Two Antibiotics, Ampicillin and Tetracycline, Exert Different Effects in HT-29 Colorectal Adenocarcinoma Cells in Terms of Cell Viability and Migration Capacity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Cell Viability Assay
2.3. Cells’ Morphology Assessment
2.4. Scratch Assay
2.5. Nuclear Staining
2.6. In Ovo Irritant Potential Assessment by the Means of Chorioallantoic Membrane (HET-CAM) Assay
3. Results
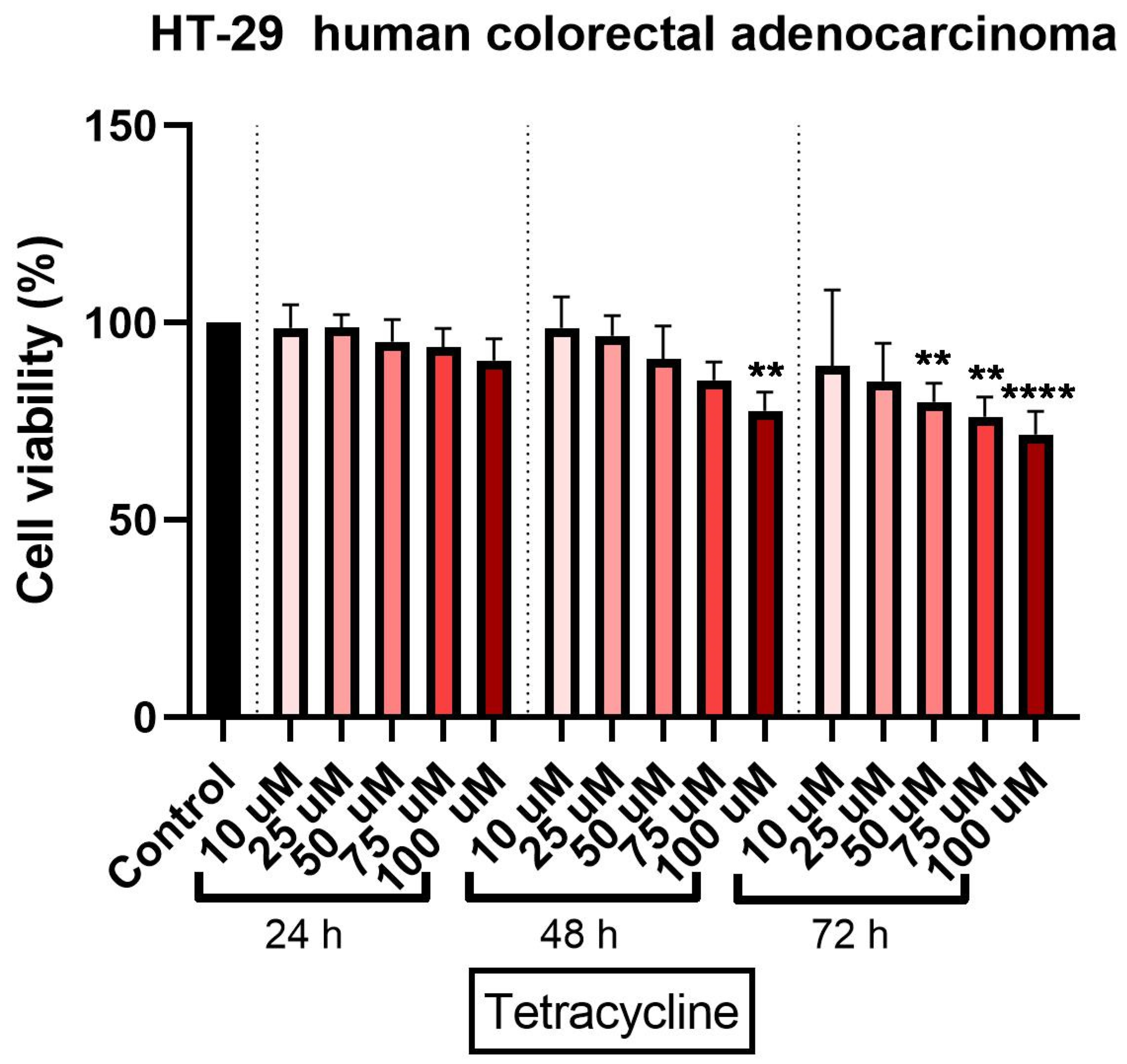
3.1. Tetracycline Affects Cell Viability in a Dose and Time-Dependent Manner
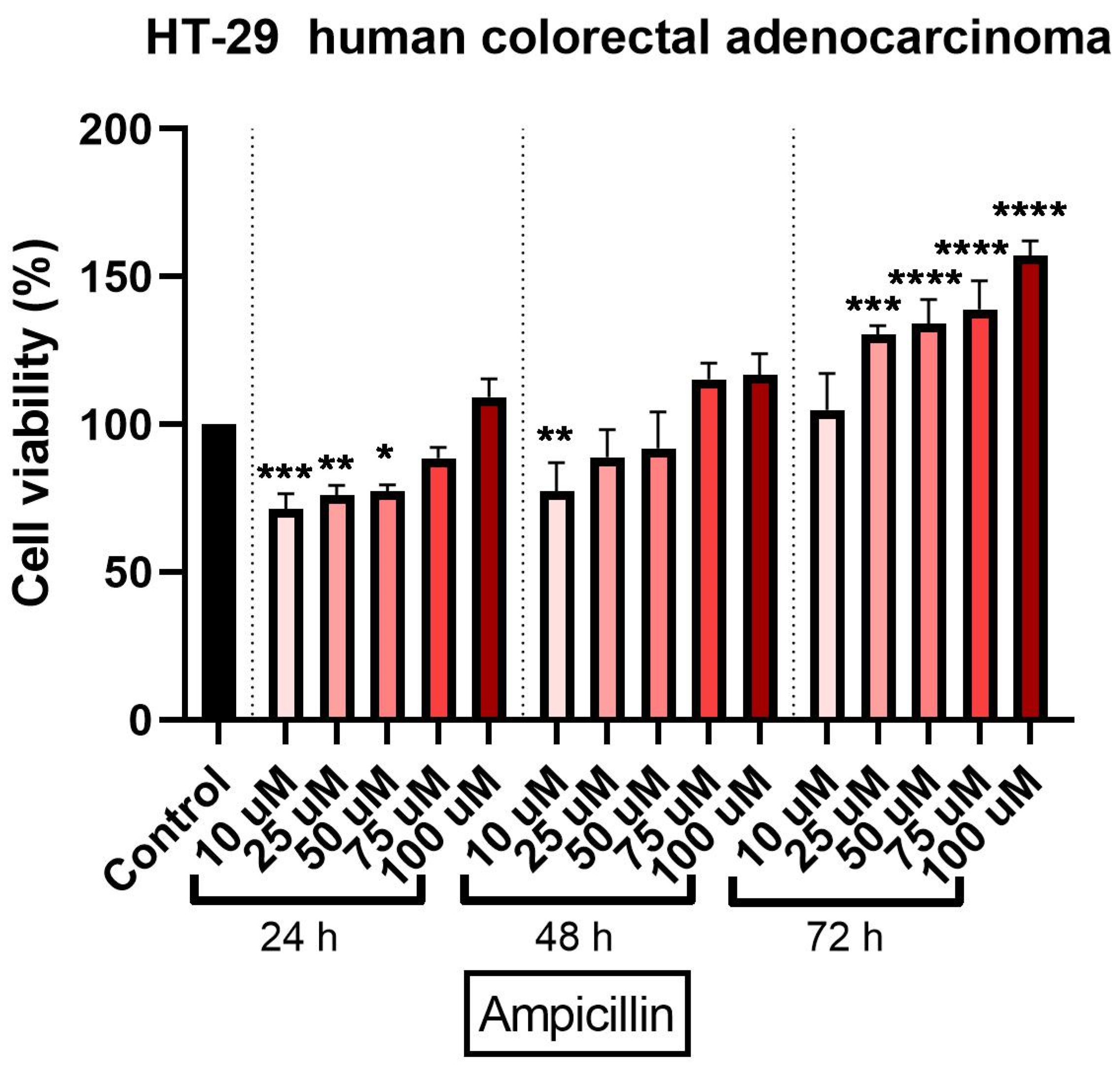
3.2. Ampicillin Interfered Dose- and Time-Dependent in HT-29 Cells’ Viability
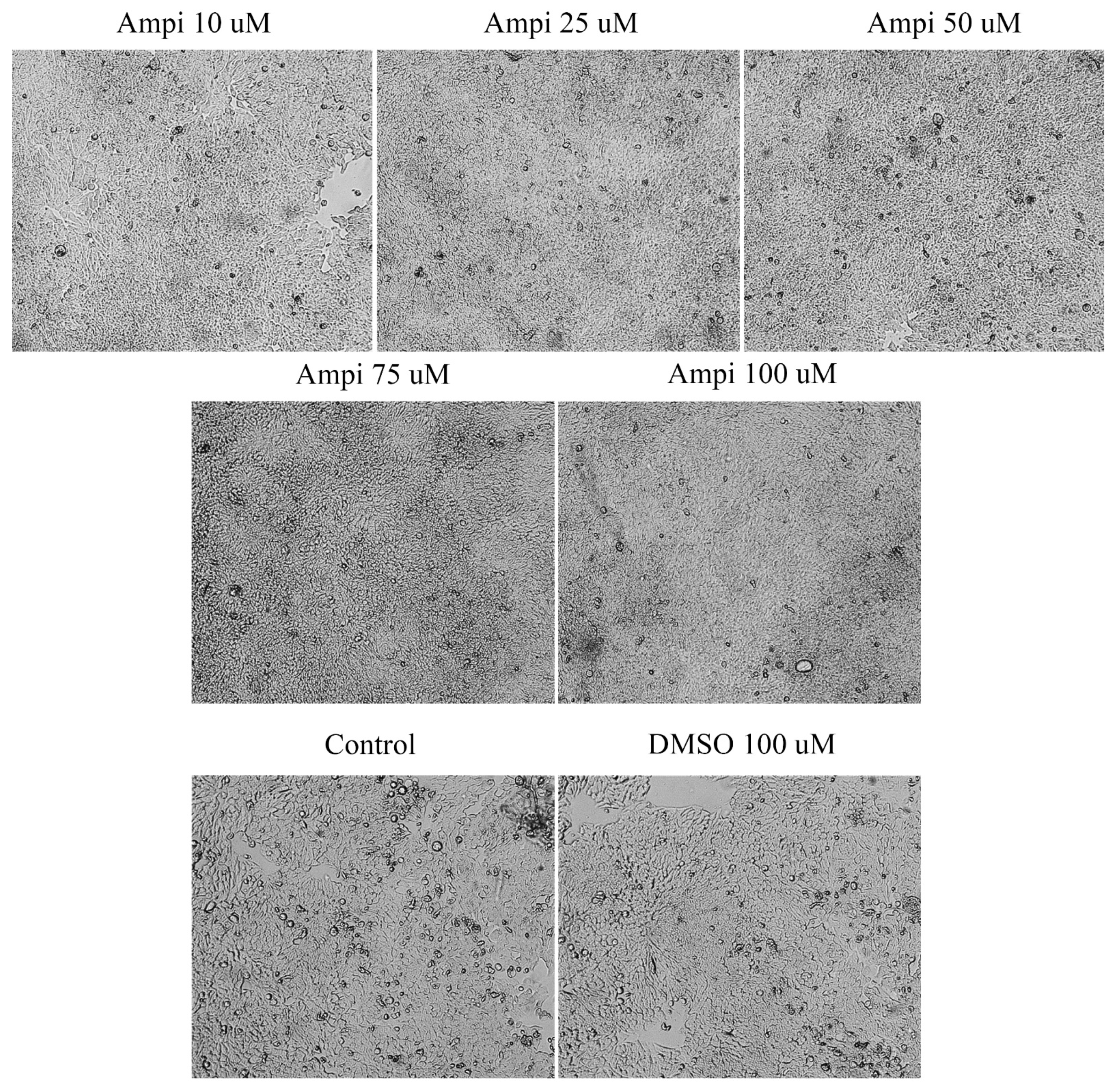
3.3. Tetracycline and Ampicillin Treatment Determined Morphological and Cell Shape Changes in HT-29 Cells
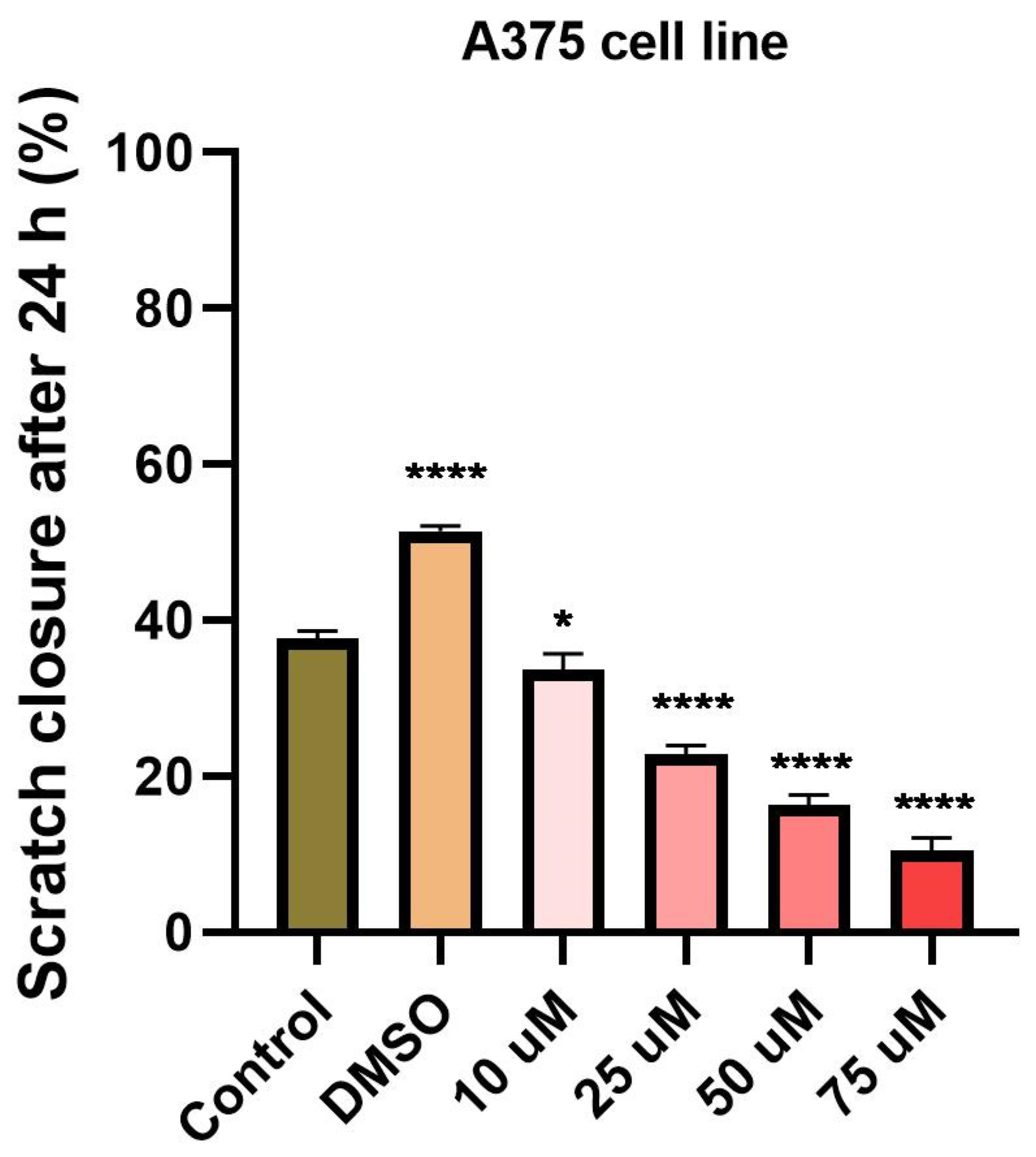
3.4. Tetracycline and Ampicillin Influenced Cell Migration
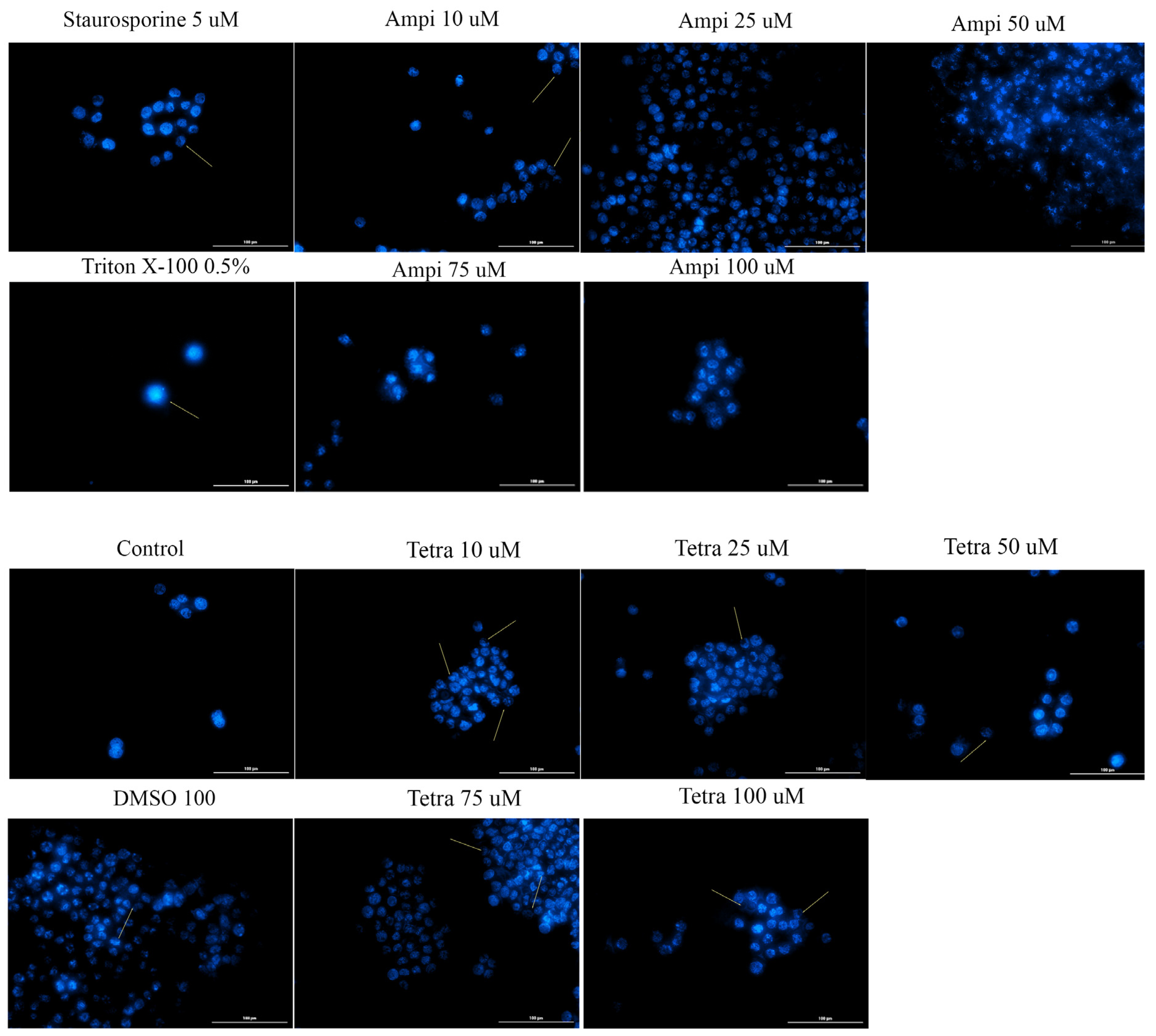
3.5. Tetracycline and Ampicillin Induced Apoptotic-Like Features in the Nucleus of HT-29 Cells
3.6. Tetracycline and Ampicillin Exerted a Slight Irritant Effect in Ovo
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Charmsaz, S.; Collins, D.M.; Perry, A.S.; Prencipe, M. Novel Strategies for Cancer Treatment: Highlights from the 55th IACR Annual Conference. Cancers 2019, 11, 1125. [Google Scholar] [CrossRef] [Green Version]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA. Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Simin, J.; Fornes, R.; Liu, Q.; Olsen, R.S.; Callens, S.; Engstrand, L.; Brusselaers, N. Antibiotic Use and Risk of Colorectal Cancer: A Systematic Review and Dose–Response Meta-Analysis. Br. J. Cancer 2020, 123, 1825–1832. [Google Scholar] [CrossRef] [PubMed]
