Survival Impact of Aggressive Treatment and PD-L1 Expression in Oligometastatic NSCLC
Abstract
1. Introduction
2. Materials
2.1. Study Design
2.2. Patients and Pathology Specimens
2.3. Treatments
2.4. End Points
2.5. Statistical Analysis
3. Results
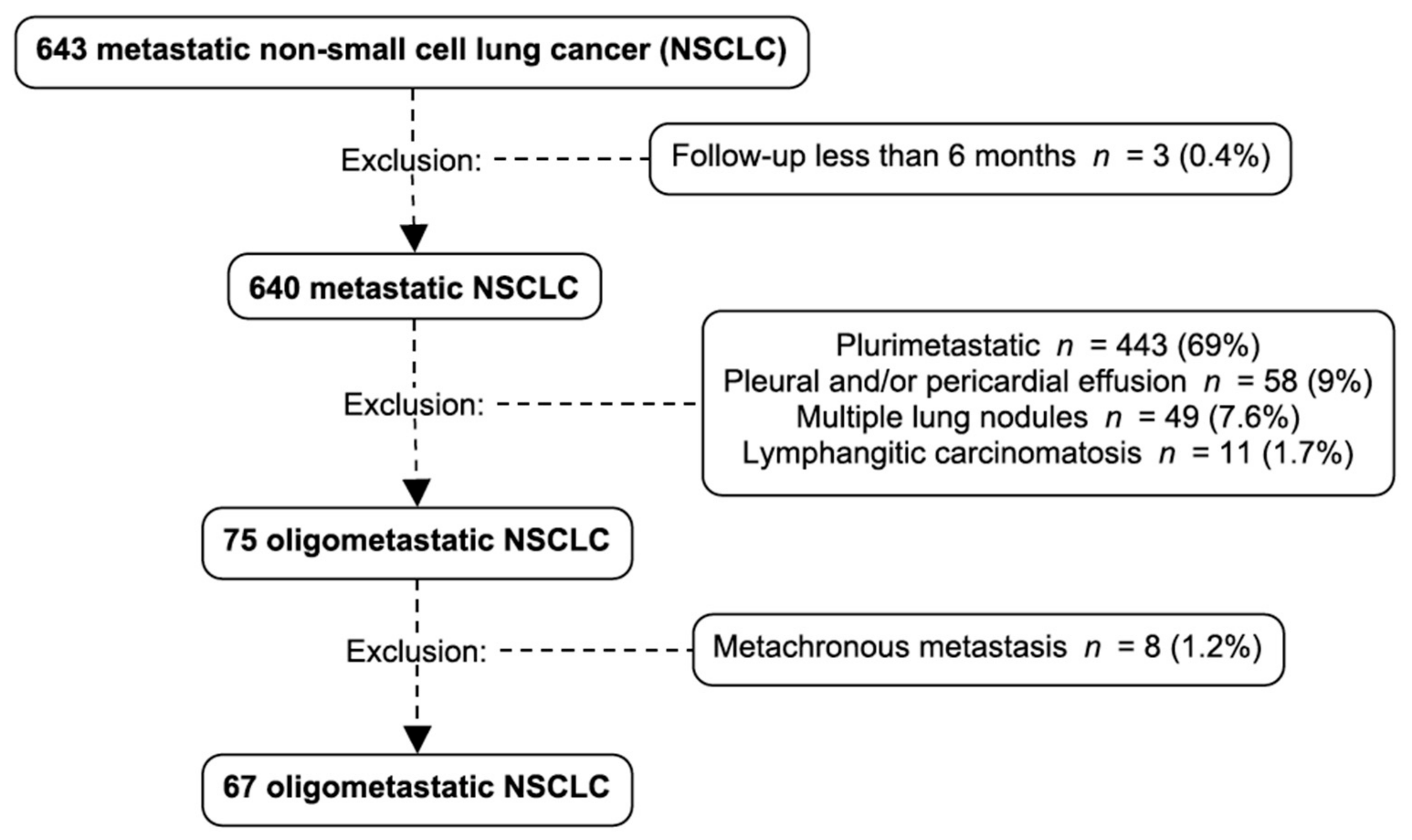
3.1. Oligometastatic Non-Small Cell Lung Cancer (NSCLC) Prevalence
3.2. Metastatic Sites Prevalence
3.3. Treatments
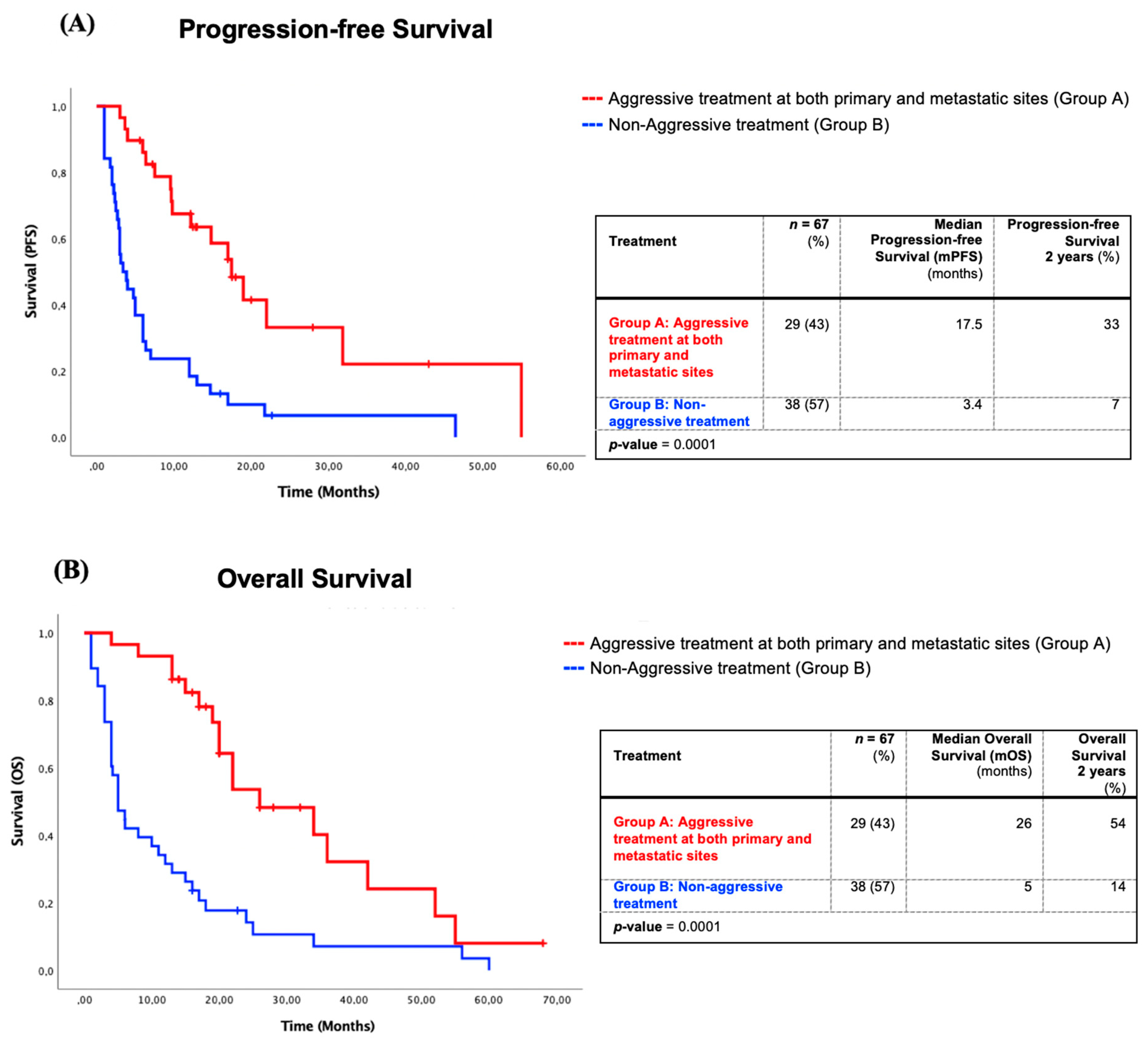
3.3.1. Survival of Patients with Aggressive Treatment at Both Metastatic and Primary Sites Compared to Non-Aggressive Treatments
3.3.2. Survival Impact of Four Management Approaches in Oligometastatic NSCLC
3.3.3. Overall Survival (OS) and Progression-Free Survival (PFS) Analyzed by “Modified” Stage of Patients with Aggressive Treatment at Both Sites versus Other Treatments
- Stage I: The mOS for the “modified” stage I oligometastatic NSCLC, which refers to the combined stage T and N of the IASLC staging eighth edition, radically treated at both sites (Group A) was 42 months compared with 16 months for other forms of treatments (Group B) (p = 0.316). The mPFS was the same in both group (p = 0.669) (Table S2).
