Co-Creating a Feasible, Acceptable and Safe Home-Based High-Intensity Interval Training Programme for People with Parkinson’s: The HIIT-Home4Parkinson’s Study
Abstract
1. Introduction
- To develop a home-based HIIT programme which was feasible and achievable for PwP;
- To develop the intervention components (including clinician and participant materials, guides and recording documents);
- To develop a first draft of the intervention logic model which would identify the key programme components for process evaluation in future studies.
2. Materials and Methods
2.1. Stage 1: Exercise Protocol and Logic Model Initial Proposal
2.2. Stage 2: Evolving the Initial Proposal
2.2.1. Co-Creator Recruitment Strategy and Eligibility
2.2.2. Focus Group Methodology
2.2.3. Focus Group Data Analysis
2.2.4. Exercise Testing Methodology
2.2.5. Exercise Environment
2.2.6. Exercise Session 1
2.2.7. Sessions 1–3: HIIT Programme Testing
2.2.8. Exercise Testing Data Analysis
2.3. Stage 3: Final Drafting
3. Results
3.1. Stage 1—The Initial Proposal
3.1.1. Key Principles
3.1.2. Key Uncertainties
3.2. Stage 2—Evolving the Initial Proposal
3.2.1. Co-Creator Recruitment
3.2.2. Attendance (Figure 1)
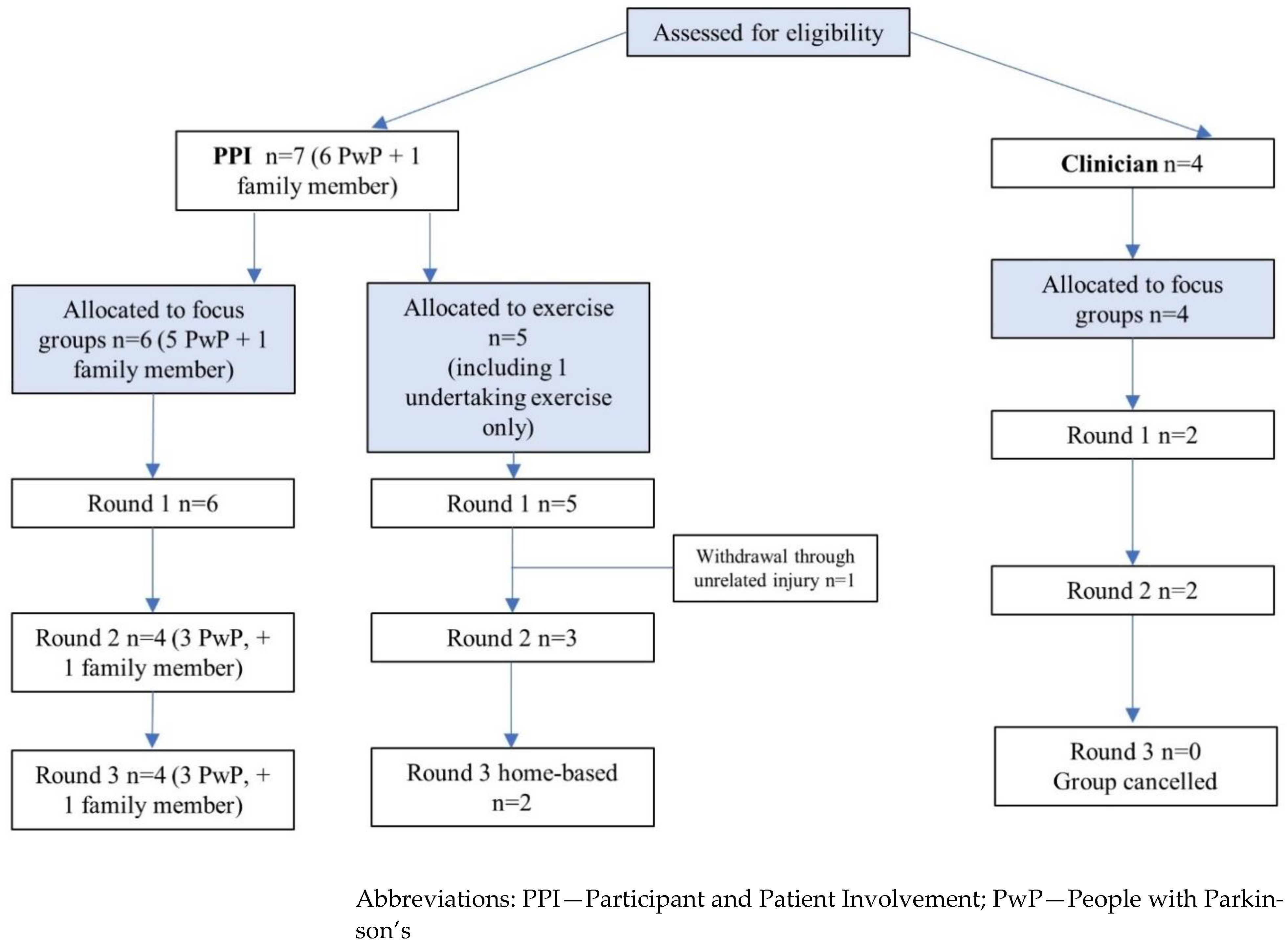
3.2.3. HIIT Testing Results (Table 2)
| Participant | HIIT Sets Undertaken/Completed | % of Sets Achieving Mean Target Intensity | Mean (SD) % of HRmax | Mean RPE | Adverse Effects |
|---|---|---|---|---|---|
| 1 | 8/8 | 100 | 83.6 (±4.3) | 13.4 (Somewhat hard) | Mild shoulder pain |
| 2 | 7/8 | 88 | 80.6 (±5.0) | 14.5 (Hard) | Mild shoulder pain |
| 3 | 8/8 | 50 | 71.9 (±6.5) | 13.4 (Somewhat hard) | None |
| 4 | 12/12 | 92 | 83.0 (±7.1) | 14.9 (Hard) | Mild knee pain/calf cramp |
| 5 | 4/4 | 100 | 86.3 (±1.6) | 17.5 (Very hard) | None |
| Overall | 39/40 | 86 | 80.8 (±7.2) | 14.7 (Hard) | - |
3.2.4. Qualitative Data
3.3. Stage 3: Final Drafting
3.4. The Final Co-Created HH4P Programme
3.4.1. Individualised HIIT Protocol (Table 4)
| Phase | Protocol/Bouts | Intensity | Accumulated Time (min/s) |
|---|---|---|---|
| Warm up | 10 min: stretching/fast marching on the spot | 50–60% HRmax | 10.0 |
| HIIT set 1 | Group 1: Whole body (45 s work/15 s rest) × 3 | ≥75% HRmax | 12.45 |
| 2-min rest/active recovery | Rest/slow walking on the spot | 50% HRmax | 14.45 |
