Associations between Physical Activity and Health Parameters in Adolescent Pupils in Egypt
Abstract
:1. Introduction
1.1. Aim of the Study
- Describe a range of anthropometric (weight, height, body mass index and body fat) and physiological (cholesterol level, systolic blood pressure, diastolic blood pressure and heart rate) parameters of a sample of Egyptian secondary school pupils;
- Assess the association between the PA intervention and pupils’ anthropometric parameters; and,
- Explore the association between the PA intervention and pupils’ physiological parameters.
2. Methods
2.1. Procedures and Participants
2.2. PA Intervention
2.3. Data Collection Tools
2.4. Data Analysis
3. Results
Were the two groups (intervention and control) similar at baseline?
Were the two groups (intervention and control) similar after 3 months?
Were the control pupils similar at baseline and after 3 months?
Were the intervention pupils similar at baseline and after 3 months?
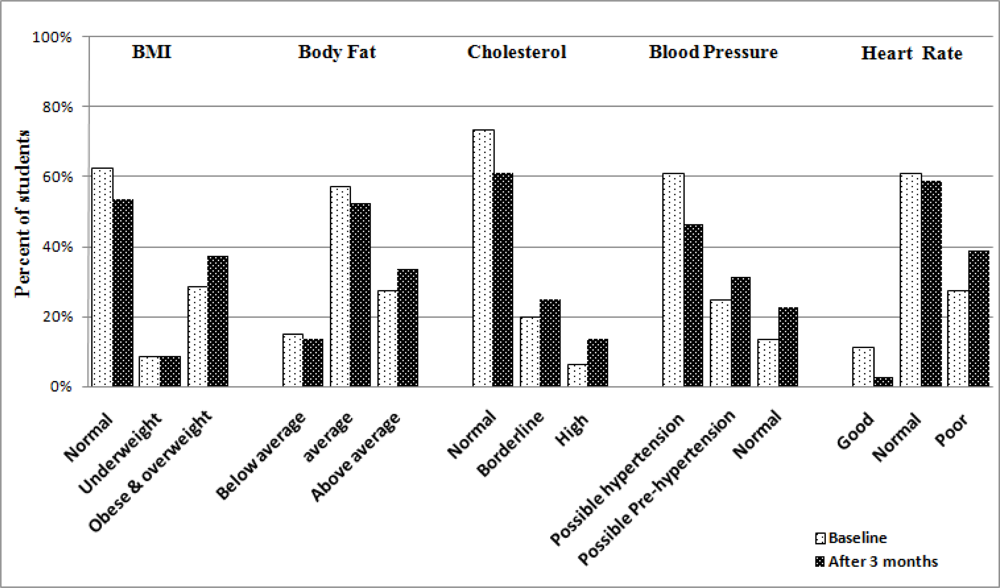
Intervention and control pupils’ health parameters after 3 months
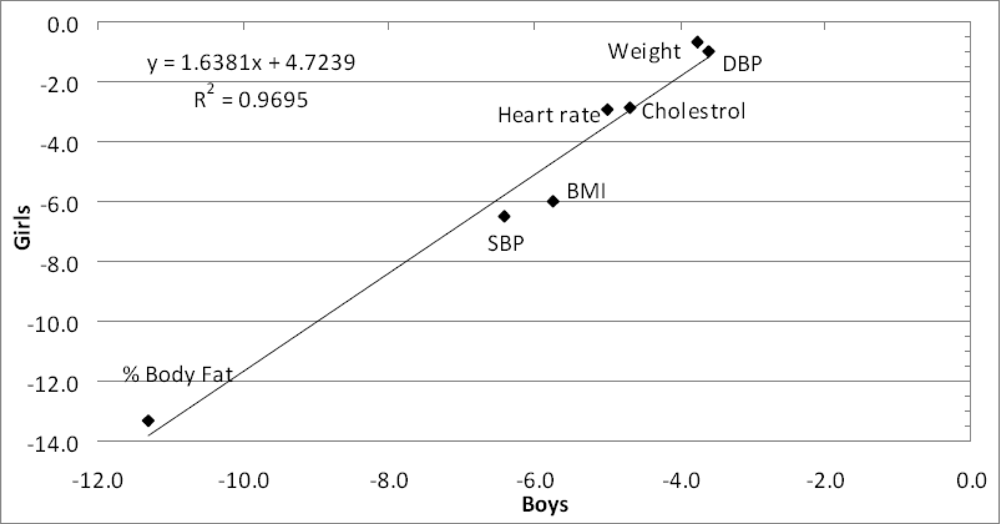
Were the actual mean changes on each of the seven parameters similar for boys and girls?
4. Discussion
Acknowledgments
References
- World Health Organization, Available online: https://apps.who.int/infobase/report.aspx (accessed on August 21, 2009).
- .
