Factors Associated with “Survivor Identity” in Men with Breast Cancer
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants and Recruitment
2.2. Measures
2.3. Analyses
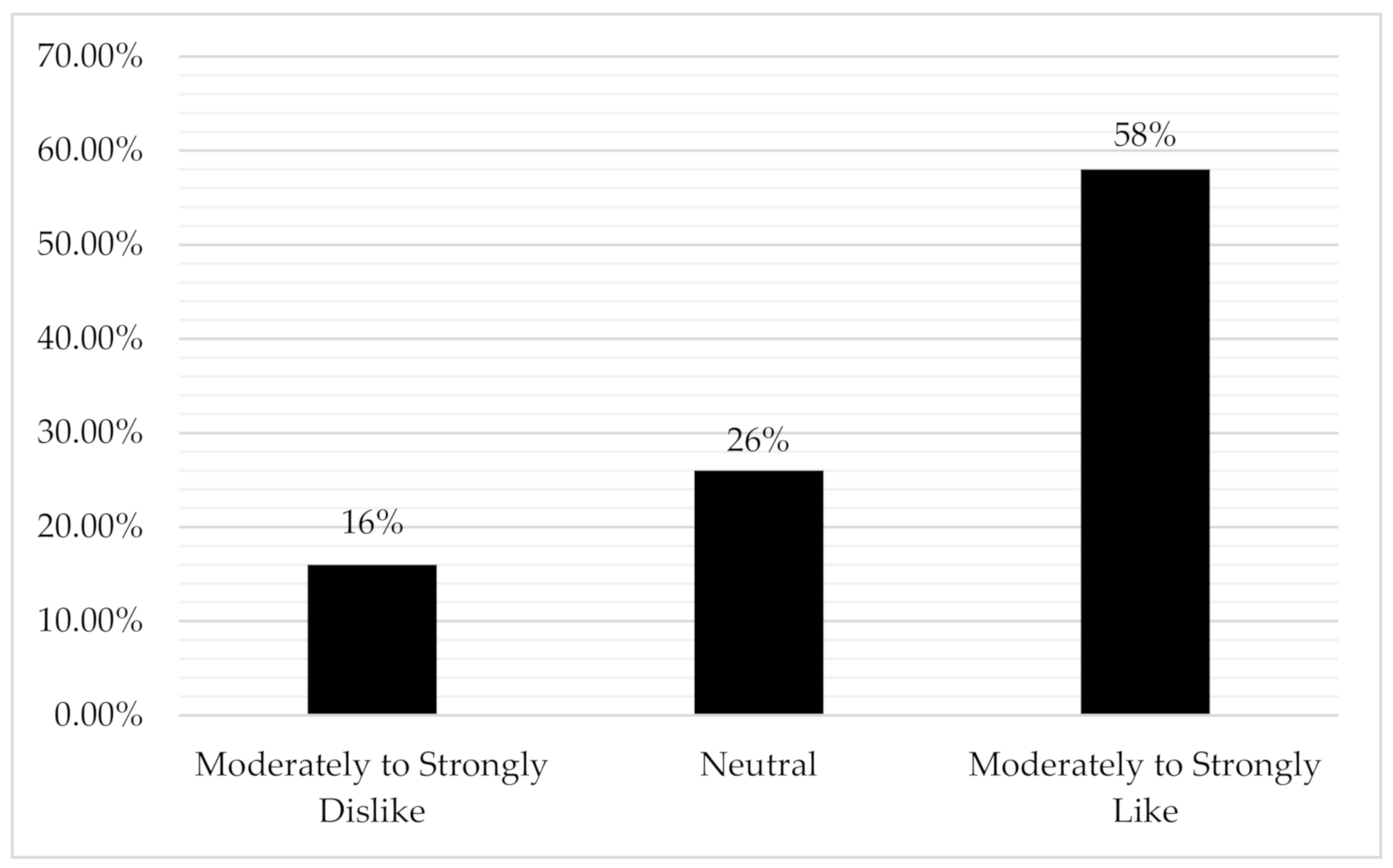
3. Results
3.1. How Males with Breast Cancer Feel about “Survivor” Identity
“A lot of female identity since puberty is associated with the breasts, from both a beauty aspect and a potentially nurturing, mothering role. Losing one or both breasts is potentially very destructive to a woman’s sense of self and her role in life. Men don’t share this level of identification, but the scar remaining from a male mastectomy can be disfiguring and a source of embarrassment at, for example, the beach, if one allows it to be”.
