Sequential Use of Carfilzomib and Pomalidomide in Relapsed Multiple Myeloma: A Report from the Canadian Myeloma Research Group (CMRG) Database
Abstract
:1. Introduction
2. Materials and Methods
2.1. CMRG Database
2.2. Patients
2.3. Endpoints and Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Palumbo, A.; Anderson, K. Multiple myeloma. N. Engl. J. Med. 2011, 364, 1046–1060. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, S.K.; Therneau, T.M.; Gertz, M.A.; Lacy, M.Q.; Dispenzieri, A.; Rajkumar, S.V.; Fonseca, R.; Witzig, T.E.; Lust, J.A.; Larson, D.R.; et al. Clinical course of patients with relapsed multiple myeloma. Mayo Clin. Proc. 2004, 79, 867–874. [Google Scholar] [CrossRef] [PubMed]
- Binder, M.; Nandakumar, B.; Rajkumar, S.V.; Kapoor, P.; Buadi, F.K.; Dingli, D.; Lacy, M.Q.; Gertz, M.A.; Hayman, S.R.; Leung, N.; et al. Mortality trends in multiple myeloma after the introduction of novel therapies in the United States. Leukemia 2021, 36, 801–808. [Google Scholar] [CrossRef] [PubMed]
- San Miguel, J.F.; Schlag, R.; Khuageva, N.K.; Dimopoulos, M.A.; Shpilberg, O.; Kropff, M.; Spicka, I.; Petrucci, M.T.; Palumbo, A.; Samoilova, O.S.; et al. Bortezomib plus melphalan and prednisone for initial treatment of multiple myeloma. N. Engl. J. Med. 2008, 359, 906–917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benboubker, L.; Dimopoulos, M.A.; Dispenzieri, A.; Catalano, J.; Belch, A.R.; Cavo, M.; Pinto, A.; Weisel, K.; Ludwig, H.; Bahlis, N.; et