- El-Zanaty, F; Way, A. Egypt Demographic and Health Survey 2005; Cairo, Ministry of Health and Population, National Population Council, El-Zanaty and Associates, and ORC Macro: Cairo, Egypt, 2006. [Google Scholar]
- Ashour, Z; Ibrahim, MM; Appel, LJ; Ibrahim, AS; Whelton, PK. The Egyptian National Hypertension Project (NHP). Design and rationale. The NHP Investigative Team. Hypertension 1995, 26, 880–885. [Google Scholar]
- Ekvall, VK. Pediatric Nutrition in Chronic Diseases and Developmental Disorders: Prevention, Assessment, and Treatment, 2nd ed; Oxford University Press: New York, NY, USA, 2005. [Google Scholar]
- Fuster, V; Topol, EJ; Nabel, EG. Atherothrombosis and Coronary Artery Disease; Lippincott Williams & Wilkins: New York, NY, USA, 2005. [Google Scholar]
- Hedley, AA; Ogden, CL; Johnson, CL; Carroll, MD; Curtin, LR; Flegal, KM. Prevalence of overweight and obesity among US children, adolescents, and adults, 1999–2002. JAMA 2004, 291, 2847–2850. [Google Scholar]
- Centers for Disease Control and Prevention.
- Chen, JL; Wu, Y. Cardiovascular risk factors in Chinese American children: associations between overweight, acculturation, and physical activity. J. Pediatr. Health Care 2008, 22, 103–110. [Google Scholar]
- Al-Sendi, AM; Shetty, P; Musaiger, AO; Myatt, M. Relationship between body composition and blood pressure in Bahraini adolescents. Br. J. Nutr 2003, 90, 837–844. [Google Scholar]
- Lauer, RM; Clarke, WR; Mahoney, LT; Witt, J. Childhood predictors for high adult blood pressure. The Muscatine Study. Pediatr Clin North Am 1993, 40, 23–40. [Google Scholar]
- Field, AE; Cook, NR; Gillman, MW. Weight status in childhood as a predictor of becoming overweight or hypertensive in early adulthood. Obes. Res 2005, 13, 163–169. [Google Scholar]
- American College of Sports Medicine; Durstine, JL. ACSM’s Resource Manual for Guidelines for Exercise Testing And Prescription, 2nd ed; Lea & Febiger: Philadelphia, PA, USA, 1993. [Google Scholar]
- Dobbins, M; De Corby, K; Robeson, P; Husson, H; Tirilis, D. School-based physical activity programs for promoting physical activity and fitness in children and adolescents aged 6–18. Cochrane Database Syst Rev 2009, 1, CD007651. [Google Scholar]
- Motlagh, ME; Kelishadi, R; Ardalan, G; Gheiratmand, R; Majdzadeh, R; Heidarzadeh, A. Rationale, methods and first results of the Iranian national programme for prevention of chronic diseases from childhood: CASPIAN Study. East. Mediterr. Health J 2009, 15, 302–314. [Google Scholar]
- Jackson, RT; Rashed, M; Al-Hamad, N; Hwalla, N; Al-Somaie, M. Comparison of BMI-for-age in adolescent girls in 3 countries of the Eastern Mediterranean Region. East. Mediterr. Health J 2007, 13, 430–440. [Google Scholar]
- Gharib, NM; Shah, P. Anthropometry and body composition of school children in Bahrain. Ann. Saudi. Med 2009, 29, 258–269. [Google Scholar]
- Saleh, EA; Mahfouz, AA; Tayel, KY; Naguib, MK; Bin-al-Shaikh, NM. Hypertension and its determinants among primary-school children in Kuwait: an epidemiological study. East. Mediterr. Health J 2000, 6, 333–337. [Google Scholar]
- Keller, K. Encyclopedia of Obesity, 1st ed; SAGE: Newbury Park, CA, USA, 2008. [Google Scholar]
- Goran, MI; Sothern, M. Handbook of Pediatric Obesity: Etiology, Pathophysiology, and Prevention; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Esteghamati, A; Khalilzadeh, O; Rashidi, A; Meysamie, A; Haghazali, M; Asgari, F; Abbasi, M; Rastad, S; Gouya, M. M. Association between physical activity and insulin resistance in Iranian adults: National Surveillance of Risk Factors of Non-Communicable Diseases (SuRFNCD-2007). Prev. Med 2009, 49, 402–406. [Google Scholar]
- Drukteinis, JS; Roman, MJ; Fabsitz, RR; Lee, ET; Best, LG; Russell, M; Devereux, RB. Cardiac and systemic hemodynamic characteristics of hypertension and prehypertension in adolescents and young adults: the Strong Heart Study. Circulation 2007, 115, 221–227. [Google Scholar]
- Izdebska, E; Izdebski, J; Cybulska, I; Makowiecka-Ciesla, M; Trzebski, A. Moderate exercise training reduces arterial chemoreceptor reflex drive in mild hypertension. J. Physiol. Pharmacol 2006, 57, 93–102. [Google Scholar]
- Reinehr, T; de Sousa, G; Toschke, AM; Andler, W. Long-term follow-up of cardiovascular disease risk factors in children after an obesity intervention. Am. J. Clin. Nutr 2006, 84, 490–496. [Google Scholar]
- Sobieszczańska, M; Kałka, DPW; Adamus, J. Physical activity in basic and primary prevention of cardiovascular disease. Pol. Merkur. Lekarski j 2009, 26, 659–664. [Google Scholar]
- World Health Organization, Available online: https://apps.who.int/bmi/index.jsp?introPage=intro_3.html (accessed on August 20, 2009).
