Green Blends Based on Ionic Liquids with Improved Performance for Membrane Technology: Perspectives for Environmental Applications
Abstract
:1. Introduction
2. Results and Discussion
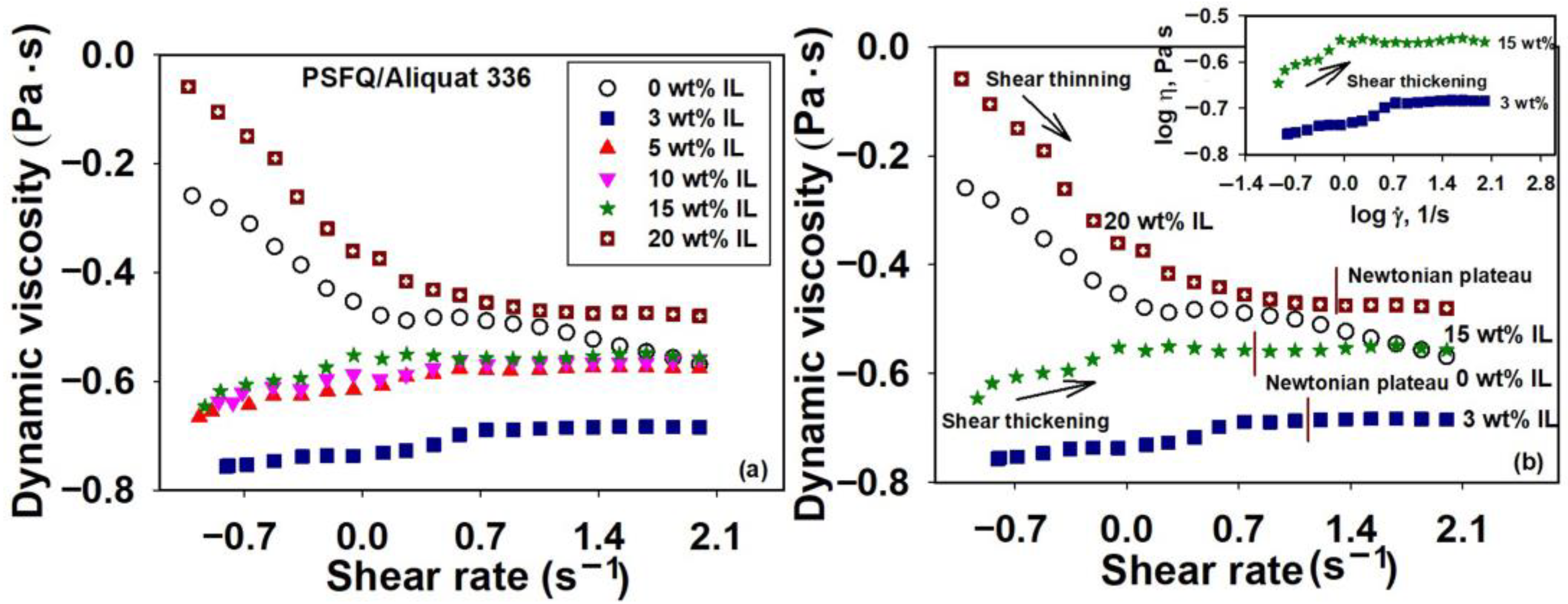
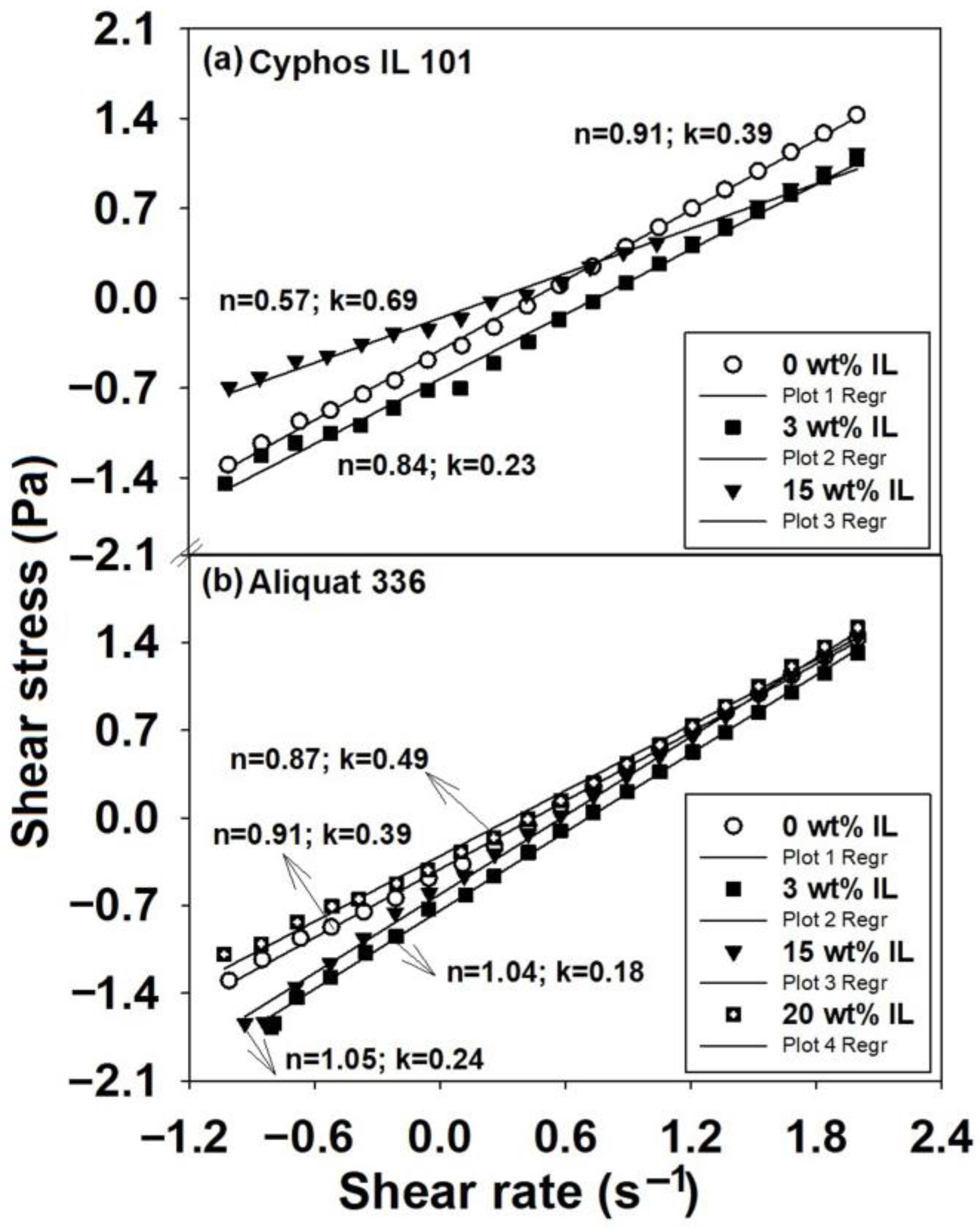
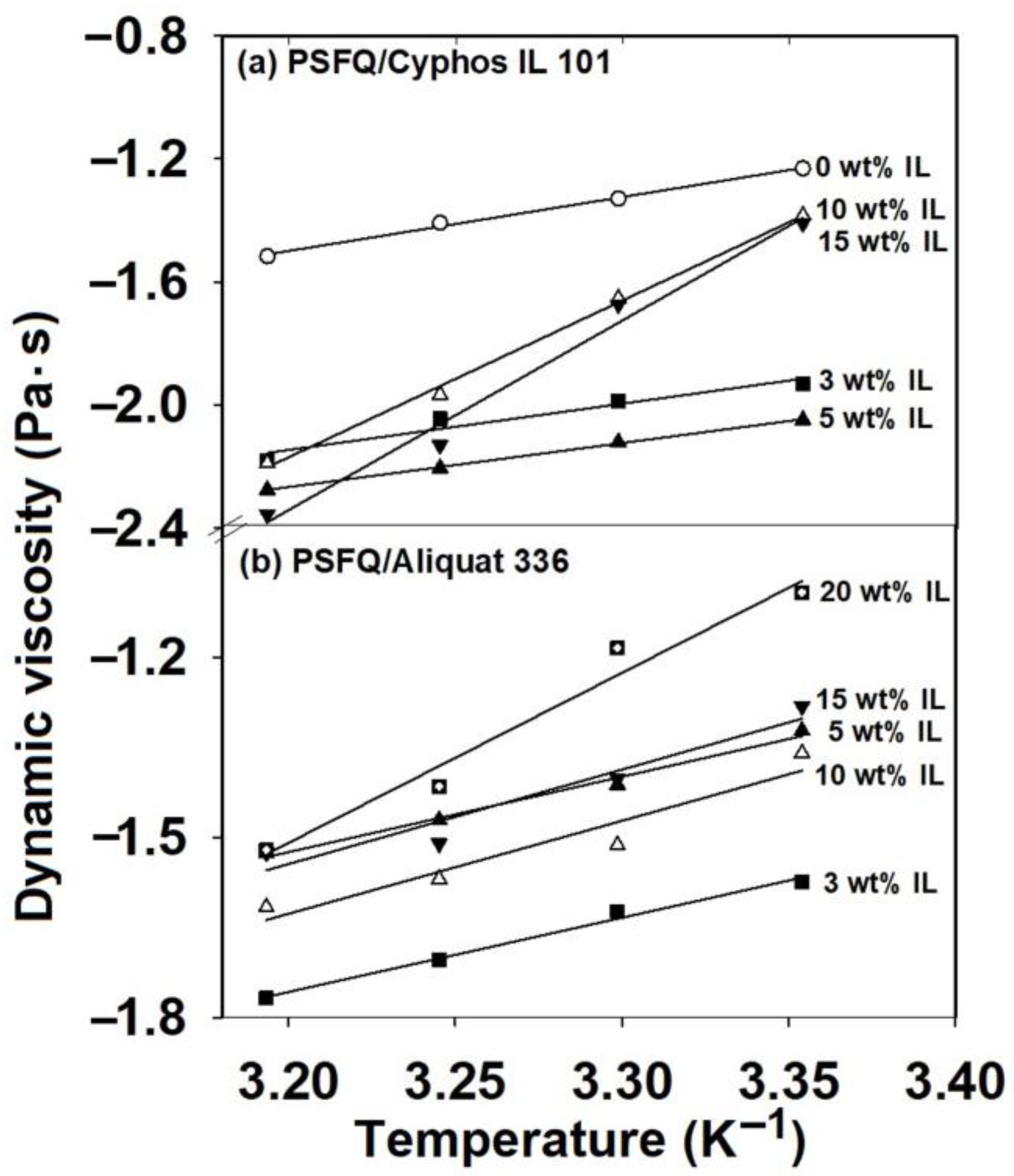
2.1. Rheological Response of Polysulfone Blends Based on Ionic Liquids
2.2. Tuning of Microstructure in Polysulfone Blends Based on Ionic Liquids by Location of the Compatibility Limit
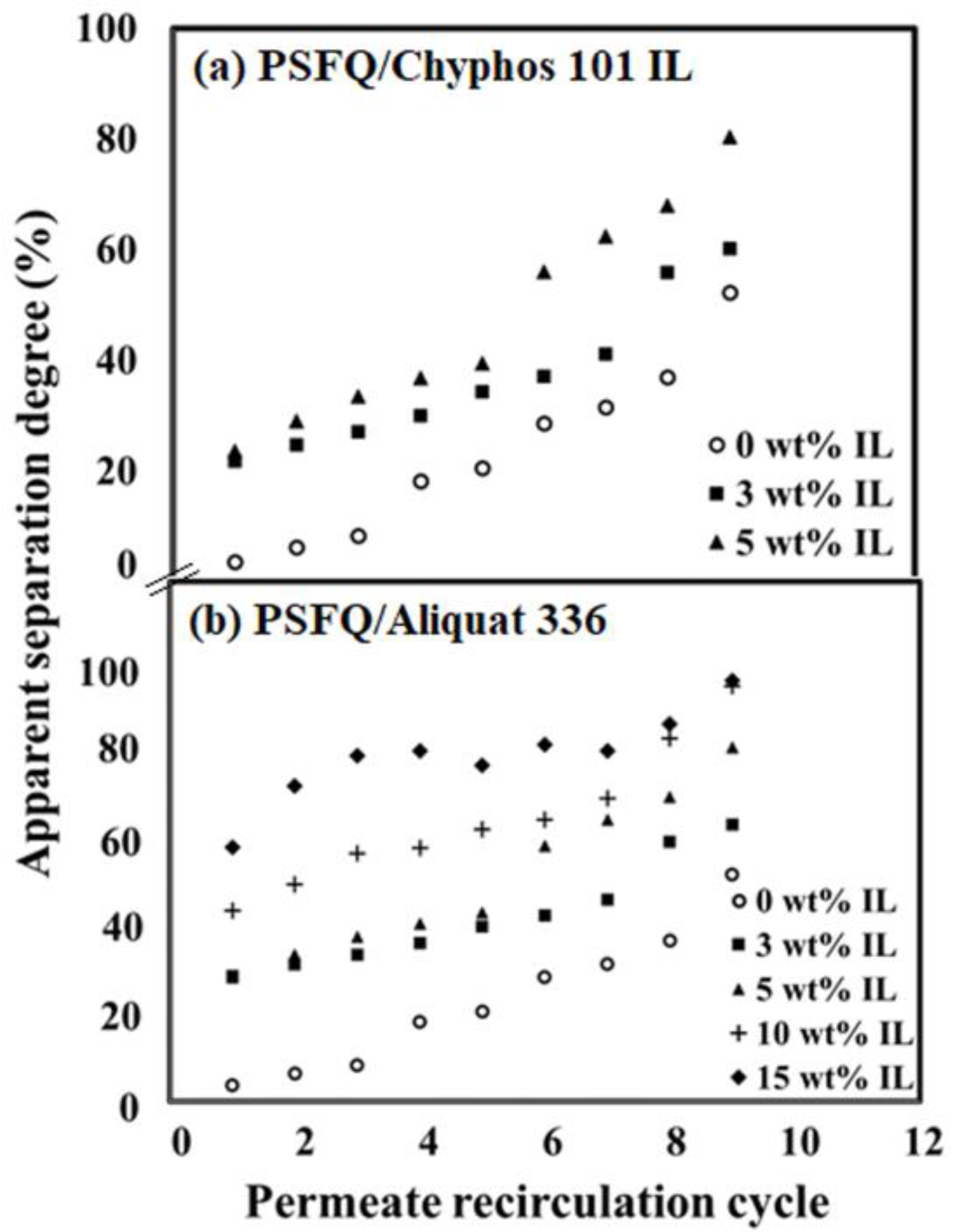
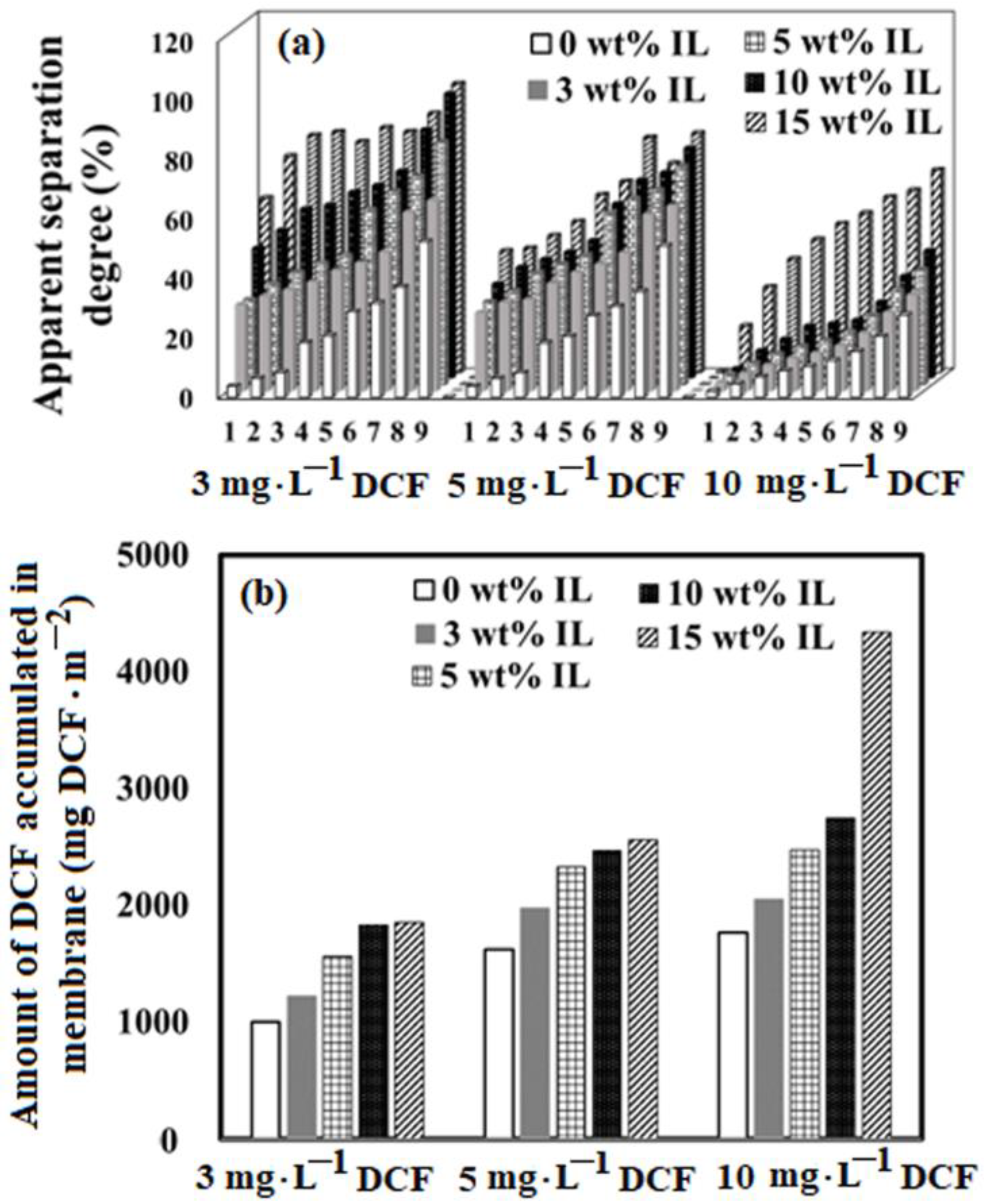
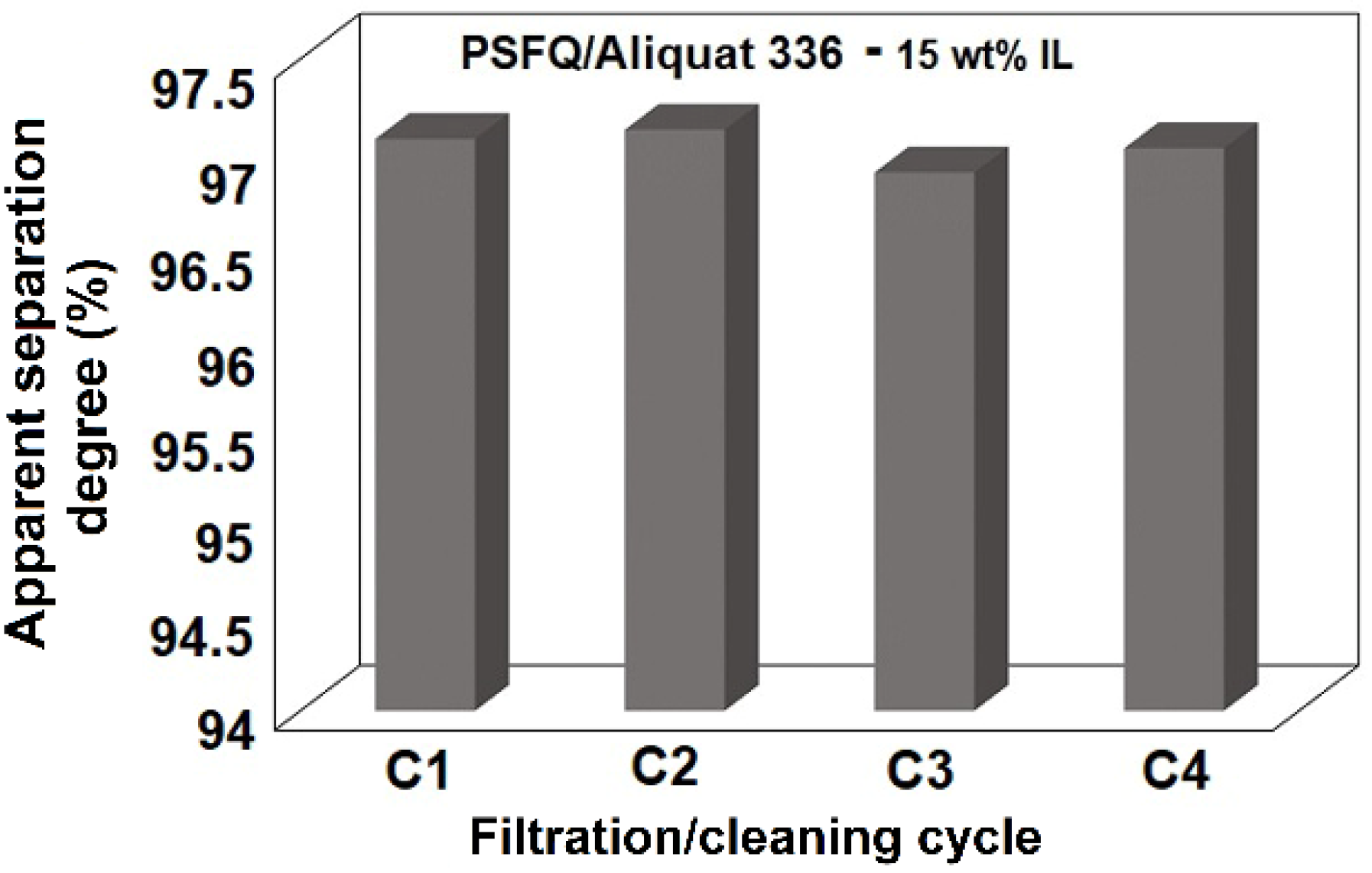
2.3. Membrane Efficiency in the Treatment Process of Water Containing Diclofenac
3. Materials and Methods
3.1. Materials
3.2. Membrane Preparation
3.3. Characterization and Testing
3.3.1. Rheological Investigations
3.3.2. Differential Scanning Calorimetry
3.3.3. Scanning Electron Microscopy
3.3.4. Ultrafiltration Tests
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Singh, N.B.; Nagpal, G.; Agrawal, S.; Rachna. Water Purification by Using Adsorbents: A Review. Environ. Technol. Innov. 2018, 11, 187–240. [Google Scholar] [CrossRef]
- Han, W.; Fu, F.; Cheng, Z.; Tang, B.; Wu, S. Studies on the Optimum Conditions Using Acid-Washed Zero-Valent Iron/Aluminum Mixtures in Permeable Reactive Barriers for the Removal of Different Heavy Metal Ions from Wastewater. J. Hazard. Mater. 2016, 302, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Cantwell, M.G.; Katz, D.R.; Sullivan, J.C.; Shapley, D.; Lipscomb, J.; Epstein, J.; Juhl, A.R.; Knudson, C.; O’Mullan, G.D. Spatial Patterns of Pharmaceuticals and Wastewater Tracers in the Hudson River Estuary. Water Res. 2018, 137, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Bi, E. Adsorption Site-Dependent Transport of Diclofenac in Water Saturated Minerals and Reference Soils. Chemosphere 2019, 236, 124256. [Google Scholar] [CrossRef]
- Obotey Ezugbe, E.; Rathilal, S. Membrane Technologies in Wastewater Treatment: A Review. Membranes 2020, 10, 89. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, N.N.R.; Ang, W.L.; Leo, C.P.; Mohammad, A.W.; Hilall, N. Current Advances in Membrane Technologies for Saline Wastewater Treatment: A Comprehensive Review. Desalination 2021, 517, 115170. [Google Scholar] [CrossRef]
