Impact of Oral Hygiene Practices in Reducing Cardiometabolic Risk, Incidence, and Mortality: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Inclusion and Exclusion Criteria
2.2. Search Strategy
2.3. Data Extraction and Synthesis
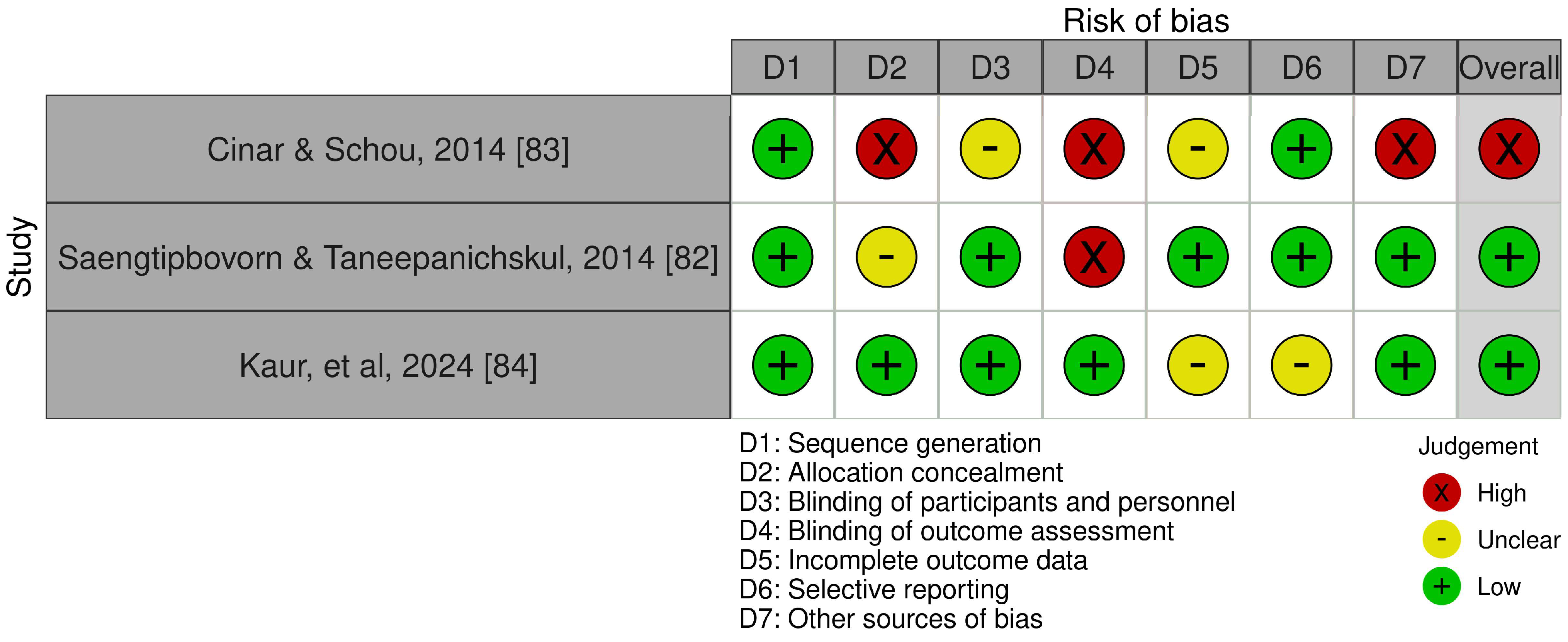
2.4. Quality Appraisal and Risk of Bias
3. Results
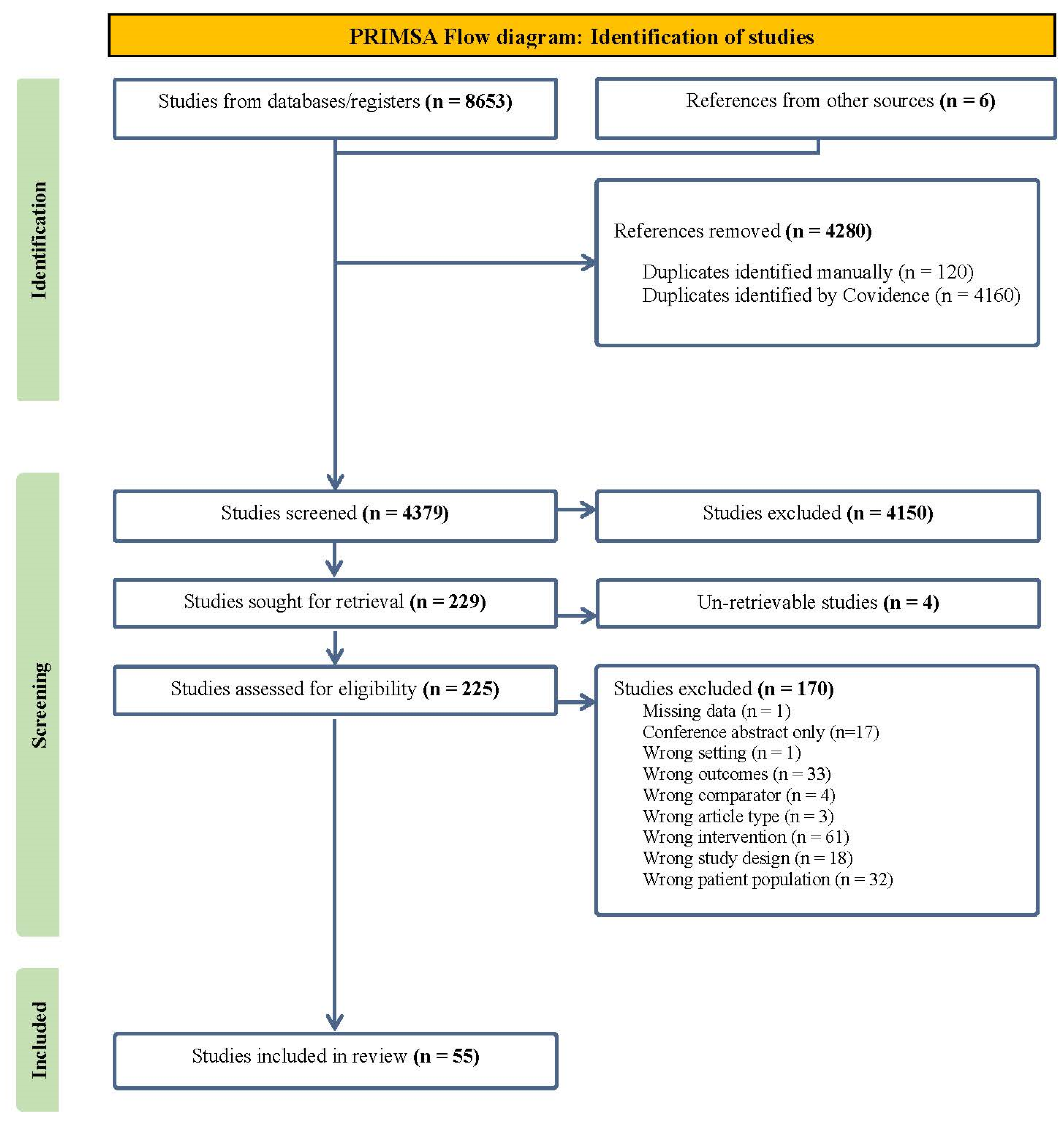
3.1. Selection Process
3.2. Characteristics
3.3. Toothbrushing Frequency
3.4. Toothbrushing Device
3.5. Toothbrushing Technique
3.6. Timing
3.7. Duration
3.8. Interdental Cleaning
3.9. Mouthwash and Toothpaste
4. Discussion
4.1. Main Findings
4.2. Risk Factor Comparison
5. Clinical Significance
6. Strengths and Limitations
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Agrawal, A.; Rao, G. Novel Approaches for Early Diagnosis and Prevention of Cardiometabolic Diseases. J. Clin. Prev. Cardiol. 2023, 12, 23–26. [Google Scholar] [CrossRef]
- Barnett, K.; Mercer, S.W.; Norbury, M.; Watt, G.; Wyke, S.; Guthrie, B. Epidemiology of multimorbidity and implications for health care, research, and medical education: A cross-sectional study. Lancet 2012, 380, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Roth, G.A.; Mensah, G.A.; Johnson, C.O.; Addolorato, G.; Ammirati, E.; Baddour, L.M.; Barengo, N.C.; Beaton, A.Z.; Benjamin, E.J.; Benziger, C.P.; et al. Global Burden of Cardiovascular Diseases and Risk Factors, 1990–2019: Update From the GBD 2019 Study. J. Am. Coll. Cardiol. 2020, 76, 2982–3021. [Google Scholar] [CrossRef] [PubMed]
- Ong, K.L.; Stafford, L.K.; McLaughlin, S.A.; Boyko, E.J.; Vollset, S.E.; Smith, A.E.; Dalton, B.E.; Duprey, J.; Cruz, J.A.; Hagins, H.; et al. Global, regional, and national burden of diabetes from 1990 to 2021, with projections of prevalence to 2050: A systematic analysis for the Global Burden of Disease Study 2021. Lancet 2023, 402, 203–234. [Google Scholar] [CrossRef] [PubMed]
- Bello, A.K.; Okpechi, I.G.; Levin, A.; Ye, F.; Damster, S.; Arruebo, S.; Donner, J.-A.; Caskey, F.J.; Cho, Y.; Davids, M.R.; et al. An update on the global disparities in kidney disease burden and care across world countries and regions. Lancet Glob. Health 2024, 12, e382–e395. [Google Scholar] [CrossRef]
- Heart, Stroke and Vascular Disease: Australian Facts. Australian Institute of Health and Welfare. June 2024. Available online: https://www.aihw.gov.au/reports/heart-stroke-vascular-diseases/hsvd-facts/contents/about (accessed on 1 September 2022).
- Australian Institute of Health and Welfare. Diabetes: Australian Facts. 17 June 2024. Available online: https://www.aihw.gov.au/reports/diabetes/diabetes/contents/impact-of-diabetes/diabetes-deaths (accessed on 19 August 2024).
- Australian Institute of Health and Welfare. Chronic Kidney Disease: Australian Facts, About. 17 June 2024. Available online: https://www.aihw.gov.au/reports/chronic-kidney-disease/chronic-kidney-disease/contents/about (accessed on 19 August 2024).
