Drug Treatment for Advanced Hepatocellular Carcinoma: First-Line and Beyond
Abstract
:1. Background
2. Systemic Therapies for Advanced HCC
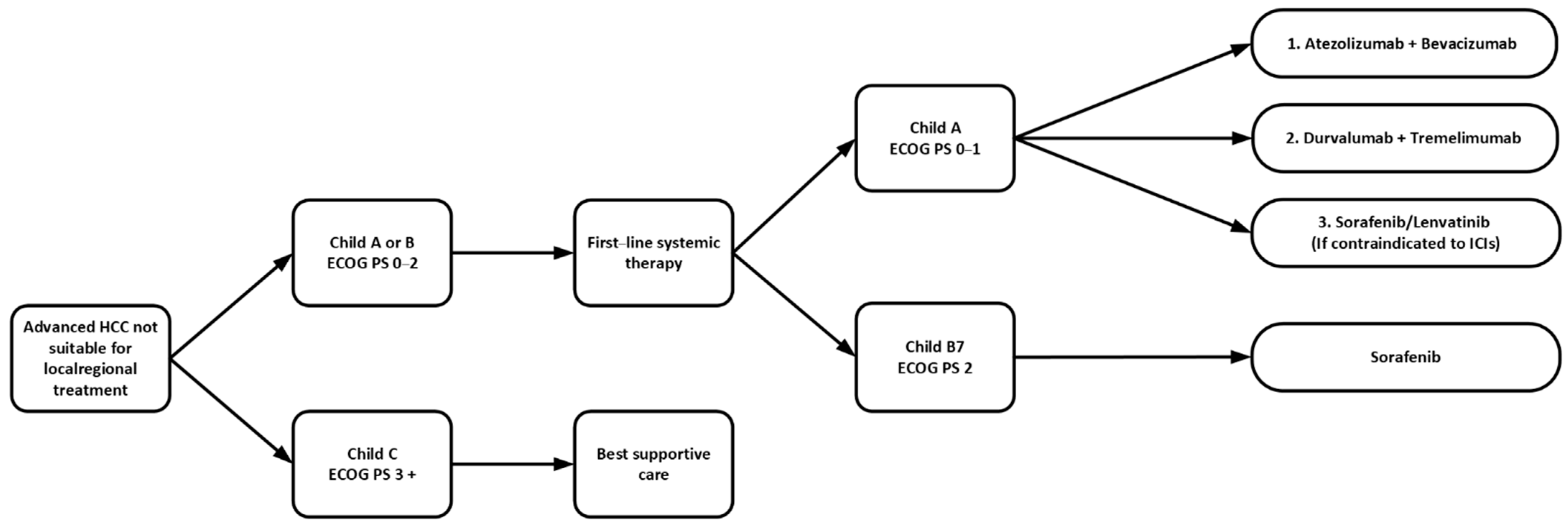
2.1. First-Line Systemic Therapy
2.1.1. Single Agent Multikinase Inhibitor
Sorafenib
Lenvatinib
2.1.2. Combining ICI with an Anti-VEGF Antibody
Atezolizumab and Bevacizumab
Atezolizumab and Cabozantinib
2.1.3. PD-1 and CTLA-4 Antibodies Combination
Durvalumab and Tremelimumab
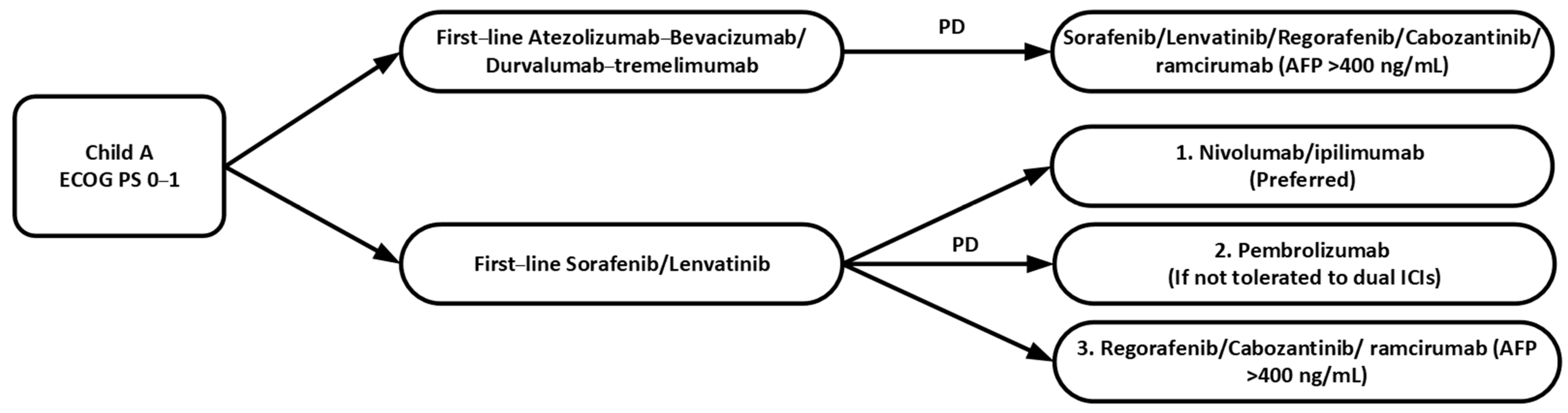
2.1.4. Summary in First-Line Systemic Therapy
2.2. Second-Line Systemic Therapy
2.2.1. Single-Agent Multikinase Inhibitor
Regorafenib
Cabozantinib
2.2.2. Single VEGF Antibody
Ramucirumab
2.2.3. Single ICI
Pembrolizumab
2.2.4. PD-1 and CTLA-4 Antibodies Combination
Nivolumab and Ipilimumab
2.2.5. Summary in Second-Line Systemic Therapy
3. Ongoing Trials in Combination Systemic Treatment
4. Discussion and Future Perspectives
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Villanueva, A.; Llovet, J.M. Targeted therapies for hepatocellular carcinoma. Gastroenterology 2011, 140, 1410–1426. [Google Scholar] [CrossRef] [PubMed]
- Vitale, A.; Peck-Radosavljevic, M.; Giannini, E.G.; Vibert, E.; Sieghart, W.; Van Poucke, S.; Pawlik, T.M. Personalized treatment of patients with very early hepatocellular carcinoma. J. Hepatol. 2017, 66, 412–423. [Google Scholar] [CrossRef]
- Kulik, L.; El-Serag, H.B. Epidemiology and Management of Hepatocellular Carcinoma. Gastroenterology 2019, 156, 477–491.e471. [Google Scholar] [CrossRef] [PubMed]
- Kudo, M. Management of Hepatocellular Carcinoma in Japan as a World-Leading Model. Liver Cancer 2018, 7, 134–147. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef]
- Ikeda, K.; Kudo, M.; Kawazoe, S.; Osaki, Y.; Ikeda, M.; Okusaka, T.; Tamai, T.; Suzuki, T.; Hisai, T.; Hayato, S.; et al. Phase 2 study of lenvatinib in patients with advanced hepatocellular carcinoma. J. Gastroenterol. 2017, 52, 512–519. [Google Scholar] [CrossRef]
- Bruix, J.; Qin, S.; Merle, P.; Granito, A.; Huang, Y.H.; Bodoky, G.; Pracht, M.; Yokosuka, O.; Rosmorduc, O.; Breder, V.; et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2017, 389, 56–66. [Google Scholar] [CrossRef]
- Abou-Alfa, G.K.; Meyer, T.; Cheng, A.L.; El-Khoueiry, A.B.; Rimassa, L.; Ryoo, B.Y.; Cicin, I.; Merle, P.; Chen, Y.; Park, J.W.; et al. Cabozantinib in Patients with Advanced and Progressing Hepatocellular Carcinoma. N. Engl. J. Med. 2018, 379, 54–63. [Google Scholar] [CrossRef]
- Zhu, A.X.; Park, J.O.; Ryoo, B.Y.; Yen, C.J.; Poon, R.; Pastorelli, D.; Blanc, J.F.; Chung, H.C.; Baron, A.D.; Pfiffer, T.E.; et al. Ramucirumab versus placebo as second-line treatment in patients with advanced hepatocellular carcinoma following first-line therapy with sorafenib (REACH): A randomised, double-blind, multicentre, phase 3 trial. Lancet Oncol. 2015, 16, 859–870. [Google Scholar] [CrossRef]
- Greten, T.F.; Lai, C.W.; Li, G.; Staveley-O’Carroll, K.F. Targeted and Immune-Based Therapies for Hepatocellular Carcinoma. Gastroenterology 2019, 156, 510–524. [Google Scholar] [CrossRef]
- Zhu, A.X.; Finn, R.S.; Edeline, J.; Cattan, S.; Ogasawara, S.; Palmer, D.; Verslype, C.; Zagonel, V.; Fartoux, L.; Vogel, A.; et al. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): A non-randomised, open-label phase 2 trial. Lancet Oncol. 2018, 19, 940–952. [Google Scholar] [CrossRef]