- Saravanan, A.; Senthil Kumar, P.; Jeevanantham, S.; Karishma, S.; Tajsabreen, B.; Yaashikaa, P.R.; Reshma, B. Effective Water/Wastewater Treatment Methodologies for Toxic Pollutants Removal: Processes and Applications Towards Sustainable Development. Chemosphere 2021, 280, 130595. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, F.H.; Kumar, M.; Peinemann, K.V. Pebax®1657/Graphene Oxide Composite Membranes for Improved Water Vapor Separation. J. Membr. Sci. 2017, 525, 187–194. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Liu, Y.; Wang, J.; Gascon, J.; Li, J.; van der Bruggen, B. Metal-Organic Frameworks Based Membranes for Liquid Separation. Chem. Soc. Rev. 2017, 46, 7124–7144. [Google Scholar] [CrossRef] [PubMed]
- Razali, M.; Kim, J.F.; Attfield, M.; Budd, P.M.; Drioli, E.; Lee, Y.M.; Szekely, G. Sustainable Wastewater Treatment and Recycling in Membrane Manufacturing. Green Chem. 2015, 17, 5196–5205. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Tian, M.; Bi, W.; Row, K.H. Application of Ionic Liquids in High Performance Reversed-Phase Chromatography. Int. J. Mol. Sci. 2009, 10, 2591–2610. [Google Scholar] [CrossRef]
- Wang, J.; Luo, J.; Feng, S.; Li, H.; Wan, Y.; Zhang, X. Recent Development of Ionic Liquid Membranes. Green Energy Environ. 2016, 1, 43–61. [Google Scholar] [CrossRef] [Green Version]
- Yan, X.; Anguille, S.; Bendahan, M.; Moulin, P. Ionic Liquids Combined with Membrane Separation Processes: A Review. Sep. Purif. Technol. 2019, 222, 230–253. [Google Scholar] [CrossRef]
- ul Mustafa, M.Z.; bin Mukhtar, H.; Md Nordin, N.A.H.; Mannan, H.A.; Nasir, R.; Fazil, N. Recent Developments and Applications of Ionic Liquids in Gas Separation Membranes. Chem. Eng. Technol. 2019, 42, 2580–2593. [Google Scholar] [CrossRef]
- Foong, C.Y.; Wirzal, M.D.H.; Bustam, M.A. A Review on Nanofibers Membrane with Amino-Based Ionic Liquid for Heavy Metal Removal. J. Mol. Liq. 2020, 297, 111793. [Google Scholar] [CrossRef]
- Dai, Z.; Noble, R.D.; Gin, D.L.; Zhang, X.; Deng, L. Combination of Ionic Liquids with Membrane Technology: A New Approach for CO2 Separation. J. Memb. Sci. 2016, 497, 1–20. [Google Scholar] [CrossRef]