- Arthur, J.C.; Perez-Chanona, E.; Mühlbauer, M.; Tomkovich, S.; Uronis, J.M.; Fan, T.-J.; Campbell, B.J.; Abujamel, T.; Dogan, B.; Rogers, A.B.; et al. Intestinal Inflammation Targets Cancer-Inducing Activity of the Microbiota. Science 2012, 338, 120–123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dik, V.K.; van Oijen, M.G.H.; Smeets, H.M.; Siersema, P.D. Frequent Use of Antibiotics Is Associated with Colorectal Cancer Risk: Results of a Nested Case-Control Study. Dig. Dis. Sci. 2016, 61, 255–264. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cao, Y.; Wu, K.; Mehta, R.; Drew, D.A.; Song, M.; Lochhead, P.; Nguyen, L.H.; Izard, J.; Fuchs, C.S.; Garrett, W.S.; et al. Long-Term Use of Antibiotics and Risk of Colorectal Adenoma. Gut 2018, 67, 672–678. [Google Scholar] [CrossRef]
- Saus, E.; Iraola-Guzmán, S.; Willis, J.R.; Brunet-Vega, A.; Gabaldón, T. Microbiome and Colorectal Cancer: Roles in Carcinogenesis and Clinical Potential. Mol. Aspects Med. 2019, 69, 93–106. [Google Scholar] [CrossRef] [PubMed]
- Pancu, D.F.; Scurtu, A.; Macasoi, I.G.; Marti, D.; Mioc, M.; Soica, C.; Coricovac, D.; Horhat, D.; Poenaru, M.; Dehelean, C. Antibiotics: Conventional Therapy and Natural Compounds with Antibacterial Activity—A Pharmaco-Toxicological Screening. Antibiotics 2021, 10, 401. [Google Scholar] [CrossRef]
- Gao, Y.; Shang, Q.; Li, W.; Guo, W.; Stojadinovic, A.; Mannion, C.; Man, Y.-G.; Chen, T. Antibiotics for Cancer Treatment: A Double-Edged Sword. J. Cancer 2020, 11, 5135–5149. [Google Scholar] [CrossRef]