- Boyd, L.; Wilkins, E. Periodontal Disease Development. In Clinical Practice of The Dental Hygienist; Jones & Bartlett Learning, LLC: Burlington, VT, USA, 2020; pp. 659–706. [Google Scholar]
- Eke, P.I.; Borgnakke, W.S.; Genco, R.J. Recent epidemiologic trends in periodontitis in the USA. Periodontology 2000 2020, 82, 257–267. [Google Scholar] [CrossRef]
- Nazir, M.A. Prevalence of periodontal disease, its association with systemic diseases and prevention. Int. J. Health Sci. 2017, 11, 72–80. [Google Scholar]
- Zhao, X.; Wang, J.; Xu, Y.; Zhou, J.; Hu, L. The role of periodontal disease in atherosclerotic cardiovascular disease. Biocell 2023, 47, 1431–1438. [Google Scholar] [CrossRef]
- Bui, F.Q.; Almeida-da-Silva, C.L.C.; Huynh, B.; Trinh, A.; Liu, J.; Woodward, J.; Asadi, H.; Ojcius, D.M. Association between periodontal pathogens and systemic disease. Biomed. J. 2019, 42, 27–35. [Google Scholar] [CrossRef]
- Păunică, I.; Giurgiu, M.; Dumitriu, A.S.; Păunică, S.; Pantea Stoian, A.M.; Martu, M.-A.; Serafinceanu, C. The Bidirectional Relationship between Periodontal Disease and Diabetes Mellitus—A Review. Diagnostics 2023, 13, 681. [Google Scholar] [CrossRef]
- Wahid, A.; Chaudhry, S.; Ehsan, A.; Butt, S.; Ali Khan, A. Bidirectional Relationship between Chronic Kidney Disease & Periodontal Disease. Pak. J. Med. Sci. 2013, 29, 211–215. [Google Scholar] [CrossRef] [PubMed]
- He, I.; Poirier, B.; Jensen, E.; Kaur, S.; Hedges, J.; Jesudason, S.; Jamieson, L.; Sethi, S. Demystifying the connection between periodontal disease and chronic kidney disease—An umbrella review. J. Periodontal Res. 2023, 58, 874–892. [Google Scholar] [CrossRef] [PubMed]
- Salhi, L.; De Carvalho, B.; Reners, M. Update on the Roles of Oral Hygiene and Plaque Control on Periodontal Disease. In Periodontitis: Advances in Experimental Research, 13th ed.; Santi-Rocca, J., Ed.; Springer International Publishing: Cham, Switzerland, 2022; pp. 329–339. ISBN 978-3-030-96881-6. [Google Scholar]
- Macarelli, C.; Boyd, L. Oral Infection Control: Toothbrushes and Toothbrushing. In Clinical Practice of the Dental Hygienist; Jones & Bartlett Learning, LLC: Burlington, VT, USA, 2020; pp. 929–983. [Google Scholar]
- Perry, K. Dentifrices and Mouthrinses. In Clinical Practice of the Dental Hygienist; Jones & Bartlett Learning, LLC: Burlington, VT, USA, 2020; pp. 1023–1059. [Google Scholar]
- Moravec, L.; Boyd, L. Oral Infection Control: Interdental Care. In Clinical Practice of the Dental Hygienist; Jones & Bartlett Learning, LLC: Burlington, VT, USA, 2020; pp. 984–1022. [Google Scholar]
- Berger, D.; Rakhamimova, A.; Pollack, A.; Loewy, Z. Oral Biofilms: Development, Control, and Analysis. High. Throughput 2018, 7, 24. [Google Scholar] [CrossRef] [PubMed]
- Miller, C.S.; Emecen-Huyja, P.; Gehrig, J.; Shin, D. Host Immune Response to Periodontal Pathogens. In Foundations of Periodontics for the Dental Hygienist; Wolters Kluwer | Lippincott WIlliams & Wilkins: Philadelphia, PN, USA, 2019; pp. 279–300. [Google Scholar]
- Tadakamadla, J.; Kumar, S.; Mamatha, G.P. Comparative evaluation of oral health status of chronic kidney disease (CKD) patients in various stages and healthy controls. Spec. Care Dent. 2014, 34, 122–126. [Google Scholar] [CrossRef] [PubMed]
- Joseph, P.; Prabhakar, P.; Holtfreter, B.; Pink, C.; Suvan, J.; Kocher, T.; Pitchika, V. Systematic review and meta-analysis of randomized controlled trials evaluating the efficacy of non-surgical periodontal treatment in patients with concurrent systemic conditions. Clin. Oral. Investig. 2023, 28, 21. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, C.; Zhang, P.; Fu, W.; Zhang, J.; Zhang, Z.; Mao, J.; Yang, Y.; Zou, L. Association Between the Frequency of Tooth Brushing and the Risk of Cardiovascular Disease: A Systematic Review and Meta-analysis. Angiology 2024. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Boutron, I.; Page, M.J.; Higgins, J.P.T.; Altman, D.G.; Lundh, A.; Hróbjartsson, A. Chapter 7: Considering bias and conflicts of interest among the included studies. In Cochrane Handbook for Systematic Reviews of Interventions Version 64; Higgins, J.P.T., Thomas, J., Chandler, J., Cumpston, M., Page, M., Welsh, V., Eds.; Cochrane: Hoboken, NJ, USA, 2023; pp. 8:1–8:73, updated August 2023; Available online: www.training.cochrane.org/handbook (accessed on 1 January 2020).
- Wells, G.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses; Ottawa Hospital Research Institute: Ottawa, ON, Canada, 2021; Available online: https://www.ohri.ca//programs/clinical_epidemiology/oxford.asp (accessed on 1 September 2024).
- Covidence Systematic Review Software; Veritas Health Innovation: Melbourne, Australia, 2023. Available online: https://www.covidence.org/ (accessed on 1 September 2024).
- Luo, H.; Wu, B.; Kamer, A.R.; Adhikari, S.; Sloan, F.; Plassman, B.L.; Tan, C.; Qi, X.; Schwartz, M.D. Oral Health, Diabetes, and Inflammation: Effects of Oral Hygiene Behaviour. Int. Dent. J. 2022, 72, 484–490. [Google Scholar] [CrossRef]
- Wang, Y.; Jiang, Y.; Chen, Y.; Yu, L.; Zhou, J.; Wang, N.; Liu, T.; Fu, C. Associations of oral hygiene with incident hypertension and type 2 diabetes mellitus: A population-based cohort study in Southwest China. J. Clin. Hypertens. 2022, 24, 483–492. [Google Scholar] [CrossRef]
- Chang, Y.; Lee, J.S.; Lee, K.-J.; Woo, H.G.; Song, T.-J. Improved oral hygiene is associated with decreased risk of new-onset diabetes: A nationwide population-based cohort study. Diabetologia 2020, 63, 924–933. [Google Scholar] [CrossRef]
- Assante, S.M.; Morimoto, S.; Tedesco, T.K.; Gimenez, T.; Ramalho, K.M. Correlations between dental assistance/oral health and clinical intercurrences in an end-stage kidney disease patients: A historical cohort study. Minerva Stomatol. 2020, 69, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, A.; Panat, S.R. Oral health behavior and HbA1c in Indian adults with type 2 diabetes. J. Oral. Sci. 2012, 54, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Almas, K.; Al-Lazzam, S.; Al-Quadairi, A. The effect of oral hygiene instructions on diabetic type 2 male patients with periodontal diseases. J. Contemp. Dent. Pract. 2003, 4, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Woo, H.G.; Lee, J.S.; Song, T.J. Better oral hygiene is associated with lower risk of stroke. J. Periodontol. 2021, 92, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Woo, H.G.; Park, J.; Lee, J.S.; Song, T.J. Improved oral hygiene care is associated with decreased risk of occurrence for atrial fibrillation and heart failure: A nationwide population-based cohort study. Eur. J. Prev. Cardiol. 2020, 27, 1835–1845. [Google Scholar] [CrossRef]
- de Souza, C.M.; Braosi, A.P.R.; Luczyszyn, S.M.; Olandoski, M.; Kotanko, P.; Craig, R.G.; Trevilatto, P.C.; Pecoits-Filho, R. Association among oral health parameters, periodontitis, and its treatment and mortality in patients undergoing hemodialysis. J. Periodontol. 2014, 85, e169–e178. [Google Scholar] [CrossRef]
- Hirano, K.; Shimbo, T.; Komatsu, Y.; Kobayashi, D. Frequency of tooth brushing as a predictive factor for future kidney function decline. J. Nephrol. 2022, 35, 191–199. [Google Scholar] [CrossRef]
- Kobayashi, D.; Mizuno, A.; Mitsui, R.; Shimbo, T. Frequency of daily tooth brushing and subsequent cardiovascular events. Coron. Artery Dis. 2020, 31, 545–549. [Google Scholar] [CrossRef]
- Joshipura, K.J.; Munoz-Torres, F.J.; Morou-Bermudez, E.; Patel, R.P. Over-the-counter mouthwash use and risk of pre-diabetes/diabetes. Nitric Oxide 2017, 71, 14–20. [Google Scholar] [CrossRef]
- Joshipura, K.; Munoz-Torres, F.; Fernandez-Santiago, J.; Patel, R.P.; Lopez-Candales, A. Over-the-counter mouthwash use, nitric oxide and hypertension risk. Blood Press 2020, 29, 103–112. [Google Scholar] [CrossRef]
- Kuwabara, M.; Motoki, Y.; Sato, H.; Fujii, M.; Ichiura, K.; Kuwabara, K.; Nakamura, Y. Low frequency of toothbrushing practices is an independent risk factor for diabetes mellitus in male and dyslipidemia in female: A large-scale, 5-year cohort study in Japan. J. Cardiol. 2017, 70, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-Y.; Kim, S.-H.; Kang, S.-H.; Yoon, C.-H.; Lee, H.-J.; Yun, P.-Y.; Youn, T.-J.; Chae, I.-H. Improved oral hygiene care attenuates the cardiovascular risk of oral health disease: A population-based study from Korea. Eur. Heart J. 2019, 40, 1138–1145. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.C.; Ruospo, M.; Wong, G.; Craig, J.C.; Petruzzi, M.; De Benedittis, M.; Ford, P.; Johnson, D.W.; Tonelli, M.; Natale, P.; et al. Dental Health and Mortality in People With End-Stage Kidney Disease Treated With Hemodialysis: A Multinational Cohort Study. Am. J. Kidney Dis. 2015, 66, 666–676. [Google Scholar] [CrossRef] [PubMed]
- Reichert, S.; Schlitt, A.; Beschow, V.; Lutze, A.; Lischewski, S.; Seifert, T.; Dudakliewa, T.; Gawe, R.; Werdan, K.; Hofmann, B.; et al. Use of floss/interdental brushes is associated with lower risk for new cardiovascular events among patients with coronary heart disease. J. Periodontal Res. 2015, 50, 180–188. [Google Scholar] [CrossRef]
- Song, T.J.; Jeon, J.; Kim, J. Cardiovascular risks of periodontitis and oral hygiene indicators in patients with diabetes mellitus. Diabetes Metab. 2021, 47, 101252. [Google Scholar] [CrossRef]
- Song, T.-J.; Chang, Y.; Jeon, J.; Kim, J. Oral health and longitudinal changes in fasting glucose levels: A nationwide cohort study. PLoS ONE 2021, 16, e0253769. [Google Scholar] [CrossRef]
- Zhuang, Z.; Gao, M.; Lv, J.; Yu, C.; Guo, Y.; Bian, Z.; Yang, L.; Du, H.; Chen, Y.; Ning, F.; et al. Associations of toothbrushing behaviour with risks of vascular and nonvascular diseases in Chinese adults. Eur. J. Clin. Investig. 2021, 51, e13634. [Google Scholar] [CrossRef]
- Winning, L.; Patterson, C.C.; Neville, C.E.; Kee, F.; Linden, G.J. Periodontitis and incident type 2 diabetes: A prospective cohort study. J. Clin. Periodontol. 2017, 44, 266–274. [Google Scholar] [CrossRef]
- Jangam, T.G. Oral Hygiene Home Care Practices and Cardiovascular Disease Mortality: A Cohort Study; Boston University School of Medicine: Boston, MA, USA, 2017; Available online: https://open.bu.edu/bitstream/handle/2144/26760/JANGAM_bu_0017N_13238.pdf?sequence=5 (accessed on 21 July 2023).