al. Lenalidomide and dexamethasone in transplant-ineligible patients with myeloma. N. Engl. J. Med. 2014, 371, 906–917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dimopoulos, M.A.; Oriol, A.; Nahi, H.; San-Miguel, J.; Bahlis, N.J.; Usmani, S.Z.; Rabin, N.; Orlowski, R.Z.; Komarnicki, M.; Suzuki, K.; et al. Daratumumab, Lenalidomide, and Dexamethasone for Multiple Myeloma. N. Engl. J. Med. 2016, 375, 1319–1331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Palumbo, A.; Chanan-Khan, A.; Weisel, K.; Nooka, A.K.; Masszi, T.; Beksac, M.; Spicka, I.; Hungria, V.; Munder, M.; Mateos, M.V.; et al. Daratumumab, Bortezomib, and Dexamethasone for Multiple Myeloma. N. Engl. J. Med. 2016, 375, 754–766. [Google Scholar] [CrossRef] [PubMed]
- Dimopoulos, M.A.; Moreau, P.; Palumbo, A.; Joshua, D.; Pour, L.; Hájek, R.; Facon, T.; Ludwig, H.; Oriol, A.; Goldschmidt, H.; et al. Carfilzomib and dexamethasone versus bortezomib and dexamethasone for patients with relapsed or refractory multiple myeloma (ENDEAVOR): A randomised, phase 3, open-label, multicentre study. Lancet Oncol. 2016, 17, 27–38. [Google Scholar] [CrossRef]
- Miguel, J.S.; Weisel, K.; Moreau, P.; Lacy, M.; Song, K.; Delforge, M.; Karlin, L.; Goldschmidt, H.; Banos, A.; Oriol, A.; et al. Pomalidomide plus low-dose dexamethasone versus high-dose dexamethasone alone for patients with relapsed and refractory multiple myeloma (MM-003): A randomised, open-label, phase 3 trial. Lancet Oncol. 2013, 14, 1055–1066. [Google Scholar] [CrossRef] [Green Version]
