Real-World Treatment Patterns and Clinical Outcomes in Canadian Patients with AML Unfit for First-Line Intensive Chemotherapy
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Study Population
3.2. Treatment Patterns
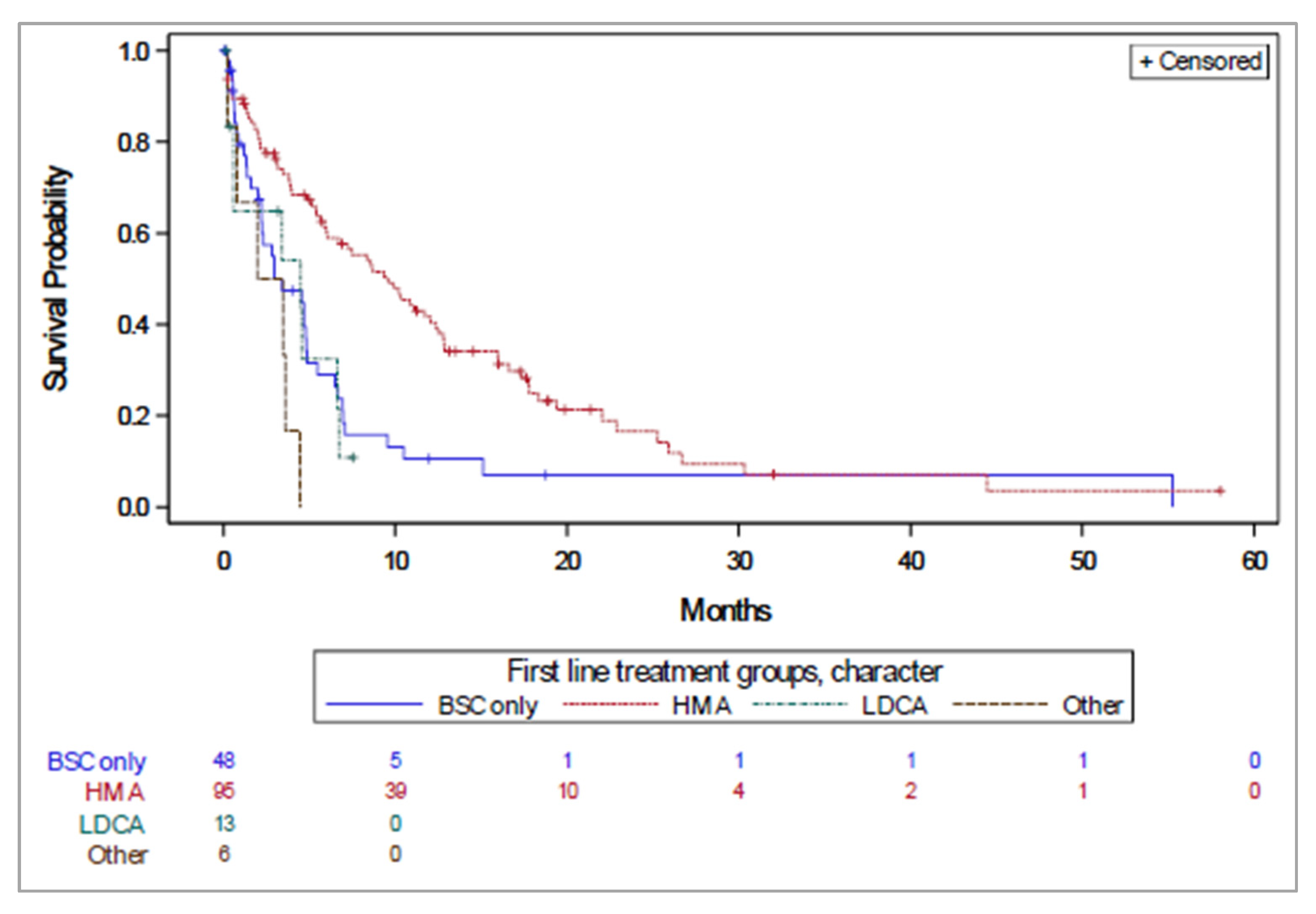
3.3. Overall Survival
3.4. Treatment Response
3.5. Healthcare Resource Utilization
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Döhner, H.; Weisdorf, D.J.; Bloomfield, C.D. Acute Myeloid Leukemia. N. Engl. J. Med. 2015, 373, 1136–1152. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute. Cancer Stat Facts: Leukemia—Acute Myeloid Leukemia (AML). Available online: https://seer.cancer.gov/statfacts/html/amyl.html (accessed on 4 April 2022).