| HIIT set 2 | Group 2: Upper body (45 s work/15 s rest) × 3 | ≥75% HRmax | 17.30 |
| 2-min rest/active recovery | Rest/slow walking on the spot | 50% HRmax | 19.30 |
| HIIT set 3 | Group 3: Whole body (45 s work/15 s rest) × 3 | ≥75% HRmax | 22.15 |
| 2-min rest/active recovery | Rest/slow walking on the spot | 50% HRmax | 24.15 |
| HIIT set 4 | Group 4: Lower body (45 s work/15 s rest) ×3 | ≥75% HRmax | 27.0 |
| Cool down | 5 min walking on the spot (voluntary speed)/stretching | Back to resting | 32.0 |
| Menu of HIIT exercises | |||
| Group 1: Whole body Running on the spot, or Skipping (without rope), or Star jumps | Group 2: Upper body Boxing (Crosses, jabs, hooks and uppercuts), or Over-head shoulder press with choice of hand-held weights, or Front arm-raise with choice of resistance bands | Group 3: Whole body (Select different exercise from choice 1 if possible) Running on the spot, or Skipping (without rope), or Star jumps | Group 4: Lower body (Performed with balance aids/supports as required) Body-weight squats, or Chair sit to standing position, or Forward lunges |
3.4.2. HIIT Exercise Resources
3.4.3. Adaptations
- The potential to double (or halve) exercise frequency, thereby modifying the intensity.
- The potential to “bounce” (inclusion of lower body) during shoulder exercises and boxing, thereby increasing the intensity.
- Utilising home-based balance aids for support/leverage to maximise safety and achievability during lower-body exercises.
- The potential to add an extra coordinative aspect, thereby increasing the difficulty level (and potentially providing additional benefit to static and dynamic balance) [35]. For example, boxing exercises could also involve an element of foot rotation if required.
- To account for physiological adaptation to the programme, co-creators recommended that exercise target intensity should be titrated to maintain a suitable relative intensity, depending on progression. This will be addressed in fortnightly check-ins, and target exercise intensity titrated up (or down) accordingly. Increased intensity will be achieved by increasing movement amplitude and/or frequency, or through exercise adaptations (for example “bouncing” during boxing).
