Long-Term Results of Postoperative Hypofractionated Accelerated Breast and Lymph Node Radiotherapy (HypoAR) with Hypofractionated Boost
Abstract
:1. Introduction
2. Materials and Methods
2.1. Radiotherapy Schemes
2.2. Toxicity Evaluation
2.3. Statistical Analysis
3. Results
3.1. Radiation Toxicity
3.2. Second Carcinomas
3.3. Control of In Situ Carcinoma
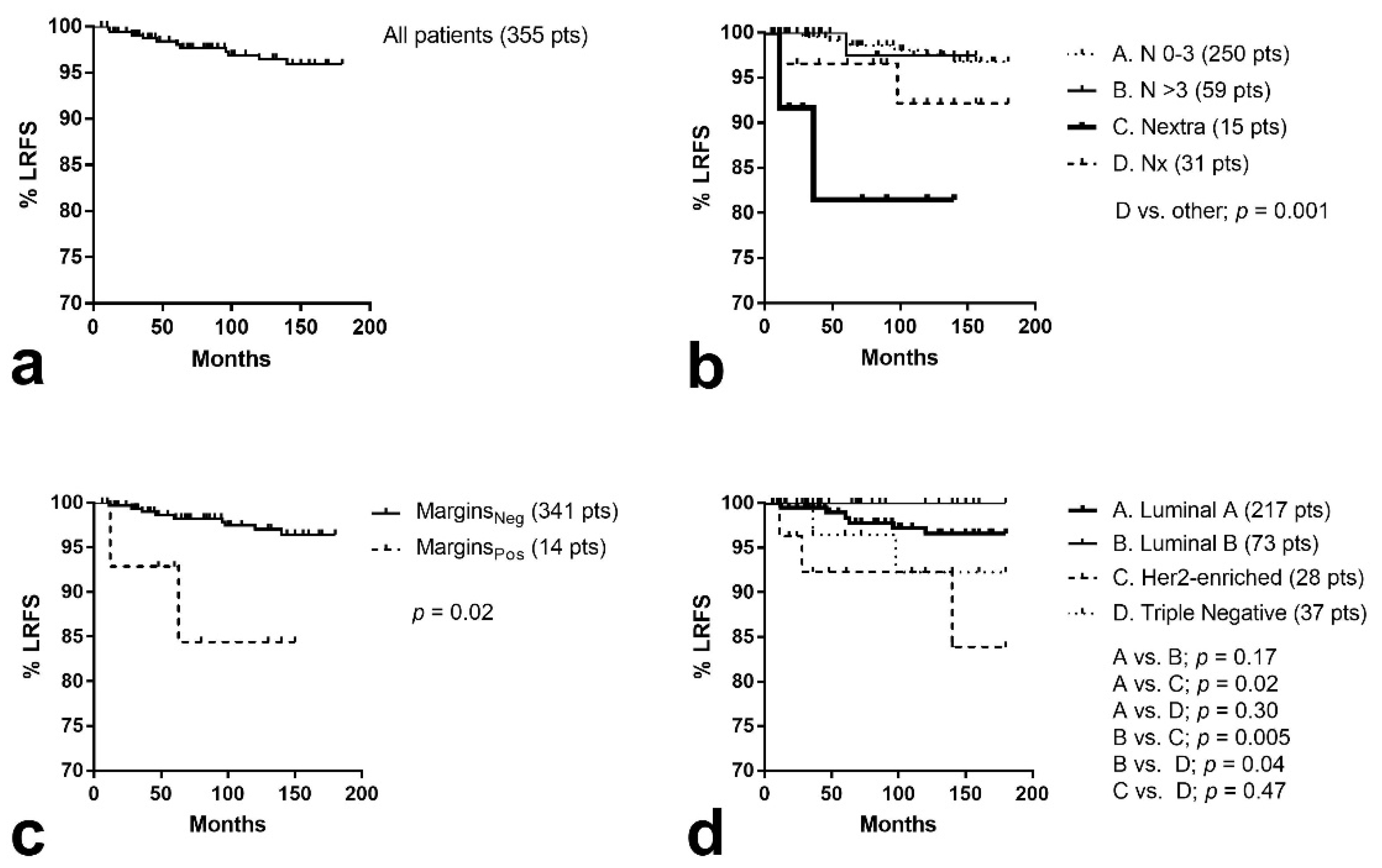
3.4. Locoregional Control
