Adjuvant ICIs Plus Targeted Therapies Reduce HCC Recurrence after Hepatectomy in Patients with High Risk of Recurrence
Abstract
1. Introduction
2. Methods
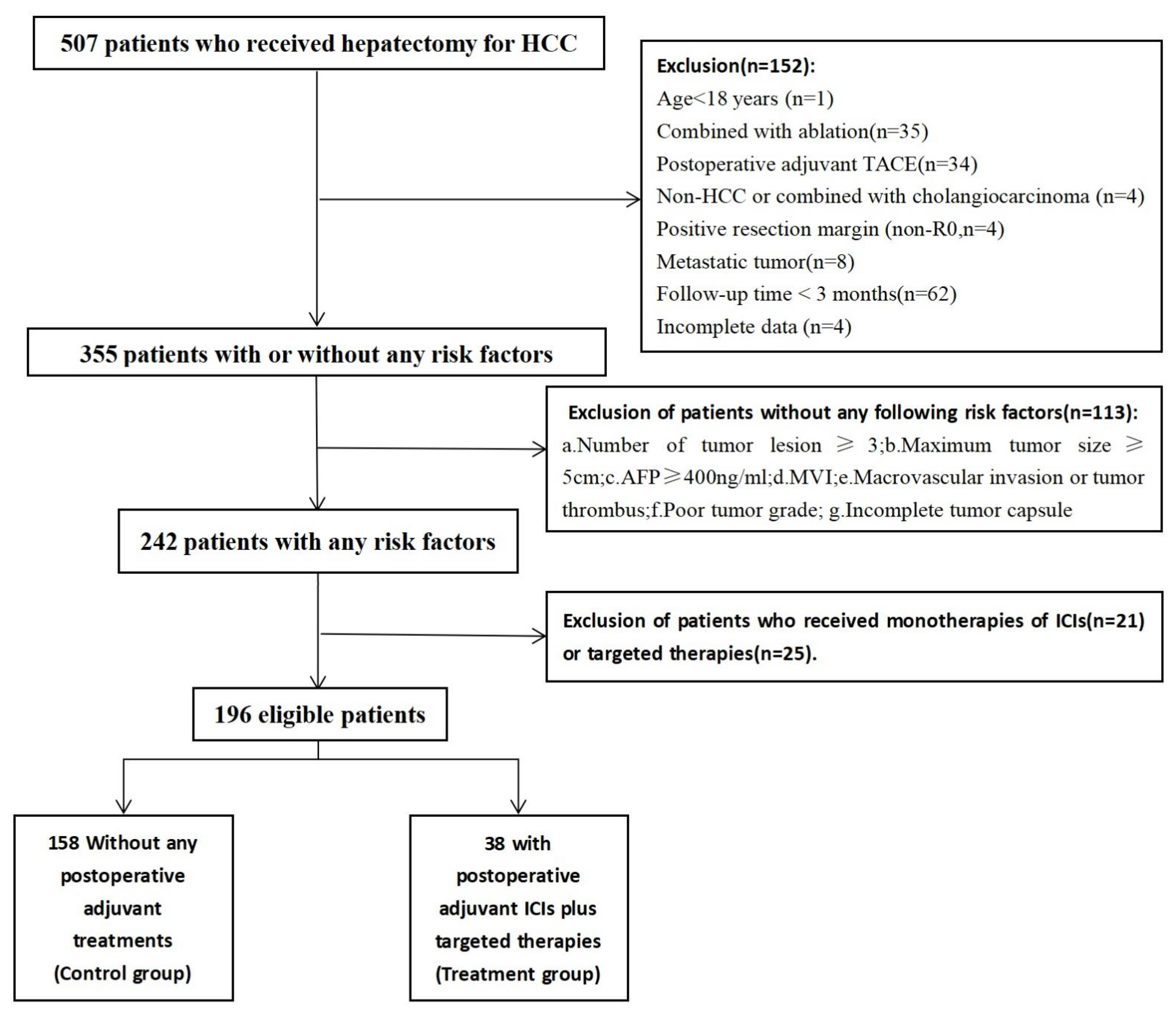
2.1. Study Population
2.2. Treatments
2.3. Outcomes and Follow-Up Assessment
2.4. Statistical Methods
3. Results
3.1. Baseline Characteristics and Follow-Up Status
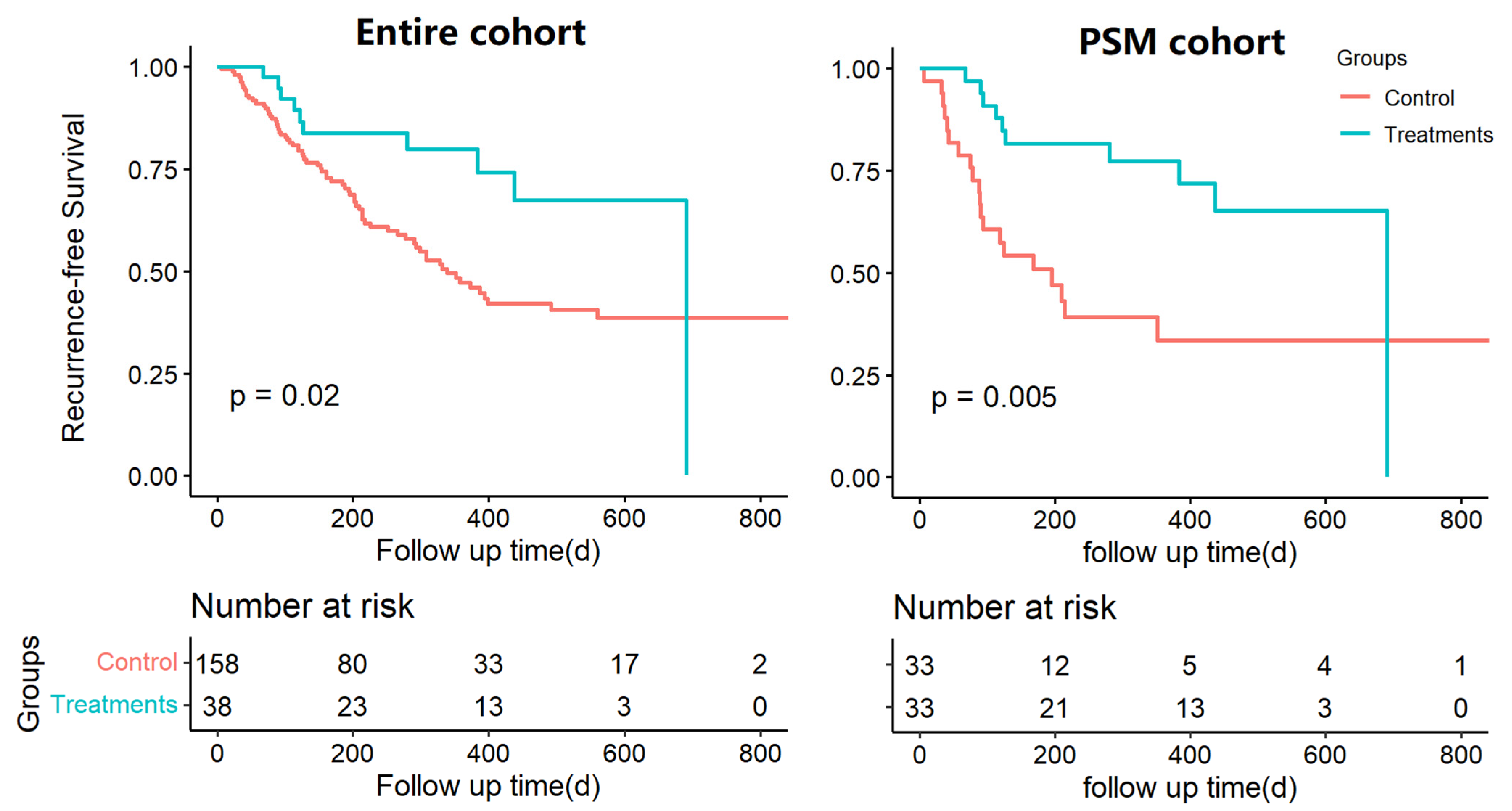
3.2. Survival Analysis of Postoperative Adjuvant Treatments in the Overall and Propensity-Matched Cohorts
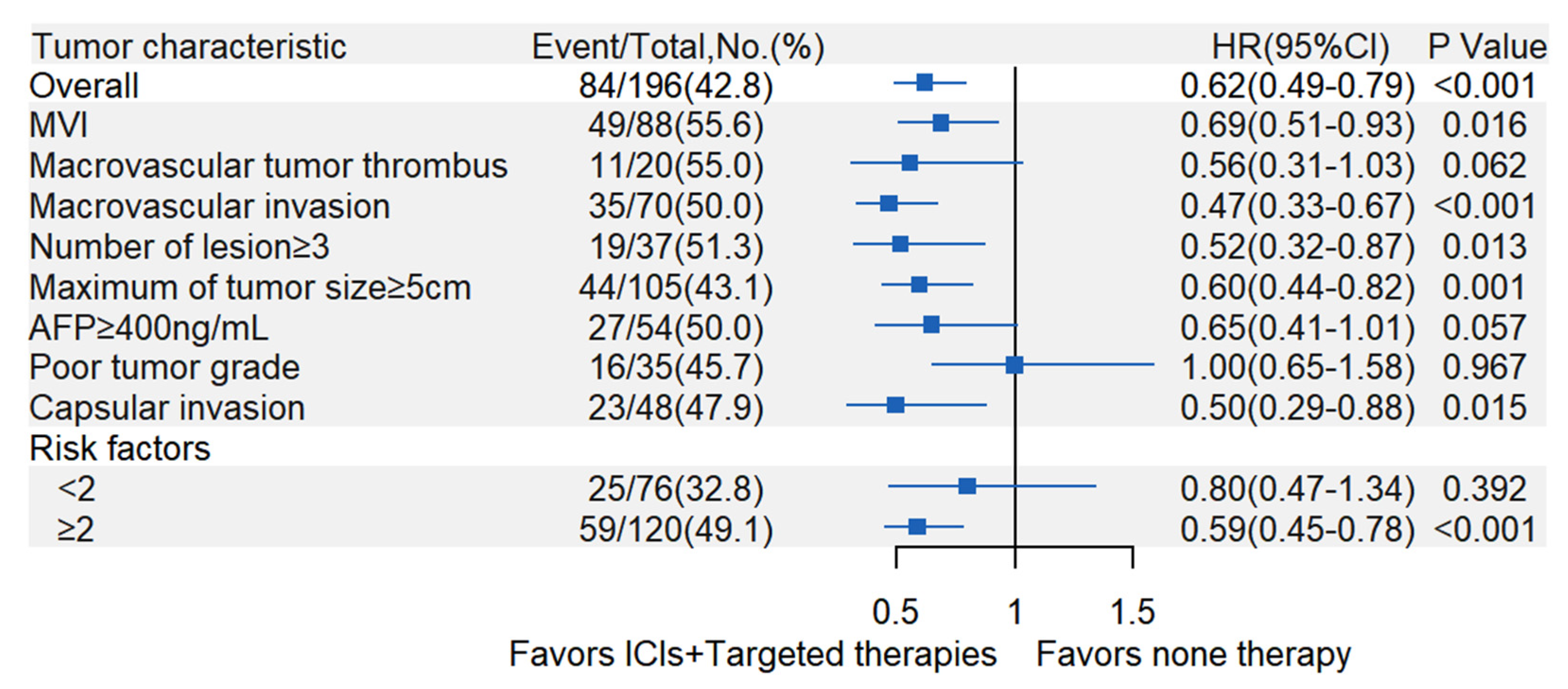
3.3. Exploratory Subgroup Analyses of Postoperative Adjuvant ICIs Plus Targeted Therapies
3.4. Safety of Postoperative Adjuvant Therapies
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lim, K.; Chow, P.; Allen, J.; Siddiqui, F.J.; Chan, E.S.; Tan, S. Systematic review of outcomes of liver resection for early hepatocellular carcinoma within the Milan criteria. Br. J. Surg. 2012, 99, 1622–1629. [Google Scholar] [CrossRef]
- Belghiti, J.; Panis, Y.; Faroes, O.; Benhamou, J.P.; Fekete, F. Intrahepatic Recurrence After Resection of Hepatocellular Carcinoma Complicating Cirrhosis. Ann. Surg. 1991, 214, 114–117. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver. European Organisation for Research and Treatment of Cancer. EASL–EORTC clinical practice guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2012, 56, 908–943. [Google Scholar] [CrossRef] [PubMed]
- Li, W.-F.; Yen, Y.-H.; Liu, Y.-W.; Wang, C.-C.; Yong, C.-C.; Lin, C.-C.; Cheng, Y.-F.; Wang, J.-H.; Lu, S.-N. Preoperative predictors of early recurrence after resection for hepatocellular carcinoma. Am. J. Surg. 2022, 223, 945–950. [Google Scholar] [CrossRef] [PubMed]
- Xia, W.; Peng, T.; Guan, R.; Zhou, Y.; Zeng, C.; Lin, Y.; Wu, Z.; Tan, H. Development of a novel prognostic nomogram for the early recurrence of liver cancer after curative hepatectomy. Ann. Transl. Med. 2021, 9, 1541. [Google Scholar] [CrossRef] [PubMed]
- Xing, H.; Zhang, W.-G.; Cescon, M.; Liang, L.; Li, C.; Wang, M.-D.; Wu, H.; Lau, W.Y.; Zhou, Y.-H.; Gu, W.-M.; et al. Defining and predicting early recurrence after liver resection of hepatocellular carcinoma: A multi-institutional study. Hpb 2020, 22, 677–689. [Google Scholar] [CrossRef]