- Martins Lopes, M.S.; Machado, L.M.; Ismael Amaral Silva, P.A.; Tome Uchiyama, A.A.; Yen, C.T.; Ricardo, E.D.; Mutao, T.S.; Pimenta, J.R.; Shimba, D.S.; Hanriot, R.M.; et al. Antibiotics, Cancer Risk and Oncologic Treatment Efficacy: A Practical Review of the Literature. Ecancermedicalscience 2020, 14, 1106. [Google Scholar] [CrossRef]
- Petrelli, F.; Ghidini, M.; Ghidini, A.; Perego, G.; Cabiddu, M.; Khakoo, S.; Oggionni, E.; Abeni, C.; Hahne, J.C.; Tomasello, G.; et al. Use of Antibiotics and Risk of Cancer: A Systematic Review and Meta-Analysis of Observational Studies. Cancers 2019, 11, 1174. [Google Scholar] [CrossRef] [Green Version]
- Rinninella, E.; Raoul, P.; Cintoni, M.; Franceschi, F.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. What Is the Healthy Gut Microbiota Composition? A Changing Ecosystem across Age, Environment, Diet, and Diseases. Microorganisms 2019, 7, 14. [Google Scholar] [CrossRef] [Green Version]
- Boursi, B.; Mamtani, R.; Haynes, K.; Yang, Y.-X. Recurrent Antibiotic Exposure May Promote Cancer Formation – Another Step in Understanding the Role of the Human Microbiota? Eur. J. Cancer 2015, 51, 2655–2664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seidlitz, E.; Saikali, Z.; Singh, G. Use of Tetracyclines for Bone Metastases. Bone Metastasis 2005, 293–303. [Google Scholar] [CrossRef]
- Sapadin, A.N.; Fleischmajer, R. Tetracyclines: Nonantibiotic properties and their clinical implications. J. Am. Acad. Dermatol. 2006, 54, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, H.; Inoue, H.; Mitsuke, Y.; Badran, A.; Ikegaya, S.; Ueda, T. Doxycycline Induces Apoptosis by Way of Caspase-3 Activation with Inhibition of Matrix Metalloproteinase in Human T-Lymphoblastic Leukemia CCRF-CEM Cells. J. Lab. Clin. Med. 2002, 140, 382–386. [Google Scholar] [CrossRef] [PubMed]