- Stage II: The mOS of “modified” stage II of oligometastatic NSCLC patients of Group A was 34 months vs. 6 months for group B and was statistically significant (p = 0.024). The mPFS of the “modified” stage II patients treated radically at both sites was not reached vs. 6.0 months of group B patients (p = 0.016) (Table S2).
- Stage III: The mOS and mPFS for “modified” stage III was significantly higher in Group A with an mOS of 22 months vs. 4 months for Group B (p = 0.001). The mPFS of the “modified” stage III of Groups A and B were respectively 19 months vs. 3 months (p = 0.003) (Table S2).
3.4. Epidermal Growth Factor Receptor (EGFR) Mutation Status and Anaplastic Lymphoma Kinase (ALK) Expression Profile
3.5. Programmed Cell Death Ligand 1 (PD-L1) Expression Profile
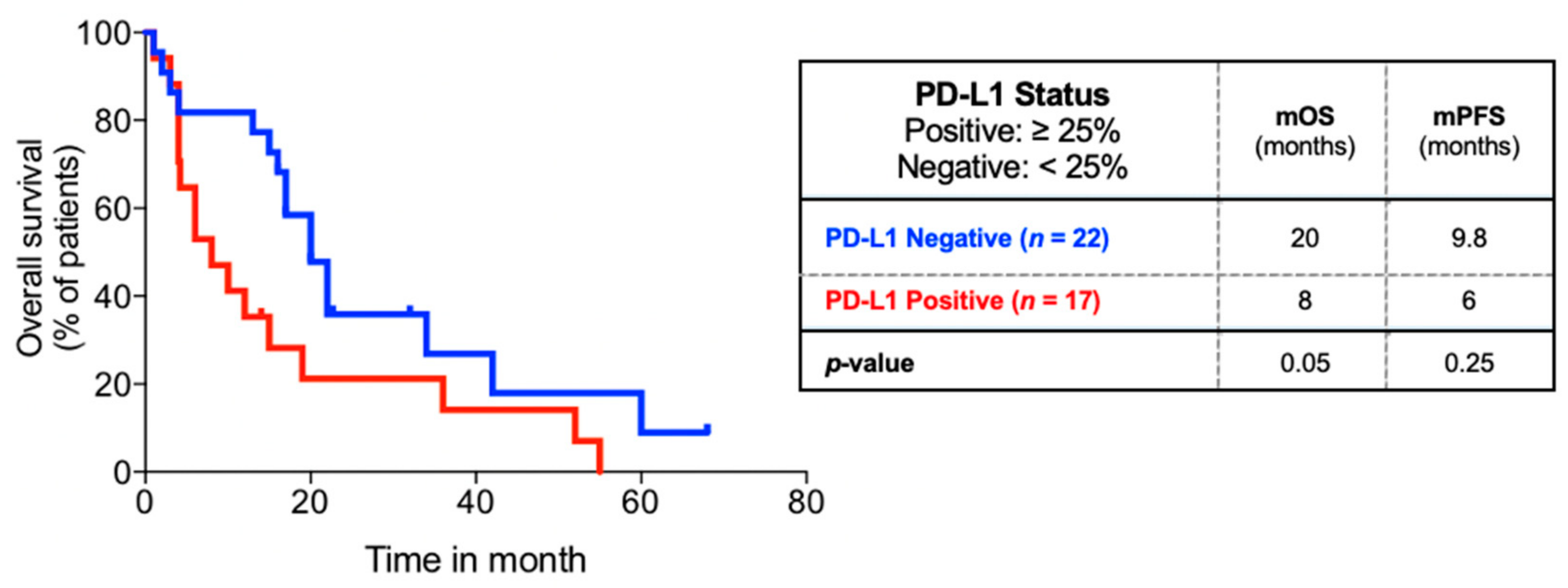
3.6. PD-L1 Status and Clinical Outcomes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Eberhardt, W.E.E.; Mitchell, A.; Crowley, J.; Kondo, H.; Kim, Y.T.; Turrisi, A., III; Goldstraw, P.; Rami-Porta, R.; On behalf of the International Association for the Study of Lung Cancer Staging and Prognostic Factors Committee; Advisory Board Members, and Participating Institutions. The IASLC Lung Cancer Staging Project: Proposals for the Revision of the M Descriptors in the Forthcoming Eighth Edition of the TNM Classification of Lung Cancer. J. Thorac. Oncol. 2015, 10, 1515–1522. [Google Scholar] [CrossRef] [PubMed]
- Hellman, S.; Weichselbaum, R.R. Oligometastases. J. Clin. Oncol. 1995, 13, 8–10. [Google Scholar] [CrossRef] [PubMed]
- Dingemans, A.M.C.; Hendriks, L.E.L.; Berghmans, T.; Levy, A.; Hasan, B.; Faivre-Finn, C.; Giaj-Levra, M.; Giaj-Levra, M.; Girard, N.; Greillier, L.; et al. Definition of Synchronous Oligometastatic Non-Small Cell Lung Cancer-A Consensus Report. J. Thorac. Oncol. 2019, 14, 2109–2119. [Google Scholar] [CrossRef] [PubMed]
- Laurie, S.A.; Banerji, S.; Blais, N.; Brule, S.; Cheema, P.K.; Cheung, P.; Daaboul, N.; Hao, D.; Hirsh, V.; Juergens, R.; et al. Canadian consensus: Oligoprogressive, pseudoprogressive, and oligometastatic non-small-cell lung cancer. Curr. Oncol. 2019, 26, e81–e93. [Google Scholar] [CrossRef] [PubMed]
- Torok, J.A.; Gu, L.; Tandberg, D.J.; Wang, X.; Harpole Jr, D.H.; Kelsey, C.R.; Salama, J.K. Patterns of Distant Metastases After Surgical Management of Non-Small-cell Lung Cancer. Clin. Lung Cancer 2017, 18, e57–e70. [Google Scholar] [CrossRef]
- Parikh, R.B.; Cronin, A.M.; Kozono, D.E.; Oxnard, G.R.; Mak, R.H.; Jackman, D.M.; Lo, P.C.; Baldini, E.H.; Johnson, B.E.; Chen, A.B. Definitive primary therapy in patients presenting with oligometastatic non-small cell lung cancer. Int. J. Radiat. Oncol. Biol. Phys. 2014, 89, 880–887. [Google Scholar] [CrossRef]
