The Role of Cytoreductive Nephrectomy in Renal Cell Carcinoma with Sarcomatoid Histology: A Case Series and Review of the Literature
Abstract
:1. Introduction
2. Case Presentation
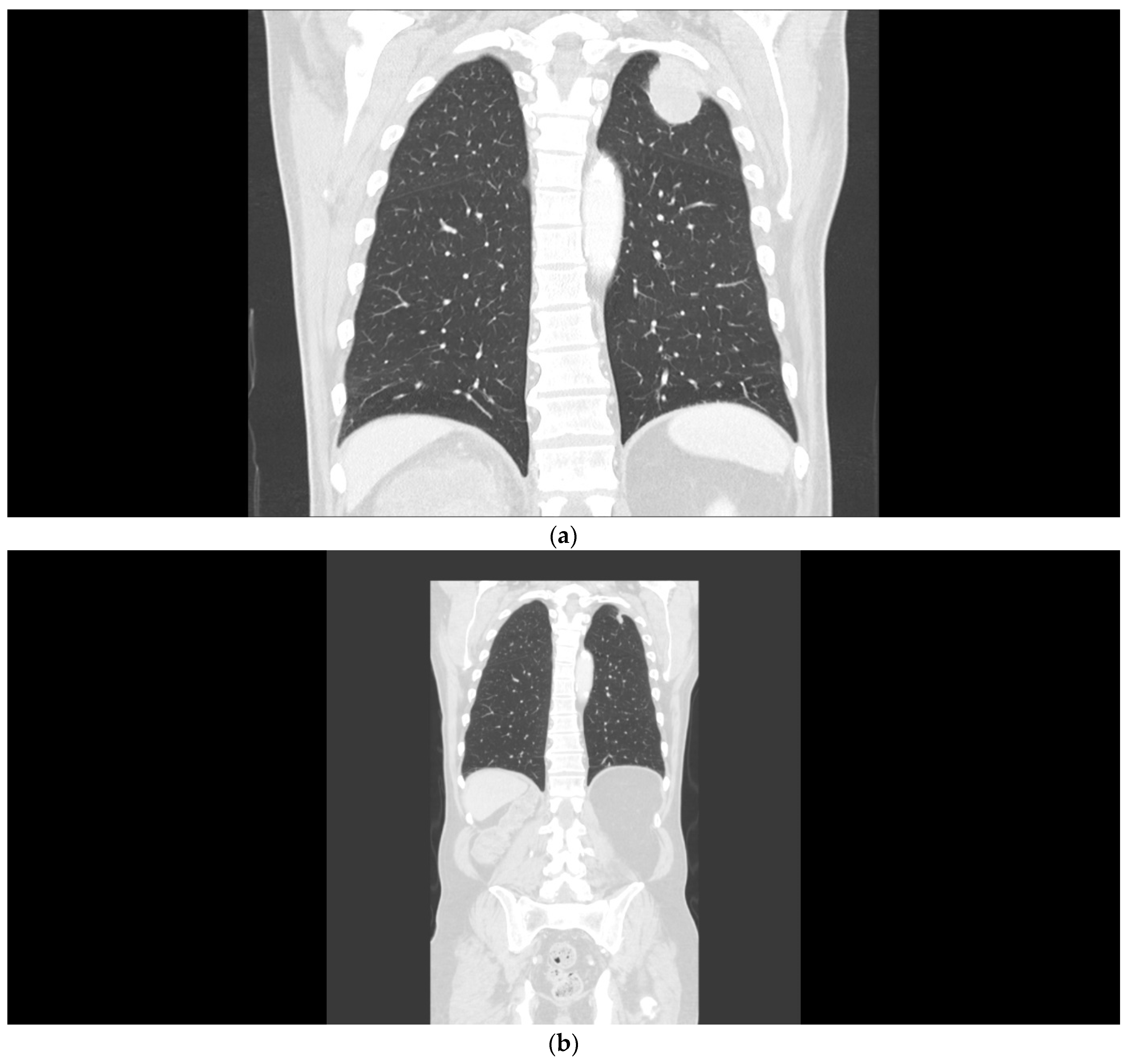
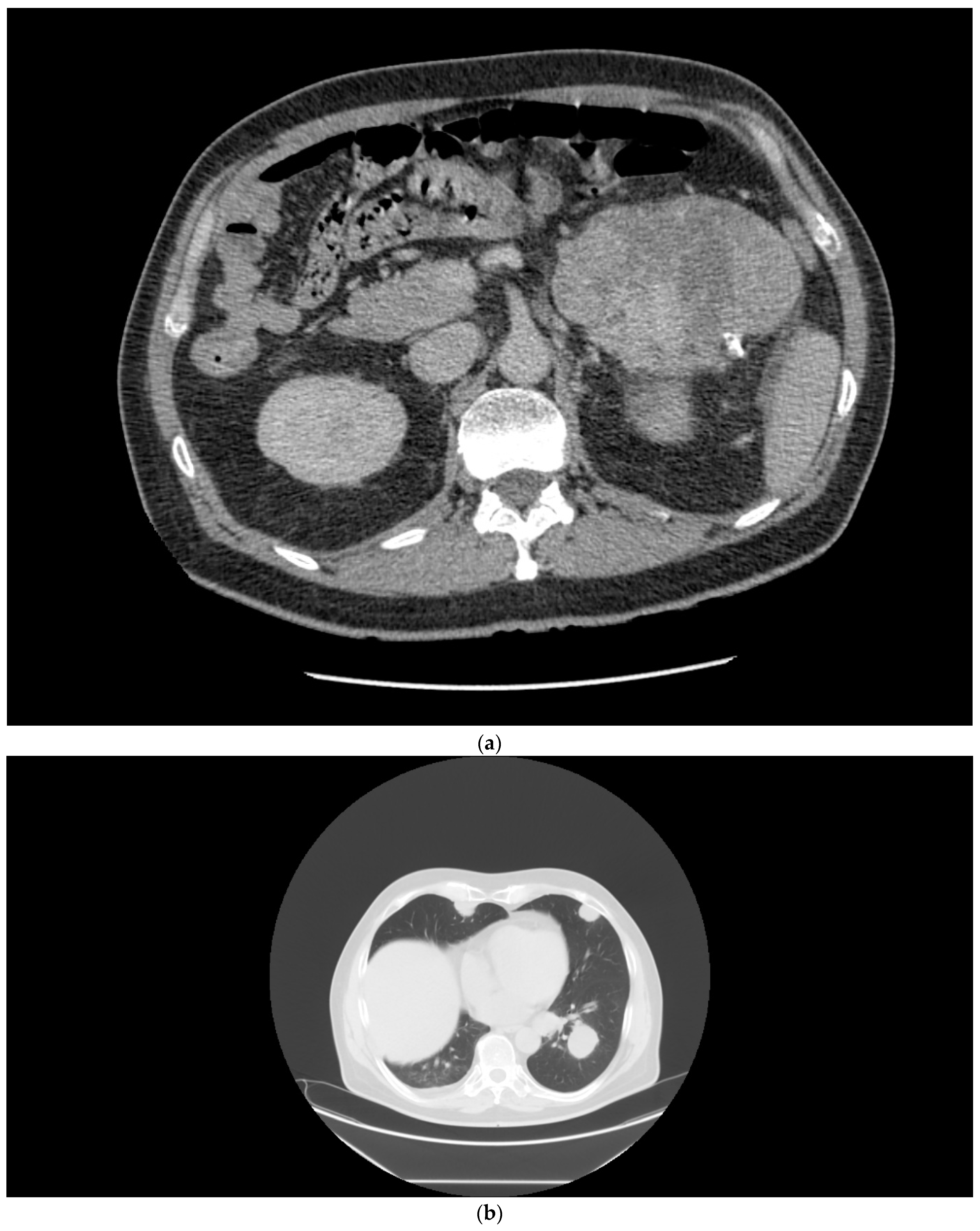
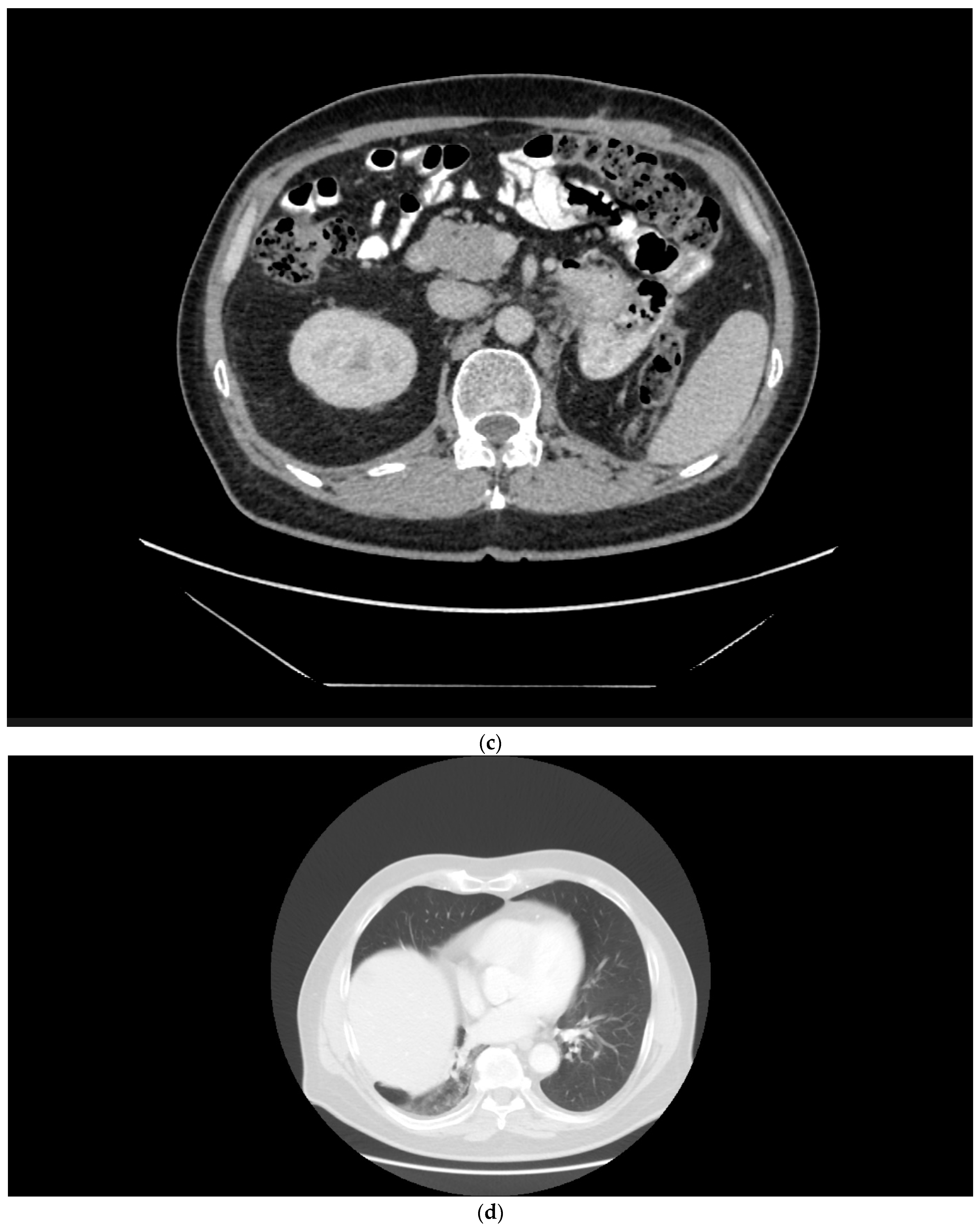
2.1. Case 1
2.2. Case 2
2.3. Case 3
2.4. Case 4
2.5. Case 5
2.6. Case 6
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.; Zargar-Shoshtari, K.; Dhillon, J.; Lin, H.Y.; Yue, B.; Fishman, M.; Sverrisson, E.F.; Spiess, P.E.; Gupta, S.; Poch, M.A.; et al. Using percentage of sarcomatoid differentiation as a prognostic factor in renal cell carcinoma. Clin. Genitourin. Cancer 2015, 13, 225–230. [Google Scholar] [CrossRef] [PubMed]
- de Velasco, G.; McKay, R.R.; Lin, X.; Moreira, R.B.; Simantov, R.; Choueiri, T.K. Comprehensive Analysis of Survival Outcomes in Non-Clear Cell Renal Cell Carcinoma Patients Treated in Clinical Trials. Clin. Genitourin. Cancer 2017, 15, 652–660.e651. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.Y.; Thompson, R.H.; Lohse, C.M.; Leibovich, B.C.; Boorjian, S.A.; Cheville, J.C.; Costello, B.A. A novel prognostic model for patients with sarcomatoid renal cell carcinoma. BJU Int. 2015, 115, 405–411. [Google Scholar] [CrossRef] [Green Version]
- Kyriakopoulos, C.E.; Chittoria, N.; Choueiri, T.K.; Kroeger, N.; Lee, J.L.; Srinivas, S.; Knox, J.J.; Bjarnason, G.A.; Ernst, S.D.; Wood, L.A.; et al. Outcome of patients with metastatic sarcomatoid renal cell carcinoma: Results from the International Metastatic Renal Cell Carcinoma Database Consortium. Clin. Genitourin. Cancer 2015, 13, e79–e85. [Google Scholar] [CrossRef]
- Adibi, M.; Thomas, A.Z.; Borregales, L.D.; Merrill, M.M.; Slack, R.S.; Chen, H.C.; Sircar, K.; Murugan, P.; Tamboli, P.; Jonasch, E.; et al. Percentage of sarcomatoid component as a prognostic indicator for survival in renal cell carcinoma with sarcomatoid dedifferentiation. Urol. Oncol. 2015, 33, 427.e17–427.e23. [Google Scholar] [CrossRef] [Green Version]
- Moch, H.; Cubilla, A.L.; Humphrey, P.A.; Reuter, V.E.; Ulbright, T.M. The 2016 WHO Classification of Tumours of the Urinary System and Male Genital Organs-Part A: Renal, Penile, and Testicular Tumours. Eur. Urol. 2016, 70, 93–105. [Google Scholar] [CrossRef]
