Prevalence and Severity of Periodontitis in Patients with Ulcerative Colitis: A Case–Control Study
Abstract
1. Introduction
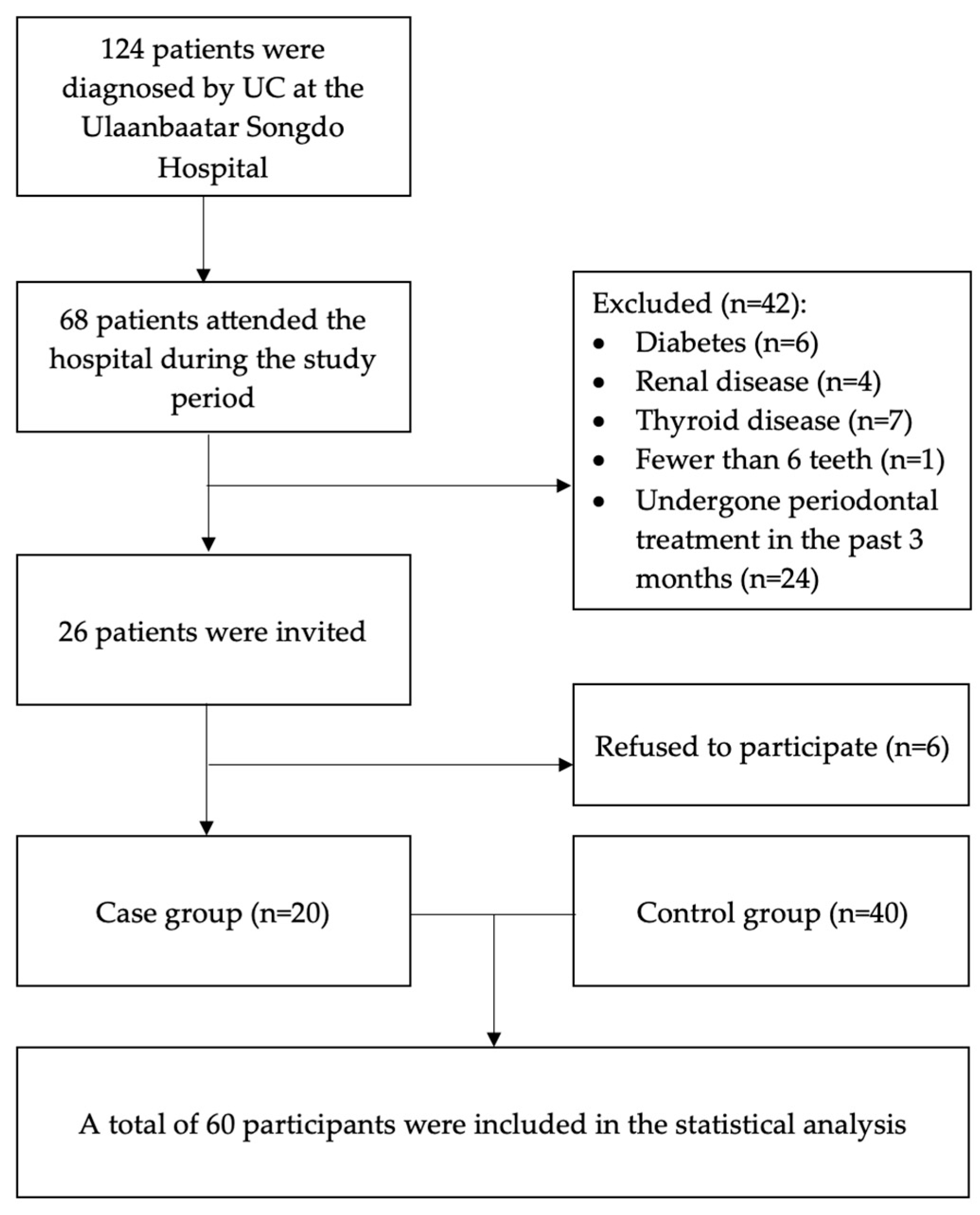
2. Materials and Methods
2.1. Study Participants
2.2. Ethical Considerations
2.3. Diagnosis of UC and Data Collection
2.4. Periodontal Examination
- Stage I/II periodontitis:
- Interdental CAL of 1–4 mm;
- Maximum probing depth ≤ 5 mm;
- No tooth loss due to periodontitis.