- Moreau, P.; Joshua, D.; Chng, W.J.; Palumbo, A.; Goldschmidt, H.; Hájek, R.; Facon, T.; Ludwig, H.; Pour, L.; Niesvizky, R.; et al. Impact of prior treatment on patients with relapsed multiple myeloma treated with carfilzomib and dexamethasone vs bortezomib and dexamethasone in the phase 3 ENDEAVOR study. Leukemia 2017, 31, 115–122. [Google Scholar] [CrossRef]
- Orlowski, R.Z.; Moreau, P.; Niesvizky, R.; Ludwig, H.; Oriol, A.; Chng, W.J.; Goldschmidt, H.; Yang, Z.; Kimball, A.S.; Dimopoulos, M. Carfilzomib-Dexamethasone Versus Bortezomib-Dexamethasone in Relapsed or Refractory Multiple Myeloma: Updated Overall Survival, Safety, and Subgroups. Clin. Lymphoma Myeloma Leuk. 2019, 19, 522–530.e1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hájek, R.; Masszi, T.; Petrucci, M.T.; Palumbo, A.; Rosiñol, L.; Nagler, A.; Yong, K.L.; Oriol, A.; Minarik, J.; Pour, L.; et al. A randomized phase III study of carfilzomib vs low-dose corticosteroids with optional cyclophosphamide in relapsed and refractory multiple myeloma (FOCUS). Leukemia 2017, 31, 107–114. [Google Scholar] [CrossRef] [PubMed]
| Baseline Characteristics | All | CAR-POM | POM-CAR |
|---|---|---|---|
| (n = 121) | (n = 49) | (n = 72) | |