“Breast cancer is still cancer, whether viewed as traditionally a woman’s disease or not, so the diagnosis, treatment and follow up is exactly the same. In my personal situation, I did not experience any loss of identity that women may experience, only because my physical appearance did not change”.
3.2. Factors Associated with Endorsing “Survivor” Identity in Males with BC
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- National Cancer Institute. Available online: https://www.cancer.gov/publications/dictionaries/cancer-terms/def/survivor (accessed on 27 April 2020).
- Knaul, F.M.; Doubova, S.V.; Robledo, M.C.G.; Durstine, A.; Pages, G.S.; Casanova, F.; Arreola-Ornelas, H. Self-identity, lived experiences, and challenges of breast, cervical, and prostate cancer survivorship in Mexico: A qualitative study. BMC Cancer 2020, 20, 577. [Google Scholar] [CrossRef] [PubMed]
- Costa, D.S.; Mercieca-Bebber, R.; Tesson, S.; Seidler, Z.; Lopez, A.L. Patient, client, consumer, survivor or other alternatives? A scoping review of preferred terms for labelling individuals who access healthcare across settings. BMJ Open 2019, 9, e025166. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, K. The meaning of the survivor identity for women with breast cancer. Soc. Sci. Med. 2008, 67, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Berry, L.L.; Davis, S.W.; Godfrey Flynn, A.; Landercasper, J.; Deming, K.A. Is it time to reconsider the term “cancer survivor”? J. Psychosoc. Oncol. 2019, 37, 413–426. [Google Scholar] [CrossRef] [PubMed]
- Magana, A.O. Hope for Hispanic Patients in the Context of Hospice: The Impact of Narratives of Future Oriented Stories of Hope in the Experience of Anticipatory Grief for Hispanic Roman Catholic Hospice Patients in the Fort Worth Area; Brite Divinity School: Fort Worth, TX, USA, 2012. [Google Scholar]
- Rees, S. A qualitative exploration of the meaning of the term “survivor” to young women living with a history of breast cancer. Eur. J. Cancer Care 2018, 27, e12847. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.A.A.; Zago, M.M.F. Patient, cured, victim or survivor of urological cancer? A qualitative study. Rev. Lat. Am. Enferm. 2018, 26, e3089. [Google Scholar] [CrossRef] [PubMed]
- Fukui, N.; Golabi, P.; Otgonsuren, M.; de Avila, L.; Bush, H.; Younossi, Z.M. Hospice care in Medicare patients with primary liver cancer: The impact on resource utilisation and mortality. Aliment. Pharmacol. Ther. 2018, 47, 680–688. [Google Scholar] [CrossRef]
- Brown, J.M.H.; Havener, D.J.; Byrne, J.T. A Systematic Review—The Effect of Hospice and Palliative Care. Williams Honors College, Honors Research Projects. 2017. Available online: https://ideaexchange.uakron.edu/honors_research_projects/468 (accessed on 27 April 2020).