- Morris, M. Mental Health for Primary Care: A Practical Guide for Non-Specialists; Radcliffe Publishing: Oxford, UK, 2009. [Google Scholar]
- Korbonits, M. Obesity and Metabolism; Karger Publishers: London, UK, 2008. [Google Scholar]
- Mullen, MC; Shield, JE; Association, AD. Childhood and Adolescent Overweight: The Health Professional’s Guide to Identification, Treatment, and Prevention; American Dietetic Association: Chicago, IL, USA, 2004. [Google Scholar]
- Duggan, C; Watkins, JB; Walker, WA. Nutrition in Pediatrics; PMPH-USA: Hamilton, Canada, 2008. [Google Scholar]
- Roberts, CC; Levick, JR; Stanton, AW; Mortimer, PS. Assessment of truncal edema following breast cancer treatment using modified Harpenden skinfold calipers. Lymphology 1995, 28, 78–88. [Google Scholar]
- Durnin, JV; de Bruin, H; Feunekes, GI. Skinfold thicknesses: is there a need to be very precise in their location? Br. J. Nutr 1997, 77, 3–7. [Google Scholar]
- Baechle, TR; Earle, RW. Essentials of Strength Training and Conditioning; Human Kinetics: Champaign, IL, USA, 2008. [Google Scholar]
- Maud, PJ; Foster, C. Physiological Assessment of Human Fitness; Human Kinetics: Champaign, IL, USA, 2006. [Google Scholar]
- Acevedo, EO; Starks, MA. Exercise Testing and Prescription Lab Manual; Human Kinetics: Champaign, IL, USA, 2003. [Google Scholar]
- Taylor, JR; Lopez, LM. Cholesterol: point-of-care testing. Ann. Pharmacother 2004, 38, 1252–1257. [Google Scholar]
- Christofaro, DG; Fernandes, RA; Gerage, AM; Alves, MJ; Polito, MD; Oliveira, AR. Validation of the Omron HEM 742 blood pressure monitoring device in adolescents. Arq. Bras. Cardiol 2009, 92, 10–15. [Google Scholar]
- National Heart & Lung and Blood Institute.
- Craig, SR; Amin, RV; Russell, DW; Paradise, NF. Blood cholesterol screening influence of fasting state on cholesterol results and management decisions. J. Gen. Intern. Med 2000, 15, 395–399. [Google Scholar]
- Kimak, E; Ksiazek, A; Baranowicz-Gaszczyk, I; Solski, J. Disturbed lipids, lipoproteins and triglyceride-rich lipoproteins as well as fasting and nonfasting non-high-density lipoprotein cholesterol in post-renal transplant patients. Ren. Fail 2007, 29, 705–712. [Google Scholar]
- .
- Mohler, ER; Townsend, RR. Advanced Therapy in Hypertension and Vascular Disease; PMPH-USA: Hamilton, Canada, 2006. [Google Scholar]
- Hoeger, WK; Hoeger, SA. Lifetime Physical Fitness and Wellness: A Personalized Program, 10th ed; Cengage Learning: Belmont, CA, USA, 2008. [Google Scholar]