- Debien, V.; Thouvenin, J.; Lindner, V.; Barthélémy, P.; Lang, H.; Flippot, R.; Malouf, G.G. Sarcomatoid Dedifferentiation in Renal Cell Carcinoma: From Novel Molecular Insights to New Clinical Opportunities. Cancers 2019, 12, 99. [Google Scholar] [CrossRef] [Green Version]
- Voss, M.H.; Bastos, D.A.; Karlo, C.A.; Ajeti, A.; Hakimi, A.A.; Feldman, D.R.; Hsieh, J.J.; Molina, A.M.; Patil, S.; Motzer, R.J. Treatment outcome with mTOR inhibitors for metastatic renal cell carcinoma with nonclear and sarcomatoid histologies. Ann. Oncol. 2014, 25, 663–668. [Google Scholar] [CrossRef]
- Alevizakos, M.; Gaitanidis, A.; Nasioudis, D.; Msaouel, P.; Appleman, L.J. Sarcomatoid Renal Cell Carcinoma: Population-Based Study of 879 Patients. Clin. Genitourin. Cancer 2019, 17, e447–e453. [Google Scholar] [CrossRef]
- Shuch, B.; Said, J.; La Rochelle, J.C.; Zhou, Y.; Li, G.; Klatte, T.; Kabbinaavar, F.F.; Pantuck, A.J.; Belldegrun, A.S. Cytoreductive nephrectomy for kidney cancer with sarcomatoid histology--is up-front resection indicated and, if not, is it avoidable? J. Urol. 2009, 182, 2164–2171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- von Minckwitz, G.; Untch, M.; Blohmer, J.U.; Costa, S.D.; Eidtmann, H.; Fasching, P.A.; Gerber, B.; Eiermann, W.; Hilfrich, J.; Huober, J.; et al. Definition and impact of pathologic complete response on prognosis after neoadjuvant chemotherapy in various intrinsic breast cancer subtypes. J. Clin. Oncol. 2012, 30, 1796–1804. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tannir, N.M.; Signoretti, S.; Choueiri, T.K.; McDermott, D.F.; Motzer, R.J.; Flaifel, A.; Pignon, J.C.; Ficial, M.; Frontera, O.A.; George, S.; et al. Efficacy and Safety of Nivolumab Plus Ipilimumab versus Sunitinib in First-line Treatment of Patients with Advanced Sarcomatoid Renal Cell Carcinoma. Clin. Cancer Res. 2021, 27, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Rini, B.I.; Plimack, E.R.; Stus, V.; Gafanov, R.; Hawkins, R.; Nosov, D.; Pouliot, F.; Soulieres, D.; Melichar, B.; Vynnychenko, I.; et al. Pembrolizumab (pembro) plus axitinib (axi) versus sunitinib as first-line therapy for metastatic renal cell carcinoma (mRCC): Outcomes in the combined IMDC intermediate/poor risk and sarcomatoid subgroups of the phase 3 KEYNOTE-426 study. J. Clin. Oncol. 2019, 37, 4500. [Google Scholar] [CrossRef]