- De Ruysscher, D.; Wanders, R.; van Baardwijk, A.; Dingemans, A.M.C.; Reymen, B.; Houben, R.; Bootsma, G.; Pitz, C.; van Eijsden, L.; Geraedts, W.; et al. Radical treatment of non-small-cell lung cancer patients with synchronous oligometastases: Long-term results of a prospective phase II trial (Nct01282450). J. Thorac. Oncol. 2012, 7, 1547–1555. [Google Scholar] [CrossRef]
- Gomez, D.R.; Tang, C.; Zhang, J.; Blumenschein Jr, G.R.; Hernandez, M.; Lee, J.J.; Ye, R.; Palma, D.A.; Louie, A.V.; Camidge, D.R.; et al. Local Consolidative Therapy Vs. Maintenance Therapy or Observation for Patients With Oligometastatic Non-Small-Cell Lung Cancer: Long-Term Results of a Multi-Institutional, Phase II, Randomized Study. J. Clin. Oncol. 2019, 37, 1558–1565. [Google Scholar] [CrossRef]
- Villarreal-Garza, C.; de la Mata, D.; Zavala, D.G.; Macedo-Perez, E.O.; Arrieta, O. Aggressive treatment of primary tumor in patients with non-small-cell lung cancer and exclusively brain metastases. Clin. Lung Cancer 2013, 14, 6–13. [Google Scholar] [CrossRef]
- Ashworth, A.; Rodrigues, G.; Boldt, G.; Palma, D. Is there an oligometastatic state in non-small cell lung cancer? A systematic review of the literature. Lung Cancer 2013, 82, 197–203. [Google Scholar] [CrossRef]
- Patel, A.N.; Simone, C.B., 2nd; Jabbour, S.K. Risk factors and management of oligometastatic non-small cell lung cancer. Ther. Adv. Respir. Dis. 2016, 10, 338–348. [Google Scholar] [CrossRef] [PubMed]
- Reck, M.; Rodriguez-Abreu, D.; Robinson, A.G.; Hui, R.; Csoszi, T.; Fulop, A.; Gottfried, M.; Peled, N.; Tafreshi, A.; Cuffe, S.; et al. Pembrolizumab versus Chemotherapy for PD-L1-Positive Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2016, 375, 1823–1833. [Google Scholar] [CrossRef] [PubMed]
- Rebelatto, M.C.; Midha, A.; Mistry, A.; Sabalos, C.; Schechter, N.; Li, X.; Jin, X.; Steele, K.E.; Robbins, P.B.; Blake-Haskins, J.A.; et al. Development of a programmed cell death ligand-1 immunohistochemical assay validated for analysis of non-small cell lung cancer and head and neck squamous cell carcinoma. Diagn. Pathol. 2016, 11, 95. [Google Scholar] [CrossRef] [PubMed]
- Ionescu, D.N.; Downes, M.R.; Christofides, A.; Tsao, M.S. Harmonization of PD-L1 testing in oncology: A Canadian pathology perspective. Curr. Oncol. 2018, 25, e209–e216. [Google Scholar] [CrossRef]
- Brody, R.; Zhang, Y.; Ballas, M.; Siddiqui, M.K.; Gupta, P.; Barker, C.; Midha, A.; Walker, J. PD-L1 expression in advanced NSCLC: Insights into risk stratification and treatment selection from a systematic literature review. Lung Cancer 2017, 112, 200–215. [Google Scholar] [CrossRef]