- Stage III/IV periodontitis:
- Interdental CAL ≥ 5 mm;
- Probing depth ≥ 6 mm;
- Tooth mobility Grade II or higher;
- At least one tooth due to periodontitis (Stage III) or masticatory dysfunction, bite collapse, or less than 20 remaining teeth (Stage IV).
2.5. Statistical Analysis
3. Results
3.1. General Characteristics
3.2. Clinical Characteristics
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| UC | Ulcerative colitis |
| CD | Crohn’s disease |
| IBD | Inflammatory bowel disease |
| MNUMS | Mongolian National University of Medical Sciences |
| ICC | Intraclass correlation coefficient |
| PPD | Probing pocket depth |
| BOP | Bleeding on probing |
| CAL | Clinical attachment level |
| FMPS | Full-mouth plaque score |
| FMBS | Full-mouth bleeding score |
| SD | Standard deviation |
| IQR | Interquartile range |
| OR | Odds ratio |
| CI | Confidence interval |
| IL | Interleukin |
| TNF | Tumor necrosis factor |
| MMP | Matrix metalloproteinase |
References
- Abraham, C.; Cho, J.H. Inflammatory Bowel Disease. N. Engl. J. Med. 2009, 361, 2066–2078. [Google Scholar] [CrossRef]
- de Souza, H.S.P.; Fiocchi, C.; Iliopoulos, D. The IBD Interactome: An Integrated View of Aetiology, Pathogenesis and Therapy. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 739–749. [Google Scholar] [CrossRef] [PubMed]
- Molodecky, N.A.; Soon, I.S.; Rabi, D.M.; Ghali, W.A.; Ferris, M.; Chernoff, G.; Benchimol, E.I.; Panaccione, R.; Ghosh, S.; Barkema, H.W.; et al. Increasing Incidence and Prevalence of the Inflammatory Bowel Diseases with Time, Based on Systematic Review. Gastroenterology 2012, 142, 46–54.e42. [Google Scholar] [CrossRef]
- Ribaldone, D.G.; Pellicano, R.; Actis, G.C. The Gut and the Inflammatory Bowel Diseases Inside-out: Extra-Intestinal Manifestations. Minerva Gastroenterol. Dietol. 2019, 65, 309–318. [Google Scholar] [CrossRef]
- Li, C.; Wu, Y.; Xie, Y.; Zhang, Y.; Jiang, S.; Wang, J.; Luo, X.; Chen, Q. Oral Manifestations Serve as Potential Signs of Ulcerative Colitis: A Review. Front. Immunol. 2022, 13, 1013900. [Google Scholar] [CrossRef] [PubMed]
- Tabarsi, N.T.; Mortazavi, N.; Norouzi, A.; Besharat, S.; Behnampour, N.; Asgari, N. Association of Oral Manifestations with Severity of the Disease in Ulcerative Colitis Patients. BMC Gastroenterol. 2024, 24, 378. [Google Scholar] [CrossRef] [PubMed]
- Kucharski, R.; Sobocki, B.K.; Stachowska, E.; Bulman, N.; Kalinowski, L.; Kaźmierczak-Siedlecka, K. Dental Problems and Oral Microbiome Alterations in Ulcerative Colitis. Front. Immunol. 2025, 16, 1502605. [Google Scholar] [CrossRef]