- Altman, DG; Machin, D; Bryant, TN; Gardner, MJ. Statistics with Confidence, 2nd ed; BMJ Books: London, UK, 2000. [Google Scholar]
- Heath, GW; Pratt, M; Warren, CW; Kann, L. Physical activity patterns in American high school students. Results from the 1990 youth risk behavior survey. Arch. Pediatr. Adolesc. Med 1994, 148, 1131–1136. [Google Scholar]
- Sung, RY; Yu, CC; Choi, KC; McManus, A; Li, AM; Xu, SL; Chan, D; Lo, AF; Chan, JC; Fok, TF. Waist circumference and body mass index in Chinese children: cutoff values for predicting cardiovascular risk factors. Int. J. Obes. (Lond) 2007, 31, 550–558. [Google Scholar]
- Lowry, R; Galuska, DA; Fulton, JE; Wechsler, H; Kann, L. Weight management goals and practices among U.S. high school students: associations with physical activity, diet, and smoking. J. Adolesc. Health 2002, 31, 133–144. [Google Scholar]
- Pescatello, LS; VanHeest, JL. Physical activity mediates a healthier body weight in the presence of obesity. Br. J. Sports Med 2000, 34, 86–93. [Google Scholar]
- Mokdad, AH; Serdula, MK; Dietz, WH; Bowman, BA; Marks, JS; Koplan, JP. The spread of the obesity epidemic in the United States, 1991–1998. JAMA 1999, 282, 1519–1522. [Google Scholar]
- Nihiser, AJ; Lee, SM; Wechsler, H; McKenna, M; Odom, E; Reinold, C; Thompson, D; Grummer-Strawn, L. BMI measurement in schools. Pediatrics 2009, 124, S89–S97. [Google Scholar]
- Brener, ND; McManus, T; Galuska, DA; Lowry, R; Wechsler, H. Reliability and validity of self-reported height and weight among high school students. J. Adolesc Health 2003, 32, 281–287. [Google Scholar]
- Abraham, S; Luscombe, G; Boyd, C; Olesen, I. Predictors of the accuracy of self-reported height and weight in adolescent female school students. Int. J. Eat. Disord 2004, 36, 76–82. [Google Scholar]
- Morrissey, SL; Whetstone, LM; Cummings, DM; Owen, LJ. Comparison of self-reported and measured height and weight in eighth-grade students. J. Sch. Health 2006, 76, 512–515. [Google Scholar]
- Vriendt, TD; Huybrechts, I; Ottevaere, C; Trimpont, IV; Henauw, SD. Validity of Self-Reported Weight and Height of Adolescents, Its Impact on Classification into BMI-Categories and the Association with Weighing Behaviour. Int. J. Environ. Res. Public Health 2009, 6, 2696–2711. [Google Scholar] [Green Version]
- Tokmakidis, SP; Christodoulos, AD; Mantzouranis, NI. Validity of self-reported anthropometric values used to assess body mass index and estimate obesity in Greek school children. J. Adolesc. Health 2007, 40, 305–310. [Google Scholar]