- Poon, R.T.-P.; Fan, S.-T.; Wong, J. Risk Factors, Prevention, and Management of Postoperative Recurrence After Resection of Hepatocellular Carcinoma. Ann. Surg. 2000, 232, 10–24. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Jian, P.-E.; Li, S.-H.; Guo, Z.-X.; Zhang, Y.-F.; Ling, Y.-H.; Lin, X.-J.; Xu, L.; Shi, M.; Zheng, L.; et al. Adjuvant transcatheter arterial chemoembolization after curative resection for hepatocellular carcinoma patients with solitary tumor and microvascular invasion: A randomized clinical trial of efficacy and safety. Cancer Commun. 2018, 38, 61. [Google Scholar] [CrossRef]
- Wang, Z.; Ren, Z.; Chen, Y.; Hu, J.; Yang, G.; Yu, L.; Yang, X.; Huang, A.; Zhang, X.; Zhou, S.; et al. Adjuvant Transarterial Chemoembolization for HBV-Related Hepatocellular Carcinoma After Resection: A Randomized Controlled Study. Clin. Cancer Res. 2018, 24, 2074–2081. [Google Scholar] [CrossRef]
- Chen, W.; Ma, T.; Zhang, J.; Zhang, X.; Chen, W.; Shen, Y.; Bai, X.; Liang, T. A systematic review and meta-analysis of adjuvant transarterial chemoembolization after curative resection for patients with hepatocellular carcinoma. Hpb 2019, 22, 795–808. [Google Scholar] [CrossRef]
- Li, L.; Li, B.; Zhang, M. Postoperative adjuvant transarterial chemoembolization improves the prognosis of hepatocellular carcinoma patients with microvascular invasion: A systematic review and meta-analysis. Acta Radiol. 2019, 61, 723–731. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.-H.; Zhang, X.-P.; Zhou, T.-F.; Wang, K.; Wang, H.; Chai, Z.-T.; Shi, J.; Guo, W.-X.; Cheng, S.-Q. Adjuvant transarterial chemoembolization improves survival outcomes in hepatocellular carcinoma with microvascular invasion: A systematic review and meta-analysis. Eur. J. Surg. Oncol. 2019, 45, 2188–2196. [Google Scholar] [CrossRef] [PubMed]
- Nitta, H.; Beppu, T.; Imai, K.; Hayashi, H.; Chikamoto, A.; Baba, H. Adjuvant Hepatic Arterial Infusion Chemotherapy after Hepatic Resection of Hepatocellular Carcinoma with Macroscopic Vascular Invasion. World J. Surg. 2013, 37, 1034–1042. [Google Scholar] [CrossRef]
- Moran, A.; Ramos, L.F.; Picado, O.; Pendola, F.; Sleeman, D.; Dudeja, V.; Merchant, N.; Yakoub, D. Hepatocellular carcinoma: Resection with adjuvant hepatic artery infusion therapy vs resection alone. A systematic review and meta-analysis. J. Surg. Oncol. 2019, 119, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Hatano, E.; Uemoto, S.; Yamaue, H.; Yamamoto, M.; Japanese Society of Hepato-Biliary-Pancreatic Surgery. Significance of hepatic resection and adjuvant hepatic arterial infusion chemotherapy for hepatocellular carcinoma with portal vein tumor thrombus in the first branch of portal vein and the main portal trunk: A project study for hepatic surgery of the Jap. J. Hepato-Biliary-Pancreatic Sci. 2018, 25, 395–402. [Google Scholar] [CrossRef]