- Kinane, D.F.; Stathopoulou, P.G.; Papapanou, P.N. Periodontal Diseases. Nat. Rev. Dis. Primer 2017, 3, 17038. [Google Scholar] [CrossRef]
- GBD 2017 Oral Disorders Collaborators; Bernabe, E.; Marcenes, W.; Hernandez, C.R.; Bailey, J.; Abreu, L.G.; Alipour, V.; Amini, S.; Arabloo, J.; Arefi, Z.; et al. Global, Regional, and National Levels and Trends in Burden of Oral Conditions from 1990 to 2017: A Systematic Analysis for the Global Burden of Disease 2017 Study. J. Dent. Res. 2020, 99, 362–373. [Google Scholar] [CrossRef]
- Hajishengallis, G.; Chavakis, T. Local and Systemic Mechanisms Linking Periodontal Disease and Inflammatory Comorbidities. Nat. Rev. Immunol. 2021, 21, 426–440. [Google Scholar] [CrossRef]
- Genco, R.J.; Borgnakke, W.S. Risk Factors for Periodontal Disease. Periodontol. 2000 2013, 62, 59–94. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Ji, D.; Chen, X.; Wang, L.; Ye, F. Association Between Intestinal Diseases and Periodontitis: A Systematic Review and Meta-Analysis. Oral Dis. 2025, 31, 1603–1625. [Google Scholar] [CrossRef] [PubMed]
- Baima, G.; Massano, A.; Squillace, E.; Caviglia, G.P.; Buduneli, N.; Ribaldone, D.G.; Aimetti, M. Shared Microbiological and Immunological Patterns in Periodontitis and IBD: A Scoping Review. Oral Dis. 2022, 28, 1029–1041. [Google Scholar] [CrossRef]
- Agossa, K.; Dendooven, A.; Dubuquoy, L.; Gower-Rousseau, C.; Delcourt-Debruyne, E.; Capron, M. Periodontal Manifestations of Inflammatory Bowel Disease: Emerging Epidemiologic and Biologic Evidence. J. Periodontal Res. 2017, 52, 313–324. [Google Scholar] [CrossRef]
- Papageorgiou, S.N.; Hagner, M.; Nogueira, A.V.B.; Franke, A.; Jäger, A.; Deschner, J. Inflammatory Bowel Disease and Oral Health: Systematic Review and a Meta-Analysis. J. Clin. Periodontol. 2017, 44, 382–393. [Google Scholar] [CrossRef]
- She, Y.-Y.; Kong, X.-B.; Ge, Y.-P.; Liu, Z.-Y.; Chen, J.-Y.; Jiang, J.-W.; Jiang, H.-B.; Fang, S.-L. Periodontitis and Inflammatory Bowel Disease: A Meta-Analysis. BMC Oral Health 2020, 20, 67. [Google Scholar] [CrossRef]
- Agossa, K.; Roman, L.; Gosset, M.; Yzet, C.; Fumery, M. Periodontal and Dental Health in Inflammatory Bowel Diseases: A Systematic Review. Expert Rev. Gastroenterol. Hepatol. 2021, 15, 1281–1294. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Qiao, D.; Chen, R.; Zhu, F.; Gong, J.; Yan, F. The Association between Periodontitis and Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis. BioMed Res. Int. 2021, 2021, 6692420. [Google Scholar] [CrossRef]