3.4.4. Programme Delivery Support
- Baseline assessment visit to UoP will give the opportunity for uncertainties to be addressed in person.
- Pre-intervention home visit will include exercise technique and adaptations training, equipment provision and instruction, remote support instruction, partner liaison and exercise location preparation.
- Fortnightly online check-ins will provide motivation and assess for potential programme adaptations.
- Fortnightly online check-ins will monitor for adverse effects/events.
- Exercise reminder texts will be sent to participants.
- Monthly online group sessions will provide opportunities for social interaction.
- A range of internet-based and physical support resource will be available, which will include; exercise demonstration instructional video performed by a PwP, audio RAC accompaniments, contact information for live feedback, diaries and recording sheets, HIIT information sheets, loaned HR monitor and accelerometer along with equipment care and instruction leaflets.
- User-friendly diaries to record adherence and completion, substantiated by online HR data.
- Researcher contact details for immediate feedback.
3.4.5. Outcome Measures
4. Discussion
4.1. Programme Delivery Support
4.2. Outcome Measures
4.3. Limitations of the HH4P Development Process
4.4. Implications for Future Studies and HH4P
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
Appendix A. Maximal IET Procedure
Appendix B. The Final HH4P Intervention Logic Model

References
- Wen, D.; Utesch, T.; Wu, J.; Robertson, S.; Liu, J.; Hu, G.; Chen, H. Effects of different protocols of high intensity interval training for VO2max improvements in adults: A meta-analysis of randomised controlled trials. J. Sci. Med. Sport 2019, 22, 941–947. [Google Scholar] [CrossRef] [PubMed]
- Wewege, M.; Van Den Berg, R.; Ward, R.E.; Keech, A. The effects of high-intensity interval training vs. moderate-intensity continuous training on body composition in overweight and obese adults: A systematic review and meta-analysis. Obes. Rev. 2017, 18, 635–646. [Google Scholar] [CrossRef]
- Keating, C.J.; Montilla, J.Á.P.; Román, P.Á.L.; Del Castillo, R.M. Comparison of high-intensity interval training to moderate-intensity continuous training in older adults: A systematic review. J. Ageing Phys. Act. 2020, 28, 798–807. [Google Scholar] [CrossRef] [PubMed]
- Harpham, C.; Gunn, H.; Marsden, J.; Connolly, L. The feasibility, safety, physiological and clinical effects of high-intensity interval training for people with Parkinson’s: A systematic review and meta-analysis. Aging Clin. Exp. Res. 2023, 35, 497–523. [Google Scholar] [CrossRef]
- Palasz, E.; Wysocka, A.; Gasiorowska, A.; Chalimoniuk, M.; Niewiadomski, W.; Niewiadomska, G. BDNF as a promising therapeutic agent in Parkinson’s disease. Int. J. Mol. Sci. 2020, 21, 1170. [Google Scholar] [CrossRef]
- Ellis, T.; Boudreau, J.K.; DeAngelis, T.R.; Brown, L.E.; Cavanaugh, J.T.; Earhart, G.M.; Ford, M.P.; Foreman, K.B.; Dibble, L.E. Barriers to exercise in people with Parkinson disease. Phys. Ther. 2013, 93, 628–636. [Google Scholar] [CrossRef]
- Paul, S.S.; Canning, C.G.; Löfgren, N.; Sherrington, C.; Lee, D.C.; Bampton, J.; Howard, K. People with Parkinson’s disease are more willing to do additional exercise if the exercise program has specific attributes: A discrete choice experiment. J. Physiother. 2021, 67, 49–55. [Google Scholar] [CrossRef] [PubMed]
- NHS England. Supported Self-Management. 2020. Available online: https://www.england.nhs.uk/wp-content/uploads/2020/03/supported-self-management-summary-guide.pdf (accessed on 29 October 2022).
