A Scoping Review on Movement, Neurobiology and Functional Deficits in Dyslexia: Suggestions for a Three-Fold Integrated Perspective
Abstract
1. Introduction
“Reading is not exhausted merely by decoding the written word or written language, but rather anticipated by and extending into knowledge of the world. Language and reality are dynamically intertwined”[1] (p. 5).
2. What Is Dyslexia: Models and Neuronal Correlates
2.1. Models and Interpretations of the Disorder
2.2. Neural Correlates of Dyslexia
3. Reading the Cerebellum
3.1. The Cerebellum: From an Exclusively Motor Area to a Cognitive One
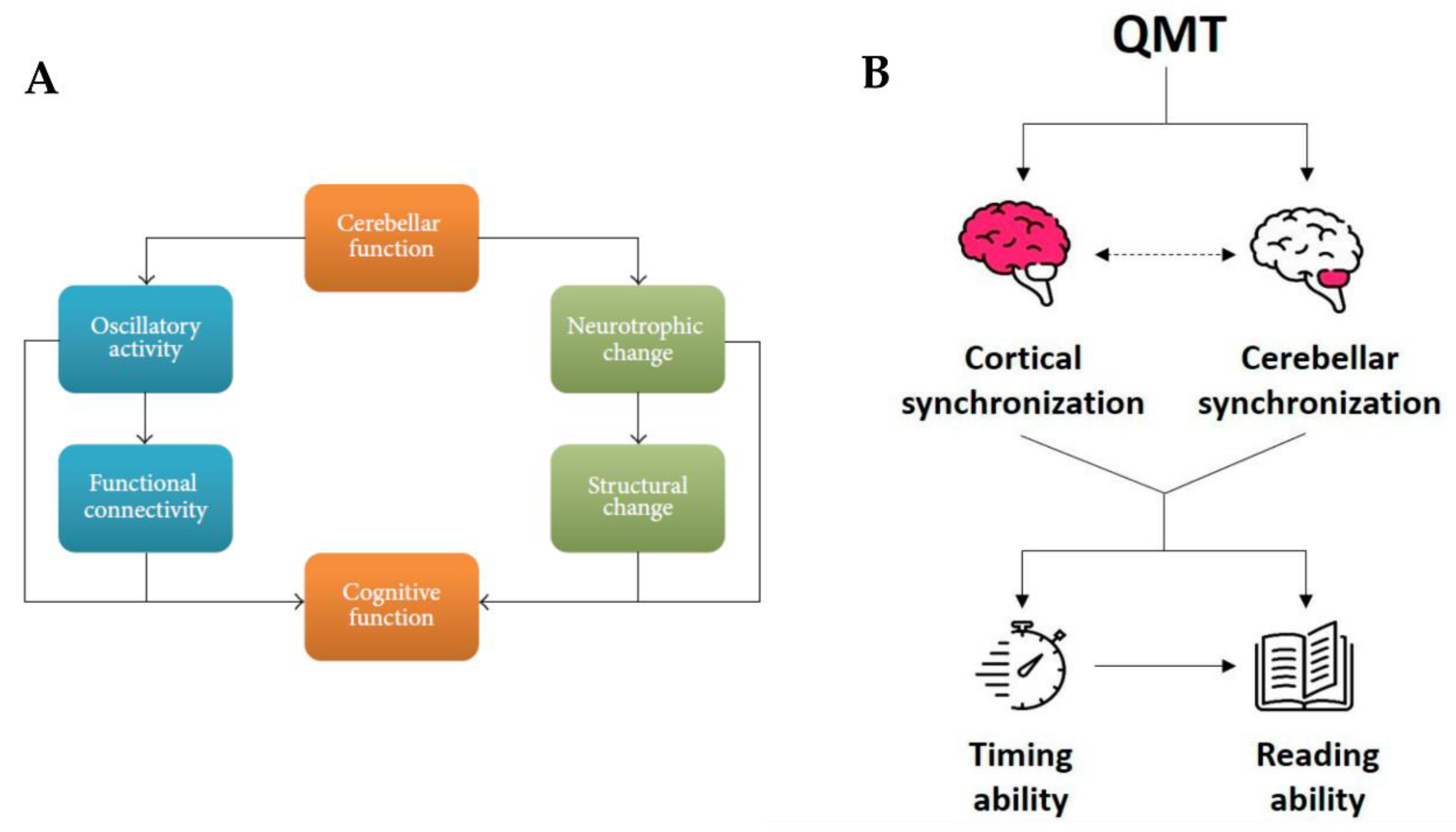
3.2. Cerebellar Involvement in Reading and Dyslexia
4. Interventions, Neuroplasticity and Dyslexia
5. Quadrato Motor Training and Dyslexia
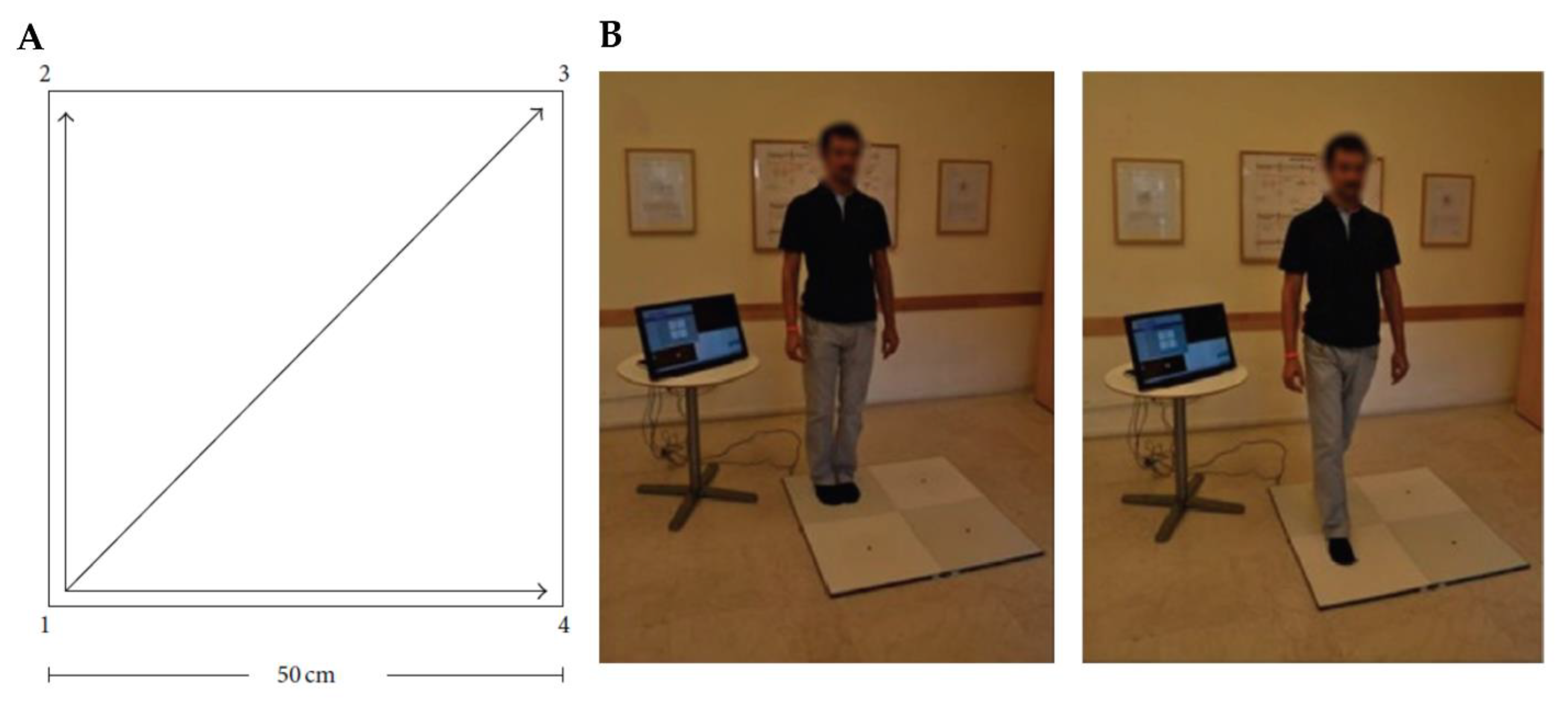
5.1. What Is Quadrato Motor Training?
5.2. Effects of Quadrato Motor Training
5.3. Quadrato Motor Training in Dyslexic Individuals
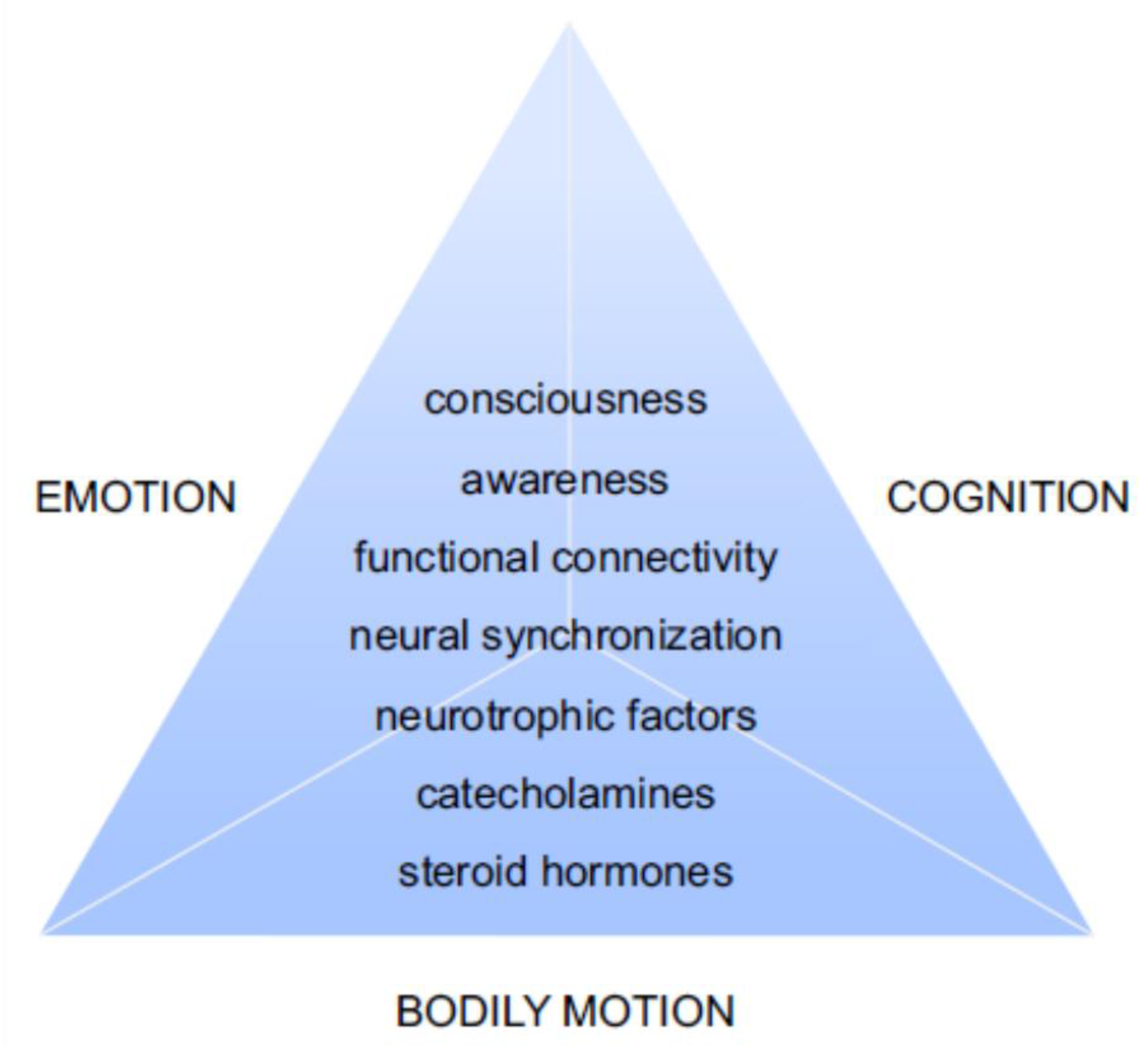
6. Discussion
6.1. Remedial Advantages of The Quadrato Motor Training
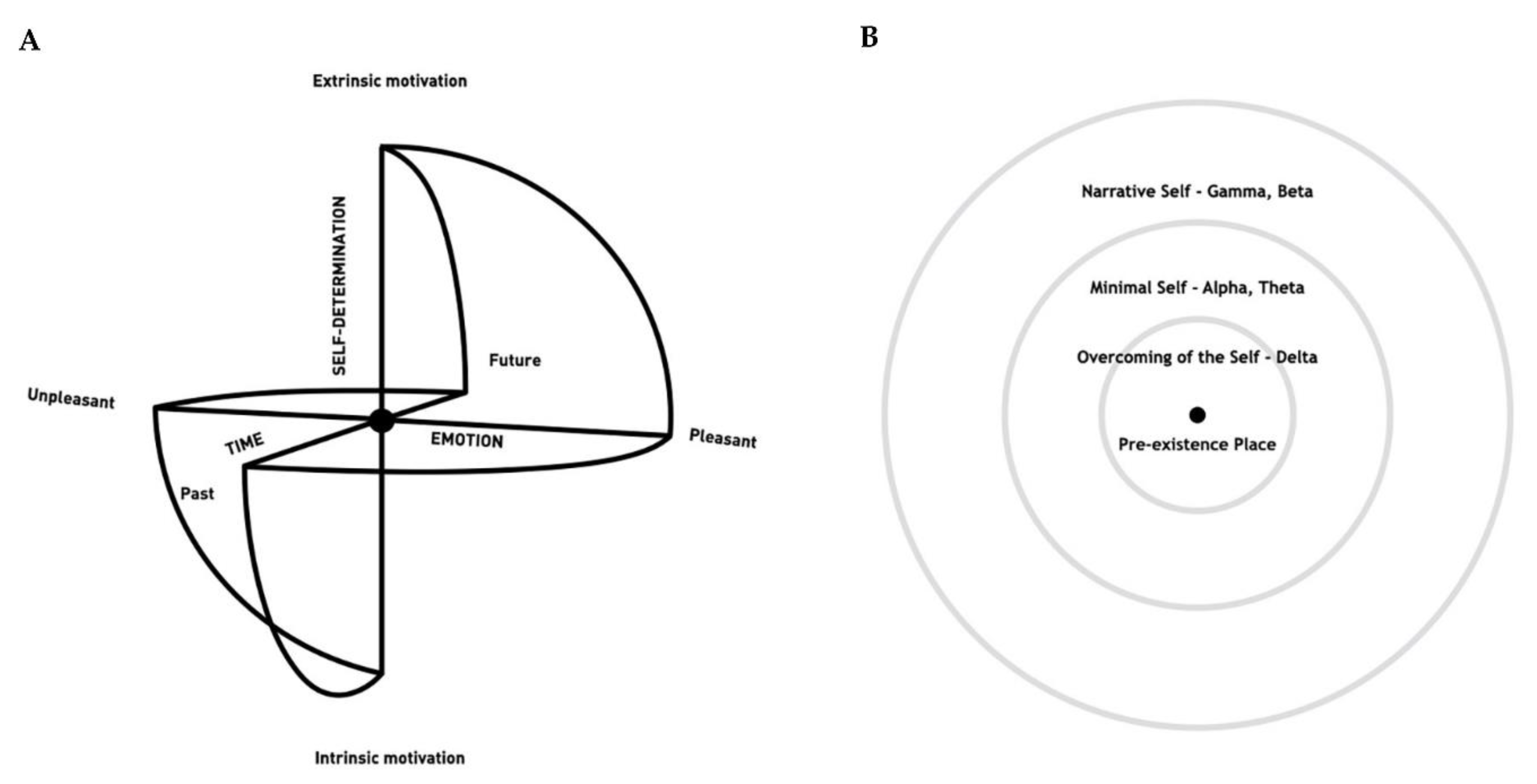
6.2. The Quadrato Motor Training within the Framework of the Sphere Model of Consciousness
7. Conclusions, Limitations and Future Directions
Author Contributions
Funding
Conflicts of Interest
References
- Freire, P. The Importance of the Act of Reading. J. Educ. 1983, 165, 5–11. [Google Scholar] [CrossRef]
- Shaywitz, S.E.; Morris, R.; Shaywitz, B.A. The Education of Dyslexic Children from Childhood to Young Adulthood. Annu. Rev. Psychol. 2008, 59, 451–475. [Google Scholar] [CrossRef]
- Grigorenko, E.L.; Compton, D.L.; Fuchs, L.S.; Wagner, R.K.; Willcutt, E.G.; Fletcher, J.M. Understanding, Educating, and Supporting Children with Specific Learning Disabilities: 50 Years of Science and Practice. Am. Psychol. 2020, 75, 37–51. [Google Scholar] [CrossRef]
- Morgan, W.P. A Case of Congenital Word Blindness. BMJ 1896, 2, 1378. [Google Scholar] [CrossRef] [PubMed]
- Kerr, J. School Hygiene, in Its Mental, Moral, and Physical Aspects. J. R. Stat. Soc. 1897, 60, 613–680. [Google Scholar] [CrossRef]
- Fletcher, J.M.; Lyon, G.R.; Fuchs, L.S.; Barnes, M.A. Learning Disabilities: From Identification to Intervention; Guilford Publications: New York, NY, USA, 2018; ISBN 1-4625-3637-9. [Google Scholar]
- Peterson, R.L.; Pennington, B.F. Developmental Dyslexia. Annu. Rev. Clin. Psychol. 2015, 11, 283–307. [Google Scholar] [CrossRef]
- Snowling, M.J.; Melby-Lervåg, M. Oral Language Deficits in Familial Dyslexia: A Meta-Analysis and Review. Psychol. Bull. 2016, 142, 498–545. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5, 5th ed.; American Psychiatric Association, Ed.; American Psychiatric Association: Washington, DC, USA, 2013; ISBN 978-0-89042-554-1. [Google Scholar]
- Lyon, G.R.; Shaywitz, S.E.; Shaywitz, B.A. A Definition of Dyslexia. Ann. Dyslexia 2003, 53, 1–14. [Google Scholar] [CrossRef]
- Scerri, T.S.; Schulte-Körne, G. Genetics of Developmental Dyslexia. Eur. Child Adolesc. Psychiatry 2010, 19, 179–197. [Google Scholar] [CrossRef]
- Stoodley, C.J.; Stein, J.F. Cerebellar Function in Developmental Dyslexia. Cerebellum 2013, 12, 267–276. [Google Scholar] [CrossRef]
- Nicolson, R.I.; Fawcett, A.J.; Dean, P. Developmental Dyslexia: The Cerebellar Deficit Hypothesis. Trends Neurosci. 2001, 24, 508–511. [Google Scholar] [CrossRef] [PubMed]
- Piek, J.P.; Dyck, M.J. Sensory-Motor Deficits in Children with Developmental Coordination Disorder, Attention Deficit Hyperactivity Disorder and Autistic Disorder. Hum. Mov. Sci. 2004, 23, 475–488. [Google Scholar] [CrossRef]
- Ben-Soussan, T.D.; Glicksohn, J.; Berkovich-Ohana, A. From Cerebellar Activation and Connectivity to Cognition: A Review of the Quadrato Motor Training. BioMed Res. Int. 2015, 2015, 954901. [Google Scholar] [CrossRef] [PubMed]
- De Fano, A.; Leshem, R.; Ben-Soussan, T.D. Creating an Internal Environment of Cognitive and Psycho-Emotional Well-Being through an External Movement-Based Environment: An Overview of Quadrato Motor Training. Int. J. Environ. Res. Public. Health 2019, 16, 2160. [Google Scholar] [CrossRef] [PubMed]
- Pesce, C.; Ben-Soussan, T.D. “Cogito Ergo Sum” or “Ambulo Ergo Sum”? New Perspectives in Developmental Exercise and Cognition Research. In Exercise-Cognition Interaction; Elsevier: Amsterdam, The Netherlands, 2016; pp. 251–282. ISBN 978-0-12-800778-5. [Google Scholar]
- Paoletti, P. Crescere Nell’eccellenza; Armando Editore: Rome, Italy, 2008; ISBN 88-6081-174-0. [Google Scholar]
- Stein, J.; Walsh, V. To See but Not to Read; the Magnocellular Theory of Dyslexia. Trends Neurosci. 1997, 20, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Snowling, M.J. From Language to Reading and Dyslexia. Dyslexia 2001, 7, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Vandermosten, M.; Boets, B.; Luts, H.; Poelmans, H.; Golestani, N.; Wouters, J.; Ghesquière, P. Adults with Dyslexia Are Impaired in Categorizing Speech and Nonspeech Sounds on the Basis of Temporal Cues. Proc. Natl. Acad. Sci. USA 2010, 107, 10389–10394. [Google Scholar] [CrossRef]
- Vidyasagar, T.R.; Pammer, K. Dyslexia: A Deficit in Visuo-Spatial Attention, Not in Phonological Processing. Trends Cogn. Sci. 2010, 14, 57–63. [Google Scholar] [CrossRef]
- Hulme, C.; Nash, H.M.; Gooch, D.; Lervåg, A.; Snowling, M.J. The Foundations of Literacy Development in Children at Familial Risk of Dyslexia. Psychol. Sci. 2015, 26, 1877–1886. [Google Scholar] [CrossRef]
- Bradley, L.; Bryant, P.E. Categorizing Sounds and Learning to Read—A Causal Connection. Nature 1983, 301, 419–421. [Google Scholar] [CrossRef]
- Shankweiler, D.; Crain, S.; Katz, L.; Fowler, A.E.; Liberman, A.M.; Brady, S.A.; Thornton, R.; Lundquist, E.; Dreyer, L.; Fletcher, J.M.; et al. Cognitive Profiles of Reading-Disabled Children: Comparison of Language Skills in Phonology, Morphology, and Syntax. Psychol. Sci. 1995, 6, 149–156. [Google Scholar] [CrossRef]
- Stanovich, K.E. The Right and Wrong Places to Look for the Cognitive Locus of Reading Disability. Ann. Dyslexia 1988, 38, 154–177. [Google Scholar] [CrossRef] [PubMed]
- Ramus, F. Neurobiology of Dyslexia: A Reinterpretation of the Data. Trends Neurosci. 2004, 27, 720–726. [Google Scholar] [CrossRef] [PubMed]
- Tallal, P.; Miller, S.; Fitch, R.H. Neurobiological Basis of Speech: A Case for the Preeminence of Temporal Processing. Ann. N. Y. Acad. Sci. 1993, 682, 27. [Google Scholar] [CrossRef]
- Eden, G.F.; VanMeter, J.W.; Rumsey, J.M.; Zeffiro, T.A. The Visual Deficit Theory of Developmental Dyslexia. NeuroImage 1996, 4, S108–S117. [Google Scholar] [CrossRef]
- Stein, J. The Magnocellular Theory of Developmental Dyslexia. Dyslexia 2001, 7, 12–36. [Google Scholar] [CrossRef]
- Nicolson, R.I.; Fawcett, A.J. Dyslexia Is More than a Phonological Disability. DYSLEXIA-CHICHESTER- 1995, 1, 19–36. [Google Scholar]
- Wolff, P.H.; Michel, G.F.; Ovrut, M.; Drake, C. Rate and Timing Precision of Motor Coordination in Developmental Dyslexia. Dev. Psychol. 1990, 26, 349. [Google Scholar] [CrossRef]
- Geuze, R.H.; Kalverboer, A.F. Tapping a Rhythm: A Problem of Timing for Children Who Are Clumsy and Dyslexic? Adapt. Phys. Act. Q. 1994, 11, 203–213. [Google Scholar] [CrossRef]
- Ivry, R.B.; Keele, S.W.; Diener, H.C. Dissociation of the Lateral and Medial Cerebellum in Movement Timing and Movement Execution. Exp. Brain Res. 1988, 73, 167–180. [Google Scholar] [CrossRef]
- Keele, S.W.; Ivry, R. Does the Cerebellum Provide a Common Computation for Diverse Tasks? A Timing Hypothesis. Ann. N. Y. Acad. Sci. 1990, 608, 179–211. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.; Ebner, T. Processing of Multiple Kinematic Signals in the Cerebellum and Motor Cortices. Brain Res. Rev. 2000, 33, 155–168. [Google Scholar] [CrossRef] [PubMed]