- Hamada, T.; Yano, K.; Wada, T.; Imamura, N.; Hiyoshi, M.; Kondo, K.; Nanashima, A. Increased Survival Benefit of Adjuvant Intra-arterial Infusion Chemotherapy in HCC Patients with Portal Vein Infiltration after Hepatectomy. World J. Surg. 2020, 44, 2770–2776. [Google Scholar] [CrossRef]
- Bruix, J.; Takayama, T.; Mazzaferro, V.; Chau, G.-Y.; Yang, J.; Kudo, M.; Cai, J.; Poon, R.T.; Han, K.-H.; Tak, W.Y.; et al. Adjuvant sorafenib for hepatocellular carcinoma after resection or ablation (STORM): A phase 3, randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2015, 16, 1344–1354. [Google Scholar] [CrossRef]
- Li, J.; Hou, Y.; Cai, X.-B.; Liu, B. Sorafenib after resection improves the outcome of BCLC stage C hepatocellular carcinoma. World J. Gastroenterol. 2016, 22, 4034–4040. [Google Scholar] [CrossRef]
- Zhuang, L.; Wen, T.; Xu, M.; Yang, J.; Wang, W.; Wu, H.; Zeng, Y.; Yan, L.; Wei, Y.; Li, B. Sorafenib combined with hepatectomy in patients with intermediate-stage and advanced hepatocellular carcinoma. Arch. Med Sci. 2017, 6, 1383–1393. [Google Scholar] [CrossRef]
- Li, Z.; Gao, J.; Zheng, S.M.; Wang, Y.; Xiang, X.; Cheng, Q.; Zhu, J. The efficacy of sorafenib in preventing hepatocellular carcinoma recurrence after resection: A systematic review and meta-analysis. Rev. Esp. Enferm. Dig. 2020, 112, 201–210. [Google Scholar] [CrossRef]
- Yau, T.; Kang, Y.-K.; Kim, T.-Y.; El-Khoueiry, A.B.; Santoro, A.; Sangro, B.; Melero, I.; Kudo, M.; Hou, M.-M.; Matilla, A.; et al. Efficacy and Safety of Nivolumab Plus Ipilimumab in Patients with Advanced Hepatocellular Carcinoma Previously Treated With Sorafenib: The CheckMate 040 Randomized Clinical Trial. JAMA Oncol. 2020, 6, e204564. [Google Scholar] [CrossRef]