- Rini, B.I.; Motzer, R.J.; Powles, T.; McDermott, D.F.; Escudier, B.; Donskov, F.; Hawkins, R.E.; Bracarda, S.; Bedke, J.; De Giorgi, U.; et al. Atezolizumab (atezo) plus bevacizumab (bev) versus sunitinib (sun) in pts with untreated metastatic renal cell carcinoma (mRCC) and sarcomatoid (sarc) histology: IMmotion151 subgroup analysis. J. Clin. Oncol. 2019, 37, 4512. [Google Scholar] [CrossRef]
- Choueiri, T.K.; Larkin, J.; Pal, S.; Motzer, R.J.; Rini, B.I.; Venugopal, B.; Alekseev, B.; Miyake, H.; Gravis, G.; Bilen, M.A.; et al. Efficacy and correlative analyses of avelumab plus axitinib versus sunitinib in sarcomatoid renal cell carcinoma: Post hoc analysis of a randomized clinical trial. ESMO Open 2021, 6, 100101. [Google Scholar] [CrossRef]
- Hwang, J.K.; Agarwal, N.; Brugarolas, J.; Zhang, T. Checking the Hippo in Sarcomatoid Renal Cell Carcinoma with Immunotherapy. Clin. Cancer Res. 2021, 27, 5–7. [Google Scholar] [CrossRef]
- Choueiri, T.K.; Larkin, J.M.G.; Pal, S.K.; Motzer, R.J.; Venugopal, B.; Alekseev, B.Y.; Miyake, H.; Gravis, G.; Bilen, M.A.; Chudnovsky, A.; et al. Efficacy and biomarker analysis of patients (pts) with advanced renal cell carcinoma (aRCC) with sarcomatoid histology (sRCC): Subgroup analysis from the phase III JAVELIN renal 101 trial of first-line avelumab plus axitinib (A plus Ax) vs sunitinib (S). Ann. Oncol. 2019, 30, 361. [Google Scholar] [CrossRef]
- Iacovelli, R.; Ciccarese, C.; Bria, E.; Bracarda, S.; Porta, C.; Procopio, G.; Tortora, G. Patients with sarcomatoid renal cell carcinoma—Re-defining the first-line of treatment: A meta-analysis of randomised clinical trials with immune checkpoint inhibitors. Eur. J. Cancer 2020, 136, 195–203. [Google Scholar] [CrossRef]
- Malouf, G.G.; Ali, S.M.; Wang, K.; Balasubramanian, S.; Ross, J.S.; Miller, V.A.; Stephens, P.J.; Khayat, D.; Pal, S.K.; Su, X.; et al. Genomic Characterization of Renal Cell Carcinoma with Sarcomatoid Dedifferentiation Pinpoints Recurrent Genomic Alterations. Eur. Urol. 2016, 70, 348–357. [Google Scholar] [CrossRef] [PubMed]