- Kim, J.; Kim, H.J.; Jeon, J.; Song, T.-J. Association between oral health and cardiovascular outcomes in patients with hypertension: A nationwide cohort study. J. Hypertens. 2022, 40, 374–381. [Google Scholar] [CrossRef]
- Guo, D.; Shi, Z.; Luo, Y.; Ding, R.; He, P. Association between oral health behavior and chronic diseases among middle-aged and older adults in Beijing, China. BMC Oral. Health 2023, 23, 97. [Google Scholar] [CrossRef]
- Isomura, E.T.; Suna, S.; Kurakami, H.; Hikoso, S.; Uchihashi, T.; Yokota, Y.; Sakata, Y.; Tanaka, S. Not brushing teeth at night may increase the risk of cardiovascular disease. Sci. Rep. 2023, 13, 10467. [Google Scholar] [CrossRef] [PubMed]
- Matsui, S.; Maruhashi, T.; Kishimoto, S.; Kajikawa, M.; Yusoff, F.M.; Nakashima, A.; Taguchi, A.; Higashi, Y. Poor tooth brushing behavior is associated with high risk of cardiovascular events: A prospective observational study. Int. J. Cardiol. 2022, 350, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Hwang, S.; Oh, H.; Rhee, M.; Kang, S.; Kim, H.; Hwang, S.-Y.; Rhee, M.-Y.; Kim, H.-Y. Association of periodontitis, missing teeth, and oral hygiene behaviors with the incidence of hypertension in middle-aged and older adults in Korea: A 10-year follow-up study. J. Periodontol. 2022, 93, 1283–1293. [Google Scholar] [CrossRef] [PubMed]
- Huh, Y.; Yoo, J.E.; Park, S.-H.; Han, K.; Kim, S.M.; Park, H.S.; Cho, K.H.; Ahn, J.-S.; Jun, S.H.; Nam, G.E. Association of Dental Diseases and Oral Hygiene Care With the Risk of Heart Failure in Patients With Type 2 Diabetes: A Nationwide Cohort Study. J. Am. Heart Assoc. 2023, 12, e029207. [Google Scholar] [CrossRef]
- Chang, Y.; Lee, J.S.; Woo, H.G.; Ryu, D.-R.; Kim, J.-W.; Song, T.-J. Improved oral hygiene care and chronic kidney disease occurrence: A nationwide population-based retrospective cohort study. Medicine 2021, 100, e27845. [Google Scholar] [CrossRef]
- Long, M.; Wang, H.; Zhou, J.; Wu, Y.; Yu, L.; Liu, T. A prospective cohort study on the association between oral hygiene behaviors and the incidence of stroke. Chin. J. Dis. Control Prev. 2023, 27, 1167–1174. [Google Scholar] [CrossRef]
- Zhou, B.J.; Jiang, C.Q.; Jin, Y.L.; Au Yeung, S.L.; Lam, T.H.; Cheng, K.K.; Zhang, W.S.; Xu, L. Association of oral health with all-cause and cause-specific mortality in older Chinese adults: A 14-year follow-up of the Guangzhou Biobank Cohort study. J. Glob. Health 2024, 14, 04111. [Google Scholar] [CrossRef]
- Vogtmann, E.; Etemadi, A.; Kamangar, F.; Islami, F.; Roshandel, G.; Poustchi, H.; Pourshams, A.; Khoshnia, M.; Gharravi, A.; Brennan, P.J.; et al. Oral health and mortality in the Golestan Cohort Study. Int. J. Epidemiol. 2017, 46, 2028–2035. [Google Scholar] [CrossRef]
- Cho, M.J.; Kim, Y.S.; Park, E.Y.; Kim, E.K. Association between Periodontal Health and Stroke: Results from the 2013-2015 Korea National Health and Nutrition Examination Survey (KNHANES). J. Dent. Sci. 2021, 16, 268–274. [Google Scholar] [CrossRef]
- Choi, J.S. Relationship between hyperglycemia and periodontal symptoms in Korean type 2 diabetes mellitus patients aged >= 50 years. Indian. J. Public. Health Res. Dev. 2019, 10, 4387–4392. [Google Scholar] [CrossRef]
- Cinar, A.B.; Oktay, I.; Schou, L. Toothbrushing: A Link Between Noncommunicable and Communicable Diseases? Oral. Health Prev. Dent. 2015, 13, 515–522. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, C.; Watt, R.; Hamer, M. Toothbrushing, inflammation, and risk of cardiovascular disease: Results from Scottish Health Survey. BMJ 2010, 340, c2451. [Google Scholar] [CrossRef]
- Frisbee, S.J.; Chambers, C.B.; Frisbee, J.C.; Goodwill, A.G.; Crout, R.J. Research Association Between Dental Hygiene, Cardiovascular Disease Risk Factors and Systemic Inflammation in Rural Adults. J. Dent. Hyg. 2010, 84, 177–184. [Google Scholar] [PubMed]
- Fujita, M.; Ueno, K.; Hata, A. Lower frequency of daily teeth brushing is related to high prevalence of cardiovascular risk factors. Exp. Biol. Med. 2009, 234, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Hwang, S.; Shim, J.; Kang, D.; Choi, J. Poor Oral Health Predicts Higher 10-Year Cardiovascular Risk A Propensity Score Matching Analysis. J. Cardiovasc. Nurs. 2018, 33, 429–436. [Google Scholar] [CrossRef]
- Kuwabara, M.; Motoki, Y.; Ichiura, K.; Fujii, M.; Inomata, C.; Sato, H.; Morisawa, T.; Morita, Y.; Kuwabara, K.; Nakamura, Y. Association between toothbrushing and risk factors for cardiovascular disease: A large-scale, cross-sectional Japanese study. BMJ Open 2016, 6, e009870. [Google Scholar] [CrossRef]
- Yoshioka, M.; Kawashima, Y.; Noma, Y.; Fukui, M.; Yanagisawa, S.; Shirayama, Y.; Nagai, K.; Hinode, D. Association between diabetes-related clinical indicators and oral health behavior among patients with type 2 diabetes. J. Med. Investig. 2021, 68, 140–147. [Google Scholar] [CrossRef]
- Huang, Y.-T.; Lin, M.-S.; James, K.; Chang, C.-H.; Tsai, W.-H.; Lin, Y.-C.; Chen, M.-Y. From self-care behaviours to cardiometabolic risks prevention for the health of farmers: Nursing implications. J. Adv. Nurs. 2023, 79, 3025–3034. [Google Scholar] [CrossRef]
- Han, S.-J.; Son, Y.-J.; Kim, B.-H. Association between diabetes mellitus and oral health status in patients with cardiovascular diseases: A nationwide population-based study. Int. J. Environ. Res. Public. Health 2021, 18, 4889. [Google Scholar] [CrossRef]
- Moon, M.-G.; Kang, S.-H.; Kim, S.-H.; Park, S.-Y.; Seol, Y.-J.; Yoon, C.-H.; Lee, H.-J.; Youn, T.-J.; Chae, I.-H.; Leira, Y.; et al. Association between toothbrushing and cardiovascular risk factors: A cross-sectional study using Korean National Health and Nutrition Examination Survey 2015–2017. BMC Oral Health 2024, 24, 4. [Google Scholar] [CrossRef]
- VanWormer, J.J.; Acharya, A.; Greenlee, R.T.; Nieto, F.J. Oral hygiene and cardiometabolic disease risk in the survey of the health of Wisconsin. Community Dent. Oral. Epidemiol. 2013, 41, 374–384. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Leveille, S.G.; Camhi, S.M.; Shi, L. Association of oral care with periodontitis and glycemic control among US adults with diabetes. BMC Oral. Health 2023, 23, 903. [Google Scholar] [CrossRef] [PubMed]
- Janket, S.-J.; Lee, C.; Surakka, M.; Jangam, T.G.; Van Dyke, T.E.; Baird, A.E.; Meurman, J.H. Oral hygiene, mouthwash usage and cardiovascular mortality during 18.8 years of follow-up. Br. Dent. J. 2023, 3, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.; Roy, P.; Thomas, A.M.; Kaur, P.; Pandian, J.D. Periodontal disease and oral hygiene practices in patients with ischemic stroke. Chrismed J. Health Res. 2015, 2, 203–207. [Google Scholar]
- Patel, J.; Kulkarni, S.; Doshi, D.; Poddar, P.; Srilatha, A.; Reddy, K.S. Periodontal disease among non-diabetic Coronary Heart Disease patients. A case-control study. Acta Bio-Medica Ateneo Parm. 2021, 92, e2021030. [Google Scholar] [CrossRef]
- Cinar, A.B.; Oktay, I.; Schou, L. Relationship between oral health, diabetes management and sleep apnea. Clin. Oral. Investig. 2013, 17, 967–974. [Google Scholar] [CrossRef]
- Afsar, B. Sociodemographic, clinical, and laboratory parameters related with presence of regular toothbrushing in hemodialysis patients. Ren. Fail. 2013, 35, 179–184. [Google Scholar] [CrossRef]
- Hiramatsu, T.; Okumura, S.; Iguchi, D.; Kojima, H. Higher dental care is positively associated with key prognosis factors in peritoneal dialysis patients: Findings from a retrospective study. Ren. Replace. Ther. 2022, 8, 6. [Google Scholar] [CrossRef]
- Saengtipbovorn, S.; Taneepanichskul, S. Effectiveness of lifestyle change plus dental care (LCDC) program on improving glycemic and periodontal status in the elderly with type 2 diabetes. BMC Oral. Health 2014, 14, 72. [Google Scholar] [CrossRef]
- Cinar, A.B.; Schou, L. Impact of empowerment on toothbrushing and diabetes management. Oral. Health Prev. Dent. 2014, 12, 337–344. [Google Scholar] [CrossRef]
- Kaur, J.; Grover, V.; Gupta, J.; Gupta, M.; Kapur, V.; Mehta, M.; Lyle, D.M.; Samujh, T.; Jain, A. Effectiveness of subgingival irrigation and powered toothbrush as home care maintenance protocol in type 2 diabetic patients with active periodontal disease: A 4-month randomized controlled trial. J. Ind. Soc. Periodontol. 2023, 27, 515–523. [Google Scholar] [CrossRef] [PubMed]
- McGuinness, L.A.; Higgins, J.P.T. Risk-of-bias VISualization (robvis): An R package and Shiny web app for visualizing risk-of-bias assessments. Res. Synth. Methods 2021, 12, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Stanimirovic, J.; Radovanovic, J.; Banjac, K.; Obradovic, M.; Essack, M.; Zafirovic, S.; Gluvic, Z.; Gojobori, T.; Isenovic, E.R. Role of C-Reactive Protein in Diabetic Inflammation. Mediat. Inflamm. 2022, 2022, 3706508. [Google Scholar] [CrossRef]
- Pant, S.; Deshmukh, A.; GuruMurthy, G.S.; Pothineni, N.V.; Watts, T.E.; Romeo, F.; Mehta, J.L. Inflammation and Atherosclerosis—Revisited. J. Cardiovasc. Pharmacol. Ther. 2014, 19, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Chen, J.; Lan, H.; Tang, Y. Role of C-Reactive Protein in Kidney Diseases. Kidney Dis. 2022, 9, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Shang, Q.; Gao, Y.; Qin, T.; Wang, S.; Shi, Y.; Chen, T. Interaction of Oral and Toothbrush Microbiota Affects Oral Cavity Health. Front. Cell. Infect. Microbiol. 2020, 10, 17. [Google Scholar] [CrossRef]
- Deo, P.N.; Deshmukh, R. Oral microbiome: Unveiling the fundamentals. J. Oral. Maxillofac. Pathol. 2019, 23, 122–128. [Google Scholar] [CrossRef]
- Marcotte, H.; Lavoie, M.C. Oral Microbial Ecology and the Role of Salivary Immunoglobulin A. Microbiol. Mol. Biol. Rev. 1998, 62, 71–109. [Google Scholar] [CrossRef]
- Bollero, P.; Di Renzo, L.; Franco, R.; Rampello, T.; Pujia, A.; Merra, G.; De Lorenzo, A.; Docimo, R. Effects of new probiotic mouthwash in patients with diabetes mellitus and cardiovascular diseases. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 5827–5836. [Google Scholar] [CrossRef]
- Mente, A.; Dehghan, M.; Rangarajan, S.; O’Donnell, M.; Hu, W.; Dagenais, G.; Wielgosz, A.; Lear, A.S.; Wei, L.; Diaz, R.; et al. Diet, cardiovascular disease, and mortality in 80 countries. Eur. Heart J. 2023, 44, 2560–2579. [Google Scholar] [CrossRef]
- Kazemi, A.; Sasani, N.; Mokhtari, Z.; Keshtkar, A.; Babajafari, S.; Poustchi, H.; Hashemian, M.; Malekzadeh, R. Comparing the risk of cardiovascular diseases and all-cause mortality in four lifestyles with a combination of high/low physical activity and healthy/unhealthy diet: A prospective cohort study. Int. J. Behav. Nutr. Phys. Act. 2022, 19, 138. [Google Scholar] [CrossRef] [PubMed]
| First Author, Year, and Region | Focused Disease | Aim | Design | Sex (M%) | Population; Mean Age (Years) | Comorbidity CVD (Yes%) | Comorbidity DM (Yes%) | Comorbidity CKD (Yes%) | Oral Hygiene Practice(s) |
|---|---|---|---|---|---|---|---|---|---|
| Afsar 2013 [80]; Turkey | CKD | To determine regular TB frequency in HD patients and second, to determine factors related to regular toothbrushing in HD patients. | Observational | 54% | 135; 52.4 ± 13.6 | 32% | 33% | N/A | TB frequency |
| Aggarwal 2012 [34]; India | T2DM | To investigate oral health attitudes, knowledge, and behavior with regard to diabetes-related factors among adults with T2DM and their attitudes to sustaining good oral health through proper oral hygiene and regular dental check-ups. | Cohort | 53.2% | 500; not reported | Not reported | N/A | Not reported | TB frequency Fluoridated Toothpaste use Mouthwash use Interdental cleaning |