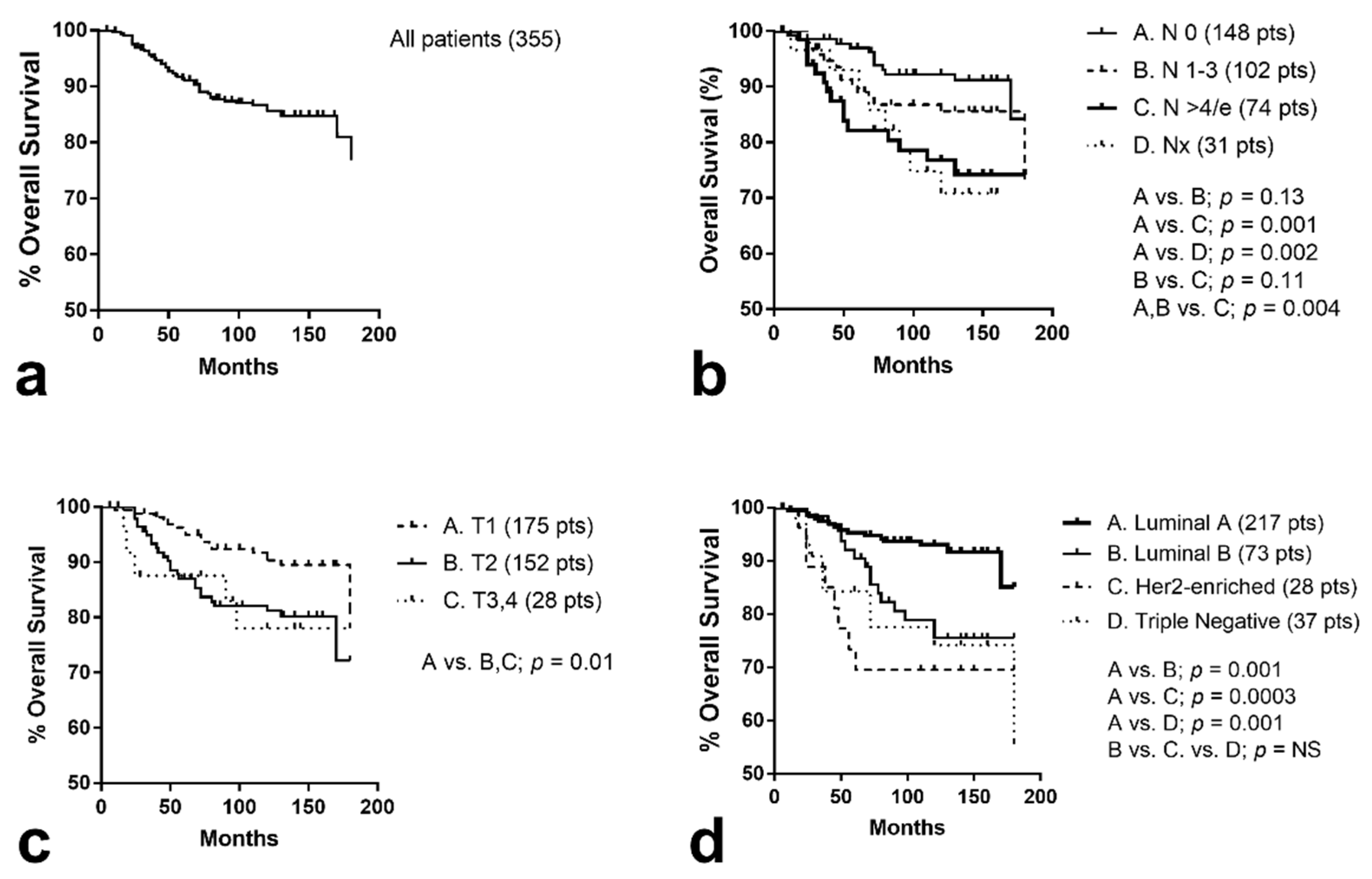
3.5. Distant Metastasis and Overall Survival
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
- The Canadian trial [19] found equivalent breast toxicity between an hypoAR schedule of 42.5 Gy delivered in 16 fractions of 2.66 Gy within 22 days and a CRT scheme of 50 Gy delivering 25 fractions of 2 Gy within 33 days. Applying the time-corrected EQD2 formula for α/β = 4 Gy, the calculated λ-value is 0.26 Gy/day.