- Brito, F.; de Barros, F.C.; Zaltman, C.; Carvalho, A.T.P.; Carneiro, A.J.d.V.; Fischer, R.G.; Gustafsson, A.; Figueredo, C.M. de S. Prevalence of Periodontitis and DMFT Index in Patients with Crohn’s Disease and Ulcerative Colitis. J. Clin. Periodontol. 2008, 35, 555–560. [Google Scholar] [CrossRef]
- Habashneh, R.A.; Khader, Y.S.; Alhumouz, M.K.; Jadallah, K.; Ajlouni, Y. The Association between Inflammatory Bowel Disease and Periodontitis among Jordanians: A Case-Control Study. J. Periodontal Res. 2012, 47, 293–298. [Google Scholar] [CrossRef]
- Vavricka, S.R.; Manser, C.N.; Hediger, S.; Vögelin, M.; Scharl, M.; Biedermann, L.; Rogler, S.; Seibold, F.; Sanderink, R.; Attin, T.; et al. Periodontitis and Gingivitis in Inflammatory Bowel Disease: A Case-Control Study. Inflamm. Bowel Dis. 2013, 19, 2768–2777. [Google Scholar] [CrossRef]
- Zhang, L.; Gao, X.; Zhou, J.; Chen, S.; Zhang, J.; Zhang, Y.; Chen, B.; Yang, J. Increased Risks of Dental Caries and Periodontal Disease in Chinese Patients with Inflammatory Bowel Disease. Int. Dent. J. 2020, 70, 227–236. [Google Scholar] [CrossRef]
- Haugbo, H.O.; Klepp, P.; Verket, A. Ulcerative Colitis and Periodontitis—A Cross-Sectional Pilot Study from a Norwegian Cohort. Acta Odontol. Scand. 2023, 81, 541–548. [Google Scholar] [CrossRef]
- Satsangi, J.; Silverberg, M.S.; Vermeire, S.; Colombel, J.-F. The Montreal Classification of Inflammatory Bowel Disease: Controversies, Consensus, and Implications. Gut 2006, 55, 749–753. [Google Scholar] [CrossRef]
- Papapanou, P.N.; Sanz, M.; Buduneli, N.; Dietrich, T.; Feres, M.; Fine, D.H.; Flemmig, T.F.; Garcia, R.; Giannobile, W.V.; Graziani, F.; et al. Periodontitis: Consensus Report of Workgroup 2 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J. Periodontol. 2018, 89 (Suppl. S1), S173–S182. [Google Scholar] [CrossRef] [PubMed]
- Grössner-Schreiber, B.; Fetter, T.; Hedderich, J.; Kocher, T.; Schreiber, S.; Jepsen, S. Prevalence of Dental Caries and Periodontal Disease in Patients with Inflammatory Bowel Disease: A Case-Control Study. J. Clin. Periodontol. 2006, 33, 478–484. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.X.W.; Brand, H.S.; Kalender, B.; De Boer, N.K.H.; Forouzanfar, T.; de Visscher, J.G.A.M. Dental and Periodontal Disease in Patients with Inflammatory Bowel Disease. Clin. Oral Investig. 2021, 25, 5273–5280. [Google Scholar] [CrossRef]