- Talcott, J.B.; Witton, C. A Sensory-Linguistic Approach to Normal and Impaired Reading Development. In Basic Functions of Language, Reading and Reading Disability; Witruk, E., Friederici, A.D., Lachmann, T., Eds.; Neuropsychology and Cognition; Springer: Boston, MA, USA, 2002; Volume 20, pp. 213–240. ISBN 978-1-4613-5350-8. [Google Scholar]
- Vicari, S.; Marotta, L.; Menghini, D.; Molinari, M.; Petrosini, L. Implicit Learning Deficit in Children with Developmental Dyslexia. Neuropsychologia 2003, 41, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Stoodley, C.J.; Fawcett, A.J.; Nicolson, R.I.; Stein, J.F. Impaired Balancing Ability in Dyslexic Children. Exp. Brain Res. 2005, 167, 370–380. [Google Scholar] [CrossRef]
- Vicari, S. Do Children with Developmental Dyslexia Have an Implicit Learning Deficit? J. Neurol. Neurosurg. Psychiatry 2005, 76, 1392–1397. [Google Scholar] [CrossRef] [PubMed]
- Howard, J.H.; Howard, D.V.; Japikse, K.C.; Eden, G.F. Dyslexics Are Impaired on Implicit Higher-Order Sequence Learning, but Not on Implicit Spatial Context Learning. Neuropsychologia 2006, 44, 1131–1144. [Google Scholar] [CrossRef]
- Stoodley, C.J.; Stein, J.F. A Processing Speed Deficit in Dyslexic Adults? Evidence from a Peg-Moving Task. Neurosci. Lett. 2006, 399, 264–267. [Google Scholar] [CrossRef]
- Stoodley, C.J.; Harrison, E.P.D.; Stein, J.F. Implicit Motor Learning Deficits in Dyslexic Adults. Neuropsychologia 2006, 44, 795–798. [Google Scholar] [CrossRef]
- Stoodley, C.J.; Ray, N.J.; Jack, A.; Stein, J.F. Implicit Learning in Control, Dyslexic, and Garden-variety Poor Readers. Ann. N. Y. Acad. Sci. 2008, 1145, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Boets, B.; Wouters, J.; van Wieringen, A.; De Smedt, B.; Ghesquière, P. Modelling Relations between Sensory Processing, Speech Perception, Orthographic and Phonological Ability, and Literacy Achievement. Brain Lang. 2008, 106, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Bucci, M.P.; Brémond-Gignac, D.; Kapoula, Z. Latency of Saccades and Vergence Eye Movements in Dyslexic Children. Exp. Brain Res. 2008, 188, 1–12. [Google Scholar] [CrossRef]
- Bucci, M.P.; Brémond-Gignac, D.; Kapoula, Z. Poor Binocular Coordination of Saccades in Dyslexic Children. Graefes Arch. Clin. Exp. Ophthalmol. 2008, 246, 417–428. [Google Scholar] [CrossRef]
- De Kleine, E.; Verwey, W.B. Representations Underlying Skill in the Discrete Sequence Production Task: Effect of Hand Used and Hand Position. Psychol. Res. PRPF 2009, 73, 685–694. [Google Scholar] [CrossRef]
- Boets, B.; Vandermosten, M.; Poelmans, H.; Luts, H.; Wouters, J.; Ghesquière, P. Preschool Impairments in Auditory Processing and Speech Perception Uniquely Predict Future Reading Problems. Res. Dev. Disabil. 2011, 32, 560–570. [Google Scholar] [CrossRef]
- Boets, B.; Vandermosten, M.; Cornelissen, P.; Wouters, J.; Ghesquière, P. Coherent Motion Sensitivity and Reading Development in the Transition From Prereading to Reading Stage: Coherent Motion Sensitivity and Dyslexia. Child Dev. 2011, 82, 854–869. [Google Scholar] [CrossRef]
- Nicolson, R.I.; Fawcett, A.J. Dyslexia, Dysgraphia, Procedural Learning and the Cerebellum. Cortex 2011, 47, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Fawcett, A.J.; Nicolson, R.I.; Dean, P. Impaired Performance of Children with Dyslexia on a Range of Cerebellar Tasks. Ann. Dyslexia 1996, 46, 259–283. [Google Scholar] [CrossRef]
- Hickok, G.; Houde, J.; Rong, F. Sensorimotor Integration in Speech Processing: Computational Basis and Neural Organization. Neuron 2011, 69, 407–422. [Google Scholar] [CrossRef]
- Buckner, R.L. The Cerebellum and Cognitive Function: 25 Years of Insight from Anatomy and Neuroimaging. Neuron 2013, 80, 807–815. [Google Scholar] [CrossRef]
- Akshoomoff, N.A.; Courchesne, E.; Press, G.A.; Iragui, V. Contribution of the Cerebellum to Neuropsychological Functioning: Evidence from a Case of Cerebellar Degenerative Disorder. Neuropsychologia 1992, 30, 315–328. [Google Scholar] [CrossRef]
- Appollonio, I.; Grafman, J.; Schwartz, V.; Massaquoi, S.; Hallett, M. Memory in Patients with Cerebellar Degeneration. Neurology 1993, 43, 1536. [Google Scholar] [CrossRef]
- Stoodley, C.J.; Schmahmann, J.D. The Cerebellum and Language: Evidence from Patients with Cerebellar Degeneration. Brain Lang. 2009, 110, 149–153. [Google Scholar] [CrossRef]
- Stoodley, C.J.; Stein, J.F. The Cerebellum and Dyslexia. Cortex 2011, 47, 101–116. [Google Scholar] [CrossRef]
- Eckert, M.A.; Leonard, C.M.; Richards, T.L.; Aylward, E.H.; Thomson, J.; Berninger, V.W. Anatomical Correlates of Dyslexia: Frontal and Cerebellar Findings. Brain 2003, 126, 482–494. [Google Scholar] [CrossRef]
- McGrath, L.M.; Pennington, B.F.; Shanahan, M.A.; Santerre-Lemmon, L.E.; Barnard, H.D.; Willcutt, E.G.; DeFries, J.C.; Olson, R.K. A Multiple Deficit Model of Reading Disability and Attention-deficit/Hyperactivity Disorder: Searching for Shared Cognitive Deficits. J. Child Psychol. Psychiatry 2011, 52, 547–557. [Google Scholar] [CrossRef]
- Pennington, B.F.; Santerre-Lemmon, L.; Rosenberg, J.; MacDonald, B.; Boada, R.; Friend, A.; Leopold, D.R.; Samuelsson, S.; Byrne, B.; Willcutt, E.G.; et al. Individual Prediction of Dyslexia by Single versus Multiple Deficit Models. J. Abnorm. Psychol. 2012, 121, 212–224. [Google Scholar] [CrossRef]
- Perry, C.; Zorzi, M.; Ziegler, J.C. Understanding Dyslexia Through Personalized Large-Scale Computational Models. Psychol. Sci. 2019, 30, 386–395. [Google Scholar] [CrossRef]
- Adams, R.M.; Kocsis, J.J.; Estes, R.E. Soft Neurological Signs in Learning-Disabled Children and Controls. Am. J. Dis. Child. 1974, 128, 614–618. [Google Scholar] [CrossRef]
- Kennard, M.A. Value of Equivocal Signs in Neurologic Diagnosis. Neurology 1960, 10, 753. [Google Scholar] [CrossRef]
- Stine, O.C.; Saratsiotis, J.B.; Mosser, R.S. Relationships between Neurological Findings and Classroom Behavior. Am. J. Dis. Child. 1975, 129, 1036–1040. [Google Scholar] [CrossRef]
- Wolff, P.H.; Hurwitz, I. Functional Implications of the Minimal Brain Damage Syndrome. In Minimal Cerebral Dysfunction in Children; Grune & Stratton, Inc.: New York, NY, USA, 1973. [Google Scholar]
- Denckla, M.B. Development of Speed in Repetitive and Successive Finger-movements in Normal Children. Dev. Med. Child Neurol. 1973, 15, 635–645. [Google Scholar] [CrossRef]
- Klicpera, C.; Wolff, P.H.; Drake, C. Bimanual Co-Ordination in Adolescent Boys with Reading Retardation. Dev. Med. Child Neurol. 2008, 23, 617–625. [Google Scholar] [CrossRef]
- Chiarenza, G.A. Motor-Perceptual Function in Children with Developmental Reading Disorders: Neuropsychophysiological Analysis. J. Learn. Disabil. 1990, 23, 375–385. [Google Scholar] [CrossRef]
- Chiarenza, G.A.; Di Pietro, S.F.; Casarotto, S. The Psychophysiology of Reading. Int. J. Psychophysiol. 2014, 94, 111–119. [Google Scholar] [CrossRef]
- Chiarenza, G.A.; Papakostopoulos, D.; Giordana, F.; Guareschi-Cazzullo, A. Movement-Related Brain Macropotentials during Skilled Performances. A Developmental Study. Electroencephalogr. Clin. Neurophysiol. 1983, 56, 373–383. [Google Scholar] [CrossRef]
- Chiarenza, G.A.; Olgiati, P.; Trevisan, C.; Casarotto, S. Preparatory and Pre-Lexical Periods in Dyslexic Children: A Reading-Related Potential Study; Elsevier: Amsterdam, The Netherlands, 2006; Volume 61, p. 303. [Google Scholar]
- Chiarenza, G.A. Quantitative EEG in Childhood Attention Deficit Hyperactivity Disorder and Learning Disabilities. Clin. EEG Neurosci. 2021, 52, 144–155. [Google Scholar] [CrossRef]