- Flynn, A.; Allen, N.E.; Dennis, S.; Canning, C.G.; Preston, E. Home-based prescribed exercise improves balance-related activities in people with Parkinson’s disease and has benefits similar to centre-based exercise: A systematic review. J. Physiother. 2019, 65, 189–199. [Google Scholar] [CrossRef]
- Lacroix, A.; Hortobagyi, T.; Beurskens, R.; Granacher, U. Effects of supervised vs. unsupervised training programs on balance and muscle strength in older adults: A systematic review and meta-analysis. Sport. Med. 2017, 47, 2341–2361. [Google Scholar] [CrossRef]
- Leask, C.F.; Sandlund, M.; Skelton, D.A.; Altenburg, T.M.; Cardon, G.; Chinapaw, M.J.; De Bourdeaudhuij, I.; Verloigne, M.; Chastin, S.F. Framework, principles and recommendations for utilising participatory methodologies in the co-creation and evaluation of public health interventions. Res. Involv. Engagem. 2019, 5, 2. [Google Scholar] [CrossRef]
- Harvey, M.; Weston, K.L.; Gray, W.K.; O’Callaghan, A.; Oates, L.L.; Davidson, R.; Walker, R.W. High-intensity interval training in people with Parkinson’s disease: A randomized, controlled feasibility trial. Clin. Rehabil. 2019, 33, 428–438. [Google Scholar] [CrossRef] [PubMed]
- Moore, G.F.; Audrey, S.; Barker, M.; Bond, L.; Bonell, C.; Hardeman, W.; Moore, L.; O’Cathain, A.; Tinati, T.; Wight, D.; et al. Process evaluation of complex interventions: Medical Research Council guidance. BMJ 2015, 350, h1258. [Google Scholar] [CrossRef] [PubMed]
- O’Cathain, A.; Croot, L.; Duncan, E.; Rousseau, N.; Sworn, K.; Turner, K.M.; Yardley, L.; Hoddinott, P. Guidance on how to develop complex interventions to improve health and healthcare. BMJ Open 2019, 9, e029954. [Google Scholar] [CrossRef] [PubMed]
- Morgan, D.L. Focus Groups as Qualitative Research; Sage Publications: New York, NY, USA, 1996; Volume 16. [Google Scholar]
- Onwuegbuzie, A.J.; Collins, K.M. A typology of mixed methods sampling designs in social science research. Qual. Rep. 2007, 12, 281–316. [Google Scholar] [CrossRef]
- Wood, A.M.; White, I.R.; Thompson, S.G. Are missing outcome data adequately handled? A review of published randomized controlled trials in major medical journals. Clin. Trials 2004, 1, 368–376. [Google Scholar] [CrossRef]
- Gundumogula, M. Importance of focus groups in qualitative research. Int. J. Humanit. Soc. Sci. 2020, 8, 299–302. [Google Scholar] [CrossRef]
- Rupert, D.J.; Poehlman, J.A.; Hayes, J.J.; Ray, S.E.; Moultrie, R.R. Virtual versus in-person focus groups: Comparison of costs, recruitment, and participant logistics. J. Med. Internet Res. 2017, 19, e6980. [Google Scholar] [CrossRef]
- Braun, V.; Clarke, V. Using thematic analysis in psychology. Qual. Res. Psychol. 2006, 3, 77–101. [Google Scholar] [CrossRef]
- Braun, V.; Clarke, V.; Weate, P. Using thematic analysis in sport and exercise research. In Routledge Handbook of Qualitative Research in Sport and Exercise; Routledge: London, UK, 2016; pp. 191–205. [Google Scholar]
- Moore, G.; Durstine, J.L.; Painter, P. American College of Sports Medicine. In Acsm’s Exercise Management for Persons with Chronic Diseases and Disabilities, 4th ed.; Human Kinetics: Champaign, IL, USA, 2016. [Google Scholar]
- Haas, B.M.; Trew, M.; Castle, P.C. Effects of respiratory muscle weakness on daily living function, quality of life, activity levels, and exercise capacity in mild to moderate Parkinson’s disease. Am. J. Phys. Med. Rehabil. 2004, 83, 601–607. [Google Scholar] [CrossRef]
- Haas, B.; Cinnamond, S.; Hunter, H.; Marsden, J. Factors associated with limited exercise capacity and feasibility of high intensity interval training in people with mild to moderate Parkinson’s disease. Int. J. Ther. Rehabil. 2016, 23, 414–422. [Google Scholar] [CrossRef]
- Borg, G. Ratings of perceived exertion and heart rates during short-term cycle exercise and their use in a new cycling strength test. Int. J. Sport. Med. 1982, 3, 153–158. [Google Scholar] [CrossRef]
- Gallo, P.M. High-Intensity Interval Training for Neurodegenerative Conditions: Indications and Recommendations for Exercise Programming. ACSM’s Health Fit. J. 2021, 25, 18–27. [Google Scholar] [CrossRef]
- Gibala, M.J.; Little, J.P.; MacDonald, M.J.; Hawley, J.A. Physiological adaptations to low-volume, high-intensity interval training in health and disease. J. Physiol. 2012, 590, 1077–1084. [Google Scholar] [CrossRef]
- McRae, G.; Payne, A.; Zelt, J.G.; Scribbans, T.D.; Jung, M.E.; Little, J.P.; Gurd, B.J. Extremely low volume, whole-body aerobic–resistance training improves aerobic fitness and muscular endurance in females. Appl. Physiol. Nutr. Metab. 2012, 37, 1124–1131. [Google Scholar] [CrossRef] [PubMed]
- Machado, A.F.; Miranda, M.L.D.J.; Rica, R.L.; Figueira, A.; Bocalini, D.S. Bodyweight high-intensity interval training: A systematic review. Rev. Bras. Med. Esporte 2018, 24, 234–237. [Google Scholar] [CrossRef]
- American College of Sports Medicine. High-Intensity Interval Training PDF Resource. 2014. Available online: https://www.acsm.org/docs/default-source/files-for-resource-library/high-intensity-interval-training.pdf (accessed on 22 April 2022).