| Age at Diagnosis, median (range) | 60 (34–82) | 63 (41–80) | 56 (34–82) |
| Age at Initiation of first therapy CAR/POM, median (range) | 65 (39–84) | 67 (48–82) | 64 (39–84) |
| Male, N (%) | 71 (58.7) | 26 (53.1) | 45 (62.5) |
| MM Subtype, N (%) | |||
| IgG | 68 (59.1) | 28 (60.9) | 40 (58.0) |
| IgA | 30 (26.1) | 13 (28.3) | 17 (24.6) |
| IgD | 1 (0.9) | 0 (0) | 1 (1.5) |
| FLC | 16 (13.9) | 5 (10.9) | 11 (15.9) |
| Unknown | 6 | 3 | 3 |
| ISS Stage, N (%) | |||
| I | 27 (29.6) | 8 (19.5) | 19 (34.6) |
| II | 35 (39.0) | 16 (39.0) | 19 (34.6) |
| III | 34 (31.5) | 17 (41.5) | 17 (30.9) |
| Unknown | 25 | 8 | 17 |
| Previous Treatment, N (%) | |||
| Lenalidomide exposed | 111 (91.7) | 41 (83.7) | 70 (97.2) |
| Lenalidomide refractory | 96 (79.3) | 33 (67.3) | 73 (87.5) |
| Daratumumab exposed | 8 (6.6) | 5 (10.2) | 3 (4.2) |
| Cytogenetics *, N (%) | |||
| High risk | 24 (25.5) | 15 (42.9) | 9 (15.3) |
| Standard risk | 70 (74.5) | 20 (57.1) | 50 (84.8) |
| Unknown | 27 | 14 | 13 |
| POM-based Regimen | |||
| PomC | 2 (1.7) | 1 (2.0) | 1 (1.4) |
| PomCD/P | 62 (51.2) | 36 (73.5) | 26 (36.1) |
| PomD | 53 (43.8) | 11 (22.5) | 42 (58.3) |
| PomVD | 4 (3.3) | 1 (2.0) | 3 (4.2) |
| CAR-based Regimen | |||
| K | 7 (5.8) | 1 (2.0) | 6 (8.3) |
| KCD | 20 (16.5) | 12 (24.5) | 8 (11.1) |
| KD/P | 76 (62.8) | 22 (44.9) | 54 (75.0) |
| KRD | 18(14.9) | 14 (28.6) | 4 (5.6) |
| Line of First Treatment, median (range) | 3 (2–9) | 3 (2–6) | 4 (2–9) |
