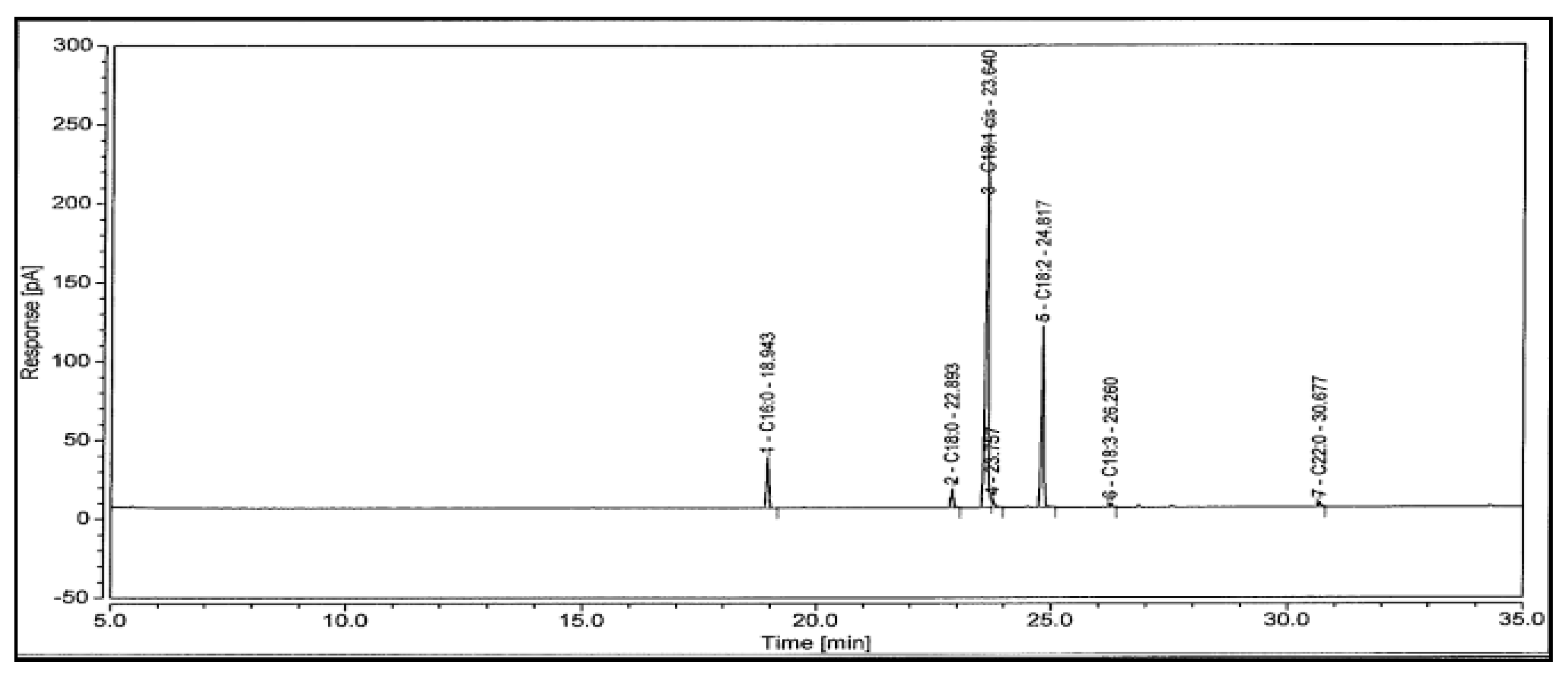
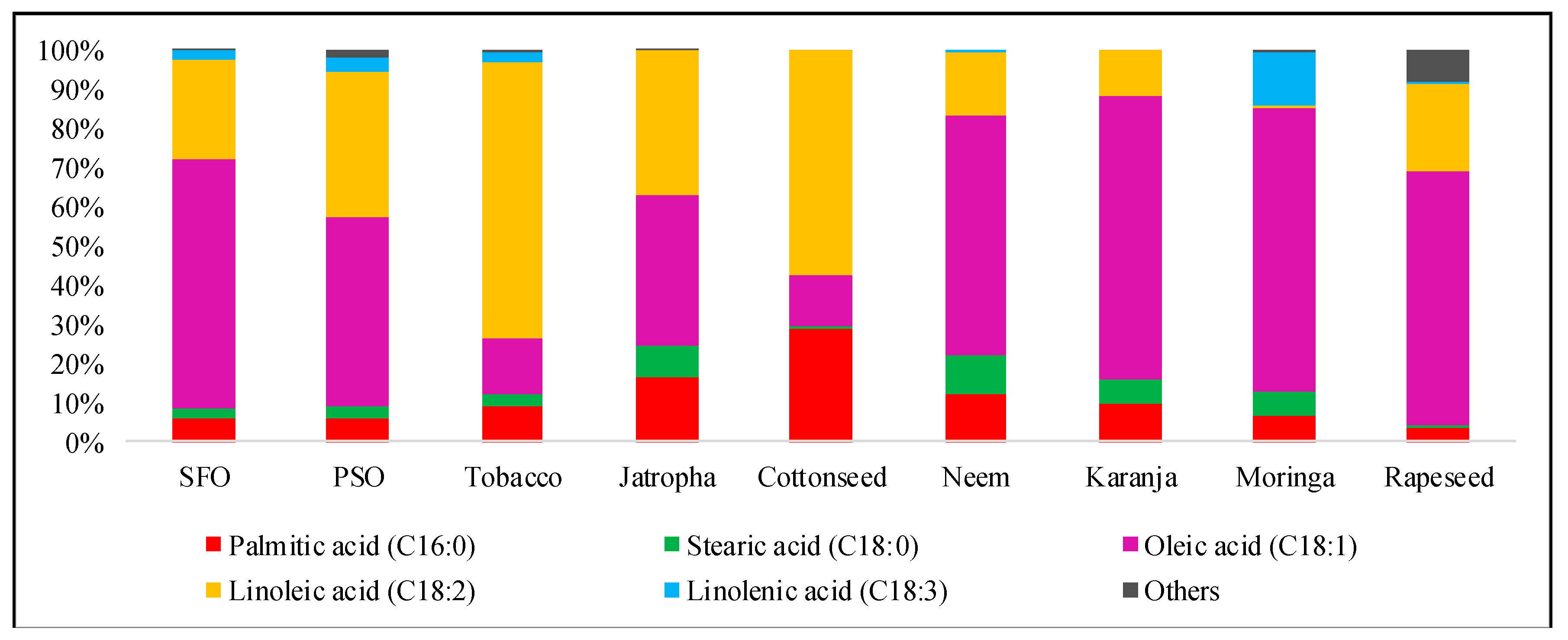
4.3. The Fatty Acid Composition of SFO Biodiesel
4.4. Optimisation of Reaction Conditions by RSM
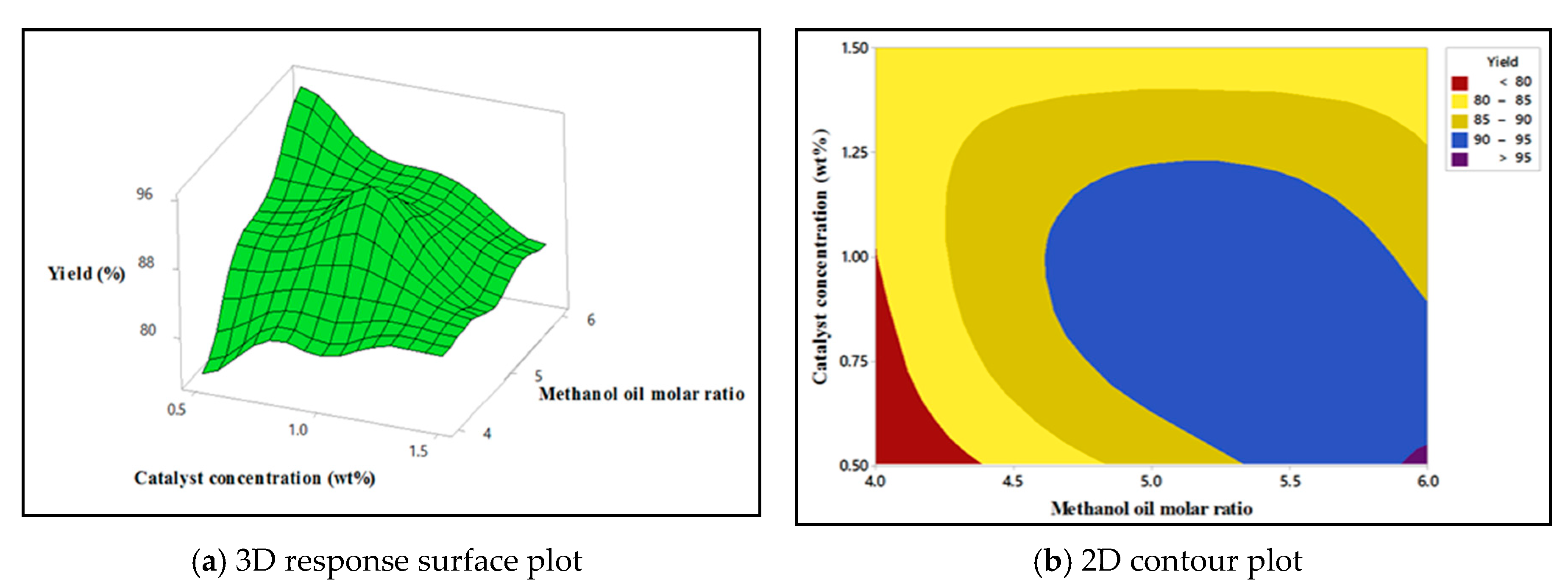
4.5. Response Surface Plots for SFO Biodiesel Production
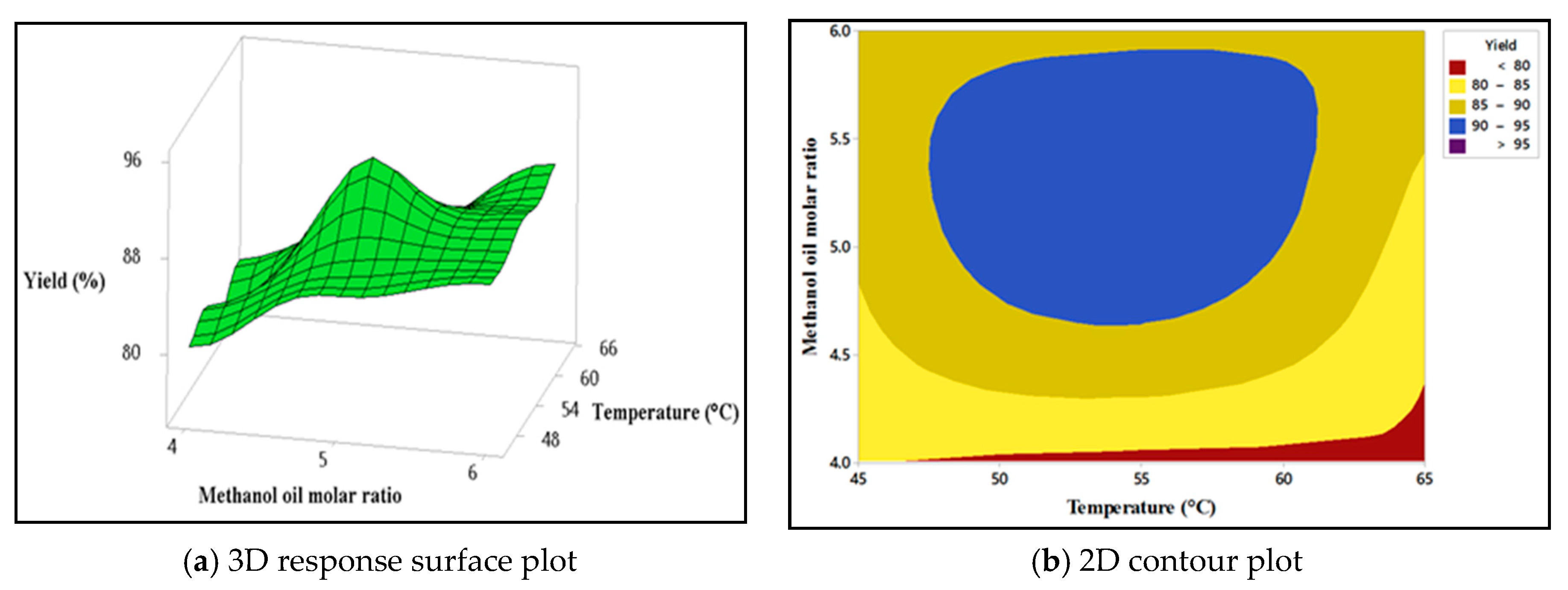
4.5.1. Interaction Effect of Methanol: Oil Molar Ratio and Catalyst Concentration
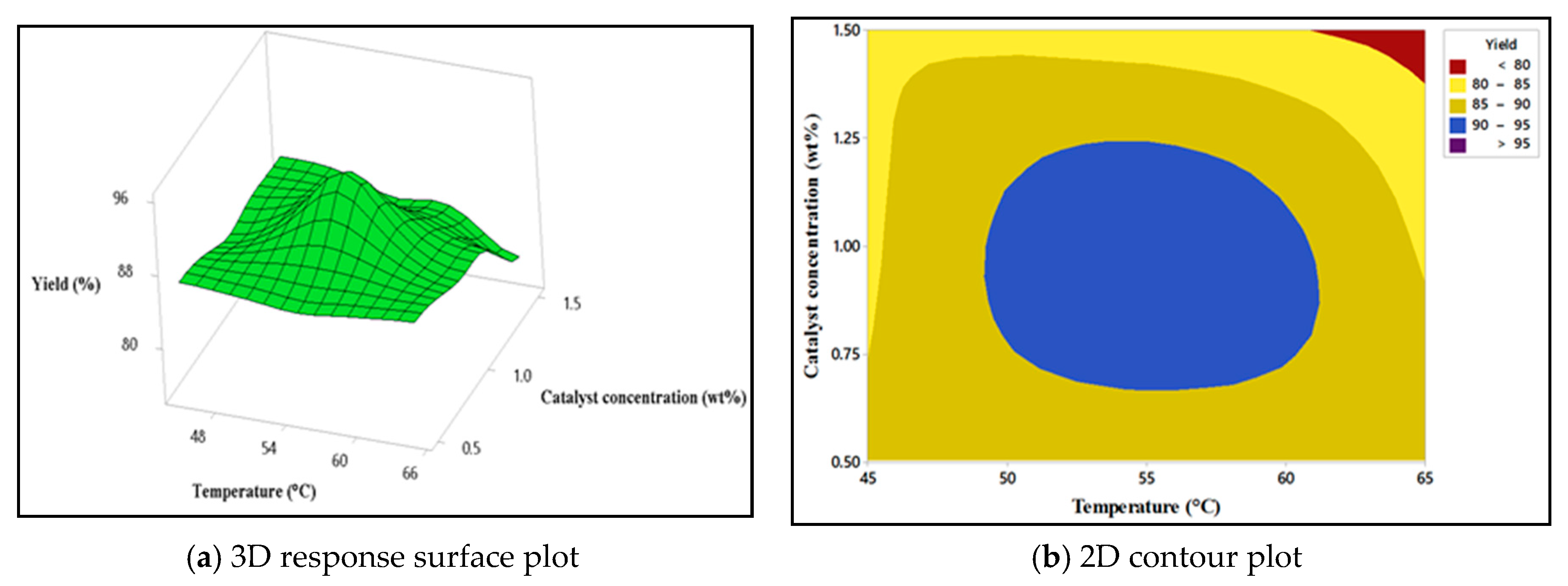
4.5.2. Interaction Effect of Catalyst Concentration and the Reaction Temperature
4.5.3. Interaction Effect of Reaction Temperature and Methanol: Oil Molar Ratio
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rahman, M.M.; Rasul, M.G.; Hassan, N.M.S.; Azad, A.K.; Uddin, M.N. Effect of small proportion of butanol additive on the performance, emission, and combustion of Australian native first-and second-generation biodiesel in a diesel engine. Environ. Sci. Pollut. Res. 2017, 24, 22402–22413. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.; Rasul, M.; Hassan, N. Study on the tribological characteristics of Australian native first generation and second generation biodiesel fuel. Energies 2017, 10, 55. [Google Scholar] [CrossRef]
- Bhuiya, M.M.K.; Rasul, M.G.; Khan, M.M.K.; Ashwath, N.; Azad, A.K.; Hazrat, M.A. Second generation biodiesel: Potential alternative to-edible oil-derived biodiesel. Energy Procedia 2014, 61, 1969–1972. [Google Scholar] [CrossRef]
- Bhuiya, M.M.K.; Rasul, M.G.; Khan, M.M.K.; Ashwath, N.; Azad, A.K.; Hazrat, M.A. Prospects of 2nd generation biodiesel as a sustainable fuel—Part 2: Properties, performance and emission characteristics. Renew. Sustain. Energy Rev. 2016, 55, 1129–1146. [Google Scholar]
- Popp, J.; Lakner, Z.; Harangi-Rákos, M.; Fári, M. The effect of bioenergy expansion: Food, energy, and environment. Renew. Sustain. Energy Rev. 2014, 32, 559–578. [Google Scholar] [CrossRef]
- NguyenThi, T.; Bazile, J.-P.; Bessières, D. Density measurements of waste cooking oil biodiesel and diesel blends over extended pressure and temperature ranges. Energies 2018, 11, 1212. [Google Scholar] [CrossRef]
- Yusop, A.; Mamat, R.; Yusaf, T.; Najafi, G.; Yasin, M.; Khathri, A. Analysis of particulate matter (pm) emissions in diesel engines using palm oil biodiesel blended with diesel fuel. Energies 2018, 11, 1039. [Google Scholar] [CrossRef]
- Ali, O.; Mamat, R.; Najafi, G.; Yusaf, T.; Safieddin Ardebili, S. Optimization of biodiesel-diesel blended fuel properties and engine performance with ether additive using statistical analysis and response surface methods. Energies 2015, 8, 14136–14150. [Google Scholar] [CrossRef]