- Finn, R.S.; Qin, S.; Ikeda, M.; Galle, P.R.; Ducreux, M.; Kim, T.-Y.; Kudo, M.; Breder, V.; Merle, P.; Kaseb, A.O.; et al. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N. Engl. J. Med. 2020, 382, 1894–1905. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Castet, F.; Heikenwalder, M.; Maini, M.K.; Mazzaferro, V.; Pinato, D.J.; Pikarsky, E.; Zhu, A.X.; Finn, R.S. Immunotherapies for hepatocellular carcinoma. Nat. Rev. Clin. Oncol. 2022, 19, 151–172. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Li, W.; Wu, X.; Zhao, F.; Wang, D.; Wu, H.; Gu, Y.; Li, X.; Qian, X.; Hu, J.; et al. Safety and Efficacy of Sintilimab and Anlotinib as First Line Treatment for Advanced Hepatocellular Carcinoma (KEEP-G04): A Single-Arm Phase 2 Study. Front. Oncol. 2022, 12, 909035. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Xu, L.; Ji, J.; Bao, D.; Hu, J.; Qian, Y.; Zhou, Y.; Chen, Z.; Li, D.; Li, X.; et al. Sintilimab combined with apatinib plus capecitabine in the treatment of unresectable hepatocellular carcinoma: A prospective, open-label, single-arm, phase II clinical study. Front. Immunol. 2022, 13, 944062. [Google Scholar] [CrossRef]
- Xu, J.; Shen, J.; Gu, S.; Zhang, Y.; Wu, L.; Wu, J.; Shao, G.; Zhang, Y.; Xu, L.; Yin, T.; et al. Camrelizumab in Combination with Apatinib in Patients with Advanced Hepatocellular Carcinoma (RESCUE): A Nonrandomized, Open-label, Phase II Trial. Clin. Cancer Res. 2021, 27, 1003–1011. [Google Scholar] [CrossRef]
- Zhao, L.; Chang, N.; Shi, L.; Li, F.; Meng, F.; Xie, X.; Xu, Z.; Wang, F. Lenvatinib plus sintilimab versus lenvatinib monotherapy as first-line treatment for advanced HBV-related hepatocellular carcinoma: A retrospective, real-world study. Heliyon 2022, 8, e09538. [Google Scholar] [CrossRef]
- Li, Q.; Cao, M.; Yuan, G.; Cheng, X.; Zang, M.; Chen, M.; Hu, X.; Huang, J.; Li, R.; Guo, Y.; et al. Lenvatinib Plus Camrelizumab vs. Lenvatinib Monotherapy as First-Line Treatment for Unresectable Hepatocellular Carcinoma: A Multicenter Retrospective Cohort Study. Front. Oncol. 2022, 12, 462. [Google Scholar] [CrossRef]
- Liu, Q.; You, N.; Li, J.; Wu, K.; Peng, X.; Wang, Z.; Wang, L.; Zhu, Y.; Zheng, L. Camrelizumab Plus Sorafenib Versus Sorafenib Monotherapy for Advanced Hepatocellular Carcinoma: A Retrospective Analysis. Front. Oncol. 2021, 11, 694409. [Google Scholar] [CrossRef]
- Chen, X.-Q.; Zhao, Y.-X.; Zhang, C.-L.; Wang, X.-T.; Zhang, X.; Chen, X.; Yuan, C.-W.; Zhao, Q.; Chen, X.-J. Effectiveness and Safety of Anlotinib with or without PD-1 Blockades in the Treatment of Patients with Advanced Primary Hepatocellular Carcinoma: A Retrospective, Real-World Study in China. Drug Des. Dev. Ther. 2022, ume 16, 1483–1493. [Google Scholar] [CrossRef]
