Lessons from Cost-Effectiveness Analysis of Smoking Cessation Programs for Cancer Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Methods for Cost-Effectiveness Analysis
2.2. Methods for Review of the Scientific Literature
3. Results
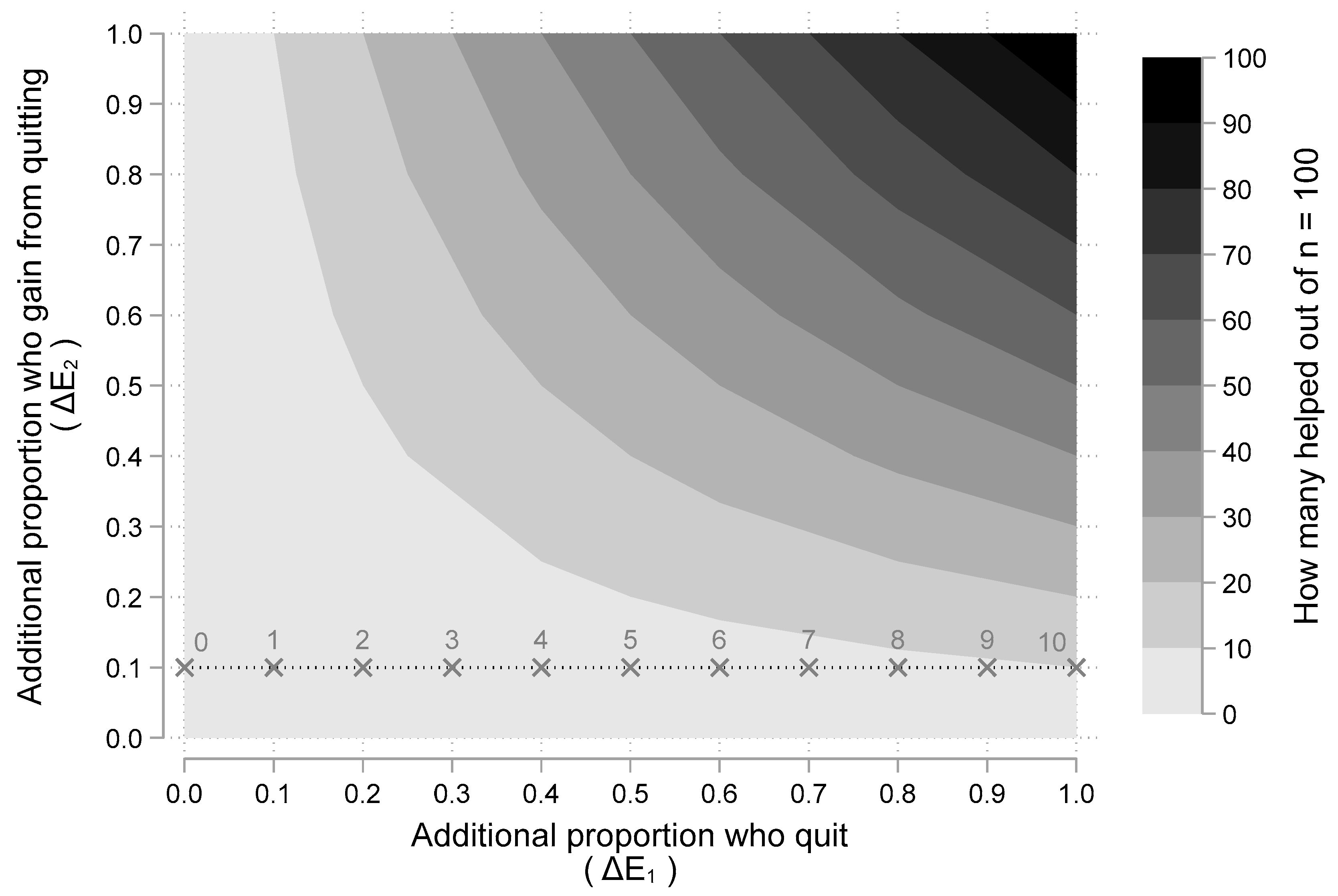
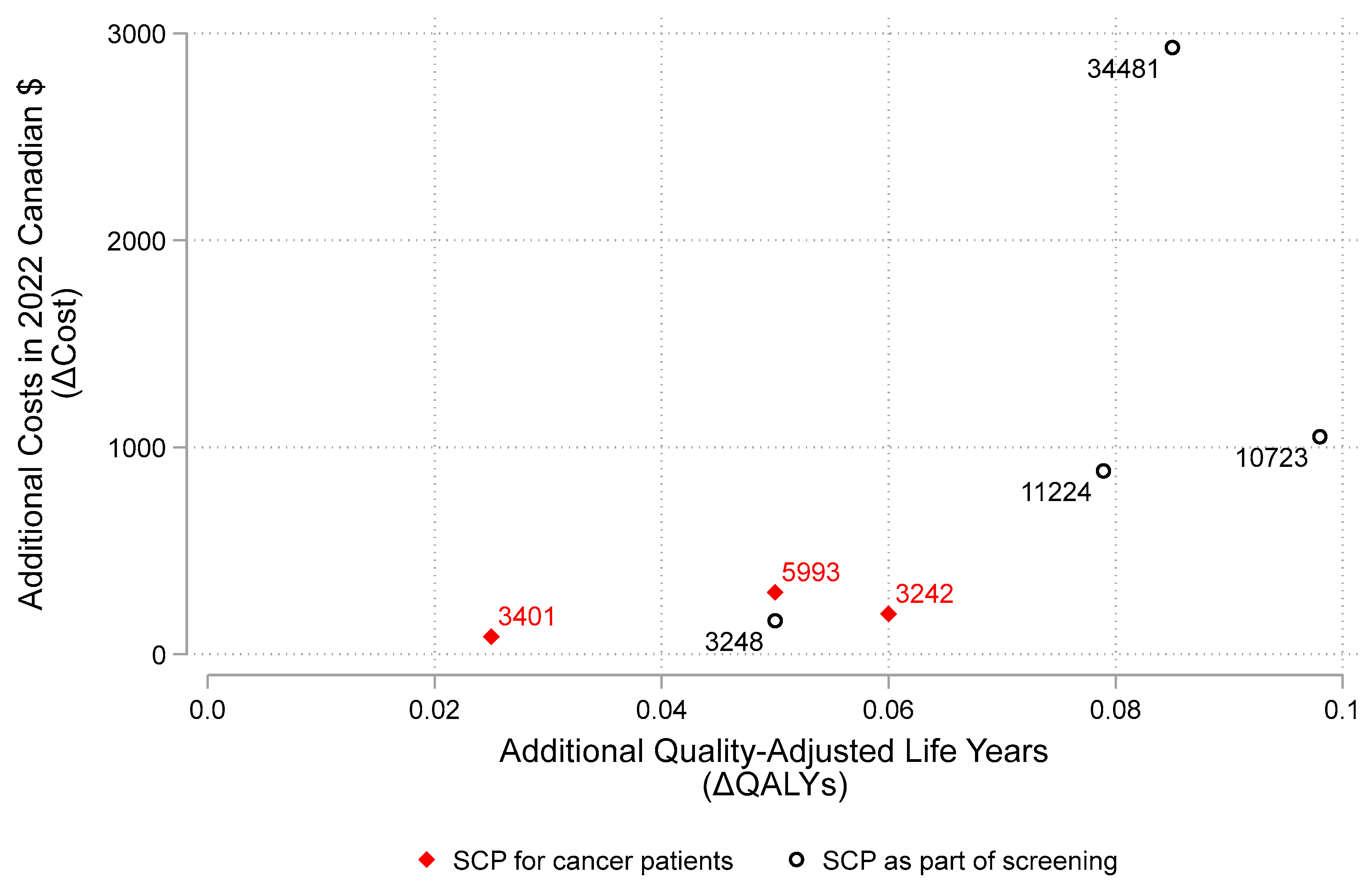
3.1. Results from the Cost-Effectiveness Model
3.2. Results from the Review of Scientific Literature
4. Discussion
4.1. Integrating Methods with Published Work: Effectiveness, Cost and Cost-Effectiveness
4.1.1. Effectiveness
4.1.2. Cost and Cost-Effectiveness
4.2. Other Issues
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Poirier, A.E.; Ruan, Y.; Grevers, X.; Walter, S.D.; Villeneuve, P.J.; Friedenreich, C.M.; Brenner, D.R.; ComPARe Study Team. Estimates of the current and future burden of cancer attributable to active and passive tobacco smoking in Canada. Prev. Med. 2019, 122, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Park, E.R.; Japuntich, S.J.; Rigotti, N.A.; Traeger, L.; He, Y.; Wallace, R.B.; Malin, J.L.; Zallen, J.P.; Keating, N.L. A snapshot of smokers after lung and colorectal cancer diagnosis. Cancer 2012, 118, 3153–3164. [Google Scholar] [CrossRef] [PubMed]