- Yau, T.; Kang, Y.-K.; Kim, T.-Y.; El-Khoueiry, A.B.; Santoro, A.; Sangro, B.; Melero, I.; Kudo, M.; Hou, M.-M.; Matilla, A.; et al. Efficacy and Safety of Nivolumab Plus Ipilimumab in Patients With Advanced Hepatocellular Carcinoma Previously Treated With Sorafenib: The CheckMate 040 Randomized Clinical Trial. JAMA Oncol. 2020, 6, e204564. [Google Scholar] [CrossRef]
- Gordan, J.D.; Kennedy, E.B.; Abou-Alfa, G.K.; Beg, M.S.; Brower, S.T.; Gade, T.P.; Goff, L.; Gupta, S.; Guy, J.; Harris, W.P.; et al. Systemic Therapy for Advanced Hepatocellular Carcinoma: ASCO Guideline. J. Clin. Oncol. 2020, 38, 4317–4345. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, S.M.; Carter, C.; Tang, L.; Wilkie, D.; McNabola, A.; Rong, H.; Chen, C.; Zhang, X.; Vincent, P.; McHugh, M.; et al. BAY 43-9006 Exhibits Broad Spectrum Oral Antitumor Activity and Targets the RAF/MEK/ERK Pathway and Receptor Tyrosine Kinases Involved in Tumor Progression and Angiogenesis. Cancer Res. 2004, 64, 7099–7109. [Google Scholar] [CrossRef]
- Dawkins, J.; Webster, R.M. The hepatocellular carcinoma market. Nat. Rev. Drug Discov. 2019, 18, 13–14. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.L.; Kang, Y.K.; Chen, Z.; Tsao, C.J.; Qin, S.; Kim, J.S.; Luo, R.; Feng, J.; Ye, S.; Yang, T.S.; et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: A phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009, 10, 25–34. [Google Scholar] [CrossRef]
- Bruix, J.; Raoul, J.L.; Sherman, M.; Mazzaferro, V.; Bolondi, L.; Craxi, A.; Galle, P.R.; Santoro, A.; Beaugrand, M.; Sangiovanni, A.; et al. Efficacy and safety of sorafenib in patients with advanced hepatocellular carcinoma: Subanalyses of a phase III trial. J. Hepatol. 2012, 57, 821–829. [Google Scholar] [CrossRef]
- Méndez-Blanco, C.; Fondevila, F.; García-Palomo, A.; González-Gallego, J.; Mauriz, J.L. Sorafenib resistance in hepatocarcinoma: Role of hypoxia-inducible factors. Exp. Mol. Med. 2018, 50, 1–9. [Google Scholar] [CrossRef]
- Al-Salama, Z.T.; Syed, Y.Y.; Scott, L.J. Lenvatinib: A Review in Hepatocellular Carcinoma. Drugs 2019, 79, 665–674. [Google Scholar] [CrossRef]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.W.; Han, G.; Jassem, J.; et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: A randomised phase 3 non-inferiority trial. Lancet 2018, 391, 1163–1173. [Google Scholar] [CrossRef]
- Vogel, A.; Cervantes, A.; Chau, I.; Daniele, B.; Llovet, J.M.; Meyer, T.; Nault, J.C.; Neumann, U.; Ricke, J.; Sangro, B.; et al. Hepatocellular carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2019, 30, 871–873. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef]
- Finn, R.S.; Qin, S.; Ikeda, M.; Galle, P.R.; Ducreux, M.; Kim, T.Y.; Kudo, M.; Breder, V.; Merle, P.; Kaseb, A.O.; et al. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N. Engl. J. Med. 2020, 382, 1894–1905. [Google Scholar] [CrossRef] [PubMed]
- Finn, R.S.; Qin, S.; Ikeda, M.; Galle, P.R.; Ducreux, M.; Kim, T.-Y.; Lim, H.Y.; Kudo, M.; Breder, V.V.; Merle, P.; et al. IMbrave150: Updated overall survival (OS) data from a global, randomized, open-label phase III study of atezolizumab (atezo) + bevacizumab (bev) versus sorafenib (sor) in patients (pts) with unresectable hepatocellular carcinoma (HCC). J. Clin. Oncol. 2021, 39, 267. [Google Scholar] [CrossRef]
