Effect of (Poly)phenols as Potential Agents in the Treatment of Psoriasis: A Systematic Review of the Evidence and Future Applications
Abstract
1. Introduction
1.1. Epidemiology and Pathophysiology of Psoriasis
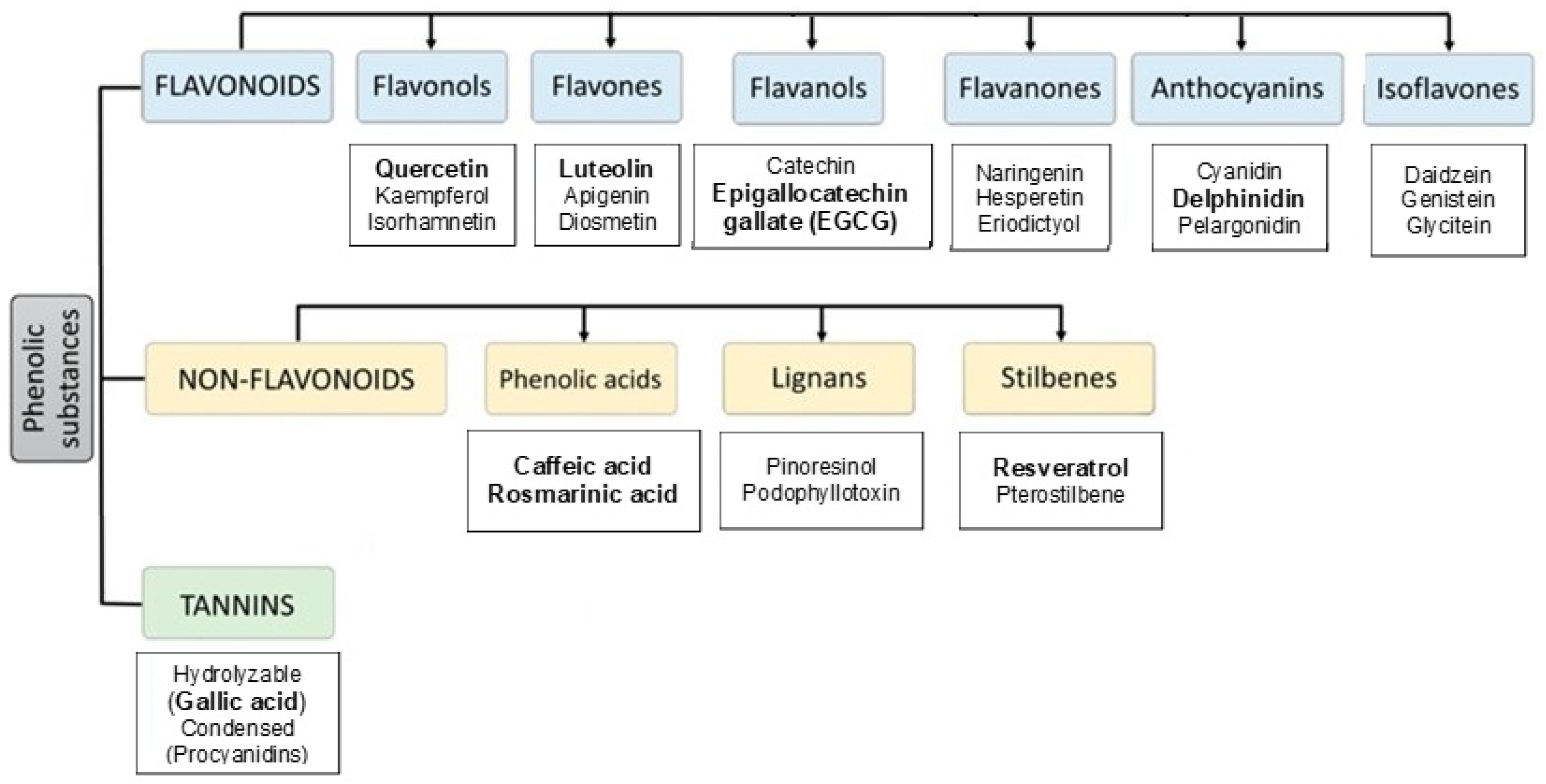
1.2. Categorizing Polyphenols
2. Materials and Methods
2.1. Protocol Registration
2.2. Data Sources and Search Strategy
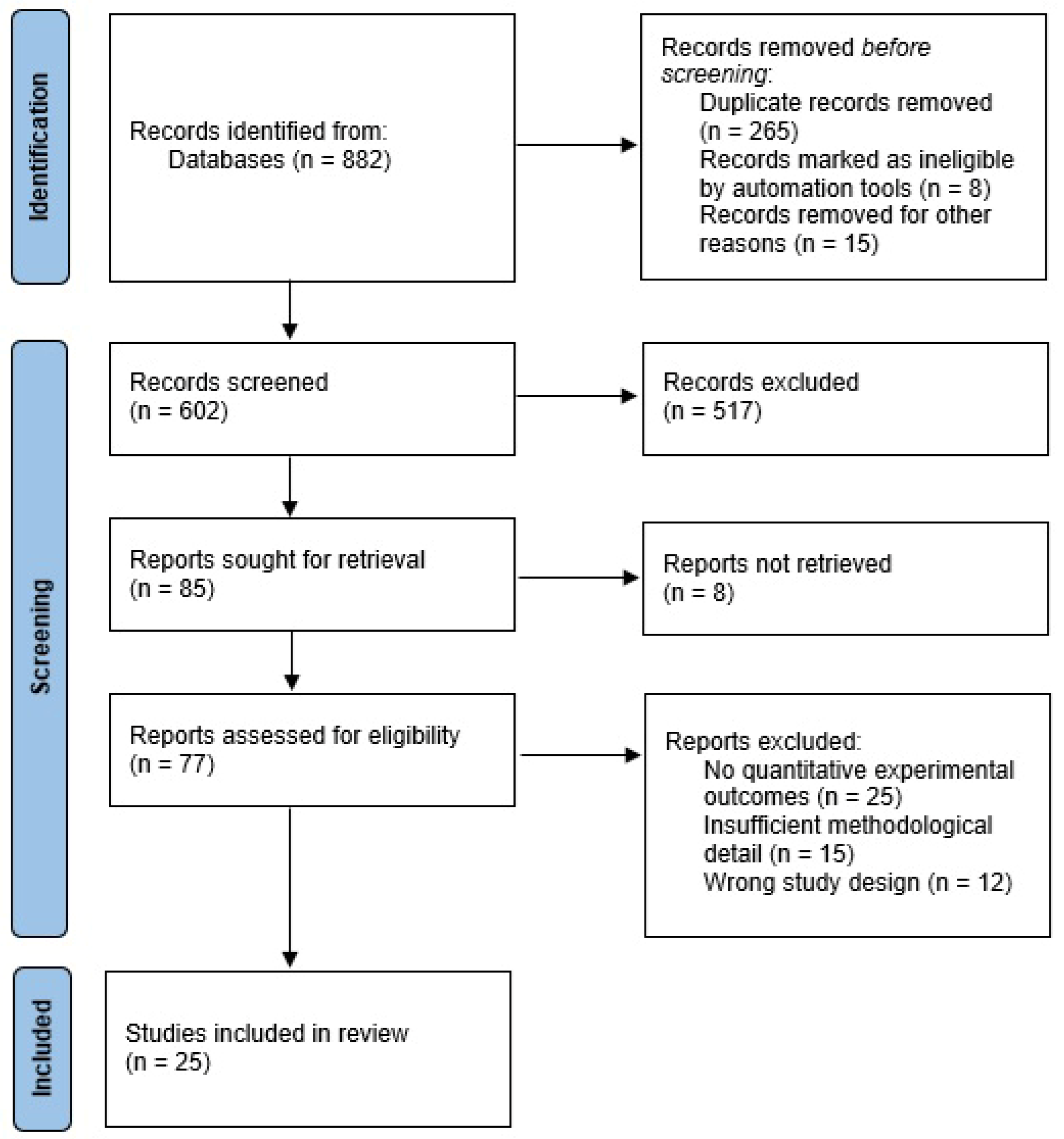
2.3. Study Selection and Data Acquisition
3. Results
3.1. Study Characteristics and Quality Assessment
3.2. Polyphenol Distribution
3.3. Study Characteristics and Therapeutic Outcomes
3.4. Dose–Response Relationships and Bioavailability Considerations
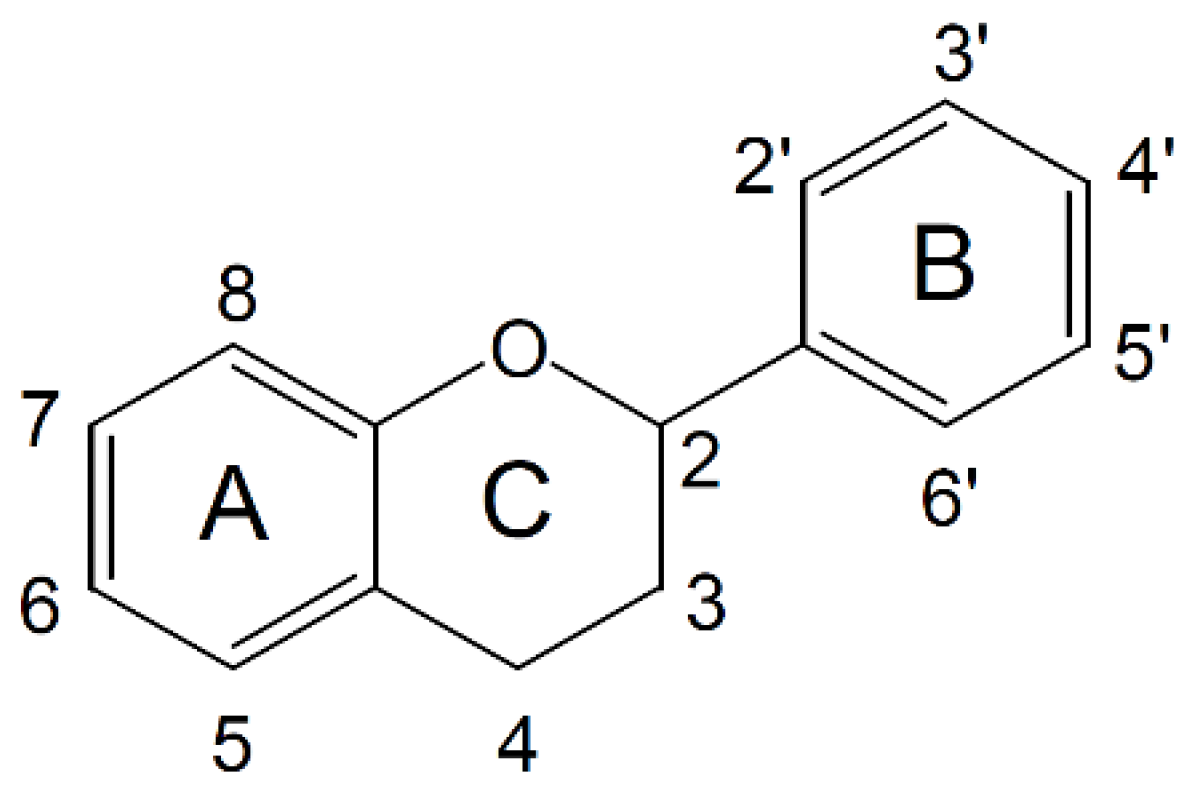
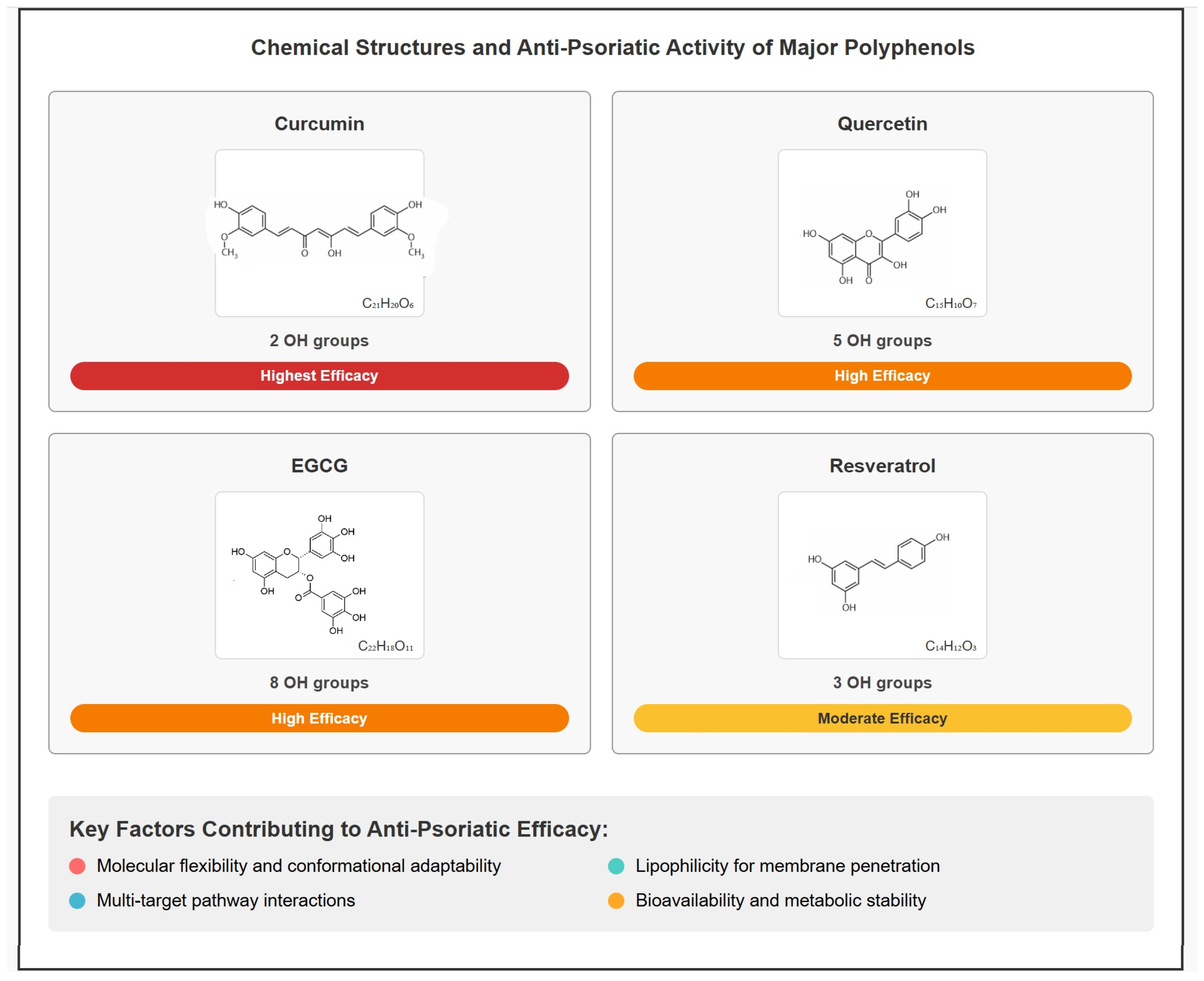
3.5. Structure–Activity Relationships of Major Polyphenols
4. Discussion
4.1. Mechanistic Analysis of Polyphenolic Anti-Psoriatic Effects
4.1.1. NF-kappaB Pathway Modulation: A Central Mechanism
4.1.2. IL-17/Th17 Axis Suppression: Targeting Adaptive Immunity
4.1.3. Keratinocyte Proliferation Control: Addressing Hyperproliferation
4.1.4. Oxidative Stress Reduction: Addressing Fundamental Pathophysiology
4.1.5. Multi-Target vs. Single-Target Approaches: Mechanistic Comparison
4.1.6. Structure–Activity Considerations
4.2. Comparative Analysis of Polyphenol Efficacy
4.3. Dose–Response Relationships and Bioavailability
4.4. Safety Considerations and Clinical Implementation
4.5. Limitations and Research Gaps
5. Conclusions and Future Applications
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Gottlieb, A.; Merola, J.F. Psoriatic Arthritis for Dermatologists. J. Dermatol. Treat. 2020, 31, 662–679. [Google Scholar] [CrossRef]
- Damiani, G.; Bragazzi, N.L.; Karimkhani Aksut, C.; Wu, D.; Alicandro, G.; McGonagle, D.; Guo, C.; Dellavalle, R.; Grada, A.; Wong, P.; et al. The Global, Regional, and National Burden of Psoriasis: Results and Insights From the Global Burden of Disease 2019 Study. Front. Med. 2021, 8, 743180. [Google Scholar] [CrossRef] [PubMed]
- Andres-Ejarque, R.; Ale, H.B.; Grys, K.; Tosi, I.; Solanky, S.; Ainali, C.; Catak, Z.; Sreeneebus, H.; Saklatvala, J.; Dand, N.; et al. Author Correction: Enhanced NF-ΚB Signaling in Type-2 Dendritic Cells at Baseline Predicts Non-Response to Adalimumab in Psoriasis. Nat. Commun. 2021, 12, 4741. [Google Scholar] [CrossRef] [PubMed]
- Elmets, C.A.; Lim, H.W.; Stoff, B.; Connor, C.; Cordoro, K.M.; Lebwohl, M.; Armstrong, A.W.; Davis, D.M.R.; Elewski, B.E.; Gelfand, J.M.; et al. Joint American Academy of Dermatology–National Psoriasis Foundation Guidelines of Care for the Management and Treatment of Psoriasis with Phototherapy. J. Am. Acad. Dermatol. 2019, 81, 775–804. [Google Scholar] [CrossRef] [PubMed]
- Menter, A.; Strober, B.E.; Kaplan, D.H.; Kivelevitch, D.; Prater, E.F.; Stoff, B.; Armstrong, A.W.; Connor, C.; Cordoro, K.M.; Davis, D.M.R.; et al. Joint AAD-NPF Guidelines of Care for the Management and Treatment of Psoriasis with Biologics. J. Am. Acad. Dermatol. 2019, 80, 1029–1072. [Google Scholar] [CrossRef]
- Mason, A.R.; Mason, J.; Cork, M.; Dooley, G.; Hancock, H. Topical Treatments for Chronic Plaque Psoriasis. Cochrane Database Syst. Rev. 2013, 2013, CD005028. [Google Scholar] [CrossRef]