- Garces, Y.I.; Yang, P.; Parkinson, J.; Zhao, X.; Wampfler, J.A.; Ebbert, J.O.; Sloan, J.A. The relationship between cigarette smoking and quality of life after lung cancer diagnosis. Chest 2004, 126, 1733–1741. [Google Scholar] [CrossRef]
- Passarelli, M.N.; Newcomb, P.A.; Hampton, J.M.; Trentham-Dietz, A.; Titus, L.J.; Egan, K.M.; Baron, J.A.; Willett, W.C. Cigarette smoking before and after breast cancer diagnosis: Mortality from breast cancer and smoking-related diseases. J. Clin. Oncol. 2016, 34, 1315. [Google Scholar] [CrossRef]
- Nia, P.S.; Weyler, J.; Colpaert, C.; Vermeulen, P.; Van Marck, E.; Van Schil, P. Prognostic value of smoking status in operated non-small cell lung cancer. Lung Cancer 2005, 47, 351–359. [Google Scholar]
- Tseng, T.S.; Lin, H.Y.; Moody-Thomas, S.; Martin, M.; Chen, T. Who tended to continue smoking after cancer diagnosis: The national health and nutrition examination survey 1999–2008. BMC Public Health 2012, 12, 784. [Google Scholar] [CrossRef] [PubMed]
- Evans, W.K.; Warren, G.W.; Dresler, C. Ignoring the Obvious: Smoking Cessation Improves Survival. J. Thorac. Oncol. 2022, 17, 596–598. [Google Scholar] [CrossRef]
- Warren, G.W.; Silverman, C.; Halligan, M. Challenges and Adaptations for Providing Smoking Cessation for Patients with Cancer across Canada during the COVID-19 Pandemic. Curr. Oncol. 2022, 29, 2263–2271. [Google Scholar] [CrossRef]
