The Epidemiology of Myeloproliferative Neoplasms in New Zealand between 2010 and 2017: Insights from the New Zealand Cancer Registry
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Cohort
2.2. The New Zealand Cancer Registry
2.3. Statistical Analysis
3. Results
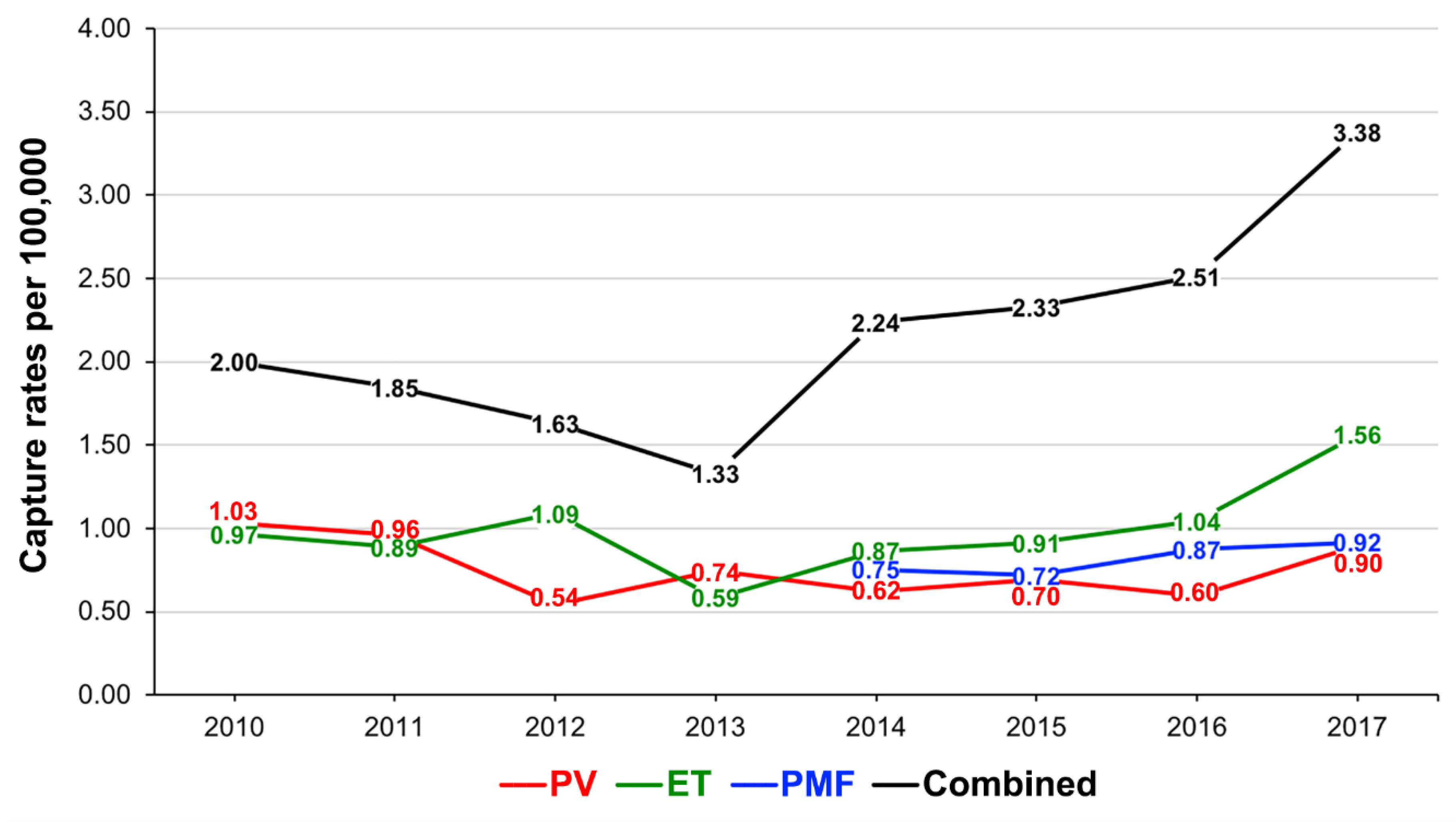
3.1. Capture Rates of PV, ET and PMF
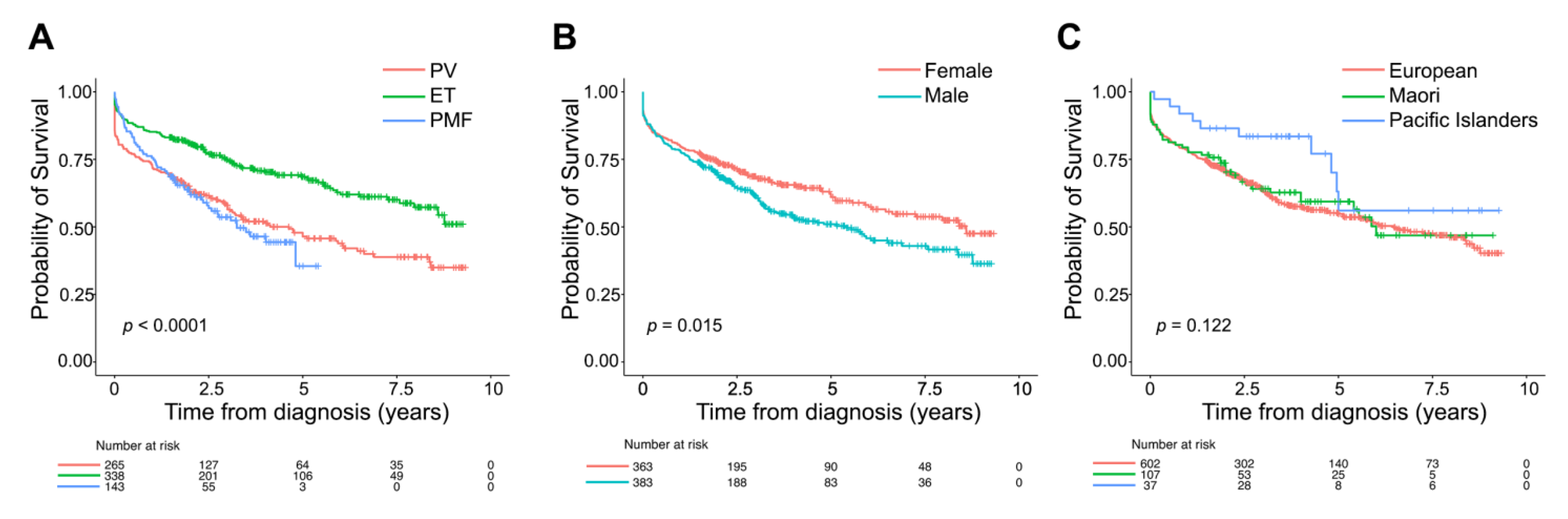
3.2. Overall Characteristics of Patients with MPN
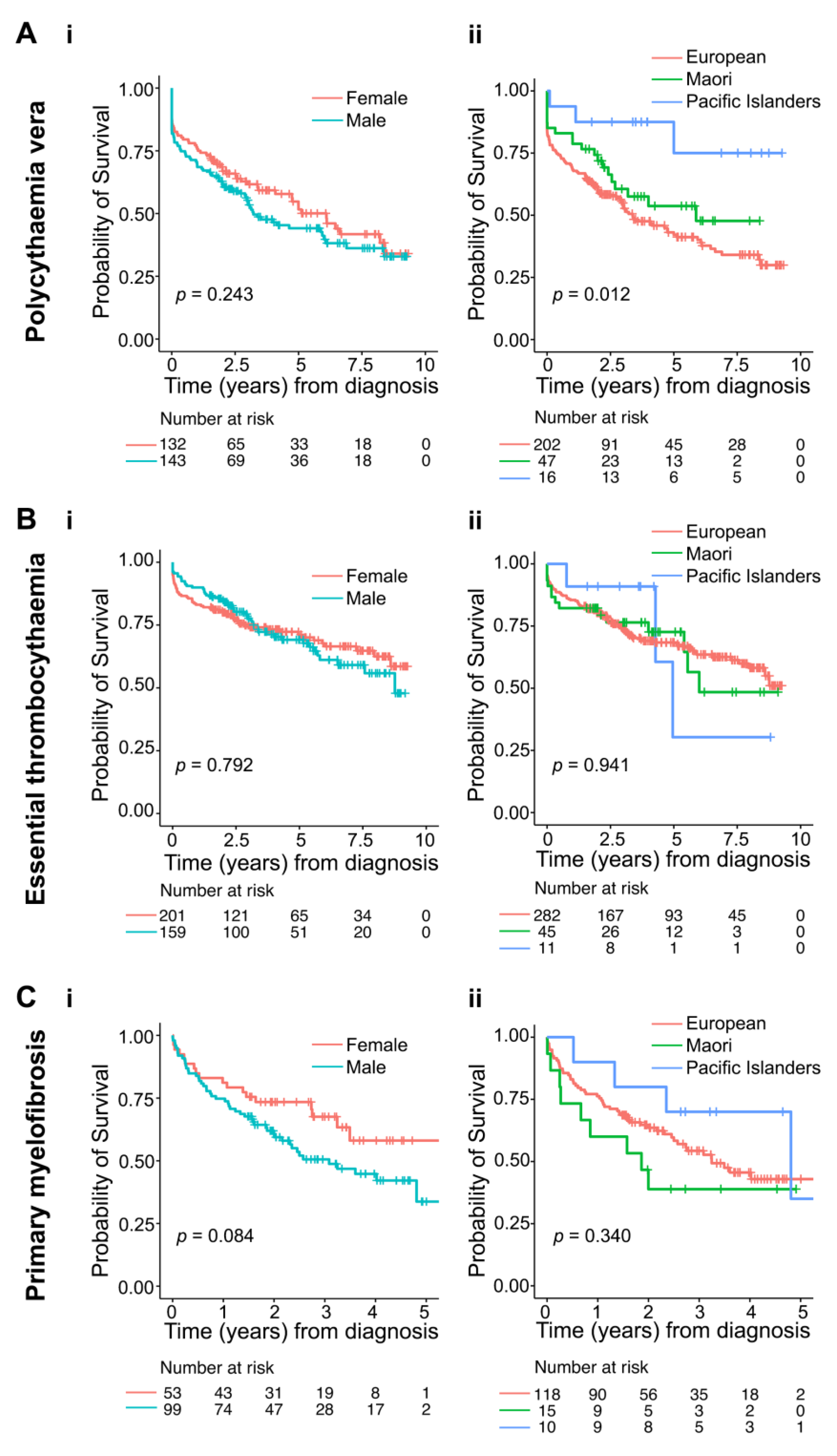
3.3. Epidemiology of Polycythaemia Vera
3.4. Epidemiology of Essential Thrombocythaemia
3.5. Epidemiology of Primary Myelofibrosis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tefferi, A.; Barbui, T. Polycythemia vera and essential thrombocythemia: 2021 update on diagnosis, risk-stratification and management. Am. J. Hematol. 2020, 95, 1599–1613. [Google Scholar] [CrossRef]
- Tefferi, A. Primary myelofibrosis: 2017 update on diagnosis, risk-stratification, and management. Am. J. Hematol. 2016, 91, 1262–1271. [Google Scholar] [CrossRef]
- Harrison, C.N.; Koschmieder, S.; Foltz, L.; Guglielmelli, P.; Flindt, T.; Koehler, M.; Mathias, J.; Komatsu, N.; Boothroyd, R.N.; Spierer, A.; et al. The impact of myeloproliferative neoplasms (MPNs) on patient quality of life and productivity: Results from the international MPN Landmark survey. Ann. Hematol. 2017, 96, 1653–1665. [Google Scholar] [CrossRef]
- Titmarsh, G.J.; Duncombe, A.S.; McMullin, M.F.; O’Rorke, M.; Mesa, R.; De Vocht, F.; Horan, S.; Fritschi, L.; Clarke, M.; Anderson, L.A. How common are myeloproliferative neoplasms? A systematic review and meta-analysis. Am. J. Hematol. 2014, 89, 581–587. [Google Scholar] [CrossRef] [PubMed]
- Shallis, R.M.; Wang, R.; Davidoff, A.; Ma, X.; Podoltsev, N.A.; Zeidan, A.M. Epidemiology of the classical myeloproliferative neoplasms: The four corners of an expansive and complex map. Blood Rev. 2020, 42, 100706. [Google Scholar] [CrossRef] [PubMed]
- Baade, P.D.; Ross, D.M.; Anderson, L.A.; Forsyth, C.; Fritschi, L. Changing incidence of myeloproliferative neoplasms in Australia, 2003–2014. Am. J. Hematol. 2019, 94, E107–E109. [Google Scholar] [CrossRef] [PubMed]