- Stacey, S.K.; McEleney, M. Topical Corticosteroids: Choice and Application. Am. Fam. Physician 2021, 103, 337–343. [Google Scholar]
- Menter, A.; Korman, N.J.; Elmets, C.A.; Feldman, S.R.; Gelfand, J.M.; Gordon, K.B.; Gottlieb, A.B.; Koo, J.Y.M.; Lebwohl, M.; Lim, H.W.; et al. Guidelines of Care for the Management of Psoriasis and Psoriatic Arthritis: Section 4. Guidelines of Care for the Management and Treatment of Psoriasis with Traditional Systemic Agents. J. Am. Acad. Dermatol. 2009, 61, 451–485. [Google Scholar] [CrossRef]
- Warren, R.B.; Gooderham, M.; Burge, R.; Zhu, B.; Amato, D.; Liu, K.H.; Shrom, D.; Guo, J.; Brnabic, A.; Blauvelt, A. Comparison of Cumulative Clinical Benefits of Biologics for the Treatment of Psoriasis over 16 Weeks: Results from a Network Meta-Analysis. J. Am. Acad. Dermatol. 2020, 82, 1138–1149. [Google Scholar] [CrossRef]
- Egilman, A.C.; Kesselheim, A.S.; Avorn, J.; Raymakers, A.J.N.; Rome, B.N. Use of Efficiency Frontiers to Align Prices and Clinical Benefits of Biologic Therapies for Plaque Psoriasis. JAMA Dermatol. 2024, 160, 409–416. [Google Scholar] [CrossRef]
- Thai, S.; Zhuo, J.; Zhong, Y.; Xia, Q.; Chen, X.; Bao, Y.; Dhanda, D.; Priya, L.; Wu, J.J. Real-World Treatment Patterns and Healthcare Costs in Patients with Psoriasis Taking Systemic Oral or Biologic Therapies. J. Dermatol. Treat. 2023, 34, 2176708. [Google Scholar] [CrossRef]
- Doshi, J.A.; Takeshita, J.; Pinto, L.; Li, P.; Yu, X.; Rao, P.; Viswanathan, H.N.; Gelfand, J.M. Biologic Therapy Adherence, Discontinuation, Switching, and Restarting among Patients with Psoriasis in the US Medicare Population. J. Am. Acad. Dermatol. 2016, 74, 1057–1065.e4. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Kim, M. Challenges and Future Trends in the Treatment of Psoriasis. Int. J. Mol. Sci. 2023, 24, 13313. [Google Scholar] [CrossRef]
- Farhan, M. The Promising Role of Polyphenols in Skin Disorders. Molecules 2024, 29, 865. [Google Scholar] [CrossRef] [PubMed]
- Yahfoufi, N.; Alsadi, N.; Jambi, M.; Matar, C. The Immunomodulatory and Anti-Inflammatory Role of Polyphenols. Nutrients 2018, 10, 1618. [Google Scholar] [CrossRef] [PubMed]
- Vladu, A.F.; Ficai, D.; Ene, A.G.; Ficai, A. Combination Therapy Using Polyphenols: An Efficient Way to Improve Antitumoral Activity and Reduce Resistance. Int. J. Mol. Sci. 2022, 23, 10244. [Google Scholar] [CrossRef]
- Shahi, A.; Afzali, S.; Amirzargar, A.; Mohaghegh, P.; Salehi, S.; Mansoori, Y. Potential Roles of Inflammasomes in the Pathophysiology of Psoriasis: A Comprehensive Review. Mol. Immunol. 2023, 161, 44–60. [Google Scholar] [CrossRef]
- Katsimbri, P.; Korakas, E.; Kountouri, A.; Ikonomidis, I.; Tsougos, E.; Vlachos, D.; Papadavid, E.; Raptis, A.; Lambadiari, V. The Effect of Antioxidant and Anti-Inflammatory Capacity of Diet on Psoriasis and Psoriatic Arthritis Phenotype: Nutrition as Therapeutic Tool? Antioxidants 2021, 10, 157. [Google Scholar] [CrossRef]
- Delgado, A.; Gonçalves, S.; Romano, A. Mediterranean Diet: The Role of Phenolic Compounds from Aromatic Plant Foods. Foods 2023, 12, 840. [Google Scholar] [CrossRef]
- García-Pérez, M.E.; Kasangana, P.B.; Stevanovic, T. Bioactive Polyphenols for Diabetes and Inflammation in Psoriasis Disease. Stud. Nat. Prod. Chem. 2017, 52, 231–268. [Google Scholar] [CrossRef]
