The Potential of Activated Carbon Made of Agro-Industrial Residues in NOx Immissions Abatement
Abstract
:1. Introduction
2. Materials and Methods
2.1. Samples Preparation
2.1.1. Chemical Activation
2.1.2. Physical Activation
2.2. Characterization of Activated Carbons
2.2.1. Pore Structure and Morphology Characterization
2.2.2. Characterization of Carbon Surface Chemistry and Composition
Temperature Programmed Desorption Coupled with Mass Spectrometry (TPD-MS)
Fourier Transform Infrared Spectroscopy (FTIR)
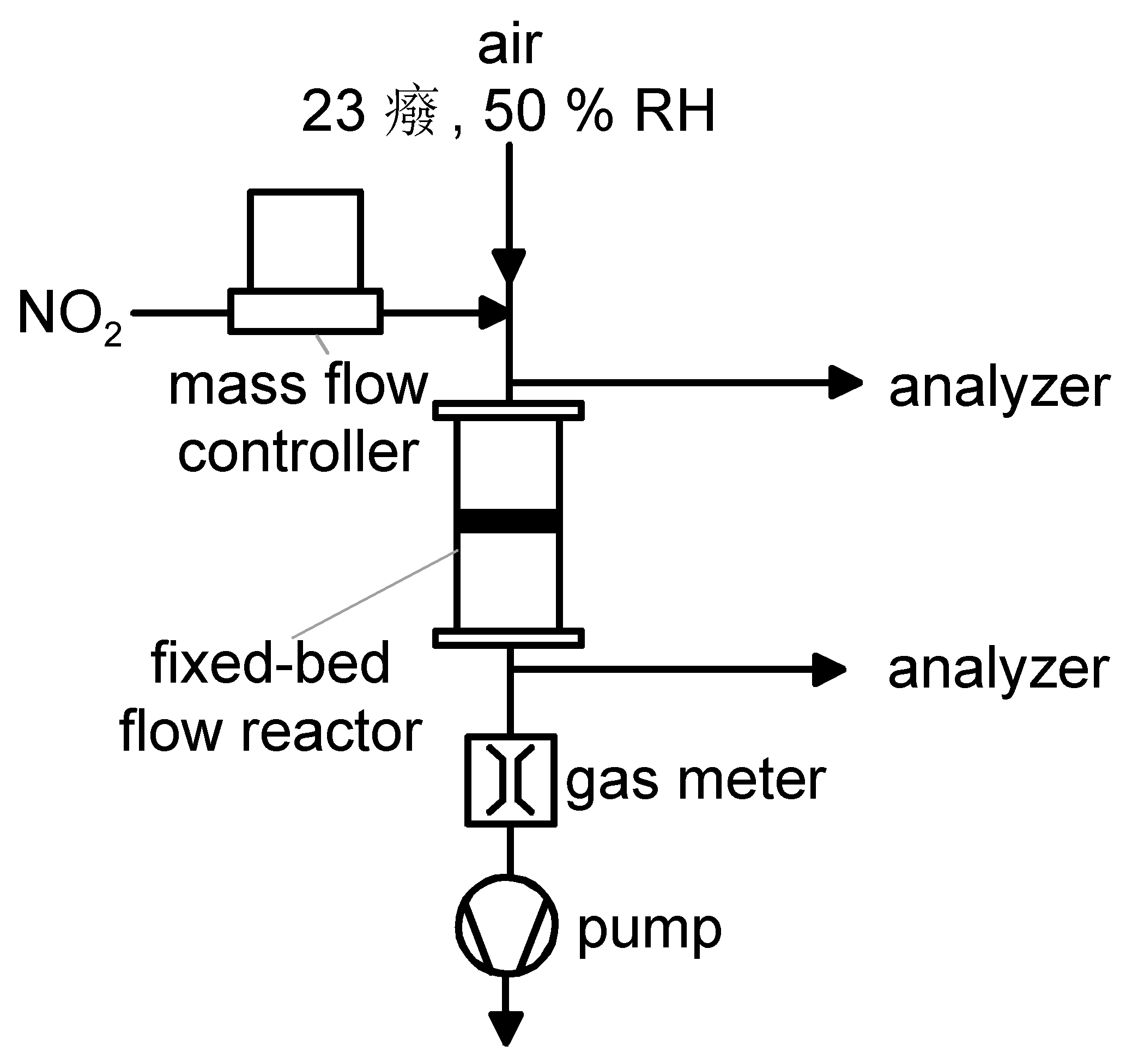
2.3. NO2 Adsorption Experiments
3. Results and Discussion
3.1. Activated Carbon Characterization
3.1.1. Morphology and Pore Structure Characterizations
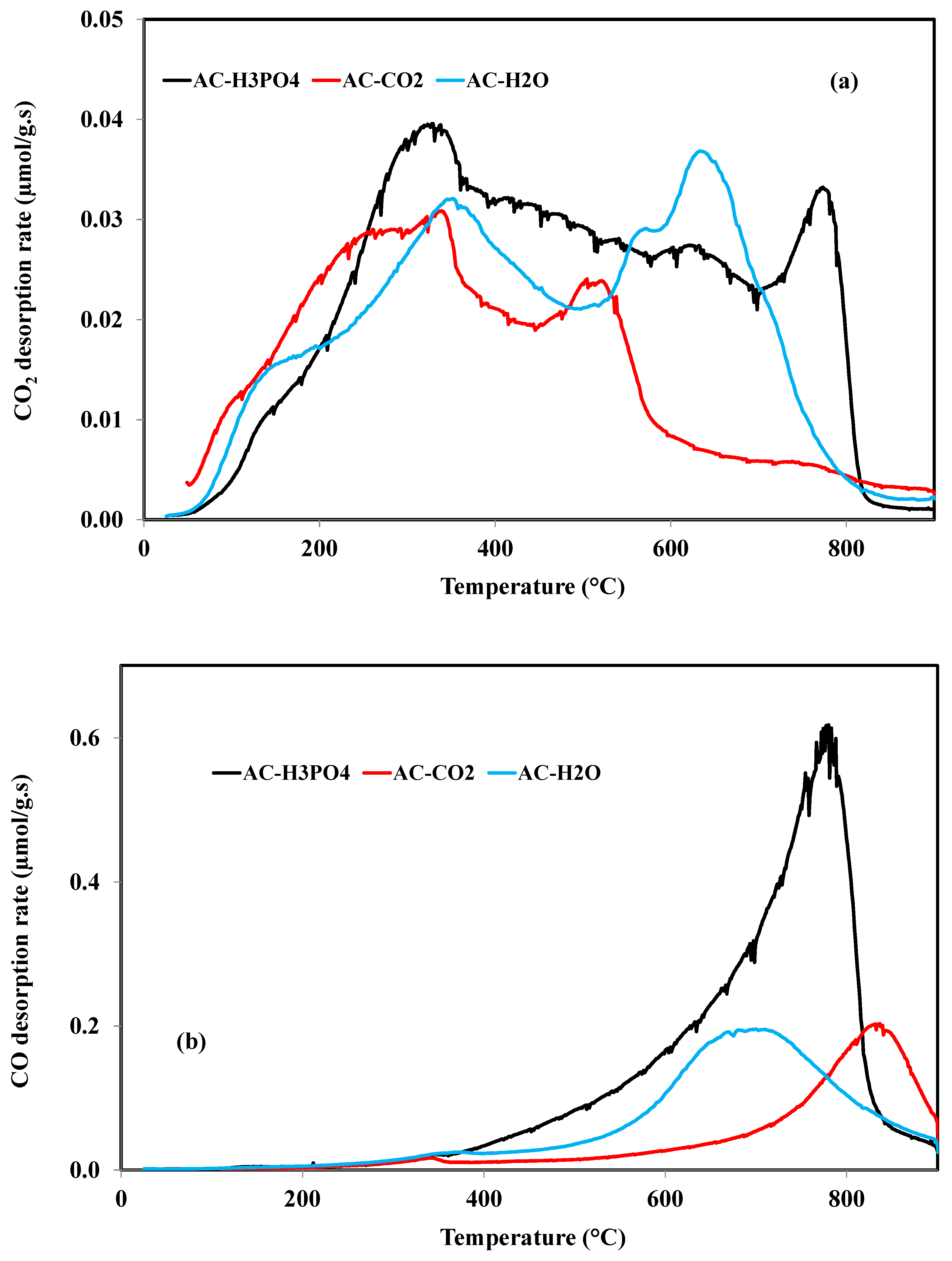
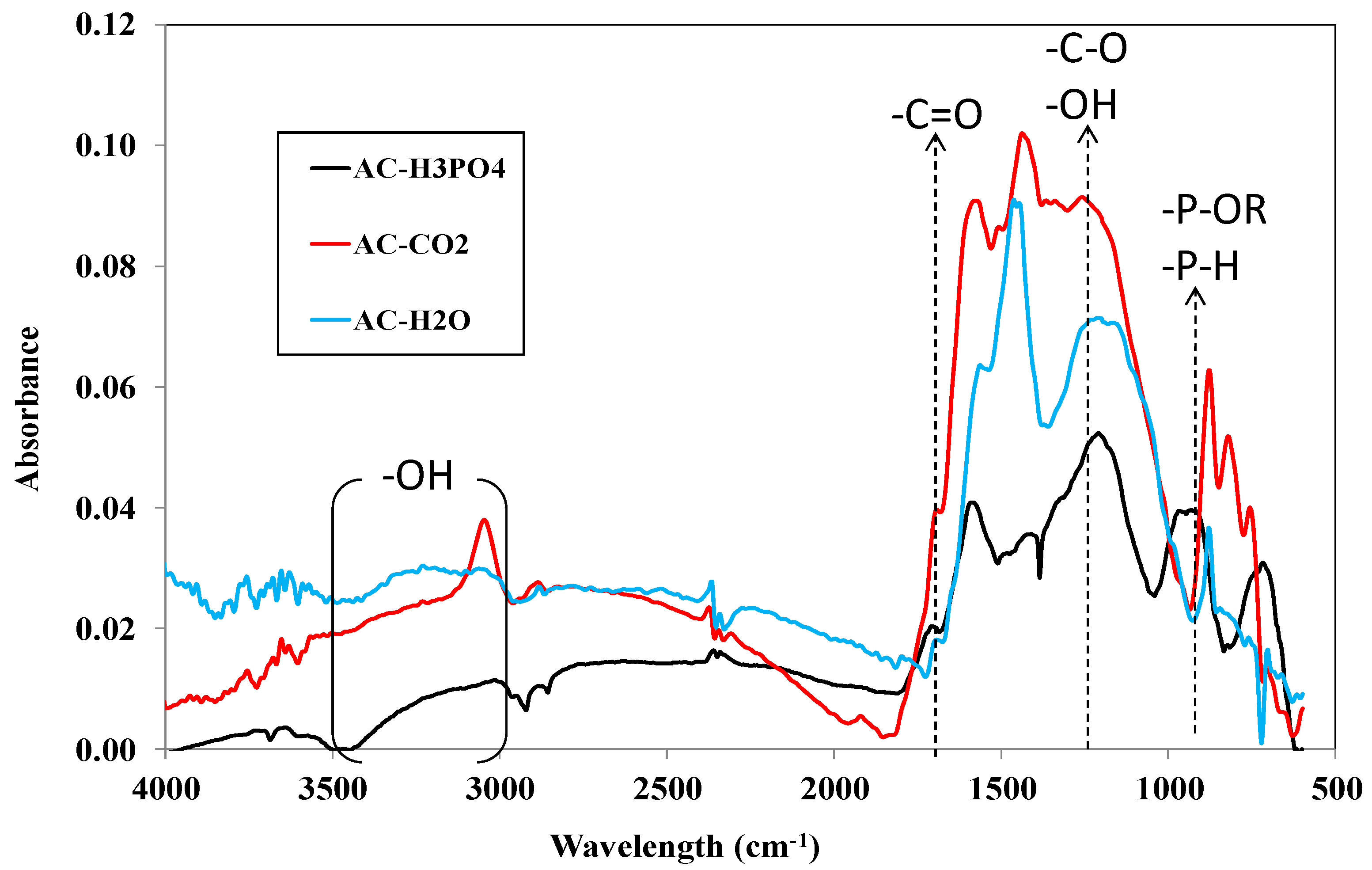
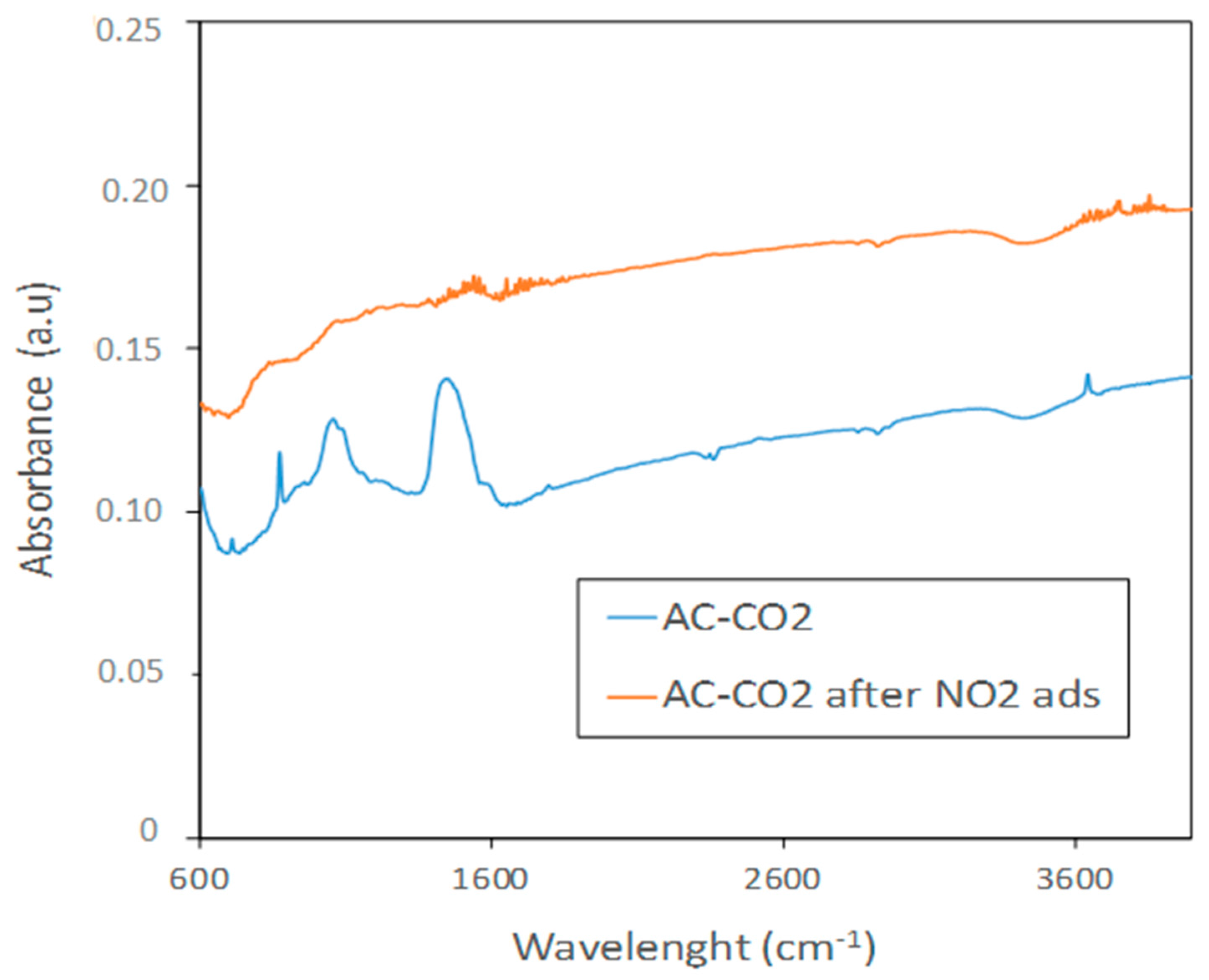
3.1.2. Characterization of Carbon Surface Chemistry
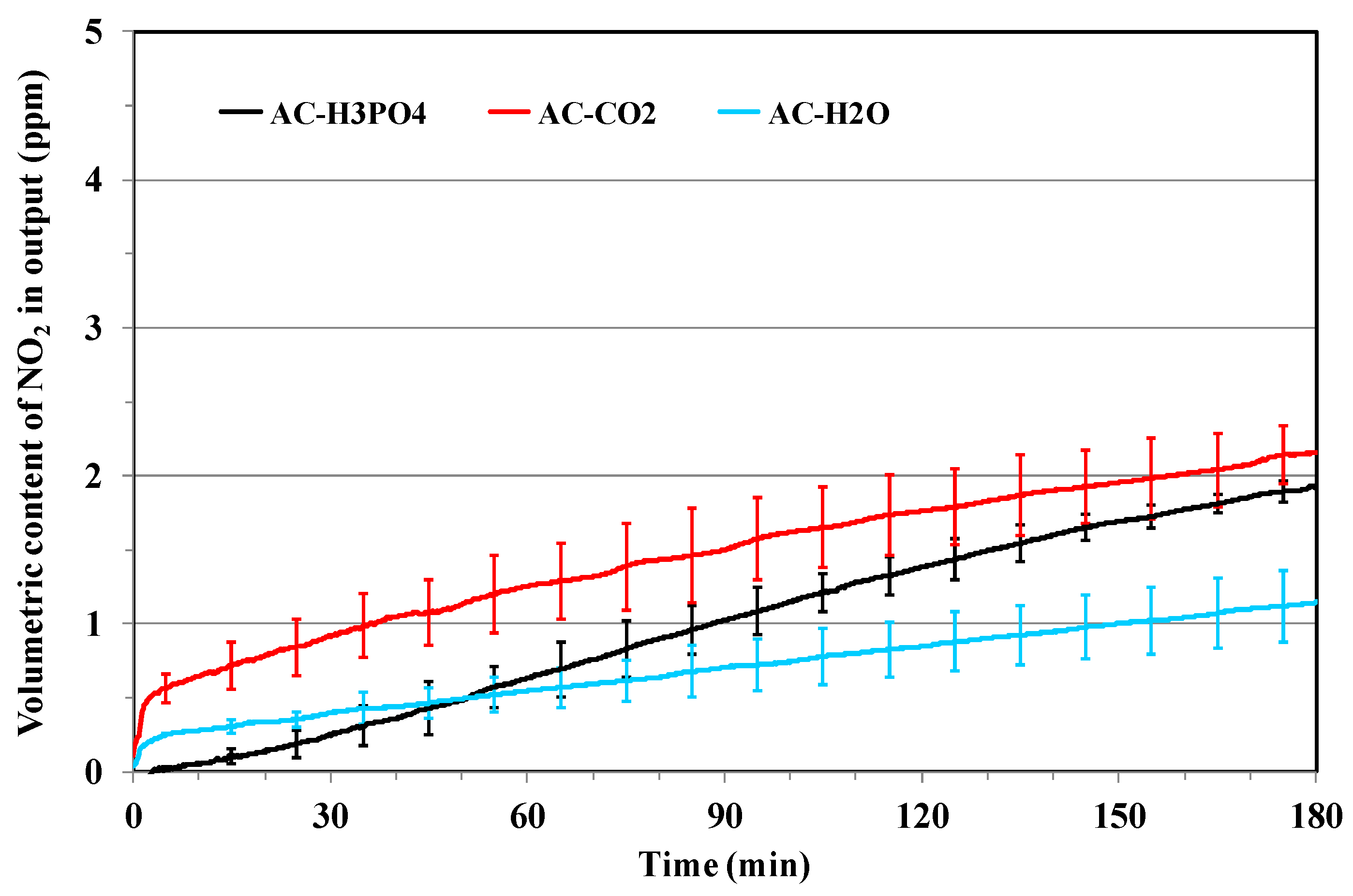
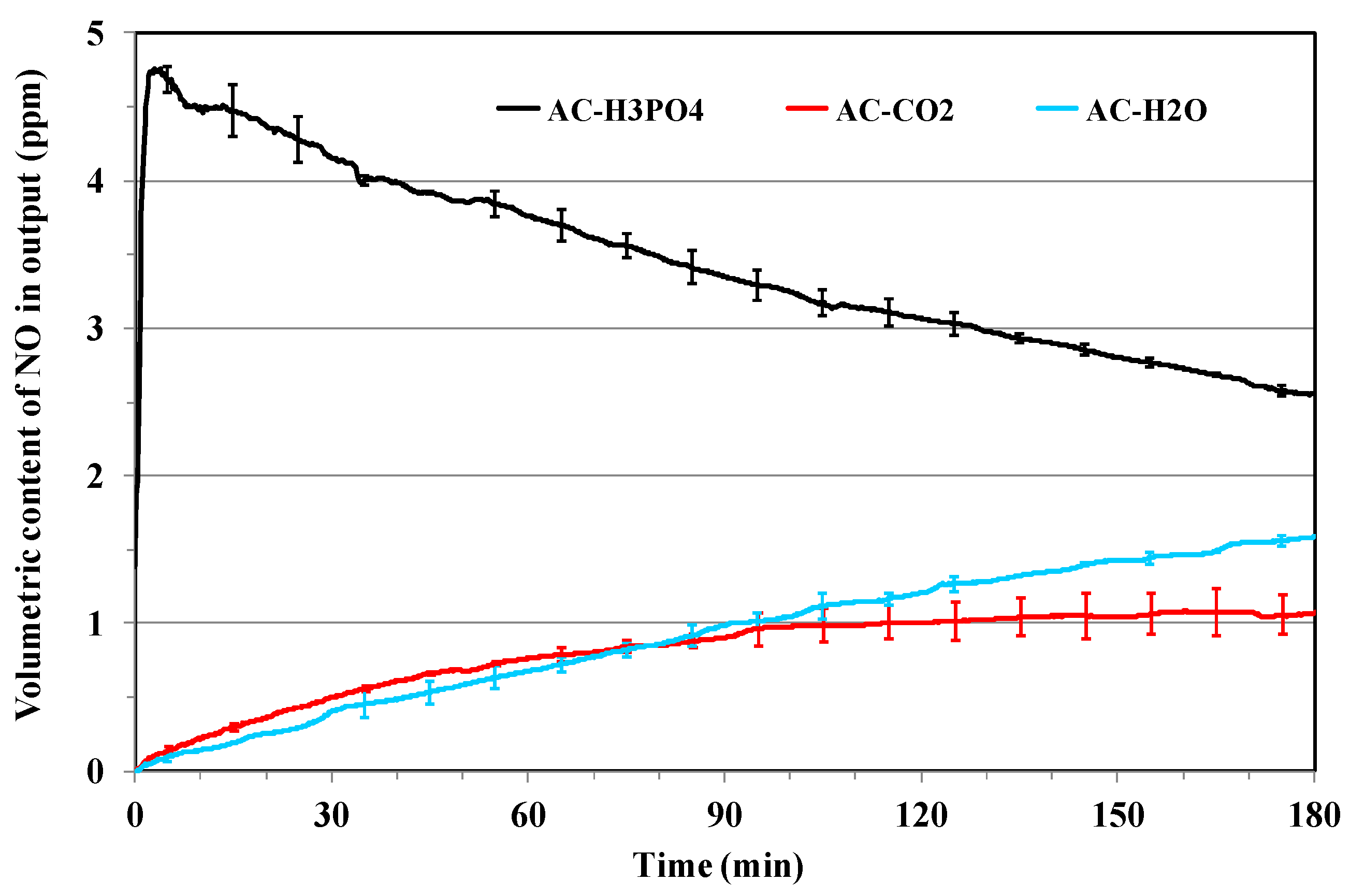
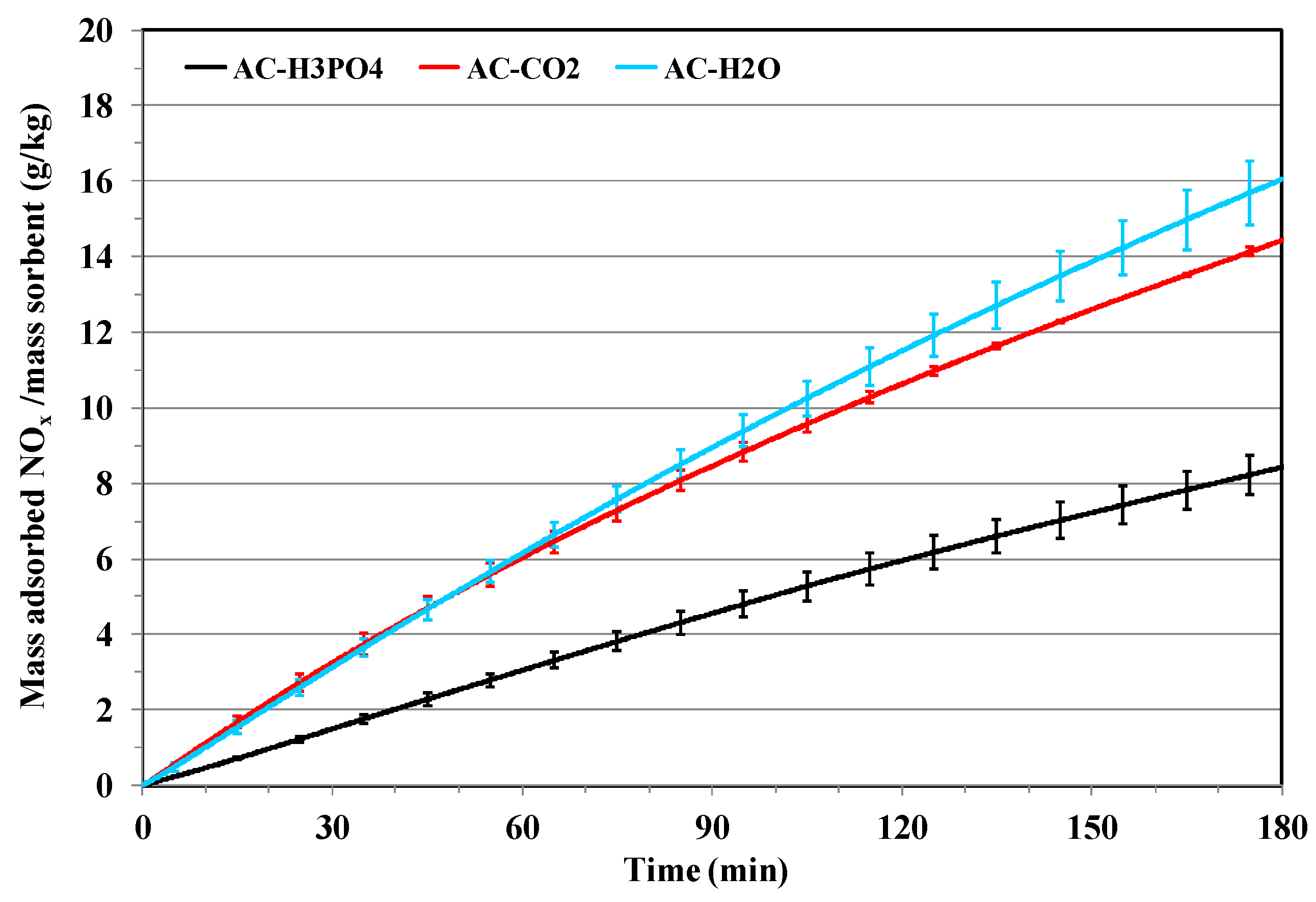
3.2. NOx Adsorption on the Different Activated Carbons
Effect of Porosity and Chemical Surface Groups
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wilkins, C.K.; Clausen, P.A.; Wolkoff, P.; Larsen, S.T.; Hammer, M.; Larsen, K.; Hansen, V.; Nielsen, G.D. Formation of strong airway irritants in mixtures of isoprene/ozone and isoprene/ozone/nitrogen dioxide. Environ. Health Perspect. 2001, 109, 937–941. [Google Scholar] [CrossRef] [PubMed]