- Teng, Y.; Ding, X.; Li, W.; Cheng, J. Clinical analysis of Sorafenib combined with domestic PD-1 inhibitors in the treatment of unresectable hepatocellular carcinoma. Chin. J Gastroenterol. Hepatol. 2022, 31, 173–176+181. [Google Scholar] [CrossRef]
- Xiang, J.; Gong, W.; Wang, C.; Sun, P.; Liu, A. Complete remission of alpha-fetoprotein-producing gastric cancer by combined tislelizumab-apatinib treatment of a patient with proficient mismatch repair: A case report. World J. Surg. Oncol. 2022, 20, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Tang, K.; Li, X.; Lu, W. Anti-PD-1-based immunotherapy plus lenvatinib to treat advanced gallbladder cancer in the elderly: A case series and review of current literature. J. Cancer Res. Clin. Oncol. 2022, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.-M.; Kim, J.M.; Choi, G.-S.; Kwon, C.H.D.; Yi, N.-J.; Lee, K.-W.; Suh, K.S.; Joh, J.-W. Characteristics of Early Recurrence After Curative Liver Resection for Solitary Hepatocellular Carcinoma. J. Gastrointest. Surg. 2019, 23, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Gabrielson, A.; Wu, Y.; Wang, H.; Jiang, J.; Kallakury, B.; Gatalica, Z.; Reddy, S.; Kleiner, D.; Fishbein, T.; Johnson, L.; et al. Intratumoral CD3 and CD8 T-cell Densities Associated with Relapse-Free Survival in HCC. Cancer Immunol. Res. 2016, 4, 419–430. [Google Scholar] [CrossRef]
- Sun, L.; Xu, G.; Liao, W.; Yang, H.; Xu, H.; Du, S.; Zhao, H.; Lu, X.; Sang, X.; Mao, Y. Clinicopathologic and prognostic significance of regulatory T cells in patients with hepatocellular carcinoma: A meta-analysis. Oncotarget 2017, 8, 39658–39672. [Google Scholar] [CrossRef] [PubMed]
- Shi, F.; Shi, M.; Zeng, Z.; Qi, R.-Z.; Liu, Z.-W.; Zhang, J.-Y.; Yang, Y.-P.; Tien, P.; Wang, F.-S. PD-1 and PD-L1 upregulation promotes CD8+ T-cell apoptosis and postoperative recurrence in hepatocellular carcinoma patients. Int. J. Cancer 2011, 128, 887–896. [Google Scholar] [CrossRef]
- Calderaro, J.; Rousseau, B.; Amaddeo, G.; Mercey, M.; Charpy, C.; Costentin, C.; Luciani, A.; Zafrani, E.-S.; Laurent, A.; Azoulay, D.; et al. Programmed death ligand 1 expression in hepatocellular carcinoma: Relationship With clinical and pathological features. Hepatology 2016, 64, 2038–2046. [Google Scholar] [CrossRef]
- Fukumura, D.; Kloepper, J.; Amoozgar, Z.; Duda, D.G.; Jain, R.K. Enhancing cancer immunotherapy using antiangiogenics: Opportunities and challenges. Nat. Rev. Clin. Oncol. 2018, 15, 325–340. [Google Scholar] [CrossRef]