- Gupta, S.C.; Tyagi, A.K.; Deshmukh-Taskar, P.; Hinojosa, M.; Prasad, S.; Aggarwal, B.B. Downregulation of Tumor Necrosis Factor and Other Proinflammatory Biomarkers by Polyphenols. Arch. Biochem. Biophys. 2014, 559, 91–99. [Google Scholar] [CrossRef] [PubMed]
- García-Pérez, M.E.; Allaeys, I.; Rusu, D.; Pouliot, R.; Janezic, T.S.; Poubelle, P.E. Picea Mariana Polyphenolic Extract Inhibits Phlogogenic Mediators Produced by TNF-α-Activated Psoriatic Keratinocytes: Impact on NF-ΚB Pathway. J. Ethnopharmacol. 2014, 151, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Parmar, K.M.; Itankar, P.R.; Joshi, A.; Prasad, S.K. Anti-Psoriatic Potential of Solanum xanthocarpum Stem in Imiquimod-Induced Psoriatic Mice Model. J. Ethnopharmacol. 2017, 198, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.; Li, B.; Luo, L.; Jiang, W.; Lu, Q.; Rong, M.; Lai, R. Curcumin Shows Excellent Therapeutic Effect on Psoriasis in Mouse Model. Biochimie 2016, 123, 73–80. [Google Scholar] [CrossRef]
- Yang, Z.; Yin, X.; Chen, C.; Huang, S.; Li, X.; Yan, J.; Sun, Q. CircOAS3 Regulates Keratinocyte Proliferation and Psoriatic Inflammation by Interacting with Hsc70 via the JNK/STAT3/NF-ΚB Signaling Pathway. Inflammation 2022, 45, 1924–1935. [Google Scholar] [CrossRef]
- Nowak-Perlak, M.; Szpadel, K.; Jabłońska, I.; Pizon, M.; Woźniak, M. Promising Strategies in Plant-Derived Treatments of Psoriasis-Update of In Vitro, In Vivo, and Clinical Trials Studies. Molecules 2022, 27, 591. [Google Scholar] [CrossRef]
- Kimmel, G.W.; Lebwohl, M. Psoriasis: Overview and Diagnosis. In Evidence-Based Psoriasis; Bhutani, T., Liao, W., Nakamura, M., Eds.; Springer: Cham, Switzerland, 2018; pp. 1–16. [Google Scholar] [CrossRef]
- Gisondi, P.; Bellinato, F.; Girolomoni, G. Topographic Differential Diagnosis of Chronic Plaque Psoriasis: Challenges and Tricks. J. Clin. Med. 2020, 9, 3594. [Google Scholar] [CrossRef]
- Roy, B.; Simard, M.; Lorthois, I.; Bélanger, A.; Maheux, M.; Duque-Fernandez, A.; Rioux, G.; Simard, P.; Deslauriers, M.; Masson, L.C.; et al. In Vitro Models of Psoriasis. In Skin Tissue Models; Academic Press: Cambridge, MA, USA, 2018; pp. 103–128. [Google Scholar] [CrossRef]
- Xie, J.; Huang, S.; Huang, H.; Deng, X.; Yue, P.; Lin, J.; Yang, M.; Han, L.; Zhang, D.K. Advances in the Application of Natural Products and the Novel Drug Delivery Systems for Psoriasis. Front. Pharmacol. 2021, 12, 644952. [Google Scholar] [CrossRef]
- Rahman, M.; Alam, K.; Ahmad, M.Z.; Gupta, G.; Afzal, M.; Akhter, S.; Kazmi, I.; Jyoti; Ahmad, F.J.; Anwar, F. Classical to Current Approach for Treatment of Psoriasis: A Review. Endocr. Metab. Immune Disord. Drug Targets 2012, 12, 287–302. [Google Scholar] [CrossRef]
- Elkhawaga, O.Y.; Ellety, M.M.; Mofty, S.O.; Ghanem, M.S.; Mohamed, A.O. Review of Natural Compounds for Potential Psoriasis Treatment. Inflammopharmacology 2023, 31, 1183–1198. [Google Scholar] [CrossRef]
- Čižmárová, B.; Hubková, B.; Tomečková, V.; Birková, A. Flavonoids as Promising Natural Compounds in the Prevention and Treatment of Selected Skin Diseases. Int. J. Mol. Sci. 2023, 24, 6324. [Google Scholar] [CrossRef]
- Durazzo, A.; Lucarini, M.; Souto, E.B.; Cicala, C.; Caiazzo, E.; Izzo, A.A.; Novellino, E.; Santini, A. Polyphenols: A Concise Overview on the Chemistry, Occurrence, and Human Health. Phytother. Res. 2019, 33, 2221–2243. [Google Scholar] [CrossRef] [PubMed]