| Almas 2003 [35]; Saudi Arabia | T2DM | To assess the effect of oral hygiene instructions on periodontal disease and to assess the glycemic changes in healthy, T2DM male Saudi patients. | Cohort | 100% | 60; 42 ± 13.6 | Not reported | N/A | Not reported | TB frequency |
| Assante 2020 [33]; Brazil | ESKD | To correlate the self-reported oral health, oral hygiene, and dental assistance to clinic intercurrences with hospitalization in adult CKD patients under dialysis. | Cohort | 49% | 77; 54 ± 16.1 | 66.2% | 2.6% | N/A | TB frequency Mouthwash use Interdental cleaning |
| Chang 2020 [37]; South Korea | CVD | To investigate the association of oral hygiene indicators with atrial fibrillation and heart failure risk in a nationwide general population-based longitudinal study. | Cohort | 61.2% | 161,286; 52.2 ± 8.7 | HTN 38.9% DL 15.9% | 9% | 7.8% | TB frequency |
| Chang 2020 [32]; South Korea | DM | To assess the association between oral hygiene indicators and the risk of new-onset diabetes. | Cohort | 57.9% | 188013; 53.3 ± 8.6 | HTN 34.6% DL 16.9% | 0 | 0.2% | TB frequency |
| Chang 2021 [36]; South Korea | CVD | To investigate the relationship between the presence of periodontal disease and indicators regarding oral hygiene with occurrence for stroke in a nationwide population longitudinal cohort. | Cohort | 59.4% | 206,602; 53.5 ± 8.6 | Hypertension 50.1% DL 24.5% | 12.6% | 0.3% | TB frequency |
| Chang 2021 [58]; South Korea | CKD | To clarify the relationship between oral hygiene indicators and CKD in a nationwide general population-based cohort. | Cohort | 60.9% | 158495; 52.3 ± 8.8 | AF 0.4% HF 1.2% HTN 39% DL 24.5 | 8.8% | N/A | TB frequency |
| Cho 2021 [62]; South Korea | CVD | To investigate the association between periodontal health and stroke amount Korean adults. | Cross- sectional | 47.3% | 9497; 55.71 ± 0.17 | Stroke 2.6% HTN 23.9% | 9.4% | Not reported | TB frequency Oral hygiene product use |
| Choi 2019 [63]; South Korea | T2DM | To investigate the relationship between HbA1c level and self-reported periodontal symptoms among patients with T2DM. | Cross- sectional | 22.4% | 156; not reported | Not reported | N/A | Not reported | TB frequency TB technique |
| Cinar 2013 [79]; Turkey | T2DM | To assess the links between tooth loss, oral health behavior, T2DM, obesity, and sleep apnea among patients with T2DM. | Prospective intervention study | 38% | 165; not reported | Not reported | N/A | Not reported | TB frequency |
| Cinar 2014 [83]; Turkey | T2DM | Assess the impact of health coaching on TB self-efficacy and frequency, and effect on diabetics’ management and quality of life in comparison to health education among patients with T2DM. | RCT | Not reported | 178; not reported | Not reported | N/A | Not reported | TB frequency |
| Cinar 2015 [64]; Turkey | T2DM | To assess the correlation between a preventive oral health behavior (toothbrushing), HDL, and self-assessed quality of life-related risk factors for non-communicable diseases and communicable diseases among patients with T2DM. | Cross-sectional | Not reported | 178; not reported | Not reported | N/A | Not reported | TB frequency |
| deOliveira 2010 [65]; Scotland | CVD | To investigate whether self-reported frequency of TB (as a proxy of periodontal disease) was associated with risk of CVD events in a sample of adults from the Scottish Health Survey. To also examine the association between frequency of TB and inflammatory markers in a subset of participants. | Cross-sectional | 46% | 11869; 50.0 ± 11.0 | HTN: 24% | 2.6% | Not reported | TB frequency |
| deSouza 2014 [38]; Brazil | CKD | To assess the prevalence and impact of oral health parameters, periodontitis, and its treatment on survival in a group of patients undergoing hemodialysis. | Cohort | 64.8% | 122; 50 ± 13 | 23% HTN 75% | 21% | N/A | Dental floss use |
| Frisbee 2010 [66]; United States | CVD | To investigate the associations between self-reported dental hygiene practices and overall dental health, cardiovascular risk factors and systemic inflammation, in adults living in rural communities. | Cross-sectional | 37.5% | 128; 41.5 ± 9.3 | Not reported | Not reported | Not reported | TB frequency Flossing frequency |
| Fujita 2009 [67]; Japan | CVD, DM | To investigate the relationship between frequency of daily teeth brushing and prevalence of cardiovascular risk factors. | Cross-sectional | 37% | 54,551; not reported | HTN 41.5% | 7.8% | Not reported | TB frequency |
| Guo 2023 [53]; China | CVD, DM | To evaluate the association between oral health behavior and various chronic diseases among middle-aged and older Chinese adults. | Cohort | 48% | 18,158; 61.3 ± 10.3 | 50.24% | 40.2% grouped as diseases of the endocrine or nutritional metabolic. | TB frequency | |
| Han 2021 [72]; South Korea | CVD, DM | To elucidate the prevalence of oral health complications and the relationship between DM and oral health status in diabetic patients with CVD. | Cross-sectional | 42.9% | 3495; not reported | Stroke 6.6% MI 3% HTN 71.2% DL 56.9% | 24.2% | 0.05% | TB frequency |
| Hiramatsu 2022 [81]; Japan | CKD | To investigate whether dental care in peritoneal dialysis patients might affect the incidence of CVD and infections such as peritonitis. | Retrospective observational | 57.6% | 165; Group A = 63.9 ± 12.0 Group B = 64.2 ± 10.5 | Not reported | 20% | N/A | TB frequency Dental floss frequency Mouth wash frequency |
| Hirano 2022 [39]; Japan | CKD | To evaluate the relationship between toothbrushing frequency and kidney function. | Cohort | 50% | 76,472; 45.9 ± 12.4 | HTN 7.6% | 2.24% | N/A | TB frequency |
| Huang 2023 [71]; Taiwan | CVD | To explore farmers’ self-care behaviors, including oral hygiene, remaining natural teeth, cardiometabolic risks, hepatitis, risk of stroke, and their determinant factors | Cross-sectional | 54.8 | 183; 66.9 ± 11.7 | Not reported | Not reported | Not reported | Dental floss use |
| Huh 2023 [57]; South Korea | CVD, T2DM | To evaluate the association of dental diseases and oral hygiene care (alone or in combination) with incident HF among patients with T2DM. | Cohort | 72.4% | No PD: 62.8 ± 11.1 Yes PD: 65.3 ± 10.1 | 5.4% | N/A | 0.84% | TB frequency |
| Hwang 2018 [68]; South Korea | CVD | To examine whether periodontal disease and/or poor oral health behavior predicted 10-year general cardiovascular risk among Korean adults with no CVD history using representative national data. | Cross-sectional | 41.2% | 8370; normal: 46.37 ± 11.92 At risk of CVD: 67.71 ± 10.6 | HTN 22.4% DL 12.6% | 8.29% | Not reported | TB frequency |
| Hwang 2022 [56]; South Korea | CVD | To investigate the association of periodontitis, missing teeth, and oral hygiene behaviors with the incidence of hypertension | Cohort | 55.1% | 104,349; 51.1 ± 8.2 | Not reported | 4.3% | Not reported | TB frequency |
| Isomura 2023 [54]; Japan | CVD | To investigate whether the timing of TB affects the risk of cardiovascular diseases. | Cohort study | 58.2% | 1583; reported as group medians Group MN: 65 Group night: 66 Group M: 68 Group none: 63 | N/A | Not reported | Not reported | TB frequency |
| Jain 2015 [77]; India | CVD | To find the prevalence of periodontal disease in stroke patients and compare with age- and gender-matched controls and to compare the oral hygiene practices followed by stroke patients and controls. | Case–control | 59.2% | 216; 59 ± 12 (Stroke) 58 ± 9 (Control) | Stroke group only: AF 3.7% Hypercholesterolemia 9.25% HTN 72% Carotid stenosis 1.85% CAD 17.6% RHD 2.77% | 44.4% (stroke group only) | 0 | TB frequency, device used, technique, timing, toothpaste use |
| Jangam 2017 [51]; United States | CVD | To examine the association between oral hygiene home care habits and cardiovascular mortality. | Cohort (part of a Ph.D. Thesis) | 63.6% | 506; 60 | 50.6% Hypertension 33.4% | 9.8% | Not reported | TB frequency, instrument used, dental floss use, mouthwash use |
| Janket 2023 [76]; Finland | CVD | Primary: To determine if brushing and flossing affect the risk of CVD mortality in multivariable adjusted models. Secondary: To determine if mouthwash usage has independent impact on CVD mortality. To determine if mouthwash usage affects some periodontal pathogens and cariogenic bacteria proportions. | Case–control | 68% | 359; Seldom/no brushing: 56.3 ± 8.6 Daily brushing only: 58.0 ± 9.6 Daily brushing and flossing: 58.9 ± 8.0 | CAD 44% HTN 31% | 7.34% | Yes, number not reported | TB frequency Dental floss use Mouthwash use |
| Joshipura 2017 [41]; Puerto Rico | DM | To evaluate longitudinally the hypothesis that regular over-the-counter mouthwash use was associated with increased risk of pre-diabetes/diabetes over a three-year period. | Cohort | 25.8% | 945; 50.6 ± 6.8 | Pre-hypertensive 30.7% HTN 47.2% | Prediabetic: 56.7% | 0 | Mouthwash use |
| Joshipura 2020 [42]; Puerto Rico | CVD | To evaluate if routine over-the-counter mouthwash use increases hypertension risk. | Cohort | 22% | 540; 49.0 ± 6.5 | 0 | 6.1% | 0 | Mouthwash use |
| Kaur 2023 [84]; India | T2DM | To evaluate the efficacy of subgingival home irrigation using water along with powered toothbrushes in diabetic patients with chronic periodontitis. | RCT | Not reported | 31; Group A 50.13 ± 6.6 Group B 55.07 ± 9.83 | 0 | N/A | 0 | TB instrument used Mouthwash use (Subgingival irrigation) |
| Kim 2022 [52]; South Korea | CVD | To investigate whether oral hygiene indicators were linked to the development of major CVDs in hypertensive patients in a longitudinal study setting. | Cohort | 63.25% | 52,677; 54.99 ± 9.51 | AF 0.90% | 7.21% | 2.88% | TB frequency |
| Kobayashi 2020 [40]; Japan | CVD | Evaluate the association between the frequency of daily toothbrushing and subsequent cardiovascular events. | Cohort | 50.3% | 71,221; 45.6 ± 12.2 | HTN 7.4% DL 4% | 2.1% | Not reported | TB frequency |
| Kuwabara 2016 [69]; Japan | CVD, DM, CKD | To clarify the association between TB and risk factors for CVD: HT, DM, DL, HUA, and CKD | Cross-sectional | 49% | 85,866; 47 ± 11.5 | HTN 15.9% DL 36.3% | 4.3% | CKD 3.4% HUA 13.5% | TB frequency |
| Kuwabara 2017 [43]; Japan | CVD, DM | To clarify the relationship between TB practices and the risk factors for CVD: DM, DL, HT, and HUA. | Cohort | 48.48% | 13,070; not reported | HTN 19.9% DL 39.2% | 4.4% | HUA 14.6% | TB frequency |
| Long 2023 [59]; China | CVD | To explore the relationship between oral health behavior and the incidence of stroke in Guizhou Province, China. | Cohort | 47.5% | 7970; 44.50 ± 15.15 | HTN 26% | 17.8% | Not reported | TB frequency |
| Luo 2022 [30]; United States | DM | To assess the association between inflammation and oral health and diabetes, as well as the mediating role of oral hygiene practice in this association. | Cohort | Not reported | 2192; 64.5 (Elevated CRP) 62.4 (Non-elevated CRP) | 0 | No exact numbers reported | 0 | Dental floss use |
| Moon 2024 [73]; Korea | CVD | To explore the association between cardiovascular risk and frequency of toothbrushing in the context of traditional risk factors and inflammatory markers. | Cross-sectional | 48.3% | 13761; 51.3 ± 0.2 | HTN 30.6% | 11% | Not reported | TB Frequency |
| Matsui 2022 [55]; Japan | CVD | To determine the association of toothbrushing behavior assessed in detail with the incidence of future cardiovascular events in a general population including patients with CVD. | Prospective observational | 68.9% | 692; 63 ± 16 | HTN 72% DL 60% CAD 19% HF 6.4% Stroke 8.4% Prior CABG 11% | 27% | 16% reported as dialysis | TB frequency and duration |