- Blondeau, P.; Iordache, V.; Poupard, O.; Genin, D.; Allard, F. Relationship between outdoor and indoor air quality in eight French schools. Indoor Air 2005, 15, 2–12. [Google Scholar] [CrossRef] [PubMed]
- Gou, X.; Wu, C.; Zhang, K.; Xu, G.; Si, M.; Wang, Y.; Wang, E.; Liu, L.; Wu, J. Low Temperature Performance of Selective Catalytic Reduction of NO with NH3 under a Concentrated CO2 Atmosphere. Energies 2015, 8, 12331–12341. [Google Scholar] [CrossRef]
- Levasseur, B.; Ebrahim, A.M.; Burress, J.; Bandosz, T.J. Interactions of NO2 at ambient temperature with cerium–zirconium mixed oxides supported on SBA-15. J. Hazard. Mater. 2011, 197, 294–303. [Google Scholar] [CrossRef] [PubMed]
- Hodjati, S.; Vaezzadeh, K.; Petit, C.; Pitchon, V.; Kiennemann, A. Absorption/desorption of NOx process on perovskites: Performances to remove NOx from a lean exhaust gas. Appl. Catal. B Environ. 2000, 26, 5–16. [Google Scholar] [CrossRef]
- Jeguirim, M.; Tschamber, V.; Brilhac, J.F.; Ehrburger, P. Interaction mechanism of NO2 with carbon black: Effect of surface oxygen complexes. J. Anal. Appl. Pyrolysis 2004, 72, 171–181. [Google Scholar] [CrossRef]
- Nowicki, P.; Pietrzak, R.; Wachowska, H. Sorption properties of active carbons obtained from walnut shells by chemical and physical activation. Catal. Today 2010, 150, 107–114. [Google Scholar] [CrossRef]
