Shifting Paradigms in TNBC Treatment: Emerging Alternatives to Capecitabine in the Post-Neoadjuvant Setting
Abstract
1. Introduction
2. Capecitabine in TNBC Residual Disease after NACT
3. Immunotherapy and PARP Inhibitors Era
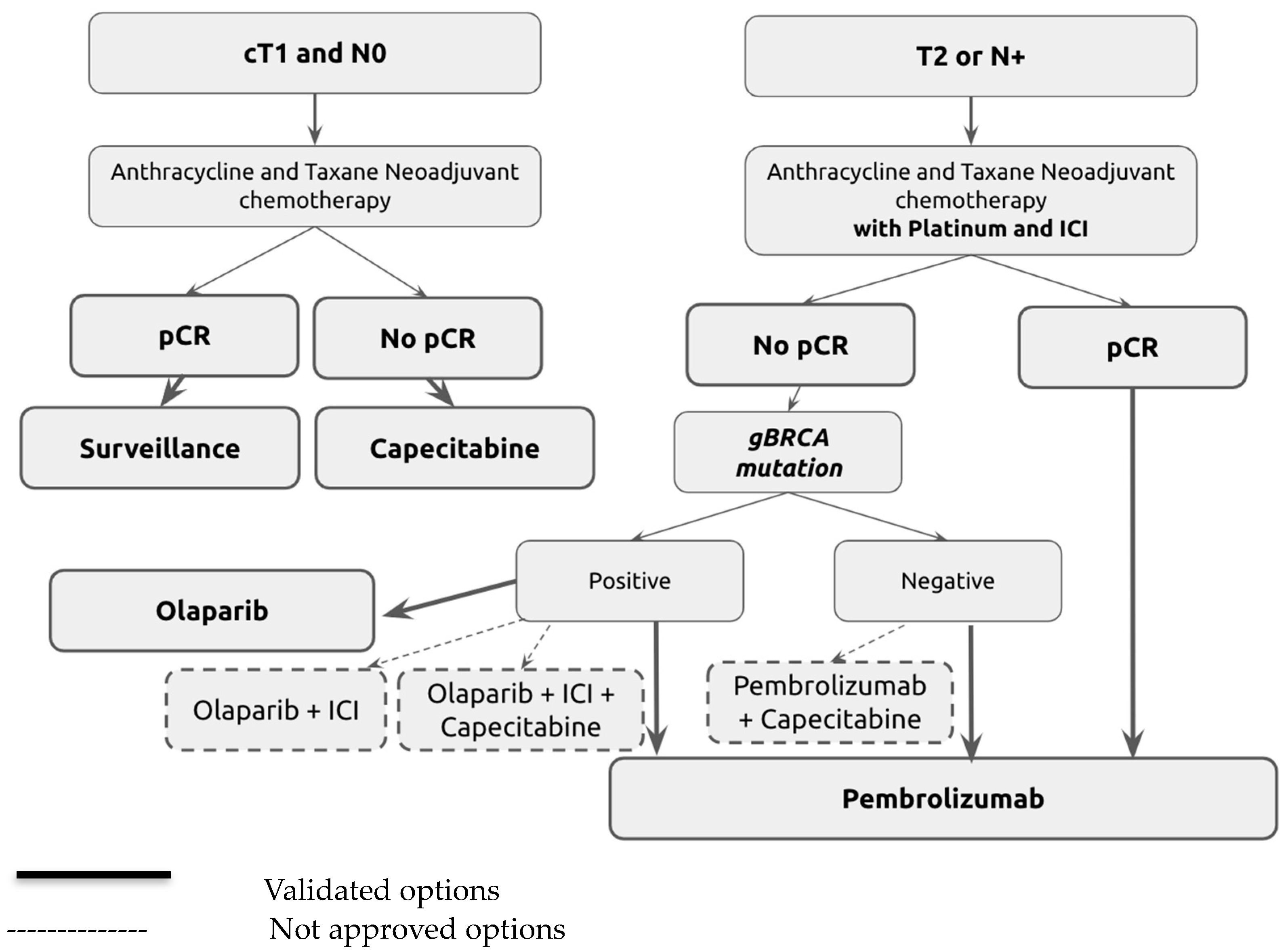
3.1. Immunotherapy in TNBC Residual Disease
3.2. PARP Inhibitors in TNBC Residual Disease
4. Future Opportunities with ADCs
5. Socio-Economic Challenges with New Therapies
Recommendations
6. Conclusions
Chemotherapy Regimens
- Anthracycline and Taxane Neoadjuvant Chemotherapy: four cycles of dose-dense AC60 or EC90 (doxorubicin 60 mg/m2 or epirubicin 90 mg/m2, plus cyclophosphamide 600 mg/m2) every 14 days followed by 12 weekly paclitaxel 80 mg/m2.
