Symptom Burden of Patients with Advanced Pancreas Cancer (APC): A Provincial Cancer Institute Observational Study
Abstract
:1. Introduction
- 1
- To describe the symptom burden of patients with APC at baseline, using the ESAS-r.
- 2
- To examine whether baseline ESAS-r is a prognostic marker for OS.
2. Materials and Methods
3. Results
3.1. Patient Characteristics
3.2. Symptom Burden at Baseline
3.3. Symptom Burden over Time
3.4. Survival Outcomes
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Brenner, D.R.; Weir, H.K.; Demers, A.A.; Ellison, L.F.; Louzado, C.; Shaw, A.; Turner, D.; Woods, R.R.; Smith, L.M. Projected Estimates of Cancer in Canada in 2020. CMAJ 2020, 192, e199–e205. [Google Scholar] [CrossRef] [Green Version]
- SEER. Cancer Stat Facts: Pancreatic Cancer. 2020. Available online: https://seer.cancer.gov/statfacts/html/pancreas.html (accessed on 12 November 2020).
- Johnson, A.M.; Wolf, S.P.; Xuan, M.; Samsa, G.; Kamal, A.; Fisher, D.A. Patient-reported symptom burden in pancreatic cancer across a multi-site palliative care collaborative. Gastroenterology 2019, 156, s318. [Google Scholar] [CrossRef]
- Moffat, G.T.; Epstein, A.S.; O’Reilly, E.M. Pancreatic Cancer—A Disease in Need: Optimizing and Integrating Supportive Care. Cancer 2019, 125, 3927–3935. [Google Scholar] [CrossRef] [PubMed]
- Torgerson, S.; Wiebe, L.A. Supportive Care of the Patient with Advanced Pancreatic Cancer. Oncol 2013, 27, 183–190. [Google Scholar]
- Tang, C.C.; Von Ah, D.; Fulton, J.S. The Symptom Experience of Patients with Advanced Pancreatic Cancer: An Integrative Review. Cancer Nurs. 2018, 41, 33–44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hammad, A.; Davis, L.E.; Mahar, A.L.; Bubis, L.D.; Zhao, H.; Earle, C.C.; Barbera, L.; Hallet, J.; Coburn, N.G. Symptom Trajectories and Predictors of Severe Symptoms in Pancreatic Adenocarcinoma at the End-of-Life: A Population Based Analysis of 2538 Patients. HPB 2019, 30, 1. [Google Scholar] [CrossRef]
- Cheung, W.Y.; Barmala, N.; Zarinehbaf, S.; Rodin, G.; Le, L.W.; Zimmermann, C. The Association of Physical and Psychological Symptom Burden with Time to Death Among Palliative Cancer Outpatients. J. Pain Symptom Manag. 2009, 37, 297–304. [Google Scholar] [CrossRef]
- Watanabe, S.M.; Nekolaichuk, C.L.; Beaumont, C. The Edmonton Symptom Assessment System, a Proposed Tool for Distress Screening in Cancer Patients: Development and Refinement. Psychooncology 2012, 21, 977–985. [Google Scholar] [CrossRef]
- Oldenmenger, W.H.; De Raaf, P.J.; De Klerk, C.; Van Der Rijt, C.C.D. Cut Points on 0–10 Numeric Rating Scales for Symptoms Included in the Edmonton Symptom Assessment Scale in Cancer Patients: A Systematic Review. J. Pain Symptom Manag. 2013, 45, 1083–1093. [Google Scholar] [CrossRef] [Green Version]
- Selby, D.; Cascella, A.; Gardiner, K.; Do, R.; Moravan, V.; Myers, J.; Chow, E. A Single Set of Numerical Cutpoints to Define Moderate and Severe Symptoms for the Edmonton Symptom Assessment System. J. Pain Symptom Manag. 2010, 39, 241–249. [Google Scholar] [CrossRef]
- Bruera, E.; Kuehn, N.; Miller, M.J.; Selmser, P.; Macmillan, K. The Edmonton Symptom Assessment System (ESAS): A Simple Method for the Assessment of Palliative Care Patients. J. Palliat. Care 1991, 7, 6–9. [Google Scholar] [CrossRef]
- Nekolaichuk, C.; Watanabe, S.; Beaumont, C. The Edmonton Symptom Assessment System: A 15-Year Retrospective Review of Validation Studies (1991–2006). Palliat. Med. 2008, 22, 111–222. [Google Scholar] [CrossRef]
- Richardson, L.A.; Jones, G.W. A Review of the Reliability and Validity of the Edmonton Symptom Assessment System. Curr. Oncol. 2009, 16, 53–64. [Google Scholar] [CrossRef] [Green Version]
