The Utility of Combined Target and Systematic Prostate Biopsies in the Diagnosis of Clinically Significant Prostate Cancer Using Prostate Imaging Reporting and Data System Version 2 Based on Biparametric Magnetic Resonance Imaging
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. bpMRI Protocol
2.3. Prostate Biopsy Protocol
2.4. csPCa
2.5. Statistical Analysis
3. Results
3.1. Patient Characteristics
3.2. PCa Detection
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Projected Cancer Statistics. Available online: https://ganjoho.jp/en/public/statistics/short_pred.html (accessed on 15 September 2018).
- Heidenreich, A.; Bastian, P.J.; Bellmunt, J.; Bolla, M.; Joniau, S.; van der Kwast, T.; Mason, M.; Matveev, V.; Wiegel, T.; Zattoni, F.; et al. European Association of Urology. European Association of Urology. EAU guidelines on prostate cancer. Part 1: Screening, diagnosis, and local treatment with curative intent-update 2013. Eur. Urol. 2014, 65, 124–137. [Google Scholar] [CrossRef]
- Bjurlin, M.A.; Meng, X.; Le Nobin, J.; Wysock, J.S.; Lepor, H.; Rosenkrantz, A.B.; Taneja, S.S. Optimization of prostate biopsy: The role of magnetic resonance imaging targeted biopsy in detection, localization and risk assessment. J. Urol. 2014, 192, 648–658. [Google Scholar] [CrossRef]
- Guichard, G.; Larré, S.; Gallina, A.; Lazar, A.; Faucon, H.; Chemama, S.; Allory, Y.; Patard, J.; Vordos, D.; Hoznek, A.; et al. Extended 21-sample needle biopsy protocol for diagnosis of prostate cancer in 1000 consecutive patients. Eur. Urol. 2007, 52, 430–435. [Google Scholar] [CrossRef]
- Gershman, B.; Van Houten, H.K.; Herrin, J.; Moreira, D.M.; Kim, S.P.; Shah, N.D.; Karnes, R.J. Impact of prostate-specific antigen (PSA) Screening trials and revised PSA screening guidelines on rates of prostate biopsy and postbiopsy complications. Eur. Urol. 2017, 71, 55–65. [Google Scholar] [CrossRef]
- Washino, S.; Okochi, T.; Saito, K.; Konishi, T.; Hirai, M.; Kobayashi, Y.; Miyagawa, T. Combination of prostate imaging reporting and data system (PI-RADS) score and prostate-specific antigen (PSA) density predicts biopsy outcome in prostate biopsy naïve patients. BJU Int. 2016, 119, 225–233. [Google Scholar] [CrossRef]
- Tan, C.H.; Wei, W.; Johnson, V.; Kundra, V. Diffusion-weighted MRI in the detection of prostate cancer: Meta-analysis. AJR Am. J. Roentgenol. 2012, 199, 822–829. [Google Scholar] [CrossRef]