- Silitonga, A.S.; Atabani, A.E.; Mahlia, T.M.I.; Masjuki, H.H.; Badruddin, I.A.; Mekhilef, S. A review on prospect of jatropha curcas for biodiesel in Indonesia. Renew. Sustain. Energy Rev. 2011, 15, 3733–3756. [Google Scholar] [CrossRef]
- Rahman, M.; Rasul, M.; Hassan, N.; Hyde, J. Prospects of biodiesel production from macadamia oil as an alternative fuel for diesel engines. Energies 2016, 9, 403. [Google Scholar]
- Sharma, R.; Gupta, A.; Abrol, G.S.; Joshi, V.K. Value addition of wild apricot fruits grown in north-west himalayan regions: A review. J. Food Sci. Technol. 2012, 51, 2917–2924. [Google Scholar] [CrossRef] [PubMed]
- Razack, S.A.; Duraiarasan, S. Response surface methodology assisted biodiesel production from waste cooking oil using encapsulated mixed enzyme. Waste Manag. 2016, 47, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Saydut, A.; Erdogan, S.; Kafadar, A.B.; Kaya, C.; Aydin, F.; Hamamci, C. Process optimization for production of biodiesel from hazelnut oil, sunflower oil and their hybrid feedstock. Fuel 2016, 183, 512–517. [Google Scholar] [CrossRef]
- Martínez, S.L.; Romero, R.; Natividad, R.; González, J. Optimization of biodiesel production from sunflower oil by transesterification using Na2O/NaX and methanol. Catal. Today 2014, 220, 12–20. [Google Scholar] [CrossRef]
- Kostić, M.D.; Djalović, I.G.; Stamenković, O.S.; Mitrović, P.M.; Adamović, D.S.; Kulina, M.K.; Veljković, V.B. Kinetic modeling and optimization of biodiesel production from white mustard (Sinapis alba L.) seed oil by quicklime-catalyzed transesterification. Fuel 2018, 223, 125–139. [Google Scholar] [CrossRef]
- Ong, H.C.; Masjuki, H.H.; Mahlia, T.M.I.; Silitonga, A.S.; Chong, W.T.; Leong, K.Y. Optimization of biodiesel production and engine performance from high free fatty acid Calophyllum inophyllum oil in Ci diesel engine. Energy Convers. Manag. 2014, 81, 30–40. [Google Scholar] [CrossRef]
- Dharma, S.; Masjuki, H.H.; Ong, H.C.; Sebayang, A.H.; Silitonga, A.S.; Kusumo, F.; Mahlia, T.M.I. Optimization of biodiesel production process for mixed Jatropha curcas–Ceiba pentandra biodiesel using response surface methodology. Energy Convers. Manag. 2016, 115, 178–190. [Google Scholar] [CrossRef]
- Gumus, M.; Kasifoglu, S. Performance and emission evaluation of a compression ignition engine using a biodiesel (apricot seed kernel oil methyl ester) and its blends with diesel fuel. Biomass Bioenergy 2010, 34, 134–139. [Google Scholar] [CrossRef]
- Fadhil, A.B. Evaluation of apricot (Prunus armeniaca L.) seed kernel as a potential feedstock for the production of liquid bio-fuels and activated carbons. Energy Convers. Manag. 2017, 133, 307–317. [Google Scholar] [CrossRef]