- Hannon, B.; Dyck, M.; Pope, A.; Swami, N.; Banerjee, S.; Mak, E.; Bryson, J.; Rodin, G.; Ridley, J.; Lo, C.; et al. Modified Edmonton Symptom Assessment System Including Constipation and Sleep: Validation in Outpatients with Cancer. J. Pain Symptom Manag. 2015, 49, 945–952. [Google Scholar] [CrossRef]
- Hui, D.; Bruera, E. The Edmonton Symptom Assessment System 25 Years Later: Past, Present, and Future Developments. J. Pain Symptom Manag. 2017, 53, 630–643. [Google Scholar] [CrossRef] [Green Version]
- Yalcin, S.; Dane, F.; Oksuzoglu, B.; Ozdemir, N.Y.; Isikdogan, A.; Ozkan, M.; Demirag, G.G.; Coskun, H.S.; Karabulut, B.; Evrensel, T.; et al. Quality of Life Study of Patients with Unresectable Locally Advanced or Metastatic Pancreatic Adenocarcinoma Treated with Gemcitabine+nab-Paclitaxel versus Gemcitabine Alone: AX-PANC-SY001, a Randomized Phase-2 Study. BMC Cancer 2020, 20, 259–270. [Google Scholar] [CrossRef] [Green Version]
- Von Hoff, D.D.; Ervin, T.; Arena, F.P.; Chiorean, E.G.; Infante, J.; Moore, M.; Seay, T.; Tjulandin, S.A.; Ma, W.W.; Saleh, M.N.; et al. Increased Survival in Pancreatic Cancer with Nab-Paclitaxel plus Gemcitabine. N. Engl. J. Med. 2013, 369, 1691–1703. [Google Scholar] [CrossRef] [Green Version]
- Conroy, T.; Desseigne, F.; Ychou, M.; Bouché, O.; Guimbaud, R.; Bécouarn, Y.; Adenis, A.; Raoul, J.-L.; Gourgou-Bourgade, S.; de la Fouchardière, C.; et al. FOLFIRINOX versus Gemcitabine for Metastatic Pancreatic Cancer. N. Engl. J. Med. 2011, 364, 1817–1825. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rizopoulos, D. JM: An R Package for the Joint Modelling of Longitudinal and Time-To-Event Data. J. Stat. Softw. 2010, 35, 1–33. [Google Scholar] [CrossRef]
- Team, R.C. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. Available online: https://www.r-project.org/ (accessed on 26 January 2021).
- Rizopoulos, D. JM: Joint Modeling of Longitudinal and Survival Data. R Package Version 1.4-8. Available online: https://CRAN.R-project.org/package=JM (accessed on 26 January 2021).
- Harrell, F.E.J. RMS: Regression Modeling Strategies. R Package Version 6.1-0. Available online: https://CRAN.R-project.org/package=rms (accessed on 26 January 2021).
- Bubis, L.D.; Davis, L.; Mahar, A.; Barbera, L.; Li, Q.; Moody, L.; Karanicolas, P.; Sutradhar, R.; Coburn, N.G. Symptom Burden in the First Year after Cancer Diagnosis: An Analysis of Patient-Reported Outcomes. J. Clin. Oncol. 2018, 36, 1103–1111. [Google Scholar] [CrossRef] [PubMed]
- Batra, A.; Yang, L.; Boyne, D.J.; Harper, A.; Cheung, W.Y.; Cuthbert, C.A. Associations between Baseline Symptom Burden as Assessed by Patient-Reported Outcomes and Overall Survival of Patients with Metastatic Cancer. Support. Care Cancer 2020, 29, 1423–1431. [Google Scholar] [CrossRef]
- McGee, S.F.; Zhang, T.; Jonker, H.; Laurie, S.A.; Goss, G.; Nicholas, G.; Albaimani, K.; Wheatley-Price, P. The Impact of Baseline Edmonton Symptom Assessment Scale Scores on Treatment and Survival in Patients With Advanced Non–Small-Cell Lung Cancer. Clin. Lung Cancer 2018, 19, e91–e99. [Google Scholar] [CrossRef]
- Reilly, C.M.; Bruner, D.W.; Mitchell, S.A.; Minasian, L.M.; Basch, E.; Dueck, A.C.; Cella, D.; Reeve, B.B. A Literature Synthesis of Symptom Prevalence and Severity in Persons Receiving Active Cancer Treatment. Support. Care Cancer 2013, 21, 1525–1550. [Google Scholar] [CrossRef] [Green Version]