- Lachmann, T. Reading Disability as a Deficit in Functional Coordination. In Basic Functions of Language, Reading and Reading Disability; Witruk, E., Friederici, A.D., Lachmann, T., Eds.; Neuropsychology and Cognition; Springer: Boston, MA, USA, 2002; Volume 20, pp. 165–198. ISBN 978-1-4613-5350-8. [Google Scholar]
- Lachmann, T.; van Leeuwen, C. Paradoxical Enhancement of Letter Recognition in Developmental Dyslexia. Dev. Neuropsychol. 2007, 31, 61–77. [Google Scholar] [CrossRef]
- Novita, S. Secondary Symptoms of Dyslexia: A Comparison of Self-Esteem and Anxiety Profiles of Children with and without Dyslexia. Eur. J. Spec. Needs Educ. 2016, 31, 279–288. [Google Scholar] [CrossRef]
- Zelazo, P.D.; Carlson, S.M. Hot and Cool Executive Function in Childhood and Adolescence: Development and Plasticity. Child Dev. Perspect. 2012, 6, 354–360. [Google Scholar] [CrossRef]
- Tsermentseli, S.; Poland, S. Cool versus Hot Executive Function: A New Approach to Executive Function. Encephalos 2016, 53, 11–14. [Google Scholar]
- Zelazo, P.; Müller, U. Executive Function in Typical and Atypical Development. In Blackwell Handbook of Childhood Cognitive Development; Goswami, U., Ed.; Blackwell Publishers Ltd.: Hoboken, NJ, USA, 2002; pp. 445–469. [Google Scholar]
- Leshem, R. Using Dual Process Models to Examine Impulsivity throughout Neural Maturation. Dev. Neuropsychol. 2016, 41, 125–143. [Google Scholar] [CrossRef]
- Nelson, J.M.; Harwood, H. Learning Disabilities and Anxiety: A Meta-Analysis. J. Learn. Disabil. 2011, 44, 3–17. [Google Scholar] [CrossRef]
- Riddick, B. Living with Dyslexia: The Social and Emotional Consequences of Specific Learning Difficulties/Disabilities; Routledge: London, UK, 2012; ISBN 0-203-43260-6. [Google Scholar]
- Livingston, E.M.; Siegel, L.S.; Ribary, U. Developmental Dyslexia: Emotional Impact and Consequences. Aust. J. Learn. Difficulties 2018, 23, 107–135. [Google Scholar] [CrossRef]
- Karami, J.; Rezaee, F.; Nosrati, R.; Abasi, M.; Siahkamari, R. The Study of Self-Esteem of Dyslexic Children in Elementary School in Kermanshah. J. Pediatr. Nurs. 2019, 5, 33–40. [Google Scholar]
- Lithari, E. Fractured Academic Identities: Dyslexia, Secondary Education, Self-Esteem and School Experiences. Int. J. Incl. Educ. 2019, 23, 280–296. [Google Scholar] [CrossRef]
- Rosalina, E. The Correlation between Self-Esteem and Student’s Reading Comprehension. Engl. Lang. Teach. Educ. J. 2019, 2, 70–78. [Google Scholar]
- Giovagnoli, S.; Mandolesi, L.; Magri, S.; Gualtieri, L.; Fabbri, D.; Tossani, E.; Benassi, M. Internalizing Symptoms in Developmental Dyslexia: A Comparison between Primary and Secondary School. Front. Psychol. 2020, 11, 461. [Google Scholar] [CrossRef]
- Zuppardo, L.; Serrano, F.; Pirrone, C.; Rodriguez-Fuentes, A. More Than Words: Anxiety, Self-Esteem and Behavioral Problems in Children and Adolescents With Dyslexia. Learn. Disabil. Q. 2021. [Google Scholar] [CrossRef]
- Dufor, O.; Serniclaes, W.; Sprenger-Charolles, L.; Démonet, J.-F. Top-down Processes during Auditory Phoneme Categorization in Dyslexia: A PET Study. NeuroImage 2007, 34, 1692–1707. [Google Scholar] [CrossRef]
- Gabrieli, J.D.E. Dyslexia: A New Synergy Between Education and Cognitive Neuroscience. Science 2009, 325, 280–283. [Google Scholar] [CrossRef]
- Rumsey, J.M.; Andreason, P.; Zametkin, A.J.; Aquino, T.; King, A.C.; Hamburger, S.D.; Pikus, A.; Rapoport, J.L.; Cohen, R.M. Failure to Activate the Left Temporoparietal Cortex in Dyslexia: An Oxygen 15 Positron Emission Tomographic Study. Arch. Neurol. 1992, 49, 527–534. [Google Scholar] [CrossRef]
- Paulesu, E.; Frith, U.; Snowling, M.; Gallagher, A.; Morton, J.; Frackowiak, R.S.J.; Frith, C.D. Is Developmental Dyslexia a Disconnection Syndrome?: Evidence from PET Scanning. Brain 1996, 119, 143–157. [Google Scholar] [CrossRef]
- Rumsey, J.; Horwitz, B.; Donohue, B.; Nace, K.; Maisog, J.; Andreason, P. Phonological and Orthographic Components of Word Recognition. A PET-RCBF Study. Brain J. Neurol. 1997, 120, 739–759. [Google Scholar] [CrossRef]
- Shaywitz, S.E.; Shaywitz, B.A.; Pugh, K.R.; Fulbright, R.K.; Constable, R.T.; Mencl, W.E.; Shankweiler, D.P.; Liberman, A.M.; Skudlarski, P.; Fletcher, J.M.; et al. Functional Disruption in the Organization of the Brain for Reading in Dyslexia. Proc. Natl. Acad. Sci. USA 1998, 95, 2636–2641. [Google Scholar] [CrossRef]
- Temple, E.; Poldrack, R.A.; Salidis, J.; Deutsch, G.K.; Tallal, P.; Merzenich, M.M.; Gabrieli, J.D.E. Disrupted Neural Responses to Phonological and Orthographic Processing in Dyslexic Children: An FMRI Study. Neuroreport 2001, 12, 299–307. [Google Scholar] [CrossRef]
- Gross-glenn, K.; Duara, R.; Barker, W.W.; Loewenstein, D.; Chang, J.-Y.; Yoshii, F.; Apicella, A.M.; Pascal, S.; Boothe, T.; Sevush, S. Positron Emission Tomographic Studies during Serial Word-Reading by Normal and Dyslexic Adults. J. Clin. Exp. Neuropsychol. 1991, 13, 531–544. [Google Scholar] [CrossRef]
- Raschle, N.M.; Chang, M.; Gaab, N. Structural Brain Alterations Associated with Dyslexia Predate Reading Onset. NeuroImage 2011, 57, 742–749. [Google Scholar] [CrossRef]
- Klingberg, T.; Hedehus, M.; Temple, E.; Salz, T.; Gabrieli, J.D.; Moseley, M.E.; Poldrack, R.A. Microstructure of Temporo-Parietal White Matter as a Basis for Reading Ability: Evidence from Diffusion Tensor Magnetic Resonance Imaging. Neuron 2000, 25, 493–500. [Google Scholar] [CrossRef]
- Deutsch, G.K.; Dougherty, R.F.; Bammer, R.; Siok, W.T.; Gabrieli, J.D.E.; Wandell, B. Children’s Reading Performance Is Correlated with White Matter Structure Measured by Diffusion Tensor Imaging. Cortex 2005, 41, 354–363. [Google Scholar] [CrossRef]
- Rimrodt, S.L.; Peterson, D.J.; Denckla, M.B.; Kaufmann, W.E.; Cutting, L.E. White Matter Microstructural Differences Linked to Left Perisylvian Language Network in Children with Dyslexia. Cortex 2010, 46, 739–749. [Google Scholar] [CrossRef]
- Pernet, C.R.; Poline, J.B.; Demonet, J.F.; Rousselet, G.A. Brain Classification Reveals the Right Cerebellum as the Best Biomarker of Dyslexia. BMC Neurosci. 2009, 10, 67. [Google Scholar] [CrossRef] [PubMed]
- Pennington, B.F. Toward an Integrated Understanding of Dyslexia: Genetic, Neurological, and Cognitivemechanisms. Dev. Psychopathol. 1999, 11, 629–654. [Google Scholar] [CrossRef] [PubMed]
- Eliez, S.; Rumsey, J.M.; Giedd, J.N.; Schmitt, J.E.; Patwardhan, A.J.; Reiss, A.L. Morphological Alteration of Temporal Lobe Gray Matter in Dyslexia: An MRI Study. J. Child Psychol. Psychiatry 2000, 41, 637–644. [Google Scholar] [CrossRef]
- Brown, W.E.; Eliez, S.; Menon, V.; Rumsey, J.M.; White, C.D.; Reiss, A.L. Preliminary Evidence of Widespread Morphological Variations of the Brain in Dyslexia. Neurology 2001, 56, 781–783. [Google Scholar] [CrossRef]
- Leonard, C.M.; Eckert, M.A.; Lombardino, L.J.; Oakland, T.; Kranzler, J.; Mohr, C.M.; King, W.M.; Freeman, A. Anatomical Risk Factors for Phonological Dyslexia. Cereb. Cortex 2001, 11, 148–157. [Google Scholar] [CrossRef]
- Rae, C.; Harasty, J.A.; Dzendrowskyj, T.E.; Talcott, J.B.; Simpson, J.M.; Blamire, A.M.; Dixon, R.M.; Lee, M.A.; Thompson, C.H.; Styles, P.; et al. Cerebellar Morphology in Developmental Dyslexia. Neuropsychologia 2002, 40, 1285–1292. [Google Scholar] [CrossRef]
- Robichon, F.; Bouchard, P.; Démonet, J.-F.; Habib, M. Developmental Dyslexia: Re-Evaluation of the Corpus Callosum in Male Adults. Eur. Neurol. 2000, 43, 233–237. [Google Scholar] [CrossRef]
- Robichon, F.; Levrier, O.; Farnarier, P.; Habib, M. Developmental Dyslexia: Atypical Cortical Asymmetries and Functional Significance: Cortical Asymmetries in Developmental Dyslexia. Eur. J. Neurol. 2000, 7, 35–46. [Google Scholar] [CrossRef]