- Barentsz, J.O.; Richenberg, J.; Clements, R.; Choyke, P.; Verma, S.; Villeirs, G.; Rouviere, O.; Logager, V.; Fütterer, J.J.; European Society of Urogenital Radiology. ESUR prostate MR guidelines 2012. Eur. Radiol. 2012, 22, 746–757. [Google Scholar] [CrossRef]
- Weinreb, J.C.; Barentsz, J.O.; Choyke, P.L.; Cornud, F.; Haider, M.A.; Macura, K.J.; Margolis, D.; Schnall, M.D.; Shtern, F.; Tempany, C.M.; et al. PI-RADS prostate imaging—Reporting and data system: 2015, Version 2. Eur. Urol. 2016, 69, 16–40. [Google Scholar] [CrossRef]
- Stanzione, A.; Imbriaco, M.; Cocozza, S.; Fusco, F.; Rusconi, G.; Nappi, C.; Mirone, V.; Mangiapia, F.; Brunetti, A.; Ragozzino, A.; et al. Biparametric 3T Magnetic resonance imaging for prostatic cancer detection in a biopsy-naïve patient population: A further improvement of PI-RADS v2? Eur. J. Radiol. 2016, 85, 2269–2274. [Google Scholar] [CrossRef] [PubMed]
- Scialpi, M.; Prosperi, E.; D’Andrea, A.; Martorana, E.; Malaspina, C.; Palumbo, B.; Orlandi, A.; Falcone, G.; Milizia, M.; Mearini, L.; et al. Biparametric versus multiparametric MRI with non-endorectal coil at 3T in the detection and localization of prostate cancer. Anticancer Res. 2017, 37, 1263–1271. [Google Scholar]
- Kuhl, C.K.; Bruhn, R.; Krämer, N.; Nebelung, S.; Heidenreich, A.; Schrading, S. Abbreviated biparametric prostate MR imaging in men with elevated prostate-specific antigen. Radiology 2017, 285, 493–505. [Google Scholar] [CrossRef]
- Schoots, I.G.; Roobol, M.J.; Nieboer, D.; Bangma, C.H.; Steyerberg, E.W.; Hunink, M.G. Magnetic resonance imaging-targeted biopsy may enhance the diagnostic accuracy of significant prostate cancer detection compared to standard transrectal ultrasound-guided biopsy: A systematic review and meta-analysis. Eur. Urol. 2015, 68, 438–450. [Google Scholar] [CrossRef]
- Schoots, I.G.; Nieboer, D.; Giganti, F.; Moore, C.M.; Bangma, C.H.; Roobol, M.J. Magnetic resonance imaging-targeted biopsy a useful addition to systematic confirmatory biopsy in men on active surveillance for low-risk prostate cancer? A systematic review and meta-analysis. BJU Int. 2018, 122, 946–958. [Google Scholar] [CrossRef]
- Mottet, N.; Bellmunt, J.; Bolla, M.; Briers, E.; Cumberbatch, M.G.; De Santis, M.; Fossati, N.; Gross, T.; Henry, A.M.; Joniau, S.; et al. EAU-ESTRO-SIOG guidelines on prostate cancer. Part 1: Screening, diagnosis, and local treatment with curative intent. Eur. Urol. 2017, 71, 618–629. [Google Scholar] [CrossRef] [PubMed]
- Buyyounouski, M.K.; Choyke, P.L.; McKenney, J.K.; Sartor, O.; Sandler, H.M.; Amin, M.B.; Kattan, M.W.; Lin, D.W. Prostate cancer—Major changes in the American Joint Committee on Cancer eighth edition cancer staging manual. CA Cancer J. Clin. 2017, 67, 245–253. [Google Scholar] [CrossRef]