- Molino, S.; Pilar Francino, M.; Ángel Rufián Henares, J. Why Is It Important to Understand the Nature and Chemistry of Tannins to Exploit Their Potential as Nutraceuticals? Food Res. Int. 2023, 173, 113329. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, 71. [Google Scholar] [CrossRef] [PubMed]
- van der Fits, L.; Mourits, S.; Voerman, J.S.A.; Kant, M.; Boon, L.; Laman, J.D.; Cornelissen, F.; Mus, A.-M.; Florencia, E.; Prens, E.P.; et al. Imiquimod-Induced Psoriasis-Like Skin Inflammation in Mice Is Mediated via the IL-23/IL-17 Axis. J. Immunol. 2009, 182, 5836–5845. [Google Scholar] [CrossRef] [PubMed]
- Antiga, E.; Bonciolini, V.; Volpi, W.; Del Bianco, E.; Caproni, M. Oral Curcumin (Meriva) Is Effective as an Adjuvant Treatment and Is Able to Reduce IL-22 Serum Levels in Patients with Psoriasis Vulgaris. Biomed. Res. Int. 2015, 2015, 283634. [Google Scholar] [CrossRef]
- Bahraini, P.; Rajabi, M.; Mansouri, P.; Sarafian, G.; Chalangari, R.; Azizian, Z. Turmeric Tonic as a Treatment in Scalp Psoriasis: A Randomized Placebo-Control Clinical Trial. J. Cosmet. Dermatol. 2018, 17, 461–466. [Google Scholar] [CrossRef]
- Sun, J.; Zhao, Y.; Jin, H.; Hu, J. Curcumin Relieves TPA-Induced Th1 Inflammation in K14-VEGF Transgenic Mice. Int. Immunopharmacol. 2015, 25, 235–241. [Google Scholar] [CrossRef]
- Skyvalidas, D.; Mavropoulos, A.; Tsiogkas, S.; Dardiotis, E.; Liaskos, C.; Mamuris, Z.; Roussaki-Schulze, A.; Sakkas, L.I.; Zafiriou, E.; Bogdanos, D.P. Curcumin Mediates Attenuation of Pro-Inflammatory Interferon γ and Interleukin 17 Cytokine Responses in Psoriatic Disease, Strengthening Its Role as a Dietary Immunosuppressant. Nutr. Res. 2020, 75, 95–108. [Google Scholar] [CrossRef]
- Varma, S.R.; Sivaprakasam, T.O.; Mishra, A.; Prabhu, S.; Rafiq, M.; Rangesh, P. Imiquimod-Induced Psoriasis-like Inflammation in Differentiated Human Keratinocytes: Its Evaluation Using Curcumin. Eur. J. Pharmacol. 2017, 813, 33–41. [Google Scholar] [CrossRef]
- Zhang, J.; Ma, Y.; Li, W. Curcumin Reduces Inflammation in Mice with the Psoriasis Model by Inhibiting NLRP3 Inflammatory Bodies. Cell. Mol. Biol. 2021, 67, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Saelee, C.; Thongrakard, V.; Tencomnao, T. Effects of Thai Medicinal Herb Extracts with Anti-Psoriatic Activity on the Expression on NF-ΚB Signaling Biomarkers in HaCaT Keratinocytes. Molecules 2011, 16, 3908–3932. [Google Scholar] [CrossRef]
- Kjær, T.N.; Thorsen, K.; Jessen, N.; Stenderup, K.; Pedersen, S.B. Resveratrol Ameliorates Imiquimod-Induced Psoriasis-like Skin Inflammation in Mice. PLoS ONE 2015, 10, e0126599. [Google Scholar] [CrossRef] [PubMed]
- Vijayalakshmi, A.; Ravichandiran, V.; Velraj, M.; Nirmala, S.; Jayakumari, S. Screening of Flavonoid “Quercetin” from the Rhizome of Smilax China Linn. for Anti–Psoriatic Activity. Asian Pac. J. Trop. Biomed. 2012, 2, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Herrera Acosta, E.; Alonso Suárez Pérez, J.; Aguilera Arjona, J.; Visioli, F. An Olive Polyphenol-Based Nutraceutical Improves Cutaneous Manifestations of Psoriasis in Humans. PharmaNutrition 2016, 4, 151–153. [Google Scholar] [CrossRef]