- Thein, M.S.; Ershler, W.B.; Jemal, A.; Yates, J.W.; Baer, M.R. Outcome of older patients with acute myeloid leukemia: An analysis of SEER data over 3 decades. Cancer 2013, 119, 2720–2727. [Google Scholar] [CrossRef] [PubMed]
- Visser, O.; Trama, A.; Maynadie, M.; Stiller, C.; Marcos-Gragera, R.; De Angelis, R.; Mallone, S.; Tereanu, C.; Allemani, C.; Ricardi, U.; et al. Incidence, survival and prevalence of myeloid malignancies in Europe. Eur. J. Cancer 2012, 48, 3257–3266. [Google Scholar] [CrossRef]
- Sasaki, K.; Ravandi, F.; Kadia, T.M.; Dinardo, C.D.; Short, N.J.; Borthakur, G.; Jabbour, E.; Kantarjian, H.M. De novo acute myeloid leukemia: A population-based study of outcome in the United States based on the Surveillance, Epidemiology, and End Results (SEER) database, 1980 to 2017. Cancer 2021, 127, 2049–2061. [Google Scholar] [CrossRef] [PubMed]
- Dohner, H.; Estey, E.; Grimwade, D.; Amadori, S.; Appelbaum, F.R.; Buchner, T.; Dombret, H.; Ebert, B.L.; Fenaux, P.; Larson, R.A.; et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood 2017, 129, 424–447. [Google Scholar] [CrossRef] [PubMed]
- Heuser, M.; Ofran, Y.; Boissel, N.; Brunet Mauri, S.; Craddock, C.; Janssen, J.; Wierzbowska, A.; Buske, C. Acute myeloid leukaemia in adult patients: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2020, 31, 697–712. [Google Scholar] [CrossRef] [PubMed]
- Auletta, J.J.; Kou, J.; Chen, M.; Shaw, B.E. Current Use and Outcome of Hematopoietic Stem Cell Transplantation. Available online: https://www.cibmtr.org/ReferenceCenter/SlidesReports/SummarySlides/pages/index.aspx (accessed on 4 April 2022).
- Juliusson, G.; Antunovic, P.; Derolf, Å.; Lehmann, S.; Möllgård, L.; Stockelberg, D.; Tidefelt, U.; Wahlin, A.; Höglund, M. Age and acute myeloid leukemia: Real world data on decision to treat and outcomes from the Swedish Acute Leukemia Registry. Blood 2009, 113, 4179–4187. [Google Scholar] [CrossRef] [PubMed]
- Appelbaum, F.R.; Gundacker, H.; Head, D.R.; Slovak, M.L.; Willman, C.L.; Godwin, J.E.; Anderson, J.E.; Petersdorf, S.H. Age and acute myeloid leukemia. Blood 2006, 107, 3481–3485. [Google Scholar] [CrossRef] [PubMed]
- Büchner, T.; Berdel, W.E.; Haferlach, C.; Haferlach, T.; Schnittger, S.; Müller-Tidow, C.; Braess, J.; Spiekermann, K.; Kienast, J.; Staib, P.; et al. Age-related risk profile and chemotherapy dose response in acute myeloid leukemia: A study by the German Acute Myeloid Leukemia Cooperative Group. J. Clin. Oncol. 2009, 27, 61–69. [Google Scholar] [CrossRef] [PubMed]
- NCCN Guidelines for Acute Myeloid Leukemia V1. 2018. Available online: https://www.nccn.org/guidelines/guidelines-process/transparency-process-and-recommendations/GetFileFromFileManager?fileManagerId=3537 (accessed on 4 April 2022).