- Baima, G.; Muwalla, M.; Testa, G.; Mazza, F.; Bebars, A.; Perotto, S.; Vernero, M.; Massano, A.; Romano, F.; Ribaldone, D.G.; et al. Periodontitis Prevalence and Severity in Inflammatory Bowel Disease: A Case-Control Study. J. Periodontol. 2023, 94, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Enver, A.; Ozmeric, N.; Isler, S.C.; Toruner, M.; Fidan, C.; Demirci, G.; Elgun, S.; Silva, A.P.B.D. Evaluation of Periodontal Status and Cytokine Levels in Saliva and Gingival Crevicular Fluid of Patients with Inflammatory Bowel Diseases. J. Periodontol. 2022, 93, 1649–1660. [Google Scholar] [CrossRef]
- Zhou, T.; Xu, W.; Wang, Q.; Jiang, C.; Li, H.; Chao, Y.; Sun, Y.; A, L. The Effect of the “Oral-Gut” Axis on Periodontitis in Inflammatory Bowel Disease: A Review of Microbe and Immune Mechanism Associations. Front. Cell. Infect. Microbiol. 2023, 13, 1132420. [Google Scholar] [CrossRef]
- Yu, J.; Lyu, J.; Zhu, T.; Li, Y.; Xia, H.; Liu, Q.; Li, L.; Chen, B. Oral-Gut Axis in Inflammation: Periodontitis Exacerbates Ulcerative Colitis via Microbial Dysbiosis and Barrier Disruption. BMC Oral Health 2025, 25, 894. [Google Scholar] [CrossRef]
- Zheng, Z.; Jin, W.; Guo, W.; Jin, Z.; Zuo, Y. Oral Fusobacterium Nucleatum Exacerbates Ulcerative Colitis via the Oral-Gut Axis: Mechanisms and Therapeutic Implications. Front. Cell. Infect. Microbiol. 2025, 15, 1564169. [Google Scholar] [CrossRef] [PubMed]
- Xi, M.; Ruan, Q.; Zhong, S.; Li, J.; Qi, W.; Xie, C.; Wang, X.; Abuduxiku, N.; Ni, J. Periodontal Bacteria Influence Systemic Diseases through the Gut Microbiota. Front. Cell. Infect. Microbiol. 2024, 14, 1478362. [Google Scholar] [CrossRef] [PubMed]
- Brito, F.; Zaltman, C.; Carvalho, A.T.P.; Fischer, R.G.; Persson, R.; Gustafsson, A.; Figueredo, C.M.S. Subgingival Microflora in Inflammatory Bowel Disease Patients with Untreated Periodontitis. Eur. J. Gastroenterol. Hepatol. 2013, 25, 239–245. [Google Scholar] [CrossRef] [PubMed]
| Variables | UC (n = 20) | Controls (n = 40) | p-Value |
|---|---|---|---|
| Age (years) | 42.9 ± 12.14 | 42.58 ± 12.06 | 0.875 |
| Gender: male, % (n) | 40 (8) | 40 (16) | 1.00 |
| Area, % (n) Urban Rural | 80 (16) 20 (4) | 90 (36) 10 (4) | 0.422 |
| Education level, % (n) Low Intermediate High | - 15 (3) 85 (17) | 32.5 (13) 67.5 (27) | 0.218 |
| Years with UC diagnosis | 1.79 (2.68) | - | |
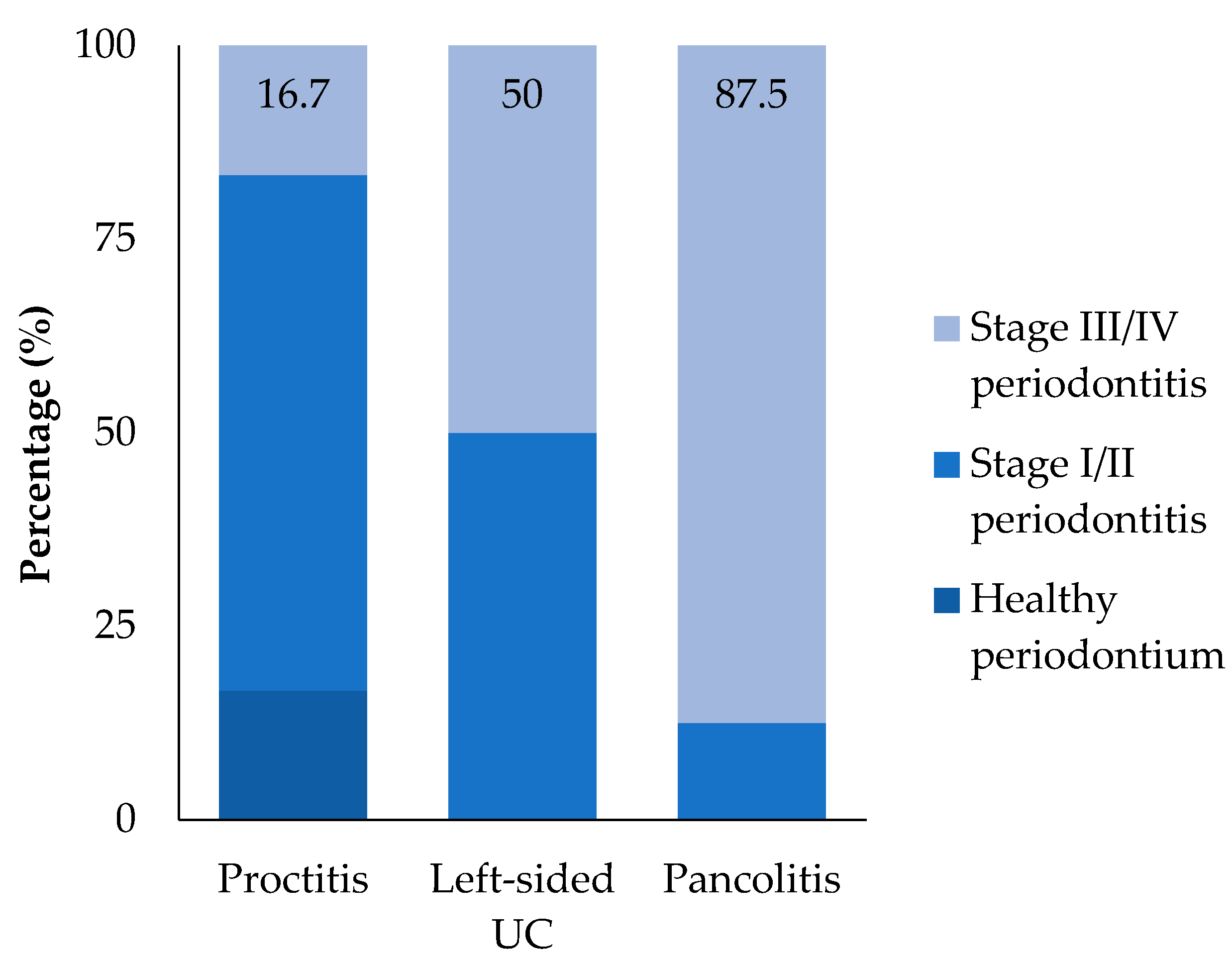
| Localization, % (n) E1 E2 E3 | 30 (6) 30 (6) 40 (8) | - | |
| Oral health practices, % (n) Flossing | 45 (9) | 55 (22) | 0.586 |