- Greten, T.F.; Abou-Alfa, G.K.; Cheng, A.L.; Duffy, A.G.; El-Khoueiry, A.B.; Finn, R.S.; Galle, P.R.; Goyal, L.; He, A.R.; Kaseb, A.O.; et al. Society for Immunotherapy of Cancer (SITC) clinical practice guideline on immunotherapy for the treatment of hepatocellular carcinoma. J. Immunother. Cancer 2021, 9, e002794. [Google Scholar] [CrossRef] [PubMed]
- Bruix, J.; Chan, S.L.; Galle, P.R.; Rimassa, L.; Sangro, B. Systemic treatment of hepatocellular carcinoma: An EASL position paper. J. Hepatol. 2021, 75, 960–974. [Google Scholar] [CrossRef] [PubMed]
- Exelixis Announces Detailed Results from Phase 3 COSMIC-312 Pivotal Trial of Cabozantinib in Combination with an Immune Checkpoint Inhibitor in Patients with Previously Untreated Advanced Liver Cancer at ESMO Asia Virtual Oncology Week 2021. Available online: https://www.businesswire.com/news/home/20211120005130/en/Exelixis-Announces-Detailed-Results-from-Phase-3-COSMIC-312-Pivotal-Trial-of-Cabozantinib-in-Combination-with-an-Immune-Checkpoint-Inhibitor-in-Patients-with-Previously-Untreated-Advanced-Liver-Cancer-at-ESMO-Asia-Virtual-Oncology-Week-2021 (accessed on 12 February 2022).
- Kelley, R.K.; Yau, T.; Cheng, A.L.; Kaseb, A.; Qin, S.; Zhu, A.X.; Chan, S.; Sukeepaisarnjaroen, W.; Breder, V.; Verset, G.; et al. VP10-2021: Cabozantinib (C) plus atezolizumab (A) versus sorafenib (S) as first-line systemic treatment for advanced hepatocellular carcinoma (aHCC): Results from the randomized phase III COSMIC-312 trial. Ann. Oncol. 2022, 33, 114–116. [Google Scholar] [CrossRef]
- Cammarota, A.; Zanuso, V.; D’Alessio, A.; Pressiani, T.; Personeni, N.; Rimassa, L. Cabozantinib plus atezolizumab for the treatment of advanced hepatocellular carcinoma: Shedding light on the preclinical rationale and clinical trials. Expert Opin. Investig. Drugs 2022, 31, 401–413. [Google Scholar] [CrossRef] [PubMed]
- Eroglu, Z.; Kim, D.W.; Wang, X.; Camacho, L.H.; Chmielowski, B.; Seja, E.; Villanueva, A.; Ruchalski, K.; Glaspy, J.A.; Kim, K.B.; et al. Long term survival with cytotoxic T lymphocyte-associated antigen 4 blockade using tremelimumab. Eur. J. Cancer 2015, 51, 2689–2697. [Google Scholar] [CrossRef]
- Antonia, S.; Goldberg, S.B.; Balmanoukian, A.; Chaft, J.E.; Sanborn, R.E.; Gupta, A.; Narwal, R.; Steele, K.; Gu, Y.; Karakunnel, J.J.; et al. Safety and antitumour activity of durvalumab plus tremelimumab in non-small cell lung cancer: A multicentre, phase 1b study. Lancet Oncol. 2016, 17, 299–308. [Google Scholar] [CrossRef]
- Kelley, R.K.; Abou-Alfa, G.K.; Bendell, J.C.; Kim, T.-Y.; Borad, M.J.; Yong, W.-P.; Morse, M.; Kang, Y.-K.; Rebelatto, M.; Makowsky, M.; et al. Phase I/II study of durvalumab and tremelimumab in patients with unresectable hepatocellular carcinoma (HCC): Phase I safety and efficacy analyses. J. Clin. Oncol. 2017, 35, 4073. [Google Scholar] [CrossRef]
- Abou-Alfa, G.K.; Chan, S.L.; Kudo, M.; Lau, G.; Kelley, R.K.; Furuse, J.; Sukeepaisarnjaroen, W.; Kang, Y.-K.; Dao, T.V.; Toni, E.N.D.; et al. Phase 3 randomized, open-label, multicenter study of tremelimumab (T) and durvalumab (D) as first-line therapy in patients (pts) with unresectable hepatocellular carcinoma (uHCC): HIMALAYA. J. Clin. Oncol. 2022, 40, 379. [Google Scholar] [CrossRef]