- Health Canada. VENCLEXTA® (Venetoclax Tablets) Product Monograph. Available online: https://pdf.hres.ca/dpd_pm/00059710.PDF (accessed on 21 January 2022).
- FDA Grants Venetoclax Breakthrough Therapy Designation for Geriatric Patients with Acute Myeloid Leukemia. The ASCO Post 2017. Available online: https://ascopost.com/News/57879 (accessed on 4 April 2022).
- European Medicines Agency. Venclyxto Summary of Product Characteristics. Available online: https://www.ema.europa.eu/en/documents/product-information/venclyxto-epar-product-information_en.pdf (accessed on 11 March 2022).
- Administration, F.D. DAURISMO (Glasdegib) Prescribing Information. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/210656s000lbl.pdf (accessed on 4 April 2022).
- Health Canada. DAURISMO (Glasdegib): Product Monograph. Available online: https://pdf.hres.ca/dpd_pm/00064470.PDF (accessed on 4 April 2022).
- European Medicines Agency. Daurismo (Glasdegib) EPAR. Available online: https://www.ema.europa.eu/en/documents/overview/daurismo-epar-medicine-overview_en.pdf (accessed on 4 April 2022).
- Miyamoto, T.; Sanford, D.; Tomuleasa, C.; Hsiao, H.-H.; Olivera, L.J.E.; Enjeti, A.K.; Gimenez Conca, A.; Castillo, T.B.D.; Girshova, L.; Martelli, M.P.; et al. Real-world treatment patterns and clinical outcomes in patients with AML unfit for first-line intensive chemotherapy. Leuk. Lymphoma 2022, 63, 928–938. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Sanford, D.; Tomuleasa, C.; Hsiao, H.H.; Olivera, L.J.E.; Enjeti, A.K.; Conca, A.G.; Del Castillo, T.B.; Girshova, L.; Martelli, M.P.; et al. Healthcare resource utilization trends in patients with acute myeloid leukemia ineligible for intensive chemotherapy receiving first-line systemic treatment or best supportive care: A multicenter international study. Eur. J. Haematol. 2022, 109, 58–68. [Google Scholar] [CrossRef] [PubMed]
- DiNardo, C.D.; Pratz, K.W.; Letai, A.; Jonas, B.A.; Wei, A.H.; Thirman, M.; Arellano, M.; Frattini, M.G.; Kantarjian, H.; Popovic, R.; et al. Safety and preliminary efficacy of venetoclax with decitabine or azacitidine in elderly patients with previously untreated acute myeloid leukaemia: A non-randomised, open-label, phase 1b study. Lancet Oncol. 2018, 19, 216–228. [Google Scholar] [CrossRef]
- Acute Myelogenous Leukemia Statistics. Available online: https://cancer.ca/en/cancer-information/cancer-types/acute-myeloid-leukemia-aml (accessed on 20 June 2022).
- NCCN. Clinical Practice Guidelines in Oncology: Acute Myeloid Leukemia Version 2. Available online: https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1411 (accessed on 8 August 2022).
