Rehabilitation Outcomes Following Hip Fracture of Home-Based Exercise Interventions Using a Wearable Device—A Randomized Controlled Pilot and Feasibility Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Setting
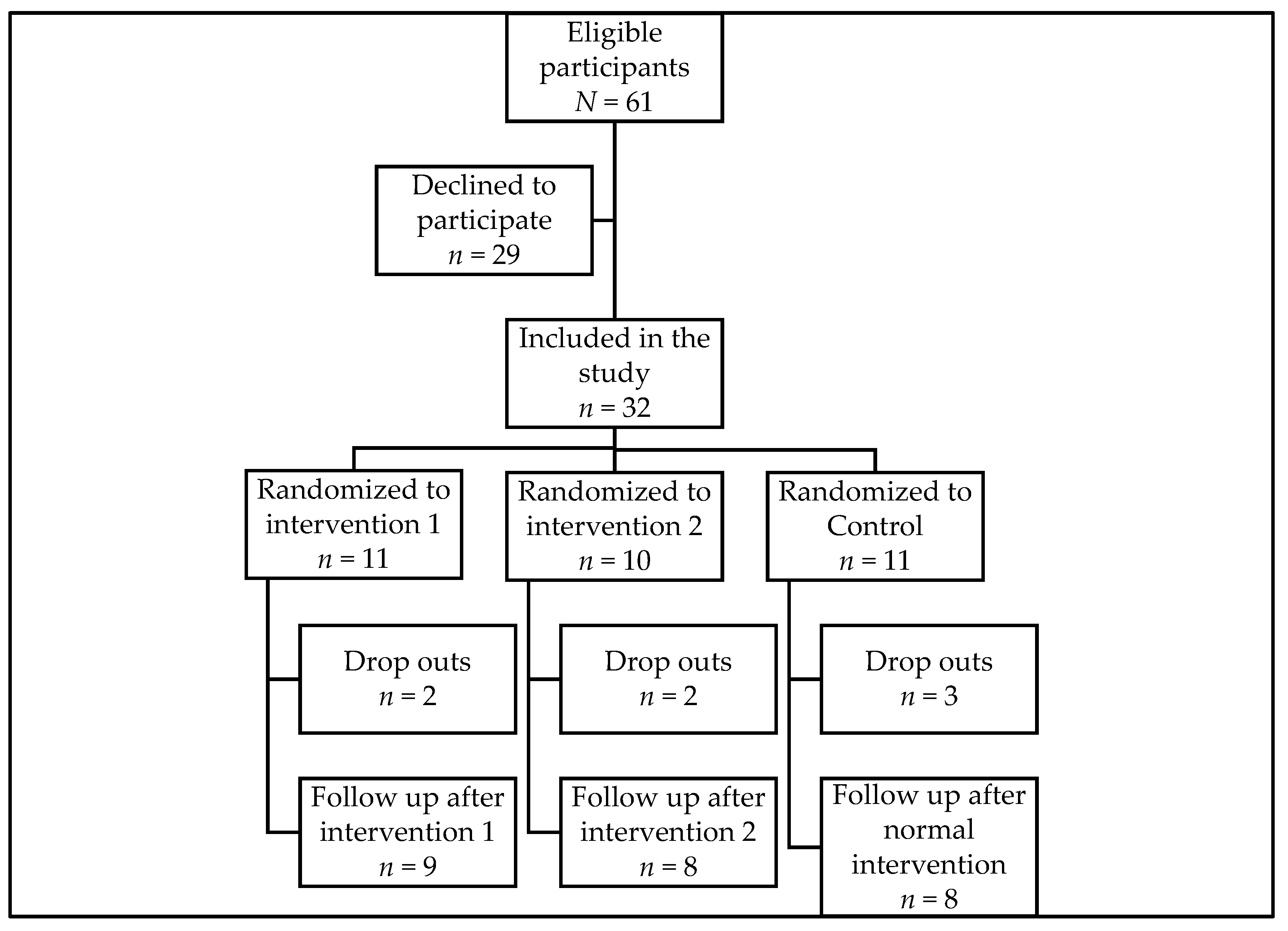
2.3. Recruitment and Participant Inclusion/Exclusion
2.4. Randomization
2.5. Intervention
2.6. Outcomes
2.7. Measurement
2.8. Procedure
2.9. Statistical Methods
3. Results
3.1. Feasibility Measures
3.2. Adverse Events
3.3. Within-Group Differences
3.4. Between-Group Differences
3.5. Adjustments before the Full RCT
4. Discussion
Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rey-Rodriguez, M.M.; Vazquez-Gamez, M.A.; Giner, M.; Garrachón-Vallo, F.; Fernández-López, L.; Colmenero, M.A.; Montoya-García, M.-J. Incidence, morbidity and mortality of hip fractures over a period of 20 years in a health area of Southern Spain. BMJ Open. 2020, 10, e037101. [Google Scholar] [CrossRef]
- Nilson, F.; Moniruzzaman, S.; Gustavsson, J.; Andersson, R. Trends in hip fracture incidence rates among the elderly in Sweden 1987–2009. J. Public Health 2012, 35, 125–131. [Google Scholar] [CrossRef]
- Rosengren, B.E.; Karlsson, M.K. The annual number of hip fractures in Sweden will double from year 2002 to 2050. Acta Orthop. 2014, 85, 234–237. [Google Scholar] [CrossRef]
- Fletcher, P.C.; Hirdes, J.P. Restriction in activity associated with fear of falling among community-based seniors using home care services. Age Ageing 2004, 33, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Banefelt, J.; Åkesson, K.E.; Spångéus, A.; Ljunggren, O.; Karlsson, L.; Ström, O.; Ortsäter, G.; Libanati, C.; Toth, E. Risk of imminent fracture following a previous fracture in a Swedish database study. Osteoporos. Int. 2019, 30, 601–609. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.J.; Um, S.H.; Kim, Y.H. Postoperative Rehabilitation after Hip Fracture: A Literature Review. Hip Pelvis 2020, 32, 125–131. [Google Scholar] [CrossRef]
- Marks, R. Hip fracture epidemiological trends, outcomes, and risk factors, 1970-2009. Int. J. Gen. Med. 2010, 3, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Siebens, H.C.; Sharkey, P.; Aronow, H.U.; Deutscher, D.; Roberts, P.; Munin, M.C.; Radnay, C.S.; Horn, S.D. Variation in Rehabilitation Treatment Patterns for Hip Fracture Treated With Arthroplasty. Pm. R. 2016, 8, 191–207. [Google Scholar] [CrossRef]
- Chen, B.; Hu, N.; Tan, J.H. Efficacy of home-based exercise programme on physical function after hip fracture: A systematic review and meta-analysis of randomised controlled trials. Int. Wound J. 2020, 17, 45–54. [Google Scholar] [CrossRef]