- Ullah, F.; Nosheen, A.; Hussain, I.; Bano, A. Base catalyzed transesterification of wild apricot kernel oil for biodiesel production. Afr. J. Biotechnol. 2009, 8, 3309–3313. [Google Scholar]
- Yadav, A.K.; Pal, A.; Dubey, A.M. Experimental studies on utilization of Prunus armeniaca L. (wild apricot) biodiesel as an alternative fuel for CI engine. Waste Biomass Valoriz. 2017, 1–9. [Google Scholar] [CrossRef]
- Hamze, H.; Akia, M.; Yazdani, F. Optimization of biodiesel production from the waste cooking oil using response surface methodology. Process Saf. Environ. Prot. 2015, 94, 1–10. [Google Scholar] [CrossRef]
- Atabani, A.E.; Silitonga, A.S.; Badruddin, I.A.; Mahlia, T.M.I.; Masjuki, H.H.; Mekhilef, S. A comprehensive review on biodiesel as an alternative energy resource and its characteristics. Renew. Sustain. Energy Rev. 2012, 16, 2070–2093. [Google Scholar] [CrossRef]
- Atadashi, I.M.; Aroua, M.K.; Abdul Aziz, A.R.; Sulaiman, N.M.N. The effects of water on biodiesel production and refining technologies: A review. Renew. Sustain. Energy Rev. 2012, 16, 3456–3470. [Google Scholar] [CrossRef]
- Banerjee, A.; Chakraborty, R. Parametric sensitivity in transesterification of waste cooking oil for biodiesel production—A review. Resour. Conserv. Recycl. 2009, 53, 490–497. [Google Scholar] [CrossRef]
- Yaakob, Z.; Mohammad, M.; Alherbawi, M.; Alam, Z.; Sopian, K. Overview of the production of biodiesel from waste cooking oil. Renew. Sustain. Energy Rev. 2013, 18, 184–193. [Google Scholar] [CrossRef]
- Targais, K.; Stobdan, T.; Yadav, A.; Singh, S.B. Extraction of apricot kernel oil in cold desert Ladakh, India. Indian J. Tradit. Knowl. 2011, 10, 304–306. [Google Scholar]
- Wang, L. Properties of manchurian apricot (Prunus mandshurica Skv.) and siberian apricot (Prunus sibirica L.) seed kernel oils and evaluation as biodiesel feedstocks. Ind. Crops Prod. 2013, 50, 838–843. [Google Scholar] [CrossRef]
- Fan, S.; Liang, T.; Yu, H.; Bi, Q.; Li, G.; Wang, L. Kernel characteristics, oil contents, fatty acid compositions and biodiesel properties in developing siberian apricot (Prunus sibirica L.) seeds. Ind. Crops Prod. 2016, 89, 195–199. [Google Scholar] [CrossRef]
- Rahman, M.M.; Hassan, M.H.; Kalam, M.A.; Atabani, A.E.; Memon, L.A.; Rahman, S.M.A. Performance and emission analysis of Jatropha curcas and Moringa oleifera methyl ester fuel blends in a multi-cylinder diesel engine. J. Clean. Prod. 2014, 65, 304–310. [Google Scholar] [CrossRef]
- Kafuku, G.; Mbarawa, M. Alkaline catalyzed biodiesel production from moringa oleifera oil with optimized production parameters. Appl. Energy 2010, 87, 2561–2565. [Google Scholar] [CrossRef]