- Saikali, Z.; Singh, G. Doxycycline and Other Tetracyclines in the Treatment of Bone Metastasis. Anticancer. Drugs 2003, 14, 773–778. [Google Scholar] [CrossRef]
- Sharma, S.K.; Singh, L.; Singh, S. Comparative Study between Penicillin and Ampicillin. Sch. J. App. Med. Sci. 2013, 1, 291–294. [Google Scholar]
- Kaushik, D.; Mohan, M.; Borade, D.M.; Swami, O.C. Ampicillin: Rise Fall and Resurgence. J. Clin. Diagn. Res. 2014, 8, ME01–ME3. [Google Scholar] [CrossRef]
- Mohammed, T.J. Ampicillin Inhibition Effect on HCT116 Cell Line Ampicillin Inhibition Effect on HCT116 Cell Line. Al-Qadisiyah J. Pure Sci. 2017, 22, 185–190. [Google Scholar]
- Ferraz, R.; Costa-Rodrigues, J.; Fernandes, M.H.; Santos, M.M.; Marrucho, I.M.; Rebelo, L.P.N.; Prudêncio, C.; Noronha, J.P.; Petrovski, Ž.; Branco, L.C. Antitumor Activity of Ionic Liquids Based on Ampicillin. ChemMedChem 2015, 10, 1480–1483. [Google Scholar] [CrossRef] [Green Version]
- Bartlett, J.G. Antibiotic Use in Relation to the Risk of Breast Cancer. Infect. Dis. Clin. Pract. 2004, 12, 263. [Google Scholar] [CrossRef] [Green Version]
- Felice, F.; Zambito, Y.; Belardinelli, E.; Fabiano, A.; Santoni, T.; Di Stefano, R. Effect of Different Chitosan Derivatives on in Vitro Scratch Wound Assay: A Comparative Study. Int. J. Biol. Macromol. 2015, 76, 236–241. [Google Scholar] [CrossRef] [PubMed]
- Batista-duharte, A.; Murillo, G.J.; Betancourt, J.E.; Oliver, P.; Damiana, T. The Hen’s Egg Test on Chorioallantoic Membrane: An Alternative Assay for the Assessment of the Irritating Effect of Vaccine Adjuvants. Int. J. Toxicol. 2016, 35, 627–633. [Google Scholar] [CrossRef] [PubMed]