- Hegde, P.S.; Wallin, J.J.; Mancao, C. Predictive markers of anti-VEGF and emerging role of angiogenesis inhibitors as immunotherapeutics. Semin. Cancer Biol. 2018, 52, 117–124. [Google Scholar] [CrossRef]
- Gao, Q.; Anwar, I.J.; Abraham, N.; Barbas, A.S. Liver Transplantation for Hepatocellular Carcinoma after Downstaging or Bridging Therapy with Immune Checkpoint Inhibitors. Cancers 2021, 13, 6307. [Google Scholar] [CrossRef] [PubMed]
- Sové, R.J.; Verma, B.K.; Wang, H.; Ho, W.J.; Yarchoan, M.; Popel, A.S. Virtual clinical trials of anti-PD-1 and anti-CTLA-4 immunotherapy in advanced hepatocellular carcinoma using a quantitative systems pharmacology model. J. Immunother. Cancer 2022, 10, e005414. [Google Scholar] [CrossRef] [PubMed]
- Verma, B.K.; Subramaniam, P.; Vadigepalli, R. Modeling the Dynamics of Human Liver Failure Post Liver Resection. Processes 2018, 6, 115. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.-Y.; Ouyang, Q.; Cen, W.-J.; Wang, F.; Tang, W.-T.; Shao, J.-Y. A Model Based on Artificial Intelligence Algorithm for Monitoring Recurrence of HCC after Hepatectomy. Am. Surg. 2021, 00031348211063549. [Google Scholar] [CrossRef] [PubMed]
| Adjuvant ICIs | Targeted Therapies | Entire Cohort (n = 38) | PSM Cohort (n = 33) |
|---|---|---|---|
| Sintilimab | Anlotinib [24] | 9 | 9 |
| Apatinib [25] | 7 | 6 | |
| Lenvatinib [27] | 4 | 3 | |
| Sorafenib [31] | 1 | 1 | |
| Camrelizumab | Anlotinib [30] | 3 | 3 |
| Apatinib [26] | 7 | 6 | |
| Lenvatinib [28] | 2 | 2 | |
| Sorafenib [29] | 1 | 1 | |
| Tislelizumab | Apatinib [32] | 1 | 1 |
| Lenvatinib [33] | 2 | 0 | |
| Atezolizumab | Bevacizumab [22] | 1 | 1 |
| Entire Cohort (n = 196) | PSM Cohort (n = 66) | |||||
|---|---|---|---|---|---|---|
| Characteristic | Adjuvant ICIs Plus Targeted Therapies | p | Adjuvant ICIs Plus Targeted Therapies | p | ||
| No (n = 158) | Yes (n = 38) | No (n = 33) | Yes (n = 33) | |||
| Gender (%) | 0.933 | >0.99 | ||||
| female | 20 (12.7) | 4 (10.5) | 3 (9.1) | 3 (9.1) | ||
| male | 138 (87.3) | 34 (89.5) | 30 (90.9) | 30 (90.9) | ||
| Age (y, median [IQR]) | 55.0 [47.0, 63.0] | 50.5 [44.0, 54.8] | 0.026 | 49.0 [44.0, 56.0] | 50.0 [44.0, 55.0] | 0.847 |
| Diabetes (%) | 18 (11.4) | 7 (18.4) | 0.371 | 4 (12.1) | 7 (21.2) | 0.509 |