- Alptekin, E.; Canakci, M. Determination of the density and the viscosities of biodiesel–diesel fuel blends. Renew. Energy 2008, 33, 2623–2630. [Google Scholar] [CrossRef]
- Encinar, J.M.; González, J.F.; Rodríguez-Reinares, A. Biodiesel from used frying oil. Variables affecting the yields and characteristics of the biodiesel. Ind. Eng. Chem. Res. 2005, 44, 5491–5499. [Google Scholar] [CrossRef]
- Graboski, M.S.; McCormick, R.L. Combustion of fat and vegetable oil derived fuels in diesel engines. Prog. Energy Combust. Sci. 1998, 24, 125–164. [Google Scholar] [CrossRef]
- Silitonga, A.S.; Ong, H.C.; Mahlia, T.M.I.; Masjuki, H.H.; Chong, W.T. Biodiesel conversion from high FFA crude Jatropha curcas, Calophyllum inophyllum and Ceiba pentandra oil. Energy Procedia 2014, 61, 480–483. [Google Scholar] [CrossRef]
- Silitonga, A.S.; Ong, H.C.; Masjuki, H.H.; Mahlia, T.M.I.; Chong, W.T.; Yusaf, T.F. Production of biodiesel from Sterculia foetida and its process optimization. Fuel 2013, 111, 478–484. [Google Scholar] [CrossRef]
- Keera, S.T.; El Sabagh, S.M.; Taman, A.R. Transesterification of vegetable oil to biodiesel fuel using alkaline catalyst. Fuel 2011, 90, 42–47. [Google Scholar] [CrossRef]
- Hasni, K.; Ilham, Z.; Dharma, S.; Varman, M. Optimization of biodiesel production from Brucea javanica seeds oil as novel non-edible feedstock using response surface methodology. Energy Convers. Manag. 2017, 149, 392–400. [Google Scholar] [CrossRef]
- Anwar, M.; Rasul, M.G.; Ashwath, N. Production optimization and quality assessment of papaya (Carica papaya) biodiesel with response surface methodology. Energy Convers. Manag. 2018, 156, 103–112. [Google Scholar] [CrossRef]
- Benjumea, P.; Agudelo, J.R.; Agudelo, A.F. Effect of the degree of unsaturation of biodiesel fuels on engine performance, combustion characteristics, and emissions. Energy Fuels 2011, 25, 77–85. [Google Scholar] [CrossRef]
- Altun, Ş. Effect of the degree of unsaturation of biodiesel fuels on the exhaust emissions of a diesel power generator. Fuel 2014, 117, 450–457. [Google Scholar] [CrossRef]
- Wang, L.; Yu, H. Biodiesel from siberian apricot (Prunus sibirica L.) seed kernel oil. Bioresour. Technol. 2012, 112, 355–358. [Google Scholar] [CrossRef] [PubMed]
- Kate, A.E.; Lohani, U.C.; Pandey, J.P.; Shahi, N.C.; Sarkar, A. Traditional and mechanical method of the oil extraction from wild apricot kernel: A comparative study. Res. J. Chem. Environ. Sci. 2014, 2, 54–60. [Google Scholar]
- Atabani, A.E.; César, A.D.S. Calophyllum inophyllum L.—A prospective non-edible biodiesel feedstock. Study of biodiesel production, properties, fatty acid composition, blending and engine performance. Renew. Sustain. Energy Rev. 2014, 37, 644–655. [Google Scholar] [CrossRef]
