Spontaneous Regression of Metastatic Renal Cell Carcinoma after SARS-CoV-2 Infection: A Report of Two Cases
Abstract
:1. Introduction
2. Case Description
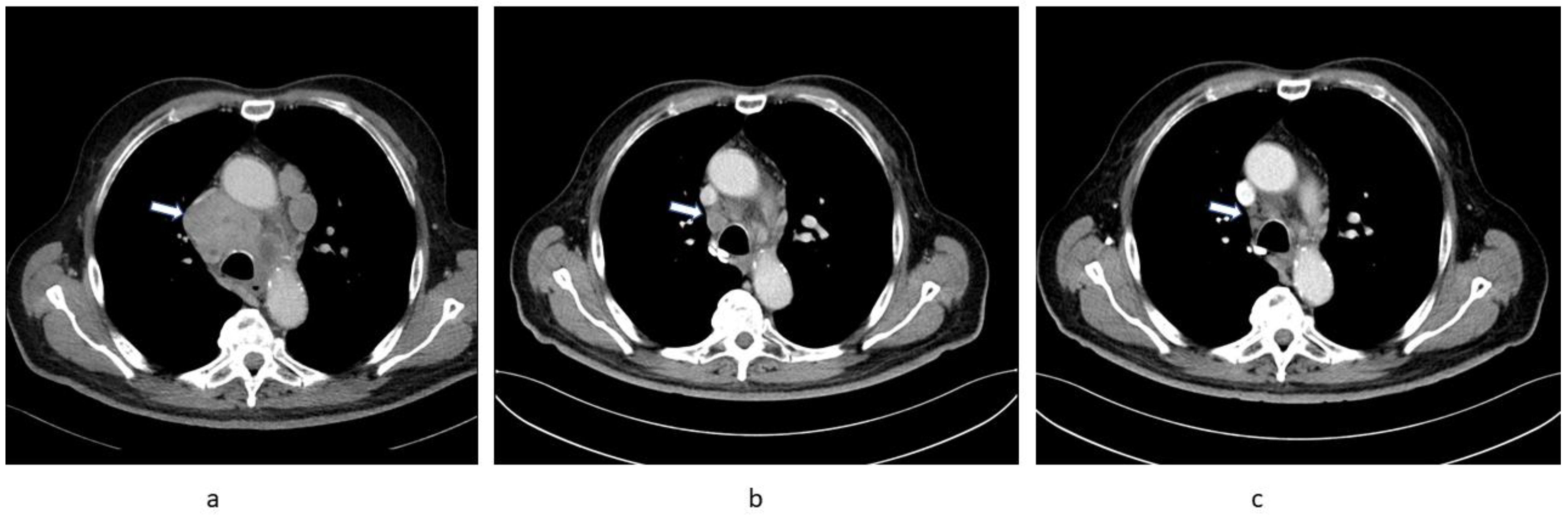
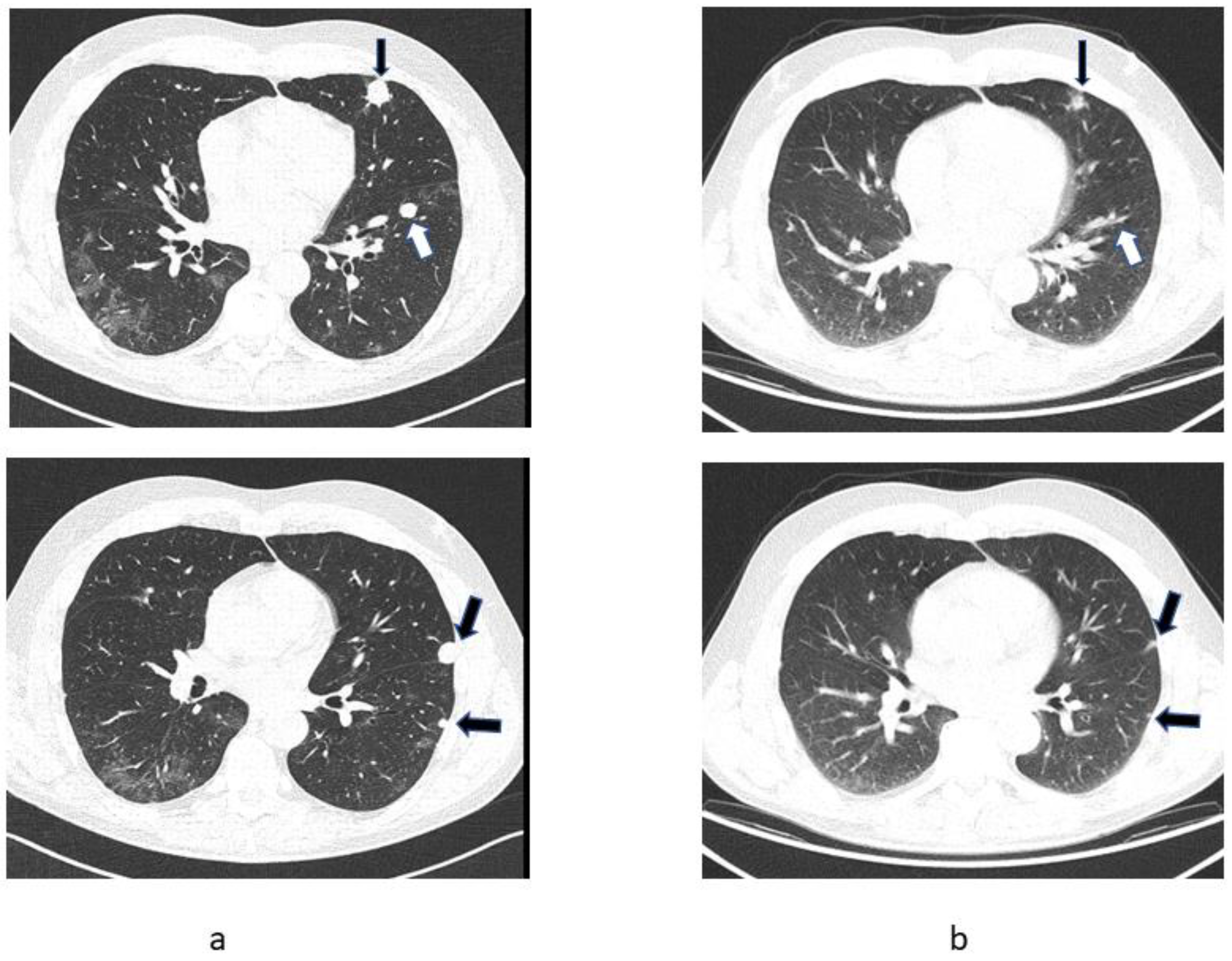
2.1. Patient 1
2.2. Patient 2
3. Discussion
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bumpus, H.C. The Apparent Disappearance of Pulmonary Metastasis in a Case of Hypernephroma Following Nephrectomy. J. Urol. 1928, 20, 185–192. [Google Scholar] [CrossRef]
- Jenkins, G.D. Regression of Pulmonary Metastasis Following Nephrectomy for Hypernephroma: Eight Year Follow-up. J. Urol. 1959, 82, 37–40. [Google Scholar] [CrossRef]
- Melichar, B.; Vaněčková, J.; Morávek, P.; Urminská, H.; Podhola, M. Spontaneous regression of renal cell carcinoma lung metastases in a patient with psoriasis. Acta Oncol. 2009, 48, 925–927. [Google Scholar] [CrossRef] [PubMed]
- Behzad, N. Immunology of the Spontaneous Remission of Cancer. Int. J. Cancer Clin. Res. 2019, 6, 112. [Google Scholar] [CrossRef]
- Challenor, S.; Tucker, D. SARS-CoV-2-induced remission of Hodgkin lymphoma. Br. J. Haematol. 2021, 192, 415. [Google Scholar] [CrossRef] [PubMed]
- Sollini, M.; Gelardi, F.; Carlo-Stella, C.; Chiti, A. Complete remission of follicular lymphoma after SARS-CoV-2 infection: From the “flare phenomenon” to the “abscopal effect”. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 2652–2654. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef] [Green Version]
- Tay, M.Z.; Poh, C.M.; Rénia, L.; Macary, P.A.; Ng, L.F.P. The trinity of COVID-19: Immunity, inflammation and intervention. Nat. Rev. Immunol. 2020, 20, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Jordan, S.C.; Zakowski, P.; Tran, H.P.; Smith, E.A.; Gaultier, C.; Marks, G.; Zabner, R.; Lowenstein, H.; Oft, J.; Bluen, B.; et al. Compassionate Use of Tocilizumab for Treatment of SARS-CoV-2 Pneumonia. Clin. Infect. Dis. 2020, 71, 3168–3173. [Google Scholar] [CrossRef] [PubMed]