- Pietrzak, R. Sawdust pellets from coniferous species as adsorbents for NO2 removal. Bioresour. Technol. 2010, 101, 907–913. [Google Scholar] [CrossRef] [PubMed]
- Belala, Z.; Belhachemi, M.; Jeguirim, M. Activated Carbon Prepared from Date Pits for the Retention of NO2 at Low Temperature. Int. J. Chem. React. Eng. 2014, 12, 717–726. [Google Scholar] [CrossRef]
- Ghouma, I.; Jeguirim, M.; Dorge, S.; Limousy, L.; Matei Ghimbeu, C.; Ouederni, A. Activated carbon prepared by physical activation of olive stones for the removal of NO2 at ambient temperature. C. R. Chim. 2015, 18, 63–74. [Google Scholar] [CrossRef]
- Belhachemi, M.; Jeguirim, M.; Limousy, L.; Addoun, F. Comparison of NO2 removal using date pits activated carbon and modified commercialized activated carbon via different preparation methods: Effect of porosity and surface chemistry. Chem. Eng. J. 2014, 253, 121–129. [Google Scholar] [CrossRef]
- Heschel, W.; Ahnert, F. Multicomponent adsorption of NO2, SO2 and butane from air on activated carbon. Adsorpt. Sci. Technol. 2002, 20, 353–370. [Google Scholar]
- Labaki, M.; Issa, M.; Smeekens, S.; Heylen, S.; Kirschhock, C.E.A.; Villani, K.; Jeguirim, M.; Habermacher, D.; Brilhac, J.F.; Martens, J.A. Modeling of NOx adsorption–desorption–reduction cycles on a ruthenium loaded Na–Y zeolite. Appl. Catal. B Environ. 2010, 97, 13–20. [Google Scholar] [CrossRef]