- Tan, X.M.; Rodrigue, D. A Review on Porous Polymeric Membrane Preparation. Part I: Production Techniques with Polysulfone and Poly (Vinylidene Fluoride). Polymers 2019, 11, 1160. [Google Scholar] [CrossRef] [Green Version]
- Ayaz, M.; Muhammad, A.; Younas, M.; Khan, A.L.; Rezakazemi, M. Enhanced Water Flux by Fabrication of Polysulfone/Alumina Nanocomposite Membrane for Copper(Ii) Removal. Macromol. Res. 2019, 27, 565–571. [Google Scholar] [CrossRef]
- Ursino, C.; Castro-Muñoz, R.; Drioli, E.; Gzara, L.; Albeirutty, M.H.; Figoli, A. Progress of Nanocomposite Membranes for Water Treatment. Membranes 2018, 8, 18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Filimon, A.; Avram, E.; Dunca, S. Surface and Interface Properties of Functionalized Polysulfones: Cell-Material Interaction and Antimicrobial Activity. Polym. Eng. Sci. 2015, 55, 2184–2194. [Google Scholar] [CrossRef]
- Miculescu, M.; Thakur, V.K.; Miculescu, F.; Voicu, S.I. Graphene-Based Polymer Nanocomposite Membranes: A Review. Polym. Adv. Technol. 2016, 27, 844–859. [Google Scholar] [CrossRef]
- Perreault, F.; Tousley, M.E.; Elimelech, M. Thin-Film Composite Polyamide Membranes Functionalized with Biocidal Graphene Oxide Nanosheets. Environ. Sci. Technol. Lett. 2014, 1, 71–76. [Google Scholar] [CrossRef]
- Richards, H.L.; Baker, P.G.L.; Iwuoha, E. Metal Nanoparticle Modified Polysulfone Membranes for Use in Wastewater Treatment: A Critical Review. J. Surf. Eng. Mater. Adv. Technol. 2012, 2, 183–193. [Google Scholar] [CrossRef] [Green Version]
- Ioan, S.; Filimon, A.; Avram, E. Conformational and Viscometric Behavior of Quaternized Polysulfones in Dilute Solution. Polym. Eng. Sci. 2006, 46, 827–836. [Google Scholar] [CrossRef]
- Filimon, A.; Avram, E.; Ioan, S. Influence of Mixed Solvents and of Temperature on the Solution Properties of Quaternized Polysulfone. J. Macromol. Sci. Part B 2007, 46, 503–520. [Google Scholar] [CrossRef]
- Filimon, A.; Albu, R.M.; Avram, E.; Ioan, S. Effect of Alkil Side Chain on The Conformational Properties of Polysulfone with Quaternary Groups. J. Macromol. Sci. Part B 2010, 49, 207–217. [Google Scholar] [CrossRef]