- Ekvall Hansson, E.; Fänge, A.M.; Rogmark, C. Assessing the Outcome of Rehabilitation after Hip Fracture with a Wearable Device-A Study Protocol for a Randomized Control Trial in Community Healthcare. Int. J. Environ. Res. Public Health 2021, 18, 10165. [Google Scholar] [CrossRef]
- Fairhall, N.J.; Dyer, S.M.; Mak, J.C.; Diong, J.; Kwok, W.S.; Sherrington, C. Interventions for improving mobility after hip fracture surgery in adults. Cochrane Database Syst. Rev. 2022, 9, Cd001704. [Google Scholar] [PubMed]
- McDonough, C.M.; Harris-Hayes, M.; Kristensen, M.T.; Overgaard, J.A.; Herring, T.B.; Kenny, A.M.; Mangione, K.K. Physical Therapy Management of Older Adults With Hip Fracture. J. Orthop. Sport. Phys. 2021, 51, Cpg1–Cpg81. [Google Scholar] [CrossRef] [PubMed]
- Hulsbæk, S.; Juhl, C.; Røpke, A.; Bandholm, T.; Kristensen, M.T. Exercise Therapy Is Effective at Improving Short- and Long-Term Mobility, Activities of Daily Living, and Balance in Older Patients Following Hip Fracture: A Systematic Review and Meta-Analysis. J. Gerontol. A. Biol. Sci. Med. Sci. 2022, 77, 861–871. [Google Scholar] [CrossRef] [PubMed]
- Littbrand, H.; Rosendahl, E.; Lindelöf, N. The HIFE Program, 2nd ed.; Umeå University: Umeå, Sweden, 2014. [Google Scholar]
- Bull, F.C.; Al-Ansari, S.S.; Biddle, S.; Borodulin, K.; Buman, M.P.; Cardon, G.; Carty, C.; Chaput, J.P.; Chastin, S.; Chou, R.; et al. World Health Organization 2020 guidelines on physical activity and sedentary behaviour. Br. J. Sport. Med. 2020, 54, 1451–1462. [Google Scholar] [CrossRef]
- Ekvall Hansson, E.; Tornberg, Å. Coherence and reliability of a wearable inertial measurement unit for measuring postural sway. BMC Res. Notes 2019, 12, 201. [Google Scholar] [CrossRef]
- Craig, P.; Dieppe, P.; Macintyre, S.; Michie, S.; Nazareth, I.; Petticrew, M. Developing and evaluating complex interventions: The new Medical Research Council guidance. BMJ 2008, 337, a1655. [Google Scholar] [CrossRef]
- Thabane, L.; Ma, J.; Chu, R.; Cheng, J.; Ismaila, A.; Rios, L.P.; Robson, R.; Thabane, M.; Giangregorio, L.; Goldsmith, C.H. A tutorial on pilot studies: The what, why and how. BMC Med. Res. Methodol. 2010, 10, 1. [Google Scholar] [CrossRef]
- Ekvall Hansson, E.; Valkonen, E.; Olsson Möller, U.; Chen Lin, Y.; Magnusson, M.; Fransson, P.-A. Gait Flexibility among Older Persons Significantly More Impaired in Fallers Than Non-Fallers—A Longitudinal Study. Int. J. Environ. Res. Public Health 2021, 18, 7074. [Google Scholar] [CrossRef]
- Jansen, C.-P.; Klenk, J.; Nerz, C.; Todd, C.; Labudek, S.; Kramer-Gmeiner, F.; Becker, C.; Schwenk, M. Association between everyday walking activity, objective and perceived risk of falling in older adults. Age Ageing 2021, 50, 1586–1592. [Google Scholar] [CrossRef]