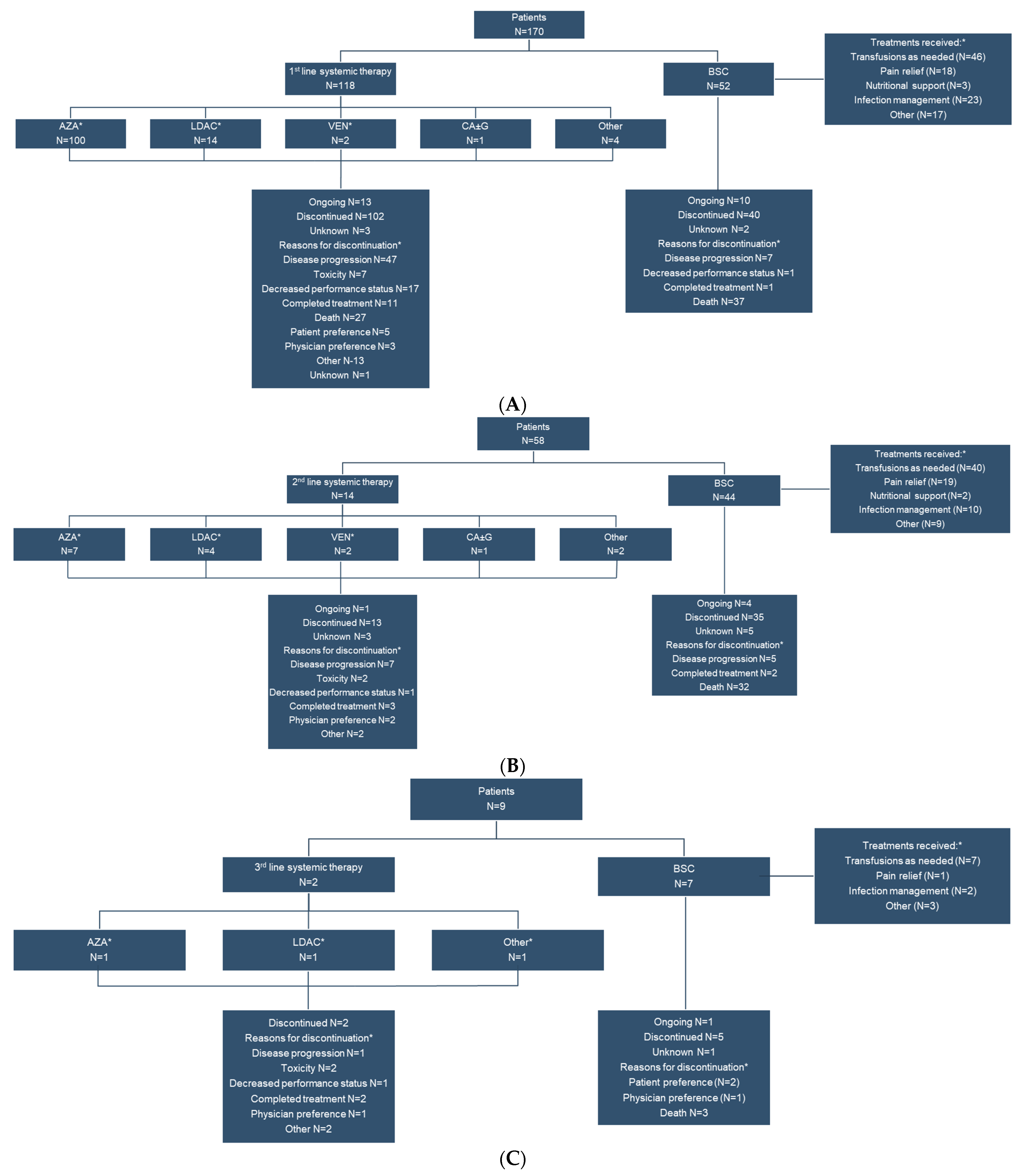
| Overall (n = 170) | 1st Line Systemic Therapy (n = 118) | BSC (n = 52) | |
|---|---|---|---|
| Female gender (n [%]) * | 65 (38.2) | 41 (34.7) | 24 (46.2) |
| Mean (SD) age at diagnosis (years) | 74.3 (7.01) | 74.3 (6.90) | 74.3 (7.37) |
| ≤75 years (n [%]) | 99 (58.2) | 71 (60.2) | 28 (53.8) |
| Secondary AML | |||
| Yes | 73 (42.9) | 44 (37.3) | 29 (55.8) |
| No | 79 (46.5) | 59 (50.0) | 20 (38.5) |
| Unknown | 18 (10.6) | 15 (12.7) | 3 (5.8) |
| ECOG performance status | |||
| 0–1 | 41 (24.1) | 27 (22.9) | 14 (26.9) |
| ≥2 | 43 (23.5) | 20 (17.0) | 23 (44.2) |
| Unknown | 86 (50.6) | 71 (60.2) | 15 (28.8) |
| AML classification—WHO (n [%]) | |||
| AML with recurrent abnormalities | 13 (7.6) | 11 (9.3) | 2 (3.8) |
| AML with MDS-related changes | 76 (44.7) | 53 (44.9) | 23 (44.2) |
| AML not otherwise specified | 41 (24.1) | 28 (23.7) | 13 (25.0) |
| Myeloid sarcoma | 1 (1.2) | 0 | 2 (3.8) |
| Unknown | 33 (19.4) | 22 (18.6) | 11 (21.2) |
| Cytogenetic risk (n [%]) | |||
| Favourable | 26 (15.3) | 19 (16.1) | 7 (13.5) |
| Intermediate | 47 (27.6) | 38 (32.2) | 9 (17.3) |
| Poor | 61 (35.9) | 46 (39.0) | 15 (28.8) |
| Unknown | 36 (21.2) | 15 (12.7) | 21 (40.4) |
| Molecular features identified (n [%]) | |||
| Any | 40 (23.5) | 30 (25.4) | 10 (19.2) |
| IDH2 | 1 (2.5) | 1 (3.3) | 0 |
| TP53 | 2 (5.0) | 2 (6.7) | 0 |
| TET2 | 1 (2.5) | 1 (3.3) | 0 |
| RUNX1 | 5 (12.5) | 3 (10.0) | 2 (20.0) |
| DNMT3A | 1 (2.5) | 1 (3.3) | 0 |
| ASXL 1 | 3 (7.5) | 3 (10.0) | 0 |
| FLT3TKD | 2 (5.0) | 2 (6.7) | 0 |
| JAK2 | 7 (17.5) | 3 (10.0) | 4 (40.0) |
| NPM1 | 13 (32.5) | 10 (33.3) | 3 (30.0) |
| SRSF2 | 2 (5.0) | 2 (6.7) | 0 |
| MLLPTD | 2 (5.0) | 1 (3.3) | 1 (10.0) |
| Other | 11 (27.5) | 9 (30.0) | 2 (20.0) |
| None | 56 (32,9) | 42 (35.6) | 14 (26.9) |
| Unknown | 74 (43.5) | 46 (39.0) | 28 (53.8) |
| Co-morbidities (n [%]) | |||
| Myocardial infarction | 2 (1.2) | 1 (0.8) | 1 (1.9) |
| Angina/coronary artery disease | 22 (12.9) | 12 (10.2) | 10 (19.2) |