- Steinbrink, C.; Ackermann, H.; Lachmann, T.; Riecker, A. Contribution of the Anterior Insula to Temporal Auditory Processing Deficits in Developmental Dyslexia. Hum. Brain Mapp. 2009, 30, 2401–2411. [Google Scholar] [CrossRef] [PubMed]
- Pernet, C.; Andersson, J.; Paulesu, E.; Demonet, J.F. When All Hypotheses Are Right: A Multifocal Account of Dyslexia. Hum. Brain Mapp. 2009, 30, 2278–2292. [Google Scholar] [CrossRef]
- Eckert, M. Neuroanatomical Markers for Dyslexia: A Review of Dyslexia Structural Imaging Studies. Neuroscientist 2004, 10, 362–371. [Google Scholar] [CrossRef]
- Miall, R.C.; Weir, D.J.; Wolpert, D.M.; Stein, J.F. Is the Cerebellum a Smith Predictor? J. Mot. Behav. 1993, 25, 203–216. [Google Scholar] [CrossRef] [PubMed]
- Wolpert, D.M.; Miall, R.C.; Kawato, M. Internal Models in the Cerebellum. Trends Cogn. Sci. 1998, 2, 338–347. [Google Scholar] [CrossRef]
- Miall, R.C.; King, D. State Estimation in the Cerebellum. Cerebellum 2008, 7, 572–576. [Google Scholar] [CrossRef]
- Leiner, H.C.; Leiner, A.L.; Dow, R.S. Does the Cerebellum Contribute to Mental Skills? Behav. Neurosci. 1986, 100, 443. [Google Scholar] [CrossRef]
- Leiner, H.C. Solving the Mystery of the Human Cerebellum. Neuropsychol. Rev. 2010, 20, 229–235. [Google Scholar] [CrossRef]
- Raichle, M.E.; Fiez, J.A.; Videen, T.O.; MacLeod, A.-M.K.; Pardo, J.V.; Fox, P.T.; Petersen, S.E. Practice-Related Changes in Human Brain Functional Anatomy during Nonmotor Learning. Cereb. Cortex 1994, 4, 8–26. [Google Scholar] [CrossRef]
- Desmond, J.E.; Gabrieli, J.D.E.; Glover, G.H. Dissociation of Frontal and Cerebellar Activity in a Cognitive Task: Evidence for a Distinction between Selection and Search. NeuroImage 1998, 7, 368–376. [Google Scholar] [CrossRef]
- Kim, S.-G.; Uğurbil, K.; Strick, P.L. Activation of a Cerebellar Output Nucleus During Cognitive Processing. Science 1994, 265, 949–951. [Google Scholar] [CrossRef]
- Selemon’, L.D.; Goldman-Rakic, P.S. Longitudinal Topography and Lnterdigitation of Corticostriatal Projections in the Rhesus Monkey. J. Neurosci. 1985, 5, 776–794. [Google Scholar] [CrossRef]
- Selemon, L.; Goldman-Rakic, P. Common Cortical and Subcortical Targets of the Dorsolateral Prefrontal and Posterior Parietal Cortices in the Rhesus Monkey: Evidence for a Distributed Neural Network Subserving Spatially Guided Behavior. J. Neurosci. 1988, 8, 4049–4068. [Google Scholar] [CrossRef]
- Strick, P.L.; Dum, R.P.; Fiez, J.A. Cerebellum and Nonmotor Function. Annu. Rev. Neurosci. 2009, 32, 413–434. [Google Scholar] [CrossRef]
- Badre, D.; Wagner, A.D. Selection, Integration, and Conflict Monitoring: Assessing the Nature and Generality of Prefrontal Cognitive Control Mechanisms. Neuron 2004, 41, 473–487. [Google Scholar] [CrossRef]
- Hart, H.; Radua, J.; Nakao, T.; Mataix-Cols, D.; Rubia, K. Meta-Analysis of Functional Magnetic Resonance Imaging Studies of Inhibition and Attention in Attention-Deficit/Hyperactivity Disorder: Exploring Task-Specific, Stimulant Medication, and Age Effects. JAMA Psychiatry 2013, 70, 185–198. [Google Scholar] [CrossRef]
- Brunoni, A.R.; Vanderhasselt, M.-A. Working Memory Improvement with Non-Invasive Brain Stimulation of the Dorsolateral Prefrontal Cortex: A Systematic Review and Meta-Analysis. Brain Cogn. 2014, 86, 1–9. [Google Scholar] [CrossRef]
- Hertrich, I.; Dietrich, S.; Blum, C.; Ackermann, H. The Role of the Dorsolateral Prefrontal Cortex for Speech and Language Processing. Front. Hum. Neurosci. 2021, 15, 645209. [Google Scholar] [CrossRef]
- Ito, M. Control of Mental Activities by Internal Models in the Cerebellum. Nat. Rev. Neurosci. 2008, 9, 304–313. [Google Scholar] [CrossRef]
- Berman, K.F.; Ostrem, J.L.; Randolph, C.; Gold, J.; Goldberg, T.E.; Coppola, R.; Carson, R.E.; Herscovitch, P.; Weinberger, D.R. Physiological Activation of a Cortical Network during Performance of the Wisconsin Card Sorting Test: A Positron Emission Tomography Study. Neuropsychologia 1995, 33, 1027–1046. [Google Scholar] [CrossRef]
- Nagahama, Y.; Fukuyama, H.; Yamauchi, H.; Matsuzaki, S.; Konishi, J.; Shibasaki, H.; Kimura, J. Cerebral Activation during Performance of a Card Sorting Test. Brain 1996, 119, 1667–1675. [Google Scholar] [CrossRef]
- Paulesu, E.; Frith, C.D.; Frackowiak, R.S. The Neural Correlates of the Verbal Component of Working Memory. Nature 1993, 362, 342–345. [Google Scholar] [CrossRef]
- Grasby, P.; Frith, C.D.; Friston, K.J.; Simpson, J.; Fletcher, P.C.; Frackowiak, R.S.; Dolan, R.J. A Graded Task Approach to the Functional Mapping of Brain Areas Implicated in Auditory—Verbal Memory. Brain 1994, 117, 1271–1282. [Google Scholar] [CrossRef]
- Paulesu, E.; Connelly, A.; Frith, C.; Friston, K.; Heather, J.; Myers, R.; Gadian, D.; Frackowiak, R. Functional MR Imaging Correlations with Positron Emission Tomography. Initial Experience Using a Cognitive Activation Paradigm on Verbal Working Memory. Neuroimaging Clin. N. Am. 1995, 5, 207–225. [Google Scholar]
- Desmond, J.E.; Sum, J.M.; Wagner, A.D.; Demb, J.B.; Shear, P.K.; Glover, G.H.; Gabrieli, J.D.E.; Morrell, M.J. Functional MRI Measurement of Language Lateralization in Wada-Tested Patients. Brain 1995, 118, 1411–1419. [Google Scholar] [CrossRef]
- Awh, E.; Jonides, J.; Smith, E.E.; Schumacher, E.H.; Koeppe, R.A.; Katz, S. Dissociation of Storage and Rehearsal in Verbal Working Memory: Evidence From Positron Emission Tomography. Psychol. Sci. 1996, 7, 25–31. [Google Scholar] [CrossRef]
- Desmond, J.E.; Gabrieli, J.D.E.; Wagner, A.D.; Ginier, B.L.; Glover, G.H. Lobular Patterns of Cerebellar Activation in Verbal Working-Memory and Finger-Tapping Tasks as Revealed by Functional MRI. J. Neurosci. 1997, 17, 9675–9685. [Google Scholar] [CrossRef]
- Jonides, J.; Schumacher, E.H.; Smith, E.E.; Lauber, E.J.; Awh, E.; Minoshima, S.; Koeppe, R.A. Verbal Working Memory Load Affects Regional Brain Activation as Measured by PET. J. Cogn. Neurosci. 1997, 9, 462–475. [Google Scholar] [CrossRef]
- de Zubicaray, G.I.; Williams, S.C.R.; Wilson, S.J.; Rose, S.E.; Brammer, M.J.; Bullmore, E.T.; Simmons, A.; Chalk, J.B.; Semple, J.; Brown, A.P.; et al. Prefrontal Cortex Involvement in Selective Letter Generation: A Functional Magnetic Resonance Imaging Study. Cortex 1998, 34, 389–401. [Google Scholar] [CrossRef]
- Diamond, A. Close Interrelation of Motor Development and Cognitive Development and of the Cerebellum and Prefrontal Cortex. Child Dev. 2000, 71, 44–56. [Google Scholar] [CrossRef]
- Palva, S.; Palva, J.M. New Vistas for α-Frequency Band Oscillations. Trends Neurosci. 2007, 30, 150–158. [Google Scholar] [CrossRef]
- Kujala, J.; Pammer, K.; Cornelissen, P.; Roebroeck, A.; Formisano, E.; Salmelin, R. Phase Coupling in a Cerebro-Cerebellar Network at 8–13 Hz during Reading. Cereb. Cortex 2007, 17, 1476–1485. [Google Scholar] [CrossRef]
- Palva, J.M.; Monto, S.; Kulashekhar, S.; Palva, S. Neuronal Synchrony Reveals Working Memory Networks and Predicts Individual Memory Capacity. Proc. Natl. Acad. Sci. USA 2010, 107, 7580–7585. [Google Scholar] [CrossRef] [PubMed]
- Silberstein, R.B.; Danieli, F.; Nunez, P.L. Fronto-Parietal Evoked Potential Synchronization Is Increased During Mental Rotation. NeuroReport 2003, 14, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, A. Transient Hypofrontality as a Mechanism for the Psychological Effects of Exercise. Psychiatry Res. 2006, 145, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Fink, A.; Graif, B.; Neubauer, A.C. Brain Correlates Underlying Creative Thinking: EEG Alpha Activity in Professional vs. Novice Dancers. NeuroImage 2009, 46, 854–862. [Google Scholar] [CrossRef]
- Watson, T.C.; Becker, N.; Apps, R.; Jones, M.W. Back to Front: Cerebellar Connections and Interactions with the Prefrontal Cortex. Front. Syst. Neurosci. 2014, 8, 4. [Google Scholar] [CrossRef]