- Turajlic, S.; Xu, H.; Litchfield, K.; Rowan, A.; Horswell, S.; Chambers, T.; O’Brien, T.; Lopez, J.I.; Watkins, T.B.K.; Nicol, D.; et al. Deterministic Evolutionary Trajectories Influence Primary Tumor Growth: TRACERx Renal. Cell 2018, 173, 595–610.e511. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kawakami, F.; Sircar, K.; Rodriguez-Canales, J.; Fellman, B.M.; Urbauer, D.L.; Tamboli, P.; Tannir, N.M.; Jonasch, E.; Wistuba, I.I.; Wood, C.G.; et al. Programmed cell death ligand 1 and tumor-infiltrating lymphocyte status in patients with renal cell carcinoma and sarcomatoid dedifferentiation. Cancer 2017, 123, 4823–4831. [Google Scholar] [CrossRef] [PubMed]
- Joseph, R.W.; Millis, S.Z.; Carballido, E.M.; Bryant, D.; Gatalica, Z.; Reddy, S.; Bryce, A.H.; Vogelzang, N.J.; Stanton, M.L.; Castle, E.P.; et al. PD-1 and PD-L1 Expression in Renal Cell Carcinoma with Sarcomatoid Differentiation. Cancer Immunol. Res. 2015, 3, 1303–1307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, S.; Cheville, J.C.; Jungbluth, A.A.; Zhang, Y.; Zhang, L.; Chen, Y.B.; Tickoo, S.K.; Fine, S.W.; Gopalan, A.; Al-Ahmadie, H.A.; et al. JAK2/PD-L1/PD-L2 (9p24.1) amplifications in renal cell carcinomas with sarcomatoid transformation: Implications for clinical management. Mod. Pathol. 2019, 32, 1344–1358. [Google Scholar] [CrossRef]
- Yoshida, T.; Ohe, C.; Ikeda, J.; Atsumi, N.; Ohsugi, H.; Sugi, M.; Higasa, K.; Saito, R.; Tsuta, K.; Matsuda, T.; et al. Eosinophilic features in clear cell renal cell carcinoma correlate with outcomes of immune checkpoint and angiogenesis blockade. J. Immunother. Cancer 2021, 9, e002922. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Lu, R.; Kapur, P.; Jaiswal, B.S.; Hannan, R.; Zhang, Z.; Pedrosa, I.; Luke, J.J.; Zhang, H.; Goldstein, L.D.; et al. An Empirical Approach Leveraging Tumorgrafts to Dissect the Tumor Microenvironment in Renal Cell Carcinoma Identifies Missing Link to Prognostic Inflammatory Factors. Cancer Discov. 2018, 8, 1142–1155. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Z.; Kim, T.B.; Peng, B.; Karam, J.; Creighton, C.; Joon, A.; Kawakami, F.; Trevisan, P.; Jonasch, E.; Chow, C.W.; et al. Sarcomatoid Renal Cell Carcinoma Has a Distinct Molecular Pathogenesis, Driver Mutation Profile, and Transcriptional Landscape. Clin. Cancer Res. 2017, 23, 6686–6696. [Google Scholar] [CrossRef] [Green Version]
- Singla, N.; Ghandour, R.A.; Margulis, V. Is cytoreductive nephrectomy relevant in the immunotherapy era? Curr. Opin. Urol. 2019, 29, 526–530. [Google Scholar] [CrossRef] [PubMed]