- American College of Sports Medicine. A Road Map to Effective Recovery PDF Resource. 2019. Available online: https://www.acsm.org/docs/default-source/files-for-resource-library/a-road-map-to-effective-muscle-recovery.pdf (accessed on 25 April 2022).
- Thaut, M.H.; McIntosh, G.C.; Hoemberg, V. Neurobiological foundations of neurologic music therapy: Rhythmic entrainment and the motor system. Front. Psychol. 2015, 5, 1185. [Google Scholar] [CrossRef]
- Yoo, G.E.; Kim, S.J. Rhythmic auditory cueing in motor rehabilitation for stroke patients: Systematic review and meta-analysis. J. Music. Ther. 2016, 53, 149–177. [Google Scholar] [CrossRef] [PubMed]
- Karageorghis, C.I.; Jones, L.; Priest, D.L.; Akers, R.I.; Clarke, A.; Perry, J.M.; Reddick, B.T.; Bishop, D.T.; Lim, H.B. Revisiting the relationship between exercise heart rate and music tempo preference. Res. Q. Exerc. Sport 2011, 82, 274–284. [Google Scholar] [CrossRef]
- Kara, B.; Genc, A.; Colakoglu, B.D.; Cakmur, R. The effect of supervised exercises on static and dynamic balance in Parkinson’s disease patients. NeuroRehabilitation 2012, 30, 351–357. [Google Scholar] [CrossRef]
- Spaulding, S.J.; Barber, B.; Colby, M.; Cormack, B.; Mick, T.; Jenkins, M.E. Cueing and gait improvement among people with Parkinson’s disease: A meta-analysis. Arch. Phys. Med. Rehabil. 2013, 94, 562–570. [Google Scholar] [CrossRef] [PubMed]
- Forte, R.; Tocci, N.; De Vito, G. The Impact of Exercise Intervention with Rhythmic Auditory Stimulation to Improve Gait and Mobility in Parkinson Disease: An Umbrella Review. Brain Sci. 2021, 11, 685. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.T.; Tai, H.L.; Yang, C.C.; Chen, Y.S. Acute Effects of Self-Selected Music Intervention on Golf Performance and Anxiety Level in Collegiate Golfers: A Crossover Study. Int. J. Environ. Res. Public Health 2020, 17, 7478. [Google Scholar] [CrossRef] [PubMed]
- Karageorghis, C.I.; Terry, P.C.; Lane, A.M. Development and initial validation of an instrument to assess the motivational qualities of music in exercise and sport: The Brunel Music Rating Inventory. J. Sport. Sci. 1999, 17, 713–724. [Google Scholar] [CrossRef] [PubMed]
- Gunn, H.; Andrade, J.; Paul, L.; Miller, L.; Creanor, S.; Stevens, K.; Green, C.; Ewings, P.; Barton, A.; Berrow, M.; et al. A self-management programme to reduce falls and improve safe mobility in people with secondary progressive MS: The BRiMS feasibility RCT. Health Technol. Assess. 2019, 23, 1–166. [Google Scholar] [CrossRef]
- Caglar, A.T.; Gurses, H.N.; Mutluay, F.K.; Kiziltan, G. Effects of home exercises on motor performance in patients with Parkinson’s disease. Clin. Rehabil. 2005, 19, 870–877. [Google Scholar] [CrossRef]
- Song, J.; Paul, S.S.; Caetano, M.J.D.; Smith, S.; Dibble, L.E.; Love, R.; Schoene, D.; Menant, J.C.; Sherrington, C.; Lord, S.R.; et al. Home-based step training using videogame technology in people with Parkinson’s disease: A single-blinded randomised controlled trial. Clin. Rehabil. 2018, 32, 299–311. [Google Scholar] [CrossRef]
- Van Den Helder, J.; Van Dronkelaar, C.; Tieland, M.; Mehra, S.; Dadema, T.; Visser, B.; Kröse, B.J.; Engelbert, R.H.; Weijs, P.J. A digitally supported home-based exercise training program and dietary protein intervention for community dwelling older adults: Protocol of the cluster randomised controlled VITAMIN trial. BMC Geriatr. 2018, 18, 183. [Google Scholar] [CrossRef]
- O’Callaghan, A.; Harvey, M.; Houghton, D.; Gray, W.K.; Weston, K.L.; Oates, L.L.; Romano, B.; Walker, R.W. Comparing the influence of exercise intensity on brain-derived neurotrophic factor serum levels in people with Parkinson’s disease: A pilot study. Aging Clin. Exp. Res. 2020, 32, 1731–1738. [Google Scholar] [CrossRef]