- The START-B trial [25] found less breast toxicity produced by an hypoAR schedule of 40 Gy delivered in 15 fractions within 19 days compared to the CRT scheme of 50 Gy delivering 25 fractions of 2 Gy within 33 days. This implies a λ-value of lower than 0.4 Gy.
References
- Litière, S.; Werutsky, G.; Fentiman, I.S.; Rutgers, E.; Christiaens, M.R.; Van Limbergen, E.; Baaijens, M.H.; Bogaerts, J.; Bartelink, H. Breast conserving therapy versus mastectomy for stage I-II breast cancer: 20 year follow-up of the EORTC 10801 phase 3 randomised trial. Lancet Oncol. 2012, 13, 412–419. [Google Scholar] [CrossRef]
- Early Breast Cancer Trialists’ Collaborative Group (EBCTCG); Darby, S.; McGale, P.; Correa, C.; Taylor, C.; Arriagada, R.; Clarke, M.; Cutter, D.; Davies, C.; Ewertz, M.; et al. Effect of radiotherapy after breast-conserving surgery on 10-year recurrence and 15-year breast cancer death: Meta-analysis of individual patient data for 10,801 women in 17 randomised trials. Lancet 2011, 378, 1707–1716. [Google Scholar] [CrossRef] [Green Version]
- Athas, W.F.; Adams-Cameron, M.; Hunt, W.C.; Amir-Fazli, A.; Key, C.R. Travel distance to radiation therapy and receipt of radiotherapy following breast-conserving surgery. J. Natl. Cancer Inst. 2000, 92, 269–271. [Google Scholar] [CrossRef] [Green Version]
- Fletcher, G.H. Hypofractionation: Lessons from complications. Radiother. Oncol. 1991, 20, 10–15. [Google Scholar] [CrossRef]
- Baillet, F.; Housset, M.; Maylin, C.; Boisserie, G.; Bettahar, R.; Delanian, S.; Habib, F. The use of a specific hypofractionated radiation therapy regimen versus classical fractionation in the treatment of breast cancer: A randomized study of 230 patients. Int. J. Radiat. Oncol. Biol. Phys. 1990, 19, 1131–1133. [Google Scholar] [CrossRef]
- Brenner, D.J.; Hall, E.J. Fractionation and protraction for radiotherapy of prostate carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 1999, 43, 1095–1101. [Google Scholar] [CrossRef]
- Yarnold, J.; Ashton, A.; Bliss, J.; Homewood, J.; Harper, C.; Hanson, J.; Haviland, J.; Bentzen, S.; Owen, R. Fractionation sensitivity and dose response of late adverse effects in the breast after radiotherapy for early breast cancer: Long-term results of a randomised trial. Radiother. Oncol. 2005, 75, 9–17. [Google Scholar] [CrossRef] [Green Version]
- START Trialists’ Group; Bentzen, S.M.; Agrawal, R.K.; Aird, E.G.; Barrett, J.M.; Barrett-Lee, P.J.; Bliss, J.M.; Brown, J.; Dewar, J.A.; Dobbs, H.J.; et al. The UK Standardisation of Breast Radiotherapy (START) Trial A of radiotherapy hypofractionation for treatment of early breast cancer: A randomised trial. Lancet Oncol. 2008, 9, 331–341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Whelan, T.J.; Pignol, J.P.; Levine, M.N.; Julian, J.A.; MacKenzie, R.; Parpia, S.; Shelley, W.; Grimard, L.; Bowen, J.; Lukka, H.; et al. Long-term results of hypofractionated radiation therapy for breast cancer. N. Engl. J. Med. 2010, 362, 513–520. [Google Scholar] [CrossRef] [Green Version]
- Koukourakis, M.I.; Giatromanolaki, A.; Kouroussis, C.; Kakolyris, S.; Sivridis, E.; Frangiadaki, C.; Retalis, G.; Georgoulias, V.; Tumor and Angiogenesis Research Group. Hypofractionated and accelerated radiotherapy with cytoprotection (HypoARC): A short, safe, and effective postoperative regimen for high-risk breast cancer patients. Int. J. Radiat. Oncol. Biol. Phys. 2002, 52, 144–155. [Google Scholar] [CrossRef]