- Starbek Zorko, M.; Štrukelj, B.; Švajger, U.; Kreft, S.; Lunder, T. Efficacy of a Polyphenolic Extract from Silver Fir (Abies alba) Bark on Psoriasis: A Randomised, Double-Blind, Placebo-Controlled Trial. Pharmazie 2018, 73, 56–60. [Google Scholar] [CrossRef]
- An, J.; Li, Z.; Dong, Y.; Ren, J.; Huo, J. Amentoflavone Protects against Psoriasis-like Skin Lesion through Suppression of NF-ΚB-Mediated Inflammation and Keratinocyte Proliferation. Mol. Cell. Biochem. 2016, 413, 87–95. [Google Scholar] [CrossRef]
- Di, T.T.; Ruan, Z.T.; Zhao, J.X.; Wang, Y.; Liu, X.; Wang, Y.; Li, P. Astilbin Inhibits Th17 Cell Differentiation and Ameliorates Imiquimod-Induced Psoriasis-like Skin Lesions in BALB/c Mice via Jak3/Stat3 Signaling Pathway. Int. Immunopharmacol. 2016, 32, 32–38. [Google Scholar] [CrossRef]
- Wu, J.; Li, H.; Li, M. Effects of Baicalin Cream in Two Mouse Models: 2,4-Dinitrofluorobenzene-Induced Contact Hypersensitivity and Mouse Tail Test for Psoriasis. Int. J. Clin. Exp. Med. 2015, 8, 2128. [Google Scholar]
- Chamcheu, J.C.; Pal, H.C.; Siddiqui, I.A.; Adhami, V.M.; Ayehunie, S.; Boylan, B.T.; Noubissi, F.K.; Khan, N.; Syed, D.N.; Elmets, C.A.; et al. Prodifferentiation, Anti-Inflammatory and Antiproliferative Effects of Delphinidin, a Dietary Anthocyanidin, in a Full-Thickness Three-Dimensional Reconstituted Human Skin Model of Psoriasis. Skin Pharmacol. Physiol. 2015, 28, 177–188. [Google Scholar] [CrossRef]
- Weng, Z.; Patel, A.B.; Vasiadi, M.; Therianou, A.; Theoharides, T.C. Luteolin Inhibits Human Keratinocyte Activation and Decreases NF-ΚB Induction That Is Increased in Psoriatic Skin. PLoS ONE 2014, 9, e90739. [Google Scholar] [CrossRef]
- Zhang, M.; Li, N.; Cai, R.; Gu, J.; Xie, F.; Wei, H.; Lu, C.; Wu, D. Rosmarinic Acid Protects Mice from Imiquimod Induced Psoriasis-like Skin Lesions by Inhibiting the IL-23/Th17 Axis via Regulating Jak2/Stat3 Signaling Pathway. Phytother. Res. 2021, 35, 4526–4537. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, X.; Mei, L.; Wang, H.; Fang, F. Epigallocatechin-3-Gallate (EGCG) Inhibits Imiquimod-Induced Psoriasis-like Inflammation of BALB/c Mice. BMC Complement. Altern. Med. 2016, 16, 334. [Google Scholar] [CrossRef] [PubMed]
- Tsiogkas, S.G.; Apostolopoulou, K.; Mavropoulos, A.; Grammatikopoulou, M.G.; Dardiotis, E.; Zafiriou, E.; Bogdanos, D.P. Gallic Acid Diminishes Pro-Inflammatory Interferon-γ- and Interleukin-17-Producing Sub-Populations in Vitro in Patients with Psoriasis. Immunol. Res. 2023, 71, 475–487. [Google Scholar] [CrossRef] [PubMed]
- Belcaro, G.; Luzzi, R.; Hu, S.; Cesarone, M.R.; Dugall, M.; Ippolito, E.; Corsi, M.; Caporale, S. Improvement in Signs and Symptoms in Psoriasis Patients with Pycnogenol® Supplementation. Panminerva Med. 2014, 56, 41–48. [Google Scholar] [PubMed]
- García-Pérez, M.E.; Royer, M.; Duque-Fernandez, A.; Diouf, P.N.; Stevanovic, T.; Pouliot, R. Antioxidant, Toxicological and Antiproliferative Properties of Canadian Polyphenolic Extracts on Normal and Psoriatic Keratinocytes. J. Ethnopharmacol. 2010, 132, 251–258. [Google Scholar] [CrossRef]
- Rendon, A.; Schäkel, K. Psoriasis Pathogenesis and Treatment. Int. J. Mol. Sci. 2019, 20, 1475. [Google Scholar] [CrossRef]