- Zouaoui, N.; Labaki, M.; Jeguirim, M. Diesel soot oxidation by nitrogen dioxide, oxygen and water under engine exhaust conditions: Kinetics data related to the reaction mechanism. C. R. Chim. 2014, 17, 672–680. [Google Scholar] [CrossRef]
- Bennici, S.; Gervasini, A.; Ravasio, N.; Zaccheria, F. Optimization of Tailoring of CuOx Species of Silica Alumina Supported Catalysts for the Selective Catalytic Reduction of NOx. J. Phys. Chem. B 2003, 107, 5168–5176. [Google Scholar] [CrossRef]
- Limousy, L.; Mahzoul, H.; Brilhac, J.F.; Gilot, P.; Garin, F.; Maire, G. SO2 sorption on fresh and aged SOx traps. Appl. Catal. B Environ. 2003, 42, 237–249. [Google Scholar] [CrossRef]
- Sager, U.; Schmidt, F. Adsorption of Nitrogen Oxides, Water Vapour and Ozone onto Activated Carbon. Adsorpt. Sci. Technol. 2009, 27, 135–145. [Google Scholar] [CrossRef]
- Sager, U.; Suhartiningsih; Schmidt, F. Einfluss der NO2-Dosierung auf Adsorptionsfiltertests. Chem. Ing. Tech. 2010, 82, 1737–1742. [Google Scholar] [CrossRef]
- Sager, U.; Schmidt, W.; Schmidt, F. Suhartiningsih Catalytic reduction of nitrogen oxides via nanoscopic oxide catalysts within activated carbons at room temperature. Adsorption 2013, 19, 1027–1033. [Google Scholar] [CrossRef]