- Maestri, M.; Pallozzi, M.; Santopaolo, F.; Cerrito, L.; Pompili, M.; Gasbarrini, A.; Ponziani, F.R. Durvalumab: An investigational agent for unresectable hepatocellular carcinoma. Expert Opin. Investig. Drugs 2022, 31, 347–360. [Google Scholar] [CrossRef]
- Wilhelm, S.M.; Dumas, J.; Adnane, L.; Lynch, M.; Carter, C.A.; Schütz, G.; Thierauch, K.-H.; Zopf, D. Regorafenib (BAY 73-4506): A new oral multikinase inhibitor of angiogenic, stromal and oncogenic receptor tyrosine kinases with potent preclinical antitumor activity. Int. J. Cancer 2011, 129, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Q.; Chen, W.; Ren, M.; Wang, J.; Zhang, H.; Deng, D.Y.B.; Zhang, L.; Shang, C.; Chen, Y. Cabozantinib Suppresses Tumor Growth and Metastasis in Hepatocellular Carcinoma by a Dual Blockade of VEGFR2 and MET. Clin. Cancer Res. 2014, 20, 2959–2970. [Google Scholar] [CrossRef] [PubMed]
- Rimassa, L.; Assenat, E.; Peck-Radosavljevic, M.; Pracht, M.; Zagonel, V.; Mathurin, P.; Rota Caremoli, E.; Porta, C.; Daniele, B.; Bolondi, L.; et al. Tivantinib for second-line treatment of MET-high, advanced hepatocellular carcinoma (METIV-HCC): A final analysis of a phase 3, randomised, placebo-controlled study. Lancet Oncol. 2018, 19, 682–693. [Google Scholar] [CrossRef]
- Firtina Karagonlar, Z.; Koc, D.; Iscan, E.; Erdal, E.; Atabey, N. Elevated hepatocyte growth factor expression as an autocrine c-Met activation mechanism in acquired resistance to sorafenib in hepatocellular carcinoma cells. Cancer Sci. 2016, 107, 407–416. [Google Scholar] [CrossRef]
- Rimassa, L.; Kelley, R.K.; Meyer, T.; Ryoo, B.Y.; Merle, P.; Park, J.W.; Blanc, J.F.; Lim, H.Y.; Tran, A.; Borgman-Hagey, A.E.; et al. Outcomes based on plasma biomarkers for the phase III CELESTIAL trial of cabozantinib (C) versus placebo (P) in advanced hepatocellular carcinoma (aHCC). Ann. Oncol. 2019, 30, v257–v258. [Google Scholar] [CrossRef]
- Kelley, R.K.; Meyer, T.; Rimassa, L.; Merle, P.; Park, J.W.; Yau, T.; Chan, S.L.; Blanc, J.F.; Tam, V.C.; Tran, A.; et al. Serum Alpha-fetoprotein Levels and Clinical Outcomes in the Phase III CELESTIAL Study of Cabozantinib versus Placebo in Patients with Advanced Hepatocellular Carcinoma. Clin. Cancer Res. 2020, 26, 4795–4804. [Google Scholar] [CrossRef]
- FDA Approves Ramucirumab for Hepatocellular Carcinoma. Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-ramucirumab-hepatocellular-carcinoma (accessed on 12 February 2022).
- Zhu, A.X.; Kang, Y.K.; Yen, C.J.; Finn, R.S.; Galle, P.R.; Llovet, J.M.; Assenat, E.; Brandi, G.; Pracht, M.; Lim, H.Y.; et al. Ramucirumab after sorafenib in patients with advanced hepatocellular carcinoma and increased α-fetoprotein concentrations (REACH-2): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019, 20, 282–296. [Google Scholar] [CrossRef]
- Finn, R.S.; Ryoo, B.Y.; Merle, P.; Kudo, M.; Bouattour, M.; Lim, H.Y.; Breder, V.; Edeline, J.; Chao, Y.; Ogasawara, S.; et al. Pembrolizumab as Second-Line Therapy in Patients with Advanced Hepatocellular Carcinoma in KEYNOTE-240: A Randomized, Double-Blind, Phase III Trial. J. Clin. Oncol. 2020, 38, 193–202. [Google Scholar] [CrossRef] [PubMed]
- FDA Grants Accelerated Approval to Nivolumab and Ipilimumab Combination for Hepatocellular Carcinoma. Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-nivolumab-and-ipilimumab-combination-hepatocellular-carcinoma (accessed on 12 February 2022).