- Danubio, ME; Miranda, G; Vinciguerra, MG; Vecchi, E; Rufo, F. Comparison of self-reported and measured height and weight: implications for obesity research among young adults. Econ. Hum. Biol 2008, 6, 181–190. [Google Scholar]
- Stray-Pedersen, M; Helsing, RM; Gibbons, L; Cormick, G; Holmen, TL; Vik, T; Belizan, JM. Weight status and hypertension among adolescent girls in Argentina and Norway: data from the ENNyS and HUNT studies. BMC Public Health 2009, 9, 398. [Google Scholar]
- Galal, OM. The nutrition transition in Egypt: obesity, undernutrition and the food consumption context. Public Health Nutr 2002, 5, 141–148. [Google Scholar]
- Okuda, M; Sugiyama, S; Kunitsugu, I; Hinoda, Y; Okuda, Y; Shirabe, K; Yoshitake, N; Hobara, T. Use of Body Mass Index and Percentage Overweight Cutoffs to Screen Japanese Children and Adolescents for Obesity-Related Risk Factors. J. Epidemiol 2009, 20, 46–53. [Google Scholar]
- Lightwood, J; Bibbins-Domingo, K; Coxson, P; Wang, YC; Williams, L; Goldman, L. Forecasting the future economic burden of current adolescent overweight: an estimate of the coronary heart disease policy model. Am. J. Public Health 2009, 99, 2230–2237. [Google Scholar]
- Ribas, SA; da Silva, LC. Dyslipidemia in schoolchildren from private schools in Belem. Arq Bras Cardiol 2009, 92, 412–417. [Google Scholar]
- Anglum, A. Primary care management of childhood and adolescent hypertension. J. Am. Acad. Nurse Pract 2009, 21, 529–534. [Google Scholar]
- Paschoal, MA; Trevizan, PF; Scodeler, NF. Heart rate variability, blood lipids and physical capacity of obese and non-obese children. Arq. Bras. Cardiol 2009, 93, 239–246. [Google Scholar]
- Murphy, MH; Blair, SN; Murtagh, EM. Accumulated versus continuous exercise for health benefit: a review of empirical studies. Sports Med 2009, 39, 29–43. [Google Scholar]
- Nielsen, GA; Andersen, LB. The association between high blood pressure, physical fitness, and body mass index in adolescents. Prev. Med 2003, 36, 229–234. [Google Scholar]
- Gamble, HL; Parra, GR; Beech, BM. Moderators of physical activity and obesity during adolescence. Eat Behav 2009, 10, 232–236. [Google Scholar]
- Kim, Y; Lee, S. Physical activity and abdominal obesity in youth. Appl. Physiol. Nutr. Metab 2009, 34, 571–581. [Google Scholar]
- Ekelund, U; Franks, PW; Sharp, S; Brage, S; Wareham, NJ. Increase in physical activity energy expenditure is associated with reduced metabolic risk independent of change in fatness and fitness. Diabetes Care 2007, 30, 2101–2106. [Google Scholar]