| Palmer 2015 [45]; Europe arm: (France, Hungary, Italy, Poland, Portugal, and Spain); and South American arm: (Argentina) | CKD | To investigate whether oral disease using standardized assessments of dental disease and preventative dental health practices are associated with early death in patients undergoing haemodialysis. | Cohort | 57.7% | 4205; 61.6 ± 15.6 | MI: 12.5% Stroke: 10.3% | 32% | N/A | TB frequency and duration Dental floss use Mouthwash use |
| Park 2019 [44]; South Korea | CVD | To evaluate whether oral hygiene behavior can alleviate cardiovascular risk associated with oral health status using a nationwide population-based cohort. | Cohort | 58% | 247,969; 52 | HTN 19.9% DL 24% | 6% | 0.7% | TB frequency |
| Patel 2021 [78]; India | CVD | To assess and compare the periodontal health status among CHD patients with age- and gender-matched controls. | Case–control | 63.6% | 1616; 48.30 ± 7.73 | 50% | 0 | Not reported | TB frequency and method |
| Reichert 2015 [46]; Germany | CVD | To investigate whether oral hygiene habits, severe periodontitis, presence of periodontopathogens in the subgingival biofilm, or certain IL-6 genotypes represent independent risk factors for the incidence of new cardiovascular events among inpatients suffering from CHD. | Cohort | 74% | 942; 68.8 (median) | HTN 87.6% | 34.20% | Not reported | Dental floss or interdental brush use TB frequency |
| Saengtipbovorn 2014 [82]; Thailand | T2DM | To assess the effectiveness of a Lifestyle Change plus Dental Care program to improve glycemic and periodontal status in the elderly with T2DM. | Quasi-experimental RCT | 34.35% | 131; Intervention: 63.83 ± 4.51 Control: 64.06 ± 5.53 | 0 | N/A | 0 | TB Interdental cleaning Mouth rinse use |
| Song 2021 [47]; South Korea | DM, CVD | Evaluate periodontitis and poor oral hygiene as independent risk factors for either cerebral or MI in the diabetes population. | Cohort | 64.75% | 17,009; 55.65 ± 9.66 | 0 | N/A | 0 | TB frequency |
| Song 2021 [48]; South Korea | DM (risk factor) | To assess the association between oral hygiene indicators of periodontitis, tooth loss, and TB with longitudinal fasting glucose levels in non-diabetic subjects. | Cohort | 58% | 91,963; 56.16 ± 7.6 | HTN 36.2% | Not reported | 9.1% | TB frequency |
| VanWormer 2013 [74]; United States | DM | To examine the degree oral hygiene habits were associated with CVD and T2DM risk levels among American adults. | Cross-sectional | 43% | 1008; not reported | 0 | 0 | 0 | TB and flossing frequency |
| Vogtmann 2017 [61]; Korea | CVD | To test the association between oral health, oral care, and denture use with overall and cause-specific mortality. | Cohort | 46.4% | 50024; 51.9 ± 8.9 | HTN 30.6% | 11% | Not reported | TB frequency |
| Wang 2022 [31]; China | DM, CVD (risk factor) | To investigate the relationship between oral health and incident hypertension/T2DM. | Cohort | 47.5% | 8139; 44.52 ± 15.16 | HTN 26.1% DL 57.7% | 8.4% | Not discussed | TB frequency |
| Winning 2017 [50]; UK | T2DM | The aim of this study was to investigate periodontitis as a risk factor for incident T2DM in a group of men aged 58–72 years. | Cohort | 100% | 63.7 ± 3.0 | ACVD 9.09% HTN 29.45% | 0 | Not reported | TB frequency |
| Yoshioka 2021 [70]; Japan | T2DM | To clarify the association between oral health behavior and diabetes-related clinical indicators among patients with T2DM. | Cross-sectional | 74.9% | 74; 63.7 ± 12.4 | HTN medications 56.8% | N/A | Not reported | TB timing |
| Zhang 2023 [75]; United States | DM | To examine the associations between oral health behaviors, specifically flossing and preventive dental care, and periodontitis and glycemic control, among US dentate adults with diabetes. | Cohort | 51.1% | 892; 60.0 ± 0.4 | Not reported | 100% | Not reported | Flossing |
| Zhuang 2021 [49]; China | CVD, T2DM, CKD | To investigate the associations between a self-reported measure of TB behavior [as a proxy of oral hygiene] and the risk of cardiovascular events, as well as examine the association of TB behavior with nonvascular diseases such as DM and CKD. | Cohort | 41% | 487,198; 51.5 ± 10.6 | 0 | 3.2% | Not reported | TB frequency |
| Zhou 2024 [60]; China | CVD | To examine the associations of oral health with all-cause and cause-specific mortality in middle-aged and older Chinese adults. | Cohort | 27.6% | 28006; 62.0 ± 7.1 | Not reported | 14.6% | Not reported | TB frequency |
| First Author and Year | Strategy | Comparator/Variable | Findings |
|---|---|---|---|
| Afsar 2013 [80] | OR and 95% CI | Regular TB vs. Irregular TB | TB was infrequent in study participants. Depression, cognitive function, education, and chronic inflammation influenced TB in participants. Serum hs-CRP levels showed inverse association with regular TB. |
| Aggarwal 2012 [34] | % and p values. | Good DM Control HbA1c ≤ 6.0% Moderate DM Control HbA1c 6.1–7.5% Poor DM Control HbA1c ≥ 7.5% | Maintenance of good oral hygiene was poor among participants. 49% reported TB once daily. 2× daily TB was lowest within the poor HBa1c category. Patients without DM complications had a higher rate of 2× daily TB (p < 0.001). Only 38.4% patients were aware that diabetes affects oral health. 52.8% reported that their dentist did not know they had diabetes. |
| Almas 2003 [35] | Mean ± SD, %, and p values. | Group 1: Healthy [non-diabetic] with periodontal disease Group 2: T2DM with early to moderate periodontitis Group 3: T2DM with advanced periodontitis | HbA1c and GCF levels decreased significantly after one week of oral hygiene instructions: HbA1c G1: p < 0.004 G2: p < 0.002 G3: p < 0.000 GCF G1, G2, G3: p < 0.000 The severity of periodontal disease increased with an increase in HbA1c and GCF levels. * Limited change in CPITN measurements. * Mean plaque score decreases in all groups, with the largest in group 3. Structured oral hygiene instructions can have a positive effect on the metabolic control of type 2 diabetes. * Exact numbers not reported. |
| Assante 2020 [33] | Pearson’s Correlation [r] | TB times per day Use of Dental Floss Use of Mouthwash | Strong correlations between oral hygiene habits and intercurrent hospitalization of ESKD patients undergoing dialysis. Those not using dental floss and mouthwash needed 7.7% and 30.8% more hospitalizations than those who were using floss and mouthwash, respectively. Negative correlation [r = −0.89] between times of TB per day and patient hospitalization. Negative correlation [r = −0.98] between oral health self-report and patient hospitalization. Positive correlation [r = 0.9] between number of remaining teeth and oral diseases. |
| Chang 2020 [37] | HR Kaplan–Meier survival curves | TB times/day: 0–1× daily, 2× daily, ≥3× daily. | The risk of AF and HF was lower in groups with frequent TB. Fewer teeth was associated with increased risk of AF and HF. Kaplan–Meier survival curves showed that the risk for AF and HF was lower in subjects without periodontal disease. Frequent TB (≥3× daily) was significantly associated with attenuated risk of AF and was related with decreased risk of HF occurrence. Optimal oral health, gained by frequent TB may decrease risk of AF and HF. |
| Chang 2020 [32] | HR | Periodontal disease No Yes Teeth brush times/day 0–1 2 ≥3 Dental visit history No yes Dental scaling No Yes Number of missing teeth 0 1–7 8–14 ≥15 | 16.1% of cohort developed new-onset DM at the 10-year follow-up. Frequent TB may reduce the risk of new-onset DM. The presence of periodontal disease and lower remaining teeth may increase the risk of new-onset DM. After accounting for all confounders: Frequent TB (≥3× daily) was negatively correlated with occurrence of new-onset DM (p < 0.001). Periodontal disease was associated with new-onset DM (p < 0.001). Number of missing teeth (≥15 teeth) was positively associated with occurrence of new-onset DM (p < 0.001). |
| Chang 2021 [36] | HR Kaplan–Meier survival curves | Periodontal disease No Yes Teeth brush times/day 0–1 2 ≥3 Dental visit history No yes Dental scaling No Yes Number of missing teeth 0 1–7 8–14 ≥15 Number of dental caries 0 1–3 ≥4 | Frequent TB (≥3× daily) was related to decreased risk of stroke (p < 0.001) In the sensitivity analysis, frequent TB was associated with risk of cerebral infarction. Only TB frequency (≥3× daily) was associated with risk of cerebral and subarachnoid hemorrhage in the multivariable analysis. Kaplan–Meier survival curves demonstrated that the risk for stroke depended on number of daily TB, dental visit history, dental scaling, number of teeth loss, and number of dental caries. Regular and frequent daily oral hygiene care were negatively and infrequent oral hygiene care positively related with risk of occurrence for stroke, respectively. |
| Chang 2021 [58] | t-tests and logistic regression analysis were performed for statistical analysis | Periodontal disease No Yes Teeth brush times/day 0–1 2 ≥3 Tooth loss 0 1–7 8–14 15–21 ≥22 Professional scaling No Yes | Increased TB frequency may reduce the risk of CKD. Kaplan–Meir survival curves showed periodontal disease and fewer remaining teeth were positively related with an elevated risk of CKD (p =< 0.001). The risk of CKD had a negative correlation as the frequency of TB (p =< 0.001). Increased number of TBs (≥3 times a day) was inversely associated with the development of CKD. The subgroup analysis found having BMI ≥ 25 and TB at least 3 times a day were more strongly associated with occurrence of CKD (p for interaction < 0.05). |
| Cho 2021 [62] | OR Logistic regression analysis | Daily TB 0 1–2 ≥3 Use of oral hygiene products 0 1–2 ≥3 Self-perceived oral health Poor Good Tooth ache Yes No Chewing level Poor Fair Good Untreated tooth Poor Good Number of residual teeth 0–19 ≥20 CPI 4 3 2 1 0 | Daily TB frequency had a significant negative effect impact on periodontal health and stroke. The stroke group showed lower daily TB frequency and less use of other oral hygiene products than the no-stroke group (p < 0.001). |
| Choi 2019 [63] | OR | TB/day ≤2 ≥3 TB technique Non-rolling stroke rolling stroke Recent dental checkups <2 years ago >2 years ago Interproximal cleaning devices No Yes Periodic dental scaling No Yes HbA1c level Good control [<7.0] Poor control [≥7.0] | HbA1c control and experiencing periodontal symptoms were correlated in patients ≥ 50 with T2DM. Participants who brushed their teeth ≥ 3 times daily had more periodontal symptoms (p < 0.05). Participants with poorly controlled HbA1c level had more periodontal symptoms than those with good control (p < 0.05). * * Authors do not disclose/define periodontal symptoms. Does not correlate with TB frequency and T2DM. |
| Cinar 2013 [79] | Pearson’s Correlation (r) | No. teeth lost in upper jaw <mean ≥mean No. teeth lost in lower jaw <mean ≥mean Total no. teeth lost <mean ≥mean TB frequency Daily Less than daily Gingival bleeding on TB Rare At least occasionally | Oral care with early diagnosis and monitoring of glycemic level can help prevent complications of T2DM. TB frequency was negatively correlated with fasting blood glucose (rs = 0.18, p < 0.005) and positively with HDL (rs = 0.23, p < 0.001]. Patients who TB less than daily have an increased risk of HDL (≤39 mg/dL, %), 78%, compared to those who TB daily, 53% (p < 0.05). Patients who TB less than daily have an increased risk of LDL (≥95 mg/dL, %), 50%, compared to those who TB daily, 45%. Patients who TB less than daily have an increased risk of HbA1c (≥6.5%), 79%, compared to those who TB daily, 68%. Patients who TB less than daily have an increased risk of probability of sleep apnea, 47%, compared to those who TB daily, 30% (p < 0.05). |