- Warren, G.W.; Alberg, A.J.; Cummings, K.M.; Dresler, C. Smoking Cessation after a Cancer Diagnosis Is Associated with Improved Survival. J. Thorac. Oncol. 2020, 15, 705–708. [Google Scholar] [CrossRef]
- Warren, G.W. Mitigating the Adverse Health Effects and Costs Associated with Smoking after a Cancer Diagnosis. Transl. Lung Cancer Res. 2019, 8, S59–S66. [Google Scholar] [CrossRef]
- Warren, G.W.; Cartmell, K.B.; Garrett-Mayer, E.; Salloum, R.G.; Cummings, K.M. Attributable Failure of First-Line Cancer Treatment and Incremental Costs Associated with Smoking by Patients with Cancer. JAMA Netw. Open 2019, 2, e191703. [Google Scholar] [CrossRef]
- Iragorri, N.; Essue, B.; Timmings, C.; Keen, D.; Bryant, H.; Warren, G.W. The Cost of Failed First-Line Cancer Treatment Related to Continued Smoking in Canada. Curr. Oncol. 2020, 27, 307–312. [Google Scholar] [CrossRef]
- The Commonwealth Fund. State and Federal Efforts to Enhance Access to Basic Health Care. Available online: https://www.commonwealthfund.org/publications/newsletter-article/state-and-federal-efforts-enhance-access-basic-health-care (accessed on 29 June 2022).