- Sager, U.; Däuber, E.; Asbach, C.; Bathen, D.; Schmidt, F.; Weidenthaler, C.; Tseng, J.C.; Schmidt, W. Differences between the adsorption of NO2 and NO on modified activated carbon/Unterschiede bei der Adsorption von NO2 und NO an modifizierter Aktivkohle. Air Qual. Control 2014, 74, 181–184. [Google Scholar]
- Sager, U.; Däuber, E.; Bathen, D.; Asbach, C.; Schmidt, F.; Tseng, J.-C.; Pommerin, A.; Weidenthaler, C.; Schmidt, W. Influence of the degree of infiltration of modified activated carbons with CuO/ZnO on the separation of NO2 at ambient temperatures. Adsorpt. Sci. Technol. 2016, 34, 307–319. [Google Scholar] [CrossRef]
- Limousy, L.; Ghouma, I.; Ouederni, A.; Jeguirim, M. Amoxicillin removal from aqueous solution using activated carbon prepared by chemical activation of olive stone. Environ. Sci. Pollut. Res. 2017, 24, 9993–10004. [Google Scholar] [CrossRef] [PubMed]
- Travis, W.; Gadipelli, S.; Guo, Z. Superior CO2 adsorption from waste coffee ground derived carbons. RSC Adv. 2015, 5, 29558–29562. [Google Scholar] [CrossRef]
- Jagtoyen, M.; Derbyshire, F. Activated carbons from yellow poplar and white oak by H3PO4 activation. Carbon 1998, 36, 1085–1097. [Google Scholar] [CrossRef]
- Srinivas, G.; Krungleviciute, V.; Guo, Z.-X.; Yildirim, T. Exceptional CO2 capture in a hierarchically porous carbon with simultaneous high surface area and pore volume. Energy Environ. Sci. 2013, 7, 335–342. [Google Scholar] [CrossRef]