- Filimon, A.; Avram, E.; Dunca, S.; Stoica, I.; Ioan, S. Surface Properties and Antibacterial Activity of Quaternized Polysulfones. J. Appl. Polym. Sci. 2009, 112, 1808–1816. [Google Scholar] [CrossRef]
- Filimon, A.; Stoica, I.; Onofrei, M.D.; Bargan, A.; Dunca, S. Quaternized Polysulfones-Based Blends: Surface Properties and Performance in Life Quality and Environmental Applications. Polym. Test. 2018, 71, 285–295. [Google Scholar] [CrossRef]
- Avram, E. Polymers with Pendent Functional Groups. VI. A Comparative Study on The Chloromethylation of Linear Polystyrene and Polysulfone with Paraformaldehyde/Me3SiCl. Polym. Plast. Technol. Eng. 2001, 40, 275–281. [Google Scholar] [CrossRef]
- Luca, C.; Avram, E.; Petrariu, I. Quaternary Ammonium Polyelectrolytes. V. Amination Studies of Chloromethylated Polystyrene with N,N-Dimethylalkylamines. J. Macromol. Sci. Part A 1988, 25, 345–361. [Google Scholar] [CrossRef]
- Bhadra, B.N.; Seo, P.W.; Jhung, S.H. Adsorption of Diclofenac Sodium from Water Using Oxidized Activated Carbon. Chem. Eng. J. 2016, 301, 27–34. [Google Scholar] [CrossRef]
- Yu, H.; Nie, E.; Xu, J.; Yan, S.W.; Cooper, W.J.; Song, W.H. Degradation of Diclofenac by Advanced Oxidation and Reduction Processes: Kinetic Studies, Degradation Pathways and Toxicity Assessments. Water Res. 2013, 47, 1908–1918. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Chen, N.; Li, L.; Wang, Q.; Duan, W. Ionic Liquid Modified Poly(Vinyl Alcohol) with Improved Thermal Processability and Excellent Electrical Conductivity. Ind. Eng. Chem. Res. 2018, 57, 5472–5481. [Google Scholar] [CrossRef]
- Graessley, W.W. Polymeric Liquids and Networks: Structure and Properties; Garland Science: New York, NY, USA, 2004. [Google Scholar]
- Daoud, M.; Cotton, J.P.; Farnoux, B.; Jannink, G.; Sarma, G.; Benoit, H.; Dupressix, R.; Picot, C.; de Gennes, P.G. Solutions of Flexible Polymers. Neutron Experiments and Interpretation. Macromolecules 1975, 8, 804–818. [Google Scholar] [CrossRef]
- Abdel-Alim, A.H.; Balke, S.T.; Hamielec, A.E. Flow Properties of Polystyrene Solutions Under High Shear Rates. J. Appl. Polym. Sci. 1973, 17, 1431–1442. [Google Scholar] [CrossRef]