- Koukourakis, M.I.; Tsoutsou, P.G.; Abatzoglou, I.M.; Sismanidou, K.; Giatromanolaki, A.; Sivridis, E. Hypofractionated and accelerated radiotherapy with subcutaneous amifostine cytoprotection as short adjuvant regimen after breast-conserving surgery: Interim report. Int. J. Radiat. Oncol. Biol. Phys. 2009, 74, 1173–1180. [Google Scholar] [CrossRef]
- Koukourakis, M.I.; Panteliadou, M.; Abatzoglou, I.M.; Sismanidou, K.; Sivridis, E.; Giatromanolaki, A. Postmastectomy hypofractionated and accelerated radiation therapy with (and without) subcutaneous amifostine cytoprotection. Int. J. Radiat. Oncol. Biol. Phys. 2013, 85, e7–e13. [Google Scholar] [CrossRef] [PubMed]
- Maciejewski, B.; Withers, H.R.; Taylor, J.M.; Hliniak, A. Dose fractionation and regeneration in radiotherapy for cancer of the oral cavity and oropharynx: Tumor dose-response and repopulation. Int. J. Radiat. Oncol. Biol. Phys. 1989, 16, 831–843. [Google Scholar] [CrossRef]
- Thames, H.D.; Bentzen, S.M.; Turesson, I.; Overgaard, M.; Van den Bogaert, W. Time-dose factors in radiotherapy: A review of the human data. Radiother. Oncol. 1990, 19, 219–235. [Google Scholar] [CrossRef]
- Common Terminology Criteria for Adverse Events (CTCAE), Version 5.0. Published 27 November 2017. Available online: https://www.eortc.be/services/doc/ctc/CTCAE_v5_Quick_Reference_5x7.pdf (accessed on 1 June 2021).
- LENT SOMA tables. Radiother. Oncol. 1995, 35, 17–60. [CrossRef]
- Woodward, S.G.; Varshney, K.; Anne, P.R.; George, B.J.; Willis, A.I. Trends in Use of Hypofractionated Whole Breast Radiation in Breast Cancer: An Analysis of the National Cancer Database. Int. J. Radiat. Oncol. Biol. Phys. 2021, 109, 449–457. [Google Scholar] [CrossRef]
- Brunt, A.M.; Haviland, J.S.; Sydenham, M.; Agrawal, R.K.; Algurafi, H.; Alhasso, A.; Barrett-Lee, P.; Bliss, P.; Bloomfield, D.; Bowen, J.; et al. Ten-Year Results of FAST: A Randomized Controlled Trial of 5-Fraction Whole-Breast Radiotherapy for Early Breast Cancer. J. Clin. Oncol. 2020, 38, 3261–3272. [Google Scholar] [CrossRef] [PubMed]
- Arsenault, J.; Parpia, S.; Goldberg, M.; Rakovitch, E.; Reiter, H.; Doherty, M.; Lukka, H.; Sussman, J.; Wright, J.; Julian, J.; et al. Acute Toxicity and Quality of Life of Hypofractionated Radiation Therapy for Breast Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2020, 107, 943–948. [Google Scholar] [CrossRef] [PubMed]
- Haviland, J.S.; Owen, J.R.; Dewar, J.A.; Agrawal, R.K.; Barrett, J.; Barrett-Lee, P.J.; Dobbs, H.J.; Hopwood, P.; Lawton, P.A.; Magee, B.J.; et al. START Trialists’ Group. The UK Standardisation of Breast Radiotherapy (START) trials of radiotherapy hypofractionation for treatment of early breast cancer: 10-year follow-up results of two randomised controlled trials. Lancet Oncol. 2013, 14, 1086–1094. [Google Scholar] [CrossRef]
- Graham, P.; Fourquet, A. Placing the boost in breast-conservation radiotherapy: A review of the role, indications and techniques for breast-boost radiotherapy. Clin. Oncol. (R. Coll. Radiol.) 2006, 18, 210–219. [Google Scholar] [CrossRef]
- Bartelink, H.; Maingon, P.; Poortmans, P.; Weltens, C.; Fourquet, A.; Jager, J.; Schinagl, D.; Oei, B.; Rodenhuis, C.; Horiot, J.C.; et al. Whole-breast irradiation with or without a boost for patients treated with breast-conserving surgery for early breast cancer: 20-year follow-up of a randomised phase 3 trial. Lancet Oncol. 2015, 16, 47–56. [Google Scholar] [CrossRef]
- Radiotherapy Dose Fractionation. 3rd ed. March 2019. Available online: https://www.rcr.ac.uk/system/files/publication/field_publication_files/brfo193_radiotherapy_dose_fractionation_third-edition.pdf (accessed on 1 June 2021).