- García-Martínez, N.; Andreo-Martínez, P.; Quesada-Medina, J.; de los Ríos, A.P.; Chica, A.; Beneito-Ruiz, R.; Carratalá-Abril, J. Optimization of non-catalytic transesterification of tobacco (Nicotiana tabacum) seed oil using supercritical methanol to biodiesel production. Energy Convers. Manag. 2017, 131, 99–108. [Google Scholar] [CrossRef]
- Goyal, P.; Sharma, M.P.; Jain, S. Optimization of conversion of high free fatty acid Jatropha curcas oil to biodiesel using response surface methodology. ISRN Chem. Eng. 2012, 2012, 8. [Google Scholar] [CrossRef]
- Lin, L.; Zhou, C.; Vittayapadung, S.; Shen, X.; Dong, M. Opportunities and challenges for biodiesel fuel. Appl. Energy 2011, 88, 1020–1031. [Google Scholar] [CrossRef]
- Demirbaş, A. Biodiesel fuels from vegetable oils via catalytic and non-catalytic supercritical alcohol transesterifications and other methods: A survey. Energy Convers. Manag. 2003, 44, 2093–2109. [Google Scholar] [CrossRef]
- Shankar, V.; Jambulingam, R. Waste crab shell derived CaO impregnated Na-ZSM-5 as a solid base catalyst for the transesterification of neem oil into biodiesel. Sustain. Environ. Res. 2017, 27, 273–278. [Google Scholar] [CrossRef]
- Patel, R.L.; Sankhavara, C.D. Biodiesel production from karanja oil and its use in diesel engine: A review. Renew. Sustain. Energy Rev. 2017, 71, 464–474. [Google Scholar] [CrossRef]
- Rashid, U.; Anwar, F.; Moser, B.R.; Knothe, G. Moringa oleifera oil: A possible source of biodiesel. Bioresour. Technol. 2008, 99, 8175–8179. [Google Scholar] [CrossRef] [PubMed]
- Silitonga, A.S.; Masjuki, H.H.; Mahlia, T.M.I.; Ong, H.C.; Chong, W.T.; Boosroh, M.H. Overview properties of biodiesel diesel blends from edible and non-edible feedstock. Renew. Sustain. Energy Rev. 2013, 22, 346–360. [Google Scholar] [CrossRef]
- Gupta, J.; Agarwal, M.; Dalai, A.K. Optimization of biodiesel production from mixture of edible and nonedible vegetable oils. Biocatal. Agric. Biotechnol. 2016, 8, 112–120. [Google Scholar] [CrossRef]
- Silva, G.F.; Camargo, F.L.; Ferreira, A.L.O. Application of response surface methodology for optimization of biodiesel production by transesterification of soybean oil with ethanol. Fuel Process. Technol. 2011, 92, 407–413. [Google Scholar] [CrossRef]
- Vicente, G.; Martínez, M.; Aracil, J. Optimisation of integrated biodiesel production. Part I. A study of the biodiesel purity and yield. Bioresour. Technol. 2007, 98, 1724–1733. [Google Scholar] [CrossRef] [PubMed]


| Factors/Variables | Unit | Symbol Coded | Range and Levels | ||