- Qin, S.; Chen, Z.; Fang, W.; Ren, Z.; Xu, R.; Ryoo, B.-Y.; Meng, Z.; Bai, Y.; Chen, X.; Liu, X.; et al. Pembrolizumab plus best supportive care versus placebo plus best supportive care as second-line therapy in patients in Asia with advanced hepatocellular carcinoma (HCC): Phase 3 KEYNOTE-394 study. J. Clin. Oncol. 2022, 40, 383. [Google Scholar] [CrossRef]
- Allen, E.; Jabouille, A.; Rivera, L.B.; Lodewijckx, I.; Missiaen, R.; Steri, V.; Feyen, K.; Tawney, J.; Hanahan, D.; Michael, I.P.; et al. Combined antiangiogenic and anti-PD-L1 therapy stimulates tumor immunity through HEV formation. Sci. Transl. Med. 2017, 9, eaak9679. [Google Scholar] [CrossRef] [PubMed]
- Khan, K.A.; Kerbel, R.S. Improving immunotherapy outcomes with anti-angiogenic treatments and vice versa. Nat. Rev. Clin. Oncol. 2018, 15, 310–324. [Google Scholar] [CrossRef] [PubMed]
- Yi, M.; Jiao, D.; Qin, S.; Chu, Q.; Wu, K.; Li, A. Synergistic effect of immune checkpoint blockade and anti-angiogenesis in cancer treatment. Mol. Cancer 2019, 18, 60. [Google Scholar] [CrossRef] [PubMed]
- Kudo, M. Scientific Rationale for Combined Immunotherapy with PD-1/PD-L1 Antibodies and VEGF Inhibitors in Advanced Hepatocellular Carcinoma. Cancers 2020, 12, 89. [Google Scholar] [CrossRef]
- Finn, R.S.; Ikeda, M.; Zhu, A.X.; Sung, M.W.; Baron, A.D.; Kudo, M.; Okusaka, T.; Kobayashi, M.; Kumada, H.; Kaneko, S.; et al. Phase Ib Study of Lenvatinib Plus Pembrolizumab in Patients With Unresectable Hepatocellular Carcinoma. J. Clin. Oncol. 2020, 38, 2960–2970. [Google Scholar] [CrossRef] [PubMed]
- Finn, R.S.; Zhu, A.X. Evolution of Systemic Therapy for Hepatocellular Carcinoma. Hepatology 2021, 73 (Suppl. 1), 150–157. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Zhang, Y.; Jia, R.; Yue, C.; Chang, L.; Liu, R.; Zhang, G.; Zhao, C.; Zhang, Y.; Chen, C.; et al. Anti-PD-1 Antibody SHR-1210 Combined with Apatinib for Advanced Hepatocellular Carcinoma, Gastric, or Esophagogastric Junction Cancer: An Open-label, Dose Escalation and Expansion Study. Clin. Cancer Res. 2019, 25, 515–523. [Google Scholar] [CrossRef]
- Xu, J.; Shen, J.; Gu, S.; Zhang, Y.; Wu, L.; Wu, J.; Shao, G.; Zhang, Y.; Xu, L.; Yin, T.; et al. Camrelizumab in Combination with Apatinib in Patients with Advanced Hepatocellular Carcinoma (RESCUE): A Nonrandomized, Open-label, Phase II Trial. Clin. Cancer Res. 2021, 27, 1003–1011. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, J.; Shen, J.; Gu, S.; Wu, L.; Wu, J.; Shao, G.; Zhang, Y.; Xu, L.; Yin, T.; et al. Update on overall survival (OS) of RESCUE: An open-label, phase 2 trial of camrelizumab (C) in combination with apatinib (A) in patients with advanced hepatocellular carcinoma (HCC). J. Clin. Oncol. 2021, 39, 4076. [Google Scholar] [CrossRef]
- Kelley, R.K.; Greten, T.F. Hepatocellular Carcinoma—Origins and Outcomes. N. Engl. J. Med. 2021, 385, 280–282. [Google Scholar] [CrossRef] [PubMed]
- Pfister, D.; Núñez, N.G.; Pinyol, R.; Govaere, O.; Pinter, M.; Szydlowska, M.; Gupta, R.; Qiu, M.; Deczkowska, A.; Weiner, A.; et al. NASH limits anti-tumour surveillance in immunotherapy-treated HCC. Nature 2021, 592, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Chan, L.L.; Chan, S.L. Novel Perspectives in Immune Checkpoint Inhibitors and the Management of Non-Alcoholic Steatohepatitis-Related Hepatocellular Carcinoma. Cancers 2022, 14, 1526. [Google Scholar] [CrossRef]
- Sun, H.; Zhu, M.S.; Wu, W.R.; Shi, X.D.; Xu, L.B. Role of anti-angiogenesis therapy in the management of hepatocellular carcinoma: The jury is still out. World J. Hepatol. 2014, 6, 830–835. [Google Scholar] [CrossRef]
- Niu, L.; Liu, L.; Yang, S.; Ren, J.; Lai, P.B.S.; Chen, G.G. New insights into sorafenib resistance in hepatocellular carcinoma: Responsible mechanisms and promising strategies. Biochim. Biophys. Acta Rev. Cancer 2017, 1868, 564–570. [Google Scholar] [CrossRef]
- Singh, A.; Settleman, J. EMT, cancer stem cells and drug resistance: An emerging axis of evil in the war on cancer. Oncogene 2010, 29, 4741–4751. [Google Scholar] [CrossRef]