- Anthracycline and Taxane Neoadjuvant Chemotherapy with Platinum and ICIs (KN522 Protocol): Weekly paclitaxel (80 mg/m2) and carboplatin (AUC five every 3 weeks, or 1.5 once weekly) for the first 12 weeks, plus four cycles of pembrolizumab (200 mg) followed by four cycles of doxorubicin (60 mg/m2) or epirubicin (90 mg/m2), plus cyclophosphamide (600 mg/m2) administered once every 3 weeks and four cycles of pembrolizumab (200 mg).
- Adjuvant Capecitabine: Oral capecitabine at a dose of 1250 mg/m2, twice per day, on days 1 to 14 every 3 weeks for six or eight cycles.
- Adjuvant Olaparib: Oral olaparib 300 mg twice per day for 1 year.
- Adjuvant Pembrolizumab: Pembrolizumab at a dose of 200 mg for nine cycles.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| TNBC | Triple-negative breast cancer |
| NACT | Neoadjuvant chemotherapy |
| pCR | Pathological complete response |
| DFS | Disease-free survival |
| MBC | Metastatic breast cancer |
| PARPi | Poly (ADP-ribose) polymerase inhibitor |
| ICI | Immune checkpoint inhibitor |
| Dato-DXd | Datopotamab deruxtecan |
| AUC | Area under the curve |
| SG | Sacituzumab govitecan |
| T-DXd | Trastuzumab deruxtecan |
References
- Harbeck, N.; Gnant, M. Breast cancer. Lancet 2017, 389, 1134–1150. [Google Scholar] [CrossRef] [PubMed]
- von Minckwitz, G.; Untch, M.; Blohmer, J.U.; Costa, S.D.; Eidtmann, H.; Fasching, P.A.; Gerber, B.; Eiermann, W.; Hilfrich, J.; Huober, J.; et al. Definition and impact of pathologic complete response on prognosis after neoadjuvant chemotherapy in various intrinsic breast cancer subtypes. J. Clin. Oncol. 2012, 30, 1796–1804. [Google Scholar] [CrossRef] [PubMed]
- Cortazar, P.; Zhang, L.; Untch, M.; Mehta, K.; Costantino, J.P.; Wolmark, N.; Bonnefoi, H.; Cameron, D.; Gianni, L.; Valagussa, P.; et al. Pathological complete response and long-term clinical benefit in breast cancer: The CTNeoBC pooled analysis. Lancet 2014, 384, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Loibl, S.; André, F.; Bachelot, T.; Barrios, C.H.; Bergh, J.; Burstein, H.J.; Cardoso, M.J.; Carey, L.A.; Dawood, S.; Del Mastro, L.; et al. Early breast cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann. Oncol. 2024, 35, 159–182. [Google Scholar] [CrossRef] [PubMed]
- Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Increasing the dose intensity of chemotherapy by more frequent administration or sequential scheduling: A patient-level meta-analysis of 37,298 women with early breast cancer in 26 randomised trials. Lancet 2019, 393, 1440–1452. [Google Scholar] [CrossRef] [PubMed]
- Schmid, P.; Cortes, J.; Pusztai, L.; McArthur, H.; Kümmel, S.; Bergh, J.; Denkert, C.; Park, Y.H.; Hui, R.; Harbeck, N.; et al. Pembrolizumab for Early Triple-Negative Breast Cancer. N. Engl. J. Med. 2020, 382, 810–821. [Google Scholar] [CrossRef] [PubMed]