- Singla, N.; Hakimi, A.A.; Margulis, V. Editorial: The evolving role of cytoreductive nephrectomy. Curr. Opin. Urol. 2019, 29, 505–506. [Google Scholar] [CrossRef]
- Escudier, B.; Porta, C.; Schmidinger, M.; Rioux-Leclercq, N.; Bex, A.; Khoo, V.; Grünwald, V.; Gillessen, S.; Horwich, A. Renal Cell Carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2019, 30, 706–720. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Conti, S.L.; Thomas, I.C.; Hagedorn, J.C.; Chung, B.I.; Chertow, G.M.; Wagner, T.H.; Brooks, J.D.; Srinivas, S.; Leppert, J.T. Utilization of cytoreductive nephrectomy and patient survival in the targeted therapy era. Int. J. Cancer 2014, 134, 2245–2252. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mathieu, R.; Pignot, G.; Ingles, A.; Crepel, M.; Bigot, P.; Bernhard, J.C.; Joly, F.; Guy, L.; Ravaud, A.; Azzouzi, A.R.; et al. Nephrectomy improves overall survival in patients with metastatic renal cell carcinoma in cases of favorable MSKCC or ECOG prognostic features. Urol. Oncol. 2015, 33, 339.e9–339.e15. [Google Scholar] [CrossRef] [PubMed]
- Heng, D.Y.; Wells, J.C.; Rini, B.I.; Beuselinck, B.; Lee, J.L.; Knox, J.J.; Bjarnason, G.A.; Pal, S.K.; Kollmannsberger, C.K.; Yuasa, T.; et al. Cytoreductive nephrectomy in patients with synchronous metastases from renal cell carcinoma: Results from the International Metastatic Renal Cell Carcinoma Database Consortium. Eur. Urol. 2014, 66, 704–710. [Google Scholar] [CrossRef] [PubMed]
- Choueiri, T.K.; Xie, W.; Kollmannsberger, C.; North, S.; Knox, J.J.; Lampard, J.G.; McDermott, D.F.; Rini, B.I.; Heng, D.Y.C. The Impact of Cytoreductive Nephrectomy on Survival of Patients with Metastatic Renal Cell Carcinoma Receiving Vascular Endothelial Growth Factor Targeted Therapy. J. Urol. 2011, 185, 60–66. [Google Scholar] [CrossRef]
- Alhalabi, O.; Karam, J.A.; Tannir, N.M. Evolving role of cytoreductive nephrectomy in metastatic renal cell carcinoma of variant histology. Curr. Opin. Urol. 2019, 29, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Kassouf, W.; Sanchez-Ortiz, R.; Tamboli, P.; Tannir, N.; Jonasch, E.; Merchant, M.M.; Matin, S.; Swanson, D.A.; Wood, C.G. Cytoreductive nephrectomy for metastatic renal cell carcinoma with nonclear cell histology. J. Urol. 2007, 178, 1896–1900. [Google Scholar] [CrossRef]