| Congestive heart failure | 11 (6.5) | 8 (6.8) | 3 (5.8) |
| Arrhythmias | 14 (8.2) | 9 (7.6) | 5 (9.6) |
| Restrictive lung disease or COPD | 8 (4.7) | 8 (6.8) | 0 |
| Liver cirrhosis (Child Pugh A, B or C) | 1 (0.6) | 1 (0.8) | 0 |
| Elevated transaminases unrelated to cirrhosis | 1 (0.6) | 1 (0.8) | 0 |
| CKD stage 3, 4 or 5 | 2 (1.2) | 0 | 2 (3.8) |
| Other | 99 (58.2) | 69 (58.5) | 30 (57.7) |
| Unknown | 15 (8.8) | 9 (7.6) | 6 (11.5) |
| None | 39 (22.9) | 27 (22.9) | 12 (23.1) |
| Overall (n = 170) | LDAC (n = 14) | HMA (n = 97) | Other (n = 7) | BSC Only (n = 52) | |
|---|---|---|---|---|---|
| Median (95% CI) OS (months) | 8.6 (6.2–11.1) | 6.4 (5.0–14.2) | 13.1 (10.0–15.2) | NE | 3.0 (2.2–4.9) |
| Median (95% CI) PFS (months) | 5.8 (4.4–7.2) | 5.5 (1.4–12.9) | 9.7 (7.2–11.4) | 3.6 (1.5–NE) | 2.4 (1.2–3.2) |
| 2-year (95% CI) OS (%) | 20.5 (13.8–28.2) | 11.7 (0.7–39.4) | 26.9 (17.2–37.4) | 62.5 (14.2–89.3) | 6.7 (1.4–17.9) |
| 5-year (95% CI) OS (%) * | 3.2 (0.3–12.7) | 0 | 4.7 (0.5–17.4) | 0 | 0 |
| Overall (n = 170) | 1st Line Systemic Therapy (n = 118) | BSC Only (n = 52) | |
|---|---|---|---|
| Alive at end of study | 48 (28.2) | 37 (31.4) | 11 (21.2) |
| Cause of death (n [%]): | |||
| AML progression | 84 (68.9) | 56 (69.1) | 28 (68.3) |
| Infection | 21 (17.2) | 16 (19.8) | 5 (12.2) |
| Multi-organ failure | 1 (0.8) | 16 (19.8) | 1 (2.4) |
| Other comorbid conditions | 5 (0.1) | 3 (3.7) | 2 (4.9) |
| Unrelated to a disease | 1 (0.8) | 3 (3.7) | 1 (2.4) |
| Unknown | 10 (8.2) | 6 (7.4) | 4 (9.8) |
| Best Overall Response (n, %) | First Line Therapy (n = 118) | Second Line Therapy (n = 14) |
|---|---|---|
| CR | 10 (8.5) | 1 (7.1) |
| Cri | 8 (6.8) | 0 |
| PR | 18 (15.3) | 4 (28.6) |
| SD | 21 (17.8) | 0 |
| PD | 12 (10.2) | 2 (14.3) |
| Unknown | 49 (41.5) | 7 (50.0) |
| First Line Systemic Therapy (n = 118) | BSC (n = 52) | |
|---|---|---|
| Outpatient consultation (N [%]): | ||
| Yes * | 96 (81.4) | 43 (82.7) |
| No | 18 (15.3) | 5 (9.6) |
| Unknown | 4 (3.4) | 4 (7.7) |