| Subsequent Treatments, N (%) | |||
| Lenalidomide | 8 (6.6) | 4 (8.2) | 4 (5.6) |
| Pomalidomide | 71 (58.7) | 49 (100) | 22 (30.6) |
| Bortezomib | 20 (16.5) | 6 (12.2) | 14 (19.4) |
| Carfilzomib | 76 (62.8) | 4 (8.2) | 72 (100) |
| Ixazomib | 12 (9.9) | 3 (6.1) | 9 (12.5) |
| Anti-CD38 | 43 (35.5) | 16 (32.7) | 27 (37.5) |
| Anti-BCMA | 9 (7.4) | 3 (6.1) | 6 (8.3) |
| Therapy Response | All (n = 121) | CAR-POM (n = 49) | POM-CAR (n = 72) |
|---|---|---|---|
| Carfilzomib-based | |||
| ORR (≥PR), n (%) * | 62 (52.1) | 29 (62.0) | 33 (47.1) |
| ≥VGPR, n (%) | 25 (21.0) α | 18 (38.3) β | 7 (10.0) β |
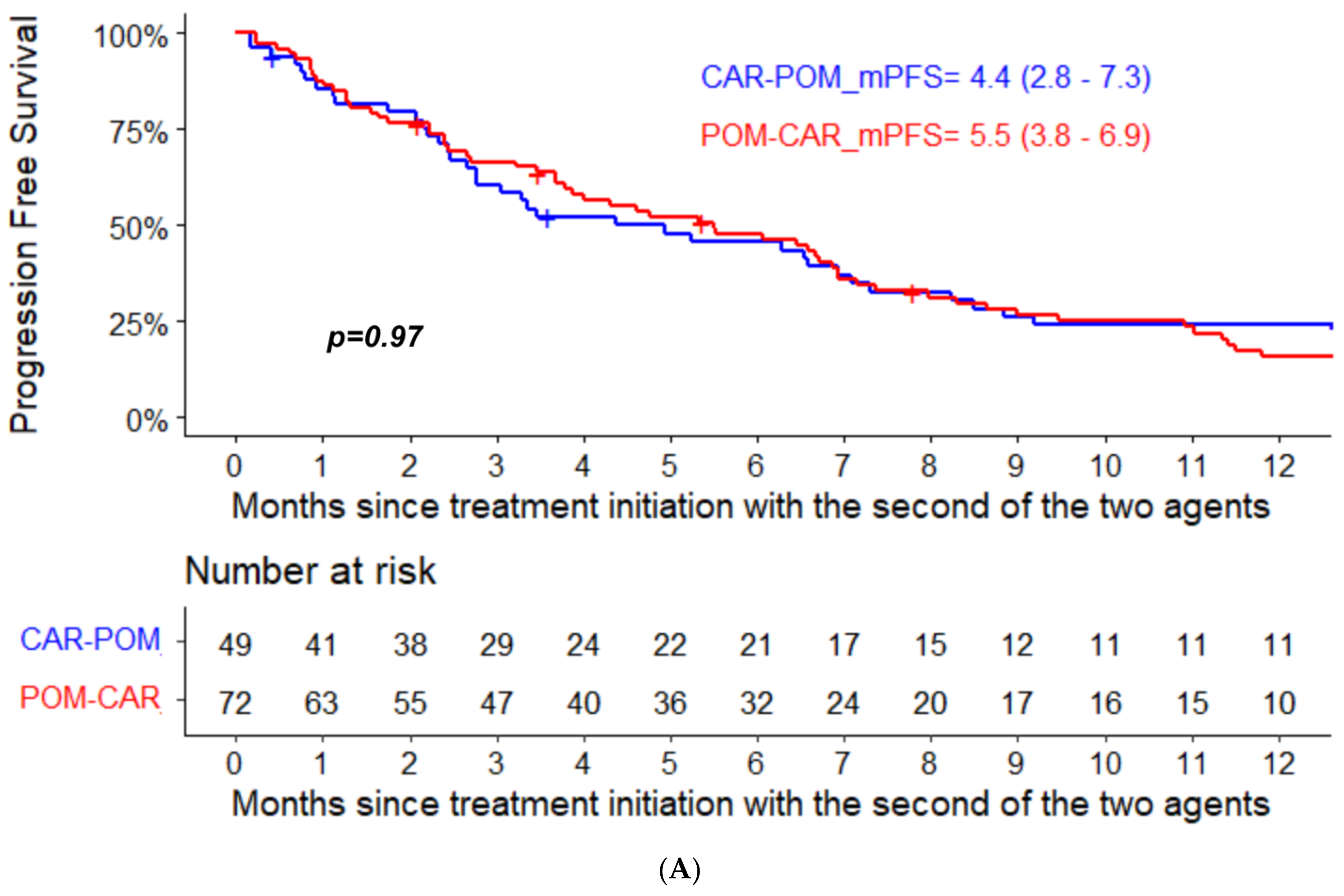
| Median PFS (95% CI) | 6.4 (5.1–7.4) | 7.4 (5.9–11.3) | 5.5 (3.8–6.9) |
| Pomalidomide-based | |||
| ORR (≥PR), n (%) | 57 (49.6) | 22 (48.9) | 35 (50.0) |
| ≥VGPR, n (%) | 19 (16.5) α | 5 (11.1) γ | 14 (20.3) γ |
| Median PFS (95% CI) | 5.7 (4.0–8.2) | 4.4 (2.8–7.3) | 6.4 (4.0–12.8) |
| ** | All (n = 78) | CAR-POM (n = 40) | POM-CAR (n = 38) |
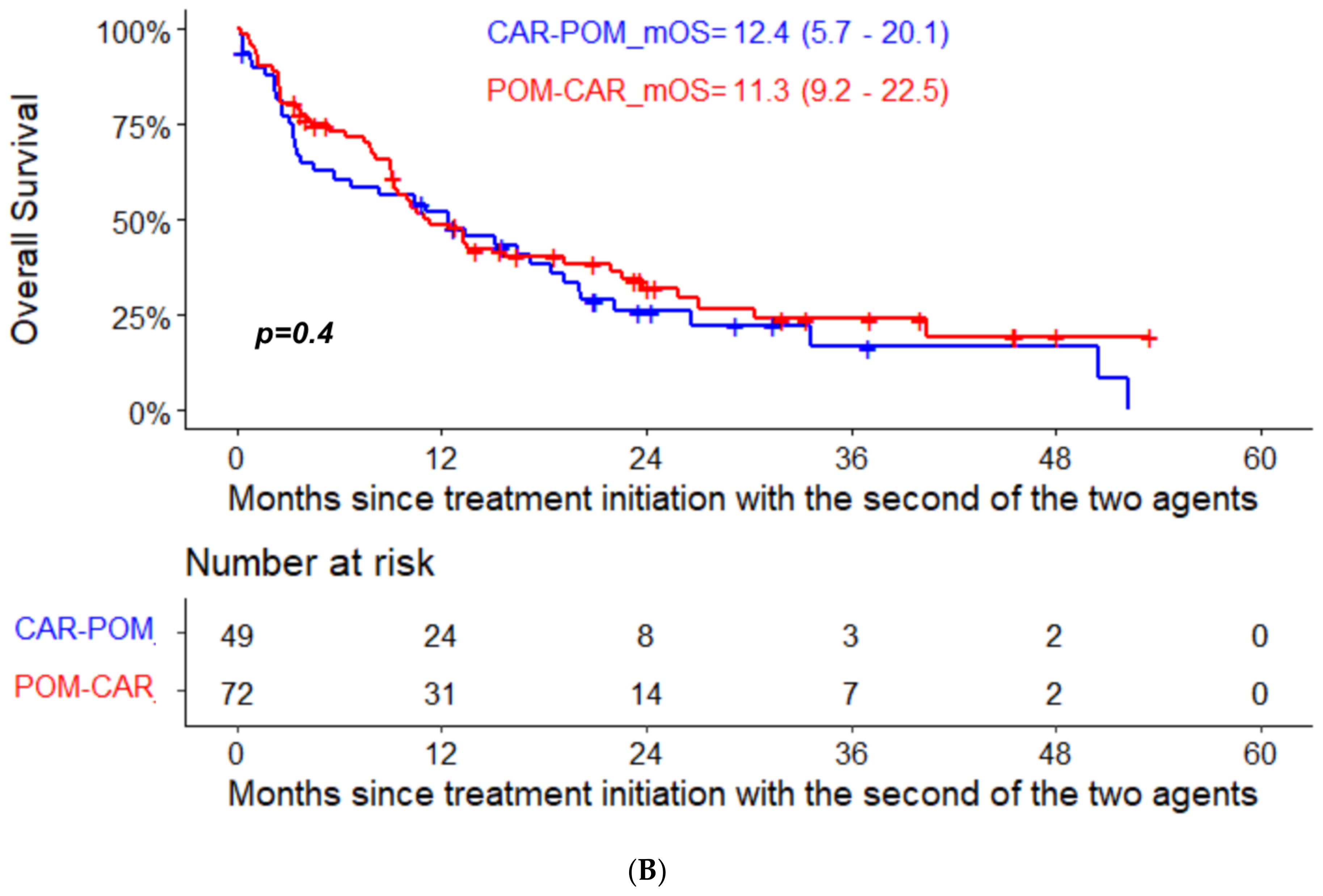
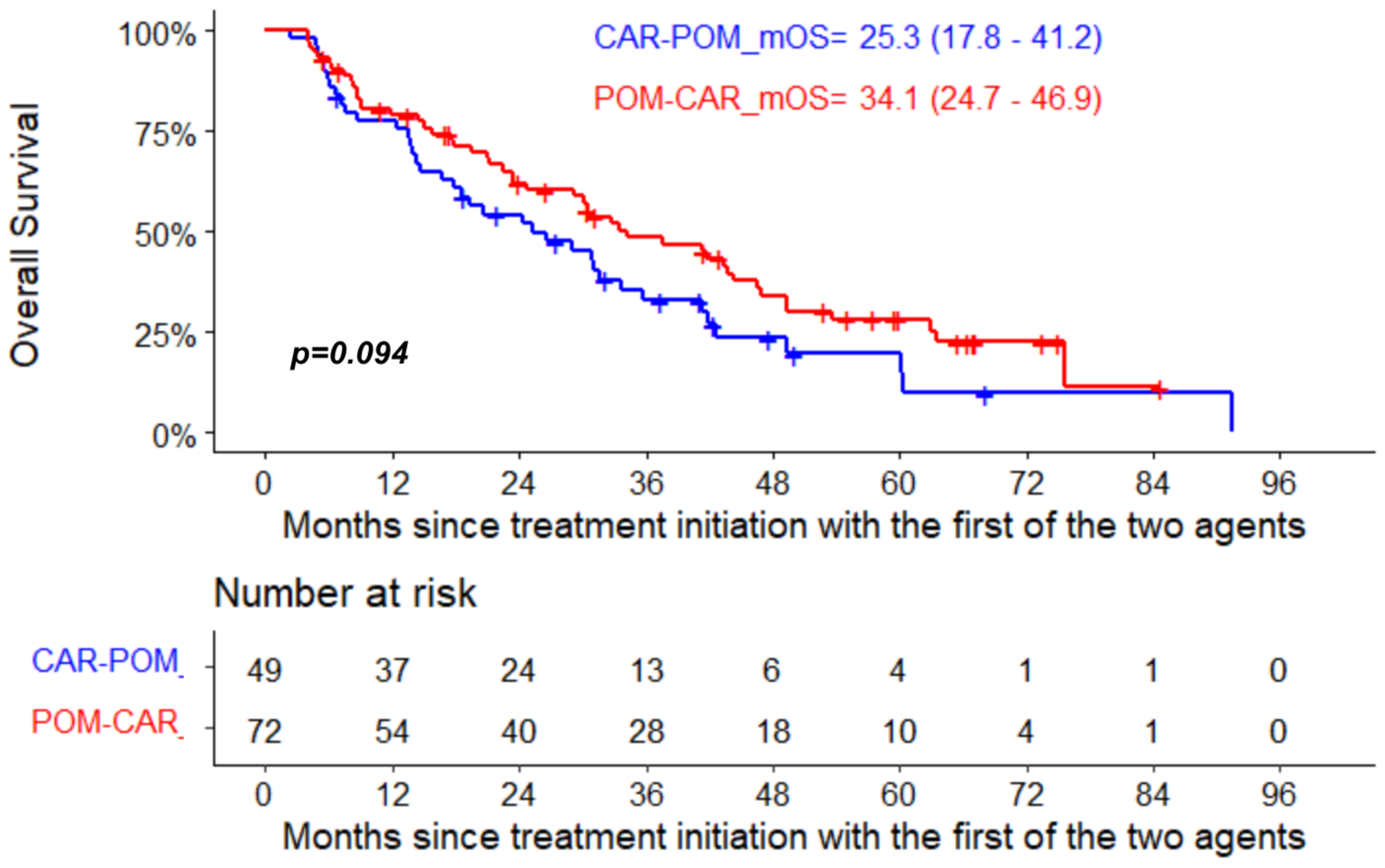
| Median PFS 2 (95% CI) ** | 13.0 (10.4–24.2) | 13.2 (10.4–24.2) | 13.0 (8.2–31.1) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
McCurdy, A.; Venner, C.P.; Masih-Khan, E.; Louzada, M.; LeBlanc, R.; Sebag, M.; Song, K.; Jimenez-Zepeda, V.H.; Kotb, R.; Kardjadj, M.; et al. Sequential Use of Carfilzomib and Pomalidomide in Relapsed Multiple Myeloma: A Report from the Canadian Myeloma Research Group (CMRG) Database. Curr. Oncol. 2022, 29, 1575-1582. https://doi.org/10.3390/curroncol29030132