- Falkner, B; Gidding, SS; Portman, R; Rosner, B. Blood pressure variability and classification of prehypertension and hypertension in adolescence. Pediatrics 2008, 122, 238–242. [Google Scholar]
- Peters, RM; Flack, JM. Salt sensitivity and hypertension in African Americans: implications for cardiovascular nurses. Prog. Cardiovasc. Nurs 2000, 15, 138–144. [Google Scholar]
- National Cholesterol Education Program: highlights of the Report of the Expert Panel on Blood Cholesterol Levels in Children and Adolescents. US Department of Health and Human Services, Public Health Service. National Institutes of Health, National Heart, Lung, and BloodInstitute. J. Am. Osteopath Assoc 1992, 92, 380–388. [Google Scholar]
- Tuomainen, P; Peuhkurinen, K; Kettunen, R; Rauramaa, R. Regular physical exercise, heart rate variability and turbulence in a 6-year randomized controlled trial in middle-aged men: the DNASCO study. Life Sci 2005, 77, 2723–2734. [Google Scholar]
- Buchheit, M; Gindre, C. Cardiac parasympathetic regulation: respective associations with cardiorespiratory fitness and training load. Am. J. Physiol. Heart Circ. Physiol 2006, 291, H451–H458. [Google Scholar]
- Gamelin, FX; Baquet, G; Berthoin, S; Thevenet, D; Nourry, C; Nottin, S; Bosquet, L. Effect of high intensity intermittent training on heart rate variability in prepubescent children. Eur. J. Appl. Physiol 2009, 105, 731–738. [Google Scholar]
- Ortega, FB; Ruiz, JR; Castillo, MJ; Sjostrom, M. Physical fitness in childhood and adolescence: a powerful marker of health. Int. J. Obes. (Lond) 2008, 32, 1–11. [Google Scholar]
- Fogelholm, M. How physical activity can work? Int. J. Pediatr. Obes 2008, 3, 10–14. [Google Scholar]
- Hardman, AE. Exercise in the prevention of atherosclerotic, metabolic and hypertensive diseases: a review. J. Sports Sci 1996, 14, 201–218. [Google Scholar]
- Sallis, JF; Patrick, K. Physical activity guidelines for adolescents: consensus statement. Pediatr. Exerc. Sci 1994, 6, 302–314. [Google Scholar]
- Khatib, O. Noncommunicable diseases: risk factors and regional strategies for prevention and care. East. Mediterr. Health J 2004, 10, 778–788. [Google Scholar]
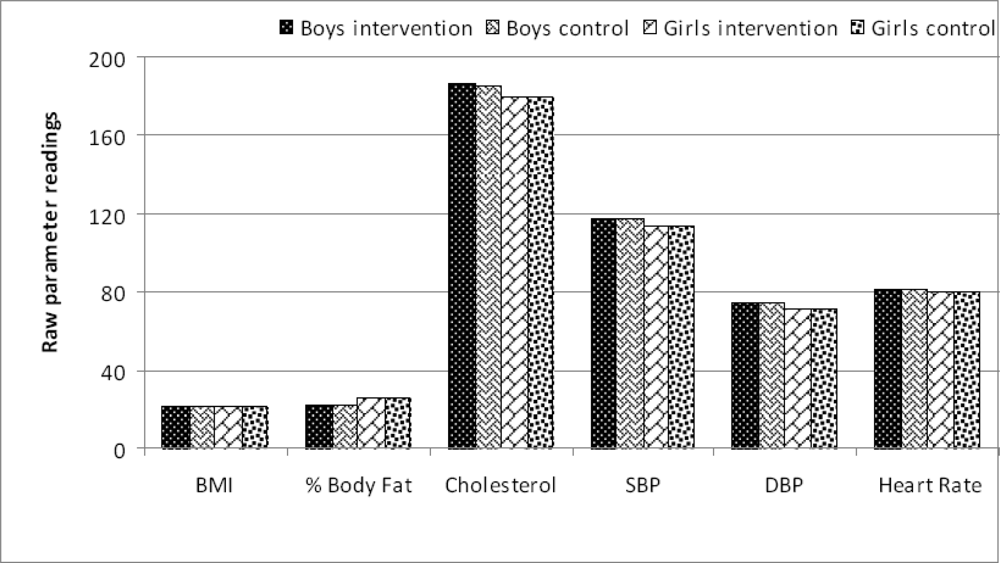
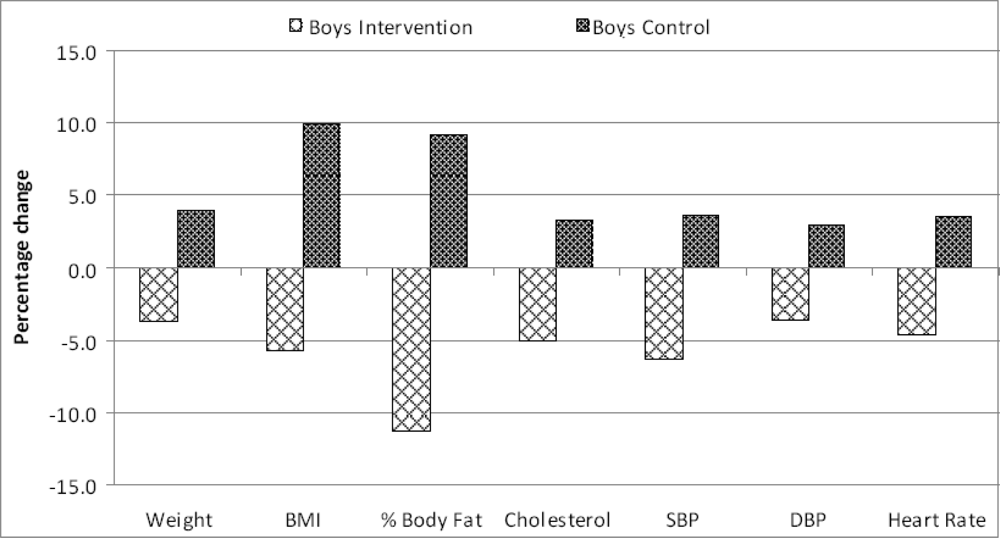
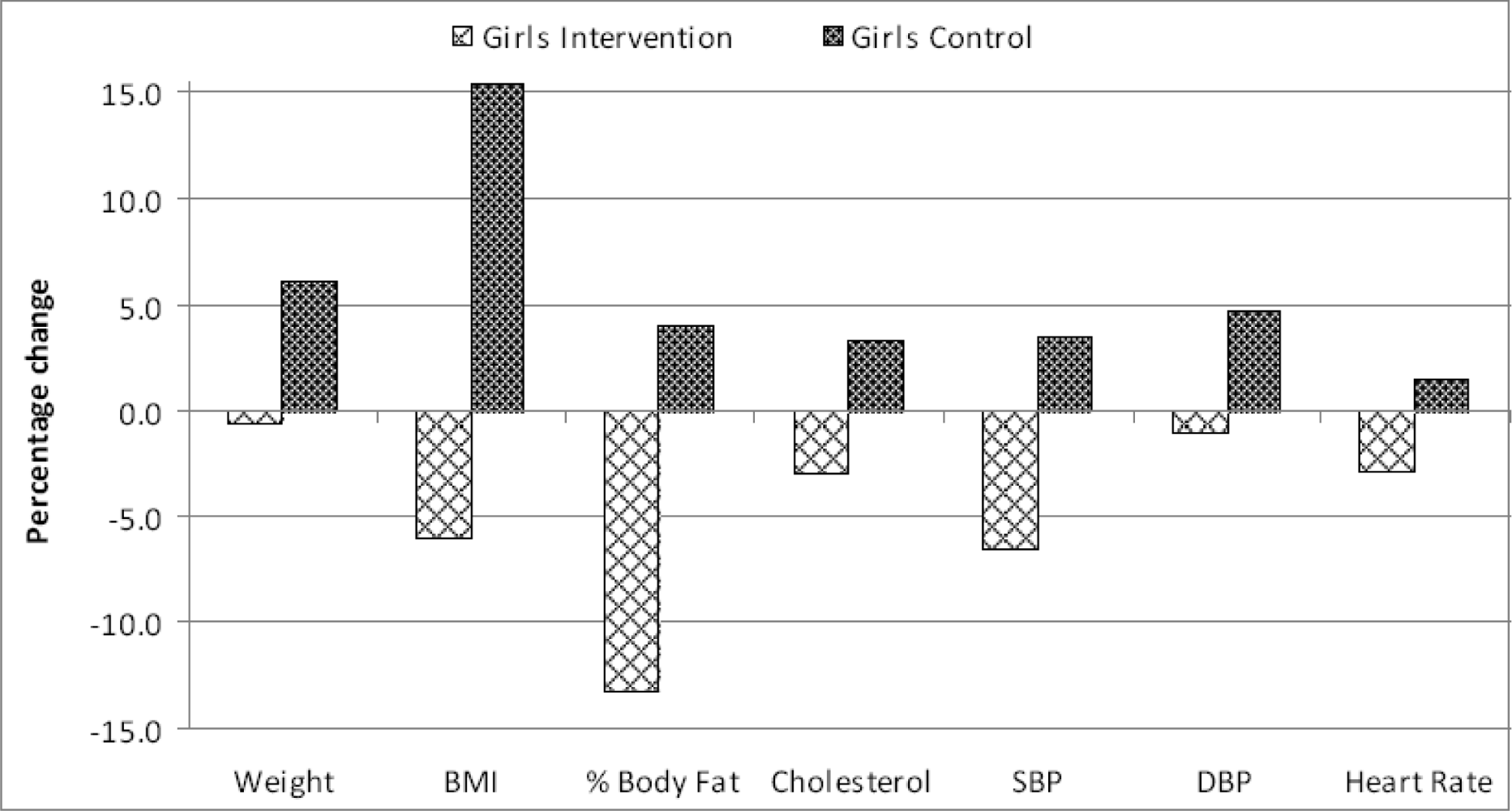
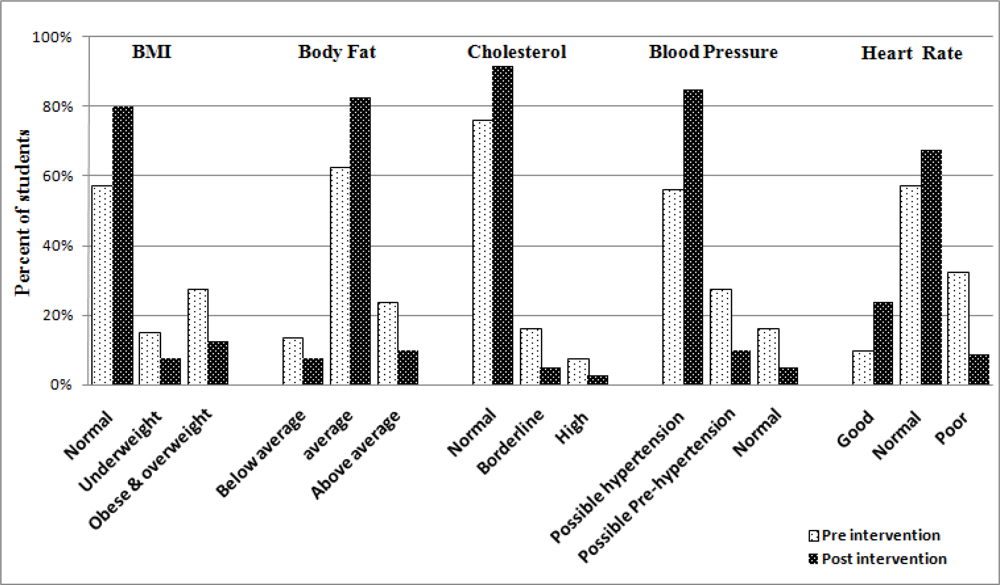
| Parameter | Control Mean (SD) | Intervention Mean (SD) | Differencea | 95% Confidence Limits | P - value | |
|---|---|---|---|---|---|---|
| Min | Max | |||||
| Boys | ||||||
| BMI† | 21.2 (3.6) | 20.9 (4.1) | +0.3 | −1.45 | +2.05 | NS |
| % Body Fat | 21.9 (5.4) | 22.1(6.3) | −0.2 | −2.86 | +2.46 | NS |