- Moulard, O.; Mehta, J.; Fryzek, J.; Olivares, R.; Iqbal, U.; Mesa, R.A. Epidemiology of myelofibrosis, essential thrombocythemia, and polycythemia vera in the European Union. Eur. J. Haematol. 2014, 92, 289–297. [Google Scholar] [CrossRef]
- Srour, S.A.; Devesa, S.S.; Morton, L.M.; Check, D.P.; Curtis, R.E.; Linet, M.S.; Dores, G.M. Incidence and patient survival of myeloproliferative neoplasms and myelodysplastic/myeloproliferative neoplasms in the United States, 2001–2012. Br. J. Haematol. 2016, 174, 382–396. [Google Scholar] [CrossRef] [PubMed]
- Roaldsnes, C.; Holst, R.; Frederiksen, H.; Ghanima, W. Myeloproliferative neoplasms: Trends in incidence, prevalence and survival in Norway. Eur. J. Haematol. 2017, 98, 85–93. [Google Scholar] [CrossRef]
- Heppner, J.; Nguyen, L.T.; Guo, M.; Naugler, C.; Rashid-Kolvear, F. Incidence of myeloproliferative neoplasms in Calgary, Alberta, Canada. BMC Res. Notes 2019, 12, 286. [Google Scholar] [CrossRef]
- Byun, J.M.; Kim, Y.J.; Youk, T.; Yang, J.J.; Yoo, J.; Park, T.S. Real world epidemiology of myeloproliferative neoplasms: A population based study in Korea 2004–2013. Ann. Hematol. 2017, 96, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Gale, R.P.; Zhang, Y.; Qin, T.; Chen, H.; Zhang, P.; Zhang, T.; Liu, L.; Qu, S.; Xiao, Z. Unique features of primary myelofibrosis in Chinese. Blood 2012, 119, 2469–2473. [Google Scholar] [CrossRef]
- Bai, J.; Ai, L.; Zhang, L.; Yang, F.C.; Zhou, Y.; Xue, Y. Incidence and risk factors for myelofibrotic transformation among 272 Chinese patients with JAK2-mutated polycythemia vera. Am. J. Hematol. 2015, 90, 1116–1121. [Google Scholar] [CrossRef] [PubMed]
- Fu, R.; Xuan, M.; Lv, C.; Zhang, L.; Li, H.; Zhang, X.; Zhang, D.; Sun, T.; Xue, F.; Liu, X.; et al. External validation and clinical evaluation of the International Prognostic Score of Thrombosis for Essential Thrombocythemia (IPSET-thrombosis) in a large cohort of Chinese patients. Eur. J. Haematol. 2014, 92, 502–509. [Google Scholar] [CrossRef] [PubMed]