- Mármol, I.; Sánchez-de-Diego, C.; Pradilla Dieste, A.; Cerrada, E.; Rodriguez Yoldi, M.J. Colorectal Carcinoma: A General Overview and Future Perspectives in Colorectal Cancer. Int. J. Mol. Sci. 2017, 18, 197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Francino, M.P. Antibiotics and the Human Gut Microbiome: Dysbioses and Accumulation of Resistances. Front. Microbiol. 2016, 6, 1543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.; Haines, C.; Watson, A.J.M.; Hart, A.R.; Platt, M.J.; Pardoll, D.M.; Cosgrove, S.E.; Gebo, K.A.; Sears, C.L. Oral Antibiotic Use and Risk of Colorectal Cancer in the United Kingdom, 1989–2012: A Matched Case-Control Study. Gut 2019, 68, 1971–1978. [Google Scholar] [CrossRef]
- Song, M.; Nguyen, L.H.; Emilsson, L.; Chan, A.T.; Ludvigsson, J.F. Antibiotic Use Associated With Risk of Colorectal Polyps in a Nationwide Study. Clin. Gastroenterol. Hepatol. Off. Clin. Pract. J. Am. Gastroenterol. Assoc. 2020, 19, 1426–1435.e6. [Google Scholar] [CrossRef] [PubMed]
- Olejniczak, A.; Szaryńska, M.; Kmieć, Z. In vitro characterization of spheres derived from colorectal cancer cell lines. Int. J. Oncol. 2018, 52, 599–612. [Google Scholar] [CrossRef] [Green Version]
- Maghiari, A.L.; Coricovac, D.; Pinzaru, I.A.; Macașoi, I.G.; Marcovici, I.; Simu, S.; Navolan, D.; Dehelean, C. High Concentrations of Aspartame Induce Pro-Angiogenic Effects in Ovo and Cytotoxic Effects in HT-29 Human Colorectal Carcinoma Cells. Nutrients 2020, 12, 3600. [Google Scholar] [CrossRef]
- Ahmed, D.; Eide, P.W.; Eilertsen, I.A.; Danielsen, S.A.; Eknæs, M.; Hektoen, M.; Lind, G.E.; Lothe, R.A. Epigenetic and genetic features of 24 colon cancer cell lines. Oncogenesis 2013, 2, e71. [Google Scholar] [CrossRef] [PubMed]