- Li, S.; Zhu, R.; Li, D.; Li, N.; Zhu, X. Prognostic factors of oligometastatic non-small cell lung cancer: A meta-analysis. J. Thorac. Dis. 2018, 10, 3701–3713. [Google Scholar] [CrossRef]
- Uruga, H.; Bozkurtlar, E.; Huynh, T.G.; Muzikansky, A.; Goto, Y.; Gomez-Caraballo, M.; Hata, A.N.; Gainor, J.F.; Mark, E.J.; Engelman, J.A.; et al. Programmed Cell Death Ligand (PD-L1) Expression in Stage II and III Lung Adenocarcinomas and Nodal Metastases. J. Thorac. Oncol. 2017, 12, 458–466. [Google Scholar] [CrossRef]
- Li, M.; Li, A.; Zhou, S.; Xu, Y.; Xiao, Y.; Bi, R.; Yang, W. Heterogeneity of PD-L1 expression in primary tumors and paired lymph node metastases of triple negative breast cancer. BMC Cancer 2018, 18, 4. [Google Scholar] [CrossRef]
- Basu, A.; Yearley, J.H.; Annamalai, L.; Pryzbycin, C.; Rini, B. Association of PD-L1, PD-L2, and Immune Response Markers in Matched Renal Clear Cell Carcinoma Primary and Metastatic Tissue Specimens. Am. J. Clin. Pathol. 2019, 151, 217–225. [Google Scholar] [CrossRef]
- Lantuejoul, S.; Damotte, D.; Hofman, V.; Adam, J. Programmed death ligand 1 immunohistochemistry in non-small cell lung carcinoma. J. Thorac. Dis. 2019, 11, S89–S101. [Google Scholar] [CrossRef]
- Palma, D.A.; Olson, R.; Harrow, S.; Gaede, S.; Louie, A.V.; Haasbeek, C.; Mulroy, L.; Lock, M.; Rodrigues, G.B.; Yaremko, B.P.; et al. Stereotactic Ablative Radiotherapy for the Comprehensive Treatment of Oligometastatic Cancers: Long-Term Results of the SABR-COMET Phase II Randomized Trial. J. Clin. Oncol. 2020, 38, 2830–2838. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Brain (n = 50) 1 Metastases (n = 37) 2 Metastases (n = 9) 3 Metastases (n = 4) | Bone (n = 4) | Adrenal (n = 6) | Liver (n = 2) | Contralateral Lung (n = 5) | Total N (n = 67) |
|---|---|---|---|---|---|---|
| Sex | ||||||
| Male | 29 (43) | 2 (3) | 3 (5) | 2 (3) | 2 (3) | 38 (57) |
| Age at diagnosis | ||||||
| < 65 | 34 (51) | 3 (5) | 3 (5) | 1 (2) | 4 (6) | 45 (67) |
| ≥65 | 16 (24) | 1 (2) | 3 (5) | 1 (2) | 1 (2) | 22 (33) |
| Smoking history | ||||||
| Positive | 50 (75) | 2 (3) | 6 (9) | 2 (3) | 5 (8) | 65 (97) |
| ECOG | ||||||
| 0 | 13 (19) | 1 (2) | 1 (2) | 0 (0) | 2 (3) | 17 (25) |
| 1 | 20 (30) | 3 (5) | 2 (3) | 2 (3) | 1 (2) | 28 (42) |
| 2 | 5 (8) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 5 (8) |
| 3 | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| 4 | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| N/A | 12 (18) | 0 (0) | 3 (5) | 0 (0) | 2 (3) | 17 (25) |
| Histology | ||||||
| Adenocarcinoma | 36 (54) | 3 (5) | 4 (6) | 0 (0) | 5 (8) | 48 (2) |