- Srinivas, G.; Burress, J.; Yildirim, T. Graphene oxide derived carbons (GODCs): Synthesis and gas adsorption properties. Energy Environ. Sci. 2012, 5, 6453–6459. [Google Scholar] [CrossRef]
- Román, S.; Ledesma, B.; Álvarez-Murillo, A.; Al-Kassir, A.; Yusaf, T. Dependence of the Microporosity of Activated Carbons on the Lignocellulosic Composition of the Precursors. Energies 2017, 10, 542. [Google Scholar] [CrossRef]
- Guizani, C.; Jeguirim, M.; Gadiou, R.; Escudero Sanz, F.J.; Salvador, S. Biomass char gasification by H2O, CO2 and their mixture: Evolution of chemical, textural and structural properties of the chars. Energy 2016, 112, 133–145. [Google Scholar] [CrossRef]
- Figueiredo, J.L.; Pereira, M.F.R.; Freitas, M.M.A.; Órfão, J.J.M. Modification of the surface chemistry of activated carbons. Carbon 1999, 37, 1379–1389. [Google Scholar] [CrossRef]
- Reddy, K.S.K.; Al Shoaibi, A.; Srinivasakannan, C. A comparison of microstructure and adsorption characteristics of activated carbons by CO2 and H3PO4 activation from date palm pits. New Carbon Mater. 2012, 27, 344–351. [Google Scholar] [CrossRef]
- El-Hendawy, A.-N.A. Influence of HNO3 oxidation on the structure and adsorptive properties of corncob-based activated carbon. Carbon 2003, 41, 713–722. [Google Scholar] [CrossRef]