- Möller U, O.; Fänge A, M.; Kristensson, J.; Smedberg, D.; Falkvall, F.; Hansson E, E. Modern technology against falls—A description of the MoTFall project. Health Inform. J. 2021, 27, 14604582211011514. [Google Scholar] [CrossRef]
- Cho, Y.S.; Jang, S.H.; Cho, J.S.; Kim, M.J.; Lee, H.D.; Lee, S.Y.; Moon, S.B. Evaluation of Validity and Reliability of Inertial Measurement Unit-Based Gait Analysis Systems. Ann. Rehabil. Med. 2018, 42, 872–883. [Google Scholar] [CrossRef] [PubMed]
- Kobsar, D.; Charlton, J.M.; Tse, C.T.F.; Esculier, J.F.; Graffos, A.; Krowchuk, N.M.; Thatcher, D.; Hunt, M.A. Validity and reliability of wearable inertial sensors in healthy adult walking: A systematic review and meta-analysis. J. Neuroeng. Rehabil. 2020, 17, 62. [Google Scholar] [CrossRef] [PubMed]
- Piirtola, M.; Era, P. Force platform measurements as predictors of falls among older people—A review. Gerontology 2006, 52, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Johansson, J.; Nordström, A.; Gustafson, Y.; Westling, G.; Nordström, P. Increased postural sway during quiet stance as a risk factor for prospective falls in community-dwelling elderly individuals. Age Ageing 2017, 46, 964–970. [Google Scholar] [CrossRef]
- Fredriksson, L.; Hellström, K. Reliability, Validity, and Reference Values for Functional Balance Test for Geriatric Patients (FBG). Phys. Occup. Ther. Geriatr. 2012, 30, 177–188. [Google Scholar] [CrossRef]
- Collin, C.; Wade, D.T.; Davies, S.; Horne, V. The Barthel ADL Index: A reliability study. Int. Disabil. Stud. 1988, 10, 61–63. [Google Scholar] [CrossRef]
- Hormozi, S.; Alizadeh-Khoei, M.; Sharifi, F.; Taati, F.; Aminalroaya, R.; Fadaee, S.; Angooti-Oshnari, L.; Saghebi, H. Iranian Version of Barthel Index: Validity and Reliability in Outpatients’ Elderly. Int. J. Prev. Med. 2019, 10, 130. [Google Scholar]
- Unnanuntana, A.; Jarusriwanna, A.; Nepal, S. Validity and responsiveness of Barthel index for measuring functional recovery after hemiarthroplasty for femoral neck fracture. Arch. Orthop. Trauma Surg. 2018, 138, 1671–1677. [Google Scholar] [CrossRef]
- Devlin, N.J.; Brooks, R. EQ-5D and the EuroQol Group: Past, Present and Future. Appl. Health Econ. Health Policy 2017, 15, 127–137. [Google Scholar] [CrossRef]
- Emrani, Z.; Akbari Sari, A.; Zeraati, H.; Olyaeemanesh, A.; Daroudi, R. Health-related quality of life measured using the EQ-5D–5 L: Population norms for the capital of Iran. Health Qual. Life Outcomes 2020, 18, 108. [Google Scholar] [CrossRef]
- Pérez-Ros, P.; Martínez-Arnau, F.M. EQ-5D-3L for Assessing Quality of Life in Older Nursing Home Residents with Cognitive Impairment. Life 2020, 10, 100. [Google Scholar] [CrossRef] [PubMed]
- EUROQOL. EQ-5D-5L|Choosing a Value Set. Available online: https://euroqol.org/eq-5d-instruments/eq-5d-3l-about/valuation/choosing-a-value-set/ (accessed on 1 December 2020).