| Cinar 2014 [83] | Compared numerical data, percentage, and used p values. | Health Coaching Health Education | 70% of HC participants increased their TB 2× daily (p < 0.05). 0.6% improvement in HbA1c levels observed in the HC group (p < 0.05). HC is more effective at improving TB behavior than HE, contributing significantly to DM management. |
| Cinar 2015 [64] | OR Pearson’s Correlation (r) | TB frequency Daily Less than daily Gingival bleeding on TB Favorable Unfavorable | 27% of participants brushed 2× daily. Self-reported gingival bleeding was negatively correlated with QoL and TB frequency. TB was positively correlated with HDL and QoL. Participants who brushed < 1× daily were more likely to have unfavorable HDL and QoL than those who brushed daily [OR = 1.84 CI = 1.02–3.32 vs. OR 1.53 CI = 1.07–2.18, p < 0.05]. |
| deOliveira 2010 [65] | HR Regression models | TB frequency 2× daily 1× daily <1× daily | TB is associated with CVD. Participants who reported less frequent TB had a 70% increased risk of a cardiovascular disease event. Less frequent TB was associated with increased concentrations of CRP and fibrinogen. |
| deSouza 2014 [38] | Uni- and multivariate analysis HR Kaplan–Meier Curves | Frequency of dental visits Rarely Annually/biannually Quarterly Use of dental floss Decayed teeth Filled teeth | Oral health status was poor amongst participants. Dental floss use was irregular in 40% of participants. The univariate analysis showed a lack of dental floss use and low frequency of visits to the dentist were positively associated with an increased risk for patient mortality (HR 1.99 [0.85–4.14] p = 0.066). After the multivariate analysis, the significance of this correlation disappeared (HR 0.19 [0.2–3.10] p = 0.731). |
| Frisbee 2010 [66] | Univariate analysis using Pearson’s chi-square statistic. Univariate ANOVA analysis | Frequency of TB At least daily ≤1× daily Frequency of flossing 2–6 times a week ≤1× a week Frequency of dental care Every 6 months Less than 6 months | Dental hygiene (defined as TB, flossing preventative care) was significantly related to total cholesterol, mean arterial pressure, adiponectin, CRP, fibrinogen, IL-1B, and tPAI-1. In both analyses, there was a statistically significant independent association between dental hygiene and systemic inflammation. Systemic inflammation from periodontal disease could begin before the onset of the clinical signs associated with poor dental hygiene. Better overall dental health was associated with lower levels of CRP levels and higher levels of adiponectin and fibrinogen. |
| Fujita 2009 [67] | Kappa coefficient was calculated. Pearson’s chi-square tests OR | Frequency of TB After every meal 1–2 times a day Hardly ever | Lower frequency of daily TB was significantly related to higher prevalence of DM and high TG and/or low HDL-C in both men and women, as well as a higher prevalence in hypertension for women. Total cholesterol status was not significantly affected by TB frequency. |
| Guo 2023 [53] | Multivariate logistic models Logistic regression OR | Frequency of TB ≥2× daily ≤1× daily | Poor oral hygiene practices were associated with higher risk of chronic diseases. TB ≤1× daily increased the risk of hypertension, ischemic heart disease, cerebrovascular disease, and diabetes. |
| Han 2021 [72] | Kolmogorov−Smirnov test Chi-square test, Independent t-test Logistic regression | No DM DM | The DM group had a significantly higher number of people with a lower TB frequency (p < 0.001) compared with the non-DM group. There was a correlation between increased prevalence of oral health diseases and fewer remaining teeth in those living with the combination of CVD and DM. The incidence of periodontitis was found to be 1.4 times higher in the DM group compared to the non-DM group after adjusting for confounding variables. |
| Hiramatsu 2022 [81] | Compared numerical data, percentage, and used p values. ANOVA testing Regression analysis | Dental care score ≥ 7 Dental care score < 7 | Oral hygiene practices were grouped and given a dental score. These scores then stratified the groups by Group A (≥7-good dental care) and Group B (<7 poor dental care). Participants in Group B had lower daily TB frequency than those in Group A [1.33 ± 0.58 vs. 2.54 ± 0.50 p = 0.001]. Increased albumin and lower CRP were associated with Group A. The multiple regression analysis found TB frequency was the most important factor for all-cause peritonitis [t value = −2.182, p = 0.048], streptococcal peritonitis [t value = −2.739, p = 0.006], CHF [t value = −1.739, p = 0.059], and pneumonia [t value = −2.916, p = 0.004]. |
| Hirano 2022 [39] | Multivariate analysis Compared numerical data and percentage and used p values. | TB frequency Once a day or not everyday 1–2 times/day After every meal | Participants who brushed 1–2× daily had a lower prevalence of cancer and DM. Participants who brushed after every meal had a lower prevalence of hypertension. Those who brushed their teeth 1–2× daily had a higher eGFR and milder stage of CKD than those who brushed their teeth ≤ 1× daily. Those who brushed their teeth after every meal fell within a more severe CKD risk category, suggesting more severe proteinuria, than those who brushed their teeth 1–2× daily. Frequent TB was associated with a decreased risk of composite renal outcomes defined by a 25% eGFR reduction from baseline values, an eGFR of <15 mL/min/1.73 m2 (stage 5 CKD), and a requirement for regular dialysis. TB 1–2× daily was associated with better kidney function than TB after every meal. |
| Huang 2023 [71] | Compared numerical data and percentage and used p values. Hierarchical multiple linear regression analyses | Dental floss use before bed Seldom/occasional Usually/always | Having a lower remaining teeth was significantly associated with seldom or occasional dental floss use after dinner/before bed [t = 4.93, p < 0.001], and high risk of stroke [t = 8.38, p < 0.001]. The regression analyses showed that the determinant factors associated with the remaining natural teeth were high risk of stroke [t = −2.6, p < 001], and dental floss use [t = 1.98, p < 0.05]. |
| Huh 2023 [57] | Kaplan–Meier curves HR Compared numerical data | Periodontal disease No Yes Dental caries No Yes No. missing teeth 0 1–7 8–14 ≥15 Professional dental cleaning <1 yearly ≥1 yearly Daily TB 0–1 ≥2 | Oral diseases and lower TB frequency were associated with HF incidence amongst patients with T2DM. HR for HF decreased by 10% among participants who brushed their teeth ≥ 2 daily (HR 0.90 [95% CI, 0.82–0.98] p = 0.018). Individuals with ≥15 missing teeth and lower TB frequency had an increased HF risk (HR 1.50 [95% CI, 1.09–2.06] for 0–1 time of daily TB). These associations were not observed among individuals with ≥15 missing teeth and higher TB frequency (HR 1.16 [95% CI, 0.92–1.47] for ≥2 times of daily TB). The HRs for HF increased by 20% among individuals with both periodontal diseases and dental caries (HR 1.20 [95% CI, 1.01–1.43]; 5-year RD 0.20, SE 0.10). |
| Hwang 2018 [68] | OR Compared numerical data and percentage and used p values. | Perceived oral health. Good Moderate Not good Diagnosed periodontal disease Yes No Frequency of TB ≥3 times a day <3 times a day Dental visit experience in a year Yes No Using oral hygiene supplies Yes No Preventative dental treatment Yes No | TB < 3 times per day was an independent predictor of intermediate/high CVD risk. Diagnosed periodontal disease and low TB frequency predicted a higher 10-year cardiovascular risk among people with no history of CVD. |
| Hwang 2022 [56] | HR Log-rank test Compared numerical data and percentage and used p values. | Periodontal disease No Yes Number of missing teeth 0 1–7 8–14 ≥15 Dental scaling No Yes Teeth brush times/day 0–1 2 ≥3 | TB frequency (≥3 daily) at baseline (HR: 0.85) was significantly associated with incident hypertension. Periodontitis was significantly associated with incident hypertension (HR: 1.02; 95% CI: 1.00–1.04). The risk of hypertension was lower in the higher frequency TB group (≥3 vs. 0–1 times/day; HR: 0.85; 95% CI: 0.83–0.88; p for trend < 0.0001). In the middle-aged group (40–64), frequency of TB [for ≥3 group; HR: 0.85; 95% CI: 0.83–0.88] was significant. In the older group (≥65 years), weaker effects were observed for frequency of TB (for ≥3 group; HR: 0.93; 95% CI: 0.87–0.99). Protective effects of TB disappeared in the group with fewer remaining teeth and periodontitis. |
| Isomura 2023 [54] | Kaplan–Meier curves HR Compared numerical data | TB frequency: Group MN (morning and night) Group M (morning only) Group Night (night only) Group None (not brushing) | Univariate and multivariate analyses of cardiovascular events showed significantly higher survival estimates in Groups MN (p = 0.021) and Night (p = 0.004) than in Group None. TB only in the morning is inadequate to prevent CVD. There were no significant differences in cardiovascular events, hospitalization events, or life prognosis among the groups based on the number of teeth, caries, or periodontal disease. |
| Jain 2015 [77] | Chi-square and Fisher’s exact tests OR | Type of teeth cleaning Toothbrush Finger/twig Method of cleaning Horizontal Vertical/circular Material used Toothpaste Other Frequency of cleaning/day Once More than once Time of TB Before meals After meals Decayed teeth Missing teeth Filled teeth | Those living with stroke were more likely to use finger/twig to clean their teeth compared to the control group (17.4% vs. 1.9% [p ≤ 0.0001]). Those living with stroke were less likely to brush more than once a day compared to the control group (7.7% vs. 26.9% [p < 0.0001]). Those living with stroke had a greater number of missing teeth compared to control group [(4.02 ± 5.85) vs. (1.81 ± 3.89) (p = 0.002)]. Those with poor oral hygiene practices were significantly more likely to develop stroke and periodontal disease. |
| Jangam 2017 [51] | Univariate analysis T-test/Chi-squared test Multiple regression model Logistic regression model OR p value | Oral Hygiene Score 0 = low 1 = medium 2 = high Dental check-ups 1 = once a year >1 = longer duration between check-ups Electronic toothbrush and sweets Yes No Sugary beverage intake Yes No Number of teeth | An inverse correlation exists between oral hygiene habits and CVD mortality (p = 0.02). All oral hygiene habits, particularly flossing (p = 0.03), decreased the risk of CVD outcomes. * There was an association between individual oral habits and CVD: flossing, TB, number of teeth, last dental visit, and use of electronic toothbrush were each associated with CVD. However, mouthwash use (p = 0.60) was the only oral habit that had no independent association with CVD. This study found that the odds of a person with a high OH score (OR = 0.21) dying due to CVD were lower compared to those with a low oral hygiene score (OR = 0.59). * CVD outcomes not defined. |
| Janket 2023 [76] | HR Compared numerical data Kaplan–Meier curves | Oral hygiene self-care practice: Better (daily TB and flossing) Good (daily TB only) Poor (no TB/flossing) Baseline CAD Mouthwash use No Yes | Daily TB and flossing had significantly greater benefits on CVD mortality risk, over TB alone. CVD mortality risk was the lowest in the best OHS group (both TB and flossing) (HR = 0.25 [CI: 0.07–0.89]; p = 0.03) and in the TB-only group (HR = 0.72 [CI: 0.37–1.41]; p = 0.34). In a stratified analysis, the CAD group had enough CVD mortality and beneficial effects of OHS remained (HR = 0.50 [0.24–1.06]; p = 0.07). CRP was marginally significant between groups. SLZ (an innate oral immunity marker) had a significant inverse correlation with OHS. The effect of independent mouthwash usage on CVD mortality was not meaningful (HR 0.95 [0.45–2.01]; p = 0.89). |