- Griffiths, C.E.M.; Armstrong, A.W.; Gudjonsson, J.E.; Barker, J.N.W.N. Psoriasis. Lancet 2021, 397, 1301–1315. [Google Scholar] [CrossRef]
- Pal, H.C.; Chamcheu, J.C.; Adhami, V.M.; Wood, G.S.; Elmets, C.A.; Mukhtar, H.; Afaq, F. Topical Application of Delphinidin Reduces Psoriasiform Lesions in the Flaky Skin Mouse Model by Inducing Epidermal Differentiation and Inhibiting Inflammation. Br. J. Dermatol. 2014, 172, 354. [Google Scholar] [CrossRef]
- Pleńkowska, J.; Gabig-Cimińska, M.; Mozolewski, P. Oxidative Stress as an Important Contributor to the Pathogenesis of Psoriasis. Int. J. Mol. Sci. 2020, 21, 6206. [Google Scholar] [CrossRef]
- Choi, D.I.; Park, J.H.; Choi, J.Y.; Piao, M.S.; Suh, M.S.; Lee, J.B.; Yun, S.J.; Lee, S.C. Keratinocytes-Derived Reactive Oxygen Species Play an Active Role to Induce Type 2 Inflammation of the Skin: A Pathogenic Role of Reactive Oxygen Species at the Early Phase of Atopic Dermatitis. Ann. Dermatol. 2020, 33, 26. [Google Scholar] [CrossRef] [PubMed]
- Na, H.K.; Surh, Y.J. Modulation of Nrf2-Mediated Antioxidant and Detoxifying Enzyme Induction by the Green Tea Polyphenol EGCG. Food Chem. Toxicol. 2008, 46, 1271–1278. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Jiao, Y.; Xue, J.; Zhang, Q.; Yang, H.; Xing, L.; Chen, G.; Wu, J.; Zhang, S.; Zhu, W.; et al. Metformin Sensitizes Non-Small Cell Lung Cancer Cells to an Epigallocatechin-3-Gallate (EGCG) Treatment by Suppressing the Nrf2/HO-1 Signaling Pathway. Int. J. Biol. Sci. 2017, 13, 1560–1569. [Google Scholar] [CrossRef] [PubMed]
- Chamcheu, J.C.; Siddiqui, I.A.; Adhami, V.M.; Esnault, S.; Bharali, D.J.; Babatunde, A.S.; Adame, S.; Massey, R.J.; Wood, G.S.; Longley, B.J.; et al. Chitosan-Based Nanoformulated (−)-Epigallocatechin-3-Gallate (EGCG) Modulates Human Keratinocyte-Induced Responses and Alleviates Imiquimod-Induced Murine Psoriasiform Dermatitis. Int. J. Nanomed. 2018, 13, 4189–4206. [Google Scholar] [CrossRef]
- Hadidi, M.; Liñán-Atero, R.; Tarahi, M.; Christodoulou, M.C.; Aghababaei, F. The Potential Health Benefits of Gallic Acid: Therapeutic and Food Applications. Antioxidants 2024, 13, 1001. [Google Scholar] [CrossRef]
- Shojaee, M.S.; Moeenfard, M.; Farhoosh, R. Kinetics and Stoichiometry of Gallic Acid and Methyl Gallate in Scavenging DPPH Radical as Affected by the Reaction Solvent. Sci. Rep. 2022, 12, 8765. [Google Scholar] [CrossRef]
- Yang, D.J.; Moh, S.H.; Son, D.H.; You, S.; Kinyua, A.W.; Ko, C.M.; Song, M.; Yeo, J.; Choi, Y.H.; Kim, K.W. Gallic Acid Promotes Wound Healing in Normal and Hyperglucidic Conditions. Molecules 2016, 21, 899. [Google Scholar] [CrossRef]
- Geana, E.I.; Ciucure, C.T.; Tamaian, R.; Marinas, I.C.; Gaboreanu, D.M.; Stan, M.; Chitescu, C.L. Antioxidant and Wound Healing Bioactive Potential of Extracts Obtained from Bark and Needles of Softwood Species. Antioxidants 2023, 12, 1383. [Google Scholar] [CrossRef]
- Cas, M.D.; Ghidoni, R. Dietary Curcumin: Correlation between Bioavailability and Health Potential. Nutrients 2019, 11, 2147. [Google Scholar] [CrossRef]
- Yang, B.; Dong, Y.; Wang, F.; Zhang, Y. Nanoformulations to Enhance the Bioavailability and Physiological Functions of Polyphenols. Molecules 2020, 25, 4613. [Google Scholar] [CrossRef]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and Other Phenolic Compounds from Medicinal Plants for Pharmaceutical and Medical Aspects: An Overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef] [PubMed]