| Cholesterol§ | 185.3 (18.3) | 186.5 (17.1) | −1.2 | −9.11 | +6.71 | NS |
| SBP‡ | 116.9 (10.2) | 117.2 (8.9) | −0.3 | −4.56 | +3.96 | NS |
| DBP# | 74.3 (7.2) | 74.8 (6.8) | −0.5 | −3.63 | +2.63 | NS |
| Heart RateΨ | 80.7 (7.6) | 81.1(8.2) | −0.4 | −3.96 | +3.16 | NS |
| Girls | ||||||
| BMI† | 21.4 (3.8) | 21.6 (4.5) | −0.2 | −2.09 | +1.69 | NS |
| % Body Fat | 25.1 (8.1) | 25.6 (7.4) | −0.5 | −3.99 | +2.99 | NS |
| Cholesterol§ | 182.4 (14.3) | 179.4 (19.5) | +3.0 | −4.82 | +10.82 | NS |
| SBP‡ | 114.6 (11.3) | 113.7 (9.1) | +0.9 | −3.64 | +5.44 | NS |
| DBP# | 70.6 (6.1) | 71.3 (5.9) | −0.7 | −3.39 | +1.99 | NS |
| Heart RateΨ | 81.2 (9.1) | 80.5 (7.3) | +0.6 | −3.05 | +4.25 | NS |
| Parameter | Control Mean (SD) | Intervention Mean (SD) | Differencea | 95% Confidence Limits | P-value | |
|---|---|---|---|---|---|---|
| Min | Max | |||||
| Boys | ||||||
| BMI† | 23.3 (3.5) | 19.7 (3.1) | +3.6 | +2.12 | +5.07 | <0.05 |
| % Body Fat | 23.9 (10.1) | 19.6 (8.2) | +4.3 | +0.23 | +8.37 | <0.05 |
| Cholesterol§ | 191.4 (19) | 177.2 (19.3) | +14.2 | +5.60 | +22.80 | <0.05 |
| SBP‡ | 121.1 (8.3) | 109.7 (7.6) | +11.4 | +7.85 | +14.95 | <0.05 |
| DBP# | 76.5 (7.2) | 72.1 (6.8) | +4.4 | +1.27 | +7.53 | <0.05 |
| Heart RateΨ | 83.5 (8.3) | 77.3 (9.5) | +6.2 | +2.16 | +10.24 | <0.05 |
| Girls | ||||||
| BMI† | 24.7 (3.7) | 20.3 (3.6) | +4.4 | +2.77 | +6.04 | <0.05 |
| % Body Fat | 26.1 (7.9) | 22.2 (9.8) | +3.9 | –0.15 | +7.95 | NS |
| Cholesterol§ | 188.3 (16.5) | 174.1 (20.2) | +14.2 | +5.82 | +22.58 | <0.05 |
| SBP‡ | 118.6 (7.5) | 106.3 (8.3) | +12.3 | +8.73 | +15.87 | <0.05 |
| DBP# | 73.9 (6.1) | 70.6 (5.9) | +3.3 | +0.61 | +5.99 | <0.05 |
| Heart RateΨ | 82.2 (9.8) | 78.2 (7.6) | +4 | +0.13 | +7.87 | <0.05 |
| Parameter | Baseline Mean (SD) | After 3 months Mean (SD) | Differencea | 95% Confidence Limits | P-value | |
|---|---|---|---|---|---|---|
| Min | Max | |||||
| Boys | ||||||
| Weight¥ | 60.9 (9.3) | 63.3 (8.9) | +2.4 | –1.9 | +6.7 | NS |
| BMI† | 21.2 (3.6) | 23.3 (3.5) | +2.1 | +0.4 | +3.8 | <0.05 |
| % Body Fat | 21.9 (5.4) | 23.9 (10.1) | +2.0 | –1.9 | +5.9 | NS |
| Cholesterol§ | 185.3 (18.3) | 191.4 (19) | +6.1 | –2.8 | +15.0 | NS |
| SBP‡ | 116.9 (10.2) | 121.1 (8.3) | +4.2 | –0.2 | +8.6 | NS |
| DBP# | 74.3 (7.2) | 76.5 (7.2) | +2.2 | –1.2 | +5.6 | NS |
| Heart RateΨ | 80.7 (7.6) | 83.5 (8.3) | +2.8 | –1.0 | +6.6 | NS |
| Girls | ||||||
| Weight¥ | 61.1 (11) | 64.8 (11.4) | +3.7 | –1.6 | +9.0 | NS |
| BMI† | 21.4 (3.8) | 24.7 (3.7) | +3.3 | +1.5 | +5.1 | < 0.05 |