- Blum, K.A.; Gupta, S.; Tickoo, S.K.; Chan, T.A.; Russo, P.; Motzer, R.J.; Karam, J.A.; Hakimi, A.A. Sarcomatoid renal cell carcinoma: Biology, natural history and management. Nat. Rev. Urol. 2020, 17, 659–678. [Google Scholar] [CrossRef] [PubMed]
- Adashek, J.J.; Zhang, Y.; Skelton, W.P.t.; Bilotta, A.; Chahoud, J.; Zemp, L.; Li, J.; Dhillon, J.; Manley, B.; Spiess, P.E. Dissecting Outcomes: Should Cytoreductive Nephrectomy Be Performed for Patients with Metastatic Renal Cell Carcinoma with Sarcomatoid Dedifferentiation? Front. Oncol. 2020, 10, 627025. [Google Scholar] [CrossRef]
- Singla, N.; Hutchinson, R.C.; Ghandour, R.A.; Freifeld, Y.; Fang, D.; Sagalowsky, A.I.; Lotan, Y.; Bagrodia, A.; Margulis, V.; Hammers, H.J.; et al. Improved survival after cytoreductive nephrectomy for metastatic renal cell carcinoma in the contemporary immunotherapy era: An analysis of the National Cancer Database. Urol. Oncol. 2020, 38, 604.e9–604.e17. [Google Scholar] [CrossRef]
- Dilme, R.V.; Rivas, J.G.; Campi, R.; Puente, J.; Jerez, T.; Enikeev, D.; Esperto, F.; Sierra, J.M. Cytoreductive Nephrectomy in the Management of Metastatic Renal Cell Carcinoma: Is There Still a Debate? Curr. Urol. Rep. 2021, 22, 54. [Google Scholar] [CrossRef]
- Mejean, A.; Ravaud, A.; Thezenas, S.; Colas, S.; Beauval, J.B.; Bensalah, K.; Geoffrois, L.; Thiery-Vuillemin, A.; Cormier, L.; Lang, H.; et al. Sunitinib Alone or after Nephrectomy in Metastatic Renal-Cell Carcinoma. N. Engl. J. Med. 2018, 379, 417–427. [Google Scholar] [CrossRef] [PubMed]
- Bex, A.; Albiges, L.; Ljungberg, B.; Bensalah, K.; Dabestani, S.; Giles, R.H.; Hofmann, F.; Hora, M.; Kuczyk, M.A.; Lam, T.B.; et al. Updated European Association of Urology Guidelines for Cytoreductive Nephrectomy in Patients with Synchronous Metastatic Clear-cell Renal Cell Carcinoma. Eur. Urol. 2018, 74, 805–809. [Google Scholar] [CrossRef] [PubMed]
- Bex, A.; Mulders, P.; Jewett, M.; Wagstaff, J.; van Thienen, J.V.; Blank, C.U.; van Velthoven, R.; Del Pilar Laguna, M.; Wood, L.; van Melick, H.H.E.; et al. Comparison of Immediate vs Deferred Cytoreductive Nephrectomy in Patients with Synchronous Metastatic Renal Cell Carcinoma Receiving Sunitinib: The SURTIME Randomized Clinical Trial. JAMA Oncol. 2019, 5, 164–170. [Google Scholar] [CrossRef]