- Reyes-Gibby, C.C.; Chan, W.; Abbruzzese, J.L.; Xiong, H.Q.; Ho, L.; Evans, D.B.; Varadhachary, G.; Bhat, S.; Wolff, R.A.; Crane, C. Patterns of Self-Reported Symptoms in Pancreatic Cancer Patients Receiving Chemoradiation. J. Pain Symptom Manag. 2007, 34, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Dai, W.F.; Beca, J.; Guo, H.; Isaranawatchai, W.; Schwartz, D.; Naipaul, R.; Arias, J.; Qiao, Y.; Gavura, S.; Redmond-Misner, R.; et al. Are Population-Based Patient-Reported Outcomes Associated with Overall Survival in Patients with Advanced Pancreatic Cancer? Cancer Med. 2020, 9, 215–224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sherman, D.W.; Mcmillan, S.C. The Physical Health of Patients with Advanced Pancreatic Cancer and the Psychological Health of Their Family Caregivers When Newly Enrolled in Hospice. J. Hosp. Palliat. Nurs. 2015, 17, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Cuthbert, C.A.; Boyne, D.J.; Yuan, X.; Hemmelgarn, B.R.; Cheung, W.Y. Patient-Reported Symptom Burden and Supportive Care Needs at Cancer Diagnosis: A Retrospective Cohort Study. Support. Care Cancer 2020, 28, 5889–5899. [Google Scholar] [CrossRef]
- Graham, J.; Gingerich, J.; Lambert, P.; Alamri, A.; Czaykowski, P. Baseline Edmonton Symptom Assessment System and Survival in Metastatic Renal Cell Carcinoma. Curr. Oncol. 2018, 25, e319–e323. [Google Scholar] [CrossRef] [Green Version]
- Lee, G.; Kim, H.S.; Lee, S.W.; Park, Y.R.; Kim, E.H.; Lee, B.; Hu, Y.J.; Kim, K.A.; Kim, D.A.; Cho, H.Y.; et al. Pre-Screening of Patient-Reported Symptoms Using the Edmonton Symptom Assessment System in Outpatient Palliative Cancer Care. Eur. J. Cancer Care 2020, 29, e13305. [Google Scholar] [CrossRef] [PubMed]
- Barbera, L.; Seow, H.; Howell, D.; Sutradhar, R.; Earle, C.; Liu, Y.; Stitt, A.; Husain, A.; Sussman, J.; Dudgeon, D. Symptom Burden and Performance Status in a Population-Based Cohort of Ambulatory Cancer Patients. Cancer 2010, 116, 5767–5776. [Google Scholar] [CrossRef]
- Mendoza, T.R.; Kehl, K.L.; Bamidele, O.; Williams, L.A.; Shi, Q.; Cleeland, C.S.; Simon, G. Assessment of Baseline Symptom Burden in Treatment-Naïve Patients with Lung Cancer: An Observational Study. Support. Care Cancer 2019, 27, 3439–3447. [Google Scholar] [CrossRef] [PubMed]
- Gourgou-Bourgade, S.; Bascoul-Mollevi, C.; Desseigne, F.; Ychou, M.; Bouché, O.; Guimbaud, R.; Bécouarn, Y.; Adenis, A.; Raoul, J.L.; Boige, V.; et al. Impact of FOLFIRINOX Compared with Gemcitabine on Quality of Life in Patients with Metastatic Pancreatic Cancer: Results from the PRODIGE 4/ACCORD 11 Randomized Trial. J. Clin. Oncol. 2013, 31, 23–29. [Google Scholar] [CrossRef]
- Schönherr, C.; Götze, T.O.; zur Hausen, G.; Reichart, A.; Pauligk, C.; Schlag, R.; Siegler, G.M.; Dörfel, S.; Aldaoud, A.; Hahn, L.; et al. Quality of Life (QoL) in Patients with Metastatic Pancreatic Cancer Receiving First-Line Nab-Paclitaxel/Gemcitabine Chemotherapy: Results of the Large QoL Study AIO-QoliXane/PARAGON. Ann. Oncol. 2018, 29, 252. [Google Scholar] [CrossRef]
- Royal, R.E.; Levy, C.; Turner, K.; Mathur, A.; Hughes, M.; Kammula, U.S.; Sherry, R.M.; Topalian, S.L.; Yang, J.C.; Lowy, I.; et al. Phase 2 Trial of Single Agent Ipilimumab (Anti-CTLA-4) for Locally Advanced or Metastatic Pancreatic Adenocarcinoma. J. Immunother. 2010, 33, 828–833. [Google Scholar] [CrossRef]
- O’Reilly, E.M.; Oh, D.Y.; Dhani, N.; Renouf, D.J.; Lee, M.A.; Sun, W.; Fisher, G.; Hezel, A.; Chang, S.C.; Vlahovic, G.; et al. Durvalumab with or Without Tremelimumab for Patients with Metastatic Pancreatic Ductal Adenocarcinoma: A Phase 2 Randomized Clinical Trial. JAMA Oncol. 2019, 5. [Google Scholar] [CrossRef]
- AstraZeneca. Study of Tremelimumab in Patients With Advanced Solid Tumors. Available online: https://clinicaltrials.gov/ct2/show/results/NCT02527434 (accessed on 17 July 2021).