- Sopik, V.; Nofech-Mozes, S.; Sun, P.; Narod, S.A. The relationship between local recurrence and death in early-stage breast cancer. Breast Cancer Res. Treat. 2016, 155, 175–185. [Google Scholar] [CrossRef] [PubMed]
- Haviland, J.S.; Mannino, M.; Griffin, C.; Porta, N.; Sydenham, M.; Bliss, J.M.; Yarnold, J.R.; START Trialists’ Group. Late normal tissue effects in the arm and shoulder following lymphatic radiotherapy: Results from the UK START (Standardisation of Breast Radiotherapy) trials. Radiother. Oncol. 2018, 126, 155–162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fragomeni, S.M.; Sciallis, A.; Jeruss, J.S. Molecular Subtypes and Local-Regional Control of Breast Cancer. Surg. Oncol. Clin. N. Am. 2018, 27, 95–120. [Google Scholar] [CrossRef] [PubMed]
- Kirova, Y.M.; Gambotti, L.; De Rycke, Y.; Vilcoq, J.R.; Asselain, B.; Fourquet, A. Risk of second malignancies after adjuvant radiotherapy for breast cancer: A large-scale, single-institution review. Int. J. Radiat. Oncol. Biol. Phys. 2007, 68, 359–363. [Google Scholar] [CrossRef]
- Schneider, U.; Besserer, J.; Mack, A. Hypofractionated radiotherapy has the potential for second cancer reduction. Theor. Biol. Med. Model. 2010, 7, 4. [Google Scholar] [CrossRef] [Green Version]
- Koukourakis, M.I. Radiation damage and radioprotectants: New concepts in the era of molecular medicine. Br. J. Radiol. 2012, 85, 313–330. [Google Scholar] [CrossRef] [Green Version]
- Murray Brunt, A.; Haviland, J.S.; Wheatley, D.A.; Sydenham, M.A.; Alhasso, A.; Bloomfield, D.J.; Chan, C.; Churn, M.; Cleator, S.; Coles, C.E.; et al. Hypofractionated breast radiotherapy for 1 week versus 3 weeks (FAST-Forward): 5-year efficacy and late normal tissue effects results from a multicentre, non-inferiority, randomised, phase 3 trial. Lancet 2020, 395, 1613–1626. [Google Scholar] [CrossRef]
| All Cases | Schedule A | Schedule B | p-Value | |
|---|---|---|---|---|
| 367 | 290 | 77 | ||
| Age | ||||
| Median | 56 | 56 | 60 | 0.74 |
| Range | 26–84 | 26–84 | 29–81 | |
| Performance status | ||||
| 0 | 367 | 290 | 77 | |
| Histology | ||||
| NOS (*) | 338 | 264 | 74 | 0.41 |
| Lobular | 23 | 21 | 2 | |
| Myeloid | 4 | 3 | 1 | |
| Mucinous | 2 | 2 | 0 | |
| T-stage | ||||
| Tis | 12 | 6 | 6 | 0.22 |
| T1 | 175 | 147 | 28 | 0.01 (#) |
| T2 | 153 | 115 | 38 | |
| T3 | 19 | 16 | 3 | |
| T4 | 8 | 6 | 2 | |
| Multifocality | ||||
| No | 342 | 272 | 70 | 0.37 |
| Yes | 25 | 18 | 7 | |
| Lymphovascular space invasion | ||||
| No | 337 | 266 | 71 | 0.89 |
| Yes | 30 | 24 | 6 | |
| Resection Margins | ||||