| Number of visits (median [range]) | 10 (1–105) | 6 (1–80) |
| Hospitalization (N [%]): | ||
| Yes * | 79 (66.9) | 37 (71.2) |
| No | 36 (30.5) | 8 (15.4) |
| Unknown | 3 (2.5) | 7 (13.5) |
| Number of hospitalizations (N [%]): | ||
| 1 | 50 (63.3) | 27 (73.0) |
| 2 | 19 (24.1) | 7 (18.9) |
| ≥3 | 10 (12.7) | 3 (8.1) |
| Duration of stay (days, median [range]) | ||
| Overall | 7 (1–100) | 9 (1–92) |
| In ICU | 0 (0–31) | 2 (0–38) |
| Reason for hospitalization: † | ||
| Progression/relapse-related | 22 (16.4) | 19 (34.5) |
| Infection-related | 70 (52.2) | 32 (58.2) |
| Transfusion-related | 9 (6.7) | 4 (7.3) |
| Treatment administration-related | 14 (10.4) | 3 (5.5) |
| Other AML-related | 25 (18.7) | 10 (18.2) |
| Other | 38 (28.4) | 10 (18.2) |
| RBC/PLT transfusion (N [%]): | ||
| Yes | 96 (81.4) | 39 (75.0) |
| No | 16 (13.6) | 7 (13.5) |
| Unknown | 6 (5.1) | 6 (11.5) |
| If yes, number of RBC transfusions (median [range]) | 10 (2–180) | 6 (1–100) |
| If yes, number of PLT transfusions (median [range]) | 1.5 (0–50) | 1.5 (0–200) |
| Systemic Therapy | BSC | |
|---|---|---|
| First Line Therapy | n = 118 | n = 52 |
| Antibiotic or antiviral use (N [%]): | ||
| Yes * | 63 (53.4) | 29 (55.8) |
| No | 54 (45.8) | 16 (30.8) |
| Unknown | 1 (0.8) | 7 (13.5) |
| Reason for use:† | ||
| Prophylaxis | 16 (25.4) | 3 (10.3) |
| Curative | 51 (81.0) | 27 (93.1) |
| Unknown | 3 (4.8) | 0 |
| Antifungal use (N [%]): | ||
| Yes* | 21 (17.8) | 3 (5.8) |
| No | 96 (81.4) | 44 (84.6) |
| Unknown | 1 (0.8) | 5 (9.6) |
| Reason for use:† | ||
| Prophylaxis | 11 (52.5) | 1 (33.3) |
| Curative | 8 (38.1) | 2 (66.7) |
| Unknown | 2 (9.5) | 0 |
| Second Line Therapy | n = 14 | n = 44 |
| Antibiotic or antiviral use (N [%]): | ||
| Yes* | 6 (42.9) | 17 (38.6) |
| No | 7 (50.0) | 21 (47.7) |
| Unknown | 1 (7.1) | 6 (13.6) |
| Reason for use:† | ||
| Prophylaxis | 1 (16.7) | 6 (35.3) |
| Curative | 6 (100.0) | 10 (58.8) |
| Unknown | 0 | 2 (11.8) |
| Antifungal use (N [%]): | ||
| Yes * | 2 (14.3) | 8 (18.2) |