- Poggio, F.; Bruzzone, M.; Ceppi, M.; Pondé, N.F.; La Valle, G.; Del Mastro, L.; de Azambuja, E. Platinum-based neoadjuvant chemotherapy in triple-negative breast cancer: A systematic review and meta-analysis. Ann. Oncol. 2018, 29, 1497–1508. [Google Scholar] [CrossRef] [PubMed]
- Masuda, N.; Lee, S.-J.; Ohtani, S.; Im, Y.-H.; Lee, E.-S.; Yokota, I.; Kuroi, K.; Im, S.-A.; Park, B.-W.; Kim, S.-B.; et al. Adjuvant Capecitabine for Breast Cancer after Preoperative Chemotherapy. N. Engl. J. Med. 2017, 376, 2147–2159. [Google Scholar] [CrossRef]
- Kuroi, K.; Toi, M.; Ohno, S.; Nakamura, S.; Iwata, H.; Masuda, N.; Sato, N.; Tsuda, H.; Kurosumi, M.; Akiyama, F. Prognostic significance of subtype and pathologic response in operable breast cancer; a pooled analysis of prospective neoadjuvant studies of JBCRG. Breast Cancer 2013, 22, 486–495. [Google Scholar] [CrossRef]
- von Minckwitz, G.; Schneeweiss, A.; Loibl, S.; Salat, C.; Denkert, C.; Rezai, M.; Blohmer, J.U.; Jackisch, C.; Paepke, S.; Gerber, B.; et al. Neoadjuvant carboplatin in patients with triple-negative and HER2-positive early breast cancer (GeparSixto; GBG 66): A randomised phase 2 trial. Lancet Oncol. 2014, 15, 747–756. [Google Scholar] [CrossRef]
- Sikov, W.M.; Berry, D.A.; Perou, C.M.; Singh, B.; Cirrincione, C.T.; Tolaney, S.M.; Kuzma, C.S.; Pluard, T.J.; Somlo, G.; Port, E.R.; et al. Impact of the Addition of Carboplatin and/or Bevacizumab to Neoadjuvant Once-per-Week Paclitaxel Followed by Dose-Dense Doxorubicin and Cyclophosphamide on Pathologic Complete Response Rates in Stage II to III Triple-Negative Breast Cancer: CALGB 40603 (Alliance). J. Clin. Oncol. 2015, 33, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Loibl, S.; O’Shaughnessy, J.; Untch, M.; Sikov, W.M.; Rugo, H.S.; McKee, M.D.; Huober, J.; Golshan, M.; von Minckwitz, G.; Maag, D.; et al. Addition of the PARP inhibitor veliparib plus carboplatin or carboplatin alone to standard neoadjuvant chemotherapy in triple-negative breast cancer (BrighTNess): A randomised, phase 3 trial. Lancet Oncol. 2018, 19, 497–509. [Google Scholar] [CrossRef]
- Miles, D.; Minckwitz, G.; Seidman, A.D. Combination Versus Sequential Single-Agent Therapy in Metastatic Breast Cancer. Oncol. 2002, 7, 13–19. [Google Scholar] [CrossRef]
- Lehmann, B.D.; Bauer, J.A.; Chen, X.; Sanders, M.E.; Chakravarthy, A.B.; Shyr, Y.; Pietenpol, J.A. Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J. Clin. Investig. 2011, 121, 2750–2767. [Google Scholar] [CrossRef]
- Mayer, I.A.; Zhao, F.; Arteaga, C.L.; Symmans, W.F.; Park, B.H.; Burnette, B.L.; Tevaarwerk, A.J.; Garcia, S.F.; Smith, K.L.; Makower, D.F.; et al. Randomized Phase III Postoperative Trial of Platinum-Based Chemotherapy Versus Capecitabine in Patients With Residual Triple-Negative Breast Cancer Following Neoadjuvant Chemotherapy: ECOG-ACRIN EA1131. J. Clin. Oncol. 2021, 39, 2539–2551. [Google Scholar] [CrossRef]