- EUROQOL. EQ-5D-5L|Valuation|Crosswalk Index Value Calculator. Available online: https://euroqol.org/eq-5d-instruments/eq-5d-5l-about/valuation-standard-value-sets/crosswalk-index-value-calculator/ (accessed on 1 December 2020).
- Lyu, S.; Freivalds, A.; Downs, D.S.; Piazza, S.J. Assessment of postural sway with a pendant-mounted wearable sensor. Gait Posture 2022, 92, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Pajala, S.; Era, P.; Koskenvuo, M.; Kaprio, J.; Törmäkangas, T.; Rantanen, T. Force Platform Balance Measures as Predictors of Indoor and Outdoor Falls in Community-Dwelling Women Aged 63–76 Years. J. Gerontol. Ser. A 2008, 63, 171–178. [Google Scholar] [CrossRef]
- Gerdhem, P.; Ringsberg, K.A.; Akesson, K. The relation between previous fractures and physical performance in elderly women. Arch. Phys. Med. Rehabil. 2006, 87, 914–917. [Google Scholar] [CrossRef] [PubMed]
- Kuijlaars, I.A.R.; Sweerts, L.; Nijhuis-van der Sanden, M.W.G.; van Balen, R.; Staal, J.B.; van Meeteren, N.L.U.; Hoogeboom, T.J. Effectiveness of Supervised Home-Based Exercise Therapy Compared to a Control Intervention on Functions, Activities, and Participation in Older Patients After Hip Fracture: A Systematic Review and Meta-analysis. Arch. Phys. Med. Rehabil. 2019, 100, 101–114.e106. [Google Scholar] [CrossRef]
- Hansen, C.; Beckbauer, M.; Romijnders, R.; Warmerdam, E.; Welzel, J.; Geritz, J.; Emmert, K.; Maetzler, W. Reliability of IMU-Derived Static Balance Parameters in Neurological Diseases. Int. J. Environ. Res. Public Health 2021, 18, 3644. [Google Scholar] [CrossRef]
- Rosendahl, E.; Lindelöf, N.; Littbrand, H.; Yifter-Lindgren, E.; Lundin-Olsson, L.; Håglin, L.; Gustafson, Y.; Nyberg, L. High-intensity functional exercise program and proteinenriched energy supplement for older persons dependent in activities of daily living: A randomised controlled trial. Aust. J. Physiother. 2006, 52, 105–113. [Google Scholar] [CrossRef]
- Thorngren, K.-G. Optimized Treatment of Hip Fractures. In Biomechanics and Biomaterials in Orthopedics; Poitout, D.G., Ed.; Springer: London, UK, 2004; pp. 515–527. [Google Scholar]
- Lee, S.Y.; Jung, S.H.; Lee, S.-U.; Ha, Y.-C.; Lim, J.-Y. Effect of Balance Training After Hip Fracture Surgery: A Systematic Review and Meta-analysis of Randomized Controlled Studies. J. Gerontol. Ser. A 2018, 74, 1679–1685. [Google Scholar] [CrossRef]
- Knauf, T.; Buecking, B.; Hack, J.; Barthel, J.; Bliemel, C.; Aigner, R.; Ruchholtz, S.; Eschbach, D. Development of the Barthel Index 5 years after hip fracture: Results of a prospective study. Geriatr. Gerontol. Int. 2019, 19, 809–814. [Google Scholar] [CrossRef]
- Chiang, M.H.; Huang, Y.Y.; Kuo, Y.J.; Huang, S.W.; Jang, Y.C.; Chu, F.L.; Chen, Y.P. Prognostic Factors for Mortality, Activity of Daily Living, and Quality of Life in Taiwanese Older Patients within 1 Year Following Hip Fracture Surgery. J. Pers. Med. 2022, 12, 102. [Google Scholar] [CrossRef]
- Amarilla-Donoso, F.J.; Roncero-Martin, R.; Lavado-Garcia, J.M.; Toribio-Felipe, R.; Moran-Garcia, J.M.; Lopez-Espuela, F. Quality of life after hip fracture: A 12-month prospective study. PeerJ 2020, 8, e9215. [Google Scholar] [CrossRef] [PubMed]