| Large Cell Carcinoma | 3 (5) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 3 (5) |
| Squamous Cell Carcinoma | 2 (3) | 0 (0) | 2 (3) | 2 (3) | 0 (0) | 6 (9) |
| Unspecified | 9 (13) | 1 (2) | 0 (0) | 0 (0) | 0 (0) | 10 (15) |
| PD-L1 status * | ||||||
| Positive | 16 (41) | 0 (0) | 1 (3) | 0 (0) | 0 (0) | 17 (44) |
| Negative | 18 (46) | 2 (5) | 0 (0) | 0 (0) | 2 (5) | 22 (56) |
| Insufficient tissue | 16 | 2 | 5 | 2 | 3 | 28 |
| EGFRstatus | ||||||
| Positive | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Negative | 29 (43) | 4 (6) | 4 (6) | 1 (1) | 3 (4) | 41 (61) |
| Insufficient tissue | 21 (31) | 0 (0) | 2 (4) | 1 (1) | 2 (3) | 26 (39) |
| ALKstatus | ||||||
| Positive | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Negative | 35 (52) | 4 (6) | 5 (7) | 1 (1) | 4 (6) | 49 (73) |
| Insufficient tissue | 15 (22) | 0 | 1 (1) | 1 (1) | 1 (1) | 18 (27) |
| Metastatic Site | Brain (n = 50) | Bone (n = 4) | Adrenal gland (n = 6) | Liver (n = 2) | Contralateral lung (n = 5) | Total (n = 67) |
|---|---|---|---|---|---|---|
| Aggressive treatment | 52 (78) | |||||
| Group A | 29 (43) | |||||
| Metastatic site + Primary site + Adjuvant chemotherapy (Group A1) | 4 (6) | 1 (2) | 0 (0.0) | 1 (2) | 1 (2) | 7 (10) |
| Metastatic site + Primary sites (Group A2) | 21 (31) | 0 (0.0) | 1 (2) | 0 (0) | 0 (0) | 22 (33) |
| Group B1 | 4 (6) | |||||
| Primary site only | 1 (2) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 1 (2) |
| Primary site + adjuvant chemotherapy | 0 (0) | 1 (2) | 0 (0) | 0 (0) | 2 (3) | 3 (5) |
| Group B2 | 19 (28) | |||||
| Metastatic site only | 11 (16) | 0 (0) | 0 (0) | 0 (0) | 1 (2) | 12 (18) |
| Metastatic site + palliative chemotherapy +/− palliative RT | 6 (9) | 1 (2) | 0 (0) | 0 (0) | 0 (0) | 7 (10) |
| Palliative treatment (Group B3) | 15 (22) | |||||
| Watch & Wait | 0 (0) | 0 (0) | 2 (3) | 0 (0) | 0 (0) | 2 (3) |
| Palliative chemotherapy | 0 (0) | 0 (0) | 2 (3) | 1 (2) | 1 (2) | 4 (6) |
| Palliative tyrosine-kinase inhibitor | 0 (0) | 1 (2) | 0 (0) | 0 (0) | 0 (0) | 1 (2) |
| Palliative chemotherapy + RT | 1 (2) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 1 (2) |
| Palliative RT | 6 (9) | 0 (0) | 1 (2) | 0 (0) | 0 (0) | 7 (10) |
| Staging by metastatic site | ||||||
| 8th IASLC classification TN staging (p = 0.053) | ||||||
| T1–4 N0 | 22 (33) | 2 (3) | 2 (3) | 2 (3) | 3 (5) | 31(46) |
| T1–4 N1 | 3 (5) | 0 (0) | 0 (0) | 0 (0) | 1 (2) | 4 (6) |
| T1–4 N2 | 17 (25) | 2 (3) | 3 (5) | 0 (0) | 1 (2) | 23 (34) |
| T1–4 N3 | 8 (12) | 0 (0) | 1 (2) | 0 (0) | 0 (0) | 9 (13) |
| “Modified” Stage based on the combined 8th IASLC Edition T and N stages (p = 0.142) | ||||||