- Weiss, G.J.; Blaydorn, L.; Beck, J.; Bornemann-Kolatzki, K.; Urnovitz, H.; Schütz, E.; Khemka, V. Phase Ib/II Study of Gemcitabine, Nab-Paclitaxel, and Pembrolizumab in Metastatic Pancreatic Adenocarcinoma. Investig. New Drugs 2018, 36, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Renouf, D.J.; Knox, J.J.; Kavan, P.; Jonker, D.; Welch, S.; Couture, S.; Lemay, F.; Tehfe, M.; Harb, M.; Aucoin, N.; et al. LBA65—The Canadian Cancer Trials Group PA.7 trial: Results of a randomized phase II study of gemcitabine (GEM) and nab-paclitaxel (Nab-P) vs GEM, nab-P, durvalumab (D) and tremelimumab (T) as first line therapy in metastatic pancreatic ductal adenocarcinoma (mPDAC). In proceedings of the 2020 ESMO Conference. Ann. Oncol. 2020, 31 (Suppl. 4), s1142–s1215. [Google Scholar]
- Nct. A Study of Nivolumab by Itself or Nivolumab Combined With Ipilimumab in Patients With Advanced or Metastatic Solid Tumors. Available online: https://clinicaltrials.gov/show/NCT01928394 (accessed on 17 July 2021).
- Luchini, C.; Brosens, L.A.A.; Wood, L.D.; Chatterjee, D.; Shin, J., II; Sciammarella, C.; Fiadone, G.; Malleo, G.; Salvia, R.; Kryklyva, V.; et al. Comprehensive Characterisation of Pancreatic Ductal Adenocarcinoma with Microsatellite Instability: Histology, Molecular Pathology and Clinical Implications. Gut 2021, 70, 148–156. [Google Scholar] [CrossRef]
- Marabelle, A.; Le, D.T.; Ascierto, P.A.; Di Giacomo, A.M.; de Jesus-Acosta, A.; Delord, J.P.; Geva, R.; Gottfried, M.; Penel, N.; Hansen, A.R.; et al. Efficacy of Pembrolizumab in Patients with Noncolorectal High Microsatellite Instability/ Mismatch Repair–Deficient Cancer: Results from the Phase II KEYNOTE-158 Study. J. Clin. Oncol. 