- Abu-Ghanem, Y.; van Thienen, J.V.; Blank, C.; Aarts, M.J.B.; Jewett, M.; de Jong, I.J.; Lattouf, J.B.; van Melick, H.H.E.; Wood, L.; Mulders, P.; et al. Cytoreductive nephrectomy and exposure to sunitinib—A post hoc analysis of the Immediate Surgery or Surgery after Sunitinib Malate in Treating Patients with Metastatic Kidney Cancer (SURTIME) trial. BJU Int. 2022, 130, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Meerveld-Eggink, A.; Graafland, N.; Wilgenhof, S.; Van Thienen, J.V.; Lalezari, F.; Grant, M.; Szabados, B.; Abu-Ghanem, Y.; Kuusk, T.; Boleti, E.; et al. Primary Renal Tumour Response in Patients Treated with Nivolumab and Ipilimumab for Metastatic Renal Cell Carcinoma: Real-world Data Assessment. Eur. Urol. Open. Sci. 2022, 35, 54–58. [Google Scholar] [CrossRef] [PubMed]
- Chakiryan, N.H.; Gore, L.R.; Reich, R.R.; Dunn, R.L.; Jiang, D.D.; Gillis, K.A.; Green, E.; Hajiran, A.; Hugar, L.; Zemp, L.; et al. Survival Outcomes Associated with Cytoreductive Nephrectomy in Patients with Metastatic Clear Cell Renal Cell Carcinoma. JAMA Netw. Open 2022, 5, e2212347. [Google Scholar] [CrossRef]
- Labbate, C.; Hatogai, K.; Werntz, R.; Stadler, W.M.; Steinberg, G.D.; Eggener, S.; Sweis, R.F. Complete response of renal cell carcinoma vena cava tumor thrombus to neoadjuvant immunotherapy. J. Immunother. Cancer 2019, 7, 66. [Google Scholar] [CrossRef] [Green Version]
- Pignot, G.; Thiery-Vuillemin, A.; Walz, J.; Lang, H.; Bigot, P.; Werle, P.; Balssa, L.; Geoffrois, L.; Leblanc, L.; Albigès, L.; et al. Nephrectomy after Complete Response to Immune Checkpoint Inhibitors for Metastatic Renal Cell Carcinoma: A New Surgical Challenge? Eur. Urol. 2020, 77, 761–763. [Google Scholar] [CrossRef]
- Kuusk, T.; Abu-Ghanem, Y.; Mumtaz, F.; Powles, T.; Bex, A. Perioperative therapy in renal cancer in the era of immune checkpoint inhibitor therapy. Curr. Opin. Urol. 2021, 31, 262–269. [Google Scholar] [CrossRef]
- Sejima, T.; Masago, T.; Yoshida, M.; Nishi, T.; Kawabata, Y.; Tajima, Y.; Yumioka, T.; Honda, M.; Takenaka, A. Pathological eradication of recurrent metastatic renal cell carcinoma with sarcomatoid component by nivolumab plus ipilimumab combination therapy. Int. Cancer Conf. J. 2021, 10, 285–289. [Google Scholar] [CrossRef]
- Park, J.J.; Kellezi, O.; Hamasha, R.; Ali, A.; Alva, A.S. Immunotherapy in metastatic sarcomatoid renal cell carcinoma: A single institution experience. Cancer Treat. Res. Commun. 2020, 25, 100251. [Google Scholar] [CrossRef] [PubMed]