- Zoladz, J.A.; Majerczak, J.; Zeligowska, E.; Mencel, J.; Jaskolski, A.; Jaskolska, A.; Marusiak, J. Moderate-intensity interval training increases serum brain-derived neurotrophic factor level and decreases inflammation in Parkinson’s disease patients. J. Physiol. Pharmacol. 2014, 65, 441–448. [Google Scholar]
- Wang, X.; Zeng, F.; Jin, W.S.; Zhu, C.; Wang, Q.H.; Bu, X.L.; Luo, H.B.; Zou, H.Q.; Pu, J.; Zhou, Z.H.; et al. Comorbidity burden of patients with Parkinson’s disease and Parkinsonism between 2003 and 2012: A multicentre, nationwide, retrospective study in China. Sci. Rep. 2017, 7, 1671. [Google Scholar] [CrossRef]
- Kang, S.J.; Ko, K.J. Association between resting heart rate, VO2max and carotid intima-media thickness in middle-aged men. IJC Heart Vasc. 2019, 23, 100347. [Google Scholar] [CrossRef] [PubMed]
- Uygur, M.; Bellumori, M.; Knight, C.A. Effects of a low-resistance, interval bicycling intervention in Parkinson’s Disease. Physiother. Theory Pract. 2017, 33, 897–904. [Google Scholar] [CrossRef] [PubMed]
- Ben-Tovim, D.I.; Dougherty, M.L.; O’Connell, T.J.; McGrath, K.M. Patient journeys: The process of clinical redesign. Med. J. Aust. 2008, 188, S14–S17. [Google Scholar] [CrossRef]
- Hall, D.A.; Smith, H.; Heffernan, E.; Fackrell, K. Core Outcome Measures in Tinnitus International Delphi (COMiT’ID) Research Steering Group, Recruiting and retaining participants in e-Delphi surveys for core outcome set development: Evaluating the COMiT’ID study. PLoS ONE 2018, 13, e0201378. [Google Scholar] [CrossRef] [PubMed]
- de Souto Barreto, P.; Ferrandez, A.M.; Saliba-Serre, B. Are older adults who volunteer to participate in an exercise study fitter and healthier than non-volunteers? The participation bias of the study population. J. Phys. Act. Health 2013, 10, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Eldridge, S.M.; Chan, C.L.; Campbell, M.J.; Bond, C.M.; Hopewell, S.; Thabane, L.; Lancaster, G.A. CONSORT 2010 statement: Extension to randomised pilot and feasibility trials. BMJ 2016, 355, i5239. [Google Scholar] [CrossRef] [PubMed]
- Calvert, M.; Blazeby, J.; Altman, D.G.; Revicki, D.A.; Moher, D.; Brundage, M.D.; CONSORT PRO Group. Reporting of patient-reported outcomes in randomized trials: The CONSORT PRO extension. JAMA 2013, 309, 814–822. [Google Scholar] [CrossRef]
- Robergs, R.A.; Ghiasvand, F.; Parker, D. Biochemistry of exercise-induced metabolic acidosis. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2004, 287, R502–R516. [Google Scholar] [CrossRef]
- Engel, F.A.; Faude, O.; Kölling, S.; Kellmann, M.; Donath, L. Verbal encouragement and between-day reliability during high-intensity functional strength and endurance performance testing. Front. Physiol. 2019, 10, 460. [Google Scholar] [CrossRef]
- Mezzani, A. Cardiopulmonary exercise testing: Basics of methodology and measurements. Ann. Am. Thorac. Soc. 2017, 14 (Suppl. 1), S3–S11. [Google Scholar] [CrossRef]
| Exercise co-Creator Baseline Characteristics | Maximal Exercise Testing Results | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Participant | Age (y) | Sex | Height/Weight (cm/kg) | Estimated Weekly MVPA (min) | H & Y Stage | Time Since Diagnosis (y) | Medication/on or off | Age Adjusted HRmax (BPM) | Time/Reason for Termination | VO2max (ml/kg/min) | HRmax (BPM) | RPE on Termination | RER on Termination | HIIT Target Heart Rate (BPM) |
| 1 | 63 | M | 176.5/89.8 | 180 | 3 | 3 | Sinemet/Ropinirole: No on/off periods | 157 | 16.56/Volitional exhaustion | 35 | 136 | 18 | 1.19 | 103 |
| 2 | 62 | F | 164.5/67.8 | 480 | 2 | 2 | Madopar: On | 158 | 9.45/Volitional exhaustion | 22 | 144 | 19 | 1.19 | 108 |