- Miele, E.; Abballe, L.; Spinelli, G.P.; Besharat, Z.M.; Catanzaro, G.; Chiacchiarini, M.; Vacca, A.; Po, A.; Capalbo, C.; Ferretti, E. BRAF mutant colorectal cancer: ErbB2 expression levels as predictive factor for the response to combined BRAF/ErbB inhibitors. BMC Cancer 2020, 20, 129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fife, R.S.; Rougraff, B.T.; Proctor, C.; Sledge, G.W.J. Inhibition of Proliferation and Induction of Apoptosis by Doxycycline in Cultured Human Osteosarcoma Cells. J. Lab. Clin. Med. 1997, 130, 530–534. [Google Scholar] [CrossRef]
- Dezube, B.J.; Krown, S.E.; Lee, J.Y.; Bauer, K.S.; Aboulafia, D.M. Randomized Phase II Trial of Matrix Metalloproteinase Inhibitor COL-3 in AIDS-Related Kaposi’s Sarcoma: An AIDS Malignancy Consortium Study. J. Clin. Oncol. 2006, 24, 1389–1394. [Google Scholar] [CrossRef]
- Onoda, T.; Ono, T.; Dhar, D.K.; Yamanoi, A.; Nagasue, N. Tetracycline Analogues (Doxycycline and COL-3) Induce Caspase-Dependent and -Independent Apoptosis in Human Colon Cancer Cells. Int. J. Cancer 2006, 118, 1309–1315. [Google Scholar] [CrossRef] [PubMed]
- Sagar, J.; Sales, K.; Dijk, S.; Taanman, J.; Seifalian, A.; Winslet, M. Does doxycycline work in synergy with cisplatin and oxaliplatin in colorectal cancer? World J. Surg. Oncol. 2009, 7, 2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ray, S.D.; Farris, F.F.; Hartmann, A.C. Hormesis. In Encyclopedia of Toxicology, 3rd ed.; Academic Press: Bethesda, MD, USA, 2014; pp. 944–948. [Google Scholar] [CrossRef]
- Mathieu, A.; Fleurier, S.; Frénoy, A.; Dairou, J.; Bredeche, M.F.; Sanchez-Vizuete, P.; Song, X.; Matic, I. Discovery and Function of a General Core Hormetic Stress Response in E. coli Induced by Sublethal Concentrations of Antibiotics. Cell Rep. 2016, 17, 46–57. [Google Scholar] [CrossRef] [Green Version]