- Witten, T.A.; Cohen, M.H. Crosslinking in Shear-Thickening Ionomers. Macromolecules 1985, 18, 1915–1918. [Google Scholar] [CrossRef]
- Reiner, M. Deformation, Strain and Flow, 2nd ed.; H.K. Lewis & Co. Ltd: London, UK, 1960. [Google Scholar]
- Bauer, W.H.; Collins, E.A. Thixotropy and dilatancy. In Rheology. Theory and Applications; Eirich, F.E., Ed.; Academic Press: New York, NY, USA, 1967; Volume IV, pp. 423–459. [Google Scholar]
- Freundlich, H.; Roder, H.L. Dilatancy and Its Relation To Thixotropy. Trans. Faraday Soc. 1938, 34, 308–316. [Google Scholar] [CrossRef]
- Camargo, D.; Andrade, R.S.; Ferreira, G.A.; Mazzer, H.; Cardozo-Filho, L.; Iglesias, M. Investigation of the Rheological Properties of Protic Ionic Liquids. J. Phys. Org. Chem. 2016, 29, 604–612. [Google Scholar] [CrossRef]
- Moosavi, M.; Daneshvar, A. Investigation of the Rheological Properties of Two Imidazolium-Based Ionic Liquids. J. Mol. Liq. 2014, 190, 59–67. [Google Scholar] [CrossRef]
- Jacquemin, J.; Anouti, M.; Lemordant, D. Physico-Chemical Properties of Non-Newtonian Shear Thickening Diisopropyl-Ethylammonium-Based Protic Ionic Liquids and Their Mixtures with Water and Acetonitrile. J. Chem. Eng. Data 2011, 56, 556–564. [Google Scholar] [CrossRef] [Green Version]
- Gusain, R.; Bakshi, P.S.; Panda, S.; Sharma, O.P.; Gardas, R.; Khatri, O.P. Physicochemical and Tribophysical Properties of Trioctylalkylammonium bis(Salicylato) Borate (N888 N-Bscb) Ionic Liquids: Effect of Alkyl Chain Length. Phys. Chem. Chem. Phys. 2017, 19, 6433–6442. [Google Scholar] [CrossRef] [PubMed]
- Doublier, J.-L.; Wood, P.J. Rheological Properties of Aqueous Solutions of (1->3)(1->4)-P-D-Glucan from Oats (Avena sativa L.). Cereal Chem. 1995, 72, 335–340. [Google Scholar]
- Li, H.; Shen, X.; Gong, G.; Wang, D. Compatibility Studies with Blends Based on Hydroxypropylcellulose and Polyacrylonitrile. Carbohydr. Polym. 2008, 73, 191–200. [Google Scholar] [CrossRef]
- Lu, F.; Cheng, B.; Song, J.; Liang, Y. Rheological Characterization of Concentrated Cellulose Solutions in 1-Allyl-3-Methylimidazolium Chloride. J. Appl. Polym. Sci. 2012, 124, 3419–3425. [Google Scholar] [CrossRef]