- van Malenstein, H.; Dekervel, J.; Verslype, C.; Van Cutsem, E.; Windmolders, P.; Nevens, F.; van Pelt, J. Long-term exposure to sorafenib of liver cancer cells induces resistance with epithelial-to-mesenchymal transition, increased invasion and risk of rebound growth. Cancer Lett. 2013, 329, 74–83. [Google Scholar] [CrossRef]
- Chen, X.; Lingala, S.; Khoobyari, S.; Nolta, J.; Zern, M.A.; Wu, J. Epithelial mesenchymal transition and hedgehog signaling activation are associated with chemoresistance and invasion of hepatoma subpopulations. J. Hepatol. 2011, 55, 838–845. [Google Scholar] [CrossRef] [PubMed]
- Ruiz de Galarreta, M.; Bresnahan, E.; Molina-Sánchez, P.; Lindblad, K.E.; Maier, B.; Sia, D.; Puigvehi, M.; Miguela, V.; Casanova-Acebes, M.; Dhainaut, M.; et al. β-Catenin Activation Promotes Immune Escape and Resistance to Anti-PD-1 Therapy in Hepatocellular Carcinoma. Cancer Discov. 2019, 9, 1124–1141. [Google Scholar] [CrossRef]
- Singal, A.G.; Hoshida, Y.; Pinato, D.J.; Marrero, J.; Nault, J.C.; Paradis, V.; Tayob, N.; Sherman, M.; Lim, Y.S.; Feng, Z.; et al. International Liver Cancer Association (ILCA) White Paper on Biomarker Development for Hepatocellular Carcinoma. Gastroenterology 2021, 160, 2572–2584. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Dai, X.; Dai, J.; Ding, C.; Zhang, Z.; Lin, Z.; Hu, J.; Lu, M.; Wang, Z.; Qi, Y.; et al. AFP promotes HCC progression by suppressing the HuR-mediated Fas/FADD apoptotic pathway. Cell Death Dis. 2020, 11, 822. [Google Scholar] [CrossRef]
- Lai, S.-C.; Su, Y.-T.; Chi, C.-C.; Kuo, Y.-C.; Lee, K.-F.; Wu, Y.-C.; Lan, P.-C.; Yang, M.-H.; Chang, T.-S.; Huang, Y.-H. DNMT3b/OCT4 expression confers sorafenib resistance and poor prognosis of hepatocellular carcinoma through IL-6/STAT3 regulation. J. Exp. Clin. Cancer Res. 2019, 38, 474. [Google Scholar] [CrossRef]
- Tan, W.; Luo, X.; Li, W.; Zhong, J.; Cao, J.; Zhu, S.; Chen, X.; Zhou, R.; Shang, C.; Chen, Y. TNF-α is a potential therapeutic target to overcome sorafenib resistance in hepatocellular carcinoma. EBioMedicine 2019, 40, 446–456. [Google Scholar] [CrossRef]
- Haber, P.K.; Torres-Martin, M.; Dufour, J.-F.; Verslype, C.; Marquardt, J.; Galle, P.R.; Vogel, A.; Meyer, T.; Labgaa, I.; Roberts, L.R.; et al. Molecular markers of response to anti-PD1 therapy in advanced hepatocellular carcinoma. J. Clin. Oncol. 2021, 39, 4100. [Google Scholar] [CrossRef]
- Buti, S.; Bersanelli, M.; Perrone, F.; Tiseo, M.; Tucci, M.; Adamo, V.; Stucci, L.S.; Russo, A.; Tanda, E.T.; Spagnolo, F.; et al. Effect of concomitant medications with immune-modulatory properties on the outcomes of patients with advanced cancer treated with immune checkpoint inhibitors: Development and validation of a novel prognostic index. Eur. J. Cancer 2021, 142, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Meriggi, F.; Zaniboni, A. Antibiotics and steroids, the double enemies of anticancer immunotherapy: A review of the literature. Cancer Immunol. Immunother. 2021, 70, 1511–1517. [Google Scholar] [CrossRef]
| Agent | Regimen(s) | Targets | Indication | Sample Size | OS (Months) | OS (HR, 95% CI) | PFS (Months) | ORR (%) | TRAEs (%) | Year of Publication | Year of Approval (if Applicable) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Sorafenib vs. Placebo (SHARP) [6] | Sorafenib 400 mg BID | VEGFR, c-KIT, PDGFR, RET and Ras/Raf/MEK/ERK | First-line as monotherapy | 602 | Sorafenib vs placebo: 10.7 vs. 7.9 | HR 0.69; 95% CI 0.55 to 0.87; p < 0.001 | 5.5 vs. 2.8 | PRR 2% vs. 1% | Grade 3/4: diarrhoea 8% vs. 2%, HFS 8% vs. <1% | 2007 | 2007 |
| Sorafenib vs. Placebo (Asia–Pacific) [17] | Sorafenib 400 mg BID | VEGFR, c-KIT, PDGFR, RET and Ras/Raf/MEK/ERK | First-line as monotherapy | 226 | Sorafenib vs placebo: 6.5 vs. 4.2 | HR 0.68; 95% CI 0.50 to 0.93; p = 0.014 | 2.8 vs. 1.4 | PRR 3.3% | Grade 3/4: diarrhoea 6%, HFS 11%, fatigue 3% | 2007 | 2007 |
| Lenvatinib vs. Sorafenib (REFLECT) [21] | Lenvatinib 12 mg or 8 mg/d # | VEGFR, PDGFR, FGFR, KIT and RET | First-line as monotherapy | 954 | Lenvatinib vs. sorafenib: 13.6 vs. 12.3 | HR 0.92; 95% CI 0.79 to 1.06 | 7.4 vs. 3.7 | 24% vs. 9% | Hypertension 23% vs. 14%, HFS (37% vs. 52% any grade, 3% vs. 11% grade 3/4) | 2018 | 2018 (noninferiority trial) |