| Hypertention (%) | 30 (19.0) | 6 (15.8) | 0.823 | 7 (21.2) | 6 (18.2) | >0.99 |
| HbsAg (%) | 0.739 | >0.99 | ||||
| positive | 140 (88.6) | 35 (92.1) | 32 (97.0) | 31 (93.9) | ||
| negative | 18 (11.4) | 3 (7.9) | 1 (3.0) | 2 (6.1) | ||
| Cirrhosis (%) | 82 (51.9) | 26 (68.4) | 0.098 | 18 (54.5) | 22 (66.7) | 0.450 |
| Child-Pugh grade (%) | 0.459 | 0.063 | ||||
| A | 146 (92.4) | 37 (97.4) | 28 (84.8) | 33 (100.0) | ||
| B | 12 (7.6) | 1 (2.6) | 5 (15.2) | 0 (0.0) | ||
| BCLC-stage (%) | <0.001 | 0.469 | ||||
| A | 73 (46.2) | 4 (10.5) | 8 (24.2) | 4 (12.1) | ||
| B | 12 (7.6) | 6 (15.8) | 3 (9.1) | 6 (18.2) | ||
| C | 49 (31.0) | 25 (65.8) | 20 (60.6) | 20 (60.6) | ||
| Preoperative locoregional therapies(%) | 22 (13.9) | 13 (34.2) | 0.007 | 9 (27.3) | 9 (27.3) | >0.99 |
| ECOG score ≥1 (%) | 9 (5.7) | 3 (7.9) | 0.896 | 1 (3.0) | 3 (9.1) | 0.606 |
| Maximum tumor size(cm, median [IQR]) | 4.5 [2.6, 6.6] | 3.6 [1.1, 5.7] | 0.048 | 5.5 [2.7, 8.1] | 3.8 [1.7, 5.7] | 0.068 |
| Number of lesion (%) | 0.046 | >0.99 | ||||
| <3 | 133 (84.2) | 26 (68.4) | 25 (75.8) | 25 (75.8) | ||
| ≥3 | 25 (15.8) | 12 (31.6) | 8 (24.2) | 8 (24.2) | ||
| Macrovascular invasion (%) | 46 (29.1) | 24 (63.2) | <0.001 | 21 (63.6) | 19 (57.6) | 0.801 |
| Macrovascular tumor thrombus (%) | 15 (9.5) | 5 (13.2) | 0.710 | 5 (15.2) | 4 (12.1) | >0.99 |
| AFP (ng/mL, median [IQR]) | 39.6 [4.8, 599.0] | 11.3 [4.1, 169.7] | 0.177 | 45.9 [6.4, 487.0] | 12.6 [4.1, 293.1] | 0.145 |
| Poor tumor grade (%) | 27 (17.1) | 8 (21.1) | 0.736 | 7 (21.2) | 6 (18.2) | >0.99 |
| Incomplete Capsule (%) | 40 (25.3) | 8 (21.1) | 0.735 | 7 (21.2) | 8 (24.2) | >0.99 |
| MVI (%) | 72 (45.6) | 16 (42.1) | 0.838 | 18 (54.5) | 16 (48.5) | 0.805 |
| Location of recurrence | 0.138 | 0.043 | ||||
| intrahepatic | 56 (35.4) | 7 (18.4) | 17 (51.5) | 7 (21.2) | ||
| extrahepatic | 4 (2.5) | 1 (2.6) | 0 (0.0) | 1 (3.0) | ||
| both | 14 (8.9) | 2 (5.3) | 3 (9.1) | 2 (6.1) | ||
| Follow up time (d, mean (SD)) | 363.9 (231.6) | 371.2 (189.7) | 0.858 | 331.6 (233.7) | 394.2 (189.2) | 0.237 |
| Characteristics | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| HR | 95%CI | p | aHR | 95%CI | p | |
| Gender, male/female | 0.75 | 0.40–1.37 | 0.340 | |||
| Age, >60 y/<60 y | 0.95 | 0.59–1.51 | 0.817 | |||
| Hypertention, yes/no | 1.07 | 0.63–1.82 | 0.815 | |||
| Diabetes, yes/no | 0.80 | 0.40–1.60 | 0.523 | |||
| HBV infection, yes/no | 2.34 | 0.95–5.78 | 0.058 | 2.17 | 0.86–5.46 | 0.097 |