- Milte, R.; Crotty, M.; Miller, M.D.; Whitehead, C.; Ratcliffe, J. Quality of life in older adults following a hip fracture: An empirical comparison of the ICECAP-O and the EQ-5D-3 L instruments. Health Qual. Life Outcomes 2018, 16, 173. [Google Scholar] [CrossRef] [PubMed]
- Forsat, N.D.; Palmowski, A.; Palmowski, Y.; Boers, M.; Buttgereit, F. Recruitment and Retention of Older People in Clinical Research: A Systematic Literature Review. J. Am. Geriatr. Soc. 2020, 68, 2955–2963. [Google Scholar] [CrossRef] [PubMed]
- Omar, A.; Cumal, A.; Vellani, S.; Krassikova, A.; Lapenskie, J.; Bayly, M.; Welch, V.A.; Ghogomu, E.; Iaboni, A.; McGilton, K.S. Health and social interventions to restore physical function of older adults post-hip fracture: A scoping review. BMJ Open 2021, 11, e053992. [Google Scholar] [CrossRef]

| Categories | Examples of Exercises |
|---|---|
| A: Static and dynamic exercises in combination with lower-limb strength exercises | Squats, body weight transfer, sit to stand, lunges, step-up, stair walking |
| B: Dynamic balance exercises in walking | Walking exercises with various difficulty levels, step over, change of walking surface |
| C: Static and dynamic balance exercises in standing | Weight transfers, turning head in different directions, reaching for an object, catching and throwing a ball, kicking a ball |
| D: Lower-limb strength exercises with continuous balance support | Squats, sit to stand, heel raises, weight transfer sideways, stair walking |
| E: Walking with continuous balance support | Walking in different directions, walking and turning |
| Training Session | High Intensity | Medium Intensity | Low Intensity |
|---|---|---|---|
| Strength training exercises | Sets of 8–12 RM | Sets of 13–15 RM | Sets of >15 RM |
| Balance training exercises | Postural stability fully challenged * | Postural stability not fully challenged or fully challenged in a minority of the exercises | Postural stability not challenged |
| Measure | Total Sample n = 32 Median (IQR) | Intervention 1 n = 11 Median (IQR) | Intervention 2 n = 10 Median (IQR) | Control n = 11 Median (IQR) |
|---|---|---|---|---|
| Women/men | 25/7 | 10/1 | 7/3 | 8/3 |
| Age (years) | 85 (12) | 83 (13) | 86 (16) | 85 (16) |
| Intervention days | 77 (38) | 58 (33) | 77.5 (26) | 80.5 (19) |
| FBG * | 6 (3) | 7 (3) | 5,5 (2) | 8 (2) |
| BI | 62.5 (24) | 60 (20) | 55 (18) | 75 (20) |
| EQ5D index | 0.508 (0.211) | 0.465 (0.226) | 0.487 (0.382) | 0.556 (0.110) |
| EQ5D VAS in mm | 50 (30) | 40 (45) | 44 (43) | 50 (25) |
| PSOEAP mm/s | 15 (22) | 15 (38.3) | 17 (13.5) | 7 (19) |
| PSOEML mm/s | 4 (4) | 4.5 (10.3) | 5.0 (2.5) | 3.0 (2.3) |
| PSCEAP mm/s | 10 (19.5) | 16.5 (38.5) | 16 (16) | 9.5 (8.8) |
| PSCEML mm/s | 5 (8) | 5 (18) | 5 (4) | 7 (7.5) |
| Within-Group Differences, Intervention 1 (n = 9) | Within-Group Differences, Intervention 2 (n = 8) | Within-Group Differences, Control (n = 8) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Measure | Median Change | CI | p-Value | Median Change | CI | p-Value | Median Change | CI | p-Value |