- Tutt, A.N.J.; Garber, J.E.; Kaufman, B.; Viale, G.; Fumagalli, D.; Rastogi, P.; Gelber, R.D.; de Azambuja, E.; Fielding, A.; Balmaña, J.; et al. Adjuvant Olaparib for Patients with—or—Mutated Breast Cancer. N. Engl. J. Med. 2021, 384, 2394–2405. [Google Scholar] [CrossRef] [PubMed]
- Bonadio, R.C.; Tarantino, P.; Testa, L.; Punie, K.; Pernas, S.; Barrios, C.; Curigliano, G.; Tolaney, S.M.; Barroso-Sousa, R. Management of patients with early-stage triple-negative breast cancer following pembrolizumab-based neoadjuvant therapy: What are the evidences? Cancer Treat Rev. 2022, 110, 102459. [Google Scholar] [CrossRef]
- Shitara, K.; Van Cutsem, E.; Bang, Y.-J.; Fuchs, C.; Wyrwicz, L.; Lee, K.-W.; Kudaba, I.; Garrido, M.; Chung, H.C.; Lee, J.; et al. Efficacy and Safety of Pembrolizumab or Pembrolizumab Plus Chemotherapy vs Chemotherapy Alone for Patients With First-line, Advanced Gastric Cancer: The KEYNOTE-062 Phase 3 Randomized Clinical Trial. JAMA Oncol. 2020, 6, 1571–1580. [Google Scholar] [CrossRef] [PubMed]
- Janjigian, Y.Y.; Shitara, K.; Moehler, M.; Garrido, M.; Salman, P.; Shen, L.; Wyrwicz, L.; Yamaguchi, K.; Skoczylas, T.; Campos Bragagnoli, A.; et al. First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): A randomised, open-label, phase 3 trial. Lancet. 2021, 398, 27–40. [Google Scholar] [CrossRef]
- Burtness, B.; Harrington, K.J.; Greil, R.; Soulières, D.; Tahara, M.; de Castro, G., Jr.; Psyrri, A.; Basté, N.; Neupane, P.; Bratland, A.; et al. Pembrolizumab alone or with chemotherapy versus cetuximab with chemotherapy for recurrent or metastatic squamous cell carcinoma of the head and neck (KEYNOTE-048): A randomised, open-label, phase 3 study. Lancet 2019, 394, 1915–1928. [Google Scholar] [CrossRef]
- Pusztai, L.; Barlow, W.E.; Ganz, P.A.; Henry, N.L.; White, J.; Jagsi, R. Abstract OT1-02-04: SWOG S1418/NRG-BR006: A randomized, phase III trial to evaluate the efficacy and safety of MK-3475 as adjuvant therapy for triple receptor-negative breast cancer with > 1 cm residual invasive cancer or positive lymph nodes (>pN1mic) after neoadjuvant chemotherapy. Cancer Res. 2018, 78, OT1-02-0. [Google Scholar]
- Khoury, K.; Isaacs, C.; Gatti-Mays, M.; Donahue, R.N.; Schlom, J.; Wang, H.; Gallagher, C.; Graham, D.; Warren, R.; Dilawari, A.; et al. Abstract OT3-04-01: Nivolumab or capecitabine or combination therapy as adjuvant therapy for triple negative breast cancer (TNBC) with residual disease following neoadjuvant chemotherapy: The OXEL study. Cancer Res. 2019, 79, OT3-04. [Google Scholar] [CrossRef]
- Park, I.H.; Kim, G.M.; Kim, J.H.; Kim, H.; Park, K.H.; Park, Y.H.; Baek, S.K.; Sim, S.H.; Ahn, H.K.; Lee, G.-W.; et al. Randomized, phase II trial to evaluate the efficacy and safety of atezolizumab plus capecitabine adjuvant therapy compared to capecitabine monotherapy for triple receptor-negative breast cancer (TNBC) with residual invasive cancer after neoadjuvant chemotherapy (MIRINAE trial, KCSG-BR18-21). J. Clin. Oncol. 2020, 38, TPS597. [Google Scholar] [CrossRef]