- Cheung, S.Y.; Delfabbro, P. Are you a cancer survivor? A review on cancer identity. J. Cancer Surviv. 2016, 10, 759–771. [Google Scholar] [CrossRef]
- Thong, M.S.; Wolschon, E.M.; Koch-Gallenkamp, L.; Waldmann, A.; Waldeyer-Sauerland, M.; Pritzkuleit, R.; Bertram, H.; Kajüter, H.; Eberle, A.; Holleczek, B.; et al. “Still a Cancer Patient”—Associations of Cancer Identity With Patient-Reported Outcomes and Health Care Use Among Cancer Survivors. JNCI Cancer Spectr. 2018, 2, pky031. [Google Scholar] [CrossRef]
- Kelly, K.M.; Shah, N.; Shedlosky-Shoemaker, R.; Porter, K.; Agnese, D. Living post treatment: Definitions of those with history and no history of cancer. J. Cancer Surviv. 2011, 5, 158–166. [Google Scholar] [CrossRef]
- Morris, B.A.; Lepore, S.J.; Wilson, B.; Lieberman, M.A.; Dunn, J.; Chambers, S.K. Adopting a survivor identity after cancer in a peer support context. J. Cancer Surviv. 2014, 8, 427–436. [Google Scholar] [CrossRef]
- Deber, R.B.; Kraetschmer, N.; Urowitz, S.; Sharpe, N. Patient, consumer, client, or customer: What do people want to be called? Health Expect. 2005, 8, 345–351. [Google Scholar] [CrossRef]
- Gucalp, A.; Traina, T.A.; Eisner, J.R.; Parker, J.S.; Selitsky, S.R.; Park, B.H.; Elias, A.D.; Baskin-Bey, E.S.; Cardoso, F. Male breast cancer: A disease distinct from female breast cancer. Breast Cancer Res. Treat. 2019, 173, 37–48. [Google Scholar] [CrossRef]
- Muldbücker, P.; Steinmann, D.; Christiansen, H.; de Zwaan, M.; Zimmermann, T. Are women more afraid than men? Fear of recurrence in couples with cancer—Predictors and sex-role-specific differences. J. Psychosoc. Oncol. 2020, 39, 89–104. [Google Scholar] [CrossRef]
- Esserman, L.; Shieh, Y.; Thompson, I. Rethinking screening for breast cancer and prostate cancer. JAMA 2009, 302, 1685–1692. [Google Scholar] [CrossRef]
- Howlader, N.; Noone, A.M.; Krapcho, M.; Miller, D.; Brest, A.; Yu, M.; Ruhl, J.; Tatalovich, Z.; Mariotto, A.; Lewis, D.R.; et al. Table 1.11: Median Age of Cancer Patients at Diagnosis, 2013–2017, by Primary Cancer Site, Race and Sex. In Cancer Statistics Review, 1975–2017. Available online: https://seer.cancer.gov/csr/1975_2017/ (accessed on 27 April 2020).
- Rawla, P. Epidemiology of Prostate Cancer. World J. Oncol. 2019, 10, 63–89. [Google Scholar] [CrossRef]
- Cancer.Net—Breast Cancer: Statistics. Available online: https://www.cancer.net/cancer-types/breast-cancer/statistics#:~:text=The%205%2Dyear%20survival%20rate (accessed on 15 August 2020).
- Baer, L.; Formenti, S.C. Breast cancer clinical and translational research: Analogies and implications for prostate cancer. Rev. Urol. 2007, 9 (Suppl. 2), S28–S39. [Google Scholar]
- Catton, C.; Milosevic, M.; Warde, P.; Bayley, A.; Crook, J.; Bristow, R.; Gospodarowicz, M. Recurrent prostate cancer following external beam radiotherapy: Follow-up strategies and management. Urol. Clin. N. Am. 2003, 30, 751–763. [Google Scholar] [CrossRef]
- Dong, Y.; Peng, C.Y. Principled missing data methods for researchers. Springerplus 2013, 2, 222. [Google Scholar] [CrossRef]
- Bartlett, J.W.; Harel, O.; Carpenter, J.R. Asymptotically Unbiased Estimation of Exposure Odds Ratios in Complete Records Logistic Regression. Am. J. Epidemiol. 2015, 182, 730–736. [Google Scholar] [CrossRef]
- American Cancer Society. Cancer Facts & Figures. Available online: https://www.cancer.org/cancer/pancreatic-cancer/detection-diagnosis-staging/survival-rates.html (accessed on 25 January 2021).