| Brushing frequency, % (n) ≤Once/day Twice/day ≥Three times/day | 45 (9) 55 (11) - | 27.5 (11) 72.5 (29) - | 0.246 |
| Smoking, %(n) Yes No | 25 (5) 75 (15) | 85 (34) 15 (6) | 0.481 |
| Variables | UC (n = 20) | Controls (n = 40) | p-Value |
|---|---|---|---|
| Diagnosis, % (n) | 0.011 | ||
| Healthy periodontium | 5 (1) | 27.5 (11) | |
| Stage I/II periodontitis | 40 (8) | 52.5 (21) | |
| Stage III/IV periodontitis | 55 (11) | 20 (8) | |
| Periodontal parameters, | |||
| median (IQR) | |||
| Number of teeth | 24 (5) | 25 (5) | 0.482 |
| FMPS | 71.4 (24.62) | 54.1 (21.85) | 0.002 |
| FMBS | 27.68 (15.73) | 23.7 (17.79) | 0.433 |
| CAL | 4 (2) | 2.5 (4) | 0.010 |
| Number of PPD ≥ 4 mm | 3.5 (5) | 1 (3) | <0.001 |
| Variables | OR (95% CI) | p-Value |
|---|---|---|
| Age (years) | 1.13 (1.06–1.20) | <0.001 |
| Sex: Male | 0.47 (0.14–1.54) | 0.211 |
| Smoking | 1.43 (0.36–5.66) | 0.611 |
| FMPS (%) | 1.09 (1.04–1.16) | <0.001 |
| FMBS (%) | 1.03 (0.99–1.07) | 0.106 |
| UC | Stage III/IV Periodontitis | |
|---|---|---|
| OR (95% CI) | p-Value | |
| Crude | 4.0 (1.24–12.88) | 0.020 |
| Adjusted for age | 9.14 (1.72–48.65) | 0.010 |
| Adjusted for age and smoking | 8.93 (1.60–49.81) | 0.012 |
| Adjusted for age and FMPS | 4.86 (0.77; 30.50) | 0.092 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soronzonbold, A.; Khishigdorj, O.; Duger, D.; Batbayar, B. Prevalence and Severity of Periodontitis in Patients with Ulcerative Colitis: A Case–Control Study. Int. J. Environ. Res. Public Health 2025, 22, 1355. https://doi.org/10.3390/ijerph22091355
Soronzonbold A, Khishigdorj O, Duger D, Batbayar B. Prevalence and Severity of Periodontitis in Patients with Ulcerative Colitis: A Case–Control Study. International Journal of Environmental Research and Public Health. 2025; 22(9):1355. https://doi.org/10.3390/ijerph22091355
Chicago/Turabian StyleSoronzonbold, Angar, Oyunkhishig Khishigdorj, Davaadorj Duger, and Bayarchimeg Batbayar. 2025. "Prevalence and Severity of Periodontitis in Patients with Ulcerative Colitis: A Case–Control Study" International Journal of Environmental Research and Public Health 22, no. 9: 1355. https://doi.org/10.3390/ijerph22091355
APA StyleSoronzonbold, A., Khishigdorj, O., Duger, D., & Batbayar, B. (2025). Prevalence and Severity of Periodontitis in Patients with Ulcerative Colitis: A Case–Control Study. International Journal of Environmental Research and Public Health, 22(9), 1355. https://doi.org/10.3390/ijerph22091355