| Stage I (T1–2 N0 M1b) | 13 (20) | 2 (3) | 0 (0) | 1 (2) | 2 (3) | 18 (27) |
| Stage II (T1–2 N1 M1b and T3–4 N0 M1b) | 10 (15) | 0 (0) | 0 (0) | 1 (2) | 2 (3) | 13 (19) |
| Stage III (T1–2 N2 M1b and T3–4 N1 M1b) | 27 (40) | 2 (3) | 6 (9) | 0 (0) | 1 (2) | 36 (54) |
| Demographic and Tumoral Characteristics | Subgroup | PD-L1 Positive (≥25%) n (%) | PD-L1 Negative (<25%) n (%) | p-value |
|---|---|---|---|---|
| Sex | Men | 8 (21) | 13 (33) | 0.455 |
| Women | 9 (23) | 9 (23) | ||
| Age at diagnosis | ≥50 y.o. | 15 (38) | 19 (49) | 0.862 |
| <50 y.o. | 2 (5) | 3 (8) | ||
| Smoking history | ≤25 pack-years | 2 (5) | 2 (8) | 0.371 |
| >25 to <50 pack-years | 8 (21) | 14 (36) | ||
| ≥50 pack-years | 5 (13) | 6 (15) | ||
| N/A | 2 (5) | 0 | ||
| ECOG at diagnosis | 0 | 3 (8) | 7 (18) | 0.719 |
| 1 | 6 (15) | 8 (21) | ||
| 2 | 1 (3) | 1 (3) | ||
| N/A | 7 (18) | 6 (15) | ||
| T staging | 1 | 1 (3) | 9 (23) | 0.065 |
| 2 | 6 (15) | 8 (21) | ||
| 3 | 4 (10) | 2 (5) | ||
| 4 | 4 (10) | 2 (5) | ||
| N/A | 2 (5) | 1 (3) | ||
| N staging | 0 | 8 (21) | 15 (38) | 0.470 |
| 1 | 1 (3) | 1 (3) | ||
| 2 | 5 (13) | 5 (13) | ||
| 3 | 3 (8) | 1 (3) | ||
| Histology | Adenocarcinoma | 11 (28) | 17 (44) | 0.849 |
| Squamous Cell Carcinoma | 1 (3) | 1 (3) | ||
| Large Cell Carcinoma | 1 (3) | 1 (3) | ||
| Poorly Differentiated | 4 (10) | 3 (8) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gauvin, C.; Krishnan, V.; Kaci, I.; Tran-Thanh, D.; Bédard, K.; Albadine, R.; Leduc, C.; Gaboury, L.; Blais, N.; Tehfe, M.; et al. Survival Impact of Aggressive Treatment and PD-L1 Expression in Oligometastatic NSCLC. Curr. Oncol. 2021, 28, 593-605. https://doi.org/10.3390/curroncol28010059
Gauvin C, Krishnan V, Kaci I, Tran-Thanh D, Bédard K, Albadine R, Leduc C, Gaboury L, Blais N, Tehfe M, et al. Survival Impact of Aggressive Treatment and PD-L1 Expression in Oligometastatic NSCLC. Current Oncology. 2021; 28(1):593-605. https://doi.org/10.3390/curroncol28010059
Chicago/Turabian StyleGauvin, Camille, Vimal Krishnan, Imane Kaci, Danh Tran-Thanh, Karine Bédard, Roula Albadine, Charles Leduc, Louis Gaboury, Normand Blais, Mustapha Tehfe, and et al. 2021. "Survival Impact of Aggressive Treatment and PD-L1 Expression in Oligometastatic NSCLC" Current Oncology 28, no. 1: 593-605. https://doi.org/10.3390/curroncol28010059
APA StyleGauvin, C., Krishnan, V., Kaci, I., Tran-Thanh, D., Bédard, K., Albadine, R., Leduc, C., Gaboury, L., Blais, N., Tehfe, M., Routy, B., & Florescu, M. (2021). Survival Impact of Aggressive Treatment and PD-L1 Expression in Oligometastatic NSCLC. Current Oncology, 28(1), 593-605. https://doi.org/10.3390/curroncol28010059