| 3 | 49 | M | 176/77.7 | 350 | 3 | 6 | Ropinirole/Madopar/Opicapone/On | 171 | 14.50/Volitional exhaustion/HR & VO2 plateaux | 36 | 166 | 18 | 1.25 | 125 |
| 4 | 61 | M | 177/75.2 | 210 | 2 | 6 | Madopar/Rasigiline/On | 159 | 13.06/HR and VO2 plateaux | 31 | 163 | 18 | 1.25 | 122 |
| 5 | 64 | M | 177/80.6 | 140 | 3 | 20 | Subcutaneous levodopa/Metazopine/On | 156 | 4.00/Equipment malfunction | - * | - | - | - | 117 ** |
| Focus group co-creator characteristics | ||||||||||||||
| Group | Participants | |||||||||||||
| PPI: | Exercise co-creators 1, 2, 3, 4 + 1 PwP (3.5 years diagnosed, H & Y 2, teacher of remedial exercise classes, regular weekly MVPA) + 1 family member (spouse of participant 2) | |||||||||||||
| Clinician: | 1 qualified, registered physiotherapist, with current and over 10 years’ experience of leading exercise classes for PwP including high-intensity programmes + 1 student physiotherapist with current and three years’ experience of assisting with delivering exercise classes for PwP | |||||||||||||
| Round | Event | Attending Co-Creators | Main Thematic Categories (in Bold Type) and Sub-Categories |
|---|---|---|---|
| 1 | PPI focus group | 2 academic researchers 5 PwP, 1 family member | HIIT exercise concerns: Time restraints/Individual capacity/On-off periods/Space restraints/Coordination Programme delivery concerns: Supervision/Injury/Motivation/No social element/Screening HIIT exercise opportunities/motivators: Exercise suggestions/Initial protocol suitable intervals/Potential adaptations/Equipment suggestions Programme delivery opportunities/motivators: Home-based convenience/Remote support suggestions/Evidence of change as a motivator/Initial protocol suitable duration and frequency |
| Clinician focus group | 2 academic researchers 1 qualified clinician 1 student clinician | HIIT exercise barriers and considerations: Parkinson’s severity differentiation/Reduced movement amplitude Programme delivery concerns: Remote support/Licensing/Motivation/Outcome measures (PDQ-39 unsuitable)/Physio costs HIIT exercise opportunities and facilitators: Rhythmic cueing/Differentiation possibilities/Exercise suggestions: boxing variations Programme delivery opportunities and motivators: Remote support suggestions/Outcome measures/Motivation/Training/Social motivation | |
| Post exercise interviews | 5 PwP (Undertaken separately with 1 academic researcher) | Exercises and timings: Achievable/Challenging but enjoyable/Acceptable duration/Visible heart rate monitor useful/Intensity effort dependent Pre-programme training: No fitness training required, but may be for some PwP/Technique instruction important Safety: Balance issues during squats for those of Hoehn and Yahr 3/Support required for lower-body exercises RAC accompaniment: Correct tempo/Helps to maintain cadence/Enhances enjoyment/Not personal style preference/Differing styles required Other thoughts: Online group sessions may increase motivation | |
| 2 | PPI focus group | 2 academic researchers 3 PwP 1 family member | Outcome measures—acceptability and deemed importance: Brain-derived neurotrophic factor acceptable and important/VO2max acceptable procedure/Physical activity, 2 weeks accelerometery acceptable 30 s sit to stand acceptable but not suitable as fortnightly “motivator”/Rate of perceived exertion concern with judgement/Unified Parkinson’s Disease Rating Scale part III important and acceptable/Adherence, completion, adverse effects and events self-report diary acceptable/Ox-PAQ acute important and acceptable Specific points regarding the HIIT protocol: Challenging and enjoyable/Alternative exercises—core exercises, leg raises/Accompaniment acceptable, but requires variation/Pre-recorded verbal encouragement acceptable/Visible heart rate monitor acceptable and useful/Squats & sit to stand require support/Fixture and fittings damage potential |