- Schmahmann, J.D.; Sherman, J.C. The Cerebellar Cognitive Affective Syndrome. Brain J. Neurol. 1998, 121, 561–579. [Google Scholar] [CrossRef]
- Schmahmann, J.D. Disorders of the Cerebellum: Ataxia, Dysmetria of Thought, and the Cerebellar Cognitive Affective Syndrome. J. Neuropsychiatry Clin. Neurosci. 2004, 16, 367–378. [Google Scholar] [CrossRef]
- Hölzel, B.K.; Carmody, J.; Vangel, M.; Congleton, C.; Yerramsetti, S.M.; Gard, T.; Lazar, S.W. Mindfulness Practice Leads to Increases in Regional Brain Gray Matter Density. Psychiatry Res. Neuroimaging 2011, 191, 36–43. [Google Scholar] [CrossRef]
- Chávez-Eakle, R.A.; Graff-Guerrero, A.; García-Reyna, J.-C.; Vaugier, V.; Cruz-Fuentes, C. Cerebral Blood Flow Associated with Creative Performance: A Comparative Study. NeuroImage 2007, 38, 519–528. [Google Scholar] [CrossRef]
- Vandervert, L.R. Cognitive Functions of the Cerebellum Explain How Ericsson’s Deliberate Practice Produces Giftedness. High Abil. Stud. 2007, 18, 89–92. [Google Scholar] [CrossRef]
- Vandervert, L.R.; Schimpf, P.H.; Liu, H. How Working Memory and the Cerebellum Collaborate to Produce Creativity and Innovation. Creat. Res. J. 2007, 19, 1–18. [Google Scholar] [CrossRef]
- Schlosser, R.; Hutchinson, M.; Joseffer, S.; Rusinek, H.; Saarimaki, A.; Stevenson, J.; Dewey, S.L.; Brodie, J.D. Functional Magnetic Resonance Imaging of Human Brain Activity in a Verbal Fluency Task. J. Neurol. Neurosurg. Psychiatry 1998, 64, 492–498. [Google Scholar] [CrossRef]
- Ackermann, H.; Riecker, A.; Mathiak, K.; Erb, M.; Grodd, W.; Wildgruber, D. Rate-Dependent Activation of a Prefrontal-Insular-Cerebellar Network during Passive Listening to Trains of Click Stimuli: An FMRI Study. Neuroreport 2001, 12, 4087–4092. [Google Scholar] [CrossRef] [PubMed]
- Wildgruber, D.; Ackermann, H.; Grodd, W. Differential Contributions of Motor Cortex, Basal Ganglia, and Cerebellum to Speech Motor Control: Effects of Syllable Repetition Rate Evaluated by FMRI. NeuroImage 2001, 13, 101–109. [Google Scholar] [CrossRef]
- Marien, P.; Engelborghs, S.; Fabbro, F.; De Deyn, P.P. The Lateralized Linguistic Cerebellum: A Review and a New Hypothesis. Brain Lang. 2001, 79, 580–600. [Google Scholar] [CrossRef] [PubMed]
- Stoodley, C.J. The Cerebellum and Cognition: Evidence from Functional Imaging Studies. Cerebellum 2012, 11, 352–365. [Google Scholar] [CrossRef]
- Stoodley, C.J.; Valera, E.M.; Schmahmann, J.D. Functional Topography of the Cerebellum for Motor and Cognitive Tasks: An FMRI Study. NeuroImage 2012, 59, 1560–1570. [Google Scholar] [CrossRef] [PubMed]
- Keren-Happuch, E.; Chen, S.A.; Ho, M.R.; Desmond, J.E. A Meta-analysis of Cerebellar Contributions to Higher Cognition from PET and FMRI Studies. Hum. Brain Mapp. 2014, 35, 593. [Google Scholar]
- Booth, J.R.; Wood, L.; Lu, D.; Houk, J.C.; Bitan, T. The Role of the Basal Ganglia and Cerebellum in Language Processing. Brain Res. 2007, 1133, 136–144. [Google Scholar] [CrossRef] [PubMed]
- Turkeltaub, P.E.; Gareau, L.; Flowers, D.L.; Zeffiro, T.A.; Eden, G.F. Development of Neural Mechanisms for Reading. Nat. Neurosci. 2003, 6, 767–773. [Google Scholar] [CrossRef]
- Mechelli, A.; Gorno-Tempini, M.L.; Price, C.J. Neuroimaging Studies of Word and Pseudoword Reading: Consistencies, Inconsistencies, and Limitations. J. Cogn. Neurosci. 2003, 15, 260–271. [Google Scholar] [CrossRef]
- Kibby, M.Y.; Fancher, J.B.; Markanen, R.; Hynd, G.W. A Quantitative Magnetic Resonance Imaging Analysis of the Cerebellar Deficit Hypothesis of Dyslexia. J. Child Neurol. 2008, 23, 368–380. [Google Scholar] [CrossRef]
- Leonard, C.M.; Kuldau, J.M.; Maron, L.; Ricciuti, N.; Mahoney, B.; Bengtson, M.; DeBose, C. Identical Neural Risk Factors Predict Cognitive Deficit in Dyslexia and Schizophrenia. Neuropsychology 2008, 22, 147–158. [Google Scholar] [CrossRef]
- Silani, G.; Frith, U.; Demonet, J.-F.; Fazio, F.; Perani, D.; Price, C.; Frith, C.D.; Paulesu, E. Brain Abnormalities Underlying Altered Activation in Dyslexia: A Voxel Based Morphometry Study. Brain 2005, 128, 2453–2461. [Google Scholar] [CrossRef]
- Hoeft, F.; Meyler, A.; Hernandez, A.; Juel, C.; Taylor-Hill, H.; Martindale, J.L.; McMillon, G.; Kolchugina, G.; Black, J.M.; Faizi, A.; et al. Functional and Morphometric Brain Dissociation between Dyslexia and Reading Ability. Proc. Natl. Acad. Sci. USA 2007, 104, 4234–4239. [Google Scholar] [CrossRef]
- Brambati, S.M.; Termine, C.; Ruffino, M.; Stella, G.; Fazio, F.; Cappa, S.F.; Perani, D. Regional Reductions of Gray Matter Volume in Familial Dyslexia. Neurology 2004, 63, 742–745. [Google Scholar] [CrossRef]
- Kronbichler, M.; Wimmer, H.; Staffen, W.; Hutzler, F.; Mair, A.; Ladurner, G. Developmental Dyslexia: Gray Matter Abnormalities in the Occipitotemporal Cortex. Hum. Brain Mapp. 2008, 29, 613–625. [Google Scholar] [CrossRef]
- Siok, W.T.; Niu, Z.; Jin, Z.; Perfetti, C.A.; Tan, L.H. A Structural–Functional Basis for Dyslexia in the Cortex of Chinese Readers. Proc. Natl. Acad. Sci. USA 2008, 105, 5561–5566. [Google Scholar] [CrossRef]
- Hu, W.; Lee, H.L.; Zhang, Q.; Liu, T.; Geng, L.B.; Seghier, M.L.; Shakeshaft, C.; Twomey, T.; Green, D.W.; Yang, Y.M.; et al. Developmental Dyslexia in Chinese and English Populations: Dissociating the Effect of Dyslexia from Language Differences. Brain 2010, 133, 1694–1706. [Google Scholar] [CrossRef]
- Horwitz, B.; Rumsey, J.M.; Donohue, B.C. Functional Connectivity of the Angular Gyrus in Normal Reading and Dyslexia. Proc. Natl. Acad. Sci. USA 1998, 95, 8939–8944. [Google Scholar] [CrossRef]
- Stanberry, L.I.; Richards, T.L.; Berninger, V.W.; Nandy, R.R.; Aylward, E.H.; Maravilla, K.R.; Stock, P.S.; Cordes, D. Low-Frequency Signal Changes Reflect Differences in Functional Connectivity between Good Readers and Dyslexics during Continuous Phoneme Mapping. Magn. Reson. Imaging 2006, 24, 217–229. [Google Scholar] [CrossRef]
- Shaywitz, S.E.; Shaywitz, B.A. Dyslexia (Specific Reading Disability). Biol. Psychiatry 2005, 57, 1301–1309. [Google Scholar] [CrossRef]
- Martin, J.; Colé, P.; Leuwers, C.; Casalis, S.; Zorman, M.; Sprenger-Charolles, L. Reading in French-Speaking Adults with Dyslexia. Ann. Dyslexia 2010, 60, 238–264. [Google Scholar] [CrossRef]
- Breznitz, Z.; Shaul, S.; Horowitz-Kraus, T.; Sela, I.; Nevat, M.; Karni, A. Enhanced Reading by Training with Imposed Time Constraint in Typical and Dyslexic Adults. Nat. Commun. 2013, 4, 1486. [Google Scholar] [CrossRef]
- Cavalli, E.; Colé, P.; Brèthes, H.; Lefevre, E.; Lascombe, S.; Velay, J.-L. E-Book Reading Hinders Aspects of Long-Text Comprehension for Adults with Dyslexia. Ann. Dyslexia 2019, 69, 243–259. [Google Scholar] [CrossRef]
- Landerl, K.; Wimmer, H. Development of Word Reading Fluency and Spelling in a Consistent Orthography: An 8-Year Follow-Up. J. Educ. Psychol. 2008, 100, 150–161. [Google Scholar] [CrossRef]
- Paizi, D.; Zoccolotti, P.; Burani, C. Lexical Reading in Italian Developmental Dyslexic Readers. In Reading and Dyslexia in Different Orthographies; Psychology Press: Florence, KY, USA, 2010; pp. 199–216. ISBN 0-203-85846-8. [Google Scholar]
- Geredakis, A.; Karala, M.; Ziavra, N.; Toki, E. Preliminary Measurements of Voice Parameters Using Multi Dimensional Voice Program. World J. Res. Rev. 2017, 5, 6. [Google Scholar]
- National Reading Panel (US); National Institute of Child Health and Human Development (US). Teaching Children to Read: An Evidence-Based Assessment of the Scientific Research Literature on Reading and Its Implications for Reading Instruction: Reports of the Subgroups; National Institute of Child Health and Human Development: Washinngton, DC, USA, 2000.