| No | 12 (85.7) | 30 (68.2) |
| Unknown | 0 | 6 (13.6) |
| Reason for use:† | ||
| Prophylaxis | 1 (50.0) | 4 (40.0) |
| Curative | 1 (50.0) | 3 (37.5) |
| Unknown | 0 | 1 (12.5) |
| Third Line Therapy | n = 2 | n = 7 |
| Antibiotic or antiviral use (N [%]): | ||
| Yes * | 2 (100.0) | 3 (42.9) |
| No | 0 | 4 (57.1) |
| Reason for use: † | ||
| Prophylaxis | 1 (50.0) | 0 |
| Curative | 1 (50.0) | 3 (100.0) |
| Unknown | 1 (50.0) | 0 |
| Antifungal use (N [%]): | ||
| Yes * | 2 (100.0) | 2 (28.6) |
| No | 5 (71.4) | |
| Reason for use: † | ||
| Prophylaxis | 1 (50.0) | 0 |
| Curative | 1 (50.0) | 2 (100.0) |
| Unknown | 1 (50.0) | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanford, D.; Desjardins, P.; Leber, B.; Paulson, K.; Assouline, S.; Lembo, P.M.C.; Fournier, P.-A.; Leitch, H.A. Real-World Treatment Patterns and Clinical Outcomes in Canadian Patients with AML Unfit for First-Line Intensive Chemotherapy. Curr. Oncol. 2022, 29, 6794-6806. https://doi.org/10.3390/curroncol29100535
Sanford D, Desjardins P, Leber B, Paulson K, Assouline S, Lembo PMC, Fournier P-A, Leitch HA. Real-World Treatment Patterns and Clinical Outcomes in Canadian Patients with AML Unfit for First-Line Intensive Chemotherapy. Current Oncology. 2022; 29(10):6794-6806. https://doi.org/10.3390/curroncol29100535
Chicago/Turabian StyleSanford, David, Pierre Desjardins, Brian Leber, Kristjan Paulson, Sarit Assouline, Paola M. C. Lembo, Pierre-André Fournier, and Heather A. Leitch. 2022. "Real-World Treatment Patterns and Clinical Outcomes in Canadian Patients with AML Unfit for First-Line Intensive Chemotherapy" Current Oncology 29, no. 10: 6794-6806. https://doi.org/10.3390/curroncol29100535
APA StyleSanford, D., Desjardins, P., Leber, B., Paulson, K., Assouline, S., Lembo, P. M. C., Fournier, P.-A., & Leitch, H. A. (2022). Real-World Treatment Patterns and Clinical Outcomes in Canadian Patients with AML Unfit for First-Line Intensive Chemotherapy. Current Oncology, 29(10), 6794-6806. https://doi.org/10.3390/curroncol29100535