- Srigley, J.R.; Delahunt, B.; Samaratunga, H.; Billis, A.; Cheng, L.; Clouston, D.; Evans, A.; Furusato, B.; Kench, J.; Leite, K.; et al. Controversial issues in Gleason and International Society of Urological Pathology (ISUP) prostate cancer grading: Proposed recommendations for international implementation. Pathology 2019, 51, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Clavien, P.A.; Barkun, J.; de Oliveira, M.L.; Vauthey, J.N.; Dindo, D.; Schulick, R.D.; de Santibañes, E.; Pekolj, J.; Slankamenac, K.; Bassi, C.; et al. The Clavien-Dindo classification of surgical complications: Five-year experience. Ann. Surg. 2009, 250, 187–196. [Google Scholar] [CrossRef]
- Greer, M.D.; Shih, J.H.; Lay, N.; Barrett, T.; Kayat Bittencourt, L.; Borofsky, S.; Kabakus, I.M.; Law, Y.M.; Marko, J.; Shebel, H.; et al. Validation of the dominant sequence paradigm and role of dynamic contrast-enhanced imaging in PI-RADS Version 2. Radiology 2017, 285, 859–869. [Google Scholar] [CrossRef]
- Junker, D.; Steinkohl, F.; Fritz, V.; Bektic, J.; Tokas, T.; Aigner, F.; Herrmann, T.R.W.; Michael Rieger, M.; Nagele, U. Comparison of multiparametric and biparametric MRI of the prostate: Are gadolinium-based contrast agents needed for routine examinations? World J. Urol. 2019, 37, 691–699. [Google Scholar] [CrossRef]
- Obmann, V.C.; Pahwa, S.; Tabayayong, W.; Jiang, Y.; O’Connor, G.; Dastmalchian, S.; Lu, J.; Shah, S.; Herrmann, K.A.; Paspulati, R.; et al. Diagnostic accuracy of a rapid biparametric MRI protocol for detection of histologically proven prostate cancer. Urology 2018, 122, 133–138. [Google Scholar] [CrossRef]
- Radtke, J.P.; Boxler, S.; Kuru, T.H.; Wolf, M.B.; Alt, C.D.; Popeneciu, I.V.; Steinemann, S.; Huettenbrink, C.; Bergstraesser-Gasch, C.; Klein, T.; et al. Improved detection of anterior fibromuscular stroma and transition zone prostate cancer using biparametric and multiparametric MRI with MRI-targeted biopsy and MRI-US fusion guidance. Prostate Cancer Prostatic Dis. 2015, 18, 288–296. [Google Scholar] [CrossRef]
- Gulani, V.; Calamante, F.; Shellock, F.G.; Kanal, E.; Reeder, S.B.; International Society for Magnetic Resonance in Medicine. Gadolinium deposition in the brain: Summary of evidence and recommendations. Lancet Neurol. 2017, 16, 564–570. [Google Scholar] [CrossRef]