| Bevacizumab (B) and Atezolizumab (A) vs. Sorafenib (IMbrave150) [24] | Atezolizumab IV 1200 mg and bevacizumab IV 15 mg/kg Q3W | VEGF and PD-L1 | First-line combination therapy | 501 | B and A vs. sorafenib: 19.2 vs. 13.4 | HR 0.66; 95% CI 0.52 to 0.85; p = 0.0009 | 6.8 vs. 4.3 | 30% vs. 11% | Grade 3/4: 57% vs. 55% (no statistical difference) | 2020 | 2020 |
| Durvalumab and Tremelimumab (HIMALAYA) [34] | STRIDE: Tremelimumab 300 mg, Durvalumab 1500 mg and Durvalumab 1500 mg Q4W | PDL-1 and CTLA-4 | First-line combination therapy | 1171 | STRIDE: 16.4 D: 16.6 S: 13.8 | STRIDE vs. S: HR 0.78; 96% CI 0.65 to 0.92; p = 0.0035 | STRIDE: 3.8 D: 3.7 S: 4.1 (did not reach statistical significance) | STRIDE: 20.1% D: 17% S: 5.1% | Grade 3/4: STRIDE: 25.8% D: 12.9% S: 36.9% | 2022 | Under review |
| Atezolizumab (A) and Cabozantinib (C) (COSMIC-312) [29] | Cabozantinib 60 mg daily, sorafenib 400 BID, atezolizumab IV 1200 mg Q3W and cabozantinib 40 mg daily | PD-L1 and VEGF | First-line combination therapy | 837 | A and C: 15.4 Sorafenib: 15.5 | HR 0.90; 96% CI 0.69 to 1.18; p = 0.438 | A and C: 6.8 Sorafenib: 4.2 | A and C: 11% Sorafenib: 3.7% Cabozantinib: 6.4% | Grade 3/4: A and C: 54%; HFS 7.9%, hypertension 7%, AST elevation 6.5%, ALT elevation 6.3% Sorafenib: 32%; HFS 8.2%, hypertension 6.3%, AST elevation 2.4%, ALT elevation 1.9% | 2022 | Insignificant OS benefit, not for submission for approval |
| Agent | Regimen(s) | Targets | Indication | Sample Size | OS (Months) | OS (HR, 95% CI) | PFS (Months) | ORR (%) | TRAEs (%) | Year of Publication | Year of Approval |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Regorafenib vs. Placebo (RESORCE) [8] | Regorafenib 160 mg daily and BSC 1–3W Q4W | VEGFR, FGFR, PDGFR, B-RAF, RET and KIT | Second-line monotherapy in sorafenib-experienced patients | 573 | Regorafenib vs placebo: 10.6, vs. 7.8 | HR 0.63; 95% CI 0.50 to 0.79; p < 0·0001 | 3.1 vs. 1.5 | 11% vs. 4% | Grade 3/4 HFS 13% vs. 1%, hypertension 15% vs. 5%, diarrhea 3% vs. 0, fatigue 9% vs. 5% | 2017 | 2017 |
| Pembrolizumab (KEYNOTE 224) [12] | IV 200 mg Q 3W or 400 mg Q6W | PD-1 | Second-line monotherapy in sorafenib-experienced patients | 104 | 12.9 | - | 4.9 | 17% | Grade 3/4 18.6% vs. 7.5% | 2018 | 2018 |
| Pembrolizumab (KEYNOTE 240) [44] | Pembrolizumab IV 200 mg Q 3W or placebo and BSC | PD-1 | Second-line monotherapy in sorafenib-experienced patients | 413 | Pembrolizumab vs. placebo 13.9 vs. 10.6 (no statistical significance) | HR 0.78; 95% CI 0.611 to 0.998; p = 0.0238 | 3 vs. 2.8 (no statistical significance) | 18.3% vs. 4.4% | Grade 3/4: AST elevation (7%), ALT elevation (4%), fatigue (4%), hyperbilirubinaemia (1%) | 2018 | 2018 |
| Cabozantinib vs. Placebo (CELESTIAL) [9] | Cabozantinib 60 mg daily | VEGFR, AXL, c-MET, KIT and RET | Second-line monotherapy in sorafenib-experienced patients | 707 | Cabozantinib vs. placebo: 10.2 vs. 8.0 | HR 0.76; 95% CI 0.63 to 0.92; p = 0.005 | 5.2 vs. 1.9 | 4% vs. <1% | Grade 3/4: hypertension 16% vs. 2%, HFS 17% vs. 0, AST elevation 12% vs. 7%, fatigue 10% vs. 4%, diarrhea 10% vs. 2% | 2019 | 2019 |
| Ramucirumab vs. Placebo (REACH-2) [43] | Ramucirumab IV 8 mg/kg Q2W and BSC | VEGFR2 | Second-line monotherapy in sorafenib-experienced patients with AFP ≥ 400 ng/mL | 292 | Ramucirumab vs. placebo: 8.5 vs. 7.3 | HR 0.71; 95% CI 0·531 to 0·949; p = 0·0199 | 2.8 vs. 1.6 | 5% vs. 1% | Grade 3/4: Hypertension 13% vs. 5%, AST elevation 3% vs. 5%, hyponatremia 6% vs. 0; no HFS | 2019 | 2019 |
| Nivolumab and Ipilimumab (CHECKMATE 040) [13] | Nivolumab IV 1 mg/kg then same day ipilimumab IV 3 mg/kg, Q3W × 4 and nivolumab alone (240 mg Q2W or 480 mg Q4W) | PD-1 and CTLA-4 | Second-line combination therapy in sorafenib-experienced patients | 148 | arm A: 22.8 arm B: 12.5 arm C: 12.7 | - | arm A: 17 arm B: 22.2 arm C: 16.6 | Best ORR: 32% (arm A) | Adrenal insufficiency 18%, hypothyroidism 22%, rash 35%, pneumonitis 10%, colitis 10%, infusion-related reactions 8% | 2020 | 2020 |