| Joshipura 2017 [41] | Poisson regression models IRR Percentage comparison Confidence intervals | Mouthwash use frequency Never <1× weekly 1 time per week 2 times per week 1–3× weekly 4–6× weekly 1× daily 2× daily ≥2× daily | Controlling for major DM risk factors, participants who used mouthwash ≥ 2× daily had a 55% increased risk of developing pre-DM or DM over a 3-year follow-up compared to less frequent users, and 49% higher risk compared to non-users of mouthwash. The authors hypothesize that the increased risk for diabetes among mouthwash users in their study is due to direct effects on the oral microbiome. |
| Joshipura 2020 [42] | Poisson regression models IRR Linear regression Percentage comparison Confidence intervals | Mouthwash use frequency ≤6× weekly 1× daily ≥2× daily | People who used mouthwash ≥2× daily had an 85% higher risk of being diagnosed with hypertension compared to less frequent users, and more than twice that of non-users. In unadjusted models, ≥2× daily use of mouthwash was associated with an increased risk of being diagnosed with hypertension (IRR = 1.87, 95% CI: 1.07, 3.27), compared to no use. Only using mouthwash 1× daily was associated with increase in SBP over follow-up (β = 3.98, 95% CI: 0.83, 7.13) compared to no use. Compared to no mouthwash use, its use once daily (β = 2.31, 95% CI:0.24, 4.38) and twice daily (β = 2.29, 95% CI: 0.30, 4.27) were associated with increased DBP. Almost all types of mouthwash are likely to have a detrimental impact on nitrate reducing bacteria, which may increase BP. |
| Kaur 2023 [84] | Mean ± SD values Independent samples t-test Pairwise inter group comparison Mann–Whitney test Spearman correlation | Mouthwash: Group A:Waterpik + 0.06% CHX Group B: CHX rinse 0.12% TB: Group A: Powered toothbrush Group B: Manual | Significant reduction in HbA1c levels at final follow-up (4 months) in both groups compared to baseline; Group A p = 0.003, Group B p = 0.008. Non-significant reduction in CRP and IL-B at final follow-up from baseline. |
| Kim 2022 [52] | HR Cox proportional hazard regression Compared numerical data and percentage and used p values. Cumulative incidence curves | Periodontitis No Yes Number of missing teeth 0 1–4 ≥5 Number of dental caries 0 1–4 ≥5 Teeth brush times/day 0–1 ≥2 | Frequency of TB per day (at least twice) was significantly related to a lower risk of major cardiovascular events (aHR: 0.88; 95% CI: 0.81–0.96; p = 0.002). In the secondary analysis for individual cardiovascular outcomes, TB frequently (at least two times per day) was associated with a lower risk of all stroke (aHR: 0.87; 95% CI: 0.79–0.96; p = 0.004) and hemorrhagic stroke (aHR: 0.77; 95% CI: 0.64–0.92; p = 0.003) compared with TB teeth 0–1 times per day. |
| Kobayashi 2020 [40] | Bivariable analyses Chi-square tests OR | Frequency of TB Not every day Once a day Once to twice a day After every meal | Frequent TB was inversely associated with subsequent cardiovascular events in a dose-dependent manner. Participants who brushed 1× daily may have an increased risk only for stroke, but not for ACS, compared to those who brushed their teeth after every meal. Less frequent TB may be a marker for subsequent CVD and CHD, rather than a risk factor. |
| Kuwabara 2016 [69] | ANOVA Binominal logistic regression models OR p value and confidence intervals | TB practices After every meal At least 1× daily <1× daily | Adjusted analyses found low-frequency TB was significantly associated with high prevalence of T2DM (OR = 2.03) and dyslipidemia (OR = 1.50) but not with hyperuricemia and CKD. Higher TB frequency may be beneficial for improving oral health and preventing some systemic diseases. |
| Kuwabara 2017 [43] | Logistic regression analysis OR Percentage comparison p value | TB practices After every meal At least 1× daily <1× daily | Low-frequency TB was an independent risk factor for developing DM in males (OR: 1.43) and for developing DL in females (OR: 1.18). Low-frequency TB was not a risk factor for developing DM in females, DL in males, hypertension, and HUA. TB practices may be beneficial to reduce CVD risk factor development. |
| Long 2023 [59] | Cox regression, HR | TB Frequency <1× daily 1× daily ≥2× daily | Compared to <1× daily brushing, the risk of all-cause stroke decreased by 34% (HR = 0.65, 95% CI: 0.45–0.94) for those TB 1× daily and by 50% (HR = 0.49, 95% CI: 0.30–0.78) for those TB ≥ 2× daily. Compared to <1× daily TB, the risk of ischemic stroke decreased by 29% (HR = 0.71, 95% CI: 0.46–1.08) for those TB 1× daily and by 49% (HR = 0.51, 95% CI: 0.29–0.89) for those TB ≥ 2× daily. There were no significant associations found between oral hygiene behavior and hemorrhagic stroke. |
| Luo 2022 [30] | Chi-square tests and t-tests Linear regression model Logistic regression model OR p value | Periodontitis Yes = 1 (mild, moderate, or severe) No = 0 STL (<20 teeth out of 28 teeth) Yes No | Having both STL and DM (b = −2.64), having STL alone (b = −2.08), and having DM alone (b = −1.09) were negatively associated with flossing, suggesting that those with STL or DM were less likely to floss frequently. Significant tooth loss is associated with higher systemic inflammation. Flossing may contribute to reducing systemic inflammation. |
| Moon 2024 [73] | ANOVA or Kruskal Walls test Chi-square test Tukey HSD or Steel–Dwass test Cox proportional hazards regression analysis p value and confidence intervals HR | TB frequency and duration Low frequency and short duration Low frequency or short duration Non-low frequency and non-short duration | The combination of decreased frequency and duration of TB is associated with a higher risk of cardiovascular events. Participants in the low-frequency and short-duration group had significantly higher circulating hs-CRP levels compared to the non-low and non-short group (p = 0.04). There were no significant differences in the incidences of MACEs between the non-low-frequency and non-short-duration group and the low-frequency or short-duration group. |
| Matsui 2022 [55] | Mean ± standard errors, and percentages. Generalized linear models Multivariable adjustment models | TB Frequency 0–1 daily 2 daily ≥3 daily | Those who reported more frequent toothbrushing had the following: lower systolic BP; lower non-HDL cholesterol, triglyceride, and fasting plasma glucose levels; higher HDL cholesterol levels; however, LDL cholesterol levels did not differ significantly. The estimated 10-year ASCVD risk was 13.7%, 9.1%, and 7.3% for those who brushed their teeth 0–1, 2, and ≥3 times a day, respectively. Participants who reported brushing their teeth ≥ 3 times/day had 0.48 mg/L and 0.32 mg/L lower levels of hsCRP than those who brushed their teeth 2 times/day and 0–1 time/day, respectively (unadjusted p values < 0.001 for both). WBC levels showed significant differences according to toothbrushing behaviors after multivariable adjustment (−0.20 × 103/µL, p = 0.013; and −0.18 × 103/µL, p = 0.021, respectively). |
| Palmer 2015 [45] | p value Kaplan–Meier curves Multivariable-adjusted Cox proportional hazards regression models | DMFT Very low (<5) Low (5–8.9) Moderate (9–13.9) High (14 or more) | TB daily was associated with longer survival for all-cause death (HR 0.76 (0.58–0.99)); however, it was not associated with a lower risk of cardiovascular death (HR 0.74 (0.52–1.07)). Flossing was associated with longer survival aHRs of 0.52 (95%CI, 0.32–0.85) and lower risk of cardiovascular death 0.25 (0.09–0.68). Mouthwash use was associated with longer survival aHRs of 0.79 (0.64–0.97) and lower risk of cardiovascular death 0.60 (0.44–0.82). TB for ≥2 min was associated with longer survival aHR of 0.76 (0.58–0.99) and lower risk of cardiovascular death aHR 0.74 (0.52–1.07). Tooth loss and dental disease were associated with all cause and cardiovascular mortality, while better oral hygiene practices were associated with better survival in those living with ESKD being treated with HD. |
| Park 2019 [44] | Kaplan–Meier curves HR CI Multivariable regression models | Number of dental caries 0 1–5 ≥6 Number of lost teeth 0 1–7 8–14 15–21 22–28 Number daily TB 0–1 2 ≥3 Professional clean <1× a year ≥1 a year | TB one more time a day was associated with a 9% lower risk of cardiovascular events (HR: 0.91; 95% CIs: 0.89–0.93; p< 0.001). TB three times a day or more was associated with a 19% reduction in cardiovascular events compared with TB once or less a day. Better oral hygiene practices and regular preventative dental visits are associated with a lower cardiovascular risk. The benefit of oral hygiene is independent of oral hygiene status and other confounding factors. |
| Patel 2021 [78] | Chi-square test Mann–Whitney U test ANOVA Regression analysis OR | TB method Toothbrush and paste Other TB frequency Once daily ≥2× daily | TB frequency showed a significant association with CHD (p = 0.001). Using other TB aids (neem sticks, miswak, etc.) showed lower risk of developing CHD (OR = 0.71); however, upon adjusting, it showed a 1.15 times higher risk for CHD. Lower TB frequency was associated with 3.38 times higher risk for CHD (OR = 3.38; adjusted OR = 2.54). |
| Reichert 2015 [46] | Kolmogorov–Smirnov test Shapiro–Wilk test Mann–Whitney U HR Log-rank Kapal–Meier Curves | Use of dental floss/interdental brushes No Yes | Dental flossing and interdental brush use might reduce the risk of new CVD events amongst individuals living with CHD. For those that used dental floss or interdental brushes, the bivariate analyses found significantly lower incidence for endpoints MI, stroke/TIA and death from cardiovascular causes, compared to those who did not use them (1.6% vs. 8.8%, log-rank p = 0.001). The use of dental floss and/or interdental brushes was significantly associated with a decreased risk of new cardiovascular events among those living with CVD after 1 year (p = 0.01). Dental floss/interdental brush use had an inverse association with severe periodontitis prevalence. TB frequency was not found to impact CVD outcomes. |
| Saengtipbovorn 2014 [82] | Compared numerical data and percentage and used p values. Descriptive statistics, Chi-square test, Fisher’s exact test, Multiple linear regression | Intervention group (LCDC) Control group | After the LCDC program, FPG and HbA1c decreased with statistically significant differences between the intervention and the control groups at 3 month follow-up (p = 0.003 and p =< 0.001, respectively). After the 3-month follow-up, the participants in the intervention group were more likely to use mouth rinse, salt solution, dental floss, and an inter-proximal brush. However, only salt solution and dental floss had statistically significant differences (p = 0.020 and < 0.001, respectively). The combination of LCDC in one program, improved both glycemic and periodontal status in the elderly with T2DM. |
| Song 2021 [47] | Compared numerical data and percentage and used p values. Kaplan–Meier curves HR Multivariate analysis | Periodontitis No Yes Number of missing teeth 0 1–4 ≥5 Number of dental caries 0 1–4 ≥5 Teeth brush times/day 0–1 ≥2 | Frequent TB was associated with a lower risk of cerebral or myocardial infarction in individuals living with DMs. TB 2× daily was significantly associated with a lower risk of both cerebral aHR: 0.81 (95% CI: 0.69–0.94); p = 0.007, and myocardial aHR: 0.76 (95% CI: 0.62–0.94); p = 0.012 infarction. Oral diseases are significantly associated with long-term cardiovascular events, particularly cerebral infarction. |