2020, 38, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Marabelle, A.; Fakih, M.G.; Lopez, J.; Shah, M.; Shapira-Frommer, R.; Nakagawa, K.; Chung, H.C.; Kindler, H.L.; Lopez-Martin, J.A.; Miller, W.; et al. Association of tumour mutational burden with outcomes in patients with select advanced solid tumours treated with pembrolizumab in KEYNOTE-158. Ann. Oncol. 2019, 30, v477–v478. [Google Scholar] [CrossRef]
- Al-Batran, S.E.; Hofheinz, R.D.; Reichart, A.; Pauligk, C.; Schönherr, C.; Schlag, R.; Siegler, G.; Dörfel, S.; Koenigsmann, M.; Zahn, M.O.; et al. Quality of Life and Outcome of Patients with Metastatic Pancreatic Cancer Receiving First-Line Chemotherapy with Nab-Paclitaxel and Gemcitabine: Real-Life Results from the Prospective QOLIXANE Trial of the Platform for Outcome, Quality of Life and Translational Research on Pancreatic Cancer Registry. Int. J. Cancer 2021, 148, 1478–1488. [Google Scholar] [CrossRef] [PubMed]
- Gooden, H.M.; White, K.J. Pancreatic Cancer and Supportive Care—Pancreatic Exocrine Insufficiency Negatively Impacts on Quality of Life. Support. Care Cancer 2013, 21, 1835–1841. [Google Scholar] [CrossRef]
- Steindorf, K.; Clauss, D.; Tjaden, C.; Hackert, T.; Herbolsheimer, F.; Bruckner, T.; Schneider, L.; Ulrich, C.M.; Wiskemann, J. Quality of Life, Fatigue, and Sleep Problems in Pancreatic Cancer Patients—a Randomized Trial on the Effects of Exercise. Dtsch. Arztebl. Int. 2019, 116, 471–478. [Google Scholar] [CrossRef]
- Sloan, J.A.; Zhao, X.; Novotny, P.J.; Wampfler, J.; Garces, Y.; Clark, M.M.; Yang, P. Relationship between Deficits in Overall Quality of Life and Non-Small-Cell Lung Cancer Survival. J. Clin. Oncol. 2012, 30, 1498–1504. [Google Scholar] [CrossRef]
- Gotay, C.C.; Kawamoto, C.T.; Bottomley, A.; Efficace, F. The Prognostic Significance of Patient-Reported Outcomes in Cancer Clinical Trials. J. Clin. Oncol. 2008, 26, 1355–1363. [Google Scholar] [CrossRef]