- Wang, H.; Gurau, G.; Rogers, R.D. Ionic Liquid Processing of Cellulose. Chem. Soc. Rev. 2012, 41, 1519–1537. [Google Scholar] [CrossRef]
- Scott, M.P.; Rahman, M.; Brazel, C.S. Application of Ionic Liquids as Low-Volatility Plasticizers for PMMA. Eur. Polym. J. 2003, 39, 1947–1953. [Google Scholar] [CrossRef]
- Jayasekara, R.; Harding, I.; Bowater, I.; Christie, G.B.Y.; Lonergan, G.T. Biodegradation by Composting of Surface Modified Starch and PVA Blended Films. J. Polym. Environ. 2003, 11, 49–56. [Google Scholar] [CrossRef]
- Sreedhar, B.; Chattopadhyay, D.K.; Sri Hari Karunakar, M.; Sastry, A.R.K. Thermal and Surface Characterization of Plasticized Starch Polyvinyl Alcohol Blends Crosslinked with Epichlorohydrin. J. Appl. Polym. Sci. 2006, 101, 25–34. [Google Scholar] [CrossRef]
- Urbas, A.M.; Maldovan, M.; DeRege, P.; Thomas, E.L. Bicontinuous Cubic Block Copolymer Photonic Crystals. Adv. Mater. 2002, 14, 1850–1853. [Google Scholar] [CrossRef]
- Tsehaye, M.T.; Velizarov, S.; Van der Bruggen, B. Stability of Polyethersulfone Membranes to Oxidative Agents: A Review. Polym. Degrad. Stab. 2018, 157, 15–33. [Google Scholar] [CrossRef]
- Farrokhara, M.; Dorosti, F. New High Permeable Polysulfone/Ionic Liquid Membrane for Gas Separation. Chin. J. Chem. Eng. 2020, 28, 2301–2311. [Google Scholar] [CrossRef]
- Filimon, A.; Albu, R.M.; Stoica, I.; Avram, E. Blends Based on Ionic Polysulfones with Improved Conformational and Microstructural Characteristics: Perspectives for Biomedical Applications. Compos. B. Eng. 2016, 93, 1–11. [Google Scholar] [CrossRef]
- Lessan, F.; Foudazi, R. Effect of [EMIM][BF4] Ionic Liquid on The Properties of Ultrafiltration Membranes. Polymer 2020, 210, 122977. [Google Scholar] [CrossRef]
- Idris, A.; Man, Z.; Maulud, A.S.; Khan, M.S.; Suetsugu, S. Effects of Phase Separation Behavior on Morphology and Performance of Polycarbonate Membranes. Membranes 2017, 7, 21. [Google Scholar] [CrossRef] [Green Version]
- Keshavarz, L.; Khansary, M.A.; Shirazian, S. Phase Diagram of Ternary Polymeric Solutions Containing Nonsolvent/Solvent/Polymer: Theoretical Calculation and Experimental Validation. Polymer 2015, 73, 1–8. [Google Scholar] [CrossRef]