|---|---|---|---|---|---|
| −1 | 0 | +1 | |||
| Methanol: Oil ratio | mol/mol | M | 4:1 | 5:1 | 6:1 |
| KOH catalyst concentration | wt % | C | 0.5 | 1.00 | 1.5 |
| Temperature | °C | T | 45 | 55 | 65 |
| Properties | Units | SFO This Study | SFO [18] | SFO [21] | SFO [11] | Petro Diesel |
|---|---|---|---|---|---|---|
| Kinematic Viscosity @ 40 °C | m2/s | 34.54 | 34.82 | 20.53 | 26.22 | 3.23 |
| Density | kg/m3 | 910 | 920 | 913 | 916.6 | 827.2 |
| Specific Gravity @ 15 °C | g/cm3 | 0.91 | 0.91 | 0.91 | 0.91 | 0.83 |
| Acid value | mg KOH/g | 1.65 | 2.60 | 0.68 | 0.05 | |
| Calorific value | MJ/kg | 38.4 | 39.6 | 31.5 | 45.3 | |
| Saponification number | mg KOH/g | 173 | 188 | 187 | ||
| Iodine value | mgI2/100 g | 103 | 90 | 101 |
| Non-Edible Biodiesels | Density (kg/m3) | Viscosity at 40 °C, mm2/s | Acid Value, mg KOH/g | Cetane Number (CN) | Calorific Value, MJ/Kg | Flash Point, °C | Iodine Value (IV) mgl2/100 g | Oxidation Stability (OS), h |
|---|---|---|---|---|---|---|---|---|
| SFO this study | 855.0 | 4.26 | 0.25 | 50.45 | 39.64 | 105 | 104.70 | 7.15 |
| SFO [18] | 884.3 | 4.92 | 39.95 | 111 | ||||
| SFO [21] | 857.0 | 5.20 | 0.32 | 58.70 | 38.93 | 180 | 100.70 | 6.30 |
| SFO [11] | 879.4 | 4.21 | 0.08 | 39.12 | 170 | 100.66 | ||
| Petro diesel [39] | 827.2 | 3.23 | 0.05 | 48.00 | 45.30 | 68.5 | 38.3 | 39.0 |
| Tobacco [23,38] | 888.5 | 4.23 | 51.60 | 165.4 | 136 | 0.80 | ||
| PSO [39] | 840.0 | 3.53 | 0.42 | 48.29 | 38.49 | 112 | 115.89 | 5.61 |
| Jatropha [38] | 879.5 | 4.80 | 0.40 | 51.60 | 39.23 | 135 | 104 | 2.30 |
| Rapeseed [23] | 882.0 | 4.43 | 54.40 | 37.00 | 170 | 7.60 | ||
| Cottonseed [23] | 875.0 | 4.07 | 0.16 | 54.13 | 40.43 | 150 | 1.83 | |
| Neem [23,38] | 868.0 | 5.21 | 0.65 | 39.81 | 76 | 7.10 | ||
| Karanja [38] | 931.0 | 6.13 | 0.42 | 55.00 | 43.42 | 95 | ||
| Moringa [23] | 883.0 | 5.00 | 0.18 | 67.1 | 160 | 74 | 2.3 | |
| ASTM D6751 | 880.0 | 1.9~6.0 | maximum 0.5 | minimum 47 | 93~170 | minimum 3 | ||
| EN14214 | 860~900 | 3.5~5.0 | maximum 0.5 | minimum 51 | 35 | >120 | maximum 120 | minimum 6 |
| Fatty Acids. | Formula | Molecular Weight | Structure | wt % |
|---|---|---|---|---|
| Palmitic | C16H32O2 | 256 | 16:0 | 5.85 |
| Stearic | C18H36O2 | 284 | 18:0 | 2.51 |
| Oleic | C18H34O2 | 282 | 18:1 | 63.8 |
| Linoleic | C18H32O2 | 280 | 18:2 | 25.3 |
| Linolenic | C18H30O2 | 278 | 18:3 | 0.51 |
| Behenic | C22H44O2 | 340 | 22:0 | 0.66 |
| Others | 1.29 | |||
| Total Saturated Fatty Acids (SFA) | 9.02 | |||
| Total Monounsaturated Fatty Acids (MUFA) | 63.84 | |||
| Total Polyunsaturated Fatty Acids (PUFA) | 25.85 | |||
| The degree of Unsaturation (DU) | 115.5 | |||