- Levy, D.E.; Regan, S.; Perez, G.K.; Muzikansky, A.; Friedman, E.R.; Rabin, J.; Rigotti, N.A.; Ostroff, J.S.; Park, E.R. Cost-effectiveness of Implementing Smoking Cessation Interventions for Patients with Cancer. JAMA Netw. Open. 2022, 5, e2216362. [Google Scholar] [CrossRef]
- Cadham, C.J.; Cao, P.; Jayasekera, J.; Taylor, K.L.; Levy, D.T.; Jeon, J.; Elkin, E.B.; Foley, K.L.; Joseph, A.; Kong, C.Y.; et al. Cost-effectiveness of smoking cessation interventions in the lung cancer screening setting: A simulation study. JNCI J. Natl. Cancer Inst. 2021, 113, 1065–1073. [Google Scholar] [CrossRef]
- Cantor, S.B.; Deshmukh, A.A.; Luca, N.S.; Nogueras-González, G.M.; Rajan, T.; Prokhorov, A.V. Cost-effectiveness analysis of smoking-cessation counseling training for physicians and pharmacists. Addict. Behav. 2015, 45, 79–86. [Google Scholar] [CrossRef]
- Djalalov, S.; Masucci, L.; Isaranuwatchai, W.; Evans, W.; Peter, A.; Truscott, R.; Cameron, E.; Mittmann, N.; Rabeneck, L.; Chan, K.; et al. Economic evaluation of smoking cessation in Ontario’s regional cancer programs. Cancer Med. 2018, 7, 4672–4765. [Google Scholar] [CrossRef]
- Evans, W.K.; Truscott, R.; Cameron, E.; Rana, S.; Isaranuwatchai, W.; Haque, M.; Rabeneck, L. Implementing smoking cessation within cancer treatment centres and potential economic impacts. Transl. Lung Cancer Res. 2019, 8 (Suppl. 1), S11. [Google Scholar] [CrossRef]
- Evans, W.K.; Gauvreau, C.L.; Flanagan, W.M.; Memon, S.; Yong, J.H.; Goffin, J.R.; Fitzgerald, N.R.; Wolfson, M.; Miller, A.B. Clinical impact and cost-effectiveness of integrating smoking cessation into lung cancer screening: A microsimulation model. Can. Med. Assoc. Open Access J. 2020, 8, E585–E592. [Google Scholar] [CrossRef]
- Goffin, J.R.; Flanagan, W.M.; Miller, A.B.; Fitzgerald, N.R.; Memon, S.; Wolfson, M.C.; Evans, W.K. Biennial lung cancer screening in Canada with smoking cessation—Outcomes and cost-effectiveness. Lung Cancer 2016, 101, 98–103. [Google Scholar] [CrossRef]
- Slatore, C.G.; Au, D.H.; Hollingworth, W. Cost-effectiveness of a smoking cessation program implemented at the time of surgery for lung cancer. J. Thorac. Oncol. 2009, 4, 499–504. [Google Scholar] [CrossRef]
- Villanti, A.C.; Jiang, Y.; Abrams, D.B.; Pyenson, B.S. A cost-utility analysis of lung cancer screening and the additional benefits of incorporating smoking cessation interventions. PLoS ONE 2013, 8, e71379. [Google Scholar] [CrossRef]
- Laupacis, A.; Sackett, D.L.; Roberts, R.S. An assessment of clinically useful measures of the consequences of treatment. N. Engl. J. Med. 1988, 318, 1728–1733. [Google Scholar] [CrossRef]
- Vancak, V.; Goldberg, Y.; Levine, S.Z. Guidelines to understand and compute the number needed to treat. Evid. Based Ment. Health 2021, 24, 131–136. [Google Scholar] [CrossRef]
- Park, E.R.; Japuntich, S.; Temel, J.; Lanuti, M.; Pandiscio, J.; Hilgenberg, J.; Davies, D.; Dresler, C.; Rigotti, N.A. A smoking cessation intervention for thoracic surgery and oncology clinics: A pilot trial. J. Thorac. Oncol. 2011, 6, 1059–1065. [Google Scholar] [CrossRef]
- Health Consequences of Smoking—50 Years of Progress: A Report of the Surgeon General. 2014. Available online: http://www.surgeongeneral.gov/library/reports/50-yearsof-progress/ (accessed on 29 June 2022).