- Hill, R.E.; Wakefield, C.E.; Cohn, R.J.; Fardell, J.E.; Brierley, M.E.E.; Kothe, E.; Jacobsen, P.B.; Hetherington, K.; Mercieca-Bebber, R. Survivorship Care Plans in Cancer: A Meta-Analysis and Systematic Review of Care Plan Outcomes. Oncologist 2020, 25, e351–e372. [Google Scholar] [CrossRef]
- Griffith, D.M.; Gilbert, K.; Bruce, M.A.; Thorpe, R.J. Masculinity in Men’s Health: Barrier or Portal to Healthcare. In Men’s Health in Primary Care; Heidelbaugh, J.J., Ed.; Humana Press: Cham, Switzerland; Totowa, NJ, USA, 2016; ISBN 978-3-319-26089-1. [Google Scholar]
- Barlow, M.R.; Hetzel-Riggin, M.D. Predicting posttraumatic growth in survivors of interpersonal trauma: Gender role adherence is more important Than gender. Psychol. Men Masc. 2018, 19, 446–456. [Google Scholar] [CrossRef]
- Darkeh, M.H.S.E.; Azavedo, E. Male Breast Cancer Clinical Features, Risk Factors, and Current Diagnostic and Therapeutic Approaches. Int. J. Clin. Med. 2014, 5, 1068–1086. [Google Scholar] [CrossRef]
- Losurdo, A.; Rota, S.; Gullo, G.; Masci, G.; Torrisi, R.; Bottai, G.; Zuradelli, M.; Gatzemeier, W.; Santoro, A. Controversies in clinicopathological characteristics and treatment strategies of male breast cancer: A review of the literature. Crit. Rev. Oncol. Hematol. 2017, 113, 283–291. [Google Scholar] [CrossRef]
| Demographics | n | % |
|---|---|---|
| Age (range 34–84, mean = 63, SD = 10) | ||
| Under 50 | 7 | 10 |
| 50–59 | 16 | 22.9 |
| 60–69 | 28 | 40 |
| 70–79 | 17 | 24.3 |
| 80+ | 2 | 2.9 |
| Recruited from | ||
| The Male Breast Cancer Coalition | 42 | 60 |
| Breast Cancer Network Australia | 10 | 14.3 |
| Health Care Professional | 1 | 1.4 |
| Social media (Twitter, Facebook) | 6 | 8.6 |
| Friend | 4 | 5.7 |
| Other | 7 | 10 |
| Country of Residence | ||
| USA | 32 | 45 |
| Canada | 2 | 2.9 |
| Australia | 15 | 21.4 |
| Germany | 5 | 7.1 |
| Austria | 1 | 1.4 |
| Missing | 15 | 21.4 |
| Relationship Status | ||
| Single/not in a relationship | 6 | 8.6 |
| In a committed relationship/married | 64 | 91.4 |
| Number of Children | ||
| None | 11 | 15.7 |
| One | 11 | 15.7 |
| Two | 34 | 48.6 |
| Three or greater | 14 | 20 |
| Race/Ethnicity | ||
| White | 66 | 94.3 |
| Black | 1 | 1.4 |
| Latin American | 2 | 2.9 |
| Other | 1 | 1.4 |
| Education | ||
| High school degree or equivalent/Some college | 30 | 42.9 |
| Bachelor’s degree or Associate degree | 26 | 37.1 |
| Graduate or Professional degree | 14 | 20 |
| Currently Employed | ||
| Yes | 28 | 40 |
| No | 41 | 58.6 |
| Missing | 1 | 1.4 |
| Annual Income | ||
| Under $49,000 USD | 25 | 35.7 |
| $50,000 to 99,999 USD | 29 | 41.4 |