- Price, G.L.; Davis, K.L.; Karve, S.; Pohl, G.; Walgren, R.A. Survival patterns in United States (US) medicare enrollees with non-CML myeloproliferative neoplasms (MPN). PLoS ONE 2014, 9, e90299. [Google Scholar] [CrossRef]
- Khan, I.; Shergill, A.; Saraf, S.L.; Chen, Y.F.; Patel, P.R.; Quigley, J.G.; Peace, D.; Gordeuk, V.R.; Hoffman, R.; Rondelli, D. Outcome Disparities in Caucasian and Non-Caucasian Patients With Myeloproliferative Neoplasms. Clin. Lymphoma Myeloma Leuk. 2016, 16, 350–357. [Google Scholar] [CrossRef]
- Hanna, M.Z.; Kalev-Zylinska, M.L.; Jackson, S.R.; Royle, G.; Blacklock, H.A. Distinctive features of polycythaemia vera in New Zealand Polynesians. N. Z. Med. J. 2018, 131, 38–45. [Google Scholar]
- Kleinbaum, D.G.; Klein, M. Survival Analysis A Self-Learning Text, 3rd ed.; Springer: New York, NY, USA, 2012; pp. 161–287. [Google Scholar]
- Shallis, R.M.; Zeidan, A.M.; Wang, R.; Podoltsev, N.A. Epidemiology of the Philadelphia Chromosome-Negative Classical Myeloproliferative Neoplasms. Hematol. Oncol. Clin. N. Am. 2021, 35, 177–189. [Google Scholar] [CrossRef]
- Arber, D.A.; Orazi, A.; Hasserjian, R.; Thiele, J.; Borowitz, M.J.; Le Beau, M.M.; Bloomfield, C.D.; Cazzola, M.; Vardiman, J.W. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood 2016, 127, 2391–2405. [Google Scholar] [CrossRef] [PubMed]
- Tefferi, A.; Rumi, E.; Finazzi, G.; Gisslinger, H.; Vannucchi, A.M.; Rodeghiero, F.; Randi, M.L.; Vaidya, R.; Cazzola, M.; Rambaldi, A.; et al. Survival and prognosis among 1545 patients with contemporary polycythemia vera: An international study. Leukemia 2013, 27, 1874–1881. [Google Scholar] [CrossRef] [PubMed]
- Szuber, N.; Mudireddy, M.; Nicolosi, M.; Penna, D.; Vallapureddy, R.R.; Lasho, T.L.; Finke, C.; Begna, K.H.; Elliott, M.A.; Hook, C.C.; et al. 3023 Mayo Clinic Patients With Myeloproliferative Neoplasms: Risk-Stratified Comparison of Survival and Outcomes Data Among Disease Subgroups. Mayo Clin. Proc. 2019, 94, 599–610. [Google Scholar] [CrossRef]
- Passamonti, F.; Rumi, E.; Pungolino, E.; Malabarba, L.; Bertazzoni, P.; Valentini, M.; Orlandi, E.; Arcaini, L.; Brusamolino, E.; Pascutto, C.; et al. Life expectancy and prognostic factors for survival in patients with polycythemia vera and essential thrombocythemia. Am. J. Med. 2004, 117, 755–761. [Google Scholar] [CrossRef]