McCurdy A, Venner CP, Masih-Khan E, Louzada M, LeBlanc R, Sebag M, Song K, Jimenez-Zepeda VH, Kotb R, Kardjadj M, et al. Sequential Use of Carfilzomib and Pomalidomide in Relapsed Multiple Myeloma: A Report from the Canadian Myeloma Research Group (CMRG) Database. Current Oncology. 2022; 29(3):1575-1582. https://doi.org/10.3390/curroncol29030132
Chicago/Turabian StyleMcCurdy, Arleigh, Christopher P. Venner, Esther Masih-Khan, Martha Louzada, Richard LeBlanc, Michael Sebag, Kevin Song, Victor H. Jimenez-Zepeda, Rami Kotb, Moustafa Kardjadj, and et al. 2022. "Sequential Use of Carfilzomib and Pomalidomide in Relapsed Multiple Myeloma: A Report from the Canadian Myeloma Research Group (CMRG) Database" Current Oncology 29, no. 3: 1575-1582. https://doi.org/10.3390/curroncol29030132
APA StyleMcCurdy, A., Venner, C. P., Masih-Khan, E., Louzada, M., LeBlanc, R., Sebag, M., Song, K., Jimenez-Zepeda, V. H., Kotb, R., Kardjadj, M., Mian, H., White, D., Stakiw, J., Aslam, M., Reiman, A., Gul, E., & Reece, D. (2022). Sequential Use of Carfilzomib and Pomalidomide in Relapsed Multiple Myeloma: A Report from the Canadian Myeloma Research Group (CMRG) Database. Current Oncology, 29(3), 1575-1582. https://doi.org/10.3390/curroncol29030132