| $100,000 and greater USD | 14 | 20.0 |
| Missing | 2 | 2.9 |
| Clinical Characteristics | n | % |
|---|---|---|
| Self-Reported Health Status | ||
| Excellent | 7 | 10 |
| Very Good | 28 | 40 |
| Good | 27 | 38.6 |
| Fair | 6 | 8.6 |
| Poor | 2 | 2.9 |
| Diagnosis Date | ||
| 2004 or earlier | 5 | 7.2 |
| 2005–2009 | 11 | 15.7 |
| 2010–2014 | 26 | 37.1 |
| 2015–2020 | 28 | 40 |
| Stage a | ||
| 0 | 3 | 4.3 |
| 1 | 23 | 32.9 |
| 2 | 27 | 38.6 |
| 3 | 12 | 17.1 |
| 4 | 2 | 2.9 |
| Missing | 3 | 4.3 |
| Family History of Cancer | ||
| Yes | 50 | 71.4 |
| No | 20 | 28.6 |
| Currently Receiving Treatment | ||
| Yes | 41 | 58.6 |
| No | 28 | 40 |
| Missing | 1 | 1.4 |
| Treatment History | ||
| Surgery | ||
| Past | 60 | 85.7 |
| None | 10 | 14.3 |
| Chemotherapy | ||
| Current | 6 | 8.6 |
| Past | 39 | 55.7 |
| None | 25 | 35.7 |
| Radiation | ||
| Current | 3 | 4.3 |
| Past | 32 | 45.7 |
| None | 35 | 50 |
| Hormonal | ||
| Current | 35 | 50 |
| Past | 19 | 27.1 |
| None | 16 | 22.9 |
| Immunotherapy | ||
| Current | 2 | 2.9 |
| Past | 2 | 2.9 |
| None | 66 | 94.3 |
| Co-morbid Illnesses | ||
| 0–1 | 35 | 50 |
| 2–3 | 27 | 38.6 |
| 3+ | 7 | 10 |
| Missing | 1 | 1.4 |
| Univariate Logistic Regression Analysis | ||
|---|---|---|
| Parameter | Odds Ratio (95% CI) | p |
| Age | 1.045 (0.980, 1.11) | 0.177 |
| Stage | ||
| Early (0–2) | reference | |
| Late (3–4) | 1.35 (0.246, 7.42) | 0.730 |
| Currently on Treatment | ||
| Yes | reference | |
| No | 2.81 (0.647, 12.18) | 0.168 |
| Months since Diagnosis | 1.02 (1.00, 1.04) | 0.050 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dalton, K.L.; Garland, S.N.; Miller, P.; Miller, B.; Ambrose, C.; Wassersug, R.J. Factors Associated with “Survivor Identity” in Men with Breast Cancer. Curr. Oncol. 2021, 28, 1696-1705. https://doi.org/10.3390/curroncol28030158
Dalton KL, Garland SN, Miller P, Miller B, Ambrose C, Wassersug RJ. Factors Associated with “Survivor Identity” in Men with Breast Cancer. Current Oncology. 2021; 28(3):1696-1705. https://doi.org/10.3390/curroncol28030158
Chicago/Turabian StyleDalton, Kathryn L., Sheila N. Garland, Peggy Miller, Bret Miller, Cheri Ambrose, and Richard J. Wassersug. 2021. "Factors Associated with “Survivor Identity” in Men with Breast Cancer" Current Oncology 28, no. 3: 1696-1705. https://doi.org/10.3390/curroncol28030158
APA StyleDalton, K. L., Garland, S. N., Miller, P., Miller, B., Ambrose, C., & Wassersug, R. J. (2021). Factors Associated with “Survivor Identity” in Men with Breast Cancer. Current Oncology, 28(3), 1696-1705. https://doi.org/10.3390/curroncol28030158