- Ohlrogge, K.; Ebert, K. Membranen: Grundlagen, Verfahren und Industrielle Anwendungen; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2006. [Google Scholar]
- Drioli, E.; Giorno, L.; Fontananova, E. Comprehensive Membrane Science and Engineering, 2nd ed.; Elsevier Science: Oxford, UK, 2017. [Google Scholar]
- Burrell, G.L.; Dunlop, N.F.; Separovic, F. Non-Newtonian Viscous Shear Thinning in Ionic Liquids. Soft Matter 2010, 6, 2080–2086. [Google Scholar] [CrossRef]
- Rauber, D.; Heib, F.; Schmitt, M.; Hempelmann, R. Trioctylphosphonium Room Temperature Ionic Liquids with Perfluorinated Groups−Physical Properties and Surface Behavior in Comparison with the Nonfluorinated Analogues. Colloids Surf. A Physicochem. Eng. Asp. 2018, 537, 116–125. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Zadhoush, A. Investigation of the Relation between Viscoelastic Properties of Polysulfone Solutions, Phase Inversion Process and Membrane Morphology: The Effect of Solvent Power. J. Membr. Sci. 2017, 532, 47–57. [Google Scholar] [CrossRef]
- Zhang, Z.; An, Q.; Ji, Y.; Qian, J.; Gao, C. Effect of Zero Shear Viscosity of The Casting Solution on the Morphology and Permeability of Polysulfone Membrane Prepared via the Phase-Inversion Process. Desalination 2010, 260, 43–50. [Google Scholar] [CrossRef]
- Vinodhini, P.A.; Sudha, P.N. Removal of Heavy Metal Chromium from Tannery Effluent Using Ultrafiltration Membrane. Text. Cloth. Sustain. 2016, 2, 5. [Google Scholar] [CrossRef] [Green Version]
- Li, J.F.; Xu, Z.L.; Yang, H.; Yu, L.Y.; Liu, M. Effect of TiO2 Nanoparticles on the Surface Morphology and Performance of Microporous PES Membrane. Appl. Surf. Sci. 2009, 255, 4725–4732. [Google Scholar] [CrossRef]
- Dorra, E.J.; Mourad, B.S.; Mahmoud, D. Retention of Cadmium and Zinc from Aqueous Solutions by Poly(Acrylic Acid)-Assisted Ultrafiltration. Int. J. Chem. React. 2010, 8, A133. [Google Scholar] [CrossRef]
- Khalil, E.; Sallam, A. Interaction of Two Diclofenac Acid Salts with Copolymers of Ammoniomethacrylate: Effect of Additives and Release Profiles. Drug Dev. Ind. Pharm. 1999, 25, 419–427. [Google Scholar] [CrossRef]