- Hansen, N.L.; Barrett, T.; Kesch, C.; Pepdjonovic, L.; Bonekamp, D.; O’Sullivan, R.; Distler, F.; Warren, A.; Samel, C.; Hadaschik, B.; et al. Multicentre evaluation of magnetic resonance imaging supported transperineal prostate biopsy in biopsy-naïve men with suspicion of prostate cancer. BJU Int. 2018, 122, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Mannaerts, C.K.; Kajtazovic, A.; Lodeizen, O.A.P.; Gayet, M.; Engelbrecht, M.R.W.; Jager, G.J.; Wijkstra, H.; de Reijke, T.M.; Beerlage, H.P. The added value of systematic biopsy in men with suspicion of prostate cancer undergoing multiparametric MRI-targeted biopsy. Urol. Oncol. 2019, 37, 298.e1–298.e9. [Google Scholar] [CrossRef]
- Tonttila, P.P.; Lantto, J.; Pääkkö, E.; Piippo, U.; Kauppila, S.; Lammentausta, E.; Ohtonen, P.; Vaarala, M.H. Prebiopsy multiparametric magnetic resonance imaging for prostate cancer diagnosis in biopsy-naive men with suspected prostate cancer based on elevated prostate-specific antigen values: Results from a randomized prospective blinded controlled trial. Eur. Urol. 2016, 69, 419–425. [Google Scholar] [CrossRef]
- Ploussard, G.; Beauval, J.B.; Lesourd, M.; Almeras, C.; Assoun, J.; Aziza, R.; Gautier, J.R.; Loison, G.; Portalez, D.; Salin, A.; et al. Added value of concomitant systematic biopsies for grade group prediction based on radical prostatectomy final pathology in magnetic resonance imaging positive cases undergoing fusion targeted biopsies. J. Urol. 2019, 202, 1182–1187. [Google Scholar] [CrossRef] [PubMed]
- Loeb, S.; Vellekoop, A.; Ahmed, H.U.; Catto, J.; Emberton, M.; Nam, R.; Rosario, D.J.; Scattoni, V.; Lotan, Y. Systematic review of complications of prostate biopsy. Eur. Urol. 2013, 64, 876–892. [Google Scholar] [CrossRef]
- Dalhoff, A. Global fluoroquinolone resistance epidemiology and implictions for clinical use. Interdiscip. Perspect. Infect. Dis. 2012, 2012, 976273. [Google Scholar] [CrossRef] [PubMed]
- Williamson, D.A.; Masters, J.; Freeman, J.; Roberts, S. Travel-associated extended-spectrum β-lactamase-producing Escherichia coli bloodstream infection following transrectal ultrasound-guided prostate biopsy. BJU Int. 2012, 109, E21–E22. [Google Scholar] [CrossRef]
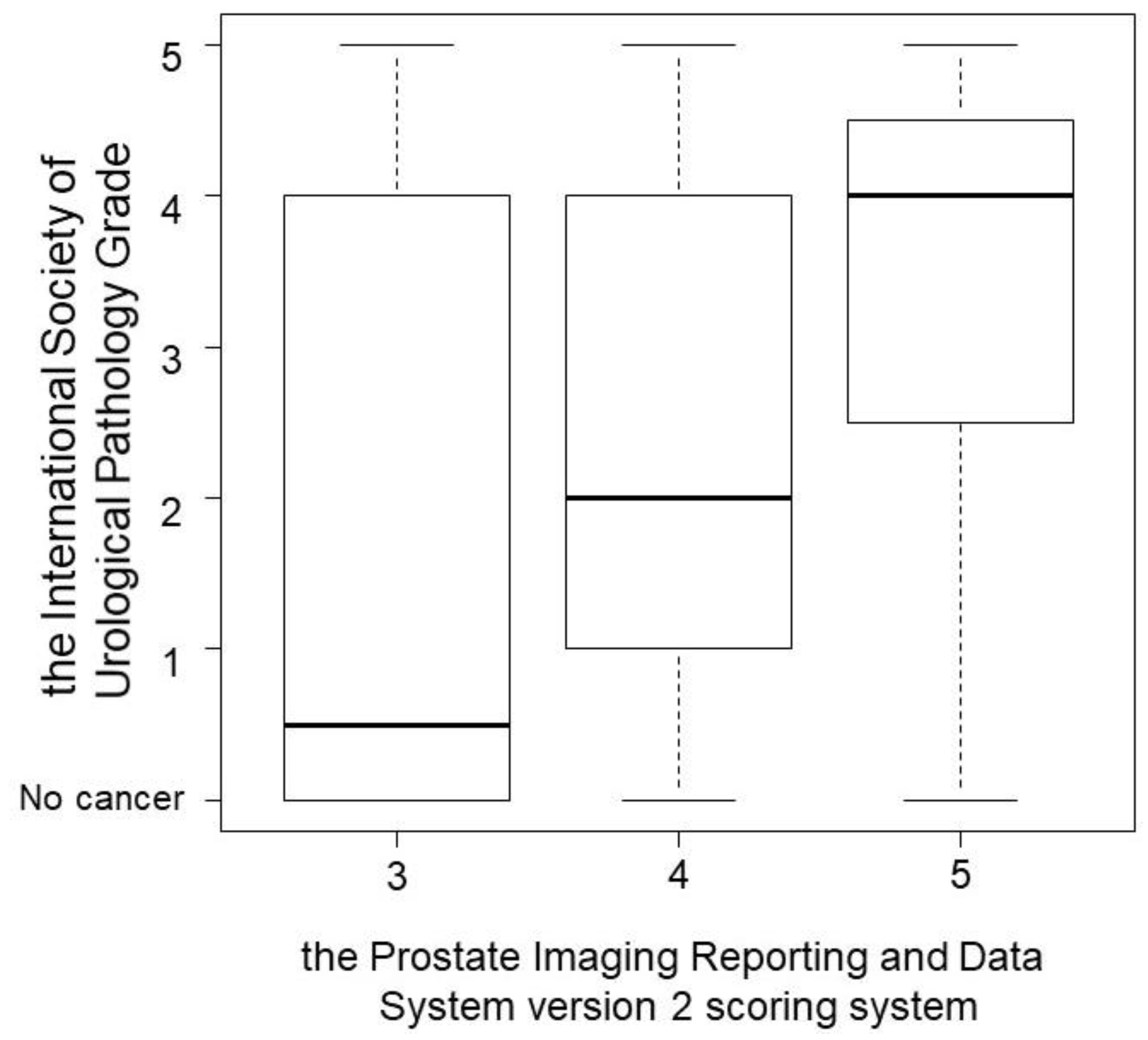
| Age (year, median, interquartile range) | 71 (67–75) |
| Body mass index (kg/m2, median, interquartile range) | 23.2 (21.5–25.3) |
| Prostate-specific antigen (ng/mL, median, interquartile range) | 8.32 (5.38–13.86) |
| Prostate volume (mL, median, interquartile range) | 30.7 (23.0–44.8) |
| Prostate-specific antigen density (ng/mL/cm3, median, interquartile range) | 0.26 (0.18–0.44) |