- Carvelli, J.; Demaria, O.; Vély, F.; Batista, L.; Benmansour, N.C.; Fares, J.; Carpentier, S.; Thibult, M.-L.; Morel, A.; Remark, R.; et al. Association of COVID-19 inflammation with activation of the C5a–C5aR1 axis. Nature 2020, 588, 146–150. [Google Scholar] [CrossRef] [PubMed]
- Jordan, S.C. Innate and adaptive immune responses to SARS-CoV-2 in humans: Relevance to acquired immunity and vaccine responses. Clin. Exp. Immunol. 2021, 204, 310–320. [Google Scholar] [CrossRef] [PubMed]
- Law, T.M.; Motzer, R.J.; Mazumdar, M.; Sell, K.W.; Walther, P.; O’Connell, M.; Khan, A.; Vlamis, V.; Vogelzang, N.J.; Bajorin, D.F. Phase iii randomized trial of interleukin-2 with or without lymphokine-activated killer cells in the treatment of patients with advanced renal cell carcinoma. Cancer 1995, 76, 824–832. [Google Scholar] [CrossRef]
- Klapper, J.A.; Downey, S.G.; Smith, F.O.; Yang, J.C.; Hughes, M.S.; Kammula, U.S.; Sherry, R.M.; Royal, R.E.; Steinberg, S.M.; Rosenberg, S. High-dose interleukin-2 for the treatment of metastatic renal cell carcinoma. Cancer 2008, 113, 293–301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Motzer, R.J.; Rini, B.I.; McDermott, D.F.; Frontera, O.A.; Hammers, H.J.; Carducci, M.A.; Salman, P.; Escudier, B.; Beuselinck, B.; Amin, A.; et al. Nivolumab plus ipilimumab versus sunitinib in first-line treatment for advanced renal cell carcinoma: Extended follow-up of efficacy and safety results from a randomised, controlled, phase 3 trial. Lancet Oncol. 2019, 20, 1370–1385. [Google Scholar] [CrossRef]
- Haineala, B.; Zgura, A.; Diaconu, C.; Mehedintu, C.; Bacinschi, X.; Anghel, R.M. Long-term Response After Stopping Immunotherapy in a Patient With Metastatic Renal Cancer. Vivo 2021, 35, 1805–1810. [Google Scholar] [CrossRef] [PubMed]
- Campesato, L.F.; Weng, C.-H.; Merghoub, T. Innate immune checkpoints for cancer immunotherapy: Expanding the scope of non T cell targets. Ann. Transl. Med. 2020, 8, 1031. [Google Scholar] [CrossRef] [PubMed]