| Negative | 335 | 279 | 74 | 0.96 |
| Positive | 14 | 11 | 3 | |
| Node involvement | ||||
| 0 | 160 | 124 | 36 | 0.45 |
| 1–3 | 120 | 79 | 23 | |
| 4–10 | 43 | 32 | 11 | |
| >10 | 16 | 14 | 2 | |
| extracapsular invasion | 15 | 14 | 1 | |
| unknown (**) | 31 | 27 | 4 | |
| Grade (invasive NOS) | ||||
| 1 | 57 | 48 | 9 | 0.27 |
| 2 | 113 | 85 | 29 | |
| 3 | 156 | 126 | 30 | |
| ER status | ||||
| Negative | 75 | 65 | 10 | 0.07 |
| Positive | 292 | 225 | 67 | |
| PgR status | ||||
| Negative | 87 | 74 | 13 | 0.11 |
| Positive | 280 | 216 | 64 | |
| HER-2status | ||||
| Negative | 263 | 202 | 56 | 0.60 |
| Positive | 104 | 88 | 21 | |
| Luminal status | ||||
| Luminal A (***) | 225 | 169 | 56 | 0.51 |
| Luminal B (****) | 76 | 61 | 15 | 0.02 (##) |
| Her-2 enriched | 28 | 23 | 5 | |
| Triple negative | 38 | 37 | 1 | |
| Surgery (invasive tumors) | ||||
| Conservative | 335 | 284 | 71 | 0.30 (###) |
| no axillary intervention | 31 | 27 | 4 | |
| axillary sampling/dissection | 324 | 257 | 67 | |
| dissected nodes 0–10 | 111 | 87 | 24 | |
| dissected nodes 11–16 | 140 | 115 | 25 | |
| dissected nodes >16 | 73 | 55 | 18 | |
| Chemotherapy (*****) | ||||
| Hormonal therapy only | 91 | 72 | 19 | 0.94 |
| Neo-adjuvant | 12 | 9 | 3 | |
| Postoperative | 264 | 209 | 55 |
| No Pts | Gy/f × f | PhysD (Gy) | EQD2n,c (Gy) | Time (Days) | Δt (Days) | EQD2n-T (Gy) | EQD2c-T (Gy) | |
|---|---|---|---|---|---|---|---|---|
| Regimen A: | 290 | |||||||
| Breast/Axilla (*) | 3.5 × 10 | 35 | 43.75 | 12 | 18 | 47.35 | 50.90 | |
| Tumor quadrant | 4 × 2 | 8 | 10.66 | 2 | 3 | 11.26 | 11.86 | |
| Total Tumor quadrant | 3.5 × 10 + 4 × 2 | 54.41 | 16 | 21 | 58.61 | 62.76 | ||
| Regimen B: | 77 | |||||||
| Breast/Axilla (*) | 2.7 × 16 | 43.2 | 48.24 | 22 | 10 | 50.24 | 52.24 | |
| Total Tumor quadrant | 3.5 × 8 + 2.7 × 8 | 35 + 24.1 (total 59.1) | 10 + 12 (total 22) | 18 | 62.7 | 66.3 |
| All Patients | Schedule A | Schedule B | p-Value | |
|---|---|---|---|---|
| 367 | 290 | 77 | ||
| Radiation dermatitis | ||||
| 0/1. None to Faint erythema | 321 (87.5) | 259 (89.3) | 62 ((80.5) | 0.06 |
| 2. Brisk erythema/Patchy moist desquamation | 46 (12.5) | 31 (10.7) | 15 (19.5) | |
| 3. Confluent moist desquamation | 0 (0) | 0 (0) | 0 (0) | |
| 4. Skin necrosis | 0 (0) | 0 (0) | 0 (0) | |
| Breast Edema (modified by authors) | ||||
| 0. None | 296 (80.6) | 237 (81.7) | 59 (76.7) | 0.58 |
| 1. Barely palpable/asymptomatic | 56 (15.3 | 43 (14.8) | 13 (16.8) | |
| 2. Moderate/tolerable | 10 (2.7) | 7 (2.4) | 3 (3.9) | |