- Tefferi, A.; Betti, S.; Barraco, D.; Mudireddy, M.; Shah, S.; Hanson, C.A.; Ketterling, R.P.; Pardanani, A.; Gangat, N.; Coltro, G.; et al. Gender and survival in essential thrombocythemia: A two-center study of 1494 patients. Am. J. Hematol. 2017, 92, 1193–1197. [Google Scholar] [CrossRef]
- Karantanos, T.; Chaturvedi, S.; Braunstein, E.M.; Spivak, J.; Resar, L.; Karanika, S.; Williams, D.M.; Rogers, O.; Gocke, C.D.; Moliterno, A.R. Sex determines the presentation and outcomes in MPN and is related to sex-specific differences in the mutational burden. Blood Adv. 2020, 4, 2567–2576. [Google Scholar] [CrossRef]
- Gangat, N.; Caramazza, D.; Vaidya, R.; George, G.; Begna, K.; Schwager, S.; Van Dyke, D.; Hanson, C.; Wu, W.; Pardanani, A.; et al. DIPSS plus: A refined Dynamic International Prognostic Scoring System for primary myelofibrosis that incorporates prognostic information from karyotype, platelet count, and transfusion status. J. Clin. Oncol. 2011, 29, 392–397. [Google Scholar] [CrossRef]
- Cervantes, F.; Dupriez, B.; Pereira, A.; Passamonti, F.; Reilly, J.T.; Morra, E.; Vannucchi, A.M.; Mesa, R.A.; Demory, J.L.; Barosi, G.; et al. New prognostic scoring system for primary myelofibrosis based on a study of the International Working Group for Myelofibrosis Research and Treatment. Blood 2009, 113, 2895–2901. [Google Scholar] [CrossRef] [PubMed]
- Chan, W.C.; Wright, C.; Riddell, T.; Wells, S.; Kerr, A.J.; Gala, G.; Jackson, R. Ethnic and socioeconomic disparities in the prevalence of cardiovascular disease in New Zealand. N. Z. Med. J. 2008, 121, 11–20. [Google Scholar] [PubMed]
- Haynes, R.; Pearce, J.; Barnett, R. Cancer survival in New Zealand: Ethnic, social and geographical inequalities. Soc. Sci. Med. 2008, 67, 928–937. [Google Scholar] [CrossRef] [PubMed]
- Varghese, C.; Liu, V.Y.; Immanuel, T.; Chien, N.; Green, T.N.; Chan, G.; Theakston, E.; Kalev-Zylinska, M.L. Ethnic differences in acute promyelocytic leukaemia between New Zealand Polynesian and European patients. Hematology 2021, 26, 215–224, in press. [Google Scholar] [CrossRef] [PubMed]