- Quinten, C.; Coens, C.; Mauer, M.; Comte, S.; Sprangers, M.A.; Cleeland, C.; Osoba, D.; Bjordal, K.; Bottomley, A. Baseline Quality of Life as a Prognostic Indicator of Survival: A Meta-Analysis of Individual Patient Data from EORTC Clinical Trials. Lancet Oncol. 2009, 10, 865–871. [Google Scholar] [CrossRef]
- Nguyen, J.; Di Giovanni, J.; Zhang, L.; Popovic, M.; Zeng, L.; Jamani, R.; Cramarossa, G.; Culleton, S.; Jon, F.; Chow, E. Projected Referral to Other Healthcare Services in an Outpatient Palliative Radiotherapy Clinic. Expert Rev. Pharmacoecon. Outcomes Res. 2012, 12, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Hui, D.; Mori, M.; Watanabe, S.M.; Caraceni, A.; Strasser, F.; Saarto, T.; Cherny, N.; Glare, P.; Kaasa, S.; Bruera, E. Referral Criteria for Outpatient Specialty Palliative Cancer Care: An International Consensus. Lancet Oncol. 2016, 17, e552–e559. [Google Scholar] [CrossRef] [Green Version]
- Zimmermann, C.; Hannon, B.; Krzyzanowska, M.K.; Li, M.; Rodin, G.; Pope, A.; Swami, N.; Giruparajah, M.; Howell, D.; Oza, A.M.; et al. Phase 2 Trial of Symptom Screening with Targeted Early Palliative Care (STEP) for Patients with Advanced Cancer. J. Clin. Oncol. 2019, 37, s11604. [Google Scholar] [CrossRef]
- Sohal, D.P.S.; Mangu, P.B.; Khorana, A.A.; Shah, M.A.; Philip, P.A.; O’Reilly, E.M.; Uronis, H.E.; Ramanathan, R.K.; Crane, C.H.; Engebretson, A.; et al. Metastatic Pancreatic Cancer: American Society of Clinical Oncology Clinical Practice Guideline. J. Clin. Oncol. 2016, 34, 2784–2796. [Google Scholar] [CrossRef]
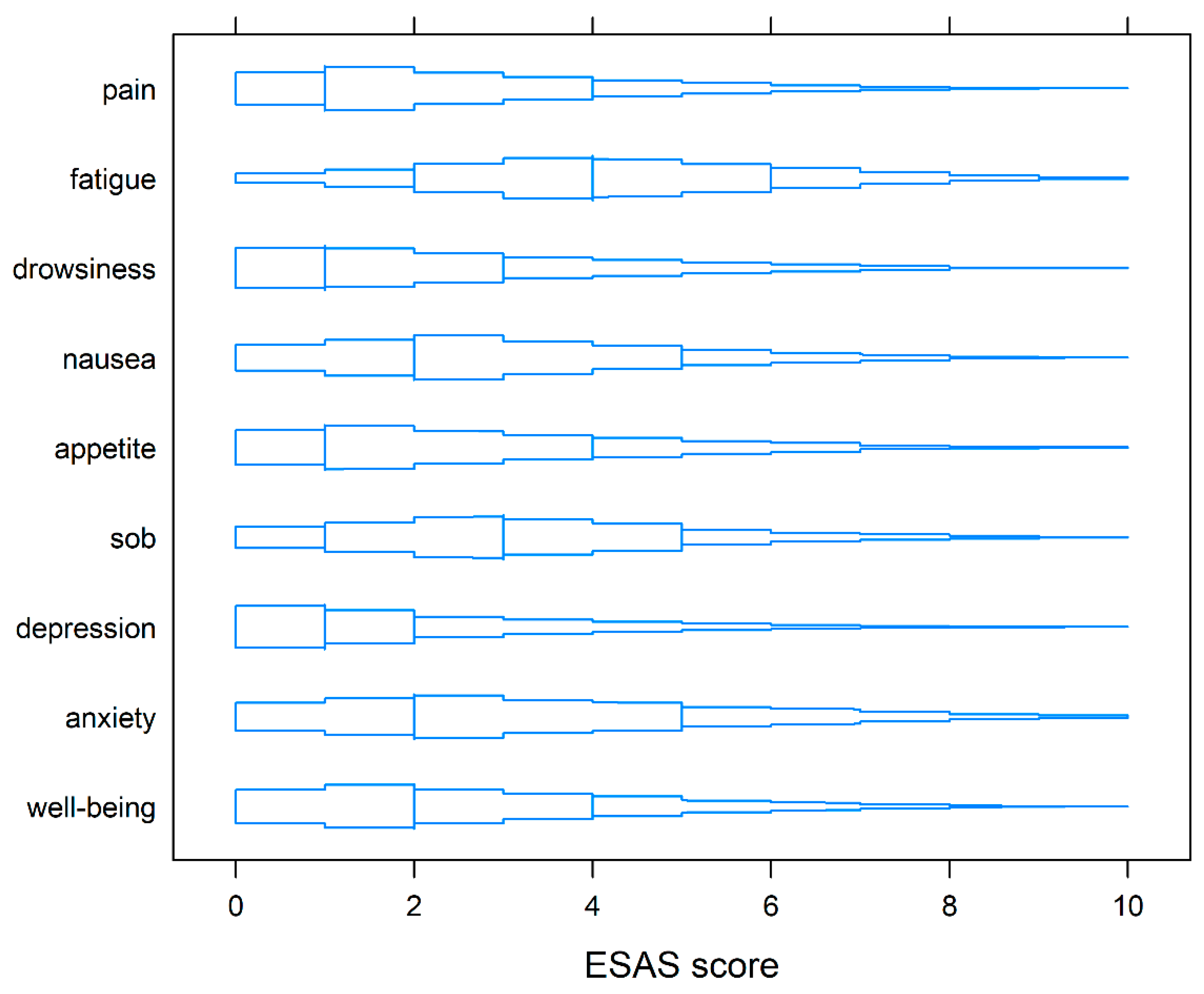
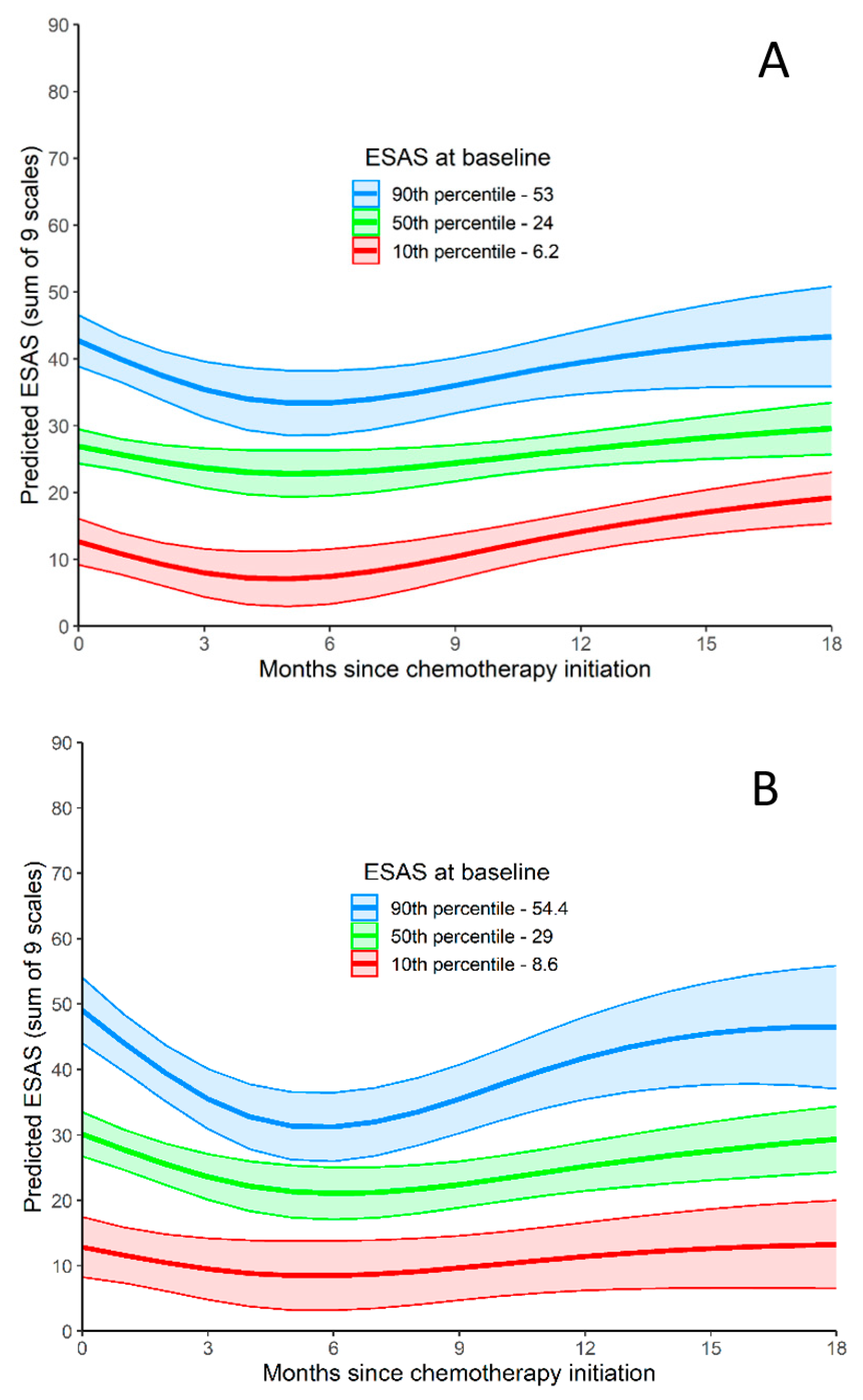
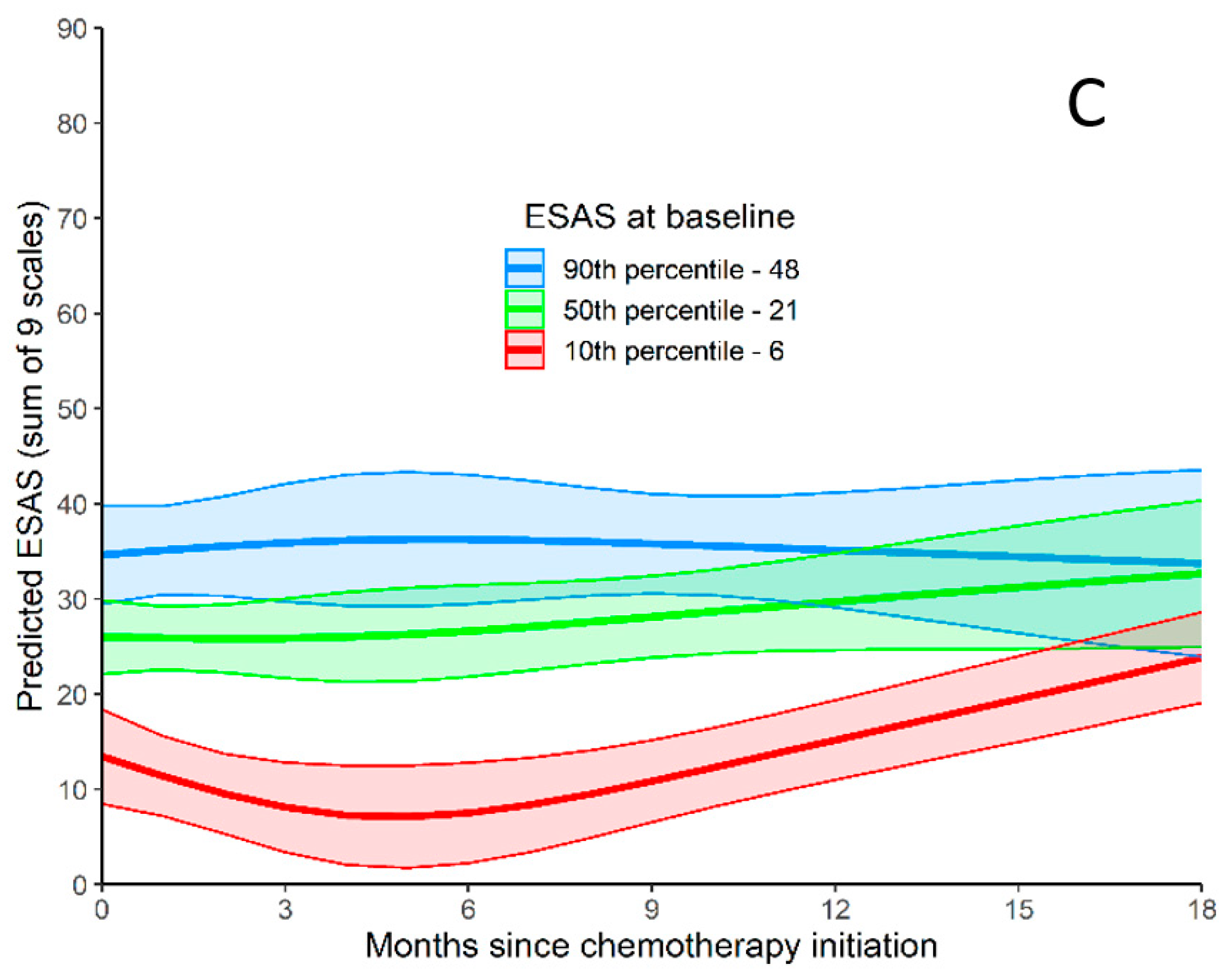