- Zillich, O.V.; Schweiggert-Weisz, U.; Eisner, P.; Kerscher, M. Polyphenols as Active Ingredients for Cosmetic Products. Int. J. Cosmet. Sci. 2015, 37, 455–464. [Google Scholar] [CrossRef] [PubMed]
| First Author and Year | Study Design | Sample | Polyphenol | Main Results |
|---|---|---|---|---|
| Antiga, 2015 [38] | RCT (level 1) | 63 psoriasis patients (PASI < 10) | Curcumin | Significant ↓ PASI score and ↓ IL-22 serum levels. |
| Bahraini, 2018 [39] | RCT (level 1) | 40 patients (mild-moderate scalp psoriasis) | Curcumin | Significant ↓ erythema, scaling, and induration vs. placebo (p < 0.05). |
| Kang, 2016 [24] | Animal study (level 6) | K14-VEGF transgenic mice (3 months) | Curcumin | ↓ All psoriasis indices and normalized TNF-alpha, IFN-gamma, IL-2, IL-12, IL-22, IL-23 levels. |
| Sun, 2015 [40] | Animal study | K14-VEGF transgenic mice (8–15 weeks) | Curcumin | Inhibited TPA-induced psoriasis-like inflammation. |
| Skyvalidas, 2020 [41] | In vitro study | T cells from 34 psoriasis patients | Curcumin | ↓ Pro-inflammatory IFN-gamma and IL-17 production at higher concentrations. |
| Kang, 2016 [24] | In vitro study | T cells | Curcumin | ↓ IL-17, IL-22, IFN-gamma, IL-2, IL-8, TNF-alpha secretion by 30–60% (10 µM). |
| Varma, 2017 [42] | In vitro study | IMQ-induced HaCaT cells | Curcumin | ↓ Cell proliferation and ↓ IL-17, TNF-alpha, IFN-gamma, IL-6 (25–50 uM). |
| Zhang, 2021 [43] | Animal study | 20 BALB/c mice (8–12 weeks, female) | Curcumin | ↓ NLRP3 expression, ↓ IL-22/IL-18 inflammation by 47%. |
| Saelee, 2011 [44] | In vitro study | HaCaT cells | Curcumin | ↓ CSF-1, IL-8, NF-kappaB2, NF-kappaB1, RelA expression. |
| Kjaer, 2015 [45] | Animal study (level 6) | 28 male BALB/c mice (6–7 weeks) | Resveratrol | ↓ NF-kappaB and ↓ IL-19, IL-17A, IL-17F gene expression in IMQ-induced psoriasis. |
| Vijayalakshmi, 2012 [46] | Animal study (level 6) | Male albino mice (25–30 g) | Quercetin | Antiproliferative activity in keratinocytes, ↓ leukocyte migration (p < 0.001), ↑ orthokeratotic regions and epidermal thickness. |
| Herrera Acosta, 2016 [47] | RCT (level 1) | 30 psoriasis patients (mild-moderate) | Olive polyphenols | ↓ Cutaneous manifestations in moderate psoriasis. PASI ↓ 27% (4.0 to 2.92 ± 1.52) after 4 weeks. |
| Zorko, 2018 [48] | RCT (level 1) | 61 patients (mild psoriasis) | Silver fir polyphenols | 40% improvement rate in elbow psoriasis. |
| García-Pérez, 2014 [22] | In vitro study | NHK and PK | Black spruce polyphenols | BS-EAc ↓ NF-kappaB pathway in TNF-alpha-activated psoriatic keratinocytes. |
| An, 2016 [49] | Animal study (level 6) | 40 male BALB/c mice (6–7 weeks) | AMF | AMF ↓ IMQ-induced TNF-alpha, IL-17A, IL-22, IL-23 serum levels. |
| Di, 2016 [50] | Animal and in vitro study (level 6) | BALB/c mice (8 weeks, 18–20 g) and Th17 cells | Astilbin | ↓ IMQ-induced keratinocyte proliferation, ↓ CD3+ infiltration, ↓ CD4+/CD8+ T cells, ↓ IL-17A, TNF-alpha, IL-6, IFN-gamma, IL-2. |
| Wu, 2015 [51] | Animal study (level 6) | 46 female BALB/c mice (6–8 weeks) and 48 male ICR mice (22–25 g) | Baicalin | 5% baicalin showed anti-inflammatory action in CHS response. |