| Long Chain Saturated Factor (LCSF) | 2.83 | |||
| Non-Edible Biodiesels | Fatty Acids (% w/w) | Ref. | |||||
|---|---|---|---|---|---|---|---|
| C16:0 | C18:0 | C18:1 | C18:2 | C18:3 | Others | ||
| SFO this study | 5.85 | 2.51 | 63.84 | 25.34 | 0.51 | 1.95 | |
| SFO | 5.62 | 1.27 | 67.31 | 24.68 | 0.08 | 1.04 | [18] |
| SFO | 4.20 | 2.32 | 71.00 | 20.15 | 1.20 | 1.13 | [21] |
| SFO | 3.87 | 0.92 | 67.21 | 27.12 | 0.11 | 0.77 | [28] |
| SFO | 3.79 | 1.01 | 65.23 | 28.92 | 0.14 | 0.91 | [42] |
| SFO | 66.20 | 28.20 | 5.60 | [43] | |||
| PSO | 6.07 | 3.13 | 47.73 | 37.25 | 1.78 | 4.04 | [39] |
| Tobacco | 8.90 | 3.50 | 14.10 | 70.10 | 1.00 | 2.40 | [45] |
| Jatropha | 16.20 | 8.20 | 38.40 | 36.80 | 0.40 | 0 | [46] |
| Rapeseed | 3.49 | 0.85 | 64.40 | 22.30 | 8.23 | 0.73 | [47] |
| Cottonseed | 28.70 | 0.90 | 13.00 | 57.40 | 0 | 0 | [48] |
| Neem | 12.00 | 10.00 | 61.00 | 16.00 | 0 | 1.00 | [49] |
| Karanja | 9.80 | 6.20 | 72.20 | 11.80 | 0 | 0 | [50] |
| Moringa | 6.50 | 6.00 | 72.20 | 1.0 | 0.65 | 13.65 | [51] |
| Wavenumber (cm−1) | Group Attribution | Vibration Type | Functional Groups | Absorption Intensity |
|---|---|---|---|---|
| 2924 | =C–H | Asymmetric stretching vibration | Alkyl | Strong |
| 2854 | –CH2 | Symmetric stretching vibration | Aromatic | Strong |
| 1742 | –C=O | Stretching | Carbonyl | Strong |
| 1460.5 | –CH2 | Shear-type vibration | Alkanes | Weak |
| 1244.8 | –CH3 | Bending vibration | Alkanes | Weak |
| 1195.7 | C–O–C | Anti-symmetric stretching vibration | Ethers | Middling |
| 1169.7 | C–O–C | Anti-symmetric stretching vibration | Ethers | Middling |
| 1120.4 | C–O–C | Anti-symmetric stretching vibration | Ethers | Weak |
| 1017.2 | C–O–C | Anti-symmetric stretching vibration | Ethers | Weak |
| 722.7 | –CH2 | Plane rocking vibration | Aromatic | Weak |
| Exp. Number | Run Order | M | C | T | Methanol: Oil (Molar Ratio) | KOH (wt %) | Temp (°C) | SFO Biodiesel Yield (%) | |
|---|---|---|---|---|---|---|---|---|---|
| Experimental | Predicted | ||||||||
| 1 | 7 | 0 | −1 | −1 | 5 | 0.5 | 45 | 86.65 | 85.27 |
| 2 | 13 | 1 | −1 | 0 | 6 | 0.5 | 55 | 95.75 | 95.89 |
| 3 | 14 | 0 | 0 | 0 | 5 | 1 | 55 | 91.65 | 92.98 |
| 4 | 12 | −1 | 0 | −1 | 4 | 1 | 45 | 80.34 | 80.85 |
| 5 | 15 | 1 | 0 | −1 | 6 | 1 | 45 | 87.71 | 88.95 |
| 6 | 6 | −1 | −1 | 0 | 4 | 0.5 | 55 | 75.24 | 76.12 |
| 7 | 9 | −1 | 1 | 0 | 4 | 1.5 | 55 | 82.27 | 82.13 |
| 8 | 11 | 0 | 0 | 0 | 5 | 1 | 55 | 93.65 | 92.98 |
| 9 | 5 | 0 | 0 | 0 | 5 | 1 | 55 | 93.65 | 92.98 |
| 10 | 8 | 0 | 1 | −1 | 5 | 1.5 | 45 | 84.58 | 84.21 |
| 11 | 4 | 1 | 1 | 0 | 6 | 1.5 | 55 | 82.11 | 81.23 |
| 12 | 3 | 1 | 0 | 1 | 6 | 1 | 65 | 89.33 | 88.82 |