- Meredith, I.; Sarfati, D.; Ikeda, T.; Blakely, T. Cancer in Pacific people in New Zealand. Cancer Causes Control 2012, 23, 1173–1184. [Google Scholar] [CrossRef]
- Dachs, G.U.; Currie, M.J.; McKenzie, F.; Jeffreys, M.; Cox, B.; Foliaki, S.; Le Marchand, L.; Robinson, B.A. Cancer disparities in indigenous Polynesian populations: Maori, Native Hawaiians, and Pacific people. Lancet Oncol. 2008, 9, 473–484. [Google Scholar] [CrossRef]
- Duncan, E.; Schofield, G.; Duncan, S.; Kolt, G.; Rush, E. Ethnicity and body fatness in New Zealanders. N. Z. Med. J. 2004, 117, U913. [Google Scholar]
- Lindholm Sorensen, A.; Hasselbalch, H.C. Smoking and philadelphia-negative chronic myeloproliferative neoplasms. Eur. J. Haematol. 2016, 97, 63–69. [Google Scholar] [CrossRef]
- Leal, A.D.; Thompson, C.A.; Wang, A.H.; Vierkant, R.A.; Habermann, T.M.; Ross, J.A.; Mesa, R.A.; Virnig, B.A.; Cerhan, J.R. Anthropometric, medical history and lifestyle risk factors for myeloproliferative neoplasms in the Iowa Women’s Health Study cohort. Int. J. Cancer 2014, 134, 1741–1750. [Google Scholar] [CrossRef]
- Leiba, A.; Duek, A.; Afek, A.; Derazne, E.; Leiba, M. Obesity and related risk of myeloproliferative neoplasms among israeli adolescents. Obesity (Silver Spring) 2017, 25, 1187–1190. [Google Scholar] [CrossRef] [PubMed]
- Duncombe, A.S.; Anderson, L.A.; James, G.; de Vocht, F.; Fritschi, L.; Mesa, R.; Clarke, M.; McMullin, M.F. Modifiable Lifestyle and Medical Risk Factors Associated With Myeloproliferative Neoplasms. Hemasphere 2020, 4, e327. [Google Scholar] [CrossRef] [PubMed]
- Podoltsev, N.A.; Wang, X.; Wang, R.; Hofmann, J.N.; Liao, L.M.; Zeidan, A.M.; Mesa, R.; Ma, X. Lifestyle factors and risk of myeloproliferative neoplasms in the NIH-AARP diet and health study. Int. J. Cancer 2020, 147, 948–957. [Google Scholar] [CrossRef]
- McMullin, M.F.; Anderson, L.A. Aetiology of Myeloproliferative Neoplasms. Cancers 2020, 12, 1810. [Google Scholar] [CrossRef]