| Song 2021 [48] | Compared numerical data and percentage and used p values. Descriptive statistics, Chi-square test Linear mixed model | TB frequency 0–2× daily ≥3× daily | Compared with TB ≤ 2 times per day, TB ≥ 3 times per day was significantly associated with decreased fasting glucose levels (β = −0.0207 mmol/L, SE = 0.0033, p < 0.001). The authors suggest that poor oral hygiene is a potential therapeutic target for glycemic control, implying that interventions to improve oral hygiene, including frequent TB, may improve long-term fasting glucose regulation. |
| VanWormer 2013 [74] | Compared numerical data, regression modeling. Point estimate ± standard error, p value | Oral hygiene index classified as follows: Excellent, good, fair, or poor. Classifications were made depending on the following factors: TB frequency <1× daily 1× daily ≥2× daily Flossing Frequency Never Rarely Some days Most days Daily | The median CVD risk score was 6.2% (range 0.4–55.8%). The initial crude model indicated a significant association between oral hygiene (F = 3.60, p = 0.018) and CVD risk score. Sensitivity analysis in this study indicated that both toothbrushing and flossing were independently associated with CVD risk score, but these predictors did not interact. Participants brushing ≥ 2× daily had a lower risk (approx. 9%) of a CVD event at 10 years compared to those brushing < 1× daily (approx. 12%). Participants flossing daily had a lower risk (approx. 8%) of a CVD event at 10 years compared to those rarely flossing (approx. 13%). The median T2DM risk score was 9.8% (range 0.1–94.2%). The initial crude model did not indicate a significant association between oral hygiene and T2DM risk score (F = 1.65, p = 0.188). The authors suggest this is due to selection bias of participants, thus leaving a smaller sample size. |
| Vogtmann 2017 [61] | Compared numerical data, percentage, p values and HR used. Cox proportional hazards | TB frequency Never 1× daily ≥2× daily Other Quintile of tooth loss Q1 (Fewer teeth missing than expected) Q2 Q3 (expected number of teeth missing) Q4 Q5 (More teeth missing than expected) | TB was inversely associated only with CVD mortality. Although brushing once a day, aHR 0.79 [95% CI: 0.69, 0.91], and the “other” category of brushing, aHR 0.76 [95% CI: 0.65, 0.88], had an inverse association, brushing twice a day was not associated with cardiovascular disease mortality, aHR 1.06 [95% CI: 0.84,1.32], compared with never brushing. Any type of TB decreased the risk of overall mortality compared with never brushing. Participants in Q5 and/or who used dentures had a higher risk of overall mortality in both adjusted and unadjusted models. |
| Wang 2022 [31] | Compared numerical data and percentage and used p values. HR Multivariable regression models | TB frequency 0× daily <1× daily 1× daily ≥2× daily | Frequent TB was associated with a reduced risk of hypertension and DM incidence. Compared with those never brushing, participants TB ≥ 2× daily had an 0.54 (95% CI: 0.40–0.72; p < 0.001) times decreased risk of hypertension and adjusted 0.64 (95% CI: 0.44–0.93; p < 0.001) times decreased risk of DM. TB at least twice a day may prevent future hypertension and DM events. |
| Winning 2017 [50] | Compared numerical data and percentage and used p values. Kaplan–Meier curves HR | Periodontal status No/mild periodontitis. Moderate periodontitis Severe periodontitis | TB frequency as part of Model 1, 2, 3, and 4 in the dose–response risk for incident T2DM was significant when comparing severe with no/mild periodontitis. Baseline moderate to severe periodontitis was an independent risk predictor of incident T2DM. |
| Yoshioka 2021 [70] | Compared numerical data and percentage and used p values. OR Shapiro–Wilk test | TB before bed Sometimes/no Always | Participants who brushed their teeth before bedtime every night had lower BMI and LDL-cholesterol than those who did not brush nightly (p =< 0.05). |
| Zhang 2023 [75] | OR and aOR; survey-weighted descriptive statistics; Chi-squared tests; Multivariable logistic regression models. Multivariable linear models. Multicollinearity between the covariates. | Flossing Yes No | The prevalence of poor glycemic control was significantly lower among flossers than non-flossers (29.1% vs. 39.6%, respectively); p < 0.01. Flossers were 39% less likely than non-flossers to have periodontitis aOR 0.61 [ 95%CI, 0.43–0.88]. Flossing was significantly associated with a HbA1c reading that was 0.3% lower than non-flossers, adjusted for covariates (β = −0.3, 95% CI −0.58, −0.02, p = 0.037). |
| Zhuang 2021 [49] | Compared numerical data, percentage, p values, and HR used. | TB frequency Rarely/Never Sometimes/Always Frequency of gum bleeding Rarely Sometimes Always | Compared with frequent TB, the multivariable adjusted HRs for less frequent TB (rarely or never) were HR 1.28 (95% CI, 1.21, 1.34) for major vascular deaths, HR 1.23 (95% CI, 1.13, 1.33) for major coronary deaths, HR 1.23 (95% CI, 1.13, 1.33) for IHD caused deaths, HR 1.13 (95% CI, 1.06, 1.1)) for cancer caused deaths, HR 1.42 (95% CI, 1.23, 1.6)) for COPD caused deaths, and HR 1.49 (95% CI, 1.05, 1.28) for liver cirrhosis. TB behavior was not associated with death from T2DM and CKD. Compared with those who brushed teeth regularly, those who never or rarely brushed teeth had higher risk of MVE HR 1.12 (95% CI, 1.09, 1.15), stroke HR 1.08 (1.05–1.12), intracerebral hemorrhage HR 1.18 (1.11–1.26), and pulmonary heart disease HR 1.22 (1.13–1.32). No significant results were observed for subarachnoid hemorrhage HR 0.97, 0.74–1.26) and IHD HR 1.00 (0.97–1.04). Those who did not brush teeth also had increased risk of cancer HR 1.09 (1.04–1.14), COPD HR 1.12 (1.05–1.20), liver cirrhosis HR 1.25 (1.09–1.44), and all-cause death HR 1.25 (1.21–1.28), but not T2DM HR 0.94 (0.86–1.03) and CKD HR 0.98 (0.81–1.18). |
| Zhou 2024 [60] | Mean/SD, One-way analysis of variance, Pearson χ test, Kaplan–Meier survival curves, HR, 95% CI, Cox proportional hazards | TB frequency <1× daily 1× daily ≥2× daily | Compared to TB ≥ 2× daily, participants with lower toothbrushing frequency had higher risks of the following: All-cause mortality:
|
| First Author, Year | S1 | S2 | S3 | S4 | C1 | O1 | O2 | O3 | Overall Score & Assessed Quality |
|---|---|---|---|---|---|---|---|---|---|
| Afsar 2013 [80] | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 8—Good | |
| Aggarwal 2012 [34] | ★ | ★ | ★ | ★ | 4—Poor | ||||
| Almas 2003 [35] | ★ | ★★ | ★ | ★ | ★ | 6—Poor | |||
| Assante 2020 [33] | ★ | ★ | ★ | ★ | ★ | ★ | 6—Good | ||
| Chang 2020 [37] | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 8—Good | |
| Chang 2020 [32] | ★ | ★ | ★ | ★ | ★ | 5—Fair | |||
| Chang 2021 [36] | ★ | ★ | ★ | ★★ | ★ | ★ | 6—Good | ||
| Chang 2021 [58] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 9—Good |
| Cho 2021 [62] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 9—Good |
| Choi 2019 [51] | ★ | ★ | ★★ | ★ | ★ | ★ | 7—Fair | ||
| Cinar 2013 [79] | ★ | ★ | ★ | ★ | ★ | 5—Poor | |||
| Cinar 2015 [64] | ★ | ★ | ★ | 3—Poor | |||||
| deOliveira 2010 [65] | ★ | ★ | ★ | ★ | ★ | ★ | ★ | 7—Good | |
| deSouza 2014 [38] | ★ | ★ | ★ | ★ | ★ | ★ | ★ | ★ | 8—Good |
| Frisbee 2010 [66] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 9—Good |
| Fujita 2009 [67] | ★ | ★ | ★ | ★ | ★ | ★ | 7—Good | ||
| Guo 2023 [53] | ★★ | ★ | ★★ | ★ | ★ | ★ | 8—Good | ||
| Han 2021 [72] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 9—Good |
| Hiramatsu 2022 [81] | ★ | ★ | ★ | ★ | ★ | ★ | ★ | 7—Fair | |
| Hirano 2022 [39] | ★ | ★ | ★ | ★★ | ★ | ★ | 7—Good | ||
| Huang 2023 [71] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 8—Good |
| Huh 2023 [57] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 9—Good |
| Hwang 2018 [68] | ★ | ★ | ★ | ★★ | ★ | ★ | 7—Good | ||
| Hwang 2022 [56] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 9—Good |
| Isomura 2023 [54] | ★ | ★★ | ★ | ★★ | ★ | ★ | ★ | 9—Good | |
| Jain 2015 [77] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 9—Good |
| Jangam 2017 [51] | ★ | ★ | ★ | ★ | ★ | 5—Poor | |||
| Janket 2023 [76] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 9—Good |
| Joshipura 2017 [41] | ★ | ★ | ★★ | ★ | ★★ | ★ | ★ | 9—Good | |
| Joshipura 2020 [42] | ★ | ★ | ★★ | ★ | ★★ | ★ | ★ | ★ | 10—Good |
| Kim 2022 [52] | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 8—Good | |
| Kobayashi 2020 [40] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | 8—Good | |
| Kuwabara 2016 [69] | ★ | ★ | ★★ | ★ | ★ | ★ | 7—Fair | ||
| Kuwabara 2017 [43] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 9—Good |
| Long 2023 [59] | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 8—Good | |
| Luo 2022 [30] | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 8—Good | |
| Matsui 2022 [55] | ★ | ★ | ★★ | ★ | ★★ | ★ | ★ | ★ | 10—Good |
| Moon 2024 [73] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 9—Good |
| Palmer 2015 [45] | ★ | ★ | ★ | ★ | ★ | ★ | 6—Fair | ||
| Park 2019 [44] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | 8—Good | |
| Patel 2021 [78] | ★ | ★ | ★ | ★ | ★ | ★ | ★ | 7—Good | |
| Reichert 2015 [46] | ★★ | ★ | ★★ | ★ | ★ | 7—Good | |||
| Song 2021 [47] | ★ | ★ | ★★ | ★ | ★ | 6—Fair | |||
| Song 2021 [48] | ★ | ★ | ★★ | ★ | ★ | ★ | 7—Fair | ||
| VanWormer 2013 [74] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | 8-Good | |
| Vogtmann 2017 [61] | ★ | ★ | ★★ | ★ | ★★ | ★ | ★ | ★ | 10—Good |
| Wang 2022 [31] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 9—Good |
| Winning 2017 [50] | ★ | ★ | ★★ | ★ | ★ | ★ | ★ | ★ | 9—Good |
| Yoshioka 2021 [70] | ★ | ★★ | ★ | ★ | ★ | 6—Fair | |||
| Zhang 2023 [75] | ★ | ★ | ★ | ★★ | ★ | ★ | 7—Good | ||
| Zhuang 2021 [49] | ★ | ★ | ★ | ★★ | ★ | ★ | 7—Good | ||
| Zhou 2024 [60] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 9—Good |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Church, L.; Franks, K.; Medara, N.; Curkovic, K.; Singh, B.; Mehta, J.; Bhatti, R.; King, S. Impact of Oral Hygiene Practices in Reducing Cardiometabolic Risk, Incidence, and Mortality: A Systematic Review. Int. J. Environ. Res. Public Health 2024, 21, 1319. https://doi.org/10.3390/ijerph21101319
Church L, Franks K, Medara N, Curkovic K, Singh B, Mehta J, Bhatti R, King S. Impact of Oral Hygiene Practices in Reducing Cardiometabolic Risk, Incidence, and Mortality: A Systematic Review. International Journal of Environmental Research and Public Health. 2024; 21(10):1319. https://doi.org/10.3390/ijerph21101319
Chicago/Turabian StyleChurch, Lauren, Kay Franks, Nidhi Medara, Karolina Curkovic, Baani Singh, Jaimit Mehta, Raied Bhatti, and Shalinie King. 2024. "Impact of Oral Hygiene Practices in Reducing Cardiometabolic Risk, Incidence, and Mortality: A Systematic Review" International Journal of Environmental Research and Public Health 21, no. 10: 1319. https://doi.org/10.3390/ijerph21101319
APA StyleChurch, L., Franks, K., Medara, N., Curkovic, K., Singh, B., Mehta, J., Bhatti, R., & King, S. (2024). Impact of Oral Hygiene Practices in Reducing Cardiometabolic Risk, Incidence, and Mortality: A Systematic Review. International Journal of Environmental Research and Public Health, 21(10), 1319. https://doi.org/10.3390/ijerph21101319