- Jesus, A.R.; Raposo, L.R.; Soromenho, M.R.C.; Agostinho, D.A.S.; Esperança, J.M.S.S.; Baptista, P.V.; Fernandes, A.R.; Reis, P.M. New Non-Toxic N-Alkyl Cholinium-Based Ionic Liquids as Excipients to Improve the Solubility of Poorly Water-Soluble Drugs. Symmetry 2021, 13, 2053. [Google Scholar] [CrossRef]
- Filimon, A.; Dobos, A.M.; Avram, E. Ionic Transport Processes in Polymer Mixture Solutions Based on Quaternized Polysulfones. J. Chem. Thermodyn. 2017, 106, 160–167. [Google Scholar] [CrossRef]
- Filimon, A.; Avram, E.; Stoica, I. Rheological and Morphological Characteristics of Multicomponent Polysulfone/Poly(Vinyl Alcohol) Systems. Polym. Int. 2014, 63, 1856–1868. [Google Scholar] [CrossRef]



| System | Mixing Ratio (wt%/wt%) | Jw (L·m−2·h−1) | w1 (g) | w2 (g) | WC (%) | ɛ (%) |
|---|---|---|---|---|---|---|
| Equations | ||||||
| PSFQ/ Cyphos IL 101 | 0 | 1529 | 0.0232 | 0.0308 | 24.7 | 2.43 |
| 3 | 1592 | 0.0228 | 0.0319 | 28.5 | 2.90 | |
| 5 | 1698 | 0.0245 | 0.0615 | 60.2 | 11.8 | |
| PSFQ/ Aliquat 336 | 0 | 1529 | 0.0232 | 0.0308 | 24.7 | 2.43 |
| 3 | 1663 | 0.0288 | 0.0492 | 41.5 | 6.51 | |
| 5 | 1779 | 0.0242 | 0.0636 | 61.9 | 12.6 | |
| 10 | 1819 | 0.0251 | 0.0689 | 63.6 | 13.9 | |
| 15 | 1930 | 0.0254 | 0.0760 | 66.5 | 16.2 |
| Polymer | |
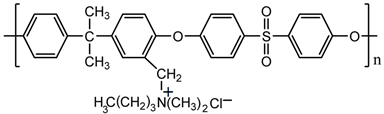 | PSFQ |
| Ionic Liquids | |
 | Cyphos IL 101 |
 | Aliquat 336 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Filimon, A.; Dobos, A.M.; Dumbrava, O.; Doroftei, F.; Lupa, L. Green Blends Based on Ionic Liquids with Improved Performance for Membrane Technology: Perspectives for Environmental Applications. Int. J. Mol. Sci. 2022, 23, 7961. https://doi.org/10.3390/ijms23147961
Filimon A, Dobos AM, Dumbrava O, Doroftei F, Lupa L. Green Blends Based on Ionic Liquids with Improved Performance for Membrane Technology: Perspectives for Environmental Applications. International Journal of Molecular Sciences. 2022; 23(14):7961. https://doi.org/10.3390/ijms23147961
Chicago/Turabian StyleFilimon, Anca, Adina Maria Dobos, Oana Dumbrava, Florica Doroftei, and Lavinia Lupa. 2022. "Green Blends Based on Ionic Liquids with Improved Performance for Membrane Technology: Perspectives for Environmental Applications" International Journal of Molecular Sciences 23, no. 14: 7961. https://doi.org/10.3390/ijms23147961