- Gonzalez-Angulo, A.M.; Timms, K.M.; Liu, S.; Chen, H.; Litton, J.K.; Potter, J.; Lanchbury, J.S.; Stemke-Hale, K.; Hennessy, B.T.; Arun, B.K.; et al. Incidence and Outcome of BRCA Mutations in Unselected Patients with Triple Receptor-Negative Breast Cancer. Clin. Cancer Res. 2011, 17, 1082–1089. [Google Scholar] [CrossRef] [PubMed]
- Lord, C.J.; Ashworth, A. PARP inhibitors: Synthetic lethality in the clinic. Science 2017, 355, 1152–1158. [Google Scholar] [CrossRef] [PubMed]
- Robson, M.; Im, S.A.; Senkus, E.; Xu, B.; Domchek, S.M.; Masuda, N.; Delaloge, S.; Li, W.; Tung, N.; Armstrong, A.; et al. Olaparib for Metastatic Breast Cancer in Patients with a Germline BRCA Mutation. N. Engl. J. Med. 2017, 377, 523–533. [Google Scholar] [CrossRef] [PubMed]
- Litton, J.K.; Rugo, H.S.; Ettl, J.; Hurvitz, S.A.; Gonçalves, A.; Lee, K.-H.; Fehrenbacher, L.; Yerushalmi, R.; Mina, L.A.; Martin, M.; et al. Talazoparib in Patients with Advanced Breast Cancer and a Germline BRCA Mutation. N. Engl. J. Med. 2018, 379, 753–763. [Google Scholar] [CrossRef] [PubMed]
- Pantelidou, C.; Sonzogni, O.; De Oliveria Taveira, M.; Mehta, A.K.; Kothari, A.; Wang, D.; Visal, T.; Li, M.K.; Pinto, J.; Castrillon, J.A.; et al. PARP Inhibitor Efficacy Depends on CD8+ T-cell Recruitment via Intratumoral STING Pathway Activation in BRCA-Deficient Models of Triple-Negative Breast Cancer. Cancer Discov. 2019, 9, 722–737. [Google Scholar] [CrossRef] [PubMed]
- Vinayak, S.; Tolaney, S.M.; Schwartzberg, L.; Mita, M.; McCann, G.; Tan, A.R.; Wahner-Hendrickson, A.E.; Forero, A.; Anders, C.; Wulf, G.M.; et al. Open-label Clinical Trial of Niraparib Combined With Pembrolizumab for Treatment of Advanced or Metastatic Triple-Negative Breast Cancer. JAMA Oncol. 2019, 5, 1132–1140. [Google Scholar] [CrossRef]
- Domchek, S.M.; Postel-Vinay, S.; Im, S.-A.; Park, Y.H.; Delord, J.-P.; Italiano, A.; Alexandre, J.; You, B.; Bastian, S.; Krebs, M.G.; et al. Olaparib and durvalumab in patients with germline BRCA-mutated metastatic breast cancer (MEDIOLA): An open-label, multicentre, phase 1/2, basket study. Lancet Oncol. 2020, 21, 1155–1164. [Google Scholar] [CrossRef]
- Pusztai, L.; Yau, C.; Wolf, D.M.; Han, H.S.; Du, L.; Wallace, A.M.; String-Reasor, E.; Boughey, J.C.; Chien, A.J.; Elias, A.D. Durvalumab with olaparib and paclitaxel for high-risk Her2-negative stage II/III breast cancer: Results from the adaptively randomized I-SPY2 trial. Cancer Cell 2021, 39, 989–998.e5. [Google Scholar]
- Chang, H.L.; Schwettmann, B.; McArthur, H.L.; Chan, I.S. Antibody-drug conjugates in breast cancer: Overcoming resistance and boosting immune response. J. Clin. Investig. 2023, 133, e172156. [Google Scholar] [CrossRef]
- Von Minckwitz, G.; Huang, C.-S.; Mano, M.S.; Loibl, S.; Mamounas, E.P.; Untch, M.; Wolmark, N.; Rastogi, P.; Schneeweiss, A.; Redondo, A.; et al. Trastuzumab Emtansine for Residual Invasive HER2-Positive Breast Cancer. N. Engl. J. Med. 2019, 380, 617–628. [Google Scholar] [CrossRef] [PubMed]