| 13 | 2 | −1 | 1 | 1 | 4 | 1 | 65 | 79.30 | 78.06 |
| 14 | 1 | 0 | −1 | 1 | 5 | 0.5 | 65 | 86.71 | 87.08 |
| 15 | 10 | 0 | 1 | 1 | 5 | 1.5 | 65 | 78.10 | 79.48 |
| Term. | Coefficients | Standard Errors | Computed T-Value | p Value |
|---|---|---|---|---|
| Constant | 92.983 | 0.893 | 104.17 | 0.000 |
| M | 4.719 | 0.547 | 8.63 | 0.000 |
| C | −2.161 | 0.547 | −3.95 | 0.011 |
| T | −0.730 | 0.547 | −1.34 | 0.239 |
| M × M | −4.490 | 0.805 | −5.58 | 0.003 |
| C × C | −4.650 | 0.805 | −5.78 | 0.002 |
| T × T | −4.323 | 0.805 | −5.37 | 0.003 |
| M × C | −5.168 | 0.773 | −6.69 | 0.001 |
| M × T | 0.665 | 0.773 | 0.86 | 0.429 |
| C × T | −1.635 | 0.773 | −2.12 | 0.088 |
| Source | Sum of Squares | Degree of Freedom | Mean Square | F-Value | p-Value | Remarks |
|---|---|---|---|---|---|---|
| Model | 532.603 | 9 | 59.178 | 24.76 | 0.001 | Significant |
| M-Methanol | 178.123 | 1 | 178.123 | 74.53 | 0.000 | Highly significant |
| C-Catalyst | 37.364 | 1 | 37.364 | 15.63 | 0.011 | Significant |
| T-Temperature | 4.263 | 1 | 4.263 | 1.78 | 0.239 | Not significant |
| M2 | 74.447 | 1 | 74.447 | 31.15 | 0.003 | Significant |
| C2 | 79.847 | 1 | 79.847 | 27.00 | 0.002 | Significant |
| T2 | 69.004 | 1 | 69.004 | 28.87 | 0.003 | Significant |
| MC | 106.823 | 1 | 106.823 | 44.69 | 0.001 | Significant |
| MT | 1.769 | 1 | 1.769 | 0.74 | 0.429 | Not significant |
| CT | 10.693 | 1 | 10.693 | 4.47 | 0.088 | Not significant |
| Lack of Fit | 9.284 | 3 | 3.095 | 2.32 | 0.315 | Not Significant |
| Pure Error | 2.667 | 2 | 1.333 | |||
| Total | 544.553 | 14 | ||||
| R2 = 0.9781 | Adj R2 = 0.9386 | |||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anwar, M.; Rasul, M.G.; Ashwath, N.; Rahman, M.M. Optimisation of Second-Generation Biodiesel Production from Australian Native Stone Fruit Oil Using Response Surface Method. Energies 2018, 11, 2566. https://doi.org/10.3390/en11102566
Anwar M, Rasul MG, Ashwath N, Rahman MM. Optimisation of Second-Generation Biodiesel Production from Australian Native Stone Fruit Oil Using Response Surface Method. Energies. 2018; 11(10):2566. https://doi.org/10.3390/en11102566
Chicago/Turabian StyleAnwar, Mohammad, Mohammad G. Rasul, Nanjappa Ashwath, and Md Mofijur Rahman. 2018. "Optimisation of Second-Generation Biodiesel Production from Australian Native Stone Fruit Oil Using Response Surface Method" Energies 11, no. 10: 2566. https://doi.org/10.3390/en11102566
APA StyleAnwar, M., Rasul, M. G., Ashwath, N., & Rahman, M. M. (2018). Optimisation of Second-Generation Biodiesel Production from Australian Native Stone Fruit Oil Using Response Surface Method. Energies, 11(10), 2566. https://doi.org/10.3390/en11102566