| 3. Requiring therapy | 5 (1.4) | 3 (1.0) | 2 (2.6) | |
| Pain (Breast/arm) | ||||
| 0. NoNone | 330 (89.9) | 263 (90.7) | 67 (87.0) | 0.31 |
| 1. Mild discomfort | 34 (9.2) | 24 (8.3) | 10 (13.0) | |
| 2. Moderate | 3 (1.0) | 3 (1.0) | 0 (0) | |
| 3. Requiring analgesics | 0 (0) | 0 (0) | 0 (0) | |
| Pneumonitis | ||||
| 0. None | 367 (100) | 290 (100) | 77 (100) | |
| Other | 0 (0) | 0 (0) | 0 (0) |
| All Patients | Schedule A | Schedule B | p-Value | |
|---|---|---|---|---|
| 367 | 290 | 77 | ||
| Breast edema | ||||
| 0. None | 311 (84.7) | 244 (84.1) | 67 (87.0) | |
| 1. Barely evident/Asymptomatic | 48 (13.1) | 41 (14.2) | 7 (9.0) | |
| 0. Evident/Tolerable | 8 (2.2) | 5 (1.7) | 3 (4.0) | |
| 1. Dysfunctional | 0 (0) | 0 (0) | 0 (0) | |
| Fibrosis/breast shrinkage | ||||
| 0. None | 310 (84.5) | 251 (86.6) | 61 (79.2) | 0.27 |
| 1. Palpable/evident in the booster field | 50 (13.6) | 34 (11.7) | 14 (18.2) | |
| 0. Marked breast shrinkage | 7 (1.9) | 5 (1.7) | 2 (2.6) | |
| 1. Very marked firmness/fixation | 0 (0) | 0 (0) | 0 (0) | |
| Skin telangiectasia (modified by authors) | ||||
| 0. None | 290 (79) | 231 (79.7) | 59 (76.6) | 0.83 |
| 1. Sparse (in the tumor bed) | 65 (17.8) | 50 (17.2) | 15 (19.5) | |
| 2. Dense (in the tumor bed) | 12 (3.2) | 9 (3.1) | 3 (3.9) | |
| 3. Outside the tumor bed | 0 (0) | 0 (0) | 0 (0) | |
| Skin atrophy or ulceration | ||||
| 0. None | 367 (100) | 290 (100) | 77 (100) | 0.20 |
| Arm lymphedema (compared to postoperative) | ||||
| 0. None | 331 (90.2) | 260 (89.7) | 71 (92.2) | |
| 1. 2–4 cm | 28 (7.6) | 25 (8.6) | 3 (3.9) | |
| 0. 4–6 cm | 8 (2.2) | 5 (1.7) | 3 (3.9) | |
| Pain (Breast/arm) | ||||
| 0. None | 353 (96.2) | 278 (95.8) | 75(97.4) | 0.76 |
| 1. Mild | 13 (3.5) | 11 (3.8) | 2 (2.6) | |
| 2. Moderate | 1 (0.2) | 1 (0.4) | 0 (0) | |
| 3. Severe | 0 (0) | 0 (0) | 0 (0) | |
| Lung fibrosis | ||||
| 0. None | 330 (89.9) | 260 (89.7) | 70 (90.9) | 0.75 |
| 1. Radiographic changes/asymptomatic | 37 (10.1) | 28 (9.6) | 7 (9.1) | |
| 2. Dense Radiographic changes/symptomatic | 0 (0) | 2 (0.7) | 0 (0) |
| Univariate | Multivariate | |||
|---|---|---|---|---|
| Variable | HR Ratio | p-Value | HR Ratio | p-Value |
| Local Progression | ||||
| T-stage (T3,4 vs. T1,2) | 2.9 | 0.38 | ||
| N (extracapsular vs. other) | 8.0 | 0.001 | 6.3 | 0.03 |
| Dissected nodes (≥16 vs. <16) | 1.0 | 0.97 | ||
| Margins (pos vs. neg) | 5.1 | 0.02 | 6.7 | 0.01 |
| Multifocality (yes vs. no) | 2.9 | 0.33 | ||
| LVI (yes vs. no) | 1.2 | 0.86 | ||
| Luminal (other vs. A, B) | 5.0 | 0.009 | 3.4 | 0.05 |