- Beberok, A.; Wrześniok, D.; Minecka, A.; Rok, J.; Delijewski, M.; Rzepka, Z.; Respondek, M.; Buszman, E. Ciprofloxacin-mediated induction of S-phase cell cycle arrest and apoptosis in COLO829 melanoma cells. Pharmacol. Rep. 2018, 70, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Shang, Q.; Li, W.; Guo, W.; Stojadinovic, A.; Mannion, C.; Man, Y.-G.; Chen, T. Antibiotics for cancer treatment: A double-edged sword. J. Cancer 2020, 11, 5135–5149. [Google Scholar] [CrossRef] [PubMed]
- Carella, A.M.; Beltrami, G.; Pica, G.; Carella, A.; Catania, G. Clarithromycin potentiates tyrosine kinase inhibitor treatment in patients with resistant chronic myeloid leukemia. Leuk. Lymphoma 2012, 53, 1409–1411. [Google Scholar] [CrossRef]
- Tsikala-Vafea, M.; Belani, N.; Vieira, K.; Khan, H.; Farmakiotis, D. Use of antibiotics is associated with worse clinical outcomes in patients with cancer treated with immune checkpoint inhibitors: A systematic review and meta-analysis. Int. J. Infect. Dis. 2021, 106, 142–154. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buchler, T.; Fiser, L.; Benesova, J.; Jirickova, H.; Votrubova, J. Spontaneous Regression of Metastatic Renal Cell Carcinoma after SARS-CoV-2 Infection: A Report of Two Cases. Curr. Oncol. 2021, 28, 3403-3407. https://doi.org/10.3390/curroncol28050294
Buchler T, Fiser L, Benesova J, Jirickova H, Votrubova J. Spontaneous Regression of Metastatic Renal Cell Carcinoma after SARS-CoV-2 Infection: A Report of Two Cases. Current Oncology. 2021; 28(5):3403-3407. https://doi.org/10.3390/curroncol28050294
Chicago/Turabian StyleBuchler, Tomas, Lukas Fiser, Jaroslava Benesova, Hana Jirickova, and Jana Votrubova. 2021. "Spontaneous Regression of Metastatic Renal Cell Carcinoma after SARS-CoV-2 Infection: A Report of Two Cases" Current Oncology 28, no. 5: 3403-3407. https://doi.org/10.3390/curroncol28050294
APA StyleBuchler, T., Fiser, L., Benesova, J., Jirickova, H., & Votrubova, J. (2021). Spontaneous Regression of Metastatic Renal Cell Carcinoma after SARS-CoV-2 Infection: A Report of Two Cases. Current Oncology, 28(5), 3403-3407. https://doi.org/10.3390/curroncol28050294