- Haslam, A.; Lythgoe, M.P.; Greenstreet Akman, E.; Prasad, V. Characteristics of Cost-effectiveness Studies for Oncology Drugs Approved in the United States From 2015-2020. JAMA Netw Open. 2021, 4, e2135123. [Google Scholar] [CrossRef]
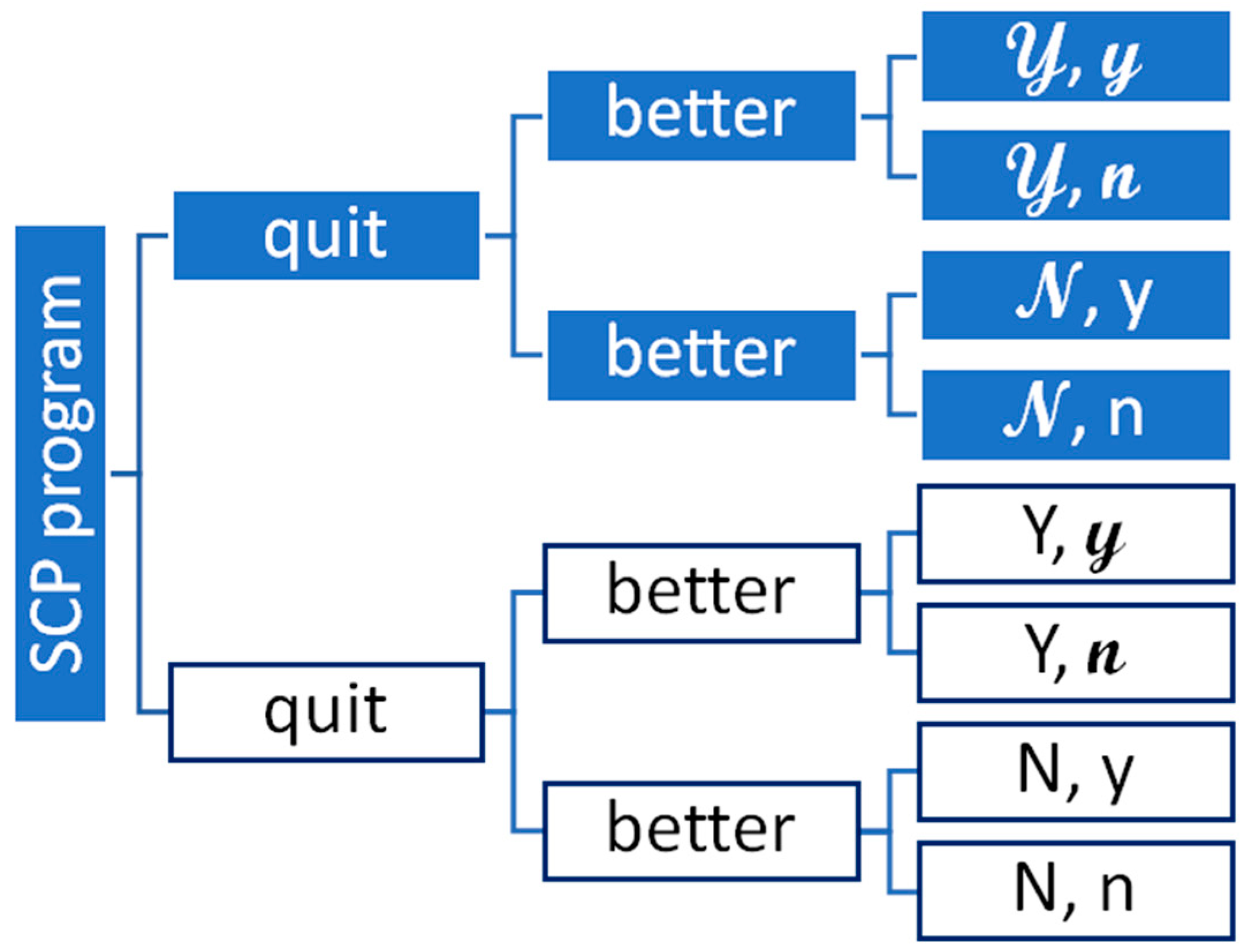


| Probability of | ||
|---|---|---|
| Option | Quitting | Not Quitting |
| SCP Program | 𝓝 | |
| No SCP program | Y | N |
| After | Doing Better | Not Doing Better |
| Quitting | 𝔂 | 𝓃 |
| Not quitting | y | n |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hoch, J.S.; Barr, H.K.; Guggenbickler, A.M.; Dewa, C.S. Lessons from Cost-Effectiveness Analysis of Smoking Cessation Programs for Cancer Patients. Curr. Oncol. 2022, 29, 6982-6991. https://doi.org/10.3390/curroncol29100549
Hoch JS, Barr HK, Guggenbickler AM, Dewa CS. Lessons from Cost-Effectiveness Analysis of Smoking Cessation Programs for Cancer Patients. Current Oncology. 2022; 29(10):6982-6991. https://doi.org/10.3390/curroncol29100549
Chicago/Turabian StyleHoch, Jeffrey S., Heather K. Barr, Andrea M. Guggenbickler, and Carolyn S. Dewa. 2022. "Lessons from Cost-Effectiveness Analysis of Smoking Cessation Programs for Cancer Patients" Current Oncology 29, no. 10: 6982-6991. https://doi.org/10.3390/curroncol29100549
APA StyleHoch, J. S., Barr, H. K., Guggenbickler, A. M., & Dewa, C. S. (2022). Lessons from Cost-Effectiveness Analysis of Smoking Cessation Programs for Cancer Patients. Current Oncology, 29(10), 6982-6991. https://doi.org/10.3390/curroncol29100549