- Agathokleous, E.; Kitao, M.; Calabrese, E.J. Human and veterinary antibiotics induce hormesis in plants: Scientific and regulatory issues and an environmental perspective. Environ. Int. 2018, 120, 489–495. [Google Scholar] [CrossRef]
- Li, L.; Li, X.; Zhong, W.; Yang, M.; Xu, M.; Sun, Y.; Ma, J.; Liu, T.; Song, X.; Dong, W.; et al. Gut Microbiota from Colorectal Cancer Patients Enhances the Progression of Intestinal Adenoma in ApcMin/+ Mice. EBioMedicine 2019, 48, 301–315. [Google Scholar] [CrossRef] [Green Version]
- Meng, Q.; Xu, J.; Goldberg, I.D.; Rosen, E.M.; Greenwald, R.A.; Fan, S. Influence of chemically modified tetracyclines on proliferation, invasion and migration properties of MDA-MB-468 human breast cancer cells. Clin. Exp. Metastasis 2000, 18, 139–146. [Google Scholar] [CrossRef]
- Chang, H.F.; Cheng, H.T.; Chen, H.Y.; Yeung, W.K.; Cheng, J.Y. Doxycycline inhibits electric field-induced migration of non-small cell lung cancer (NSCLC) cells. Sci. Rep. 2019, 9, 8094. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fife, R.S.; Sledge, G.W.J.; Sissons, S.; Zerler, B. Effects of Tetracyclines on Angiogenesis in Vitro. Cancer Lett. 2000, 153, 75–78. [Google Scholar] [CrossRef]
- Wu, M.; Liu, J.; Li, F.; Huang, S.; He, J.; Xue, Y.; Fu, T.; Feng, S.; Li, Z. Antibiotic-Induced Dysbiosis of Gut Microbiota Impairs Corneal Development in Postnatal Mice by Affecting CCR2 Negative Macrophage Distribution. Mucosal Immunol. 2020, 13, 47–63. [Google Scholar] [CrossRef] [PubMed] [Green Version]


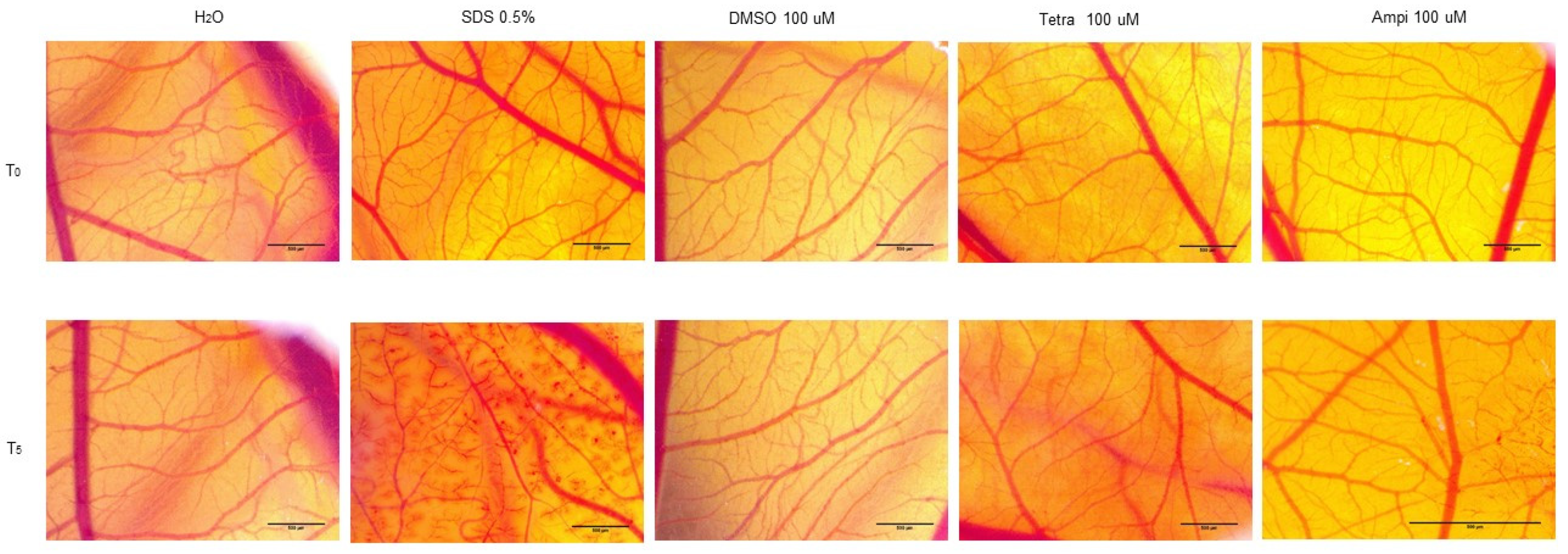
| SDS 0.5% | H2O | DMSO 100 µM | Tetra 100 µM | Ampi 100 µM | |
|---|---|---|---|---|---|