| Age (≤50 vs. >50) | 1.5 | 0.46 | ||
| RT-scheme (A vs. B) | 1.2 | 0.76 | ||
| Amifostine (yes vs. no) | 1.2 | 0.74 | ||
| Distant metastasis | ||||
| T-stage (T2,3,4 vs. T1) | 1.8 | 0.02 | 1.6 | 0.09 |
| N (other vs. 0–3) | 3.2 | 0.001 | 1.9 | 0.02 |
| Dissected nodes (≥16 vs. <16) | 1.1 | 0.79 | ||
| Margins (pos vs. neg) | 2.3 | 0.21 | ||
| Multifocality (yes vs. no) | 2.1 | 0.26 | ||
| LVI (yes vs. no) | 1.9 | 0.19 | ||
| Luminal (other vs. A, B) | 3.4 | 0.0001 | 3.1 | <0.0001 |
| Age (≤50 vs. >50) | 0.6 | 0.07 | ||
| RT-scheme (A vs. B) | 1.2 | 0.43 | ||
| Amifostine (yes vs. no) | 1.2 | 0.44 | ||
| Overall Survival | ||||
| T-stage (T2,3,4 vs. T1) | 2.1 | 0.01 | 2.0 | 0.07 |
| N (other vs. 0–3) | 3.1 | 0.004 | 2.1 | 0.02 |
| Dissected nodes (≥16 vs. <16) | 1.0 | 0.91 | ||
| Margins (pos vs. neg) | 2.7 | 0.15 | ||
| Multifocality (yes vs. no) | 2.2 | 0.15 | ||
| LVI (yes vs. no) | 1.7 | 0.30 | ||
| Luminal (other vs. A, B) | 3.5 | <0.0001 | 3.4 | <0.0001 |
| Age (≤50 vs. >50) | 0.6 | 0.12 | ||
| RT-scheme (A vs. B) | 1.2 | 0.53 | ||
| Amifostine (yes vs. no) | 1.1 | 0.76 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koukourakis, I.M.; Panteliadou, M.; Giakzidis, A.G.; Nanos, C.; Abatzoglou, I.; Giatromanolaki, A.; Koukourakis, M.I. Long-Term Results of Postoperative Hypofractionated Accelerated Breast and Lymph Node Radiotherapy (HypoAR) with Hypofractionated Boost. Curr. Oncol. 2021, 28, 3474-3487. https://doi.org/10.3390/curroncol28050300
Koukourakis IM, Panteliadou M, Giakzidis AG, Nanos C, Abatzoglou I, Giatromanolaki A, Koukourakis MI. Long-Term Results of Postoperative Hypofractionated Accelerated Breast and Lymph Node Radiotherapy (HypoAR) with Hypofractionated Boost. Current Oncology. 2021; 28(5):3474-3487. https://doi.org/10.3390/curroncol28050300
Chicago/Turabian StyleKoukourakis, Ioannis M., Marianthi Panteliadou, Axiotis G. Giakzidis, Christos Nanos, Ioannis Abatzoglou, Alexandra Giatromanolaki, and Michael I. Koukourakis. 2021. "Long-Term Results of Postoperative Hypofractionated Accelerated Breast and Lymph Node Radiotherapy (HypoAR) with Hypofractionated Boost" Current Oncology 28, no. 5: 3474-3487. https://doi.org/10.3390/curroncol28050300
APA StyleKoukourakis, I. M., Panteliadou, M., Giakzidis, A. G., Nanos, C., Abatzoglou, I., Giatromanolaki, A., & Koukourakis, M. I. (2021). Long-Term Results of Postoperative Hypofractionated Accelerated Breast and Lymph Node Radiotherapy (HypoAR) with Hypofractionated Boost. Current Oncology, 28(5), 3474-3487. https://doi.org/10.3390/curroncol28050300