- Torgesen, J.K.; Wagner, R.K.; Rashotte, C.A.; Rose, E.; Lindamood, P.; Conway, T.; Garvan, C. Preventing Reading Failure in Young Children with Phonological Processing Disabilities: Group and Individual Responses to Instruction. J. Educ. Psychol. 1999, 91, 579. [Google Scholar] [CrossRef]
- Torgesen, J.K.; Alexander, A.W.; Wagner, R.K.; Rashotte, C.A.; Voeller, K.K.; Conway, T. Intensive Remedial Instruction for Children with Severe Reading Disabilities: Immediate and Long-Term Outcomes from Two Instructional Approaches. J. Learn. Disabil. 2001, 34, 33–58. [Google Scholar] [CrossRef]
- Simos, P.G.; Fletcher, J.M.; Bergman, E.; Breier, J.I.; Foorman, B.R.; Castillo, E.M.; Davis, R.N.; Fitzgerald, M.; Papanicolaou, A.C. Dyslexia-Specific Brain Activation Profile Becomes Normal Following Successful Remedial Training. Neurology 2002, 58, 1203–1213. [Google Scholar] [CrossRef]
- Alexander, A.W.; Slinger-Constant, A.-M. Current Status of Treatments for Dyslexia: Critical Review. J. Child Neurol. 2004, 19, 744–758. [Google Scholar] [CrossRef] [PubMed]
- Leloup, G.; Anders, R.; Charlet, V.; Eula-Fantozzi, B.; Fossoud, C.; Cavalli, E. Improving Reading Skills in Children with Dyslexia: Efficacy Studies on a Newly Proposed Remedial Intervention—Repeated Reading with Vocal Music Masking (RVM). Ann. Dyslexia 2021, 71, 60–83. [Google Scholar] [CrossRef] [PubMed]
- Breznitz, Z. Enhancing the Reading of Dyslexic Children by Reading Acceleration and Auditory Masking. J. Educ. Psychol. 1997, 89, 103. [Google Scholar] [CrossRef]
- Tomatis, A. Pourquoi Mozart?: Essai; Fixot: Paris, France, 1991; ISBN 2-87645-107-7. [Google Scholar]
- Ganschow, L.; Lloyd-Jones, J.; Miles, T.R. Dyslexia and Musical Notation. Ann. Dyslexia 1994, 44, 185–202. [Google Scholar] [CrossRef]
- Oglethorpe, S. Instrumental Music for Dyslexics: A Teaching Handbook; John Wiley & Sons: Hoboken, NJ, USA, 2008; ISBN 0-470-77799-0. [Google Scholar]
- Vladikovic, J. Gifted Learners, Dyslexia, Music, and the Piano: Rude, Inattentive, Uncooperative, or Something Else? Arizona State University: Tempe, AZ, USA, 2013; ISBN 1-303-20624-2. [Google Scholar]
- Koelsch, S. Brain Correlates of Music-Evoked Emotions. Nat. Rev. Neurosci. 2014, 15, 170–180. [Google Scholar] [CrossRef]
- Zatorre, R.J.; Chen, J.L.; Penhune, V.B. When the Brain Plays Music: Auditory–Motor Interactions in Music Perception and Production. Nat. Rev. Neurosci. 2007, 8, 547–558. [Google Scholar] [CrossRef]
- Hyde, K.L.; Lerch, J.; Norton, A.; Forgeard, M.; Winner, E.; Evans, A.C.; Schlaug, G. Musical Training Shapes Structural Brain Development. J. Neurosci. 2009, 29, 3019–3025. [Google Scholar] [CrossRef]
- Eren, B. Music and Dyslexia: The Therapeutic Use of Instrument (Piano) Training with a Child with Dyslexia (A Case Study). J. Educ. Pract. 2017, 8, 97–108. [Google Scholar]
- Hudson, K.N.; Ballou, H.M.; Willoughby, M.T. Improving Motor Competence Skills in Early Childhood Has Corollary Benefits for Executive Function and Numeracy Skills. Dev. Sci. 2021, 24, e13071. [Google Scholar] [CrossRef]
- Saletta, M.; Gladfelter, A.; Vuolo, J.; Goffman, L. Interaction of Motor and Language Factors in the Development of Speech Production. In Routledge Handbook of Communication Disorders; Routledge: London, UK, 2015; pp. 381–392. ISBN 0-203-56924-5. [Google Scholar]
- Lyytinen, H.; Ahonen, T.; Eklund, K.; Guttorm, T.K.; Laakso, M.-L.; Leinonen, S.; Leppanen, P.H.; Lyytinen, P.; Poikkeus, A.-M.; Puolakanaho, A. Developmental Pathways of Children with and without Familial Risk for Dyslexia during the First Years of Life. Dev. Neuropsychol. 2001, 20, 535–554. [Google Scholar] [CrossRef]
- Viholainen, H.; Ahonen, T.; Cantell, M.; Lyytinen, P.; Lyytinen, H. Development of Early Motor Skills and Language in Children at Risk for Familial Dyslexia. Dev. Med. Child Neurol. 2002, 44, 761–769. [Google Scholar] [CrossRef] [PubMed]
- Getchell, N.; Pabreja, P.; Neeld, K.; Carrio, V. Comparing Children with and without Dyslexia on the Movement Assessment Battery for Children and the Test of Gross Motor Development. Percept. Mot. Skills 2007, 105, 207–214. [Google Scholar] [CrossRef]
- Sibley, B.A.; Etnier, J.L. The Relationship between Physical Activity and Cognition in Children: A Meta-Analysis. Pediatr. Exerc. Sci. 2003, 15, 243–256. [Google Scholar] [CrossRef]
- Ben-Yehudah, G.; Fiez, J.A. Impact of Cerebellar Lesions on Reading and Phonological Processing. Ann. N. Y. Acad. Sci. 2008, 1145, 260–274. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, D.; Nicolson, R.I.; Hambly, H. Evaluation of an Exercise-Based Treatment for Children with Reading Difficulties. Dyslexia 2003, 9, 48–71. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, D.; Nicolson, R.I. Follow-up of an Exercise-Based Treatment for Children with Reading Difficulties. Dyslexia 2007, 13, 78–96. [Google Scholar] [CrossRef] [PubMed]
- Snowling, M.J.; Hulme, C. A Critique of Claims from Reynolds, Nicolson & Hambly (2003) That DDAT Is an Effective Treatment for Children with Reading Difficulties-?Lies, Damned Lies and (Inappropriate) Statistics?? Dyslexia 2003, 9, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Rack, J.P.; Snowling, M.J.; Hulme, C.; Gibbs, S. No Evidence That an Exercise-Based Treatment Programme (DDAT) Has Specific Benefits for Children with Reading Difficulties. Dyslexia 2007, 13, 97–104. [Google Scholar] [CrossRef]
- Denton, C.A. Physical Exercise and Movement-Based Interventions for Dyslexia. Pediatrics 1999, 1150, 27–31. [Google Scholar]
- Levit-Binnun, N.; Golland, Y. Finding Behavioral and Network Indicators of Brain Vulnerability. Front. Hum. Neurosci. 2012, 6, 10. [Google Scholar] [CrossRef]
- Levit-Binnun, N.; Davidovitch, M.; Golland, Y. Sensory and Motor Secondary Symptoms as Indicators of Brain Vulnerability. J. Neurodev. Disord. 2013, 5, 26. [Google Scholar] [CrossRef] [PubMed]
- Courchesne, E.; Townsend, J.; Akshoomoff, N.A.; Saitoh, O.; Yeung-Courchesne, R.; Lincoln, A.J.; James, H.E.; Haas, R.H.; Schreibman, L.; Lau, L. Impairment in Shifting Attention in Autistic and Cerebellar Patients. Behav. Neurosci. 1994, 108, 848. [Google Scholar] [CrossRef] [PubMed]
- Lu, B.; Pang, P.T.; Woo, N.H. The Yin and Yang of Neurotrophin Action. Nat. Rev. Neurosci. 2005, 6, 603–614. [Google Scholar] [CrossRef]
- Cohen-Cory, S.; Kidane, A.H.; Shirkey, N.J.; Marshak, S. Brain-Derived Neurotrophic Factor and the Development of Structural Neuronal Connectivity. Dev. Neurobiol. 2010, 70, 271–288. [Google Scholar] [CrossRef] [PubMed]
- Mandel, R.J.; Gage, F.H.; Thal, L.J. Spatial Learning in Rats: Correlation with Cortical Choline Acetyltransferase and Improvement with NGF Following NBM Damage. Exp. Neurol. 1989, 104, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Angelucci, F.; De Bartolo, P.; Gelfo, F.; Foti, F.; Cutuli, D.; Bossù, P.; Caltagirone, C.; Petrosini, L. Increased Concentrations of Nerve Growth Factor and Brain-Derived Neurotrophic Factor in the Rat Cerebellum After Exposure to Environmental Enrichment. Cerebellum 2009, 8, 499–506. [Google Scholar] [CrossRef]
- Conner, J.M.; Franks, K.M.; Titterness, A.K.; Russell, K.; Merrill, D.A.; Christie, B.R.; Sejnowski, T.J.; Tuszynski, M.H. NGF Is Essential for Hippocampal Plasticity and Learning. J. Neurosci. 2009, 29, 10883–10889. [Google Scholar] [CrossRef]
- Fritsch, B.; Reis, J.; Martinowich, K.; Schambra, H.M.; Ji, Y.; Cohen, L.G.; Lu, B. Direct Current Stimulation Promotes BDNF-Dependent Synaptic Plasticity: Potential Implications for Motor Learning. Neuron 2010, 66, 198–204. [Google Scholar] [CrossRef]
- AL-SHAWI, R.; Hafner, A.; Chun, S.; Raza, S.; Crutcher, K.; Thrasivoulou, C.; Simons, P.; Cowen, T. ProNGF, Sortilin, and Age-related Neurodegeneration. Ann. N. Y. Acad. Sci. 2007, 1119, 208–215. [Google Scholar] [CrossRef]
- Cotman, C.W.; Berchtold, N.C. Exercise: A Behavioral Intervention to Enhance Brain Health and Plasticity. Trends Neurosci. 2002, 25, 295–301. [Google Scholar] [CrossRef]
- Gatt, J.M.; Kuan, S.A.; Dobson-Stone, C.; Paul, R.H.; Joffe, R.T.; Kemp, A.H.; Gordon, E.; Schofield, P.R.; Williams, L.M. Association between BDNF Val66Met Polymorphism and Trait Depression Is Mediated via Resting EEG Alpha Band Activity. Biol. Psychol. 2008, 79, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Voss Plasticity of Brain Networks in a Randomized Intervention Trial of Exercise Training in Older Adults. Front. Aging Neurosci. 2010, 2, 32. [CrossRef]
- Voss, M.W.; Erickson, K.I.; Prakash, R.S.; Chaddock, L.; Kim, J.S.; Alves, H.; Szabo, A.; Phillips, S.M.; Wójcicki, T.R.; Mailey, E.L.; et al. Neurobiological Markers of Exercise-Related Brain Plasticity in Older Adults. Brain. Behav. Immun. 2013, 28, 90–99. [Google Scholar] [CrossRef]
- Ernfors, P.; Lee, K.-F.; Kucera, J.; Jaenisch, R. Lack of Neurotrophin-3 Leads to Deficiencies in the Peripheral Nervous System and Loss of Limb Proprioceptive Afferents. Cell 1994, 77, 503–512. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, P.M.; Borghesani, P.R.; Levy, R.L.; Pomeroy, S.L.; Segal, R.A. Abnormal Cerebellar Development and Foliation in BDNF−/− Mice Reveals a Role for Neurotrophins in CNS Patterning. Neuron 1997, 19, 269–281. [Google Scholar] [CrossRef]
- Holschneider, D.P.; Yang, J.; Guo, Y.; Maarek, J.-M.I. Reorganization of Functional Brain Maps after Exercise Training: Importance of Cerebellar–Thalamic–Cortical Pathway. Brain Res. 2007, 1184, 96–107. [Google Scholar] [CrossRef]
- Foti, F.; Laricchiuta, D.; Cutuli, D.; De Bartolo, P.; Gelfo, F.; Angelucci, F.; Petrosini, L. Exposure to an Enriched Environment Accelerates Recovery from Cerebellar Lesion. Cerebellum 2011, 10, 104–119. [Google Scholar] [CrossRef]
- Crabbe, J.B.; Dishman, R.K. Brain Electrocortical Activity during and after Exercise: A Quantitative Synthesis. Psychophysiology 2004, 41, 563–574. [Google Scholar] [CrossRef] [PubMed]
- Swinnen, S.P. Intermanual Coordination: From Behavioural Principles to Neural-Network Interactions. Nat. Rev. Neurosci. 2002, 3, 348–359. [Google Scholar] [CrossRef] [PubMed]
- De Zeeuw, C.I.; Hoebeek, F.E.; Bosman, L.W.J.; Schonewille, M.; Witter, L.; Koekkoek, S.K. Spatiotemporal Firing Patterns in the Cerebellum. Nat. Rev. Neurosci. 2011, 12, 327–344. [Google Scholar] [CrossRef]
- Carbon, M.; Kingsley, P.B.; Tang, C.; Bressman, S.; Eidelberg, D. Microstructural White Matter Changes in Primary Torsion Dystonia: White Matter Changes in Primary Dystonia. Mov. Disord. 2008, 23, 234–239. [Google Scholar] [CrossRef]
- Carbon, M.; Argyelan, M.; Habeck, C.; Ghilardi, M.F.; Fitzpatrick, T.; Dhawan, V.; Pourfar, M.; Bressman, S.B.; Eidelberg, D. Increased Sensorimotor Network Activity in DYT1 Dystonia: A Functional Imaging Study. Brain 2010, 133, 690–700. [Google Scholar] [CrossRef]
- Dotan Ben-Soussan, T.; Glicksohn, J.; Goldstein, A.; Berkovich-Ohana, A.; Donchin, O. Into the Square and out of the Box: The Effects of Quadrato Motor Training on Creativity and Alpha Coherence. PLoS ONE 2013, 8, e55023. [Google Scholar] [CrossRef] [PubMed]
- Leshem, R.; De Fano, A.; Ben-Soussan, T.D. The Implications of Motor and Cognitive Inhibition for Hot and Cool Executive Functions: The Case of Quadrato Motor Training. Front. Psychol. 2020, 11, 940. [Google Scholar] [CrossRef]
- Swanson, H.L.; Sachse-Lee, C. A Subgroup Analysis of Working Memory in Children with Reading Disabilities: Domain-General or Domain-Specific Deficiency? J. Learn. Disabil. 2001, 34, 249–263. [Google Scholar] [CrossRef]