| FBG | 5 | 4–7 | 0.007 | 7.5 | 2–13 | 0.018 | 5.5 | 4–13 | 0.018 |
| BI | 20 | 5–40 | 0.008 | 35 | 10–40 | 0.011 | 22.5 | 15–35 | 0.012 |
| EQ5D index | 0.185 | 0.092–0.457 | 0.011 | 0.228 | 0.165–0.339 | 0.012 | 0.193 | 0.068–0.253 | 0.018 |
| EQ5D VAS | 20 | −5–35 | 0.262 | 26 | 5–45 | 0.036 | 30 | 15–45 | 0.018 |
| PSOEAP mm/s | 12 | −6–27 | 0.225 | 8 | 3–13 | 0.068 | 1 | 0–13 | 0.102 |
| PSOEML mm/s | −1 | −12–12 | 0.581 | 0 | −10–3 | 0.705 | 2 | −5–7 | 0.465 |
| PSCEAP mm/s | 7.5 | −7–20 | 0.705 | −6 | −22–2 | 0.144 | 0 | −11–3 | 1.000 |
| PSCEML mm/s | 0.5 | −4- 14 | 0.465 | 0 | −18–5 | 0.854 | 1 | −1–3 | 0.414 |
| Between-Group Differences, Intervention 1 and Intervention 2 (n = 17) | Between-Group Differences, Intervention 1 and Control (n = 17) | Between-Group Differences, Intervention 2 and Control (n = 16) | p-Value for Median Difference in Change | ||||
|---|---|---|---|---|---|---|---|
| Measure | Median Change | CI | Median Change | CI | Median Change | CI | |
| FBG | 5 | 4–10 | 5 | 5–6 | 5 | 4–10 | 0.943 |
| BI | 30 | 15–40 | 20 | 15–35 | 30 | 20–35 | 0.283 |
| EQ5D index | 0.191 | 0.137–0.339 | 0.188 | 0.104–0.253 | 0.210 | 0.123–0.256 | 0.630 |
| EQ5D VAS | 25 | 0–30 | 30 | 15–35 | 30 | 20–45 | 0.322 |
| PSOEAP mm/s | 9 | −1–13 | 6 | −1–13 | 7 | 0–13 | 0.921 |
| PSOEML mm/s | −2 | −10–3 | 0 | −5–7 | 2.5 | −5–3 | 0.724 |
| PSCEAP mm/s | −2.5 | −8–16 | 1 | −7–16 | −2 | −11–2 | 0.304 |
| PSCEML mm/s | 0 | −4–5 | 1 | −4–3 | 1 | −18–3 | 0.920 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Howell, D.F.; Malmgren Fänge, A.; Rogmark, C.; Ekvall Hansson, E. Rehabilitation Outcomes Following Hip Fracture of Home-Based Exercise Interventions Using a Wearable Device—A Randomized Controlled Pilot and Feasibility Study. Int. J. Environ. Res. Public Health 2023, 20, 3107. https://doi.org/10.3390/ijerph20043107
Howell DF, Malmgren Fänge A, Rogmark C, Ekvall Hansson E. Rehabilitation Outcomes Following Hip Fracture of Home-Based Exercise Interventions Using a Wearable Device—A Randomized Controlled Pilot and Feasibility Study. International Journal of Environmental Research and Public Health. 2023; 20(4):3107. https://doi.org/10.3390/ijerph20043107
Chicago/Turabian StyleHowell, David Flecks, Agneta Malmgren Fänge, Cecilia Rogmark, and Eva Ekvall Hansson. 2023. "Rehabilitation Outcomes Following Hip Fracture of Home-Based Exercise Interventions Using a Wearable Device—A Randomized Controlled Pilot and Feasibility Study" International Journal of Environmental Research and Public Health 20, no. 4: 3107. https://doi.org/10.3390/ijerph20043107
APA StyleHowell, D. F., Malmgren Fänge, A., Rogmark, C., & Ekvall Hansson, E. (2023). Rehabilitation Outcomes Following Hip Fracture of Home-Based Exercise Interventions Using a Wearable Device—A Randomized Controlled Pilot and Feasibility Study. International Journal of Environmental Research and Public Health, 20(4), 3107. https://doi.org/10.3390/ijerph20043107