- Bardia, A.; Tolaney, S.M.; Punie, K.; Loirat, D.; Oliveira, M.; Kalinsky, K.; Zelnak, A.; Aftimos, P.; Dalenc, F.; Sardesai, S.; et al. Biomarker analyses in the phase III ASCENT study of sacituzumab govitecan versus chemotherapy in patients with metastatic triple-negative breast cancer. Ann. Oncol. 2021, 32, 1148–1156. [Google Scholar] [CrossRef] [PubMed]
- Modi, S.; Jacot, W.; Yamashita, T.; Sohn, J.; Vidal, M.; Tokunaga, E.; Tsurutani, J.; Ueno, N.T.; Prat, A.; Chae, Y.S.; et al. Trastuzumab Deruxtecan in Previously Treated HER2-Low Advanced Breast Cancer. N. Engl. J. Med. 2022, 387, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Tarantino, P.; Hamilton, E.; Tolaney, S.M.; Cortes, J.; Morganti, S.; Ferraro, E.; Marra, A.; Viale, G.; Trapani, D.; Cardoso, F.; et al. HER2-Low Breast Cancer: Pathological and Clinical Landscape. J. Clin. Oncol. 2020, 38, 1951–1962. [Google Scholar] [CrossRef] [PubMed]
- Yip, C.H.; Evans, D.G.; Agarwal, G.; Buccimazza, I.; Kwong, A.; Morant, R.; Prakash, I.; Song, C.Y.; Taib, N.A.; Tausch, C.; et al. Global Disparities in Breast Cancer Genetics Testing, Counselling and Management. World J. Surg. 2019, 43, 1264–1270. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, F.R.; Zaric, B.; Rabea, A.; Thongprasert, S.; Lertprasertsuke, N.; Dalurzo, M.L.; Marileila Varella-Garcia. Biomarker Testing for Personalized Therapy in Lung Cancer in Low- and Middle-Income Countries. Am. Soc. Clin. Oncol. Educ. Book 2017, 37, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Cortes, J.; Perez-García, J.M.; Llombart-Cussac, A.; Curigliano, G.; El Saghir, N.S.; Cardoso, F.; Barrios, C.H.; Wagle, S.; Roman, J.; Harbeck, N.; et al. Enhancing global access to cancer medicines. CA A Cancer J. Clin. 2020, 70, 105–124. [Google Scholar] [CrossRef]
- Ruff, P.; Al-Sukhun, S.; Blanchard, C.; Shulman, L.N. Access to Cancer Therapeutics in Low- and Middle-Income Countries. Am. Soc. Clin. Oncol. Educ. Book 2016, 58–65. [Google Scholar] [CrossRef]
- Docteur, E. Towards a New Vision for Shared Responsibility in Pharmaceutical Pricing, Coverage and Reimbursement: Policy Approaches Building on Principles of Solidarity, Transparency and Sustainability; World Health Organization: Copenhagen, Denmark, 2022. [Google Scholar]
| Study Title | Trial Phase | Population | Treatment Arms | Primary Outcome |
|---|---|---|---|---|
| SWOG S1418/NRG-BR006 | Phase III | TNBC with residual disease after NACT. Residual disease is defined as ≥1 cm residual invasive carcinoma in the breast or positive lymph nodes (ypN1mi-3). | Adjuvant pembrolizumab for 1 year Observation Capecitabine was used in the adjuvant setting with pembrolizumab initiation after capecitabine completion. | Invasive DFS |
| OXEL trial (NCT03487666) | Phase II | TNBC with residual disease after NACT. Residual disease is defined as ≥1 cm residual invasive carcinoma in the breast or macroscopically positive lymph nodes. | Nivo Capecitabine Nivo + capecitabine | Changes in a peripheral immunoscore at week 6 |