- Lee Swanson, H. Age-Related Differences in Learning Disabled and Skilled Readers’ Working Memory. J. Exp. Child Psychol. 2003, 85, 1–31. [Google Scholar] [CrossRef]
- Swanson, J.W.; Swartz, M.S.; Dorn, R.A.V.; Elbogen, E.B.; Wagner, H.R.; Rosenheck, R.A.; Stroup, T.S.; McEvoy, J.P.; Lieberman, J.A. A National Study of Violent Behavior in Persons With Schizophrenia. Arch. Gen. Psychiatry 2006, 63, 490–499. [Google Scholar] [CrossRef]
- Berninger, V.W.; Nielsen, K.H.; Abbott, R.D.; Wijsman, E.; Raskind, W. Writing Problems in Developmental Dyslexia: Under-Recognized and under-Treated. J. Sch. Psychol. 2008, 46, 1–21. [Google Scholar] [CrossRef]
- Ben-Soussan, T.D.; Berkovich-Ohana, A.; Glicksohn, J.; Goldstein, A. A Suspended Act: Increased Reflectivity and Gender-Dependent Electrophysiological Change Following Quadrato Motor Training. Front. Psychol. 2014, 5, 55. [Google Scholar] [CrossRef] [PubMed]
- Kabat-Zinn, J. The Foundations of Mindfulness Practice: Attitudes and Commitment. Full Catastr. Living Using Wisdom Your Mind Face Stress Pain Illn. 2013, 19–38. [Google Scholar]
- Piervincenzi, C.; Ben-Soussan, T.D.; Mauro, F.; Mallio, C.A.; Errante, Y.; Quattrocchi, C.C.; Carducci, F. White Matter Microstructural Changes Following Quadrato Motor Training: A Longitudinal Study. Front. Hum. Neurosci. 2017, 11, 590. [Google Scholar] [CrossRef]
- Russell, T.A.; Arcuri, S.M. A Neurophysiological and Neuropsychological Consideration of Mindful Movement: Clinical and Research Implications. Front. Hum. Neurosci. 2015, 9, 282. [Google Scholar] [CrossRef]
- Clark, D.; Schumann, F.; Mostofsky, S.H. Mindful Movement and Skilled Attention. Front. Hum. Neurosci. 2015, 9, 297. [Google Scholar] [CrossRef]
- Kimmel, M.; Rogler, C.R. Affordances in Interaction—The Case of Aikido. Ecol. Psychol. 2018. [Google Scholar] [CrossRef]
- Diamond, A.; Ling, D.S. Aerobic-Exercise and Resistance-Training Interventions Have Been among the Least Effective Ways to Improve Executive Functions of Any Method Tried Thus Far. Dev. Cogn. Neurosci. 2019, 37, 100572. [Google Scholar] [CrossRef]
- Ben-Soussan, T.D.; Berkovich-Ohana, A.; Piervincenzi, C.; Glicksohn, J.; Carducci, F. Embodied Cognitive Flexibility and Neuroplasticity Following Quadrato Motor Training. Front. Psychol. 2015, 6, 1021. [Google Scholar] [CrossRef]
- Paoletti, P.; Glicksohn, J.; Ben-Soussan, T.D. Inner Design Technology: Improved Affect by Quadrato Motor Training. In The Amygdala—Where Emotions Shape Perception, Learning and Memories; InTech: Rijeka, Croatia, 2017; pp. 27–41. [Google Scholar]
- Marson, F.; Fano, A.D.; Pellegrino, M.; Pesce, C.; Glicksohn, J.; Ben-Soussan, T.D. Age-Related Differential Effects of School-Based Sitting and Movement Meditation on Creativity and Spatial Cognition: A Pilot Study. Children 2021, 8, 583. [Google Scholar] [CrossRef]
- Pesce, C. Shifting the Focus from Quantitative to Qualitative Exercise Characteristics in Exercise and Cognition Research. J. Sport Exerc. Psychol. 2012, 34, 766–786. [Google Scholar] [CrossRef]
- Tomporowski, P.D.; Pesce, C. Exercise, Sports, and Performance Arts Benefit Cognition via a Common Process. Psychol. Bull. 2019, 145, 929. [Google Scholar] [CrossRef]
- Schunk, D.H.; Zimmerman, B.J. Influencing Children’s Self-Efficacy and Self-Regulation of Reading and Writing through Modeling. Read. Writ. Q. 2007, 23, 7–25. [Google Scholar] [CrossRef]
- Kessler, E.-M.; Staudinger, U.M. Affective Experience in Adulthood and Old Age: The Role of Affective Arousal and Perceived Affect Regulation. Psychol. Aging 2009, 24, 349. [Google Scholar] [CrossRef] [PubMed]
- Ben-Soussan, T.D.; Avirame, K.; Glicksohn, J.; Goldstein, A.; Harpaz, Y.; Ben-Schachar, M. Changes in Cerebellar Activity and Inter-Hemispheric Coherence Accompany Improved Reading Performance Following Quadrato Motor Training. Front. Syst. Neurosci. 2014, 8, 81. [Google Scholar] [CrossRef] [PubMed]
- Lasaponara, S.; Mauro, F.; Ben-Soussan, T.; Carducci, F.; Tombini, M.; Quattrocchi, C.; Errante, Y.; Mallio, C.; Paoletti, P. Electrophysiological Indexes of Eyes Open and Closed Resting States Conditions Following the Quadrato Motor Training. Int J Bioelectromagn 2016, 18, 99–108. [Google Scholar]
- Lasaponara, S.; Mauro, F.; Carducci, F.; Paoletti, P.; Tombini, M.; Quattrocchi, C.C.; Mallio, C.A.; Errante, Y.; Scarciolla, L.; Ben-Soussan, T.D. Increased Alpha Band Functional Connectivity Following the Quadrato Motor Training: A Longitudinal Study. Front. Hum. Neurosci. 2017, 11, 282. [Google Scholar] [CrossRef]
- Engel, T.A.; Steinmetz, N.A.; Gieselmann, M.A.; Thiele, A.; Moore, T.; Boahen, K. Selective Modulation of Cortical State during Spatial Attention. Science 2016, 354, 1140–1144. [Google Scholar] [CrossRef]
- Sauseng, P.; Klimesch, W.; Stadler, W.; Schabus, M.; Doppelmayr, M.; Hanslmayr, S.; Gruber, W.R.; Birbaumer, N. A Shift of Visual Spatial Attention Is Selectively Associated with Human EEG Alpha Activity. Eur. J. Neurosci. 2005, 22, 2917–2926. [Google Scholar] [CrossRef]
- Travis, F.; Tecce, J.; Arenander, A.; Wallace, R.K. Patterns of EEG Coherence, Power, and Contingent Negative Variation Characterize the Integration of Transcendental and Waking States. Biol. Psychol. 2002, 61, 293–319. [Google Scholar] [CrossRef]
- Cooper, N.R.; Croft, R.J.; Dominey, S.J.; Burgess, A.P.; Gruzelier, J.H. Paradox Lost? Exploring the Role of Alpha Oscillations during Externally vs. Internally Directed Attention and the Implications for Idling and Inhibition Hypotheses. Int. J. Psychophysiol. 2003, 47, 65–74. [Google Scholar] [CrossRef]
- Takahashi, T.; Murata, T.; Hamada, T.; Omori, M.; Kosaka, H.; Kikuchi, M.; Yoshida, H.; Wada, Y. Changes in EEG and Autonomic Nervous Activity during Meditation and Their Association with Personality Traits. Int. J. Psychophysiol. 2005, 55, 199–207. [Google Scholar] [CrossRef]
- Cona, G.; Chiossi, F.; Di Tomasso, S.; Pellegrino, G.; Piccione, F.; Bisiacchi, P.; Arcara, G. Theta and Alpha Oscillations as Signatures of Internal and External Attention to Delayed Intentions: A Magnetoencephalography (MEG) Study. NeuroImage 2020, 205, 116295. [Google Scholar] [CrossRef]
- Shaw, J.C. Intention as a Component of the Alpha-Rhythm Response to Mental Activity. Int. J. Psychophysiol. 1996, 24, 7–23. [Google Scholar] [CrossRef]
- Katahira, K.; Yamazaki, Y.; Yamaoka, C.; Ozaki, H.; Nakagawa, S.; Nagata, N. EEG Correlates of the Flow State: A Combination of Increased Frontal Theta and Moderate Frontocentral Alpha Rhythm in the Mental Arithmetic Task. Front. Psychol. 2018, 9, 300. [Google Scholar] [CrossRef]
- Ben-Soussan, T.D.; Glicksohn, J.; Berkovich-Ohana, A. Attentional Effort, Mindfulness, and Altered States of Consciousness Experiences Following Quadrato Motor Training. Mindfulness 2017, 8, 59–67. [Google Scholar] [CrossRef]
- Melillo, R.; Leisman, G. Neurobehavioral Disorders of Childhood: An Evolutionary Perspective; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2004; ISBN 0-306-47814-5. [Google Scholar]
- Roberts, R.E.; Anderson, E.J.; Husain, M. White Matter Microstructure and Cognitive Function. Neuroscientist 2013, 19, 8–15. [Google Scholar] [CrossRef]
- Tang, Y.-Y.; Lu, Q.; Geng, X.; Stein, E.A.; Yang, Y.; Posner, M.I. Short-Term Meditation Induces White Matter Changes in the Anterior Cingulate. Proc. Natl. Acad. Sci. USA 2010, 107, 15649–15652. [Google Scholar] [CrossRef]
- Hölzel, B.K.; Brunsch, V.; Gard, T.; Greve, D.N.; Koch, K.; Sorg, C.; Lazar, S.W.; Milad, M.R. Mindfulness-Based Stress Reduction, Fear Conditioning, and the Uncinate Fasciculus: A Pilot Study. Front. Behav. Neurosci. 2016, 10, 124. [Google Scholar] [CrossRef]
- Froeliger, B.; Garland, E.L.; McClernon, F.J. Yoga Meditation Practitioners Exhibit Greater Gray Matter Volume and Fewer Reported Cognitive Failures: Results of a Preliminary Voxel-Based Morphometric Analysis. Evid. Based Complement. Alternat. Med. 2012, 2012, 821307. [Google Scholar] [CrossRef]
- Tang, Y.-Y.; Lu, Q.; Fan, M.; Yang, Y.; Posner, M.I. Mechanisms of White Matter Changes Induced by Meditation. Proc. Natl. Acad. Sci. USA 2012, 109, 10570–10574. [Google Scholar] [CrossRef]
- Erickson, K.I.; Leckie, R.L.; Weinstein, A.M. Physical Activity, Fitness, and Gray Matter Volume. Neurobiol. Aging 2014, 35, S20–S28. [Google Scholar] [CrossRef]
- Sexton, C.E.; Betts, J.F.; Demnitz, N.; Dawes, H.; Ebmeier, K.P.; Johansen-Berg, H. A Systematic Review of MRI Studies Examining the Relationship between Physical Fitness and Activity and the White Matter of the Ageing Brain. Neuroimage 2016, 131, 81–90. [Google Scholar] [CrossRef]
- Batouli, S.A.H.; Saba, V. At Least Eighty Percent of Brain Grey Matter Is Modifiable by Physical Activity: A Review Study. Behav. Brain Res. 2017, 332, 204–217. [Google Scholar] [CrossRef] [PubMed]
- Last, N.; Tufts, E.; Auger, L.E. The Effects of Meditation on Grey Matter Atrophy and Neurodegeneration: A Systematic Review. J. Alzheimers Dis. 2017, 56, 275–286. [Google Scholar] [CrossRef] [PubMed]
- Gothe, N.P.; Hayes, J.M.; Temali, C.; Damoiseaux, J.S. Differences in Brain Structure and Function among Yoga Practitioners and Controls. Front. Integr. Neurosci. 2018, 12, 26. [Google Scholar] [CrossRef] [PubMed]
- Barysheva, M.; Jahanshad, N.; Foland-Ross, L.; Altshuler, L.L.; Thompson, P.M. White Matter Microstructural Abnormalities in Bipolar Disorder: A Whole Brain Diffusion Tensor Imaging Study. NeuroImage Clin. 2013, 2, 558–568. [Google Scholar] [CrossRef] [PubMed]
- Mayo, C.D.; Mazerolle, E.L.; Ritchie, L.; Fisk, J.D.; Gawryluk, J.R. Alzheimer’s Disease Neuroimaging Initiative Longitudinal Changes in Microstructural White Matter Metrics in Alzheimer’s Disease. NeuroImage Clin. 2017, 13, 330–338. [Google Scholar] [CrossRef]
- Ben-Soussan, T.D.; Piervincenzi, C.; Venditti, S.; Verdone, L.; Caserta, M.; Carducci, F. Increased Cerebellar Volume and BDNF Level Following Quadrato Motor Training. Synapse 2015, 69, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Hofer, S.; Frahm, J. Topography of the Human Corpus Callosum Revisited—Comprehensive Fiber Tractography Using Diffusion Tensor Magnetic Resonance Imaging. Neuroimage 2006, 32, 989–994. [Google Scholar] [CrossRef]
- Mamah, D.; Conturo, T.E.; Harms, M.P.; Akbudak, E.; Wang, L.; McMichael, A.R.; Gado, M.H.; Barch, D.M.; Csernansky, J.G. Anterior Thalamic Radiation Integrity in Schizophrenia: A Diffusion-Tensor Imaging Study. Psychiatry Res. Neuroimaging 2010, 183, 144–150. [Google Scholar] [CrossRef]
- Van Der Werf, Y.D.; Jolles, J.; Witter, M.P.; Uylings, H.B. Contributions of Thalamic Nuclei to Declarative Memory Functioning. Cortex 2003, 39, 1047–1062. [Google Scholar] [CrossRef] [PubMed]
- Bruijn, S.M.; Van Impe, A.; Duysens, J.; Swinnen, S.P. White Matter Microstructural Organization and Gait Stability in Older Adults. Front. Aging Neurosci. 2014, 6, 104. [Google Scholar] [CrossRef] [PubMed]
- Vercruysse, S.; Leunissen, I.; Vervoort, G.; Vandenberghe, W.; Swinnen, S.; Nieuwboer, A. Microstructural Changes in White Matter Associated with Freezing of Gait in Parkinson’s Disease. Mov. Disord. 2015, 30, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Papagno, C.; Miracapillo, C.; Casarotti, A.; Romero Lauro, L.J.; Castellano, A.; Falini, A.; Casaceli, G.; Fava, E.; Bello, L. What Is the Role of the Uncinate Fasciculus? Surgical Removal and Proper Name Retrieval. Brain 2011, 134, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Von Der Heide, R.J.; Skipper, L.M.; Klobusicky, E.; Olson, I.R. Dissecting the Uncinate Fasciculus: Disorders, Controversies and a Hypothesis. Brain 2013, 136, 1692–1707. [Google Scholar] [CrossRef]