| Clinician focus group | 2 academic researchers 1 qualified clinician 1 student clinician | Outcome measures: Range of outcomes to allow for individualised motivation/Additional motivational check-ins resource intensive/Goal Attainment Scale achievable procedure but problematic Programme delivery: Fortnightly motivational check- in/Proposed programme has multiple options/Ensure choice of options to avoid overloading/Use of Smart phone application Specific points regarding the HIIT protocol: Squats and sit-to-stand balance safety and “lunge” alternative/Core exercises stimulate inadequate intensity/Shoulder exercises overly similar/Alternative exercises to facilitate clinical prescription/Modify sequence to include extra “whole body” set | |
| Post exercise interviews | 3 PwP (Undertaken separately with 1 academic researcher) | Revised exercises and sequence: Suitable duration and timings/As challenging as previous round/Felt “aerobic”/Incorrect sequencing increases risks of injury Lower-body exercise safety: Balance supports increased confidence and achievability/Overbalancing forward during squats, unsuitable for some PwP Amended RAC: Verbal encouragement acceptable and useful/Correct tempo/“Dance” style better than “Blues” for maintaining cadence Exercise sequence prompting cards: Useful/Larger versions required Other thoughts: None | |
| 3 | PPI focus group | 2 academic researchers 3 PwP 1 family member | Considerations of the home setting—increase engagement: Opportunities within initial home visit: prepare environment and engagement with partner/Partner support/Potential to exercise outside Reduce engagement: Exercise environment: Surface, available space, potential damage/Lack of partner support/Motivation/Footwear |
| Clinician focus group | Cancelled due to unavailability | N/A | |
| Post exercise interviews (Home) | 2 PwP (Undertaken separately with 1 academic researcher) | Suitability of the home-environment: Positives: Exercises appropriate for the home/User-friendly resources/Required space acceptable/Enjoyable/Good exercise adaptability Negatives: Potential over-reliance on internet access/Importance of initial visit to prepare environment/Alternative format for exercise sequence prompting cards required |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harpham, C.; Gunn, H.; Marsden, J.; Connolly, L. Co-Creating a Feasible, Acceptable and Safe Home-Based High-Intensity Interval Training Programme for People with Parkinson’s: The HIIT-Home4Parkinson’s Study. Int. J. Environ. Res. Public Health 2023, 20, 5671. https://doi.org/10.3390/ijerph20095671
Harpham C, Gunn H, Marsden J, Connolly L. Co-Creating a Feasible, Acceptable and Safe Home-Based High-Intensity Interval Training Programme for People with Parkinson’s: The HIIT-Home4Parkinson’s Study. International Journal of Environmental Research and Public Health. 2023; 20(9):5671. https://doi.org/10.3390/ijerph20095671
Chicago/Turabian StyleHarpham, Conrad, Hilary Gunn, Jonathan Marsden, and Luke Connolly. 2023. "Co-Creating a Feasible, Acceptable and Safe Home-Based High-Intensity Interval Training Programme for People with Parkinson’s: The HIIT-Home4Parkinson’s Study" International Journal of Environmental Research and Public Health 20, no. 9: 5671. https://doi.org/10.3390/ijerph20095671
APA StyleHarpham, C., Gunn, H., Marsden, J., & Connolly, L. (2023). Co-Creating a Feasible, Acceptable and Safe Home-Based High-Intensity Interval Training Programme for People with Parkinson’s: The HIIT-Home4Parkinson’s Study. International Journal of Environmental Research and Public Health, 20(9), 5671. https://doi.org/10.3390/ijerph20095671