- Ramanathan, G.; Hoover, B.M.; Fleischman, A.G. Impact of Host, Lifestyle and Environmental Factors in the Pathogenesis of MPN. Cancers 2020, 12, 2038. [Google Scholar] [CrossRef]
- Ramanathan, G.; Fleischman, A.G. The Microenvironment in Myeloproliferative Neoplasms. Hematol. Oncol. Clin. N. Am. 2021, 35, 205–216. [Google Scholar] [CrossRef]
- Jones, A.V.; Cross, N.C. Inherited predisposition to myeloproliferative neoplasms. Ther. Adv. Hematol. 2013, 4, 237–253. [Google Scholar] [CrossRef] [PubMed]
- Tashi, T.; Swierczek, S.; Prchal, J.T. Familial MPN Predisposition. Curr. Hematol. Malig. Rep. 2017, 12, 442–447. [Google Scholar] [CrossRef] [PubMed]
- Guilford, P.; Hopkins, J.; Harraway, J.; McLeod, M.; McLeod, N.; Harawira, P.; Taite, H.; Scoular, R.; Miller, A.; Reeve, A.E. E-cadherin germline mutations in familial gastric cancer. Nature 1998, 392, 402–405. [Google Scholar] [CrossRef] [PubMed]
- Kirtane, K.; Lee, S.J. Racial and ethnic disparities in hematologic malignancies. Blood 2017, 130, 1699–1705. [Google Scholar] [CrossRef]
- Mehta, A.; Fillmore, N.; Dahiya, S.; D’Souza, A.; Sweetenham, J.; Kansagra, A. Reporting of race and ethnicity at an international haematology conference. Br. J. Haematol. 2020, 191, e107–e109. [Google Scholar] [CrossRef]
- Deadmond, M.A.; Smith-Gagen, J.A. Changing incidence of myeloproliferative neoplasms: Trends and subgroup risk profiles in the USA, 1973–2011. J. Cancer Res. Clin. Oncol. 2015, 141, 2131–2138. [Google Scholar] [CrossRef] [PubMed]
| Polycythaemia Vera | |||||
| Total * | European | Māori | Pacific Islanders | p Value | |
| N (%) | 275 (100%) | 202 (73.5%) | 47 (17.1%) | 16 (5.8%) | |
| Age at diagnosis, mean (SD) | 68.6 (15.0) | 71.7 (13.9) | 59.5 (15.8) | 56.8 (12.3) | <0.001 |
| Age at diagnosis, median (IQR) | 68.0 (55.0, 77.0) | 73.0 (64.0, 82.0) | 63.0 (48.5, 69.0) | 56.0 (48.8, 62.0) | |
| Gender (%) | |||||
| Female | 132 (48.0) | 98 (48.5) | 21 (44.7) | 6 (37.5) | 0.481 |
| Male | 143 (52.0) | 104 (51.5) | 26 (55.3) | 10 (62.5) | |
| Number of patients that died (%) | 141 (51.3) | 113 (55.9) | 20 (42.6) | 3 (18.9) | 0.017 |
| Age at death, median (IQR) | 81.0 (74.4, 87.2) | 81.0 (73.1, 87.4) | 68.7 (63.2, 77.0) | 66.1 (60.1, 77.1) | 0.001 |