- Venditti, S.; Verdone, L.; Pesce, C.; Tocci, N.; Caserta, M.; Ben-Soussan, T.D. Creating Well-Being: Increased Creativity and ProNGF Decrease Following Quadrato Motor Training. BioMed Res. Int. 2015, 2015, 275062. [Google Scholar] [CrossRef]
- Caserta, M.; Ben-Soussan, T.D.; Vetriani, V.; Venditti, S.; Verdone, L. Influence of Quadrato Motor Training on Salivary ProNGF and ProBDNF. Front. Neurosci. 2019, 13, 58. [Google Scholar] [CrossRef]
- Chiarenza, G.A.; Ga, C. Movement Related Brain Macropotentials during Skilled Performance Task in Children with Learning Disabilities. In Clinical Application of Cerebral Evoked Potentials in Pediatric Medicine; Excerpta Medica: Amsterdam, The Netherlands, 1982; pp. 259–292. [Google Scholar]
- Ben-Soussan, T.D.; Glicksohn, J.; Marson, F.; Modica, M.; Pesce, C. Embodied Time: Time Production in Advanced Quadrato and Aikido Practitioners. PsyCh J. 2019, 8, 8–16. [Google Scholar] [CrossRef]
- Glicksohn, J. Entering Trait and Context into a Cognitive-Timer Model for Time Estimation. J. Environ. Psychol. 1996, 16, 361–370. [Google Scholar] [CrossRef]
- Glicksohn, J. Temporal Cognition and the Phenomenology of Time: A Multiplicative Function for Apparent Duration. Conscious. Cogn. 2001, 10, 1–25. [Google Scholar] [CrossRef]
- Ivry, R.B.; Spencer, R.M.; Zelaznik, H.N.; Diedrichsen, J. The Cerebellum and Event Timing. Ann. N. Y. Acad. Sci. 2002, 978, 302–317. [Google Scholar] [CrossRef]
- Glicksohn, J.; Ohana, A.B.; Dotan, T.B.; Goldstein, A.; Donchin, O. Time Production and EEG Alpha Revisited. NeuroQuantology 2009, 7. [Google Scholar] [CrossRef]
- Nicolson, R.I.; Fawcett, A.J.; Dean, P. Time Estimation Deficits in Developmental Dyslexia: Evidence of Cerebellar Involvement. Proc. R. Soc. Lond. B Biol. Sci. 1995, 259, 43–47. [Google Scholar]
- Wolff, P.H. Timing Precision and Rhythm in Developmental Dyslexia. Read. Writ. 2002, 15, 179–206. [Google Scholar] [CrossRef]
- Paoletti, P.; Ben-Soussan, T.D.; Giubilei, F.; Marzano, F.; Lopez, S.; Brunno, E.; Orzi, F.; Bianchi, V.; Babiloni, C.; Buttinelli, C. Subjects with Amnesic Mild Cognitive Impairment Can Do Quadrato Motor Training with Possible Beneficial Effects on Resting State Electroencephalographic Rhythms. In Proceedings of the Federation of European Neuroscience Societies (FENS), Copenhagen, Denmark, 2–6 July 2016. [Google Scholar]
- Verdone, L.; Caserta, M.; Vetriani, V.; Glicksohn, J.; Venditti, S.; Ben-Soussan, T.D. Molecular and Cognitive Effects of Quadrato Motor Training in Adult Dyslexia: A Longitudinal Case Study. Brain Body Cogn. 2019, 8, 37–44. [Google Scholar]
- Ivry, R.B.; Keele, S.W. Timing Functions of the Cerebellum. J. Cogn. Neurosci. 1989, 1, 136–152. [Google Scholar] [CrossRef]
- Tesche, C.D.; Karhu, J.J. Anticipatory Cerebellar Responses during Somatosensory Omission in Man. Hum. Brain Mapp. 2000, 9, 119–142. [Google Scholar] [CrossRef]
- Leggio, M.G.; Silveri, M.C.; Petrosini, L.; Molinari, M. Phonological Grouping Is Specifically Affected in Cerebellar Patients: A Verbal Fluency Study. J. Neurol. Neurosurg. Psychiatry 2000, 69, 102–106. [Google Scholar] [CrossRef]
- Arns, M.; Peters, S.; Breteler, R.; Verhoeven, L. Different Brain Activation Patterns in Dyslexic Children: Evidence from EEG Power and Coherence Patterns for the Double-Deficit Theory of Dyslexia. J. Integr. Neurosci. 2007, 6, 175–190. [Google Scholar] [CrossRef]
- Taurines, R.; Segura, M.; Schecklmann, M.; Albantakis, L.; Grünblatt, E.; Walitza, S.; Jans, T.; Lyttwin, B.; Haberhausen, M.; Theisen, F.M.; et al. Altered Peripheral BDNF MRNA Expression and BDNF Protein Concentrations in Blood of Children and Adolescents with Autism Spectrum Disorder. J. Neural Transm. 2014, 121, 1117–1128. [Google Scholar] [CrossRef]
- Guilford, J.P. Intelligence, Creativity, and Their Educational Implications; Edits Pub, 1968; ISBN 0-912736-09-7. [Google Scholar]
- Ben-Soussan, T.D.; Glicksohn, J. Gender-Dependent Changes in Time Production Following Quadrato Motor Training in Dyslexic and Normal Readers. Front. Comput. Neurosci. 2018, 12, 71. [Google Scholar] [CrossRef]
- Pennington, B. Controversial Therapies for Dyslexia. Perspect. Lang. Lit. Q. Publ. Int. Dyslexia Assoc. 2011, 37, 7–8. [Google Scholar]
- Miyake, A.; Friedman, N.P.; Emerson, M.J.; Witzki, A.H.; Howerter, A.; Wager, T.D. The Unity and Diversity of Executive Functions and Their Contributions to Complex “Frontal Lobe” Tasks: A Latent Variable Analysis. Cognit. Psychol. 2000, 41, 49–100. [Google Scholar] [CrossRef]
- Diamond, A.; Ling, D.S. Conclusions about Interventions, Programs, and Approaches for Improving Executive Functions That Appear Justified and Those That, despite Much Hype, Do Not. Dev. Cogn. Neurosci. 2016, 18, 34–48. [Google Scholar] [CrossRef]
- Espin, L.; García, I.; del Pino Sánchez, M.; Román, F.; Salvador, A. Effects of Psychosocial Stress on the Hormonal and Affective Response in Children with Dyslexia. Trends Neurosci. Educ. 2019, 15, 1–9. [Google Scholar] [CrossRef]
- Paoletti, P. Flussi, Territori, Luogo [Flows, Territories, Place]; MED Publishing: Madeira, Portugal, 2002. [Google Scholar]
- Paoletti, P.; Ben Soussan, T.D. The Sphere Model of Consciousness: From Geometrical to Neuro-Psycho-Educational Perspectives. Log. Univ. 2019, 13, 395–415. [Google Scholar] [CrossRef]
- Paoletti, P.; Ben-Soussan, T.D. Reflections on Inner and Outer Silence and Consciousness without Contents According to the Sphere Model of Consciousness. Front. Psychol. 2020, 11, 1807. [Google Scholar] [CrossRef]
- Paoletti, P.; Ben-Soussan, T.D.; Glicksohn, J. Inner Navigation and Theta Activity: From Movement to Cognition and Hypnosis According to the Sphere Model of Consciousness. Hypnother. Hypn. 2020. [Google Scholar] [CrossRef]
- Paoletti, P.; Leshem, R.; Pellegrino, M.; Ben-Soussan, T.D. Tackling the Electro-Topography of the Selves Through the Sphere Model of Consciousness. Front. Psychol. 2022, 1534. [Google Scholar] [CrossRef]
- Kerr, C.E.; Sacchet, M.D.; Lazar, S.W.; Moore, C.I.; Jones, S.R. Mindfulness Starts with the Body: Somatosensory Attention and Top-down Modulation of Cortical Alpha Rhythms in Mindfulness Meditation. Front. Hum. Neurosci. 2013, 7, 12. [Google Scholar] [CrossRef]
- Flash, T.; Hochner, B. Motor Primitives in Vertebrates and Invertebrates. Curr. Opin. Neurobiol. 2005, 15, 660–666. [Google Scholar] [CrossRef]
- Casasanto, D.; Boroditsky, L. Time in the Mind: Using Space to Think about Time. Cognition 2008, 106, 579–593. [Google Scholar] [CrossRef]
- Bonato, M.; Zorzi, M.; Umiltà, C. When Time Is Space: Evidence for a Mental Time Line. Neurosci. Biobehav. Rev. 2012, 36, 2257–2273. [Google Scholar] [CrossRef]
- Lakoff, G.; Johnson, M. The Metaphorical Structure of the Human Conceptual System. Cogn. Sci. 1980, 4, 195–208. [Google Scholar] [CrossRef]
- Anelli, F.; Ciaramelli, E.; Arzy, S.; Frassinetti, F. Prisms to Travel in Time: Investigation of Time-Space Association through Prismatic Adaptation Effect on Mental Time Travel. Cognition 2016, 156, 1–5. [Google Scholar] [CrossRef]
- Atance, C.M.; O’Neill, D.K. Episodic Future Thinking. Trends Cogn. Sci. 2001, 5, 533–539. [Google Scholar] [CrossRef]
- Buckner, R.L.; Carroll, D.C. Self-Projection and the Brain. Trends Cogn. Sci. 2007, 11, 49–57. [Google Scholar] [CrossRef]
- Suddendorf, T.; Corballis, M.C. The Evolution of Foresight: What Is Mental Time Travel, and Is It Unique to Humans? Behav. Brain Sci. 2007, 30, 299–313. [Google Scholar] [CrossRef]
- Schacter, D.L.; Addis, D.R.; Hassabis, D.; Martin, V.C.; Spreng, R.N.; Szpunar, K.K. The Future of Memory: Remembering, Imagining, and the Brain. Neuron 2012, 76, 677–694. [Google Scholar] [CrossRef]
- Arzy, S.; Collette, S.; Ionta, S.; Fornari, E.; Blanke, O. Subjective Mental Time: The Functional Architecture of Projecting the Self to Past and Future. Eur. J. Neurosci. 2009, 30, 2009–2017. [Google Scholar] [CrossRef]
- Barsalou, L.W.; Huttenlocher, J.; Lamberts, K. Basing Categorization on Individuals and Events. Cognit. Psychol. 1998, 36, 203–272. [Google Scholar] [CrossRef]
- Barsalou, L.W. Grounded Cognition. Annu Rev Psychol 2008, 59, 617–645. [Google Scholar] [CrossRef]
- Barsalou, L.W. Grounding Symbolic Operations in the Brain’s Modal Systems; Cambridge University Press: Cambridge, UK, 2008; pp. 9–42. [Google Scholar] [CrossRef]
- Miles, L.K.; Karpinska, K.; Lumsden, J.; Macrae, C.N. The Meandering Mind: Vection and Mental Time Travel. PLoS ONE 2010, 5, e10825. [Google Scholar] [CrossRef]
- Miles, L.; Nind, L.; Macrae, C. Moving through Time. Psychol. Sci. 2010, 21, 222. [Google Scholar] [CrossRef]
- Anelli, F.; Avanzi, S.; Arzy, S.; Mancuso, M.; Frassinetti, F. Effects of Spatial Attention on Mental Time Travel in Patients with Neglect. Cortex 2018, 101, 192–205. [Google Scholar] [CrossRef]
- Ye, J.; Qin, X.; Cui, J.; Ren, Q.; Jia, L.; Wang, Y.; Pantelis, C.; Chan, R.C.K. A Meta-Analysis of Mental Time Travel in Individuals with Autism Spectrum Disorders. J. Autism Dev. Disord. 2021. [Google Scholar] [CrossRef]
- Addis, D.R.; Wong, A.T.; Schacter, D.L. Age-Related Changes in the Episodic Simulation of Future Events. Psychol. Sci. 2008, 19, 33–41. [Google Scholar] [CrossRef]
- d’Argembeau, A.; Raffard, S.; Van der Linden, M. Remembering the Past and Imagining the Future in Schizophrenia. J. Abnorm. Psychol. 2008, 117, 247. [Google Scholar] [CrossRef]
- Ciaramelli, E.; Anelli, F.; Frassinetti, F. An Asymmetry in Past and Future Mental Time Travel Following VmPFC Damage. Soc. Cogn. Affect. Neurosci. 2021, 16, 315–325. [Google Scholar] [CrossRef]
- Dum, R.P.; Levinthal, D.J.; Strick, P.L. Motor, Cognitive, and Affective Areas of the Cerebral Cortex Influence the Adrenal Medulla. Proc. Natl. Acad. Sci. USA 2016, 113, 9922–9927. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pellegrino, M.; Ben-Soussan, T.D.; Paoletti, P. A Scoping Review on Movement, Neurobiology and Functional Deficits in Dyslexia: Suggestions for a Three-Fold Integrated Perspective. Int. J. Environ. Res. Public Health 2023, 20, 3315. https://doi.org/10.3390/ijerph20043315
Pellegrino M, Ben-Soussan TD, Paoletti P. A Scoping Review on Movement, Neurobiology and Functional Deficits in Dyslexia: Suggestions for a Three-Fold Integrated Perspective. International Journal of Environmental Research and Public Health. 2023; 20(4):3315. https://doi.org/10.3390/ijerph20043315
Chicago/Turabian StylePellegrino, Michele, Tal Dotan Ben-Soussan, and Patrizio Paoletti. 2023. "A Scoping Review on Movement, Neurobiology and Functional Deficits in Dyslexia: Suggestions for a Three-Fold Integrated Perspective" International Journal of Environmental Research and Public Health 20, no. 4: 3315. https://doi.org/10.3390/ijerph20043315
APA StylePellegrino, M., Ben-Soussan, T. D., & Paoletti, P. (2023). A Scoping Review on Movement, Neurobiology and Functional Deficits in Dyslexia: Suggestions for a Three-Fold Integrated Perspective. International Journal of Environmental Research and Public Health, 20(4), 3315. https://doi.org/10.3390/ijerph20043315