| Patient | Age (Years) | Sex (Male/ Female) | IMDC Score | Initial Stage | Size of the Primary Tumor (mm) | % Sarcomatoid Component | Number of Metastatic Sites | Treatment Duration (Days) | Time from CN to ICI Initiation (Days) | Survival from Initial Diagnosis (Months)/Latest Control | Toxicity |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 68 | M | 5 | T2bN1M1 | 100 | 100 | 3 | 720+ | 22 | 26+/(June 2022) | dermatitis |
| 2 | 58 | M | 5 | T3aN1M0 | 70 | 100 | 2 | 2419 | 99 | 90+/(May 2022) | hepatotoxicity |
| 3 | 53 | M | 3 | T3aN1M1 | 90 | 80 | 1 | 21 | 36 | 25+/(July 2022) | hepatotoxicity |
| 4 | 70 | M | 6 | T3cN1M1 | 120 | NE | 1 | 10 | NA | 0.3 | none |
| 5 | 71 | F | 1 | T3aN0M1 | 75 | 75 | 1 | 188 | 144 | 35+/(May 2022) | none |
| 6 | 66 | M | 1 | T3aN1M1 | 42 | 95 | 1 | 42 | 913 | 56 | colitis |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Studentova, H.; Rusarova, N.; Ondruskova, A.; Zemankova, A.; Student, V., Jr.; Skanderova, D.; Melichar, B. The Role of Cytoreductive Nephrectomy in Renal Cell Carcinoma with Sarcomatoid Histology: A Case Series and Review of the Literature. Curr. Oncol. 2022, 29, 5475-5488. https://doi.org/10.3390/curroncol29080433
Studentova H, Rusarova N, Ondruskova A, Zemankova A, Student V Jr., Skanderova D, Melichar B. The Role of Cytoreductive Nephrectomy in Renal Cell Carcinoma with Sarcomatoid Histology: A Case Series and Review of the Literature. Current Oncology. 2022; 29(8):5475-5488. https://doi.org/10.3390/curroncol29080433
Chicago/Turabian StyleStudentova, Hana, Nikol Rusarova, Andrea Ondruskova, Anezka Zemankova, Vladimir Student, Jr., Daniela Skanderova, and Bohuslav Melichar. 2022. "The Role of Cytoreductive Nephrectomy in Renal Cell Carcinoma with Sarcomatoid Histology: A Case Series and Review of the Literature" Current Oncology 29, no. 8: 5475-5488. https://doi.org/10.3390/curroncol29080433
APA StyleStudentova, H., Rusarova, N., Ondruskova, A., Zemankova, A., Student, V., Jr., Skanderova, D., & Melichar, B. (2022). The Role of Cytoreductive Nephrectomy in Renal Cell Carcinoma with Sarcomatoid Histology: A Case Series and Review of the Literature. Current Oncology, 29(8), 5475-5488. https://doi.org/10.3390/curroncol29080433