| IS | 19.24 | 0.07 | 4.57 | 7.36 | 9.16 |
| tH | 15 s | 300 | 300 | 300 | 202 |
| tL | 20 s | 300 | 300 | 71 | 137 |
| tC | 37 s | 300 | 150 | 235 | 178 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hut, E.-F.; Radulescu, M.; Pilut, N.; Macasoi, I.; Berceanu, D.; Coricovac, D.; Pinzaru, I.; Cretu, O.; Dehelean, C. Two Antibiotics, Ampicillin and Tetracycline, Exert Different Effects in HT-29 Colorectal Adenocarcinoma Cells in Terms of Cell Viability and Migration Capacity. Curr. Oncol. 2021, 28, 2466-2480. https://doi.org/10.3390/curroncol28040225
Hut E-F, Radulescu M, Pilut N, Macasoi I, Berceanu D, Coricovac D, Pinzaru I, Cretu O, Dehelean C. Two Antibiotics, Ampicillin and Tetracycline, Exert Different Effects in HT-29 Colorectal Adenocarcinoma Cells in Terms of Cell Viability and Migration Capacity. Current Oncology. 2021; 28(4):2466-2480. https://doi.org/10.3390/curroncol28040225
Chicago/Turabian StyleHut, Emil-Florin, Matilda Radulescu, Nicolae Pilut, Ioana Macasoi, Delia Berceanu, Dorina Coricovac, Iulia Pinzaru, Octavian Cretu, and Cristina Dehelean. 2021. "Two Antibiotics, Ampicillin and Tetracycline, Exert Different Effects in HT-29 Colorectal Adenocarcinoma Cells in Terms of Cell Viability and Migration Capacity" Current Oncology 28, no. 4: 2466-2480. https://doi.org/10.3390/curroncol28040225
APA StyleHut, E.-F., Radulescu, M., Pilut, N., Macasoi, I., Berceanu, D., Coricovac, D., Pinzaru, I., Cretu, O., & Dehelean, C. (2021). Two Antibiotics, Ampicillin and Tetracycline, Exert Different Effects in HT-29 Colorectal Adenocarcinoma Cells in Terms of Cell Viability and Migration Capacity. Current Oncology, 28(4), 2466-2480. https://doi.org/10.3390/curroncol28040225