| % Body Fat | 25.1 (8.1) | 26.1 (7.9) | +1.0 | –2.8 | +6.5 | NS |
| Cholesterol§ | 182.4 (14.3) | 188.3 (16.5) | +5.9 | –1.5 | +12.3 | NS |
| SBP‡ | 114.6 (11.3) | 118.6 (7.5) | +4.0 | –0.6 | +8.7 | NS |
| DBP# | 70.6 (6.1) | 73.9 (6.1) | +3.3 | +0.4 | +6.2 | < 0.05 |
| Heart RateΨ | 81.1 (9.1) | 82.2 (9.8) | +1.1 | –3.4 | +5.6 | NS |
| Parameter | Pre Mean (SD) | Post Mean (SD) | Differencea | 95% Confidence Limits | P - value | |
|---|---|---|---|---|---|---|
| Min | Max | |||||
| Boys | ||||||
| Weight¥ | 61.2 (8.3) | 58.9 (9.2) | −2.3 | −6.9 | +1.4 | NS |
| BMI† | 20.9 (4.1) | 19.7 (3.1) | −1.2 | −2.7 | +0.3 | NS |
| % Body Fat | 22.1 (6.3) | 19.6 (8.2) | −1.5 | −5.6 | +0.5 | NS |
| Cholesterol§ | 186.5 (17.1) | 177.2 (19.3) | −9.3 | −16.9 | –1.7 | <0.05 |
| SBP‡ | 117.2 (8.9) | 109.7 (7.6) | −7.5 | −11.0 | –4.0 | <0.05 |
| DBP# | 74.8 (6.8) | 72.1 (6.8) | −2.7 | −5.5 | +0.1 | NS |
| Heart RateΨ | 81.1 (8.2) | 77.3 (9.5) | −3.8 | −7.6 | +0.1 | NS |
| Girls | ||||||
| Weight¥ | 60.2 (9.5) | 59.8 (10.2) | −0.4 | −4.5 | +3.7 | NS |
| BMI† | 21.6 (4.5) | 20.3 (3.6) | −1.3 | −3.0 | +0.4 | NS |
| % Body Fat | 25.6 (7.4) | 22.2 (9.8) | −3.4 | −7.0 | +0.2 | NS |
| Cholesterol§ | 179.4 (19.5) | 174.1 (20.2) | −5.3 | −13.6 | +3.0 | NS |
| SBP‡ | 113.7 (9.1) | 106.3 (8.3) | −7.4 | −11.0 | +3.8 | NS |
| DBP# | 71.3 (5.9) | 70.6 (5.9) | −0.7 | −3.2 | +1.8 | NS |
| Heart RateΨ | 80.5 (7.3) | 78.2 (7.6) | −2.3 | −5.4 | +0.8 | NS |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
El Ansari, W.; El Ashker, S.; Moseley, L. Associations between Physical Activity and Health Parameters in Adolescent Pupils in Egypt. Int. J. Environ. Res. Public Health 2010, 7, 1649-1669. https://doi.org/10.3390/ijerph7041649
El Ansari W, El Ashker S, Moseley L. Associations between Physical Activity and Health Parameters in Adolescent Pupils in Egypt. International Journal of Environmental Research and Public Health. 2010; 7(4):1649-1669. https://doi.org/10.3390/ijerph7041649
Chicago/Turabian StyleEl Ansari, Walid, Said El Ashker, and Laurence Moseley. 2010. "Associations between Physical Activity and Health Parameters in Adolescent Pupils in Egypt" International Journal of Environmental Research and Public Health 7, no. 4: 1649-1669. https://doi.org/10.3390/ijerph7041649
APA StyleEl Ansari, W., El Ashker, S., & Moseley, L. (2010). Associations between Physical Activity and Health Parameters in Adolescent Pupils in Egypt. International Journal of Environmental Research and Public Health, 7(4), 1649-1669. https://doi.org/10.3390/ijerph7041649