| Pembrolizumab (KEYNOTE 394) [46] | Pembrolizumab IV 200 mg Q 3W or placebo and BSC | PD-1 | Second-line monotherapy in sorafenib-experienced patients | 453 | Pembrolizumab vs. placebo 14.6 vs. 13 | HR 0.79; 95% CI 0.63 to 0.99; p = 0.018 | 2.6 vs. 2.3 | 13.7% vs. 1.3% | Grade 3-5: 14.4% vs. 5.9% | 2022 | 2018 |
| Trial Name/ID | Phase | Regimen(s) | Targets | Comparator | Indication | Primary Endpoint(s) | Estimated Primary Completion Date |
|---|---|---|---|---|---|---|---|
| SHR-1210-III-310 | III | Camrelizumab and apatinib | PD-1 and VEGF | Sorafenib | First-line | PFS, OS | 21 December |
| NCT04444167 | I/II | AK104 (IV 6 mg/kg Q2W) and Lenvatinib # | PD-1/CTLA-4 and VEGF | N/A | First-line | ORR | 22 January |
| NCT03519997 | II | Pembrolizumab (IV 200 mg Q3W) and Bavituximab (IV 3 mg/kg weekly) | PD-1 and anti-phosphatidylserine | N/A | First-line | ORR | 22 April |
| RENOBATE | II | Nivolumab (IV 480 mg Q4W) and Regorafenib (po 80 mg daily for 21 consecutive days Q4W) | PD-1 and VEGF | N/A | First-line | ORR | 22 May |
| NCT04696055 | II | Pembrolizumab (IV 400 mg Q6W) and regorafenib (po 90 mg daily 3W on 1W off × 1 then 120 mg daily) | PD-1 and VEGF | N/A | Second-line | ORR | 22 May |
| NCT03418922 | I | Nivolumab and lenvatinib | PD-1 and VEGF | N/A | First-line | Safety | 22 June |
| LEAP-002 | III | Pembrolizumab (IV 200 mg Q3W) and Lenvatinib # | PD-1 and VEGF | Placebo and Lenvatinib # | First-line | PFS, OS | 22 July |
| NCT03941873 | I | Tislelizumab (IV 200 mg Q3W) and Sitravatinib (80 mg/120 mg daily) | PD-1 and VEGF | N/A | First or later lines | Safety | 22 August |
| IMMUNIB | II | Nivolumab (IV 240 MG Q2W up to 36 cycles) and Lenvatinib # | PD-1 and VEGF | N/A | First-line | ORR | 22 November |
| ORIENT-32 | II/III | Sintilimab (IV 200 mg Q2W) and IBI305 | PD-1 and VEGF | Sorafenib po 400 mg BID | First-line | PFS, OS | 22 December |
| CS1003-305 | III | CS1003 and lenvatinib | PD-1 and VEGF | CS1003 placebo and lenvatinib | First-line | PFS, OS | 23 June |
| AMETHISTA | IIIB | Atezolizumab (IV 1200 mg Q3W) and Bevacizumab (IV 15 mg/kg Q3W) | PDL-1 and VEGF | N/A | First-line | Grade 3 or worse NCI CTCAE v.5.0 Bleeding/ Hemorrhage | 23 July |
| NCT04442581 | II | Pembrolizumab and Cabozantinib | PD-1 and VEGF | N/A | First-line | ORR | 23 September |
| GOING | I/II | Nivolumab (1.5 mg/kg, 3 mg/kg or 240 mg Q2W) and Regorafenib (160 mg/day 3W on 1W off in the first 8W) | PD-1 and VEGF | N/A | Second-line | Safety | 23 December |
| NCT04183088 | II | Tislelizumab and Regorafenib | PD-1 and VEGF | Regorafenib ^ | First-line | Safety, ORR, PFS | 24 March |
| CHECKMATE 9DW | III | Nivolumab and ipilimumab | PD-1 and CTLA-4 | Sorafenib/lenvatinib | First-line | OS | 24 May |
| ALTN-AK105-III-02 | III | AK105 (IV 200 mg Q3W) and anlotinib (po 10 mg daily 2W on 1W off) | PD-1 and VEGF | Sorafenib po 400 mg BID | Second-line | OS | 24 June |
| IMbrave 251 | III | Atezolizumab (IV 1200 mg Q3W) and lenvatinib #/sorafenib (po 400 mg BID) | PDL-1 and VEGF | Lenvatinib #/sorafenib (po 400 mg BID) | Second-line | OS | 24 October |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feng, M.Y.; Chan, L.L.; Chan, S.L. Drug Treatment for Advanced Hepatocellular Carcinoma: First-Line and Beyond. Curr. Oncol. 2022, 29, 5489-5507. https://doi.org/10.3390/curroncol29080434
Feng MY, Chan LL, Chan SL. Drug Treatment for Advanced Hepatocellular Carcinoma: First-Line and Beyond. Current Oncology. 2022; 29(8):5489-5507. https://doi.org/10.3390/curroncol29080434
Chicago/Turabian StyleFeng, Maple Ye, Landon L. Chan, and Stephen Lam Chan. 2022. "Drug Treatment for Advanced Hepatocellular Carcinoma: First-Line and Beyond" Current Oncology 29, no. 8: 5489-5507. https://doi.org/10.3390/curroncol29080434
APA StyleFeng, M. Y., Chan, L. L., & Chan, S. L. (2022). Drug Treatment for Advanced Hepatocellular Carcinoma: First-Line and Beyond. Current Oncology, 29(8), 5489-5507. https://doi.org/10.3390/curroncol29080434