| Essential Thrombocythaemia | |||||
| Total | European | Māori | Pacific Islanders | p Value | |
| N (%) | 360 (100%) | 282 (78.3%) | 45 (12.5%) | 11 (3.1%) | |
| Age at diagnosis, mean (SD) | 65.5 (16.0) | 67.4 (15.3) | 60.5 (16.9) | 61.1 (16.6) | 0.004 |
| Age at diagnosis, median (IQR) | 69.5 (60.0, 78.0) | 70.0 (58.0, 78.0) | 62.0 (52.0, 70.0) | 64.0 (46.5, 76.0) | |
| Gender (%) | |||||
| Female | 201 (55.8) | 154 (54.6) | 27 (60.0) | 7 (63.6) | 0.818 |
| Male | 159 (44.2) | 128 (45.4) | 18 (40.0) | 4 (36.4) | |
| Number of patients that died (%) | 109 (30.3) | 91 (32.3) | 14 (31.1) | 3 (27.3) | 0.310 |
| Age at death, median (IQR) | 74.6 (66.4, 84.7) | 81.6 (75.3, 87.7) | 78.7 (64.3, 84.4) | 81.0 (78.9, 81.1) | 0.379 |
| Primary Myelofibrosis | |||||
| Total | European | Māori | Pacific Islanders | p Value | |
| N (%) | 152 (100%) | 118 (77.6%) | 15 (9.9%) | 10 (6.6%) | |
| Age at diagnosis, mean (SD) | 69.2 (12.7) | 71.6 (10.8) | 61.5 (18.4) | 65.3 (8.1) | <0.001 |
| Age at diagnosis, median (IQR) | 70.0 (59.0, 80.0) | 71.0 (65.0, 80.0) | 57.0 (53.0, 66.0) | 62.5 (59.3, 72.3) | |
| Gender (%) | |||||
| Female | 53 (34.9) | 45 (38.1) | 3 (20.0) | 2 (20.0) | 0.369 |
| Male | 99 (65.1) | 73 (61.9) | 12 (80.0) | 8 (80.0) | |
| Number of patients that died (%) | 68 (44.7) | 55 (46.6) | 9 (60.0) | 4 (40.0) | 0.063 |
| Age at death, median (IQR) | 79 (71.0, 86.0) | 75.39 (68.1, 84.8) | 60.3 (55.6, 92.0) | 75.9 (71.2, 78.7) | 0.443 |
| Univariate Analysis | Multivariate Analysis | |||||
| Polycythaemia vera | ||||||
| HR for Death | 95% CI | p Value | HR for Death | 95% CI | p Value | |
| Age | 1.07 | 1.05–1.09 | <0.001 | 1.07 | 1.05–1.1 | 0.001 |
| Gender | ||||||
| Female | Ref | Ref | ||||
| Male | 1.26 | 0.90–1.77 | 0.182 | 1.43 | 1.01–2.0 | 0.041 |
| Ethnicity * | ||||||
| European | Ref | Ref | ||||
| Māori | 0.71 | 0.44–1.15 | 0.165 | 1.46 | 0.89–2.4 | 0.136 |
| Pacific Islanders | 0.23 | 0.07–0.73 | 0.013 | 0.49 | 0.15–1.6 | 0.231 |
| Essential thrombocythaemia | ||||||
| HR for Death | 95% CI | p Value | HR for Death | 95% CI | p Value | |