| ICIs plus targeted therapies, yes/no | 0.46 | 0.24–0.90 | 0.020 | 0.62 | 0.49–0.79 | <0.001 |
| Preoperative locoregional therapies, yes/no | 1.44 | 0.84–2.45 | 0.180 | |||
| ECOG score, 1–2/0 point | 1.47 | 0.68–3.20 | 0.325 | |||
| Child-Pugh grade, B/A | 1.43 | 0.66–3.09 | 0.366 | |||
| MVI, yes/no | 2.24 | 1.45–3.46 | <0.001 | 2.04 | 1.31–3.15 | 0.001 |
| Tumor grade, poor/well-moderate | 1.15 | 0.67–1.98 | 0.618 | |||
| Capsular invasion, yes/no | 1.17 | 0.72–1.89 | 0.523 | |||
| AFP, ≥400/<400 ng/ml | 1.32 | 0.84–2.09 | 0.231 | |||
| Macrovascular invasion, yes/no | 1.49 | 0.97–2.3 | 0.069 | 2.21 | 1.39–3.52 | <0.001 |
| Macrovascular tumor thrombus, yes/no | 1.59 | 0.84–3.01 | 0.147 | |||
| Number of lesion, ≥3/<3 | 1.72 | 1.02–2.88 | 0.038 | 1.96 | 1.14–3.36 | 0.014 |
| Maximum tumor diameter, ≥5/<5 cm | 1.15 | 0.75–1.77 | 0.514 | |||
| Cirrhosis, yes/no | 1.72 | 1.10–2.68 | 0.016 | 1.69 | 1.06–2.70 | 0.026 |
| AEs | Grade1 (%) | Grade2 (%) | Grade3/4 (%) |
|---|---|---|---|
| Hepatitis | 0 | 1 (2.6) | 0 |
| Thyroiditis | 0 | 1 (2.6) | 0 |
| Dermatitis | 0 | 2 (5.3) | 0 |
| Hypopituitarism | 0 | 0 | 0 |
| Gastroenteritis | 0 | 0 | 1 (2.6) |
| Pancreatitis | 0 | 0 | 0 |
| Pneumonia | 0 | 0 | 0 |
| Myocarditis | 0 | 0 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, J.; Jiang, S.; Chen, Y.; Zhang, J.; Deng, Y. Adjuvant ICIs Plus Targeted Therapies Reduce HCC Recurrence after Hepatectomy in Patients with High Risk of Recurrence. Curr. Oncol. 2023, 30, 1708-1719. https://doi.org/10.3390/curroncol30020132
Yang J, Jiang S, Chen Y, Zhang J, Deng Y. Adjuvant ICIs Plus Targeted Therapies Reduce HCC Recurrence after Hepatectomy in Patients with High Risk of Recurrence. Current Oncology. 2023; 30(2):1708-1719. https://doi.org/10.3390/curroncol30020132
Chicago/Turabian StyleYang, Jianming, Shijie Jiang, Yewu Chen, Jian Zhang, and Yinan Deng. 2023. "Adjuvant ICIs Plus Targeted Therapies Reduce HCC Recurrence after Hepatectomy in Patients with High Risk of Recurrence" Current Oncology 30, no. 2: 1708-1719. https://doi.org/10.3390/curroncol30020132
APA StyleYang, J., Jiang, S., Chen, Y., Zhang, J., & Deng, Y. (2023). Adjuvant ICIs Plus Targeted Therapies Reduce HCC Recurrence after Hepatectomy in Patients with High Risk of Recurrence. Current Oncology, 30(2), 1708-1719. https://doi.org/10.3390/curroncol30020132