| Prostate Imaging Reporting and Data System version 2 (number, %) | |
| 3 | 22 (21.1) |
| 4 | 55 (52.9) |
| 5 | 27 (26.0) |
| Number, % | |
|---|---|
| Concordance | 13 (24.1) |
| Inconsistency | 41 (75.9) |
| Concordance ISUP grade between target and systemic biopsy | 10 (24.4) |
| Upgrading ISUP grade on target biopsy | 14 (34.1) |
| Upgrading ISUP grade on systemic biopsy | 17 (41.5) |
| Type of Complication (Number, %) | Any Grade | Grade 3 |
|---|---|---|
| Prostatitis | 2 (1.9) | 1 (1.0) |
| Hematuria | 44 (42.3) | 2 (1.9) |
| Urinary retention | 3 (2.9) | 2 (1.9) |
| Hematospermia | 1 (1.0) | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kato, D.; Ozawa, K.; Takeuchi, S.; Kawase, M.; Kawase, K.; Nakai, C.; Takai, M.; Iinuma, K.; Nakane, K.; Kato, H.; et al. The Utility of Combined Target and Systematic Prostate Biopsies in the Diagnosis of Clinically Significant Prostate Cancer Using Prostate Imaging Reporting and Data System Version 2 Based on Biparametric Magnetic Resonance Imaging. Curr. Oncol. 2021, 28, 1294-1301. https://doi.org/10.3390/curroncol28020123
Kato D, Ozawa K, Takeuchi S, Kawase M, Kawase K, Nakai C, Takai M, Iinuma K, Nakane K, Kato H, et al. The Utility of Combined Target and Systematic Prostate Biopsies in the Diagnosis of Clinically Significant Prostate Cancer Using Prostate Imaging Reporting and Data System Version 2 Based on Biparametric Magnetic Resonance Imaging. Current Oncology. 2021; 28(2):1294-1301. https://doi.org/10.3390/curroncol28020123
Chicago/Turabian StyleKato, Daiki, Kaori Ozawa, Shinichi Takeuchi, Makoto Kawase, Kota Kawase, Chie Nakai, Manabu Takai, Koji Iinuma, Keita Nakane, Hiroki Kato, and et al. 2021. "The Utility of Combined Target and Systematic Prostate Biopsies in the Diagnosis of Clinically Significant Prostate Cancer Using Prostate Imaging Reporting and Data System Version 2 Based on Biparametric Magnetic Resonance Imaging" Current Oncology 28, no. 2: 1294-1301. https://doi.org/10.3390/curroncol28020123
APA StyleKato, D., Ozawa, K., Takeuchi, S., Kawase, M., Kawase, K., Nakai, C., Takai, M., Iinuma, K., Nakane, K., Kato, H., Matsuo, M., Suzui, N., Miyazaki, T., & Koie, T. (2021). The Utility of Combined Target and Systematic Prostate Biopsies in the Diagnosis of Clinically Significant Prostate Cancer Using Prostate Imaging Reporting and Data System Version 2 Based on Biparametric Magnetic Resonance Imaging. Current Oncology, 28(2), 1294-1301. https://doi.org/10.3390/curroncol28020123