| Age | 1.11 | 1.09–0.14 | 1.113 | 1.007 | 0.981–1.034 | 0.591 |
| Gender # | ||||||
| Female | Ref | Ref | Ref | |||
| Male | 1.11 | 0.58–2.12 | 1.105 | 1.196 | 0.623–2.294 | 0.590 |
| Ethnicity *,# | ||||||
| European | Ref | Ref | Ref | |||
| Māori | 0.92 | 0.39–2.18 | 0.923 | 0.944 | 0.401–2.224 | 0.895 |
| Pacific Islanders | 0.84 | 0.11–6.43 | 0.842 | 1.320 | 0.182–9.561 | |
| Primary myelofibrosis | ||||||
| HR for Death | 95% CI | p Value | HR for Death | 95% CI | p Value | |
| Age | 1.05 | 1.03–1.08 | <0.001 | 1.06 | 1.04–1.1 | <0.001 |
| Gender | ||||||
| Female | Ref | Ref | ||||
| Male | 1.65 | 0.96–2.84 | 0.068 | 1.81 | 1.05–3.1 | 0.034 |
| Ethnicity * | ||||||
| European | Ref | |||||
| Māori | 1.45 | 0.72–2.95 | 0.300 | 2.94 | 1.37–6.3 | 0.006 |
| Pacific Islanders | 0.63 | 0.23–1.74 | 0.371 | 0.75 | 0.26–2.1 | 0.592 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Varghese, C.; Immanuel, T.; Ruskova, A.; Theakston, E.; Kalev-Zylinska, M.L. The Epidemiology of Myeloproliferative Neoplasms in New Zealand between 2010 and 2017: Insights from the New Zealand Cancer Registry. Curr. Oncol. 2021, 28, 1544-1557. https://doi.org/10.3390/curroncol28020146
Varghese C, Immanuel T, Ruskova A, Theakston E, Kalev-Zylinska ML. The Epidemiology of Myeloproliferative Neoplasms in New Zealand between 2010 and 2017: Insights from the New Zealand Cancer Registry. Current Oncology. 2021; 28(2):1544-1557. https://doi.org/10.3390/curroncol28020146
Chicago/Turabian StyleVarghese, Chris, Tracey Immanuel, Anna Ruskova, Edward Theakston, and Maggie L. Kalev-Zylinska. 2021. "The Epidemiology of Myeloproliferative Neoplasms in New Zealand between 2010 and 2017: Insights from the New Zealand Cancer Registry" Current Oncology 28, no. 2: 1544-1557. https://doi.org/10.3390/curroncol28020146
APA StyleVarghese, C., Immanuel, T., Ruskova, A., Theakston, E., & Kalev-Zylinska, M. L. (2021). The Epidemiology of Myeloproliferative Neoplasms in New Zealand between 2010 and 2017: Insights from the New Zealand Cancer Registry. Current Oncology, 28(2), 1544-1557. https://doi.org/10.3390/curroncol28020146