| MIRINAE trial (NCT03756298) | Phase II | TNBC with residual disease after NACT. Residual disease is defined as ≥1 cm residual invasive carcinoma in the breast or macroscopically positive lymph nodes. | Atez + capecitabine Capecitabine | Invasive DFS |
| BreastImmune03, NCT03818685 | Phase II | TNBC with residual disease after NACT. Residual disease is defined as an RCB score of II or III. | Nivolumab + Ipilimumab + radiotherapy Capecitabine + radiotherapy | DFS |
| Study Title | Trial Phase | Population | Treatment Arms | Primary Outcome |
|---|---|---|---|---|
| SASCIA trial (NCT04595565) | Phase III | Her2-negative BC with residual disease after NACT. * Residual disease is defined as follows: (1) macroscopic residual disease in the primary tumor (ypT > 1 mm) for TNBC; (2) CPS + EG ≥ 3 or CPS + EG 2 and ypN+ for HR + BC. | Sacituzumab govitecan. Treatment of physician’s choice (capecitabine or platinum-based chemotherapy). | Invasive DFS |
| ASCENT-05, NCT05633654 | Phase III | Residual invasive disease after surgery and neoadjuvant therapy. | Sacituzumab govitecan + pembrolizumab. Treatment of physician’s choice (pembrolizumab or pembrolizumab + capecitabine). | Invasive DFS |
| TROPION-Breast03 NCT05629585 | Phase III | Stage I-III triple-negative breast cancer without pathological complete response following neoadjuvant therapy. | Dato-DXd, with or without Durvalumab. Investigator’s choice of therapy (capecitabine, pembrolizumab, or pembrolizumab + capecitabine) | iDFS for Dato-DXd + durvalumab vs. ICT |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abahssain, H.; Souadka, A.; Alem, R.; Santoni, M.; Battelli, N.; Amela, E.; Lemaire, A.; Rodriguez, J.; Errihani, H. Shifting Paradigms in TNBC Treatment: Emerging Alternatives to Capecitabine in the Post-Neoadjuvant Setting. Curr. Oncol. 2024, 31, 3771-3782. https://doi.org/10.3390/curroncol31070278
Abahssain H, Souadka A, Alem R, Santoni M, Battelli N, Amela E, Lemaire A, Rodriguez J, Errihani H. Shifting Paradigms in TNBC Treatment: Emerging Alternatives to Capecitabine in the Post-Neoadjuvant Setting. Current Oncology. 2024; 31(7):3771-3782. https://doi.org/10.3390/curroncol31070278
Chicago/Turabian StyleAbahssain, Halima, Amine Souadka, Rania Alem, Matteo Santoni, Nicola Battelli, Eric Amela, Antoine Lemaire, Joseph Rodriguez, and Hassan Errihani. 2024. "Shifting Paradigms in TNBC Treatment: Emerging Alternatives to Capecitabine in the Post-Neoadjuvant Setting" Current Oncology 31, no. 7: 3771-3782. https://doi.org/10.3390/curroncol31070278
APA StyleAbahssain, H., Souadka, A., Alem, R., Santoni, M., Battelli, N., Amela, E., Lemaire, A., Rodriguez, J., & Errihani, H. (2024). Shifting Paradigms in TNBC Treatment: Emerging Alternatives to Capecitabine in the Post-Neoadjuvant Setting. Current Oncology, 31(7), 3771-3782. https://doi.org/10.3390/curroncol31070278