| Characteristic | n (%) |
|---|---|
| Median Age at diagnosis | 65 (range 42–88) |
| <65 | 57 (46.3) |
| ≥65 | 66 (53.7) |
| Sex | |
| Male | 65 (52.8) |
| Female | 58 (47.2) |
| Histology | |
| Adenocarcinoma | 119 (96.7) |
| Other * | 4 (3.3) |
| ECOG | |
| 0–1 | 101 (82.1) |
| 2–3 | 19 (15.4) |
| Unknown | 3 (2.4) |
| Metastatic disease | |
| Yes | 75 (61.0) |
| No | 48 (39.0) |
| Tumor Location | |
| Head/Neck | 76 (61.8) |
| Body | 26 (21.1) |
| Tail | 18 (14.6) |
| Unknown | 3 (2.4) |
| Chemotherapy Regimen | |
| FOLFIRINOX | 85 (69.1) |
| Nab-Paclitaxel + Gemcitabine | 27 (22.0) |
| Gemcitabine | 11 (8.9) |
| Number of visits with ESAS-r assessment | |
| 1 | 16 (13.0) |
| 2 | 12 (9.8) |
| 3 | 5 (4.1) |
| 4 | 9 (7.3) |
| 5 | 7 (5.7) |
| 6–10 | 23 (18.7) |
| 11–20 | 23 (18.7) |
| >21 | 28 (22.8) |
| Median Progression Free Survival | 6.7 months |
| Median OS | 10.2 months |
| Symptom | n (%) |
|---|---|
| Fatigue | 70 (56.9) |
| Anxiety | 62 (50.4) |
| Shortness of breath | 60 (48.8) |
| Nausea | 12 (9.8) |
| Variable | Univariable | Multivariable | |||||
|---|---|---|---|---|---|---|---|
| HR | 95% CI | p | HR | 95% CI | p | ||
| TSDS | per 10 | 1.10 | 0.99–1.23 | 0.081 | 0.99 | 0.84–1.18 | 0.951 |
| Physical ≥ 4 | Yes | 1.33 | 0.84–2.13 | 0.228 | 1.22 | 0.66–2.25 | 0.519 |
| No | 1 | 1 | |||||
| Psychological ≥ 4 | Yes | 1.29 | 0.88–1.90 | 0.197 | 1.15 | 0.69–1.91 | 0.588 |
| No | 1 | 1 | |||||
| Metastasis | Yes | 1.74 | 1.14–2.65 | 0.010 | |||
| No | 1 | ||||||
| CT regimen | FOLFIRINOX | 0.33 | 0.17–0.67 | 0.002 | |||
| Nab-Paclitaxel/Gem | 0.38 | 0.17–0.85 | 0.019 | ||||
| GEM | 1 | ||||||
| Age | (in years) | 1.01 | 0.99–1.03 | 0.321 | |||
| ECOG | ≥2 | 1.54 | 0.81–2.93 | 0.190 | |||
| 0–1 | 1 | ||||||
| Variable | Univariable | Multivariable | |||||