- Shen, W.; Li, Z.; Liu, Y. Surface chemical functional groups modification of porous carbon. Recent Pat. Chem. Eng. 2008, 1, 27–40. [Google Scholar] [CrossRef]
- Puziy, A.M.; Poddubnaya, O.I.; Martı́nez-Alonso, A.; Suárez-Garcı́a, F.; Tascón, J.M.D. Synthetic carbons activated with phosphoric acid: I. Surface chemistry and ion binding properties. Carbon 2002, 40, 1493–1505. [Google Scholar] [CrossRef]
- Nowicki, P.; Skibiszewska, P.; Pietrzak, R. NO2 removal on adsorbents prepared from coffee industry waste materials. Adsorption 2013, 19, 521–528. [Google Scholar] [CrossRef]
- Nowicki, P.; Wachowska, H.; Pietrzak, R. Active carbons prepared by chemical activation of plum stones and their application in removal of NO2. J. Hazard. Mater. 2010, 181, 1088–1094. [Google Scholar] [CrossRef] [PubMed]
- Nowicki, P.; Pietrzak, R. Carbonaceous adsorbents prepared by physical activation of pine sawdust and their application for removal of NO2 in dry and wet conditions. Bioresour. Technol. 2010, 101, 5802–5807. [Google Scholar] [CrossRef] [PubMed]
- Gadipelli, S.; Guo, Z.X. Graphene-based materials: Synthesis and gas sorption, storage and separation. Prog. Mater. Sci. 2015, 69, 1–60. [Google Scholar] [CrossRef]

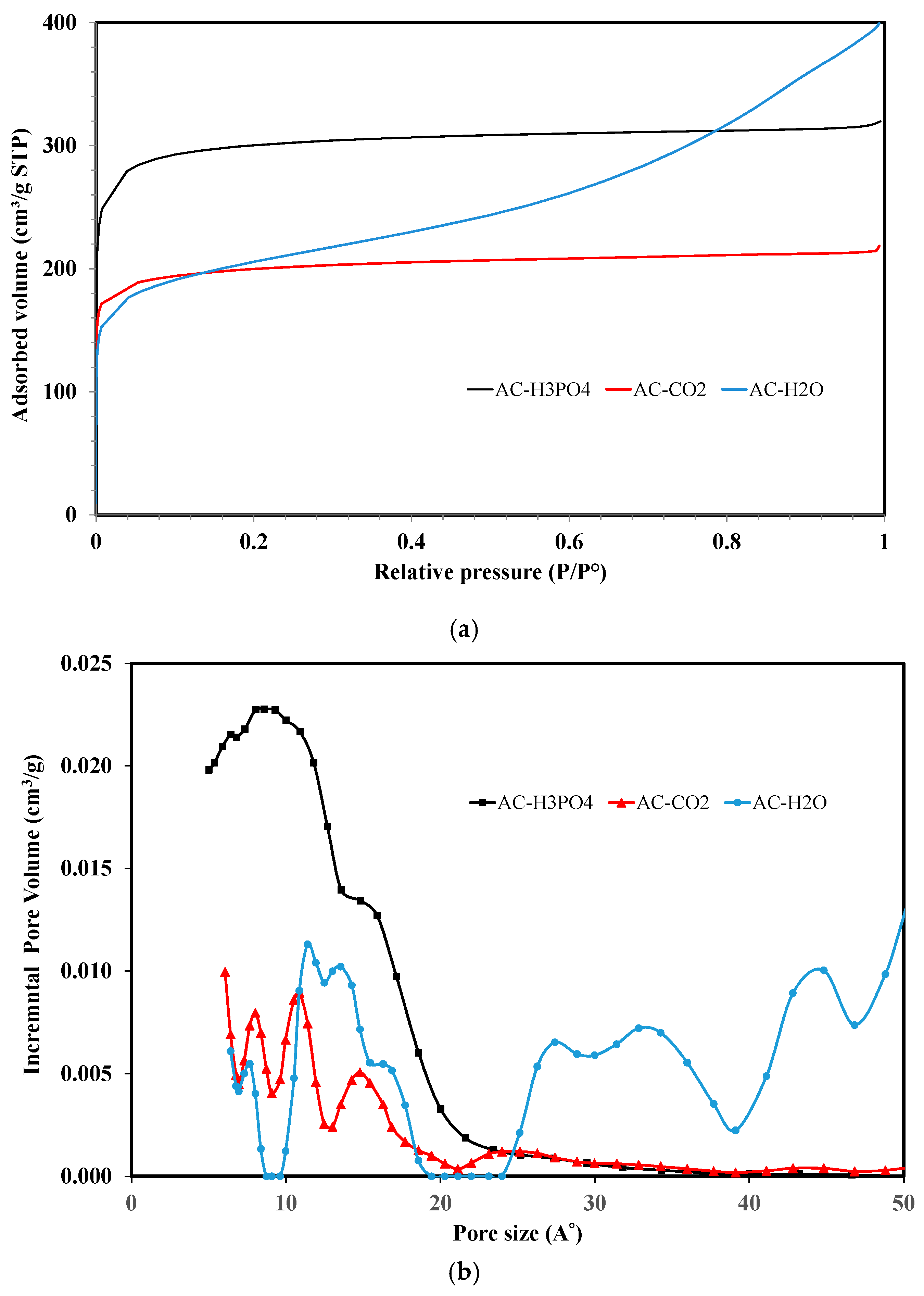

| Carbons | SBET (m2/g) | Sext (m2/g) | Sµ (m2/g) | VT pore (cm3/g) | Vmicro (cm3/g) | Vmeso (cm3/g) | % micro | Dp (A) |
|---|---|---|---|---|---|---|---|---|