| Chamcheu, 2015 [52] | In vitro study | 3D PSE model | Delphinidin | ↑ Cornification and differentiation markers, ↓ inflammation in PSE. |
| Weng, 2014 [53] | In vitro study | HaCaT and NHK | Luteolin | ↓ IL-6, IL-8, TNF-alpha, VEGF and ↓ TNF-alpha-induced NF-kappaB activation. |
| Parmar, 2017 [23] | Animal study | Swiss Albino mice (20–30 g) | Solanum polyphenols | Anti-psoriatic activity (topical 10%, oral 200–400 mg/kg), ↓ TNF-alpha, IL-1beta, IL-6, IL-17. |
| Zhang, 2021 [54] | Animal study (level 6) | BALB/c male mice (7–9 weeks, 20 ± 2 g) | Rosmarinic acid | ↓ IL-23 expression, ↓ Th17 differentiation, ↓ IL-17A production. |
| Zhang, 2016 [55] | Animal study (level 6) | BALB/c mice (6–8 weeks, 20–25 g) | EGCG | ↓ Epidermal PCNA, ↓ T cell infiltration, ↓ IL-17A/F, IL-22, IL-23, ↑ SOD/catalase. |
| Tsiogkas, 2023 [56] | In vitro study | 28 patients (moderate-severe psoriasis, PASI > 7) | Gallic acid | ↓ Pro-inflammatory IFN-gamma and IL-17-producing cell populations. |
| Belcaro, 2014 [57] | Observational study | Patients (moderate-severe psoriasis, 30–45 years) | Pycnogenol® | ↓ Affected areas, erythema, desquamation (p < 0.05), ↑ skin moisture, 30% shorter treatment time. |
| García-Pérez, 2010 [58] | In vitro study | NHK and PK | Tree bark polyphenols | Black spruce: highest antioxidant capacity, less toxic on NHK, inhibited NHK/non-lesional PK growth. |
| Compound | Effective Concentration | Clinical/Topical Dose | Route |
|---|---|---|---|
| Curcumin | 10–50 µM | 1000–2000 mg daily | Oral/Topical |
| EGCG | 30 mg/mL (3% topical) | 300 mg/kg/d, 150 mg/kg/d | Topical/Oral |
| Quercetin | 62.42 ± 10.20 µg/mL (IC50) | 25–50 mg/kg topical | Topical |
| Resveratrol | 400 mg/kg/day | 400 mg/kg daily | Oral |
| Baicalin | 1–5% cream | 5% cream optimal | Topical |
| Olive Polyphenols | 500 mg extract + vitamins | 1000 mg daily (2 capsules) | Oral |
| Silver Fir | 1–25 µg/mL (in vitro) | 2% in topical ointment | Topical |
| Black Spruce | 100–500 µg/mL (optimal at 500) | Ethyl acetate fraction | Experimental/Topical |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alalwan, T.A.; Aksal, R.; Allehdan, S.; Rondanelli, M.; Perna, S. Effect of (Poly)phenols as Potential Agents in the Treatment of Psoriasis: A Systematic Review of the Evidence and Future Applications. Nutraceuticals 2025, 5, 24. https://doi.org/10.3390/nutraceuticals5030024
Alalwan TA, Aksal R, Allehdan S, Rondanelli M, Perna S. Effect of (Poly)phenols as Potential Agents in the Treatment of Psoriasis: A Systematic Review of the Evidence and Future Applications. Nutraceuticals. 2025; 5(3):24. https://doi.org/10.3390/nutraceuticals5030024
Chicago/Turabian StyleAlalwan, Tariq A., Rojbin Aksal, Sabika Allehdan, Mariangela Rondanelli, and Simone Perna. 2025. "Effect of (Poly)phenols as Potential Agents in the Treatment of Psoriasis: A Systematic Review of the Evidence and Future Applications" Nutraceuticals 5, no. 3: 24. https://doi.org/10.3390/nutraceuticals5030024
APA StyleAlalwan, T. A., Aksal, R., Allehdan, S., Rondanelli, M., & Perna, S. (2025). Effect of (Poly)phenols as Potential Agents in the Treatment of Psoriasis: A Systematic Review of the Evidence and Future Applications. Nutraceuticals, 5(3), 24. https://doi.org/10.3390/nutraceuticals5030024