|---|---|---|---|---|---|---|---|
| HR | 95% CI | p | HR | 95% CI | p | ||
| TSDS (time-varying) | per 10 | 1.68 | 1.43–1.97 | <0.001 | 1.65 | 1.39–1.95 | <0.001 |
| Physical ≥ 4 | Yes | 0.62 | 0.35–1.07 | 0.087 | 0.66 | 0.37–1.20 | 0.171 |
| No | 1 | 1 | |||||
| Psychological ≥ 4 | Yes | 0.99 | 0.64–1.54 | 0.961 | 0.91 | 0.58–1.43 | 0.689 |
| No | 1 | 1 | |||||
| Metastasis | Yes | 2.05 | 1.3–3.22 | 0.002 | |||
| No | 1 | ||||||
| Treatment | FOLFIRINOX | 0.59 | 0.29–1.23 | 0.163 | |||
| Nab-Paclitaxel/Gem | 0.58 | 0.25–1.36 | 0.212 | ||||
| GEM | 1 | ||||||
| Age | (in years) | 1.02 | 0.99–1.04 | 0.147 | |||
| ECOG | ≥2 | 1.35 | 0.71–2.56 | 0.36 | |||
| 0–1 | 1 | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lelond, S.; Ward, J.; Lambert, P.J.; Kim, C.A. Symptom Burden of Patients with Advanced Pancreas Cancer (APC): A Provincial Cancer Institute Observational Study. Curr. Oncol. 2021, 28, 2789-2800. https://doi.org/10.3390/curroncol28040244
Lelond S, Ward J, Lambert PJ, Kim CA. Symptom Burden of Patients with Advanced Pancreas Cancer (APC): A Provincial Cancer Institute Observational Study. Current Oncology. 2021; 28(4):2789-2800. https://doi.org/10.3390/curroncol28040244
Chicago/Turabian StyleLelond, Stephanie, Julie Ward, Pascal J. Lambert, and Christina A. Kim. 2021. "Symptom Burden of Patients with Advanced Pancreas Cancer (APC): A Provincial Cancer Institute Observational Study" Current Oncology 28, no. 4: 2789-2800. https://doi.org/10.3390/curroncol28040244
APA StyleLelond, S., Ward, J., Lambert, P. J., & Kim, C. A. (2021). Symptom Burden of Patients with Advanced Pancreas Cancer (APC): A Provincial Cancer Institute Observational Study. Current Oncology, 28(4), 2789-2800. https://doi.org/10.3390/curroncol28040244