| AC-H3PO4 | 1178 | 11 | 1167 | 0.49 | 0.45 | 0.04 | 92 | 16.7 |
| AC-CO2 | 757 | 83 | 674 | 0.32 | 0.30 | 0.02 | 94 | 17.5 |
| AC-H2O | 754 | 291 | 463 | 0.58 | 0.28 | 0.30 | 48 | 32.2 |
| Sample | CO (mmol/g) | CO2 (mmol/g) | H2O (mmol/g) | H2 (mmol/g) |
|---|---|---|---|---|
| AC-H3PO4 | 3.43 | 0.72 | 3.46 | 1.80 |
| AC-CO2 | 1.06 | 0.38 | 1.77 | 0.91 |
| AC-H2O | 1.25 | 0.39 | 1.06 | 6.76 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghouma, I.; Jeguirim, M.; Sager, U.; Limousy, L.; Bennici, S.; Däuber, E.; Asbach, C.; Ligotski, R.; Schmidt, F.; Ouederni, A. The Potential of Activated Carbon Made of Agro-Industrial Residues in NOx Immissions Abatement. Energies 2017, 10, 1508. https://doi.org/10.3390/en10101508
Ghouma I, Jeguirim M, Sager U, Limousy L, Bennici S, Däuber E, Asbach C, Ligotski R, Schmidt F, Ouederni A. The Potential of Activated Carbon Made of Agro-Industrial Residues in NOx Immissions Abatement. Energies. 2017; 10(10):1508. https://doi.org/10.3390/en10101508
Chicago/Turabian StyleGhouma, Imen, Mejdi Jeguirim, Uta Sager, Lionel Limousy, Simona Bennici, Eckhard Däuber, Christof Asbach, Roman Ligotski, Frank Schmidt, and Abdelmottaleb Ouederni. 2017. "The Potential of Activated Carbon Made of Agro-Industrial Residues in NOx Immissions Abatement" Energies 10, no. 10: 1508. https://doi.org/10.3390/en10101508









